Liquid Biopsy for Subtyping Non-Hodgkin Lymphomas
Liquid Biopsy Based Approaches for Subtyping Non-Hodgkin Lymphomas: A Proposal to Address an Important Unmet Clinical Need
Dorottya Laczko 1, Narek Israelyan 1, Alex J. Rai 2
- Department of Pathology and Laboratory Medicine, Hospital of the University of Pennsylvania and Perelman School of Medicine at the University of Pennsylvania, Philadelphia, Pennsylvania, USA.
- Department of Pathology and Cell Biology, Columbia University Irving Medical Center, New York, New York USA.
* Contributed Equally
Author for correspondence: Alex J. Rai, PhD, DABCC, FAACC Professor and Director, Clinical Chemistry Service; co-Director, Automated Core Laboratory and Point-of-Care Testing Service, Department of Pathology and Cell Biology, Columbia University Irving Medical Center, 622 West 168th Street, New York, NY 10032. Email: [email protected]
OPEN ACCESS
PUBLISHED: 28 February 2025
CITATION: Laczko, D., Israelyan, N., et al., 2025. Liquid Biopsy Based Approaches for Subtyping Non-Hodgkin Lymphomas: A Proposal to Address an Important Unmet Clinical Need. Medical Research Archives, [online] 13(2). https://doi.org/10.18103/mra.v13i2.6333
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i2.6333
ISSN 2375-1924
ABSTRACT
There are over 30 subtypes of non-Hodgkin lymphoma (NHL), each with distinct clinical and pathological features, prognoses, and treatment. Diagnosis of NHLs is made primarily through tissue biopsy. These procedures are invasive and carry risk, in addition to providing limited sampling from one portion of a single tumor lesion. Differentiating between subtypes of NHL can prove to be difficult, and incorrect diagnoses are not uncommon, which leads to delays in appropriate treatment. Thus, there is an unmet clinical need in the field, which we suggest can be improved by clinical implementation of liquid biopsy biomarkers. Liquid biopsies, including analysis of blood plasma for circulating tumor cells (CTCs), circulating tumor DNA (ctDNA), and extracellular vesicles, are an emerging tool for diagnosis of tumors through blood or urine samples. In this review, we discuss the classification and significant features of the various subtypes of NHL, including their pathologic, clinical, and diagnostic criteria, and the latest progress on liquid biopsy biomarkers. The groundwork for studying and applying liquid biopsy biomarkers in non-Hodgkin lymphoma will aid in forming more accurate, less invasive diagnosis, which may ultimately help to guide earlier treatment and decrease disease burden.
Keywords:
Non-Hodgkin lymphoma, liquid biopsy, extracellular vesicles, exosomes, biomarkers
Introduction
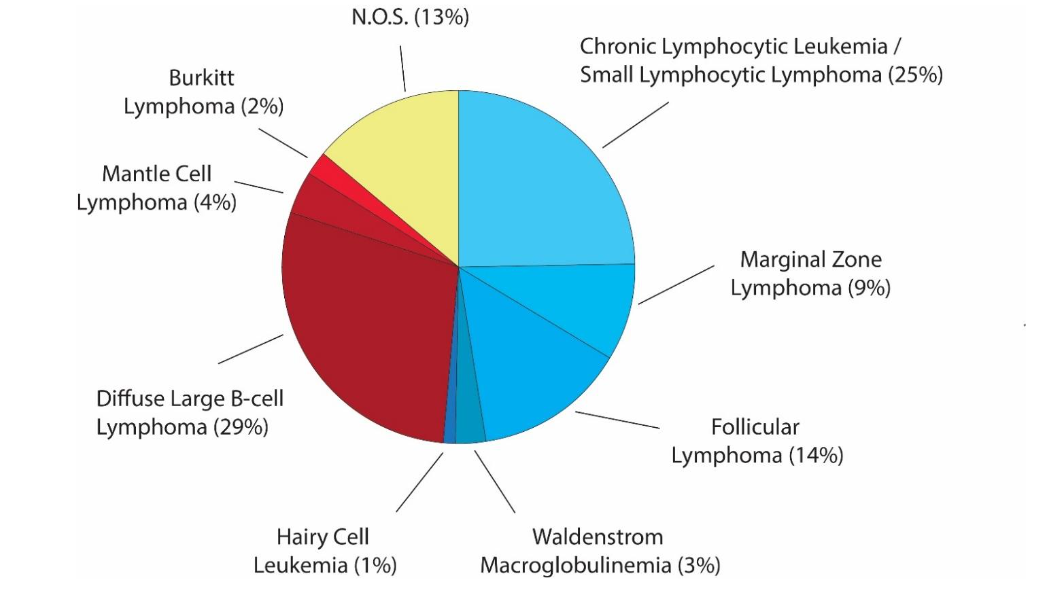
Non-Hodgkin Lymphomas (NHLs) are the most prevalent hematopoietic neoplasms in the world, and account for 4.3% of all cancers in the United States. NHL is an umbrella term for over 30 different lymphomas, primarily of B-cell origin (85%), but occasionally of T-cell origin (15%). They are frequently also divided into indolent (low-grade) and aggressive (high-grade) forms.

Diagnosis of NHLs is typically made through tissue biopsy. Such procedures are invasive and carry risk, in addition to offering only a limited sample, from one portion of a single tumor lesion. Liquid biopsies are an emerging tool for generating a molecular profile of lymphomas. This can involve evaluation of mutations through molecular subtyping, development or prognosis, or prediction of treatment response. Analysis of circulating tumor DNA (ctDNA) and cell-free DNA fragments (cfDNA) derived from blood plasma are the most commonly used liquid biopsy approaches in lymphoma. Extracellular vesicles (EVs) and their respective protein biomarkers have also emerged as important tools for predicting chemotherapy outcomes.
Ultimately, these methods can be used as either diagnostic or prognostic markers, and sometimes can serve both purposes. They may be able to provide a non-invasive means to determine the subtype of NHL, or to determine whether a particular type of lymphoma will be aggressive or indolent. Progress in this realm can fulfill the unmet clinical need of having easier, more rapid, non-invasive ways of evaluating lymphomas. In this review, we propose the utility of liquid biopsy biomarkers in differentiation between the numerous types of NHLs. We start by describing the various B-cell and T-cell lymphomas before discussing the advances and future potential of liquid biopsy-based approaches as biomarkers for NHL subtypes.
B-cell Lymphomas
BURKITT LYMPHOMA
Burkitt lymphoma (BL) is a highly aggressive form of NHL that is classically seen in children. There are strong associations with a t(8;14) translocation, seen in 75% of cases, and involving a reciprocal translocation of the c-myc gene on chromosome 8 with heavy-chain Ig on chromosome 14. This results in an overexpression of c-myc, and ultimately, subsequent increased expression of several genes involved in cellular proliferation. BL has been associated with a nearly 100% Ki-67 proliferation index, and as such, has the shortest doubling time of any human neoplasm.
Burkitt lymphoma can be further subdivided into three variants: (1) sporadic, which occurs worldwide, (2) endemic, which occurs predominantly in sub-Saharan Africa and is strongly EBV-associated, and (3) immunodeficiency-associated, which occurs primarily in the setting of HIV. Each of the three clinical variants are at risk for seeding the CNS and bone marrow, which are associated with worse outcomes. The sporadic form, which classically presents with a mass in the abdomen, is more commonly seen in adults and may have features similar to diffuse large B-cell lymphoma (DLBCL). The endemic form is most commonly seen in children and is associated with involvement of the mandible and maxilla. Unlike the other two clinical variants, endemic BL has an exquisite sensitivity to chemotherapy, with children having localized disease achieving a >90% 5-year survival rate. While sporadic and immunodeficiency-associated subtypes share a similar genetic profile, whole genome sequencing has revealed that endemic-type BL has more frequent mutations in BCL7A and BCL6, and less frequent mutations in DNMT1, SNTB2, and CTCF. Further, gene expression profiling techniques have revealed nearly identical profiles in sporadic and immunodeficiency-associated BL, but notable differences from sporadic BL, particularly in pathways B-cell receptors, TNF-a and interleukin-dependent cascades, and the RBL2-associated gene network.
