Minimally Invasive Treatment for Lumbar Spondylolisthesis
Minimally Invasive Cost-Effective Surgical Treatment of Lumbar Spondylolisthesis with Associated Spinal Stenosis
Miguelangelo Perez-Cruet¹²³, M.D., M.Sc., Jordan Black¹, M.D., M.Sc., Ishan Singhal¹, M.D., Daniel Fahim¹²⁴, M.D.
- Department of Neurosurgery, Corewell Health, William Beaumont, University Hospital, Royal Oak, MI, USA
- Department of Neurosurgery, Oakland University William Beaumont, School of Medicine, Auburn Hills, MI, USA
- Michigan Minimally Invasive Spine and Brain Surgery Specialists, Royal Oak, MI, USA
OPEN ACCESS
PUBLISHED:30 November 2024
CITATION: Cruet, M.P., et al., 2024. Minimally Invasive Cost-Effective Surgical Treatment of Lumbar Spondylolisthesis with Associated Spinal Stenosis. Medical Research Archives, [online] 12(11).
https://doi.org/10.18103/mra.v12i11.6163
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i11.6163
ISSN 2375-1924
ABSTRACT
Purpose
The paraspinal approach and functional anatomy sparing nature of the minimally invasive transforaminal lumbar interbody fusion are thought to decrease operative morbidity while improving patient recovery and long-term outcomes. This paper presents a unique minimally invasive approach for the treatment of patients with lumbar spondylolisthesis and associated spinal stenosis. The novel technique described within allows for significant reduction of spondylolisthesis, while also utilizing a substantial amount of the patient’s morselized surgical site autograft, thus limiting the use of costly bone graft extenders. We aim to validate this technique by performing a critical analysis of postoperative patient reported outcomes with comparison to traditional open midline transforaminal lumbar interbody fusion.
Methods
Data was reviewed for 19 patients who underwent the minimally invasive transforaminal lumbar interbody fusion technique described within and subsequently compared to 140 patients who underwent traditional open midline transforaminal lumbar interbody fusion; all of whom were within the control groups of an FDA approved IDE clinical trial (NCT03115983). Operative and demographic data were collected and compared including age, gender, BMI, comorbidities, operative level, procedure time, estimated blood loss, postoperative length of stay. Pre and postoperative patient reported outcome scores were also analyzed for both groups including visual analogue score (VAS) for back and leg/hip pain and Oswestry disability index (ODI).
Results
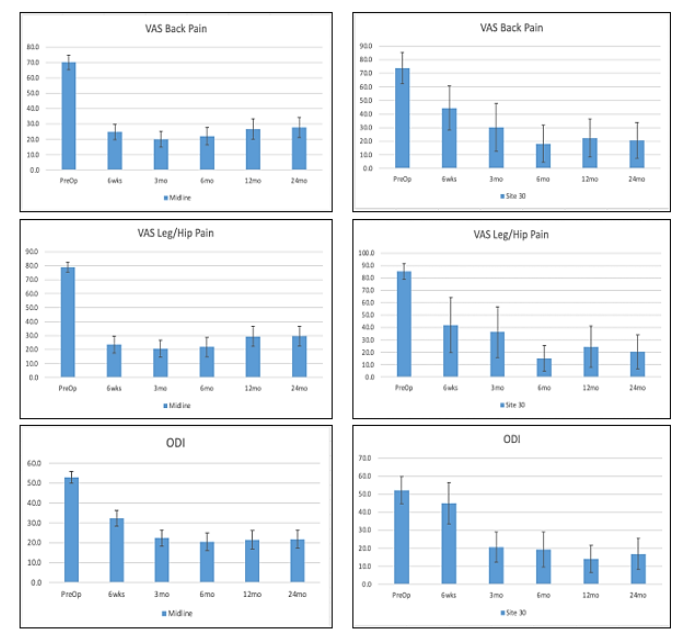
Procedure length (minutes), estimated blood loss (mL), and length of hospital stay (days) for the patients undergoing minimally invasive transforaminal lumbar interbody fusion were 156 +/- 31, 72 +/- 32, and 2.6 +/- 1.8, respectively. Compared to traditional midline transforaminal lumbar interbody fusion procedure time, estimated blood loss, and length of hospital stay of 189 +/- 78, 273 +/- 252, 3.1 +/- 1.7, respectively. VAS back and leg/hip pre-operatively were 79.3, 85.3, and 52.2 versus 20.6, 20.3 and 16.9 points, respectively at 2-year follow-up for the minimally invasive approach. Compared to traditional midline approach 69.7, 78.8, and 52.7 versus 27.9, 27.8 and 22.4 at 2-year follow-up. These improvements were all statistically significant at the 5% level.
