Pulmonary Arteriovenous Malformations in Pregnancy: Insights
Pulmonary arteriovenous malformations in pregnancy – a case report and review of literature
Pulmonary arteriovenous malformations, pregnancy, hereditary hemorrhagic telangiectasia, maternal health, fetal health.
Introduction
Arteriovenous malformations (AVM) are vascular anomalies due to aberrant communications between arterial and venous systems, most commonly affecting the lungs, brain, and liver¹. When looking specifically at pulmonary arteriovenous malformations (PAVM), approximately 3 in every 100,000 people of the general population are affected. Upwards of 80% of these cases are associated with an inherited disorder like hereditary hemorrhagic telangiectasia (HHT)²,³. HHT is an autosomal dominant condition that has a much higher prevalence, affecting up to 1 in every 5,000 people⁴. Approximately 50% of patients are asymptomatic from this disease process, particularly if PAVMs are less than 2 cm in diameter, but these are still identifiable by the third decade of life for most people²,⁵.
Despite the rarity and varied clinical presentation of PAVM, patients of reproductive age require guidance regarding the effect pregnancy may have on their disease process. Significant alterations in estrogen, vascular growth factors, cardiac output and progesterone induced vasodilation may result in PAVM growth and subsequent pulmonary hemorrhage, cardiopulmonary collapse, and maternal morbidity and mortality. Existing literature describes an increased risk of poor maternal and potentially fetal outcomes in pregnancies affected by PAVM hemorrhage, but there is still a paucity of clinical guidance regarding the optimal management of obstetric patients affected by PAVMs⁶.
We aim to discuss a case in which maternal mortality was avoided in a patient with PAVM. Through multidisciplinary collaboration and patient education, the morbidity associated with this disorder can be minimized, and maternal, fetal, and neonatal outcomes optimized. Founded on the physiologic changes of pregnancy, we justify a framework for multidisciplinary obstetric care that may improve preconception, antenatal, and postpartum management of patients affected by HHT and similar disease processes driven by arteriovenous malformations.
Case
The patient is a 31-year-old gravida 4, para 2 who presented for consultation at 30 weeks gestation. Her medical history was significant for previous embolization of more than 20 PAVM in the setting of autosomal dominant HHT, which was diagnosed via genetic testing at age 15. Her first two pregnancies were complicated by ruptured PAVM in the postpartum period that required vascular coil placement. In her second pregnancy, she also experienced ruptured PAVM during her antenatal course around 29 weeks gestation, and subsequently had a successful vaginal birth after cesarean at term.
She presented to the high-risk obstetrics clinic in the third trimester of pregnancy for coordination of care and planned delivery at a tertiary medical center with an interventional radiology team. At the time of consultation and transfer of obstetrical care, she was without recent imaging and had yet to establish care with a multidisciplinary team. We recommended urgent chest and brain imaging, maternal echocardiogram, and an assisted second stage of labor if imaging identified untreated PAVM that could not be managed prior to delivery. The patient then met with our obstetric anesthesia team and neonatal intensive care unit. Once known to all members of the multidisciplinary team that would be part of her delivery, an emergency action plan was developed.
This plan emphasized that if the patient presented with concerning symptoms, immediate chest imaging should be obtained and the interventional radiology, surgical intensive care unit, neonatal intensive care unit, and obstetric anesthesia teams should be promptly notified. If cesarean delivery was required, this should occur in an operating room with proximity to the surgical intensive care unit (ICU).