Patients with any variant of BL generally present with a rapidly growing mass, elevated lactate dehydrogenase (LDH), and high uric acid levels. Diagnosis of BL in the acute setting is primarily done using imaging studies (ultrasound and CT), while tissue biopsy of the mass is seen as the gold standard for diagnosis. A study in endemic Uganda, however, demonstrated only a 75% diagnostic accuracy due to inconsistency of local pathology procedures. Furthermore, in BL endemic areas such as sub-Saharan Africa, where the capacity for invasive tissue biopsies is severely limited, there would be an important clinical utility of ctDNA to improve the speed of diagnosis. This highlights the importance of the development of more standardized diagnostic tools, especially in resource-constrained settings.
The primary differential diagnosis of Burkitt lymphoma is diffuse large B-cell lymphoma (DLBCL). This is a more pertinent problem in adults rather than children, due to the higher prevalence of DLBCL in adults. While Burkitt lymphoma has been classically associated with c-myc rearrangement, up to 15% of DLBCL has been shown to also share this similar rearrangement. The three subtypes of BL have been shown to share a homogenous microRNA (miRNA) profile that is distinct from DLBCL.
FOLLICULAR LYMPHOMA
Follicular lymphoma (FL) is a highly common, generally indolent form of NHL. The majority of follicular lymphomas are characterized by a t(14;18) translocation, involving bcl-2 on chromosome 18 with heavy-chain Ig on chromosome 14. This results in overexpression of Bcl-2 protein, which prevents release of cytochrome c from mitochondria, and ultimately results in decreased apoptosis. FL typically presents as an asymptomatic peripheral lymphadenopathy, and thus can go unnoticed for long periods of time. Diagnosis of follicular lymphoma is preferably through biopsy of a lymph node, where immunocytochemical staining is positive for CD19, CD20, CD10, monoclonal immunoglobulin, and bcl-2.
EZH2 is one of the most commonly mutated epigenetic modifiers, present in up to 27% of follicular lymphomas. While EZH2 mutations have not been observed to have a prognostic impact, the frequencies of EZH2 mutations are decreased or eliminated following successful treatment of FL, and persistently elevated EZH2 levels are associated with treatment failure. A recent study demonstrated the efficacy of circulating tumor DNA mutation analysis of EZH2 as a rapid, real-time monitoring tool for detecting levels of EZH2 mutations.
A majority of follicular lymphomas are indolent, and exhibit an excellent response to rituximab in combination with chemotherapy. However, some follicular lymphomas can transform into DLBCL, which has a much poorer prognosis. A number of prognostic markers have been associated with the transformation of FL to DLBCL, including mutations in p53, MYC, BCL-2, and BCL-6. Studies on the utility of liquid biopsy in FL are still emerging. Hatipoğlu et al. suggested that taking pre-treatment concentrations and genotyping cfDNA could have clinical utility in FL for risk stratification and prediction for transformation. In addition, they showed that the presence of BCL2 mutation in cfDNA is associated with poor survival in treatment naïve patients.
Recently, Jimenez-Ubieto et al. evaluated minimal residual disease (MRD) in FL based on tumor cfDNA detection using an ultra-deep sequencing approach and found that positive cfDNA following first-line therapy correlated with increased risk of progression. Martin Munoz et al. reported that a positive LiqBio-MRD test within the first 6 months of FL patients who underwent chimeric antigen receptor (CAR) T-cell therapy was a useful approach to identify patients with high risk of relapse.
In a recent study by Bang et al., it was suggested that exosomal mRNA expression of BCL2 and BCL6 was correlated with disease progression both in FL and DLBCL.
MARGINAL ZONE LYMPHOMA
Marginal zone lymphoma (MZL) is a generally indolent form of NHL with strong associations with numerous inflammatory states, including autoimmune disease (Hashimoto’s thyroiditis, Sjogren’s syndrome) and chronic inflammation due to infection (H. pylori, B. burgdorferi, C. psitacci, hepatitis C, Epstein-Barr virus). Commonly, marginal zone lymphoma, and particularly gastric mucosa-associated lymphoma tissue (MALT), is associated with t(11;18) translocations which result in formation of chimeric or fusions proteins that upregulate NF-kB, leading to transcription of multiple survival-related genes.
Marginal zone lymphoma is divided into three subtypes: nodal, extranodal, and splenic. Each subtype has specific diagnostic criteria, genetic characteristics and clinical pathogenesis. The diagnosis of MZL involves immunohistochemistry staining of tumor biopsy, including markers CD20, CD10, CD5, and cyclin D1. Importantly, each of the subtypes exhibit clinically distinct outcomes. Patients with nodal marginal zone disease present with more advanced-stage disease and have lower 5-year survival rates than patients with extranodal, gastric MALT lymphoma. Further, each of the variants have the potential to progress to DLBCL.
MANTLE CELL LYMPHOMA
Mantle cell lymphoma (MCL) is classically considered an aggressive, mature form of NHL with a historically poor prognosis. It is defined by a t(11;14) translocation in which CCDN1 on chromosome 11 is translocated with heavy-chain Ig on chromosome 14, leading to overexpression of cyclin D1 protein. This ultimately leads to promotion of transition from G1 to S phase through modulation of cyclin-dependent kinase proteins.
Classically, the disease has been marked by rapid relapse and poor long-term outcomes. However, clinical data has suggested considerable heterogeneity within this disease entity, with presentations ranging from indolent to highly aggressive disease. Diagnosis is established through histological analysis of a lymph node biopsy, including mandatory evaluation of cyclin D1 expression through immunohistochemistry. A number of markers have been suggested as prognostic indicators of long-term outcomes, including mantle cell international prognostic index (MIPI), Ki-67 proliferation index, and TP53 mutations. Differentiation of mantle cell from marginal zone lymphoma and chronic lymphocytic leukemia is challenging, due to significant overlap in the tissue immunophenotypes, including of CD5, CD19, CD20.
In a prospective study, Lakhotia et al. reported that detecting immunoglobulin receptor encoding ctDNA shows good correlation with tumor burden on PET scans and the clearance of ctDNA after first round of treatment was associated with improved progression free survival.
Plasma liposomes could also be considered as MCL biomarkers. In MCL, lipid metabolism via CD36 affects drug resistance and survival, and thus, ectopic expression of CD36 causes an increase in lipid droplets and is associated with bortezomib resistance in MCL cell lines.
CHRONIC LYMPHOCYTIC LEUKEMIA (CLL) AND SMALL LYMPHOCYTIC LYMPHOMA (SLL)
CLL and SLL comprise the most common indolent forms of NHL. They are two very similar cancers, with the exception of site predominance. SLL cells are found in lymph nodes, while CLL cells are found in blood and bone marrow. Multiple causes of the disease have been suggested and evaluated, including chromosomal deletions and translocations, environmental exposures, and drugs, with numerous minor associations identified.
Diagnostic tools vary for CLL and SLL due to their site predominance. CLL diagnosis is performed primarily through flow cytometry of the blood, while lymph node biopsy is the gold standard for SLL diagnosis. The typical immunophenotype panel includes CD5, CD19, CD20, CD23, and CD19. Flow cytometry for cyclin D1 and fluorescence in situ hybridization (FISH) for t(11;14) are also often evaluated to rule out mantle cell lymphoma. Other important evaluations include TP53 mutation status and del(17p) by FISH as their presence guide selection of initial immunotherapy.
