Long-COVID: Clinical Development and Assessment Methods
Analysis of the clinical development landscape targeting long-COVID and evolution of clinical outcome assessment methods – An industry perspective
Pandey, Ramesh Chandra¹; Kumar, Saurabh¹
- Real-World Evidence, IQVIA Consulting Services, Mumbai, India
OPEN ACCESS
PUBLISHED:31 March 2025
CITATION: Pandey, RC. and Kumar, S., 2025. Analysis of the clinical development landscape targeting long-COVID and evolution of clinical outcome assessment methods – An industry perspective. Medical Research Archives, [online] 13(3). https://doi.org/10.18103/mra.v13i3.6320
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i3.6320
ISSN 2375-1924
ABSTRACT
Long-COVID is the term used for health complications seen in patients recovered from acute coronavirus disease-19 (acute COVID-19) illness. The spectrum of clinical symptoms, involvement of various organ systems, and pathophysiological mechanisms have been reviewed extensively in the literature. The pharmaceutical industry has responded to the rising disease burden due to long-COVID through clinical trials addressing specific health concerns. For this analysis, we extracted the clinical trial data from the trial registry clinicaltrials.gov using keywords specific to long-COVID OR post-COVID or post-acute sequelae of COVID-19. This universe of 5146 trials was subjected to step-by-step screening to include industry-sponsored clinical studies addressing long-COVID. We analyzed 74 long-COVID-related clinical trial activities by the pharmaceutical industry as a landscape for the evolution of the trends, clinical outcome assessment methods, and future directions. There are nearly 100 clinical studies conducted to address long-COVID since the pandemic so far. Year by year, consistent investment by pharmaceutical companies suggests that the industry recognizes the unmet needs in this field. An overview of innovative approaches in the landscape, like novel biologic products and medical technologies, has been captured. The investigators have addressed the complexity of health complications through novel assessment methods such as patient-reported outcomes (PRO) and post-COVID related functional assessment scales. Here, we highlight the top PROs and functional scales being used in these clinical studies. The lack of a specific diagnosis method remains the currently unsolved puzzle, and a prerequisite for further research and clinical development in this space.
Keywords: Long COVID, Post-COVID-19, Post-COVID Syndrome, Clinical development, Clinical outcomes, Industry
INTRODUCTION
Coronavirus disease-2019 (COVID-19) is an upper and lower respiratory tract infection caused by SARS-CoV-2 which caused a pandemic with more than 700 million cases and over 7 million deaths reported worldwide (https://data.who.int/dashboards/covid19/). Experts believe that these numbers are an underestimate due to lower and inaccurate reporting from LMIC (low- and middle-income countries)¹. Particularly, the incidence surged in three distinct waves during the period of three years between 2020 and 2022, primarily driven by different SARS-CoV-2 variants prevalent at that time²,³. The opportunities of longer follow-up post-pandemic, in patients recovered from acute COVID-19 disease resulted in identification of distinct health concern – post-COVID-19 syndrome or long-COVID. The National Institute for Health and Care Excellence (NICE; UK) defines the post-COVID-19 syndrome (or post-COVID syndrome) as a set of persistent physical, cognitive, and/or psychological symptoms that continue for more than 12 weeks after illness and which are not explained by an alternative diagnosis (https://www.nice.org.uk/guidance/ng188). Alternate definitions further sub-classify Post-COVID Syndrome into Long post-COVID symptoms and Persistent post-COVID symptoms, depending on the duration of persistence and has been reviewed elsewhere earlier⁴. However, in the literature Long-COVID has been often used interchangeably with post-COVID syndrome.
Previously, Umesh et al. (2022) reviewed multi-functional pathophysiology such as pulmonary, neuropsychological, and cardiovascular complications, as well as dysfunctional gastrointestinal, endocrine, and metabolic health, which were responsible for health concerns in long-COVID patients⁴. However, most of the industry-sponsored studies were focused on pulmonary symptoms. Although the epidemiological trends suggested a rise in cardiovascular complications, they were not addressed by most ongoing clinical studies at that time⁴. Being a chronic condition, a longer follow-up period post-pandemic provides an opportunity to revisit the initial understanding. It is essential to re-assess the landscape after 3–4 years of the pandemic waves with respect to an emerging understanding of long-COVID as a health concern, the pharma industry’s innovation and investments to address them, and new ways to evaluate health outcomes from a real-world perspective. In this study, we highlight the trends in the landscape of industry-sponsored clinical activities (observational and interventional studies) addressing long-COVID over the period, based on the data from a clinical trial registry. We also explore deeper into the study designs and review the methods to assess the clinical outcomes that have evolved with greater emphasis on patient-reported outcomes. In addition, we also discuss the recent trends in understanding of long-COVID with respect to additional understanding of pathophysiological mechanisms and how the lack of specific diagnosis biomarkers for long-COVID is a barrier to further advancement in this field.