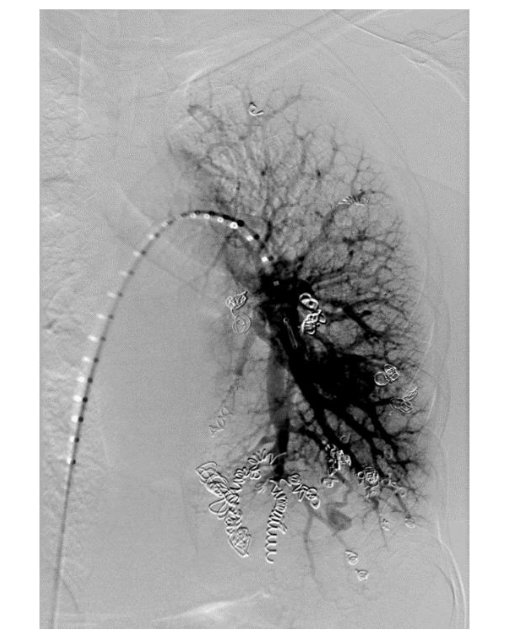
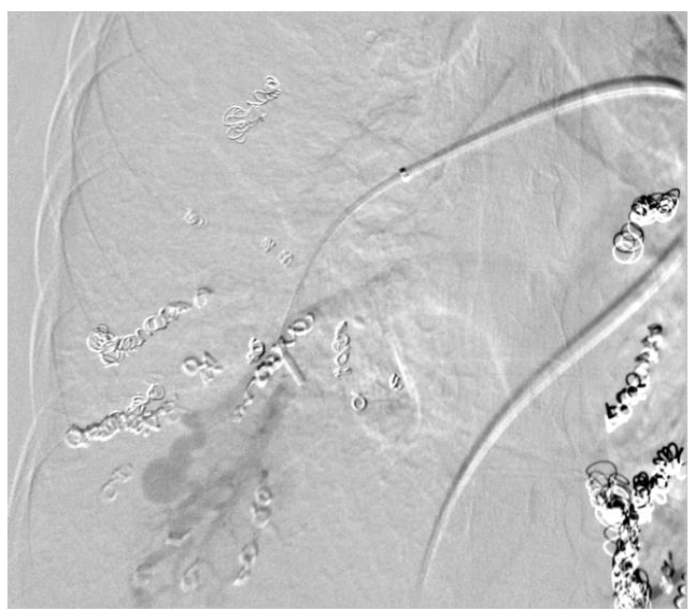
The recommended imaging studies were significant for numerous bilateral PAVM and she underwent placement of 10 total vascular coils antenatally. Unfortunately, the largest PAVM measured 32mm and was located within the left lingula, where a feeding vessel could not be identified. The next largest PAVM measured 17 mm (located in the right lower lobe) and 23 mm (located in the right upper lobe), both of which were successfully treated.
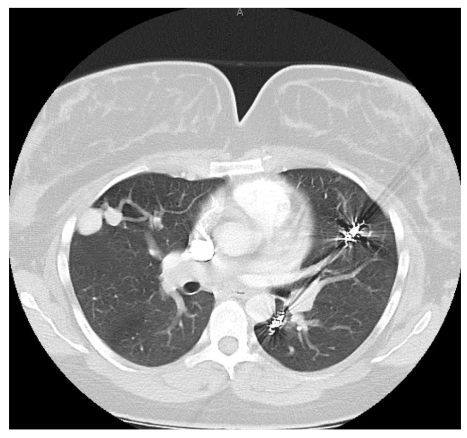
At 38 weeks 4 days estimated gestational age, the patient presented to the hospital with complete cervical dilation and fetal malpresentation. A repeat C-section was performed followed by direct admission to the surgical ICU for recovery. She was downgraded to the postpartum floor later that evening after an uneventful and brief surgical ICU course.
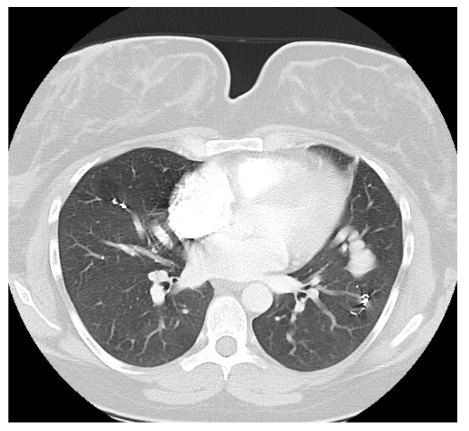
On postpartum day 1, the patient experienced an episode of acute onset hemoptysis, oxygen desaturation, tachycardia, and tachypnea. Prompt laboratory and imaging studies were obtained and pulmonology, anesthesia, and high-risk obstetrics were consulted. The patient immediately returned to the ICU where she was intubated due to persistent hypoxia. Repeat chest imaging was consistent with pulmonary hemorrhage. This was managed with urgent PAVM embolization, at which time a total of 4 vascular coils were placed in a right lower lobe PAVM that measured 17 mm. Notably, this was not one of the larger PAVMs targeted during antenatal embolization. The patient was subsequently extubated on postpartum day 3 and returned to the postpartum floor on postpartum day 4. The remainder of her hospital course was unremarkable and she was discharged home on postpartum day 5 in stable condition with appropriate follow up.
Discussion
Based on available data, the risk of PAVM rupture and subsequent hemorrhagic risk is approximately 1-2% in pregnancy and the immediate postpartum period. This is significantly higher than the overall 0.16% PAVM rupture risk in the non-pregnant state, and approaches the 2.7% lifetime risk of PAVM rupture. This drastic elevation in risk can be attributed to the numerous physiologic cardiovascular and respiratory changes of pregnancy and labor.