The cancers are characterized by the potential to transform into DLBCL through a Richter transformation resulting from a number of molecular events, including acquisition of TP53 mutations and 17p13 deletions. Clinical progression of SLL/CLL is heterogenous and depends on the presence of mutations in heavy-chain Ig variable regions, genomic changes, and comorbidities.
In a study from Yeh et al., it was demonstrated that CLL cells release abundant amounts of exosomes in plasma with a distinct CD37, CD9, and CD63 phenotype. They also provided evidence that members of miR-29 family such as miR-150, miR-155, and miR-223 are also associated with CLL.
De Luca et al. suggested that detection of serum microvesicles (MV) is a useful tool to follow disease. In their studies, the total number of MVs, mainly with CD19 and CD37 phenotype were significantly elevated compared to healthy donors and correlated with advanced clinical stage and poor prognosis.
DIFFUSE LARGE B-CELL LYMPHOMA
Diffuse large B-cell lymphoma (DLBCL) is the most common form of NHL, comprising 30-40% of cases worldwide. It is an aggressive form of NHL with an overall 5-year survival rate of 64%. Numerous mutations have been implicated in the pathogenesis of DLBCL, including in bcl-2, blc-6, and p53, among others. Further, associations have been made with infectious causes, including EBV, HHV-8, HIV, and H. pylori. DLBCL can arise sporadically, or via transformation from follicular lymphoma, marginal zone lymphoma, or CLL. Multiple variants of DLBCL exist, of which the most common include centroblastic, immunoblastic, and anaplastic variants. Further, DLBCL can also be classified by cell of origin, including activated B-cell (ABC) subtype, germinal center-B cell (GCB), and Type III, or unclassifiable by gene expression profile.
The gold standard in DLBCL diagnosis is through histological examination of either a surgical excision or needle core biopsy. Immunophenotype evaluation is used for diagnosis of DLBCL, similarly to other NHL subtypes. A number of recent studies have emerged highlighting the utility of cfDNA as both a prognostic biomarker and a predictor of treatment response in DLBCL with high cfDNA levels being associated with more aggressive disease presentation and depletion of cfDNA levels being associated with better treatment response.
Molecular subtyping of DLBCL has been shown to have important clinical utility. For example, a large subset of patients with activated B-cell DLBCL develop resistance against ibrutinib, a commonly used tyrosine kinase inhibitor in DLBCL. Recent work has identified mutations in BCL10 as the molecular mechanism which confers resistance against ibrutinib. Preemptive molecular identification of this mutation can thus guide treatment away from ibrutinib to other agents. Along this line, previous studies demonstrated that mutant L265P MyD88 is associated with ibrutinib resistance, therefore could be a useful marker to follow ibrutinib sensitivity/resistance in DLBCL patients.
The role of EVs also emerged as a biomarker to follow DLBCL patients. Metthiesen et al. showed that EVs could be used for DLBCL monitoring and early identification of residual disease as well as response to therapy. Finally, Rinaldi et al. showed promising data that miR-22 could be used as a biomarker to predict poor outcome in DLBCL patients.
PRIMARY DLBCL OF THE CENTRAL NERVOUS SYSTEM (CNS)
According to the 4th edition of the WHO classification, primary DLBCL of the CNS is defined by lymphoma arising from the brain, spinal cord, leptomeninges, or eye with exclusion of lymphomas of the dura, intravascular large B-cell lymphomas, secondary spread of systemic lymphomas, and all other lymphomas that are related to immunodeficiency. The clinical prognosis of this disease is poor. The current gold standard of the diagnosis is biopsy; however, the procedure carries a risk of surgical complication. Therefore, introduction of non-invasive methods has an important clinical significance.
Liquid biopsy of the cerebrospinal fluid (CSF) is a less invasive, non-surgical method that could potentially be used for the diagnosis of primary CNS lymphomas (PCNSL). Mutations of MYD88 gene, specifically, the p.L265P amino acid substitution can be found in up to 85% of cases, therefore it could be used as a potential biomarker to identify primary CNS lymphomas. In a study of Yamagishi et al., using digital PCR showed that MYD88 L265P mutation was successfully identified in CSF with high sensitivity and specificity. MYD88 mutation from intraocular fluid was successfully detected in a cohort of patients with vitreoretinal lymphoma as well. Besides MYD88 mutation identification in ctDNA, the diagnostic value of microRNA and inflammatory cytokines has emerged as well. miR-21, miR-19b, and miR-92a concentrations in the CSF were significantly elevated in PCNSL compared to other neurological diseases. In addition, CSF concentrations of IL-10 and IL-6 were reported to be increased in PCNSL patients compared to other brain tumors. Recently, a pilot study from Downs et al. identified methylated markers that could distinguish PCNSL from other CNS tumors with high accuracy.
WALDENSTROM MACROGLOBULINEMIA
Waldenstrom macroglobulinemia (WM) is an indolent form of non-Hodgkin lymphoma that is characterized by a monoclonal IgM protein. The mean age of diagnosis is 71 years, with a favorable 5-year survival that is >80%. Diagnosis of WM is confirmed by presence of IgM monoclonal protein associated with >10% of clonal lymphoplasmytic cells in bone marrow. Additionally, the L265P mutation in MYD88 is detectable in over 90% of patients with confirmed WM, and can be detected in peripheral blood using PCR. However, MYD88 L265P is also seen in 5% of marginal zone lymphomas, and cannot distinguish between WM and monoclonal gammopathy of unknown significance (MGUS), which is nearly always a preceding phase before the onset of WM in patients.
HAIRY CELL LEUKEMIA
Hairy cell leukemia is an indolent form of non-Hodgkin lymphoma characterized by the identification of hairy cells and presence of a BRAF V600E somatic mutation. The presence of additional mutations within hairy cell leukemia have been shown to have prognostic implications. A mutated IGHV gene confers an improved prognosis, while VH4-34 positive cases have a higher disease burden, worse response to standard therapy, shorter overall survival, and the absence of a BRAF-V600E mutation.
T-cell Lymphomas
PERIPHERAL T-CELL LYMPHOMAS
Peripheral T-cell Lymphomas (PTCL) are a heterogenous group of T-cell NHLs that accounts for approximately 5-15% of cases. Cutaneous T-cell lymphoma (CTCL), Angioimmunoblastic T cell lymphoma (AITL), Anaplastic Large Cell Lymphoma (ALCL), and PTCL not otherwise specified (PTCL-NOS) are the most frequent subtypes of PTCL. Clinically, these are aggressive lesions. Recent advances in liquid biopsy technologies for these entities are outlined below.
CUTANEOUS T-CELL LYMPHOMA
Cutaneous T-cell lymphomas (CTCLs) are primary malignancies of the skin, characterized by a cutaneous infiltration of monoclonal T cells. Mycosis fungoides, Sézary syndrome, and primary cutaneous PTCL-NOS are the most important subtypes of CTCLs. These lymphomas are defined classically by malignant, skin-homing CD4+ T cells (>90%).
Mycosis fungoides presents clinically with erythematous patches and plaques, which may last for many years without clinical progression. However, in about 30% of patients, there is progression to advanced-stage disease within 10 years. Sezary syndrome, the more aggressive form of CTCL, is defined by circulating T-cells with cerebriform (“sezary”) nuclei. It can result de novo, or via progression from mycosis fungoides.
The diagnosis of CTCL is based on clinical observations, histological examination of skin biopsies, and additional laboratory diagnostics including miRNA profiling and PCR of tissue samples, which has demonstrated high specificity and sensitivity in distinguishing CTCL from benign disorders. However, CTCL is commonly misdiagnosed, and patients can go many years with an incorrect diagnosis.
