Impact of Opioid Prescribing on Overdose Deaths: 2006-2022
Relationship Between Patients Dispensed Prescription Opioids, Opioid Treatment Admissions and Overdose Deaths: 2006-2018 and Beyond
Larry Aubry¹, Richard Lawhern²
- independent researcher with no current professional affiliations.
- Subject matter expert on U.S. regulation of prescription opioid analgesics and clinicians who employ them in pain management. He has authored or co-authored
- over 250 published papers, articles, and interviews in a mixture of peer-reviewed medical journals and mass media.
OPEN ACCESS
PUBLISHED: 31 March 2025
CITATION: Aubry, L., and Lawhern, R., 2025. Relationship Between Patients Dispensed Prescription Opioids, Opioid Treatment Admissions and Overdose Deaths: 2006-2018 and Beyond. Medical Research Archives, [online] 13(3). https://doi.org/10.18103/mra.v13i3.6440
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i3.6440
ISSN: 2375-1924
Abstract
Background: In August 2022, a paper was published by Larry Aubry and L. Thomas Carr, assessing relationships between prescription opioid sales, opioid treatment admissions and drug-related accidental deaths. This paper refines and expands the previous work by analyzing the relationship between the patients dispensed an opioid prescription to drug overdose deaths and opioid treatment admissions. It evaluates whether there is a statistically significant relationship between opioid prescribing in the current year and the subsequent year (Year + 1) and the year after that (Year + 2) opioid treatment admissions or drug overdose mortality rates. 2020-2022 data are also examined to refine understanding of the primary driving factors in U.S. accidental drug mortality. Aims: This paper seeks to more fully answer the question, “Have prevailing public health policies restricting the availability of prescription opioid analgesics been successful in reducing accidental drug overdoses, either from all drugs or specifically from prescription opioids?” Methods: Linear regression analysis has been applied to data published by the US CDC and the U.S. Food and Drug Administration from 2006 to 2018 – six years before and six years after a peak in U.S. opioid analgesic prescribing. 2020-2022 data from the CDC State Unintentional Drug Overdose Reporting System have been examined for trends and consistency with earlier data. Four measures have been applied: statistical significance of the model (overall P-value), quality of the data fit (R-squared), and the sign of the linear slope coefficient (positive or negative correlation) proportion of patients dispensed an opioid prescription to opioid treatment admissions and to deaths attributed to prescription opioid drugs. Results: No positive correlations were found between the number of patients dispensed an opioid prescription versus present-year, present-plus-year, or present-year-year+2 prescription opioid mortalities, opioid treatment admissions, and any opioid and total overdose deaths. Recent accidental drug-related deaths are dominated by non-prescription opioids, specifically illegal fentanyl and stimulants– not patients dispensed an opioid prescription. Conclusions: Current public health policy restricting the availability of clinically prescribed opioid analgesics has had no discernable effect on opioid treatment admissions or drug overdose/poisoning mortality.
Keywords: Prescription Opioid, Drug Addiction, Drug Overdose Deaths, Opioid treatment admissions.
Introduction:
For the past 40 years, the U.S. has experienced an exponential rise in the number of drug overdose deaths. Since 2021, these deaths exceeded 100,000 annually. Some commentators and policymakers attribute this increase to advertising campaigns such as “Pain as the Fifth Vital Sign” and the diversion of large quantities of pharmaceutical-grade opioids into street markets via “pill mills” during the first decade of the 21st Century. Government officials, including the Director of the United States (US) Centers for Disease Control and Prevention (CDC), have asserted that physicians “over-prescribing” opioid analgesic medications to their patients was a major cause of this debacle.
A Previous Paper: Larry Aubry and L. Thomas Carr published a paper in August 2022 assessing the relationships between opioid prescribing volume, treatment admissions, and drug-related overdose deaths. Figure 1 below offers an apt summary of the findings published in that paper.