Methods
This analysis was conducted in three steps: 1) Access a clinical trial registry to extract information about industry-sponsored studies as a dataset, 2) Sequential shortlisting and curation of the data set, and 3) Detailed analysis of the clinical studies that qualified the inclusion criteria, including type of assets, phase of development etc.
Clinical trial universe was assessed from the clinical trial registry maintained by National Library of Medicine (www.clinicaltrials.gov). The search string consisted of words and synonyms such as SARS COV-2, COVID-19, post-COVID-19, Post-acute COVID-19, Post-COVID Syndrome, COVID-19 sequelae, long COVID, persistence of symptoms and long-term health consequences (Figure 1). Timeline filter applied for the data extraction was the trial start date of 01/01/2021 or later. The data was downloaded from the registry on 30th Nov 2024 and 2nd Jan 2025 and merged. Only active clinical trials were considered for the analysis, excluding studies that were terminated, suspended, withdrawn or where the exact status was not available.
As the next step, the data universe was subjected to step-by-step shortlisting to include industry-sponsored clinical studies focusing on post-covid syndrome or long COVID (Figure 1). The shortlisting criteria included exclusions like academic sponsorship, vaccines, studies focusing on acute-COVID-19, behavioral, physiotherapy or dietary interventions, and studies where the relevance or intent of the study in relation to long-COVID could not be established by the study description. The dataset was manually curated to address the inconsistency of the classification, such as industry sponsorship, trial focus, terminologies used in the studies etc.
Finally, the data available in the included trials were analyzed in detail to review the development stage and classify innovative approaches like medical devices, algorithms, biologics, etc., based on the information retrieved from the clinical trial registry.
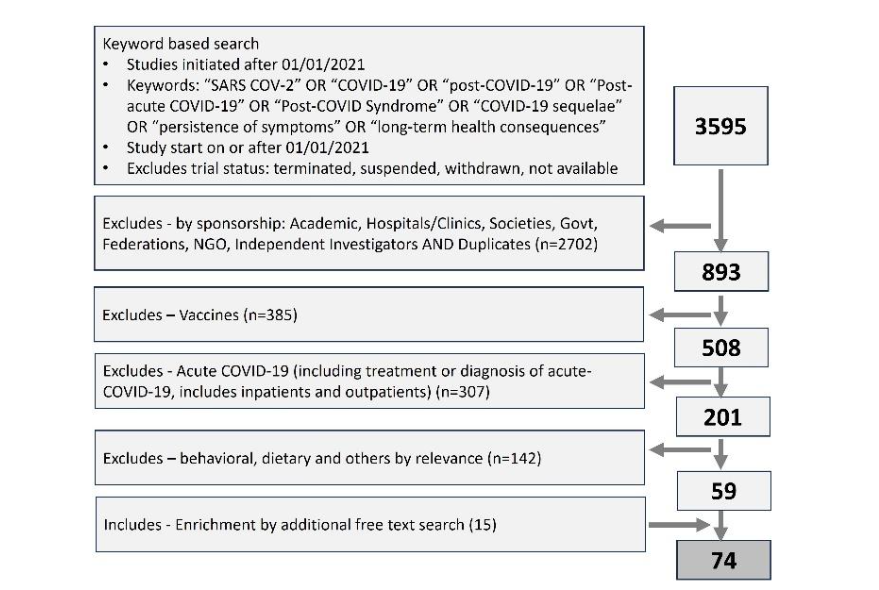
Figure 1: Prisma diagram showing the extracting and step-by-step shorting of industry sponsored clinical studies focusing on long-COVID. Clinical trial data was extracted from clinicaltrials.gov, a trial registry maintained by national library of medicine, USA.
A. INDUSTRY-SPONSORED CLINICAL STUDY LANDSCAPE ADDRESSING LONG-COVID
We analyzed 74 new initiated clinical studies, from the registry with start date on or after 01 Jan 2021. Among the 74 new clinical studies sponsored or initiated by Industry that were initiated after 2021, 81% were interventional and 19% were observational (Figure 2-A). Year-on-year (YOY) landscape demonstrates that long-COVID remains a focus for pharma industry (Figure 2-B). The landscape shows that an average of ~20 new industry-sponsored clinical studies were initiated annually over the last three years (17-25) and three studies are pre-planned for 2025 (figure 2). The type of interventions under assessment include small molecule drugs, biologic assets such as monoclonal antibodies or cell/gene therapies, MedTech interventions such as neurostimulation devices or digital therapeutics etc. (Table 1). Interestingly, there are a few innovative approaches involving leveraging of data/algorithm or machine learning to aid in the diagnosis or prognosis of the long-COVID.
