Lab-on-a-Chip Biosensors for Safer Pharmacological Research
Smarter Chips, Safer Lives: Lab-on-a-chip Biosensors for Pharmacological Applications and Healthcare Transformation
Ankit Kumar Singh¹ and Ida Tiwari²*
- Department of Applied Science and Humanities, Government Engineering College, Kaimur-821102 under Bihar Engineering University Patna, DSTTE Patna, Bihar, India
- Department of Chemistry (Centre of Advanced Study), Institute of Science, Banaras Hindu University, Varanasi-221005, India
OPEN ACCESS
PUBLISHED:31 March 2025
CITATION: Tiwari, I. and Singh, A.K., 2025. Smarter Chips, Safer Lives: Lab-on-a-chip Biosensors for Pharmacological Applications and Healthcare Transformation. Medical Research Archives, [online] 13(3).
https://doi.org/10.18103/mra.v13i3.6337
COPYRIGHT: © 2025 European Society of Medicine. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i3.6337
ISSN 2375-1924
ABSTRACT
Rapid advancements in industries like genetics, biotechnology, and medicine have raised the demand for chemical and biochemical analysis and led to the development of useful chip applications for the use of sophisticated equipment, labs, and traditional procedures. A new technique called lab-on-a-chip displays a miniature laboratory onto a tiny coin-sized chip. Reagent mixing, dilution, electrophoresis, separation, staining, and detection can all be accomplished on a sensor chip. One or several analyses are made possible by this sensor technology, which also produces high sensitivity, quick diagnostic times, improved process control, inexpensive fabrication costs, portability, and safety. The detection of drugs is currently being performed using lab-on-a-chip technology, which may accept a variety of biological and non-biological materials. A number of detection techniques were used by lab-on-a-chip devices, with immunoassays being most frequently used. The usage of real-world samples should be increased, validation should be enhanced, and practicality should be further examined in terms of providing information on cost, speed of analysis, and convenience of use. A wide variety of lab-on-a-chip techniques are already accessible, which suggests that these devices could be used as portable, quick, and affordable detection systems. Lab-on-a-chip systems can be utilised both within and outside of hospitals for clinical purposes. Numerous benefits were provided by Lab-on-a-chips over existing tests, including the ability to do point-of-care diagnostics with minimal fluid volumes, the use of small quantities of costly chemicals and samples, controlled flow rate, short diffusion distance, fast mixing time, and inexpensive fabrication costs.
Keywords: Sensor chips, toxic drugs, healthcare transformation, detection, pharmacological.
1. Introduction
The pharmaceutical sector is facing significant obstacles due to rising expenses and inefficient medication research. The low predictive capacity of current preclinical models is the reason behind drug failures in trials, hence drug development researchers have confirmed the urgent need for novel testing methods that can reliably anticipate drug safety and efficacy in people¹. According to data from the 2019 United Nations on Drugs and Crime (UNODC) World Drugs Report, 271 million persons worldwide abused drugs in 2017, resulting in approximately 585,000 drug-related deaths². This demonstrates not only the substantial number of drug users but also the considerable number of drug-related deaths that occur worldwide.
Thus, devices and methods for identifying and monitoring harmful substances are essential for ensuring human health. In this age of rapid material evolution, it is critical to describe and evaluate the toxicity associated with pharmaceutical residues. Toxic pharmaceuticals must be closely monitored to prevent any risks to human health, even if national authorities have strict control over these systems. One important element that boosts the impact of treatment is early detection³. Despite ongoing advancements in drug screening techniques, only a small percentage of drug candidates are approved for clinical use by the US Food & Drug Administration (FDA)⁴.
Drugs in various samples can be analysed using a variety of contemporary laboratory-based methods, including mass spectrometry⁵, chromatography⁶, immunoassays⁷, spectrophotometry⁸, and colorimetry⁹. These advanced methods, however, are typically linked to difficulties with sample handling, miniaturisation, or skilled lab personnel. When millions of distinct chemical combinations must be tested during drug discovery, the need for numerous read-outs puts a significant strain on the testing procedures. Small chemical volumes and parallel operation are required for a high-throughput system to handle these many samples while maintaining an inexpensive development cost¹⁰.