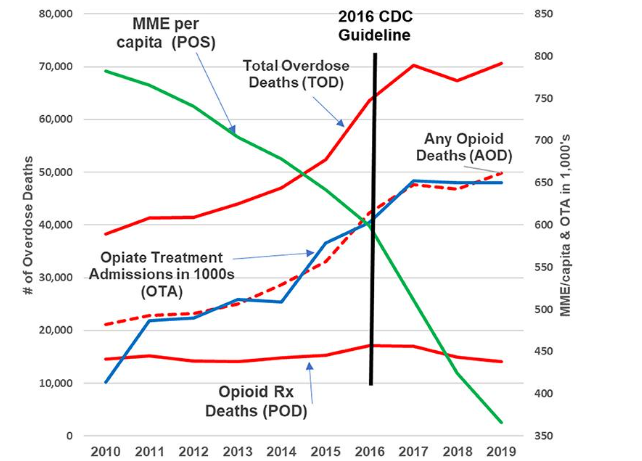
The left axis of Figure 1 displays the Number of U.S. drug poisoning/overdose deaths by year from 2010 to 2019. The right axis of the figure portrays two categories of data: a. MME/Capita (abbreviated “POS”) is the gross volume of opioid analgesics prescribed in the U.S. each year, portrayed as Morphine Milligram Equivalents per adult (green line). b. Opioid Treatment Admissions in thousands (blue line). Three categories of data on drug poisoning/overdose deaths are portrayed: c. Opioid Rx Deaths (POD – lower solid red line) involving a prescription opioid are relatively constant across the period, at approximately 14,000 to 17,000 per year. d. “Any Opioid Deaths (AOD)” are mortality reports involving legal or illegal opioids, shown on the dashed red line, rising sharply from left to right. e. “Total Overdose Deaths (TOD-upper solid red line)” includes poisoning/overdose deaths attributed to any drug of any kind. From 2010 to 2019, the total volume of prescribed opioids dropped by about 55%. Total overdose deaths involving opioids and drugs of all types rose by 70%, even as the component of this rise attributed to prescription opioids each year remained nearly constant. These characteristics track directly to the emergence of illegal fentanyl as a major and deadly contaminant in counterfeit drugs sold on U.S. streets.
Refinements of the Data and Analysis:
Following the previous paper, Larry Aubry submitted a Freedom of Information Act request to the U.S. Food and Drug Administration (FDA). The FDA provided the “Estimated number of patients who were dispensed an opioid analgesic prescription from U.S. outpatient retail pharmacies, 2006-2018” (P#) in response to the FDA Freedom of Information Act (FOIA) File 2021-2978.
Methodology: A graph of the 2006-2018 data was created, and regression analysis was conducted for three time frames to evaluate the potential time delay effects between patients dispensed an opioid prescription and drug overdose mortalities or opioid treatment admissions: a. In the same year, prescriptions were dispensed, b. In the following year, prescriptions were dispensed, and c. In the second year after, prescriptions were dispensed.
The year-by-year number of patients dispensed opioid prescriptions became the independent variable for a regression analysis from 2006 to 2018 for four dependent variables: a. Prescription Opioid Deaths (POD): Deaths in which any prescription opioid is a contributing factor. ICD-10 codes (T40.2-T40.3). Many such deaths will generate multiple reports for contributing agents in several ICD-10 codes (T40.0 for opium, T40.1 for Heroin, T40.2-T40.3 for prescription opioids, T40.4 for Synthetic Opioids other than methadone (primarily fentanyl), T40.5 for Cocaine, and T40.6 for “other and unspecified narcotics”). b. Any Opioid Deaths (AOD): Deaths in which any opioid (prescription or non-prescription) is reported as a contributing factor. ICD-10 codes (T40.0-T40.4, T40.6). c. Total Overdose Deaths (TOD): Deaths in which any drug of any kind is a contributing factor. “Includes deaths with underlying causes of unintentional drug poisoning (X40–X44), suicide drug poisoning (X60–X64), homicide drug poisoning (X85), or drug poisoning of undetermined intent (Y10–Y14), as coded in the International Classification of Diseases, 10th Revision”. d. Opiate/Opioid Treatment Admissions (OTA): Admissions to facilities licensed or certified by the State for opioid (legal or illegal) substance abuse treatment.