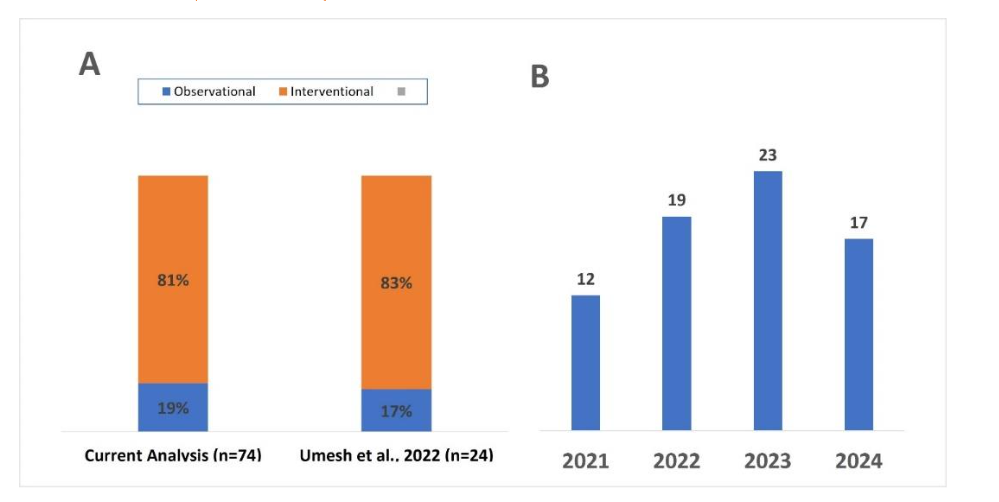
Figure 2: Overview of clinical development landscape addressing long-COVID. A) Comparison of the industry-sponsored study design with previously published report. B) Year-on-year overview of new studies initiated/sponsored by pharma companies.
Table 1: Overview of interventional assets for clinical development in long-COVID.
| Asset Study Stage | Drugs/Small Molecules | Biologic agents | Devices/Procedures |
|---|---|---|---|
| Phase III (and phase II/III) | Apabetalone, Fluvoxamine Maleate, LAU-7b, Losartan, Metformin ER/Metformin, Prospekta, Prednisolone, Sodium pyruvate (nasal spray), Ramatroban, Testofen | IgPro20, Longidaza®, Meplazumab | Devices: Wearable device, MouthLab™, Inogen One® G4, Seraph® 100, Vielight Neuro RX, Tens Eco Plus, Vimida, RD-X19™ light therapy device, Adhera®, Viraximmune Fluorospot Assay |
| Phase II (and phase I/II) | Abrocitinib, AXA1125, BC 007, Ensitrelvir, Efgartigimod, Leronlimab, Medicabilis Cannabis, Nirmatrelvir, Plitidepsin, Rintatolimod, RSLV-132, Ritonavir, Shenlingcao, S-1226, Tonabersat, TNX-102 SL, Temelimab, Vericiguat, Zofin | COVI-MSC, AER002, RegeneCyte | |
| Phase I or earlier | VIX001 | Ampion, Autologous Stem Cells, Cord Blood Stem Cells, MSC-derived exosomes | Procedures: Splenic Ultrasound Stimulation, TENS – high/low-dose, Electrical Stimulation – Sham, Stellate Ganglion Block |
| Phase IV/observational/ na* | Ethyl methyl hydroxypyridine succinate with Meldonium, KB109, Pimozide |
*Reported as per the information available in the clinical trial registry. For generics or already marketed drugs, phase IV studies are being conducted. For observational studies, medical devices, procedures or nutraceuticals clinical development phases does not apply.
Notes:
• This table excludes approaches for diagnosis, monitoring, database/algorithms etc.
• Biologic agents include monoclonal antibodies, stem cells, biological fluid derivatives including enzyme derivatives.
Although the landscape is dominated by small pharma/biotech sponsors, there are also a few large pharma players with active assets and advanced stage trials such as Pfizer (Abrocitinib and Nirmatrelvir, Phase 2) and Bayer (Vericiguat, Phase 2). Similarly, subcutaneous injection of L-proline-stabilized 20 % human immunoglobulin available as IgPro20 (CLS Behring) is being evaluated in phase 3 study for preventing Post-COVID Postural Orthostatic Tachycardia Syndrome. Advanced innovative approaches in this space include Longidaza® or Bovhyaluronidase Azoximer (NPO Petrovax Pharma), an enzyme conjugate of PH20 hyaluronidase currently in phase 3 trial for post-covid related pulmonary sequelae (https://longidaza.com) and Meplazumab (Jiangsu Pacific Meinuoke Bio Pharma Pvt Ltd), humanized anti-CD147 IgG2 monoclonal antibody that recently completed phase 3 study for preventing post-COVID symptoms. In addition, there are studies evaluating stem cells from mesenchymal origin, cord blood and autologous stem cells in post-COVID syndrome. Overall, there are 35 clinical studies for small molecules and 10 studies for biologic products investigating benefits in health conditions of post-COVID patients.
B. PARADIGM SHIFT IN LONG-COVID CLINICAL ASSESSMENT FRAMEWORK.
In the current analysis it is observed that most recent long-COVID related studies leveraged patient-reported outcomes and functional assessment scales to measure the clinical outcomes. These scales incorporate various aspects of health states such as cognitive function, physical function, quality of life, as well as functional assessments like pain index, gastrointestinal, vasomotor, and autonomic symptom scores (Table 2).