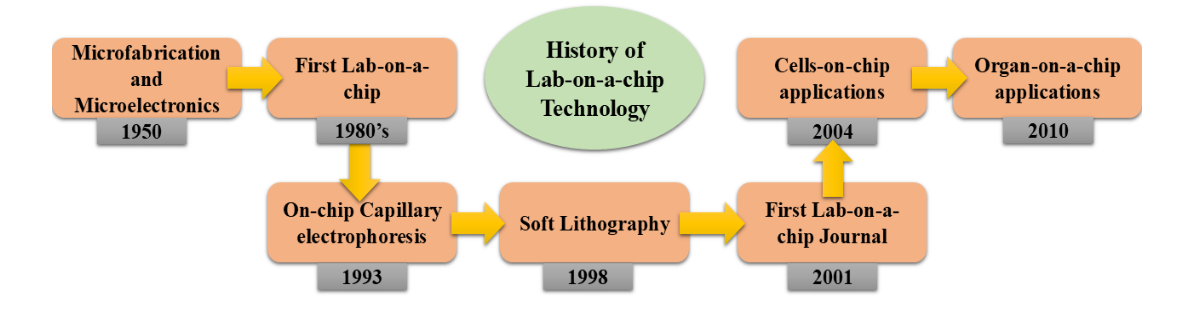
There is a growing trend of miniaturisation using chemical and biological analytical techniques. An approach that shows promise for analyte detection and for enabling wireless communication of analysis results to remote areas is chip-based sensing technology. These instruments are affordable, portable, ensure speedy findings, and can be included into biological systems that are responsive and sophisticated¹¹,¹². The advancement in the miniaturisation of these analysis techniques is directly reflected in lab-on-a-chip (LOC) technology. Devices that are capable of controlling and modifying fluid flows at the micro level are known as LOC devices. They offer a wide range of fascinating applications in a number of domains, such as drug release investigations, environmental analysis, clinical diagnostics, and food control¹³. Numerous laboratory studies, biological processes, DNA sequencing, and chemical production all make use of these microdevices. By combining microfluidic devices with biosensors, they can be used in industries like environmental monitoring, food processing and safety, pharmaceuticals, medical diagnostics, and agriculture. A few examples of applications where LOC platforms can be useful include the analysis of ions from various compositions used in forensics, explosives identification, water quality assessment, bodily fluid research, agriculture, and pollution level detection¹⁴,¹⁵. The history in the development and applications of LOC techniques is represented in Fig. 1.
Lab-on-a-chip tools provide automation and high-throughput analysis by combining various laboratory processes onto a single chip. The LOC technologies have the following key characteristics¹⁵,¹⁶,¹⁷:
(i) Transporting liquid samples containing bioparticles, such as cells, proteins, and DNA, into a field with electrode molecules that have been previously stored;
(ii) Electrode and extracting non-specific and lossy binding, as well as mixing and binding reactions of extracted bioparticles; and
(iii) Detecting thecorresponding change in chemical, physical, electrical, mechanical, or magnetic signals¹⁸. These methods provide portable equipment by performing several laboratory operations on a smaller scale, such as chemical synthesis and analysis on a single chip. Developments in micro and nanotechnology have made it possible to fabricate LOC tools in smaller sizes. This is comparable to the revolution in semiconductors, which has been significantly influenced by lithographic methods. The integration of many laboratory processes on a micro or nanoscale facilitates automation and high-throughput screening. A number of factors, such as capillary forces, electrokinetics (EKs), and pressure gradients, affect LOC manufacturing. By controlling low-volume samples, microfluidic systems can achieve high analysis rates while saving money and time. It can be applied to chemical and biological studies, as well as chemical synthesis¹⁹.
Recently, researchers have been deeply examining powerful tools for biomedical engineering research studies, such as illness modelling, drug design, and nano-drug delivery¹²,¹⁵,¹⁹. In recent years, research into various microfluidics techniques and the development of novel biomedical methods using microfluidic-based substrate have led to the production of numerous cell culture platforms for these types of approaches. Tissue chip platforms are micro instruments that replicate living tissue in vivo and show more physiologically relevant human tissue models in vitro. In vitro cell culturing, as opposed to static culture, produced optimised tissue systems more rapidly when LOC technologies were applied. Cell culture medium is prepared by these chips to replicate physiological responses and behaviours²⁰.