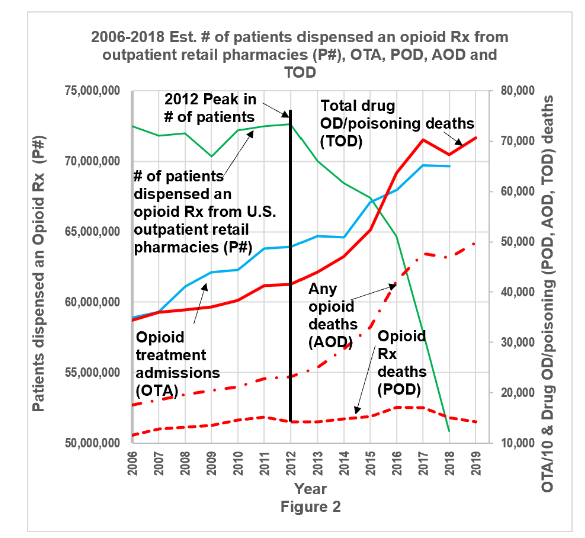
Results: Figure 2 shows the annual number of patients that were dispensed an opioid prescription from an outpatient retail pharmacy, opioid treatment admissions, and drug overdose deaths from 2006-2018. The green solid line represents the “estimated number of patients who were dispensed an opioid analgesic prescription from U.S. outpatient retail pharmacies” (P#). The blue line is Opioid Treatment Admissions. The red lines are (from top down) Total Overdose Deaths (red solid), All Opioid Deaths (red dash dot), and Prescription Opioid Deaths (red dash). The black vertical line marks the 2012 historical peak in the number of patients dispensed an opioid prescription.
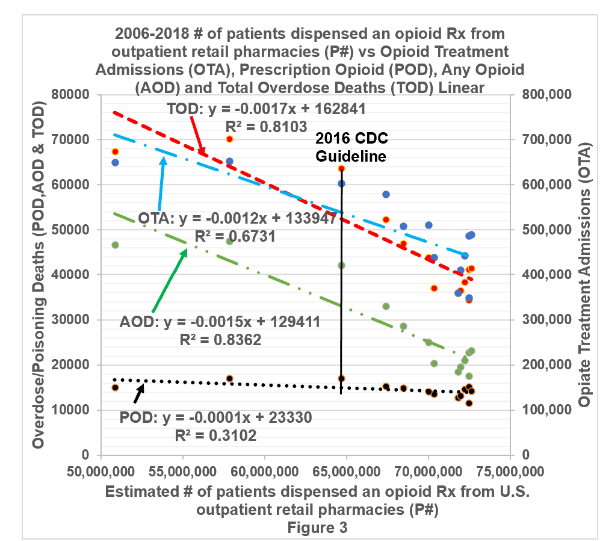
Figure 3 illustrates the linear regression of opioid treatment admissions, prescription opioid deaths, any opioid deaths and total overdose deaths as a function of the “estimated number of patients who were dispensed an opioid analgesic prescription from U.S. outpatient retail pharmacies” (P#) during the same year, from 2006-2018. The red dashed line, TOD, represents total overdose deaths. The blue dash with a single dot line, OTA, is opioid treatment admissions. The green dash with two dot line, AOD, indicates any opioid deaths. The black dotted line, POD, is prescription opioid deaths. The black solid vertical line marks the year of the 2016 CDC Guideline for Prescribing Opioids for Chronic Pain.
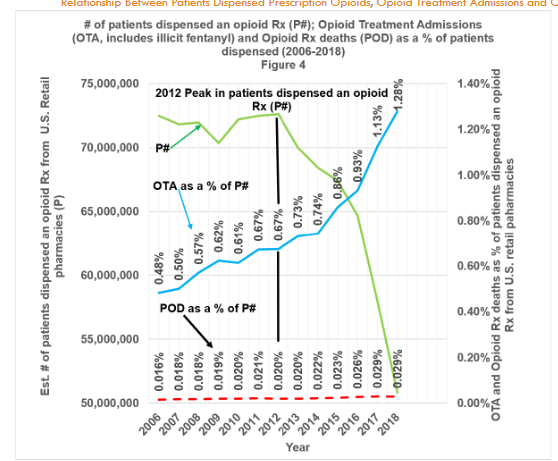
Figure 4 shows the “estimated number of patients who were dispensed an opioid analgesic prescription from U.S. outpatient retail pharmacies” (P#) as a solid green line. The blue solid line documents opioid treatment admissions (OTA), while the red dashed line documents prescription opioid deaths (POD), both as a percentage of the estimated number of patients who received an opioid analgesic prescription from U.S. outpatient retail pharmacies. The black vertical line indicates the 2012 peak in the number of patients who were dispensed an opioid prescription.