Poor disease outcomes have been associated with genes such as TOX, and miRNA profiles including miR-155 and miR-21 expression. Other biomarkers, such as LDH, are elevated in advanced disease, but are rather non-specific. However, there is currently a lack of prognostic biomarkers to predict disease progression from an indolent to an aggressive state.
ANAPLASTIC LARGE CELL LYMPHOMA
Anaplastic large cell lymphomas (ALCLs) are a group of generally aggressive lymphomas that were discovered in the 1980s. The key immunophenotypic markers of ALCL include CD30 and anaplastic lymphoma kinase (ALK). Today, classification of ALCL involves two groups: (1) those that express ALK (ALK+), which are relatively homogenous and have chromosomal abnormalities involving 2p23; (2) the remaining, heterogenous group of ALK- immunophenotypic ALCL.
ALK+ ALCL is a disease entity that more commonly affects younger males, has a better prognosis than its heterogenous ALK- counterparts (79% vs 46% overall 5-year survival, respectively), and is more responsive to chemotherapy than ALK- disease, which is more likely to relapse. Thus, identification of ALK status is a critical step in determining the prognosis and treatment plan. Immunohistochemistry of biopsy samples is currently the gold standard used for diagnosis of tumors with ALK fusions. However, there is evidence that demonstrates the utility of anti-ALK antibody titers in the plasma as a diagnostic and prognostic indicator. Anti-ALK antibodies were identified in 92% of patients with confirmed disease. Further, higher antibody titers in patients correlate with significantly lower incidence of relapses in response to chemotherapy, and with fewer circulating tumor cells.
Recent studies suggested a diagnostic role of extracellular vesicles in ALCL. Lovisa et al. found that RNY4 fragments in circulating exosomes are significantly elevated in pediatric ALCL. Increased tenascin C, osteopontin and hsp90 levels are also suggested as potential markers for ALK+ ALCL prognostic stratification.
ANGIOIMMUNOBLASTIC T-CELL LYMPHOMA
Angioimmunoblastic T-cell lymphoma (AITL) is a complex disease process with a poorly understood etiology and pathogenesis. The cell-of-origin has been identified as the follicular T helper cell, as demonstrated by molecular analyses. AITL often affects older adults >65 years, and has a poor prognosis with a 5-year median survival of 32%. Diagnosis of AITL is typically done by evaluation of a lymph node biopsy; however, the lack of specific hallmarks of the disease makes a definitive diagnosis difficult to achieve, and errors in initial diagnosis are common with AITL. A recent study evaluated the immunophenotypic features of AITL using flow cytometry on tissue biopsy and peripheral blood samples, and found an aberrant T-cell population in over 2/3 of patients with confirmed AITL, particularly with a loss of CD3 expression and positive CD10 expression. Another study demonstrated that higher serum levels of soluble IL-2R at diagnosis predicted poor outcomes in AITL.
Mutation in RHOA was found in high frequency in AITL and showed significant correlation with disease progression and relapse.
PTCL-NOS
PTCL-NOS is a heterogenous group of this entity established by diagnosis of exclusion. Based on gene expression studies it can be divided into two main categories. The first involves upregulation of GATA3 and its downstream targets (e.g., CCR4, CXCR7), and is generally associated with a poor prognosis. The second category is the TBX21 subgroup which shows upregulation of the NF-κB pathway, and has more favorable outcomes. The TBX21 group is associated with Tfh phenotype, and therefore is likely to be related to AITL at the molecular level.
The most common mutations and copy number variations in PTCL-NOS affects the TCR and NF-κB pathways, including in PLCG1, CARD11, CD28, VAV1, FYN, PTPRC, and TNFAIP3.
Studies on the utility of liquid biopsy in PTCL-NOS are limited. Ottolini et al. found evidence of recurrent RHOA mutations in a study of 25 patients diagnosed with AITL and PTCL-NOS. Miljkovic et al. used next generation sequencing of the TCR gene in PTCL patients’ serum and concluded that PTCL-NOS patients usually harbors more than one dominant TCR clone. In addition, they found that the overall sensitivity and specificity of surveillance ctDNA were lower compared to patients with DLBCL.
ADULT T-CELL LEUKEMIA/LYMPHOMA
Adult T-cell leukemia/lymphoma (ATL) is a rare, aggressive malignancy caused by infection with human T-lymphotrophic virus 1 (HTLV-1). HTLV-1 is endemic in parts of the world including Japan, Africa, South America, and the Middle East. ATL has several subclassifications, including acute, lymphoma-type, chronic, and smoldering. Acute and lymphoma-type ATL are aggressive with a poor prognosis, while chronic and smoldering subtypes are indolent, responding well to zidovudine and interferon-a treatment.
Serum levels of interleukin-2 receptor alpha (sIL-2Ra) are known to reflect tumor burden and are often used to evaluate progression of ATL. However, sIL-2R levels become elevated with inflammatory responses as well, which can be seen following certain treatments for ATL.
Cell adhesion molecule 1 (CADM1), which was initially isolated as a tumor suppressor gene in non-small cell lung cancer, has been found to be highly expressed in HTLV-1 infected T cells, and aids in adhesion of these cells into different organs. Further, CADM1 is specific to ATL lymphomas, and is not expressed in most of non-ATL lymphomas. A recent study demonstrated that a soluble, extracellular domain of CADM1 (sCDAM1), can be detected in the serum of acute-type and smoldering-type ATL patients using an immunosorbent assay, and is highly correlated with clinical parameters and s-IL2R levels.
NK/T-CELL LYMPHOMA
NK/T-cell lymphoma (NKTCL) is a rare EBV-related non-Hodgkin lymphoma of T-cell origin. It is prevalent in Asia and Latin-America. The most common predilection site is the nasal cavity but can occur at any location. Recently several frequent somatic mutations such as STAT3, JAK3, DDX3X are identified in NKTCL, which could be used as valuable diagnostic and therapeutic biomarkers.
Adequate molecular evaluation of NKTCL lesions is usually hampered by large areas of necrosis which precludes adequate molecular evaluation of the tissue. Furthermore, given the invasiveness of NKTCL, tissue sampling puts the patients at risk of complications, and therefore development of non-invasive methods of molecular profiling would have an important clinical utility.
However, the use of liquid biopsy genotyping has not been examined in NKTCL in a large patient cohort. Qi et al. performed NGS on tumor tissues and longitudinally collected plasma samples obtained from a cohort of high-risk NKTCL patients undergoing systemic chemotherapy and demonstrated that plasma cfDNA reflects tumor genome DNA of NKTCL-associated mutations. In addition, they suggested that blood cfDNA could be a promising non-invasive approach to follow treatment response and survival in NKTCL.
Liquid Biopsy
Liquid biopsy approaches include the analysis of circulating tumor cells (CTCs), circulating tumor DNA (ctDNA), and extracellular vesicles. These approaches are minimally invasive, and have been proposed as valuable tools for diagnosis and for extraction of information from the tumor in real time, allowing for prognostic evaluation along with treatment monitoring. In this review, based on the heterogeneity of several NHL subtypes, we suggest that liquid biopsy can also be used as a tool to differentiate between these subtypes.
During the formation and growth of a tumor, circulating tumor cells (CTCs) and shed into the bloodstream and can be detected through different technologies. A bead-based Immunoassay utilizing antibodies that target epithelial cell adhesion molecule (EpCAM), as employed in the CellSearch system, has become the standard for detection and enumeration of CTCs in peripheral blood. CTC detection and quantification has drawn considerable attention in solid tumors, including lung, breast, prostate and colon cancer, as both a real-time biopsy and a surrogate marker for prognosis. However, the focus of liquid biopsies in lymphoma has primarily utilized molecular analyses using ctDNA and small extracellular vesicles.