Table 2: Overview of commonly used PROs and Functional scales in long-COVID trials.
| PRO Scales | Brief Description | Reference Clinical Trial IDs |
|---|---|---|
| PROMIS-29 Variants (including PROMIS-29, PROMIS-cognitive function, PROMIS® Fatigue Score (T-Score)) | PROMIS® (Patient-Reported Outcomes Measurement Information System) is a set of person-centered measures that evaluates and monitors physical, mental, and social health in adults and children, can be administered through computer based or paper-based methods (www.healthmeasures.net). | NCT05877508, NCT06161688, NCT06163690, NCT05497089, NCT04944121, NCT05668091, NCT05592418, NCT05576662, NCT05472090, NCT06590324, NCT05497089, NCT05668091, NCT05947617, NCT05918978, NCT05633407, NCT05592418, NCT04678830 |
| Fatigue Severity Score Scale (FSS) | The FSS is a self-reported, nine-item instrument designed to assess fatigue as a symptom of a variety of different chronic conditions and has ability to measure fatigue severity or to differentiate fatigue from clinical depression, since there are similarities in the response to both. Item analysis showed excellent internal consistency and reliability¹⁰. | NCT05795816, NCT05074888, NCT06128967, NCT06251518, NCT06097442, NCT05823896, NCT04997395, NCT05228899, NCT05175339, NCT06128967, NCT05697640 |
| European Quality of Life-5 Dimensions (EQ-5D) | The EQ-5D questionnaire is used worldwide as a patient-reported outcome (PRO) instrument for the measurement and valuation of health. Several variants of EQ-5D now exist, including versions with three and five levels of severity and one for adolescent. It can be used for multiple health conditions and interventions. (www.euroqol.org) | NCT05823896, NCT05631171, NCT06097442, NCT05823896, NCT04997395, NCT05979435, NCT05668091, NCT05857124, NCT06597396, NCT06147050, NCT06383819 |
| Patient Global Impression of Change (PGI variants) (Includes sub-scales PGI-C, PGI-S and PGI-I, for change, Severity, and Improvement respectively) | The PGI, is the PRO counterpart to the Clinical Global Impression scale, (CGI), which was published in 1976 by the National Institute of Mental Health (NIMH, US). It consists of one item based on the CGI and adapted to the patient. It mainly measures Change in clinical status (PGI-C) but can also measure disease Severity (PGI-S) or disease Improvement (PGI-I). It is managed and distributed by Mapi Research Trust (ePROVIDE™ platform) (https://eprovide.mapi-trust.org). | NCT05576662, NCT05840237, NCT04944121, NCT05999435, NCT05668091, NCT05918978, NCT05633407, NCT05205577, NCT06161688 |
Modified Medical Research Council (mMRC) Dyspnea Scale
mMRC Dyspnea Scale is a simple, self-reported scale widely used in clinical research to assess the severity of breathlessness and disability in patients with respiratory diseases²¹.
Reference Clinical Trial IDs:
NCT05601180, NCT05121740, NCT06097442, NCT05713266, NCT05823896, NCT05947617, NCT06383819, NCT04949386
Chalder Fatigue Scale (CFS or CFS-11)
CFS is also referred to as the CFQ, to differentiate it from chronic fatigue syndrome or CFS. It was created by Trudie Chalder at King’s College London to measure severity of tiredness in fatiguing illnesses. CFS-11 consists of 11 items are answered on a 4-point scale and measuring two dimensions – physical fatigue and psychological fatigue²².
Reference Clinical Trial IDs:
NCT05840237, NCT05684952, NCT06251518, NCT06189006, NCT05152849, NCT06147050, NCT06251518
Montreal Cognitive Assessment (MoCA) scale
The MoCA Test is a sensitive and validated cognitive screening tool for early detection of mild to severe cognitive impairment. MoCA Full is widely used internationally by various medical professionals and available in over 100 languages and dialects. MoCA is also available as quick and fun digital self-assessment tool for the general public (www.mocacognition.com).
Reference Clinical Trial IDs:
NCT05212831, NCT05947617, NCT05823896, NCT05939622, NCT05689827, NCT05175339, NCT05592418
Generalized Anxiety Disorder – 7 item scale (GAD-7)
GAD-7 is a self-administered 7-item questionnaire designed for screening and measuring the severity of generalized anxiety disorder. The GAD-7 is validated and adopted due to its strong psychometric properties, including high internal consistency and good test-retest reliability.
(https://psychology-tools.com/test/gad-7)
Reference Clinical Trial IDs:
NCT05204615, NCT05497089, NCT04997395, NCT05947617, NCT05175339, NCT06251518, NCT06251518
Patient Health Questionnaire – 8 item scale (PHQ-8)
The PHQ-8 is a valid diagnostic and severity measure for depressive disorders in clinical research and population-based studies. It can be used as diagnostic algorithm or for defining current depression state in general population²³.