Trends in Prescription Opioid Related Deaths, 2020-2022
It has been known for many years that the mortality classification system of the International Classification of Diseases and the training of U.S. County Medical Examiners and Coroners lacks precision in distinguishing between accidental deaths involving legal versus illegal drugs. US CDC has mounted a significant effort to refine the reporting and analysis of such data, resulting in the roll-out in 2019 of the State Unintentional Drug Overdose Reporting System (SUDORS). Thus far, 36 U.S. reporting jurisdictions of the 52 carried in the US Vital Statistics System have accepted CDC funding, retrained their reporting staff and instituted revised classification and reporting systems. This represents about 48% of the U.S. population.
| National Average Rate | Maximum Rate State | Minimum Rate State | |
|---|---|---|---|
| All Drugs (Note 1) | 31.3/100K | 79.4/100K (DC) | 7.8/100K (N. Dakota) |
| Any Opioid Drugs | 25.9 (82.8%) | 69.1/100K (82.8%) (W VA) | 4.3/100K (55.6%) (S. Dakota) |
| Prescription Drugs (Note 2) | 7.8 (18.6%) | 21.5/100K (18.6%) (W VA) | ~0 (20.6%) (S. Dakota) |
Note 1: Accidental Rates of Death reports per 100K population, 33 to 36 Jurisdictions.
Note 2: “Drugs coded as “Prescription Opioids” were alfentanil, buprenorphine, butorphanol, codeine, dihydrocodeine, hydrocodone, hydromorphone, levorphanol, loperamide, meperidine, methadone, morphine, nalbuphine, noscapine, oxycodone, oxymorphone, pentazocine, prescription fentanyl, propoxyphene, remifentanil, sufentanil, tapentadol, thebaine, and tramadol. Also included as prescription opioids were brand names and metabolites (e.g., nortramadol) of these drugs and combinations of these drugs and nonopioids (e.g., acetaminophen-oxycodone). Morphine was included as prescription only if scene or witness evidence did not indicate likely heroin use and if 6-acetylmorphine was not also detected. Fentanyl was coded as a prescription opioid based on scene, toxicology, or witness evidence indicating that it was in prescribed form (e.g., a fentanyl patch was found at the scene).”
| National Average Rate (1) | Maximum Rate State | Minimum Rate State | |
|---|---|---|---|
| All Drugs | 34.4/100K | 87.0/100K (W VA) | 9.8/100K (N Dakota) |
| Any Opioid Drug | 28.2/100K (81.8%) | 74.9/100K (85.5%) (W VA) | 4.3/100K (42.7%) (S Dakota) |
| Prescription Drugs (2) | 5.4/100K (15.7%) | 17.6/100K (20.3%) (W VA) | ~0* (12.2%) (S Dakota) |
| Average | Maximum Rate State | Minimum Rate State | |
|---|---|---|---|
| National Mortality (1) | 35.0/100K | 94.5/100K (DC) | 9.4/100K (Nebraska) |
| Any Opioid Drug | 2/100K (81.8%) | 72.8/100K (81.8%) (DC) | 6.2/100K (66.9%) (Nebraska) |
| Prescription Drugs (2) | 4.3/100K (12.5%) | 11.4/100K (22.1%) (Maine) | 1.1/100K (16.0%) (Iowa) |
In these data, overall rates of U.S. accidental drug overdose mortality of all types in half of the U.S. population continue to increase at least marginally from 2020 to 2022, from 31.3 reports per 100,000 population to 35.0 reports per 100,000 population. Opioids of all types are detected in about 80% of all overdose-related accidental deaths. Rates of overdose are much higher in DC and West Virginia than in many other reporting jurisdictions across the U.S. However, the percentage of mortality reports where one or more prescription drugs are detected drops from about 18% to 12% from 2020 to 2022. Thus, we note that overdose-related mortality is not dominated by prescription drugs.