Analysis of ctDNA has emerged as a valuable detection mechanism in lymphoma. ctDNAs originate from tumor cells that have undergone apoptosis or necrosis. They generally have short half-lives ranging from minutes to hours in the blood. The importance of the utility of ctDNA lies in its ability to carry genetic information from different disease sites, providing a comprehensive picture of the heterogeneity of the lymphoma, as compared to sampling from a single site. Two main methods exist for analysis of ctDNA: PCR-based assays, involving a targeted approach in which one must know the sequence being evaluated, or next-generation sequencing (NGS) assays, which provide an untargeted and unbiased approach to sequencing genes. Studies have demonstrated the utility of ctDNA analysis in NHL, particularly in determining DLBCL cell of origin. DLBCL can arise from transcriptionally distinct subtypes depending on their cell of origin: (1) activated B cell-like (ABC), (2) germinal center B cell-like (GCB), or (3) neither ABC/GCB. The subtypes are prognostic and may predict sensitivity to therapy. Further, other studies have demonstrated the utility of ctDNA in predicting outcomes in DLBCL. In treatment responders, levels of ctDNA were dramatically reduced within one week of commencing treatment, long before clinical outcomes had been demonstrated. Additionally, while levels of ctDNA are generally higher in aggressive compared to indolent NHL, there are still limitations in differentiating types of NHL using a liquid biopsy approach.
Exosomes, a class of extracellular vesicles, are another emerging diagnostic tool in non-Hodgkin lymphoma. Exosomal cargo includes proteins, nucleic acids, lipids, and metabolites. They are found in multiple body fluids including blood and urine, and have a relatively long half-life, making them good candidates as non-invasive diagnostic biomarkers. What was previously referred to as the exosome fraction from a particular sample is now known to contain a heterogenous group of multiple types of different vesicles, including large exosomes, small exosomes, and exomeres, with distinct internal composition. An emerging technique, asymmetric-flow field-flow fractionation (AF4), has demonstrated the ability to perform separation and characterization of exosome fractions. In several studies, exosomes have emerged as potential prognostic markers, post-therapy surveillance markers, and as targets for new therapies. In a study of canine multicentric lymphoma, higher levels of small extracellular vesicles correlated with worse survival and a higher rate of disease relapse. Further, exosomes have been suggested as a prognostic marker in human DLBCL, with circulating exosomal levels of miR-99a-4p and miR-125b-5p being significantly higher in chemoresistant DLBCL. Other studies have also demonstrated a number of exosomal-derived biomarkers are useful in post-treatment surveillance of DLBCL relapse. Exosomal CD20 in DLBCL has been shown to protect the cells against anti-CD20 immunotherapy, conferring treatment resistance.
At the current time, there are no clinical-grade liquid biopsy tests used in the management of patients with lymphoma. Potentially, such testing could have multiple applications, including diagnosis, prognosis, prediction of therapeutic response, detection of recurrence, subtyping, and risk stratification. There are numerous benefits to use of liquid biopsy approaches in lymphoma, including non-invasive assessment of disease, longitudinal monitoring, and ability to sample multiple disease sites within an individual, among others. Lymphomas, specifically non-Hodgkin’s lymphomas, are a diverse group of cancers which have historically required histological analysis for their diagnosis and management. We propose a new approach of using liquid biopsy testing for the management of lymphoma patients.
Conclusion
There are numerous subtypes of non-Hodgkin lymphomas (NHLs), each with distinct pathological and clinical traits. This heterogeneity highlights the need for sophisticated diagnostic tools by making diagnosis and therapy more difficult. Since liquid biopsies offer real-time, minimally invasive insights into tumor biology, they represent a paradigm leap in the treatment of lymphomas. Liquid biopsies can be used to accurately classify subtypes, predict treatment outcomes, and provide longitudinal disease monitoring by utilizing CTCs, ctDNA/cfDNA, and EVs.
Future studies should prioritize the standardization of liquid biopsy methods and validation of their applicability across various NHL subtypes. Advances in proteomic analysis and next-generation sequencing hold promise for expanding the repertoire of actionable biomarkers. Ultimately, integrating liquid biopsies into routine clinical practice could revolutionize NHL diagnosis and treatment, paving the way for improved outcomes and personalized care strategies.
References
1. SEER, Surveillance, Epidemiology, and End Results Program. (U.S. Dept. of Health and Human Services, National Institutes of Health, National Cancer Institute, 2008-2017).
2. Al-Hamadani, M. et al. Non-Hodgkin lymphoma subtype distribution, geodemographic patterns, and survival in the US: A longitudinal analysis of the National Cancer Data Base from 1998 to 2011. Am J Hematol 90, 790-795, doi:10.1002/ajh.24086 (2015).
3. Rossi, D., Spina, V., Bruscaggin, A. & Gaidano, G. Liquid biopsy in lymphoma. Haematologica 104, 648-652, doi:10.3324/haematol.2018.206177 (2019).
4. Iversen, O. H., Iversen, U., Ziegler, J. L. & Bluming, A. Z. Cell kinetics in Burkitt lymphoma. Eur J Cancer 10, 155-163, doi:10.1016/0014-2964(74)90148-0 (1974).
5. Cairo, M. S. et al. Burkitt’s and Burkitt-like lymphoma in children and adolescents: a review of the Children’s Cancer Group experience. Br J Haematol 120, 660-670, doi:10.1046/j.1365-2141.2003.04134.x (2003).
6. Panea, R. I. et al. The whole-genome landscape of Burkitt lymphoma subtypes. Blood 134, 1598-1607, doi:10.1182/blood.2019001880 (2019).
7. Piccaluga, P. P. et al. Gene expression analysis uncovers similarity and differences among Burkitt lymphoma subtypes. Blood 117, 3596-3608, doi:10.1182/blood-2010-08-301556 (2011).
8. Ferry, J. A. Burkitt’s lymphoma: clinicopathologic features and differential diagnosis. Oncologist 11, 375-383, doi:10.1634/theoncologist.11-4-375 (2006).
9. Kalisz, K. et al. An update on Burkitt lymphoma: a review of pathogenesis and multimodality imaging assessment of disease presentation, treatment response, and recurrence. Insights Imaging 10, 56, doi:10.1186/s13244-019-0733-7 (2019).
10. Ogwang, M. D., Zhao, W., Ayers, L. W. & Mbulaiteye, S. M. Accuracy of Burkitt lymphoma diagnosis in constrained pathology settings: importance to epidemiology. Arch Pathol Lab Med 135, 445-450, doi:10.1043/2009-0443-EP.1 (2011).
11. Legason ID, Ogwang MD, Chamba C, Mkwizu E, El Mouden C, Mwinula H, Chirande L, Schuh A, Chiwanga F. A protocol to clinically evaluate liquid biopsies as a tool to speed up diagnosis of children and young adults with aggressive infection-related lymphoma in East Africa “(AI-REAL)”. BMC Cancer. 2022 May 2;22(1):484. doi: 10.1186/s12885-022-09553-w. PMID: 35501771.
12. Frost, M. et al. Comparative immunohistochemical analysis of pediatric Burkitt lymphoma and diffuse large B-cell lymphoma. Am J Clin Pathol 121, 384-392, doi:10.1309/8WYN-VUTG-V9RP-HUQH (2004).
13. Lenze, D. et al. The different epidemiologic subtypes of Burkitt lymphoma share a homogenous micro RNA profile distinct from diffuse large B-cell lymphoma. Leukemia 25, 1869-1876, doi:10.1038/leu.2011.156 (2011).
14. Freedman, A. Follicular lymphoma: 2018 update on diagnosis and management. Am J Hematol 93, 296-305, doi:10.1002/ajh.24937 (2018).