Reference Clinical Trial IDs:
NCT05204615, NCT05497089, NCT04997395, NCT05947617, NCT05175339, NCT06251518
The Composite Autonomic Symptom Score (COMPASS-31)
The COMPASS-31 scale measures neurodegenerative system symptoms through 31 patient-reported questions through six weighted domains: orthostatic intolerance, vasomotor, secretomotor, gastrointestinal, bladder and pupillomotor functions²⁴. COMPASS is developed by researchers at Mayo Clinic, Rochester (USA) and applies a norm-simplified scoring algorithm and is suitable for widespread use in autonomic research and practice²⁵.
Reference Clinical Trial IDs:
NCT05633407, NCT05823896, NCT06524739, NCT05918978, NCT05697640, NCT05877508
Fatigue Assessment Scale (FAS-10)
The FAS-10 scale is a 10-item self-report scale evaluating symptoms of chronic fatigue and can be quickly administered within 2 minutes. The FAS treats fatigue as a unidimensional construct and does not separate its measurement into different factors. A copy of the tool can be requested by the developer by direct correspondence²⁶.
Reference Clinical Trial IDs:
NCT05204615, NCT05631171, NCT05939622, NCT05689827, NCT05857124
Post-COVID-19 Functional Status Scale (PCFS)
The PCFS scale focuses on relevant aspects of daily life during follow-up after the COVID infection and objectively determines the degree of disability. The scale is intended to be used in addition to other (patient-reported) instruments; not as a stand-alone instrument replacing other relevant outcome measures²⁷. The tool is intended to be assessed at specified time points (as described in the manual: 1) at discharge; 2) in the first weeks after discharge, e.g. 4–8 weeks post-discharge; and 3) after 6 months). It can be assessed by either medical experts or trained interviewers during a short-structured interview or may be self-reported by the patient. It is available free of cost at OSF platform (https://osf.io/qgpdv/).
Reference Clinical Trial IDs:
NCT06097442, NCT05713266, NCT06590324, NCT05497089
Multidimensional Fatigue Inventory (MFI-20)
The MFI is a self-reported 20-item scale designed to evaluate five dimensions of fatigue: general fatigue, physical fatigue, reduced motivation, reduced activity, and mental fatigue. The scale leverages positively and negatively oriented items in a scale to avoid response bias (acquiescence effects); however, this procedure can also reduce the reliability of the scale²⁸.
Reference Clinical Trial IDs:
NCT05689827, NCT05939622, NCT05689827
PedsQL-MFS
The PedsQL™ Multidimensional Fatigue Scale was designed as a generic symptom-specific instrument to measure fatigue in patients with acute and chronic health conditions as well as healthy school and community populations, including pediatric and adults (https://eprovide.mapi-trust.org).
Reference Clinical Trial IDs:
NCT06147050, NCT05823896, NCT06147050
The COVID-19 Yorkshire Rehabilitation Scale (C19-YRS)
The C19-YRS is the first condition-specific, validated scale for patient assessment, monitoring and to capture persistent symptoms of Long COVID/Post-COVID syndrome. C19-YRS is a 22-item scale that includes symptom severity, functional disability, the overall health score, and additional symptoms. There is also a modified C19-YRS (C19-YRSm) based on 17-item questionnaire, developed with same subscales. There is also a Digital C19-YRS for personal use. The digital platform ELAROS has been developed by the digital health company, University of Leeds and NHS Trusts⁵.
Reference Clinical Trial IDs:
NCT04997395
COVID-19 Yorkshire Rehabilitation Scale (C19-YRS) is a post-COVID-19 specific scale developed by NHS scientists to estimate symptoms severity score, functional disability score and global health score⁵. However, the most used assessment frameworks are the Patient-Reported Outcomes Measurement Information System (PROMIS®) or its variants, and Patient Global Impression (PGI or its variants such as PGI-C, PGI-S) and European Quality of Life-5 Dimensions (EQ-5D) (Table 2). Composite Autonomic Symptom Score-31 (COMPASS-31), Montreal Cognitive Assessment (MoCA) scale, and post-COVID-19 Functional Status Scale (PCFS) are other validated methods widely used. Fatigue is recognized as the most consistent complication in post-COVID patients across studies and has been used widely in the clinical studies. In long-COVID related clinical trials, commonly used fatigue assessment tools include Fatigue Severity Score (FSS) scale, and Chalder Fatigue score (CFS-11). Similarly, commonly used psycho/cognitive functional scales for post-COVID assessments are Montreal Cognitive Assessment (MoCA) scale, PROMIS-cognitive function scale (a variant of PROMIS®), Generalized Anxiety Disorders Scale (GAD-7), Patient Health Questionnaire depression scale (PHQ-8), Depression Anxiety Stress Scale (DASS-21) etc. (Table 2). In addition to these global health assessment scores, there are also organ system-specific functional assessment tools such as mMRC Dyspnea Scale, St George’s Respiratory Questionnaire (SGRQ) specific to respiratory complications including asthma/COPD and bronchiectasis. Moreover, in addition to the specific outcome assessments, standard quality of life assessment methods such as Modified GSQ-30, PedsQL-MFS, SF-36 QoL, SF-12 QoL and WHOQOL-100 can also be used at the investigator’s discretion.