The sensing elements for target recognition and the signal-transducing elements for reporting interactions between targets and sensing elements have significantly improved as a result of the development of material sciences and sophisticated technologies for fabrication and signal-transducing processes²¹. Numerous outstanding reviews have succinctly outlined the main ideas and uses of various kinds of chemical sensors²². Thus, this study focuses on recent developments in LOC sensors for drug detection, including recent developments in biosensors for pharmacological applications, as well as advances in LOC sensors for sensing and signal-transducing elements²³.
For the extremely selective detection of toxic drugs, considerable efforts have been made to construct highly selective electrochemical biosensors¹⁵. These initiatives include the development of signal amplifiers based on nanomaterials, the investigation of biological receptors with high affinity, and the integration of microfluidic chips and biosensors²⁴. The biosensor is a phenomena analytical tool for label-free biological chemical detection and real-time biological interaction monitoring that consists of a bioreceptor, a transducer, and a signal processor²⁵. The name ‘biosensor’ itself is frequently relatively ill-defined, or at the very least, signifies different things to different people, which contributes to the
specialised staff³¹,³²,³³. Microfluidics has developed into a very interdisciplinary topic of study in recent years. The development of fully integrated ‘sample in-answer out’ LOC devices has focused on areas such as clinical diagnostics³³, but one fully integrated LOC for forensic purposes is the RapidHIT® ID System, which analyses buccal swab samples for human identification purposes and can generate a DNA profile in just 90 minutes³⁴.
The first commercially accessible LOC system was released around two years after Hopwood et al.³⁵ published the first research journal article in 2010 describing a fully integrated LOC device that might generate a DNA profile. Since the Rapid DNA Act of 2017, law enforcement has been using this type of LOC technology to analyse reference samples³⁵. This indicates that the employment of LOC systems in forensic investigations may prove to be beneficial.
Analytical instruments are made smaller by using LOC technology, which packs numerous lab tools onto a tiny single chip¹². Two essential concepts of engineering research that are miniaturization and integration are important aspects of LOC technology. The main reason for this significant growth is the quick development of integrated circuit (IC) technology. Miniaturisation makes it possible to replace traditional heavy, costly equipment with more affordable, portable alternatives¹². These days, embedded artificial intelligence technology and smartphone integration are popular.
Depending on the intended use, the literature contains a wide variety of design types. Lab-on-a-chip systems have certain basic components, but the types of components differ depending on the application. The working parts include an injector, a transporter, a preparator, a mixer, a reactor, a separator, a detector, a controller, and a power source. Since the components are represented by appropriate symbols, designers can display their LOC products consistently¹⁴. To elucidate these counters, the elements of a LOC device are shown in Fig. 2.
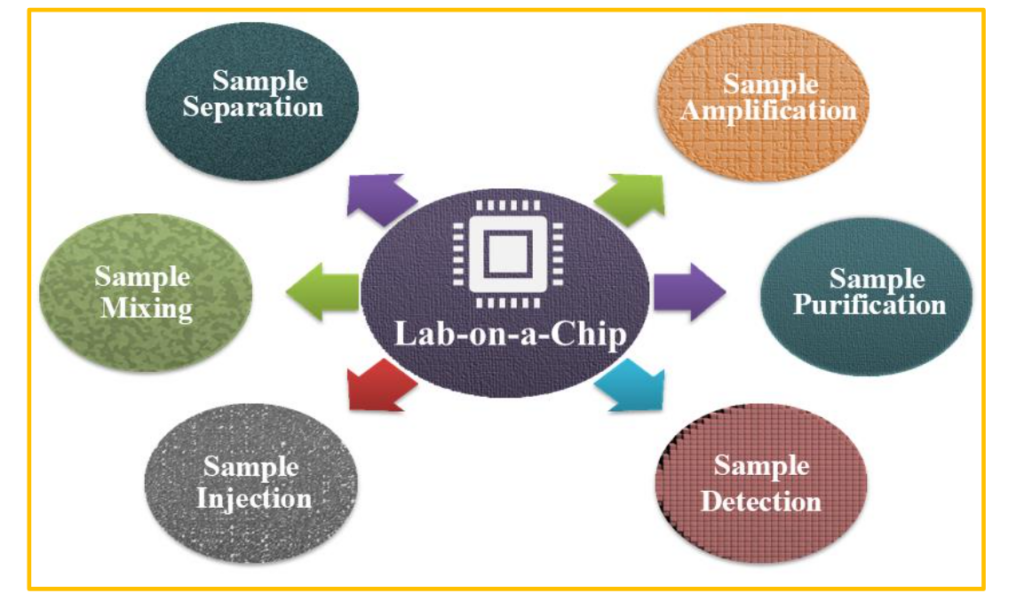
3. Key Features of Lab-on-a-chip sensors
The LOC systems differ from conventional laboratory equipment in a number of significant ways, including microfluidics, integration and miniaturization¹²,¹⁵,²⁰,³⁶.