15. Sun, R., Medeiros, L. J. & Young, K. H. Diagnostic and predictive biomarkers for lymphoma diagnosis and treatment in the era of precision medicine. Mod Pathol 29, 1118-1142, doi:10.1038/modpathol.2016.92 (2016).
16. Nagy, A. et al. Quantitative Analysis and Monitoring of EZH2 Mutations Using Liquid Biopsy in Follicular Lymphoma. Genes-Basel 11, doi:ARTN 785 10.3390/genes11070785 (2020).
17. Luminari, S., Bellei, M., Biasoli, I. & Federico, M. Follicular lymphoma – treatment and prognostic factors. Rev Bras Hematol Hemoter 34, 54-59, doi:10.5581/1516-8484.20120015 (2012).
18. Lossos, I. S. & Gascoyne, R. D. Transformation of follicular lymphoma. Best Pract Res Clin Haematol 24, 147-163, doi:10.1016/j.beha.2011.02.006 (2011).
19. Hatipoğlu T, Esmeray Sönmez E, Hu X, Yuan H, Danyeli AE, Şeyhanlı A, Önal-Süzek T, Zhang W, Akman B, Olgun A, Özkal S, Alacacıoğlu İ, Özcan MA, You H, Küçük C. Plasma Concentrations and Cancer-Associated Mutations in Cell-Free Circulating DNA of Treatment-Naive Follicular Lymphoma for Improved Non-Invasive Diagnosis and Prognosis. Front Oncol. 2022 Jun 16;12:870487. doi: 10.3389/fonc.2022.870487. PMID: 35795062; PMCID: PMC9252432.
20. Jiménez-Ubieto A, Poza M, Martin-Muñoz A, Ruiz-Heredia Y, Dorado S, Figaredo G, Rosa-Rosa JM, Rodriguez A, Barcena C, Navamuel LP, Carrillo J, Sanchez R, Rufian L, Juárez A, Rodriguez M, Wang C, de Toledo P, Grande C, Mollejo M, Casado LF, Calbacho M, Baumann T, Rapado I, Gallardo M, Sarandeses P, Ayala R, Martínez-López J, Barrio S. Real-life disease monitoring in follicular lymphoma patients using liquid biopsy ultra-deep sequencing and PET/CT. Leukemia. 2023 Mar;37(3):659-669. doi: 10.1038/s41375-022-01803-x. Epub 2023 Jan 3. PMID: 36596983.
21. Martin-Munoz et al., Dynamic Response Assessment Combining Liquid Biopsy MRD and PET/CT in Follicular Lymphoma Patients Including CAR-T Cell Therapy. Blood (2022) 140 (Supplement 1): 3514–3515.
22. Bang YH, Shim JH, Ryu KJ, Kim YJ, Choi ME, Yoon SE, Cho J, Park B, Park WY, Kim WS, Kim SJ. Clinical relevance of serum-derived exosomal messenger RNA sequencing in patients with non-Hodgkin lymphoma. J Cancer. 2022 Feb 21;13(5):1388-1397. doi: 10.7150/jca.69639. PMID: 35371331; PMCID: PMC8965112.
23. Ekstrom Smedby, K. et al. Autoimmune disorders and risk of non-Hodgkin lymphoma subtypes: a pooled analysis within the InterLymph Consortium. Blood 111, 4029-4038, doi:10.1182/blood-2007-10-119974 (2008).
24. Zucca, E., Bertoni, F., Vannata, B. & Cavalli, F. Emerging role of infectious etiologies in the pathogenesis of marginal zone B-cell lymphomas. Clin Cancer Res 20, 5207-5216, doi:10.1158/1078-0432.CCR-14-0496 (2014).
25. Liu, H. X. et al. T(11;18) is a marker for all stage gastric MALT lymphomas that will not respond to H-pylori eradication. Gastroenterology 122, 1286-1294, doi:10.1053/gast.2002.33047 (2002).
26. Bertoni, F., Rossi, D., Raderer, M. & Zucca, E. Marginal Zone Lymphomas. Cancer J 26, 336-347, doi:10.1097/PPO.0000000000000463 (2020).
27. Zucca, E. et al. Marginal zone lymphomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 31, 17-29, doi:10.1016/j.annonc.2019.10.010 (2020).
28. Nathwani, B. N. et al. Marginal zone B-cell lymphoma: A clinical comparison of nodal and mucosa-associated lymphoid tissue types. Non-Hodgkin’s Lymphoma Classification Project. J Clin Oncol 17, 2486-2492, doi:10.1200/JCO.1999.17.8.2486 (1999).
29. Shah, B. D., Martin, P. & Sotomayor, E. M. Mantle cell lymphoma: a clinically heterogeneous disease in need of tailored approaches. Cancer Control 19, 227-235, doi:10.1177/107327481201900307 (2012).
30. Lakhotia R, Melani C, Dunleavy K, Pittaluga S, Saba N, Lindenberg L, Mena E, Bergvall E, Lucas AN, Jacob A, Yusko E, Steinberg SM, Jaffe ES, Wiestner A, Wilson WH, Roschewski M. Circulating tumor DNA predicts therapeutic outcome in mantle cell lymphoma. Blood Adv. 2022 Apr 26;6(8):2667-2680. doi: 10.1182/bloodadvances.2021006397. PMID: 35143622; PMCID: PMC9043939.
31. Luanpitpong S, Janan M, Thumanu K, Poohadsuan J, Rodboon N, Klaihmon P, Issaragrisil S. Deciphering the Elevated Lipid via CD36 in Mantle Cell Lymphoma with Bortezomib Resistance Using Synchrotron-Based Fourier Transform Infrared Spectroscopy of Single Cells. Cancers (Basel). 2019 Apr 24;11(4):576. doi:10.3390/cancers11040576. PMID: 31022903; PMCID: PMC6521097.
32. Dreyling, M. & European Mantle Cell Lymphoma, N. Mantle cell lymphoma: biology, clinical presentation, and therapeutic approaches. Am Soc Clin Oncol Educ Book, 191-198, doi:10.14694/EdBook_AM.2014.34.191 (2014).
33. Dreyling, M. et al. ESMO Consensus conferences: guidelines on malignant lymphoma. part 2: marginal zone lymphoma, mantle cell lymphoma, peripheral T-cell lymphoma. Ann Oncol 24, 857-877, doi:10.1093/annonc/mds643 (2013).
34. Ladha, A., Zhao, J., Epner, E. M. & Pu, J. J. Mantle cell lymphoma and its management: where are we now? Exp Hematol Oncol 8, 2, doi:10.1186/s40164-019-0126-0 (2019).
35. Kipps, T. J. et al. Chronic lymphocytic leukaemia. Nat Rev Dis Primers 3, 16096, doi:10.1038/nrdp.2016.96 (2017).
36. Wierda, W. G. et al. Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma, Version 4.2020, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 18, 185-217, doi:10.6004/jnccn.2020.0006 (2020).
37. Rossi, D. & Gaidano, G. Richter syndrome: molecular insights and clinical perspectives. Hematol Oncol 27, 1-10, doi:10.1002/hon.880 (2009).
38. Yeh YY, Ozer HG, Lehman AM, Maddocks K, Yu L, Johnson AJ, Byrd JC. Characterization of CLL exosomes reveals a distinct microRNA signature and enhanced secretion by activation of BCR signaling. Blood. 2015 May 21;125(21):3297-305. doi: 10.1182/blood-2014-12-618470. Epub 2015 Apr 1. PMID: 25833959; PMCID: PMC4440883.