Discussion
Temporal patterns of COVID-19 acute onset showed four distinct waves of pandemic, with the fourth or the last wave occurring between July 2021 and January 2022 in the US⁶. However, COVID-19 cases continue to be reported – with 161,264 positive cases and 2,935 deaths monthly (as of 5th Jan,
(2025), mostly coming from European region (https://data.who.int/dashboards/covid19/). While under-reporting of cases mainly from (LMIC) as well as developed countries, cannot be ruled out, these numbers are still concerning. Pharma industry has responded to these public health concerns and initiated nearly 100 trials in the last 5 years. Umesh et al, 2022 reported that there were 24 active clinical studies by August 2021, sponsored by pharma industry⁴. Here, our analysis showed that 74 new clinical studies were registered on the clinicaltrials.gov portal since 2021, including 19, 23, and 17 studies initiated in the year 2022, 2023, and 2024 respectively, suggesting continued thrust from industry in the area. Our findings were consistent with previous findings that more than three-fourths of the industry sponsored studies are interventional, which is the conventional study design for the development of new drugs. Observational studies are conducted to observe epidemiological patterns or disease prevalence and are mostly driven by academic sponsorship (Figure 1-A)⁴.
The clinical development landscape reveals interesting insights with respect to innovative interventional approaches to address long-COVID as a health concerns. This includes assets such as enzyme conjugates (Longidaza®), antiviral agents (Nirmatrelvir), kinase inhibitors (Abrocitinib) and processed/stabilized immunoglobulins (IgPro20), among others. Some of these candidate drugs such as Meplazumab and IgPro20 were previously approved in US and EU market⁷,⁸. Meplazumab has shown its efficacy in acute-COVID-19, in terms of reducing virus load and cytokine levels⁹. IgPro20 is approved for primary immunodeficiency disease and tolerated well in adult as well as children¹⁰. Interestingly, emerging technological facets of innovation such as wearable devices, machine learning/algorithms and non-invasive procedures like neuromodulation are also being studied to prevent and/or treat long-COVID. This evidence reinforces that continuous innovation, and investments are being made to address long-COVID as a public health concern.
Characteristics of long-COVID have been reviewed in the literature extensively, through systematic literature reviews, meta-analyses, and retrospective large database analyses⁴,¹¹,¹². Reports have suggested that pathophysiological damages caused by acute-COVID could involve neurological, pulmonary, cardiovascular, gastrointestinal, endocrine, and metabolic systems and result in persistent clinical symptoms up to two years post initial diagnosis⁴,¹³. The exact proportion of these complications varies significantly depending on data sources, demographics, patient characteristics, and study design. Hyper-inflammatory changes and cytokine storm caused by acute-COVID-19 disease were hypothesized as key mechanisms resulting in multiple-organ dysfunctions and complications lasting over a longer duration⁴. Longitudinal follow-up of COVID-19 hospitalization survivors suggested that nearly 90% of the individuals with post-COVID sequelae returned to their respective work profiles by two years but had lower quality of life, worse exercise capacity, mental health abnormalities, and increased health-care resource usage after discharge¹⁴.
Intriguingly, the initial hypothesis of long-COVID impact on fertility and oncogenic pathogenesis did not replicate for real-world reporting over the longer observation period after COVID-19 pandemic waves¹⁵,¹⁶. The methods to assess the clinical outcomes has seen significant advancement from conventional specific symptom-based approach to patient-reported outcome based functional scales. Earlier studies related to long-COVID, assessed the clinical outcomes based on organ system-specific pathology and specific clinical endpoints⁴. In the current landscape, we observed that most of the studies have adopted to assess the disease progress through standardized patient reported outcome (PRO) scales or functional assessment tools that incorporate inputs from HCPs and/or Patients. These scales have allowed the investigators to look at long-COVID as a complex health condition, instead of the isolated perspectives. These advancements correlate with improved understanding of most symptoms such as fatigue in long-covid patients, may be attributed to several pathophysiological factors such as energy metabolism, neuropathological, cardiovascular system malfunctions acting in tandem⁴,¹³. These factors play a role in addressing complexity of the condition in an integrated manner such that they are often treated as single-dimensional construct during outcome assessment.
Mapi Research Trust (a non-profit organization focusing on patient-centered clinical outcomes research) has emerged as useful resource provider for investigators. Mapi is the exclusive distributor of over 800 COAs across therapy area made available through ePROVIDE™ platform and the PROQOLID™ database of 7000+ validated & published COAs (https://eprovide.mapi-trust.org/about/about-proqolid).