Conclusion
The minimally invasive approach described in this paper, when compared to traditional midline transforaminal lumbar interbody fusion, resulted in shorter operative time, decreased estimated blood loss, shorter length of stay, and improvements in patient reported functional outcomes. This novel minimally invasive surgical approach to transforaminal lumbar interbody fusion is an excellent surgical treatment for refractory lumbar spondylolisthesis with associated stenosis.
Introduction
Low back pain is one of the most common causes of chronic pain and disability worldwide. Studies of American adults estimate that as much as 80% of the population will suffer from back pain during their lifetime. Depending on the etiology, back pain can be managed conservatively to temper symptoms while reducing the severity and incidence of pain exacerbations; however, patients suffering from refractory lumbar spondylolisthesis with associated stenosis can be surgically treated with laminectomy and fusion using either an open or minimally invasive approach³⁷.
A disadvantage of open surgery is the need to detach paraspinous muscles from the spine and remove bone elements that are unrelated to the underlying pathology, most notably the spinous processes⁸. These structures are vital to the long-term health of the spine and their removal can potentially lead to adjacent level pathology, scar formation, and the need for additional spine surgery⁹. This paper describes a technique that allows for direct decompression of the spinal canal while sparing the paraspinous muscles and spinous process through a muscle dilating approach. In addition, the primary fusion material is the patient’s own morselized autograft harvested from the surgical site during decompression, which has been shown to reduce graft site morbidity, maintain cost effectiveness, and achieve high fusion rates¹⁰. Using percutaneous pedicle screws in combination with a unique interbody graft cage system, most spondylolisthesis can be reduced to grade 0. This is felt to improve long term patient generated outcomes by restoring canal and foraminal diameter and sagittal alignment¹¹–¹³.
Minimally invasive transforaminal lumbar interbody fusion (MI-TLIF) was developed as a safer, equally effective technique for lumbar spine fusion compared to the traditionally open transforaminal lumbar interbody fusion (TLIF). Several benefits of the MI-TLIF have been explored in the literature. MI-TLIFs have demonstrated consistently superior short-term outcomes when compared to open TLIFs as measured by blood loss, length of hospital stay, readmission rates, and postoperative recovery time¹⁴–¹⁸. Minimally invasive techniques also reduce opioid dependence in patients undergoing spine surgery by reducing the time spent in the in-patient setting¹⁹–²⁰. Procedure related adverse events are also less common following MI-TLIF compared to open TLIF, and patients treated with an MI-TLIF are more likely to report improvements in both short and long-term functional status and pain indices¹⁴–¹⁸. Radiographically, the MI-TLIF has been shown to reduce pathological lordotic misalignment of the spine and achieve fusion with similar success as the open TLIF while minimizing blood loss²¹,²².