Limitations on Analysis:
“Estimated number of patients who were dispensed an opioid analgesic prescription from U.S. outpatient retail pharmacies” data includes patients of all ages and excludes opioid products used for bulk compounding or labeled for cough/cold or opioid use disorder. Drug overdose/poisoning deaths and opiate/opioid treatment admissions are taken from the same data sources on which the CDC study of pre-2010 data appears to be based. The values employed in the present paper are as reliable and subject to the same limitations as the data used by the CDC in their analyses. Opiate/Opioid Treatment Admissions (OTA) data includes admissions for all opioids, legal and illegal. Admissions can report up to three substances of use, so in the total column, an individual admission may be counted up to three times. Admissions do not represent individuals. Thus, for example, an individual admitted to treatment twice within a calendar year would be counted as two admissions. It includes admissions at facilities that are licensed or certified by the State substance abuse agency to provide substance abuse treatment (or are administratively tracked for other reasons). It does not include all admissions to substance abuse treatment.
Drug overdose/poisoning deaths often involve alcohol and multiple drugs, where the source of drugs (e.g., legally prescribed vs. illicit/illegal) is unknown in postmortem blood toxicity screens. There is no standard for postmortem toxicology testing or drug involvement determination, potentially resulting in failure to detect IMFs or other drugs. A physician’s “best medical opinion” is relied upon in the documentation of a primary sequence/cause of the overdose death. However, death certificates may contain as many as 20 ICD-10 codes for various contributing factors.
Discussion:
From 2006-2018, prescription opioid deaths remained relatively flat. However, opioid treatment admissions, any opioid deaths, and total overdose deaths increased significantly. At the same time, the number of patients dispensed an opioid prescription from U.S. outpatient retail pharmacies decreased by 21.8 million (almost 30%) after 2012.
No direct correlation exists for the same year, as well as for the one- and two-year lag between the “estimated number of patients who were dispensed an opioid analgesic prescription from U.S. outpatient retail pharmacies” and the dependent response variables: opioid treatment admissions (cited in Congressional testimony as representing opioid addiction), prescription opioid deaths, any opioid poisoning/overdose deaths, and total overdose deaths. The correlations are negative.
Neither a direct (positive) nor an inverse (negative) correlation alone proves that a cause-and-effect relationship exists. However, correlation is necessary for establishing a cause-and-effect relationship. Opioid treatment admissions and drug overdose deaths are not predicted by, dependent on, or determined by the number of patients dispensed an opioid prescription; there is no cause-and-effect relationship. This is not to say that an individual dispensed an opioid prescription could not become addicted or die from an overdose. As the FDA states, “every drug that FDA approves – carries some risk”.
From 2006 to 2018, the mean and median death reports potentially linked to prescription opioids were 0.02% (SD 0.004%) of patients who received an opioid prescription that year. For opioid treatment admissions to which a prescription opioid may have contributed, the mean was 0.76% (SD 0.23%), and the median was 0.68%.
If we assume a one-year lag between the number of patients prescribed opioids (2006-2017) and prescription opioid deaths (2007-2018), the mean and median rates are 0.02% (SD 0.00%). For opioid treatment admissions, the mean rate would be 0.75% (SD 0.18%), with the median at 0.69%.
If we assume a two-year lag between the number of patients prescribed opioids (2006-2016) and prescription opioid deaths (2008-2018), then the mean and median rates are 0.02% (SD 0.00%). For opioid treatment admissions, the mean rate would be 0.75% (SD 0.14%), with the median at 0.70%.