• Microfluidics: The technology used in LOC devices is microfluidic, which manipulates fluids in channels that range in size from tens to hundreds of micrometres. Microfluidics makes it possible to precisely control fluid flow, mixing, and reactions, which makes sample processing and analysis more effective¹²,³¹.
• Integration: The LOC devices combine several laboratory processes onto a single chip, including sample preparation, separation, reaction, and detection. This integration speeds up and increases the accuracy of the analysis while lowering the need for manual intervention and consuming fewer samples and reagents¹².
• Miniaturization: The LOC instruments drastically cut down on the quantity of necessary samples and reagents by minimising laboratory procedures, which lowers expenses and waste production. Additionally, miniaturisation makes it possible to create POC and portable diagnostic tools¹²,³⁷.
4. Fabrication of lab-on-a-chip sensors
A wide range of inorganic, organic, and composite materials are being employed to build the fundamental structure of LOC devices. Among the most widely used materials in fabrication include crystalline minerals like silicon and glass, polymers including photoresists, acrylate, and thermoplastics, and elastomers like polydimethylsiloxane³⁸. Silicon used to rule the microfabrication industry because of its semiconducting characteristics. Due to its well-documented surface engineering capabilities based on the silanol group (SiOH), silicon is seen as a viable material for LOCs devices³⁸.
There are several techniques such as photolithography, soft lithography, laser micromachining, screen printing, 3D printing, and nano imprinting for fabrication of LOC devices³⁹,⁴⁰. The term chip refers to the initial manufacturing process, which used an enhanced type of photolithographic etching to create computer microchips and allowed for uniform control over the sizes and shapes of surface features³⁹. The various steps involved in photolithography method of LOC manufacturing is illustrated in Fig. 3.
Usually, microfabrication methods taken from the semiconductor industry are used to create LOC devices³⁷ and the general fabrication process are represented in Fig. 4. The following are the primary steps in LOC fabrication:
• Design: Computer-aided design (CAD) software is used to create the functionality and layout of the LOC devices. Reaction chambers, integrated sensors or detectors, and microfluidic channels are all part of the design.
• Master Fabrication: The planned LOC device is made into a master mould by employing either soft lithography or photolithography methods. For the microfluidic structures to be replicated, the master acts as a template.
• Replication: Polydimethylsiloxane (PDMS), a flexible and optically transparent polymer, is one of the materials used to recreate the microfluidic structures from the master mould. The entire LOC device is then created by bonding the duplicated structures to a substrate, like glass or plastic.
• Integration: As required, further parts are added to the LOC device, such as electrodes, sensors, or microvalves. These parts make it possible to identify and analyse samples as well as regulate and track fluid flow.