While most of the benefits of a MI-TLIF result from the technique itself, improvements in postoperative outcomes may also be related to the innovation in instruments and technology. The MI-TLIF is commonly defined by 3 major operative features: a paramedian/lateral incision, the use of muscle sparing dilation, and enhanced visualization; however, this still allows for significant variability between MI-TLIF procedures²³. Several contributions to the literature have explored nuances in instrument technology with the goal of improving fusion rates and patient reported outcomes while lowering rates of complication, subsidence, and pseudoarthrosis. The use of expandable cages, independent blade retractor systems, unique cage configurations, tubular dilators, and percutaneous pedicle screw systems underscores the fluidity of the MI-TLIF procedure²⁴,²⁵. Each of these variations seek to improve patient outcomes and fusion rates while mitigating the risk of complications. Notably, there is a relative paucity of clinical data regarding technical variations using existing technology and the impact on procedural adverse events and patient reported outcome.
This case series highlights a unique variation in the MI-TLIF instruments and technology with critical analysis of patient reported outcomes as part of an FDA IDE approved clinical trial. In this study, novel instruments and technology were developed that allow insertion of the patient’s own surgical site morselized autograft into the disc space around the
interbody cage device. The autograft is contained by the anulus fibrosus of the disc and allows for off-loading of the cage to promote fusion via Wolff’s law, thus minimizing cage subsidence and maximizing interbody autograft implantation to promote high fusion rates.
Methods
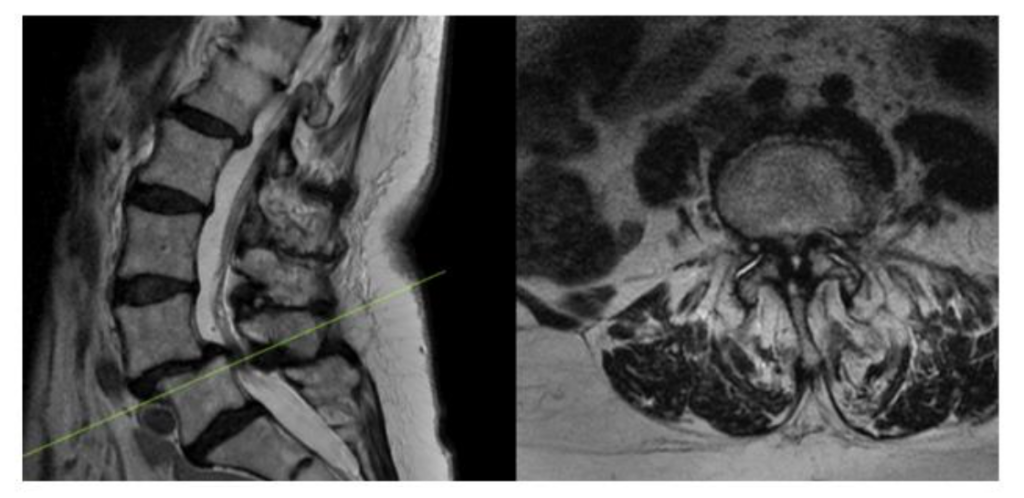
As part of an FDA approved device investigation study (NCT03115983) with Western IRB approval, between March 2016 and June 2020 all patients undergoing MI-TLIF with a single surgeon at our institution were prospectively reviewed to determine compliance with study parameters. Inclusion criteria included the presence of at least Grade 1 lumbar spondylolisthesis (per Meyerding classification) with associated spinal stenosis at one level between L1-S1 on either CT myelogram or MRI of the lumbar spine (Figure 1). All patients had to have experienced symptoms of neurogenic claudication or lumbar radiculopathy persisting for ≥6 months despite conservative management. Baseline patient reported outcomes thresholds were ≥50/100 for VAS back scores, and ≥35/100 for ODI scores. Only skeletally mature patients between 25–80 years of age were included. Patient capacity was assessed, and after the relevant risk and benefits of the procedure were discussed, the required informed consent was obtained.