If the number of patients dispensed an opioid Rx included mail-order pharmacy and bulk compounding (not addressed in the FOIA response for 2006-2018), these percentages would decrease. Opioid treatment admissions as a percentage of the number of patients prescribed opioids would be lower if OTA did not include non-Rx admissions (e.g., heroin, illicit fentanyl, etc.).
A review of Substance Abuse and Mental Health Services Administration, Center for Behavioral Health Statistics and Quality, Treatment Episode Data Set (TEDS): 2000-2010 and 2008-2018 reveals that opioid treatment admissions have been dominated by illicit and adulterated drugs, not by prescriptions. 2020 data from the State Unintentional Drug Overdose Reporting System (SUDORS) stated that 6.2% of the drug overdose deaths had “prescription opioids with no other opioids or stimulants,” with it dropping to 4.6% in 2021.
Thus, we would not expect to see a significant direct statistical relationship between opioid prescribing and treatment admissions.
The mean and median number of opioid prescriptions dispensed per patient from 2006-2018 was 3.4 (Standard Deviation 0.15), which demonstrates that a significant majority of all prescribing is for acute rather than chronic pain, with such a low exposure that the number of prescriptions cannot be attributed to clinical prescribing among patients with unremitting and chronic pain.
OTHER PERTINENT CLINICAL LITERATURE
From authoritative published sources, we are informed that: “Unlike tolerance and physical dependence, addiction is not a predictable result of opioid prescribing. Addiction occurs in only a small percentage of persons who are exposed to opioids — even among those with preexisting vulnerabilities”.
“Five-Year Trajectories of Prescription Opioid Use” — a five-year study of 3.47 million adults, published in JAMA Network Open — found approximately 3% of a population-based cohort study conducted in New South Wales, Australia, with sustained or increasing use. Study findings suggest that these adults “had greater clinical complexity and treatment needs”.
A landmark study of over 1.1 million Veterans Administration patients by Oliva, Bowe, Tavakoli, et al., has established a strong predictive model for next-year hospital admissions, suicide attempts, successful suicide, or accidental deaths involving prescription opioid medications. The incidence of such events in the clinical population who were prescribed opioids was on the order of 2%. However, for the highest-risk patient cohort (1,000 among 1,135,601 VHA patients), ten out of eleven highest risk factors related to a history of severe mental health issues or past diagnoses of a substance use disorder. Risk ratios were four to 20 times more significant than a history of clinical prescribing. Thus, it can be said with confidence that opioid prescribing in a clinical context is not a dominant risk factor in next-year patient mortality or hospital admissions.
Per the World Health Organization, we also learn, “After longer-term use, people receiving morphine may build tolerance to its pharmacological effects, that is, a decrease in effectiveness with the same dose. When this occurs, people require higher doses to achieve the same level of pain relief, but the higher doses do not significantly increase the risk of developing the serious adverse effects… How frequently tolerance occurs among patients treated with morphine is uncertain. Clinical experience in specific medical contexts, such as palliative care, suggests that occurrence is rare. In fact, patients often require higher doses of morphine or strong opioids for pain relief as their disease worsens”.
The CDC, FDA, federal advisory committee meetings, associations, publications, and peer-reviewed articles provide substantial evidence of harm and negative consequences caused by the systemic interventions to reduce prescription opioid analgesics, including lower quality of life, higher healthcare burden, and death. The effects of systemic stigma, access barriers, surveillance, investigations and fear of legal consequences that undermine pain management also impact those with sickle cell, active cancer treatment, palliative care, or end-of-life care.
The references cited offer historical evidence validating that the harms of systemic interventions are not an aberration.
The saying, “What is measured is what is valued,” is particularly relevant when looking at the 2016 CDC Guideline and its 2022 update for prescribing opioids. Importantly, none of the twelve recommendations in these guidelines emphasized the need for ongoing data collection regarding patient quality of life, outcomes, hospitalizations, or deaths resulting from the application of CDC’s guidelines. In response to FOIA request #21-01554, which sought “all information on adverse healthcare events and patient harm associated with the 2016 Guideline for Prescribing Opioids for Chronic Pain [01/01/2012 to 01/01/2021],” the CDC stated they did not possess any responsive records. In response to #23-00280-FOIA, the CDC stated, “The Opioid Rapid Response Program informs me that they do not have any information on whether continuity of care continues or not after law enforcement action or other events”. Therefore, the CDC does not have sufficient evidence to show that the benefits of their guidelines and the resultant systemic interventions outweigh the potential harm to individual patients.