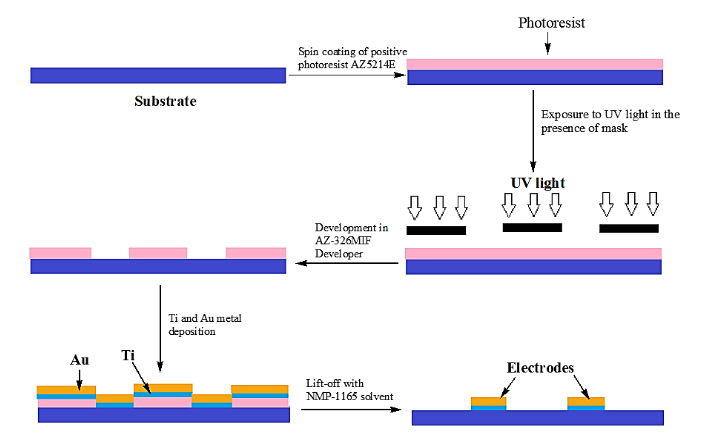
Fig. 3 Steps involved in photolithography method for fabrication of LOC⁴¹.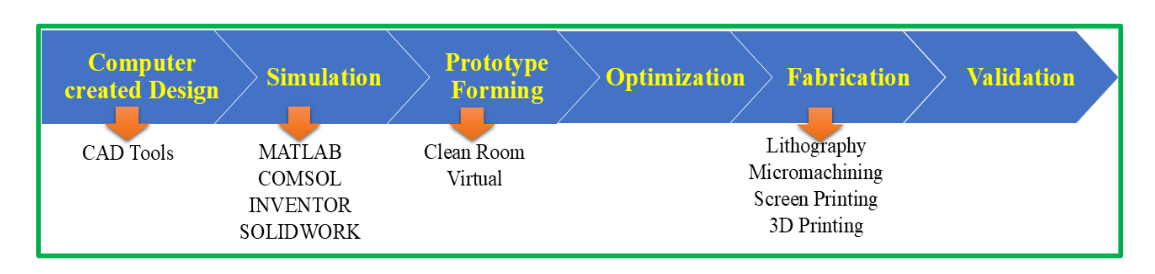
5. Nanoscale materials in fabrication of lab-on-a-chip
The choice of materials becomes crucial because LOC devices are meant to carry out very specialized tasks. This might affect the inherent characteristics of the device as well as the construction method that is chosen. The literature provides an invaluable summary of materials for LOC and microfluidics¹¹,¹². Here, few materials are briefly emphasised.
Polydimethylsiloxane
The PDMS, a silicon-based elastomer that cures from 40 °C to 70 °C, provides a practical and affordable platform for LOC. The PDMS is easily sealed, has good flexibility, gas permeability, and a low surface tension. Its high absorption capability, however, makes surface modification necessary.
Epoxy resins (SU-8)
The benefits of thermosetting materials, such as their excellent durability at high temperatures, chemical susceptibility, and transparency, are somewhat outweighed by their expensive cost.
Silicon
Many characteristics of silicon crystals are similar to those of glass, but unlike amorphous glass, which has spherical walls, silicon crystals have vertical sidewalls and opaque chips. Additionally, silicon chips have good resistance to solvents and thermal stability.
Glass
The major advantages of glass include optical transparency, chemical resistance, electroosmotic mobility, and poor electrical conductivity. Its main drawbacks, however, are its extreme hardness and high construction costs.
Several alternative metals, metal thin films, thermoplastics such as poly(methyl methacrylate) (PMMA), and hydrogels are emerging as significant materials for LOC applications in addition to the four most popular ones. A new class of LOC devices, known as paper-based microfluidics, uses hydrophobically modified cellulose and cellulose. The very porous structure of this matrix makes it possible to take advantage of capillary activities more successfully. Despite their ease of production, paper LOCs need to be improved in the areas of channel resolution, integration with other components, and, eventually, detection sensitivity. An overview of primary physical-chemical characteristics of these materials as well as the processing methods currently used to create devices are described in Table 1.
Table 1 Characteristics of the materials and processing methods commonly employed in the production of LOC devices.
| Material/Property | Elastomers | Thermosets | Thermoplastics | Silicon/Glass | Hydrogel | Paper |
|---|---|---|---|---|---|---|
| Thermal stability | medium | high | medium to high | very high | low | medium |
| Hydrophobicity | hydrophobic | hydrophobic | hydrophobic | hydrophilic | hydrophilic | amphiphilic |
| Optical transparency | high | high | medium to high | no/high | low to medium | low |
| Chemical resistance | moderate | good | moderate to good | excellent | low | low |
| Microfabrication technique | casting | casting, photopolymerization | thermo-molding | photolithography, laser assisted etching | casting, photopolymerization, 3D bioprinting | photolithography, printing |
| Throughput | high | high | high | medium to high | low to medium | high |
6. Applications of lab-on-a-chip devices
Quick developments in the field of microfluidic systems have made it possible to create low-cost, portable, disposable, and easy-to-use platforms that are ideal for LOC applications and health status monitoring¹⁴,⁴². Multidisciplinary collaboration across engineering, chemistry, physics, biochemistry, microtechnology, nanotechnology, and biotechnology is encouraged via microfluidics-based LOC systems.