Figure 1. Preoperative MRI showing L4-5 Grade 1 Spondylolisthesis with associated stenosis
Several exclusion criteria were also outlined. Patients suffering from discogenic pain, facet mediated pain, or radicular pain without anatomical abnormalities were excluded. Patients with confounding comorbidities, including peripheral vascular disease, peripheral neuropathy unrelated to spinal stenosis, osteomalacia or osteoporosis were excluded. Patients who had suffered previous back injury or had undergone any operative procedure at the level of treatment were excluded. Patients with degenerative scoliosis with a Cobb angle >10° or ankylosed segment at the operative level were excluded. Patients who were allergic to titanium or polyethylene or actively receiving immunomodulatory medications were ineligible. Morbidly obese patients (BMI > 40), patients with a history of malignancy, or patients intending to become pregnant during the study period were excluded. Patients suffering from psychiatric illness or with a current/prior diagnosis of substance abuse were excluded. 245 patients were consented and underwent MI-TLIF during that time course, and after applying our inclusion and exclusion criteria, as visualized in figure 2, nineteen patients qualified for inclusion given our study
parameters. Patients undergoing traditional open midline TLIF were also put through the same screening process and 140 patients were included for analysis from that cohort during the same time course. Preoperatively, beyond their initial CT myelogram and/or MRI lumbar spine all patients also were evaluated with anteroposterior, lateral, flexion, and extension X-rays of the lumbar spine,
Osteoporotic Self-Assessment Tool evaluation, and if there was an history of fragility fracture they also underwent a DXA or CT scan for further evaluation. Lumbar spine X-rays were also repeated at discharge, 6 weeks, 3, 6, 12, 24, 36, 48, and 60 months postoperatively in accordance with the FDA device investigation study protocol.
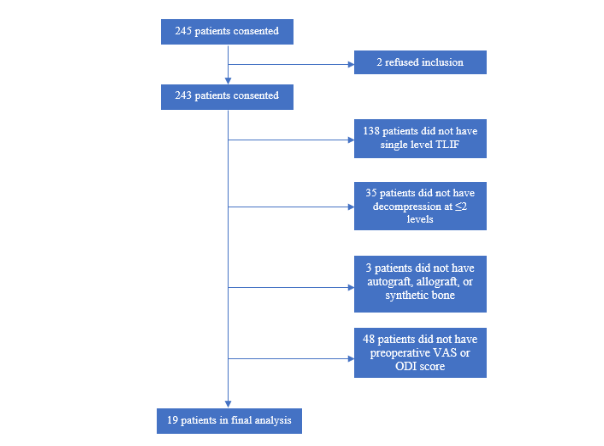
Figure 2. Flowchart illustrating MI-TLIF Patient selection details
Detailed neurological examinations, physical/medical therapy utilization, pain medication usage, leg and back pain assessments utilizing visual analog scales (VAS), Zurich Claudication Questionnaires, Oswestry Disability Index (ODI), 12-Item Short Form Health Surveys (SF-12), ability to return to work, patient overall satisfaction, and adverse events were also evaluated and recorded at 6 weeks, 3, 6, 12, 24, 36, 48, and 60 months postoperatively. Demographic, procedural, pre- and post-operative outcomes data were critically reviewed for 19 patients who underwent MI-TLIFs and TLIFs per FDA investigational device exemption (IDE) monitoring guidelines²⁶. Due to the concurrence of the COVID-19 pandemic, patient follow-up visits were transitioned from in-person office visits to scheduled telemedicine visits with visual neurological evaluations. Table 1 includes a demographic comparison of the control groups.
Due to the relatively small sample size, non-parametric Friedman ANOVA followed by Wilcoxon rank sum tests were conducted to investigate the statistical significance of functional and clinical improvements. An alpha level of 5% was used to assess statistical significance.