39. De Luca L, D’Arena G, Simeon V, Trino S, Laurenzana I, Caivano A, La Rocca F, Villani O, Mansueto G, Deaglio S, Innocenti I, Laurenti L, Molica S, Pietrantuono G, De Stradis A, Del Vecchio L, Musto P. Characterization and prognostic relevance of circulating microvesicles in chronic lymphocytic leukemia. Leuk Lymphoma. 2017 Jun;58(6):1424-1432. doi: 10.1080/10428194.2016.1243790. Epub 2016 Oct 14. PMID: 27739922.
40. Smith, A. et al. Lymphoma incidence, survival and prevalence 2004-2014: sub-type analyses from the UK’s Haematological Malignancy Research Network. Br J Cancer 112, 1575-1584, doi:10.1038/bjc.2015.94 (2015).
41. Li, S., Young, K. H. & Medeiros, L. J. Diffuse large B-cell lymphoma. Pathology 50, 74-87, doi:10.1016/j.pathol.2017.09.006 (2018).
42. Regazzo, G. et al. Diffuse large B-cell lymphoma: Time to focus on circulating blood nucleic acids? Blood Rev, 100776, doi:10.1016/j.blre.2020.100776 (2020).
43. Lauer EM, Mutter J, Scherer F. Circulating tumor DNA in B-cell lymphoma: technical advances, clinical applications, and perspectives for translational research. Leukemia. 2022 Sep;36(9):2151-2164. doi: 10.1038/s41375-022-01618-w. Epub 2022 Jun 14. PMID: 35701522; PMCID: PMC9417989.
44. Xia, M. BCL10 Gain-of-Function Mutations Aberrantly Induce Canonical and Non-Canonical NF-Kb Activation and Resistance to Ibrutinib in ABC-DLBCL. 62nd ASH Annual Meeting and Exposition (2020).
45. Mondello P, Brea EJ, De Stanchina E, Toska E, Chang AY, Fennell M, Seshan V, Garippa R, Scheinberg DA, Baselga J, Wendel HG, Younes A. Panobinostat acts synergistically with ibrutinib in diffuse large B cell lymphoma cells with MyD88 L265P mutations. JCI Insight. 2018 Nov 15;3(22):e125568. doi: 10.1172/jci.insight.125568. Erratum for: JCI Insight. 2017 Mar 23;2(6):e90196. doi: 10.1172/jci.insight.90196. PMID: 30429379; PMCID: PMC6254785.
46. Matthiesen R, Gameiro P, Henriques A, Bodo C, Moraes MCS, Costa-Silva B, Cabeçadas J, Gomes da Silva M, Beck HC, Carvalho AS. Extracellular Vesicles in Diffuse Large B Cell Lymphoma: Characterization and Diagnostic Potential. Int J Mol Sci. 2022 Nov 1;23(21):13327. doi: 10.3390/ijms232113327. PMID: 36362114; PMCID: PMC9654702.
47. Rinaldi F, Marchesi F, Palombi F, Pelosi A, Di Pace AL, Sacconi A, Terrenato I, Annibali O, Tomarchio V, Marino M, Cantonetti M, Vaccarini S, Papa E, MorettaL, Bertoni F, Mengarelli A, Regazzo G, Rizzo MG. MiR-22, a serum predictor of poor outcome and therapy response in diffuse large B-cell lymphoma patients. Br J Haematol. 2021 Nov;195(3):399-404. doi: 10.1111/bjh.17734. Epub 2021 Jul 28. PMID: 34318932.
48. Swerdlow SH, Campo E, Pileri SA, Harris NL, Stein H, Siebert R, Advani R, Ghielmini M, Salles GA, Zelenetz AD, Jaffe ES. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016 May 19;127(20):2375-90. doi: 10.1182/blood-2016-01-643569. Epub 2016 Mar 15. PMID: 26980727; PMCID: PMC4874220.
49. Yamagishi Y, Sasaki N, Nakano Y, Matushita Y, Omura T, Shimizu S, Saito K, Kobayashi K, Narita Y, Kondo A, Shiokawa Y, Nagane M, Ichimura K. Liquid biopsy of cerebrospinal fluid for MYD88 L265P mutation is useful for diagnosis of central nervous system lymphoma. Cancer Sci. 2021 Nov;112(11):4702-4710. doi: 10.1111/cas.15133. Epub 2021 Sep 27. PMID: 34523186; PMCID: PMC8586690.
50. Gu J, Jiang T, Liu S, Ping B, Li R, Chen W, Wang L, Huang X, Xu G, Chang Q. Cell-Free DNA Sequencing of Intraocular Fluid as Liquid Biopsy in the Diagnosis of Vitreoretinal Lymphoma. Front Oncol. 2022 Jul 19;12:932674. doi: 10.3389/fonc.2022.932674. PMID: 35928872; PMCID: PMC9343589.
51. Baraniskin A, Schroers R. Liquid Biopsy and Other Non-Invasive Diagnostic Measures in PCNSL. Cancers (Basel). 2021 May 28;13(11):2665. doi: 10.3390/cancers13112665. PMID: 34071407; PMCID: PMC8198992.
52. Sasayama T, Nakamizo S, Nishihara M, Kawamura A, Tanaka H, Mizukawa K, Miyake S, Taniguchi M, Hosoda K, Kohmura E. Cerebrospinal fluid interleukin-10 is a potentially useful biomarker in immunocompetent primary central nervous system lymphoma (PCNSL). Neuro Oncol. 2012 Mar;14(3):368-80. doi: 10.1093/neuonc/nor203. Epub 2011 Dec 12. PMID: 22156547; PMCID: PMC3280797.
53. Shao J, Chen K, Li Q, Ma J, Ma Y, Lin Z, Kang H, Chen B. High Level of IL-10 in Cerebrospinal Fluid is Specific for Diagnosis of Primary Central Nervous System Lymphoma. Cancer Manag Res. 2020 Jul 24;12:6261-6268. doi: 10.2147/CMAR.S255482. PMID: 32801871; PMCID: PMC7386815.
54. Downs BM, Ding W, Cope LM, Umbricht CB, Li W, He H, Ke X, Holdhoff M, Bettegowda C, Tao W, Sukumar S. Methylated markers accurately distinguish primary central nervous system lymphomas (PCNSL) from other CNS tumors. Clin Epigenetics. 2021 May 5;13(1):104. doi: 10.1186/s13148-021-01091-9. PMID: 33952317; PMCID: PMC8097855.
55. Castillo, J. J., Olszewski, A. J., Cronin, A. M., Hunter, Z. R. & Treon, S. P. Survival trends in Waldenstrom macroglobulinemia: an analysis of the Surveillance, Epidemiology and End Results database. Blood 123, 3999-4000, doi:10.1182/blood-2014-05-574871 (2014).
56. Gertz, M. A. Waldenstrom macroglobulinemia: 2021 update on diagnosis, risk stratification, and management. Am J Hematol 96, 258-269, doi:10.1002/ajh.26082 (2021).
57. Xu, L. et al. Detection of MYD88 L265P in peripheral blood of patients with Waldenstrom’s Macroglobulinemia and IgM monoclonal gammopathy of undetermined significance. Leukemia 28, 1698-1704, doi:10.1038/leu.2014.65 (2014).
58. Gertz, M. A., Fonseca, R. & Rajkumar, S. V. Waldenstrom’s macroglobulinemia. Oncologist 5, 63-67, doi:10.1634/theoncologist.5-1-63 (2000).
59. Falini, B., Martelli, M. P. & Tiacci, E. BRAF V600E mutation in hairy cell leukemia: from bench to bedside. Blood 128, 1918-1927, doi:10.1182/blood-2016-07-418434 (2016).
60. Maitre, E., Cornet, E. & Troussard, X. Hairy cell leukemia: 2020 update on diagnosis, risk stratification, and treatment. Am J Hematol 94, 1413-1422, doi:10.1002/ajh.25653 (2019).