While this analysis has highlighted the recent trends based on the data retrieved from a credible clinical trial registry, there are caveats to this approach. As there are several other clinical trial registries including country-specific, regional, as well as WHO clinical registries, the size of the clinical landscape addressing long-COVID could be much larger. We acknowledge that there may be clinical trials that were initiated before 2021 and still ongoing for longer observation. The current analysis may have missed those studies as well. The current scope excludes academic-sponsored clinical trials and hence may miss the opportunity to be inclusive of studies that were initiated by academic professionals who partner or collaborate closely with the industry but have not revealed the association. Therefore, the analysis and results from the current analysis should be interpreted directionally.
Recently, several studies attempted to identify specific biomarkers that could aid in the diagnosis of long-COVID¹⁷–¹⁹. Unfortunately, there is no consensus around any single or a set of specific biomarkers that could help diagnose long-COVID. This remains a major knowledge gap in the understanding of long-COVID in relation to its pathogenesis as well as a major barrier to specific clinical development in this indication. Most of the symptoms of long-COVID (e.g. depression, fatigue, respiratory dysfunction etc.) are redundant with other health conditions. As per current practice, long-COVID diagnosis is based on correlation of specific symptoms that cannot be explained by any other etiology and a history of acute-COVID infection. This makes the current practice of clinical differential diagnosis vague and directional. Further research is needed to establish specific diagnostic algorithm for long-COVID and etiology-attributable pathogenesis to drive further innovation in this field.
Conclusion
After more than three years since the last COVID-19 pandemic wave, acute-COVID continues to be reported in significant numbers, mainly coming from US/EU regions. Patients who recovered from acute COVID, experience post-acute sequelae or long-COVID impacting physical wellness and quality of life. These health concerns have been recognized by the pharmaceutical industry and significant investments have been made in terms of new clinical studies, innovative approaches to diagnosis or treatment, development and validation of assessment frameworks focusing on long-COVID as a complex health issue. The lack of specific biomarker sets with diagnostic potential remains a significant challenge. An option to specifically diagnose long-COVID, compared to the conventional approach of diagnosis by exclusion of other etiologies will fuel further drug development for long-COVID patients.
Declarations
Conflicts of Interest Statement:
The authors have no conflicts of interest to declare.
Authors are employed by IQVIA.
References
1. Park MB, Ranabhat CL. COVID-19 trends, public restrictions policies and vaccination status by economic ranking of countries: a longitudinal study from 110 countries. Arch Public Health 2022; 80(1):197. DOI: 10.1186/s13690-022-00936-w.
2. Thakur I, Chatterjee A, Ghosh AK, et al. A comparative study between first three waves of COVID-19 pandemic with respect to risk factors, initial clinic-demographic profile, severity and outcome. J Family Med Prim Care 2024;13(6):2455-2461. DOI: 10.4103/jfmpc.jfmpc_1884_23.
3. Bali Swain R, Lin X, Wallentin FY. COVID-19 pandemic waves: Identification and interpretation of global data. Heliyon 2024;10(3):e25090. DOI: 10.1016/j.heliyon.2024.e25090.
4. Umesh A, Pranay K, Pandey RC, Gupta MK. Evidence mapping and review of long-COVID and its underlying pathophysiological mechanism. Infection 2022;50(5):1053-1066. DOI: 10.1007/s15010-022-01835-6.
5. Sivan M, Preston N, Parkin A, et al. The modified COVID-19 Yorkshire Rehabilitation Scale (C19-YRSm) patient-reported outcome measure for Long Covid or Post-COVID-19 syndrome. J Med Virol 2022;94(9):4253-4264. DOI: 10.1002/jmv.27878.
6. Chharia A, Jeevan G, Jha RA, Liu M, Berman JM, Glorioso C. Accuracy of US CDC COVID-19 forecasting models. Front Public Health 2024;12: 1359368. DOI: 10.3389/fpubh.2024.1359368.
7. USFDA. FDA Approves First Drug to Treat Group of Rare Blood Disorders in Nearly 14 Years. FDA News Release 2020
(https://www.fda.gov/news-events/press-announcements/fda-approves-first-drug-treat-group-rare-blood-disorders-nearly-14-years).
8. Lamb YN, Syed YY, Dhillon S. Immune Globulin Subcutaneous (Human) 20% (Hizentra((R))): A Review in Chronic Inflammatory Demyelinating Polyneuropathy. CNS Drugs 2019;33(8):831-838. DOI: 10.1007/s40263-019-00655-x.
9. Bian H, Chen L, Zheng ZH, et al. Meplazumab in hospitalized adults with severe COVID-19 (DEFLECT): a multicenter, seamless phase 2/3, randomized, third-party double-blind clinical trial. Signal Transduct Target Ther 2023;8(1):46. DOI: 10.1038/s41392-023-01323-9.
10. Kanegane H, Imai K, Yamada M, et al. Efficacy and safety of IgPro20, a subcutaneous immunoglobulin, in Japanese patients with primary immunodeficiency diseases. J Clin Immunol 2014; 34(2):204-11. DOI: 10.1007/s10875-013-9985-z.