The portability of microfluidic devices makes them useful for on-site testing⁴³.
A single microfluidic biosensor can do all the analysis, including preconcentration and treatment, continuous sampling, sample separation and mixing, and more because microsystems are small⁴⁴. Additionally, these microfluidic biosensors enhance analytical performance, throughput, rapid reaction rates, portability, and real-time detection. This makes it possible to convert detection into home-testing.
techniques, which is very desirable in both developed and developing nations⁴⁵. Continuous microfluidic systems have steadily improved their performance during the past 20 years. Today, microfluidics can study biological systems from molecules to tiny multicellular organisms since technology has advanced to that stage. Because microfluidics can detect small amounts of analytes, it has given sensing devices a substantial advantage over previous techniques. This has resulted in an integration of chemical and biological processes on a single platform, with reduction in prices, waste, and energy⁴⁶.
A wide range of research fields, such as (a) system biology and analytical chemistry, (b) drug development and biological screening, (c) clinical diagnostics, (d) POC devices for environmental, and (e) medicinal applications, have recently seen a surge in interest in continuous microfluidics⁴⁵. The benefits of merging biosensors with LOC technology and microfluidics systems have sparked a lot of interest recently⁴⁶. Combining biosensors and microfluidic devices results in a comprehensive and compact substitute for conventional repeated laboratory procedures³. Microfluidic-based biosensors can also lower costs while improving detection sensitivity and specificity when compared to conventional detection techniques⁴⁷,⁴⁸.
The capacity of LOC systems to handle samples and manipulate fluids in combination with extremely sensitive biosensor designs promises to produce powerful instruments for analytical and, more precisely, diagnostic applications⁴⁹. It is beneficial for a number of reasons to combine microfluidic circuits with biosensor designs in order to improve the overall performance of sensing system. The main goal is to improve the transit of analytes from the sample volume to the biorecognition element, particularly for sensing components that are surface-bound. Microfluidic channels can analyse blood droplets or even the contents of individual cells because of their smaller sizes and volumes, which enable them to work with significantly less sample than would otherwise be possible. More importantly, the shorter distances between the biorecognition components and the analyte molecules lead to a significant increase in response time, which greatly improves the conditions for diffusion-limited processes⁵⁰,⁵¹.
Lab-on-a-chip technology is used in many different fields (c.f. Fig. 5), such as:
• Biomedical Diagnostics: For POC diagnostics, LOC devices are especially well-suited, allowing for the quick and precise identification of illnesses, infections, and genetic disorders. Even in environments with limited resources, LOC devices can deliver prompt and accurate diagnostic results by combining sample preparation, amplification, and detection onto a single chip¹⁵,¹⁹.
• Drug Discovery and Development: High-throughput screening of drug candidates is made possible by LOC technology, which speeds up the drug discovery and development process. Drug development workflow efficiency can be increased and the requirement for animal testing can be decreased by using microfluidic devices to test the toxicity and efficacy of medications on tissue models or cell cultures¹¹,²⁰,²⁹.
• Environmental Monitoring: Environmental contaminants like heavy metals, pesticides, and infectious biological agents can be monitored on-site with LOC devices. The LOC devices enable early pollution identification and cleanup by combining sample pretreatment, separation, and detection onto a single chip, allowing for quick and sensitive analysis of environmental samples⁵²,⁵³.
7. Lab-on-a-chip sensors in Pharmacological Applications
In order to eventually replace animals in preclinical testing, microfluidic LOCs may be utilised as drug testing platforms, disease modelling, biomarker identification, and distinct cellular and molecular mechanisms¹¹,⁵⁴,⁵⁵. As a result, LOCs can provide suitable conditions for assessing medication efficacy and toxicity, cellular activity, and drug metabolism⁵⁶. The use of such testing microdevices in pharmacokinetic applications has been examined in a number of studies aimed at developing innovative strategies for customised cancer treatments⁵⁷,⁵⁸,⁵⁹.