Conclusions
The CDC proclaimed in 2016 that “overprescribing opioids— largely for chronic pain — is a key driver of America’s drug-overdose epidemic”. However, in fact, stimulants (e.g., cocaine, prescription stimulants) have been the leading cause of drug overdose death since 1999, followed by synthetic opioids (e.g., illicit fentanyl, buprenorphine) and vice versa since 2010 — not prescription opioids.
Organizations representing more than 560,000 physicians and medical students stated in 2018, “Physicians must be able to practice medicine that is informed by their years of medical education, training, experience, and the available evidence, freely and without threat of criminal punishment”.
Based on real world data and the relationships that have existed since 2006, it is essential to correct/update guidelines, policies, legislation, settlements, and enforcement actions to acknowledge this material information and to prevent misrepresentation or omission of material facts, which could knowingly and willfully harm others. Patients, as it is their life, and doctors must be trusted to make decisions based on informed consent, including the associated risks. False relationships must not be the basis for systemic interventions that place a doctor at risk when treating a patient, deny a patient’s choice based on false premises, or lack the voluntary consent of the individual.
Author Contributions
The authors contributed equally to this work and share first authorship.
Conflicts of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author LA is an independent researcher with no current professional affiliations. Author RL is a subject matter expert on U.S. regulation of prescription opioid analgesics and clinicians who employ them in pain management. He has authored or co-authored over 250 published papers, articles, and interviews in a mixture of peer-reviewed medical journals and mass media.
Funding
No outside funding was involved.
References
- Jalal H, Buchanich JM, Roberts MS, Balmert LC, Zhang K, Burke DS. Changing dynamics of the drug overdose epidemic in the United States from 1979 through 2016. Science. 2018;361(6408):eaau1184. http://dx.doi.org/10.1126/science.aau1184
- National Drug Overdose (OD) deaths, 1999-2021. Live.com. National Institutes of Health on Drug Abuse. 2023. Accessed January 3, 2025. https://view.officeapps.live.com/op/view.aspx?src=https%3A%2F%2Fnida.nih.gov%2Fsites%2Fdefault%2Ffiles%2FOverdose_data_1999-2021%25201.19.23.xlsx&wdOrigin=BROWSELINK
- Centers for Disease Control and Prevention. U.S. overdose deaths decrease in 2023, first time since 2018. www.cdc.gov. May 14, 2024. Accessed January 3, 2025. https://www.cdc.gov/nchs/pressroom/nchs_press_releases/2024/20240515
- Mandell, BF. “The fifth vital sign: A complex story of politics and patient care.” Cleveland Clinic Journal of Medicine, June 2016;83(6):400-401. https://doi.org/10.3949/ccjm.83b.06016
- Scher C, Meador L, Van Cleve JH, Reid MC. Moving Beyond Pain as the Fifth Vital Sign and Patient Satisfaction Scores to Improve Pain Care in the 21st Century. Pain Management Nursing. 2018;19(2):125-129. https://doi: 10.1016/j.pmn.2017.10.010
- Duggam, G. “12 Million OxyContin Pills Shipped to a Town of 500: How Profit Fueled America’s Opioid Crisis”. CBC News, November 15, 2021. Accessed January 5, 2025. https://www.cbc.ca/documentaries/the-passionate-eye/12-million-oxycontin-pills-shipped-to-a-town-of-500-how-profit-fuelled-america-s-opioid-crisis-1.6247359
- CDC archives. CDC Releases Guideline for Prescribing Opioids for Chronic Pain. Press Release. Para. 3. Accessed January 5, 2025. https://archive.cdc.gov/#/details?url=https://www.cdc.gov/media/releases/2016/p0315-prescribing-opioids-guidelines.html