11. Al-Aly Z, Xie Y, Bowe B. High-dimensional characterization of post-acute sequelae of COVID-19. Nature 2021;594(7862):259-264. DOI: 10.103 8/s41586-021-03553-9.
12. Chen C, Haupert SR, Zimmermann L, Shi X, Fritsche LG, Mukherjee B. Global Prevalence of Post-Coronavirus Disease 2019 (COVID-19) Condition or Long COVID: A Meta-Analysis and Systematic Review. J Infect Dis 2022;226(9):1593-1607. DOI: 10.1093/infdis/jiac136.
13. Franco JVA, Garegnani LI, Metzendorf MI, Heldt K, Mumm R, Scheidt-Nave C. Post-covid-19 conditions in adults: systematic review and meta-analysis of health outcomes in controlled studies. BMJ Med 2024;3(1):e000723. DOI: 10.1136/bmjmed-2023-000723.
14. Huang L, Li X, Gu X, et al. Health outcomes in people 2 years after surviving hospitalisation with COVID-19: a longitudinal cohort study. Lancet Respir Med 2022;10(9):863-876. DOI: 10.1016/S22 13-2600(22)00126-6.
15. Kaur H, Chauhan A, Mascarenhas M. Does SARS Cov-2 infection affect the IVF outcome – A systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol 2024;292:147-157. DOI: 10.1 016/j.ejogrb.2023.11.027.
16. Tanriverdi O, Alkan A, Karaoglu T, Kitapli S, Yildiz A. COVID-19 and Carcinogenesis: Exploring the Hidden Links. Cureus 2024;16(8):e68303. DOI: 10.7759/cureus.68303.
17. Lai YJ, Liu SH, Manachevakul S, Lee TA, Kuo CT, Bello D. Biomarkers in long COVID-19: A systematic review. Front Med (Lausanne) 2023;10: 1085988. DOI: 10.3389/fmed.2023.1085988.
18. Espin E, Yang C, Shannon CP, Assadian S, He D, Tebbutt SJ. Cellular and molecular biomarkers of long COVID: a scoping review. EBioMedicine 2023;91:104552. DOI: 10.1016/j.ebiom.2023.104552.
19. Thomas C, Faghy MA, Chidley C, Phillips BE, Bewick T, Ashton RE. Blood Biomarkers of Long COVID: A Systematic Review. Mol Diagn Ther 2024; 28(5):537-574. DOI: 10.1007/s40291-024-00731-z.
20. Krupp LB, LaRocca NG, Muir-Nash J, Steinberg AD. The fatigue severity scale. Application to patients with multiple sclerosis and systemic lupus erythematosus. Arch Neurol 1989;46(10):1121-3. DOI: 10.1001/archneur.1989.00520460115022.
21. Bestall JC, Paul EA, Garrod R, Garnham R, Jones PW, Wedzicha JA. Usefulness of the Medical Research Council (MRC) dyspnoea scale as a measure of disability in patients with chronic obstructive pulmonary disease. Thorax 1999;54(7):581-6. DOI: 10.1136/thx.54.7.581.
22. Chalder T, Berelowitz G, Pawlikowska T, et al. Development of a fatigue scale. J Psychosom Res 1993;37(2):147-53. DOI: 10.1016/0022-3999(93) 90081-p.
23. Kroenke K, Strine TW, Spitzer RL, Williams JB, Berry JT, Mokdad AH. The PHQ-8 as a measure of current depression in the general population. J Affect Disord 2009;114(1-3):163-73. DOI: 10.1016 /j.jad.2008.06.026.
24. Yar T, Salem AM, Rafique N, et al. Composite Autonomic Symptom Score-31 for the diagnosis of cardiovascular autonomic dysfunction in long-term coronavirus disease 2019. J Family Community Med 2024;31(3):214-221. DOI: 10.4103/jfcm.jfcm_20_24.
25. Sletten DM, Suarez GA, Low PA, Mandrekar J, Singer W. COMPASS 31: a refined and abbreviated Composite Autonomic Symptom Score. Mayo Clin Proc 2012;87(12):1196-201. DOI: 10.1016/j.mayoc p.2012.10.013.
26. Michielsen HJ, De Vries J, Van Heck GL. Psychometric qualities of a brief self-rated fatigue measure: The Fatigue Assessment Scale. J Psychosom Res 2003;54(4):345-52. DOI: 10.1016/s0022-3999 (02)00392-6.
27. de Jong CMM, Le YNJ, Boon G, Barco S, Klok FA, Siegerink B. Eight lessons from 2 years of use of the Post-COVID-19 Functional Status scale. Eur Respir J 2023;61(5). DOI: 10.1183/13993003.0 0416-2023.
28. Mamyrbaev A, Turmukhambetova A, Bermagambetova S, et al. Assessing psychometric challenges and fatigue during the COVID-19 pandemic. J Med Life 2023;16(10):1527-1533. DOI: 10.25122/jml-2023-0244.