Table 1. Patient Demographic Data Undergoing Novel MI-TLIF versus Traditional TLIF
| Novel MI-TLIF | Traditional TLIF | |
|---|---|---|
| Sex ratio (M:F) | 7:12 | 40:100 |
| Mean Age | 65 +/- 9 | 64 +/- 9 |
| BMI | 32 +/- 6 | 30 +/- 6 |
| L4-5 index level (n, %) | 18 (95) | 120 (86) |
| L5-S1 index level (n, %) | 1 (5) | 8 (14) |
Surgical Technique
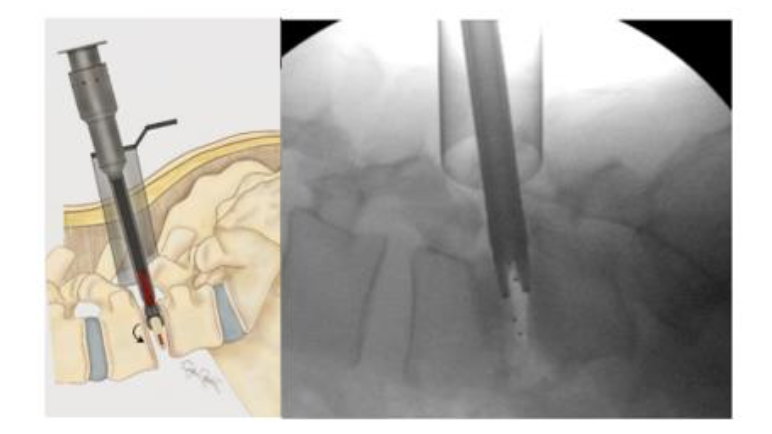
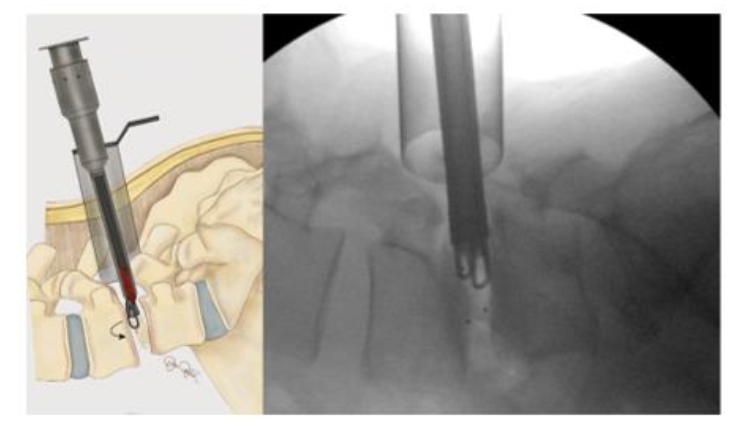
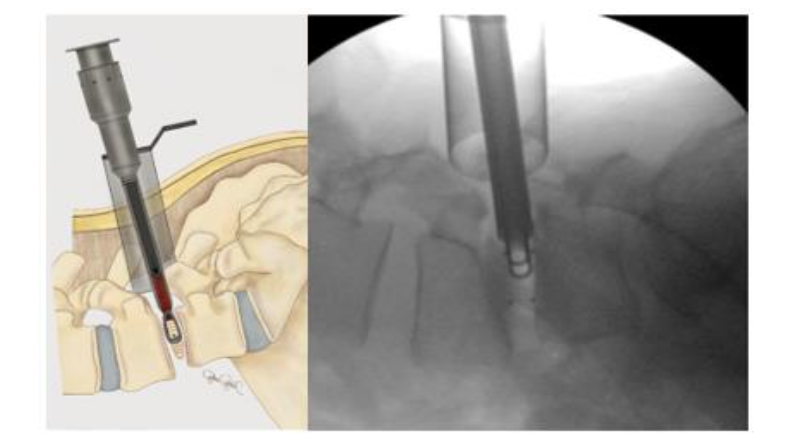
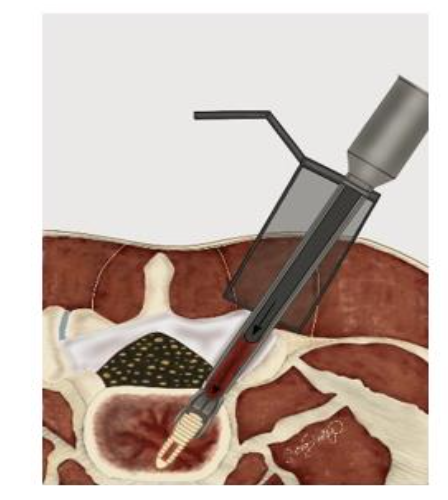
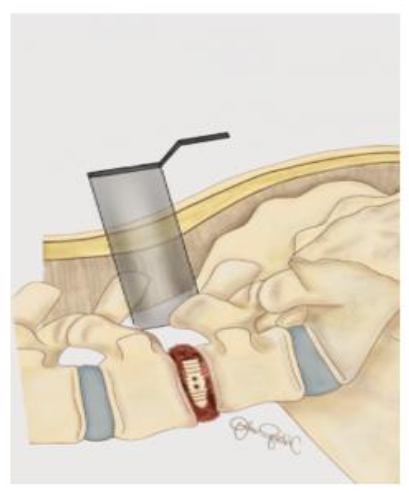
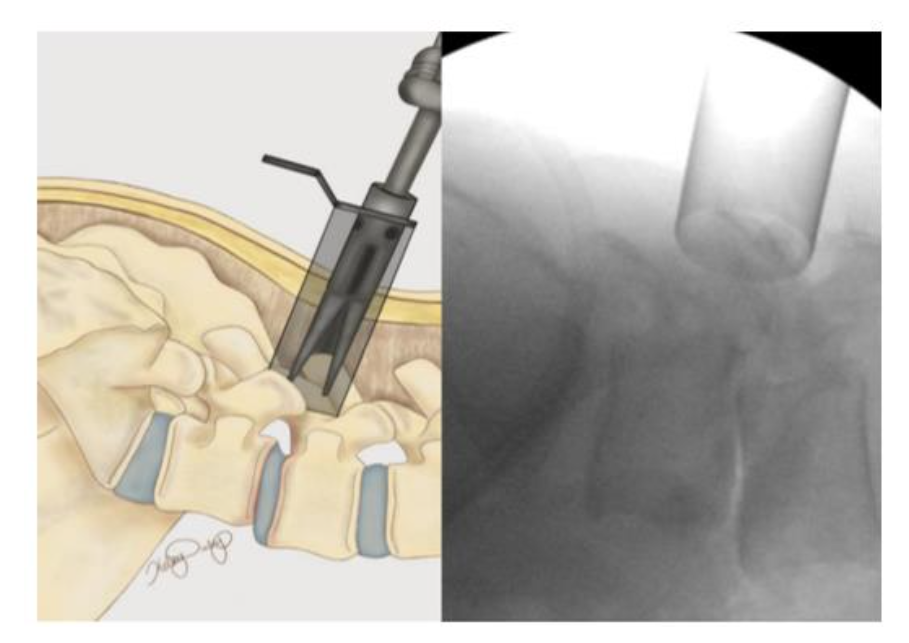
The patient is positioned prone on a spinal Jackson table the area of interest is prepped and draped in a sterile fashion. The index level is localized utilizing lateral fluoroscopy. An incision is then made 3 cm lateral to the midline overlying the appropriate disc space. The fascia is incised parallel to the spinous processes and the One-Step-Dilator (BoneBac/Thompson MIS, Sandown, NH) is used to approach the spine in a muscle sparing fashion (Figure 3). The dilator is supported by a holder and once docked on the facet, counter clock-wise rotation opens the flanges of the dilator, separating the muscle tissue. A tubular retractor of the appropriate length is then placed over the one step dilator and the dilator is removed. The tubular retractor is attached to a support arm (Walter Arm, Zimmer Spine) which has been fastened to the operative table.