- Remarks by the President in panel discussion at the National Prescription Drug Abuse and Heroin Summit. National Archives and Records Administration. Whitehouse.gov. March 29, 2016. Paragraph 6. Accessed January 5, 2025. https://obamawhitehouse.archives.gov/the-press-office/2016/03/29/remarks-president-panel-discussion-national-prescription-drug-abuse-and
- Operation Unite. Keynote Address CDC Director Dr. Tom Frieden. Lecture presented at the National Prescription Drug Abuse and Heroin Summit on March 30, 2016. Vimeo. Accessed January 5, 2025. https://vimeo.com/162843894
- Rudd, RA, Aleshire, N, Zibbell, JE, Gladden, RM. Increases in drug and opioid overdose deaths — United States, 2000–2014. Centers for Disease Control and Prevention. January 1, 2016. Accessed January 5, 2025. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6450a3.htm
- Dowell D, Zhang K, Noonan RK, Hockenberry JM. Mandatory provider review and pain clinic laws reduce the amounts of opioids prescribed and overdose death rates. Health Affairs. 2016;35(10):1876–83. p.1879,1882. http://dx.doi.org/10.1377/hlthaff.2016.0448
- Paulozzi, LJ. Jones, CM. Mack, KA, Rudd, RA. CDC. Vital signs: Overdoses of prescription opioid pain relievers — United States, 1999–2008. Morbidity and Mortality Weekly Report (MMWR). November 4, 2011. Accessed January 5, 2025. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6043a4.htm
- ASPE Issue Brief. Department of Health and Human Services, Office of the Assistant Secretary for Planning and Evaluation. Opioid Abuse in the U.S. and HHS Actions to Address Opioid-Drug Related Overdoses and Deaths.2015. P. 1–2, 5. Accessed January 5, 2025. https://aspe.hhs.gov/sites/default/files/migrated_legacy_files/56406/ib_OpioidInitiative.pdf
- Aubry L, Carr BT. Overdose, opioid treatment admissions and prescription opioid pain reliever relationships: United States, 2010–2019. Frontiers in Pain Research. 2022;3. http://dx.doi.org/10.3389/fpain.2022.884674
- Supplementary Table 1 FDA FOIA Response 2021-2978 Estimated # of patients prescribed 2006 to 2018 data and letter.pdf. Google Docs. Accessed January 5, 2025. https://drive.google.com/file/d/1IrlA-joCihIWrhNzlNHiqnykyJ9Bkvq/view?usp=sharing
- Substance Abuse and Mental Health Services Administration, Center for Behavioral Health Statistics and Quality. Treatment Episode Data Set (TEDS): 2000-2010. National Admissions to Substance Abuse Treatment Services. DASIS Series S-61, HHS Publication No. (SMA) 12-4701. Rockville, MD: Substance Abuse and Mental Health Services Administration, 2012. P. 5, 43, Table 1.1A, P. 72 Table 2.15. Accessed January 5, 2025. https://www.samhsa.gov/data/sites/default/files/2010_Treatment_Episode_Data_Set_National/2010_Treatment_Episode_Data_Set_National.pdf
- Substance Abuse and Mental Health Services Administration, Center for Behavioral Health Statistics and Quality. Treatment Episode Data Set (TEDS): 2008-2018. Admissions to and Discharges From Publicly Funded Substance Use Treatment. Rockville, MD: Substance Abuse and Mental Health Services Administration, 2020. Table 1.1a, Table 2.18. Accessed January 5, 2025. https://www.samhsa.gov/data/sites/default/files/reports/rpt31097/2018_TEDS/2018_TEDS_Tables.html#Tbl1.1a
- Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain — United States, 2016. Morbidity and Mortality Weekly Report (MMWR) Recommendations and Reports. 2016;65(No. RR-1): P. 2,16. 1–49. http://dx.doi.org/10.15585/mmwr.rr6501e1
- Lawhern RA, Doctors ‘overprescribing’ opioids isn’t the cause of the overdose epidemic — and it never was, STAT News, July 11, 2024. Accessed January 8, 2025. https://www.statnews.com/2024/07/11/doctors-overprescribing-opioids-didnt-cause-overdose-epidemic/