61. Xi, L. et al. Both variant and IGHV4-34-expressing hairy cell leukemia lack the BRAF V600E mutation. Blood 119, 3330-3332, doi:10.1182/blood-2011-09-379339 (2012).
62. Arons, E., Suntum, T., Stetler-Stevenson, M. & Kreitman, R. J. VH4-34+ hairy cell leukemia, a new variant with poor prognosis despite standard therapy. Blood 114, 4687-4695, doi:10.1182/blood-2009-01-201731 (2009).
63. Rangoonwala HI, Cascella M. Peripheral T-Cell Lymphoma. 2023 Aug 7. In:StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 32965972.
64. Wilcox, R. A. Cutaneous T-cell lymphoma: 2017 update on diagnosis, risk-stratification, and management. Am J Hematol 92, 1085-1102, doi:10.1002/ajh.24876 (2017).
65. Bagherani N, Smoller BR. An overview of cutaneous T cell lymphomas. F1000Res. 2016 Jul 28;5:F1000 Faculty Rev-1882. doi: 10.12688/f1000 research.8829.1. PMID: 27540476;PMCID: PMC4965697.
66. Trautinger, F. et al. European Organisation for Research and Treatment of Cancer consensus recommendations for the treatment of mycosis fungoides/Sezary syndrome – Update 2017. Eur J Cancer 77, 57-74, doi:10.1016/j.ejca.2017.02.027 (2017).
67. Agar, N. S. et al. Survival outcomes and prognostic factors in mycosis fungoides/Sezary syndrome: validation of the revised International Society for Cutaneous Lymphomas/European Organisation for Research and Treatment of Cancer staging proposal. J Clin Oncol 28, 4730-4739, doi:10.1200/JCO.2009.27.7665 (2010).
68. Ralfkiaer, U. et al. Diagnostic microRNA profiling in cutaneous T-cell lymphoma (CTCL). Blood 118, 5891-5900, doi:10.1182/blood-2011-06-358382 (2011).
69. Huang, Y. et al. Thymocyte selection-associated high mobility group box gene (TOX) is aberrantly over-expressed in mycosis fungoides and correlates with poor prognosis. Oncotarget 5, 4418-4425, doi:10.18632/oncotarget.2031 (2014).
70. Dulmage, B., Geskin, L., Guitart, J. & Akilov, O. E. The biomarker landscape in mycosis fungoides and Sezary syndrome. Exp Dermatol 26, 668-676, doi:10.1111/exd.13261 (2017).
71. Brunner, P. M., Jonak, C. & Knobler, R. Recent advances in understanding and managing cutaneous T-cell lymphomas. F1000Res 9, doi:10.12688/f1000research.21922.1 (2020).
72. Stein, H. et al. CD30(+) anaplastic large cell lymphoma: a review of its histopathologic, genetic, and clinical features. Blood 96, 3681-3695 (2000).
73. Medeiros, L. J. & Elenitoba-Johnson, K. S. Anaplastic Large Cell Lymphoma. Am J Clin Pathol 127, 707-722, doi:10.1309/r2q9ccuvtlrycf3h (2007).
74. Gascoyne, R. D. et al. Prognostic significance of anaplastic lymphoma kinase (ALK) protein expression in adults with anaplastic large cell lymphoma. Blood 93, 3913-3921 (1999).
75. Tsuyama, N., Sakamoto, K., Sakata, S., Dobashi, A. & Takeuchi, K. Anaplastic large cell lymphoma: pathology, genetics, and clinical aspects. J Clin Exp Hematop 57, 120-142, doi:10.3960/jslrt.17023 (2017).
76. Ait-Tahar, K. et al. Correlation of the autoantibody response to the ALK oncoantigen in pediatric anaplastic lymphoma kinase-positive anaplastic large cell lymphoma with tumor dissemination and relapse risk. Blood 115, 3314-3319, doi:10.1182/blood-2009-11-251892 (2010).
77. Lovisa F, Di Battista P, Gaffo E, Damanti CC, Garbin A, Gallingani I, Carraro E, Pillon M, Biffi A, Bortoluzzi S, Mussolin L. RNY4 in Circulating Exosomes of Patients With Pediatric Anaplastic Large Cell Lymphoma: An Active Player? Front Oncol. 2020 Feb 27;10:238. doi: 10.3389/fonc.2020.00238. PMID: 32175280; PMCID: PMC7056873.
78. Lovisa F, Garbin A, Crotti S, Di Battista P, Gallingani I, Damanti CC, Tosato A, Carraro E, Pillon M, Mafakheri E, Romanato F, Gaffo E, Biffi A, Bortoluzzi S, Agostini M, Mussolin L. Increased Tenascin C, Osteopontin and HSP90 Levels in Plasmatic Small Extracellular Vesicles of Pediatric ALK-Positive Anaplastic Large Cell Lymphoma: New Prognostic Biomarkers? Diagnostics (Basel). 2021 Feb 6;11(2):253. doi: 10.3390/diagnostics11020253. PMID: 33562105; PMCID: PMC7915848.
79. de Leval, L. et al. The gene expression profile of nodal peripheral T-cell lymphoma demonstrates a molecular link between angioimmunoblastic T-cell lymphoma (AITL) and follicular helper T (TFH) cells. Blood 109, 4952-4963, doi:10.1182/blood-2006-10-055145 (2007).
80. Vose, J., Armitage, J., Weisenburger, D. & International, T. C. L. P. International peripheral T-cell and natural killer/T-cell lymphoma study: pathology findings and clinical outcomes. J Clin Oncol 26, 4124-4130, doi:10.1200/JCO.2008.16.4558 (2008).
81. Iannitto, E., Ferreri, A. J., Minardi, V., Tripodo, C. & Kreipe, H. H. Angioimmunoblastic T-cell lymphoma. Crit Rev Oncol Hematol 68, 264-271, doi:10.1016/j.critrevonc.2008.06.012 (2008).
82. Loghavi, S. et al. Immunophenotypic and diagnostic characterization of angioimmunoblastic T-cell lymphoma by advanced flow cytometric technology. Leuk Lymphoma 57, 2804-2812, doi:10.3109/10428194.2016.1170827 (2016).
83. Shiratori, S. et al. Ultra-high level of serum soluble interleukin-2 receptor at diagnosis predicts poor outcome for angioimmunoblastic T-cell lymphoma. Leuk Lymphoma 56, 2592-2597, doi:10.3109/10428194.2014.1001985 (2015).
84. Kim SJ, Kim YJ, Yoon SE, Ryu KJ, Park B, Park D, Cho D, Kim HY, Cho J, Ko YH, Park WY, Kim WS. Circulating Tumor DNA-Based Genotyping and Monitoring for Predicting Disease Relapses of Patients with Peripheral T-Cell Lymphomas. Cancer Res Treat. 2023 Jan;55(1):291-303. doi: 10.4143/crt.2022.017. Epub 2022 Mar 2. PMID: 35240014; PMCID: PMC9873338.
85. Ottolini B, Nawaz N, Trethewey CS, Mamand S, Allchin RL, Dillon R, Fields PA, Ahearne MJ, Wagner SD. Multiple mutations at exon 2 of RHOA detected in plasma from patients with peripheral T-cell lymphoma. Blood Adv. 2020 Jun 9;4(11):2392-2403. doi: 10.1182/bloodadvances.2019001075. PMID: 32484856; PMCID: PMC7284097.
86. Oluwasanjo A, Kartan S, Johnson W, Alpdogan O, Gru A, Mishra A, Haverkos BM, Gong J, Porcu P. Peripheral T-Cell Lymphoma, not Otherwise Specified (PTCL-NOS). Cancer Treat Res. 2019;176:83-98. doi: 10.1007/978-3-319-99716-2_