Figure 3. One-step dilator (Thompson MIS/Bonebac, Sandown, NH) placement
LUMBAR EXPOSURE AND DECOMPRESSION
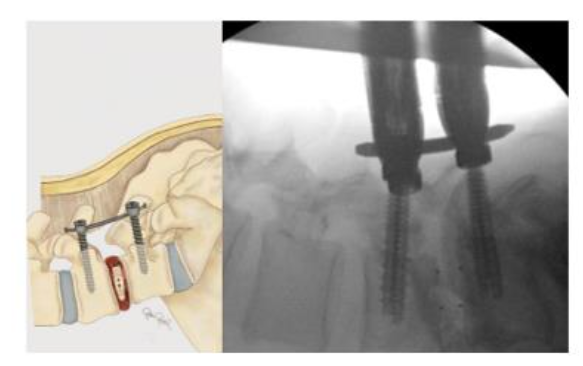
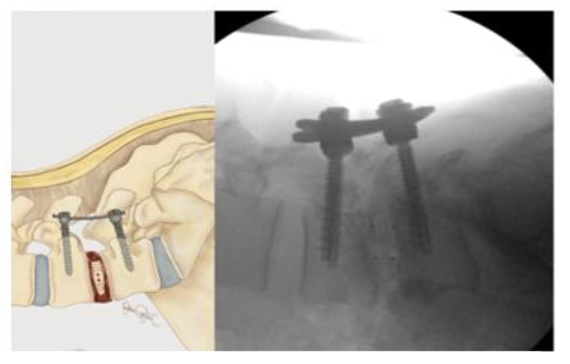
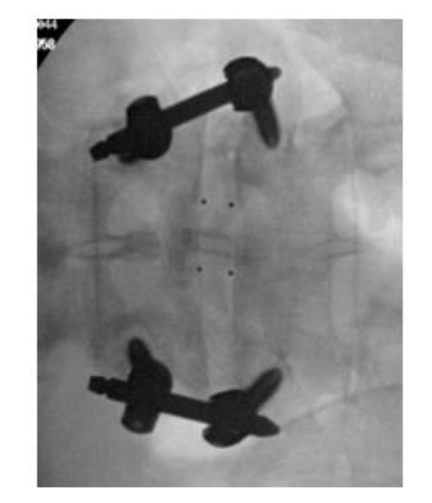
Utilizing an operative microscope the soft tissue is removed to expose the facet complex laterally, and the ipsilateral lamina medially. A high-speed drill and M8 cutting burr are used to perform the laminectomy. All drilled bone is collected using the BoneBac™ Press (BoneBac/Thompson MIS, Sandown, NH). This bone autograft avoids graft site morbidity, has exceptional handling characteristics, and can be mixed with other bone graft material as needed. After the ipsilateral laminectomy (Figure 4A) is completed, the patient and retractor are then tilted 5–10 degrees away from the surgeon to expose the base of the spinous process and the contralateral lamina which is then undercut with a cutting burr as far as the medial border of the contralateral facet complex (Figure 4B). After adequate bony decompression has been achieved, the hypertrophied ligamentum flavum is removed bilaterally, first on the ipsilateral side (Figure 4C) and then on the contralateral side (Figure 4D) which provides improved space for safely removing the contralateral ligamentum flavum, and limits durotomy. Inspection using a ball ended micro-probe instrument assures adequate direct decompression of the spinal canal (Figure 4E).

INTERBODY FUSION
Once adequate decompression is achieved, a high speed cutting burr is used to perform an ipsilateral facetectomy after which the disc space is identified using fluoroscopy, and an annulotomy is then performed to enter the disc space. A series of disc space reamers, curettes, and rongeurs help prepare the disc space for interbody arthrodesis. Care is taken to adequately prepare the vertebral end plates by removing the cartilaginous portions to promote arthrodesis. Once the disc space is thoroughly prepared, implant sizing is determined using the BoneBac/Thompson MIS reamers and trials.