For example, it was proposed to use a LOC platform to evaluate up to five drugs against osteosarcoma cells in real time⁶⁰. Other LOC microdevices were developed to perform single-cell cytotoxic and genotoxic experiments and simulate a hypoxic cancer microenvironment⁶⁰. Anti-inflammatory medicines were also assessed using a “small airway-on-a-chip” model of human lung inflammation with dynamic flow conditions⁶¹. It should be mentioned that LOC technology has been authorised by the US Food and Medicines Administration for use in pharmaceutical medicine security testing and screening⁶²,⁶³.
Jiang et al. investigated the possible drug metabolism pathway and assessed drug cytotoxicity using a microwell-based microfluidic chip⁶⁴. It had the advantage of strong integration in a more physiologically appropriate setting and was easy to use. Drug cytotoxicity was assessed using a microfluidic sidewall-attached droplet array, according to Fang et al.⁶⁵. They cultivated cells in a 3D droplet array, which has the benefit of preventing cell adhesion on the chip surface and enabling the execution of several operations on cells within droplets. Additionally, they tested the effectiveness of the anticancer drug doxorubicin, and the results showed that doxorubicin clearly reduces cell viability in a dose-dependent manner. A number of microfluidic chips have been published to assess the cytotoxicity of anticancer medications in various settings⁶⁶,⁶⁷,⁶⁸.
First, a multiple-channel array chip was created, allowing for the management of oxygen tension and the completion of cell-based high throughput toxicity studies for cisplatin and tirapazamine. The outcomes of the experiment demonstrated that cisplatin and tirapazamine exhibit opposing mechanisms of oxygen-dependent cytotoxicity.
By mimicking the composition and operations of human organs, OOC biomimetic systems are proposed as non-traditional models for assessing the efficacy or safety of medications. In vitro co-culture models can mimic the complex interactions between cells in an environment similar to that found in vivo.
By simulating microfluidic dynamics, these devices can also be used in medically appropriate ways. Additional advantages of cell co-cultures on the chip include regulated medication administration, sensor integration on the same platform, microscopic super-resolution analysis, and high-throughput analysis with reduced costs and time⁶⁹. One disadvantage of these platforms is that they are designed for specific uses, which prevents them from being used in a general way⁵⁸.
According to the standard guidelines, it may be crucial to concentrate on simulating several organs in order to identify genuine advantages and link the OOC models with particular local tissue structures and cellular phenotypes in order to eventually replicate human physiology in vivo [126]. As a result, standard open technology platforms can be obtained by assembling adaptable models for fit-for-purpose OOC with identical chemical and biological modules⁷⁰. Chips composed of various materials can be used to test the toxicity or effectiveness of medications using the OOC method. Glass or PDMS biochips are the best options for creating the right circumstances for a variety of tests that show how sensitive the organs are to medications.
These materials provide transparency for optical interrogation and may offer qualities similar to the physiological environment, making it possible to create replicas of body components. They are also very easy to process into complicated shapes⁷¹.
The use of these platforms in drug tests and pharmacological screening studies has the benefit of lowering the number of experiments for many organ types, including the heart, kidneys, liver, lungs, and central nervous system⁷², as well as cancer research⁷¹. Another benefit of LOC microfluidic devices is their ability to evaluate medications with low permeability⁷². Innovative medication delivery methods are still desperately needed, and microfluidics offers a state-of-the-art method for this.
Many studies have proven the benefits of LOCs over traditional techniques for the synthesis of sophisticated delivery systems. Fontana et al.⁷³ provided an overview of droplet microfluidic techniques as a potent tool for the creation of monodisperse drug delivery systems, including liposomes, polymersomes, microcapsules, and microspheres.
In contrast to conventional (2D, static) pharmaceutical assays, Cavero et al. described the advantages of OOCs microdevices for human-predictive biological insights on drug candidates in an Expert Opinion on Drug Safety review paper. In addition to discussing the importance of these OOC platforms for drug research and development, the authors present a wide range of them, including those related to cancer, lung, blood-brain barrier, heart, intestine, kidney, liver, pharmacokinetics, placenta, and vessel-on-chip⁷⁴.
The following provides some clear and particular instances of testing methodologies that may concentrate on one drug–one organ⁷⁵, multiple drug–one organ systems⁷⁶, and combinations of these. Kim et al.⁷⁷ investigated the pharmacokinetic profile that reduces the nephrotoxicity of gentamicin under dynamic conditions in a kidney-on-a-chip model. Using a specialised OOC, the study aimed to bridge the renal clearance gap between people and animals. Gentamicin was discovered to change cell-cell junctions, increase membrane permeability, and reduce cell viability, particularly when exposed at low levels for an extended period of time.