Some of the most notable physiologic changes to consider are the following: blood volume increase of up to 45%, increase in arterial and venous compliance, increase in cardiac output by approximately 30%, and increase in heart rate by approximately 15-20%. These changes can lead to an increased risk of rupture in patients with PAVM.
Conflict of Interest:
None
Acknowledgements:
None
References
1. Dupuis O, Delagrange L, Dupuis-Girod S. Hereditary haemorrhagic telangiectasia and pregnancy: a review of the literature. Orphanet J Rare Dis. 2020 Jan 7;15(1):5. doi: 10.1186/s13023-019-1286-z. PMID: 31910869; PMCID: PMC6947864.
2. Khurshid I, Downie GH. Pulmonary arteriovenous malformation. Postgrad Med J. 2002;78(918):191-197. doi:10.1136/pmj.78.918.191
3. Dines DE, Seward JB, Bernatz PE. Pulmonary arteriovenous fistulas. Mayo Clin Proc. 1983;58(3): 176-181.
4. Che Yusof R, Norhayati MN, Mohd Azman Y. Arteriovenous Malformation Hemorrhage in Pregnancy: A Systematic Review and Meta-Analysis. Int J Environ Res Public Health. 2022;19(20):13183. Published 2022 Oct 13. doi:10.3390/ijerph192013183
5. Esplin MS, Varner MW. Progression of pulmonary arteriovenous malformation during pregnancy: case report and review of the literature. Obstet Gynecol Surv. 1997;52(4):248-253.
doi:10.1097/00006254-199704000-00022
6. Lv X, Liu P, Li Y. Pre-existing, incidental and hemorrhagic AVMs in pregnancy and postpartum: Gestational age, morbidity and mortality, management and risk to the fetus. Interv Neuroradiol. 2016;22(2):206-211. doi:10.1177/1591019915622161
7. Christopher F Ciliberto, G.F.M., Darryl Johnston, Physiological Changes Associated with Pregnancy. Update in Anesthesia, 2008. 24(2): p. 72-76.
8. Trerotola SO, Pyeritz RE. PAVM embolization: an update. AJR Am J Roentgenol. 2010;195(4):837-845. doi:10.2214/AJR.10.5230
9. Shovlin CL, Sodhi V, McCarthy A, Lasjaunias P, Jackson JE, Sheppard MN. Estimates of maternal risks of pregnancy for women with hereditary haemorrhagic telangiectasia (Osler-Weber-Rendu syndrome): suggested approach for obstetric services. BJOG. 2008;115(9):1108-1115. doi:10.1111/j.1471-0528.2008.01786.x
10. de Gussem EM, Lausman AY, Beder AJ, et al. Outcomes of pregnancy in women with hereditary hemorrhagic telangiectasia. Obstet Gynecol. 2014; 123(3):514-520. doi:10.1097/AOG.0000000000000120
11. Fish A, Henderson K, Moushey A, Pollak J, Schlachter T. Incidence of Spontaneous Pulmonary AVM Rupture in HHT Patients. J Clin Med. 2021;10 (20):4714. Published 2021 Oct 14. doi:10.3390/jcm10204714
12. Lund CJ, Donovan JC. Blood volume during pregnancy. Significance of plasma and red cell volumes. Am J Obstet Gynecol. 1967;98(3):394-403. doi:http://doi.org/10.1016/0002-9378(67)90160-3.
13. Pritchard JA. Changes in the blood volume during pregnancy. Anesthesiology. 1965;26:393-399. doi:10.1097/00000542-196507000-00004
14. Spaanderman ME, Willekes C, Hoeks AP, Ekhart TH, Peeters LL. The effect of pregnancy on the compliance of large arteries and veins in healthy parous control subjects and women with a history of preeclampsia. Am J Obstet Gynecol. 2000;183 (5):1278-1286. doi:10.1067/mob.2000.106750
15. Charles J. Lockwood, J.A.C., Lorraine L. Dugoff, Judette Louis, Thomas R. Moore, Robert M. Silver, Robert Resnik, Creasy and Resnik’s Maternal-Fetal Medicine. 9 ed. 2022: Elsevier.
16. Lukic A, Cmelak L, Dominik Drazenovic, Hrvoje Kojundzic, Lukic IK, Vicko Gluncic. Pulmonary Arteriovenous Malformation Unmaked by Pregnancy: A Review of Pulmonary Arterious Malformations and Cardiovascular and Respiratory Changes in Pregnancy. Case Reports in Pulmonology. 2023; 2023:1-8. doi.org/10.1155/2023/5469592.