For the assessment of cardiovascular toxicity, artificial endothelialized myocardium and heart-on-a-chip models were created using three-dimensional (3D) bioprinting⁷⁸. After that, the dose-dependent reactions of endothelial cells and cardiomyocytes to doxorubicin exposure were assessed. Using multi-material 3D printing, Lind et al.⁷⁹ created cardiac micro-physiological models with strain sensors integrated into microarchitectures. Over a four-week period, the OOC platform was validated by analysing the mechanical responses of human stem cell-derived laminar cardiac tissues and the effects of isoproterenol and verapamil medications.
To replicate the intricacy of in vivo physiology, Phan et al.⁸⁴ suggested a vascularised OOC platform for extensive drug screening. In order to successfully identify both anti-angiogenic and antitumor medicines, a number of arrays of vascularised micro-tumors were developed and evaluated against up to twelve FDA-approved anti-cancer medications.
In a multi-organ human-OOC model system, the impact of hepatic metabolism on off-target cardiotoxicity was examined⁸⁰. To evaluate terfenadine and fexofenadine medications, which may have cardiac side effects reliant on hepatic metabolism, human primary hepatocytes were co-cultured with iPSC-derived cardiomyocytes. In order to study the toxicity of drug metabolites, Theobald et al. reported creating a liver–kidney-on-chip model⁸¹.
The technology used for in vitro drug screening makes it possible to simulate the flow-dependent interaction between several organ-specific cell types. The effectiveness of this OOC method was confirmed by the toxicity assessment of benzo[a]pyrene and aflatoxin B1 medications. To screen for organophosphate toxicity, a 3D tetra-culture brain-on-chip platform was suggested⁸².
By assessing the impact of drugs on barrier integrity, the study demonstrated the great usefulness of such platforms. However, Ishernharen et al. examined the growing use of OOCs in quantitative clinical pharmacology evaluation. Advances in the microphysiological system, such OOC technology, are said to have the potential to better personalise treatments, forecast drug effects, and plan preclinical and clinical trials.
A biomimetic OOC system, which mimics the biology and physiology of human organs, has demonstrated more benefits than conventional methods for medication efficacy and testing. It is explained how a “human-on-chip” system can more accurately assess the toxicity and efficacy of drugs by simulating intricate and dynamic processes such drug absorption, distribution, metabolism, and excretion.
A detailed assessment of the integration of microfluidic LOCs with pharmacological/toxicological experiments and pharmaceutical analysis was recently conducted by Ai et al.⁸⁵. The scientific community’s efforts to create “Pharm-LOC” systems that can handle the entire spectrum of pharmacological advancements, from post-marketing product management to recent drug discovery, were compiled by the authors.
Applications including drug separation and analysis on a chip, the creation of novel tools for pharmacological/toxicological models on a chip, and the use of chip-based models for drug safety and efficacy screening were the main topics of their literature review.
Several perspectives for the future challenges and breakthroughs related to Pharm-LOC advances, such as automating drug discovery, precision nanomedicine, and personalized therapy, are then highlighted.
However, its poor resemblance to real in vivo tissues and the lack of development of human disease models may be a bottleneck in the therapeutic usage of the OOCs models⁸⁶.
One of the new applications of LOC technology that has garnered increased attention is the importance of microfluidics for pharmaceutical science. In response to the need for LOC technologies for pharmaceutical science, Pharm-LOC was created. It represents chip-based platforms for the full chain of pharmacy applications, from drug discovery to post-marketing product management.
All aspects of chip-based ideas, procedures, and tools for pharmaceutical production, pharmacological/toxicological testing, and analysis are included in Pharm-LOC. This review focuses on the latest advancements in the use of LOC for pharmaceutical testing and analysis. The primary topics include the creation of innovative pharmacological and toxicological models on a chip, the isolation and analysis of drug molecules on a chip, and the use of chip-based models to assess the safety and effectiveness of drugs. We also attempt to provide an overview and outlook on the difficulties and potential innovations of Pharm-LOC development.




