Reversible Myocardial Changes in Severe Hypothyroidism
Increased Myocardial Wall Thickness in a Patient with Severe Hypothyroidism, Showed Complete Reversibility
Case Report and Literature Review
Dr. Areeg Magzoub babiker El Tayeb¹, Dr. Omnia Mutaz Mamoon Yousif², Dr. Mohamed Alaa Eldin Mohamed Ahmed³, Dr. Abdelazez Allaeldin Mohamed Ahmed⁴
- Gezira University, Sudan; Consultant Physician and Adult Cardiology, Department of Medicine and Cardiology, Alfajr College for Medical Science and Technology, Khartoum, Sudan; Department of Medicine and Cardiology, Ribat University Hospital, Khartoum, Sudan; Department of Medicine and Cardiology, Fedail Hospital, Khartoum, Sudan
- National University, Sudan; Master of Public Health and Tropical Disease, University of Medical Science and Technology, Sudan
- Sudan International University, Sudan; Imperial Hospital, Sudan
- Sudan International University, Sudan; Ribat University Hospital
OPEN ACCESS
PUBLISHED: 30 December 2024
CITATION: El Tayeb Areeg M B, Yousif O M, Alaa Eldin M, Allaeldin A. 2024. “Increased Myocardial Wall Thickness in a Patient with Severe Hypothyroidism, Showed Complete Reversibility After Thyroid Hormone Replacement Therapy” Case Report and Literature Review. Medical Research Archives, [online] 12(12). https://doi.org/10.18103/mra.v12i12.6095
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i12.6095
ISSN 2375-1924
ABSTRACT
Introduction:
Hypothyroidism affects the cardiovascular system in various ways. This may include bradycardia, diastolic hypertension, increased systemic vascular resistance (SVR), decreased cardiac contractility, and cardiac output. Increased myocardial thickness is seldomly reported. The thickness can be secondary to edema, hypertrophy, substance deposition, or the combination of all three.
Case summary:
In this case report and review we present a patient with severe hypothyroidism and massive recurrent pericardial effusion, leading to cardiac tamponade that needed drainage twice at one-month intervals. Increased myocardial wall thickness of both the septum and the posterior walls was detected. The increased thickness completely recovered in six months duration after thyroid hormone replacement and restoration of euthyroid status. We ran a review on the effects of hypothyroidism on the heart and vascular system, with a focus on the incidence, possible etiology, and reversibility of increased tissue thickness in different body organs in hypothyroid patients. These organs are the skin, the myocardium, and the carotid intima.
Conclusion:
Severe hypothyroidism can cause increased myocardial thickness that is reversible after thyroid hormone replacement therapy. Increased thickness is also seen in the skin and the carotid intima. The pathophysiology is probable of various etiology, however myocardial edema seems to be a strong reason considering the quick recovery after treatment.
Keywords:
Case report, Hypothyroidism, Myocardial thickness, Pericardial effusion, Carotid intimal thickness
Introduction
Thyroid disease is a common medical condition. It affects both genders with clear female predominance. The main reason of this predominance is the prevalence of auto-immune thyroid diseases in females compared to males, namely Grave’s and Hashimoto’s disease. Estimates showed that thyroid disease affects around (9-15%) of the adult female population and a smaller percentage of adult males. However, around the eighth decade of life, the incidence of disease in males is almost equal to that of females¹. Symptoms of hypothyroidism can vary from simple fatigue to its extreme presentation with myxoedema coma. The most common age group for hypothyroidism in many studies is 40-50 years².
Both hyperthyroidism and hypothyroidism produce changes in cardiac output, contractility, blood pressure, systemic vascular resistance, and myocardial oxygen consumption³⁴. In this case report and literature review, we will discuss the effects of hypothyroidism on the cardiovascular system as well as the skin. The focus will be the presence, possible etiology, and reversibility of increased tissue thickness in various organs including the skin, the myocardium and the vascular (carotid) intimal media.
Increased tissue thickness in hypothyroidism seems to be caused by many factors. In addition to hypertrophy, tissue edema is a main component as well as substance deposition like myosin and hyaluronic acid leading to further salt and water trapping. Tissue biopsy is the gold standard to confirm the consistency, however new modalities like cardiac magnetic resonance facilitated the recognition non-invasively.
Case History
The patient is a 72-year-old male who was known to be hypertensive with mild renal impairment, base line Creatinine = 1.3mg/dl. The patient presented with increasing shortness of breath and dizziness. His examination revealed raised jugular venous pressure, bi-basal crepitations, tender hepatomegaly and bilateral lower limb edema. His vitals were stable with a blood pressure of 150/80 and a pulse rate of 72 per minute. Initial laboratory investigations showed a Hemoglobin level of 11.9 g/dl and increased mean cell volume (MCV) of 106 FL, total white blood cell count (TWBC) of 4. Normal C reactive protein at 5mg/L, but a very high thyroid stimulating hormone (TSH) at 59 µIU/ml, with reduction in both T3 and T4 at 1.5 and 2.5 pmol/L respectively. Trans-thoracic echocardiography showed massive pericardial effusion of 6.5 cm seen more around the left ventricle as well as right atrial and right ventricular collapse (Figure 1 and 2). The left atrium was dilated at 5 cm and there was left ventricular hypertrophy with end diastolic wall thickness of the septum and the posterior wall at 33 and 18 mm respectively. The diastolic function was impaired and there was mild tricuspid regurgitation and a dilated non collapsing inferior vena cava. The left ventricular systolic function and ejection fraction were normal at 65%. The patient was diagnosed with cardiac tamponade and heart failure and was referred for diagnostic and therapeutic pericardiocentesis. Analysis of the pericardial fluid showed high protein levels but sugar, lactate dehydrogenase (LDH), and cell count were normal. Fluid culture was negative for bacterial growth, malignant cells, as well as Acid Fast Bacilli. The patient was started on Levothyroxine replacement at a dose of 150 micrograms per day as well as diuretic therapy for control of the heart failure. One month later, on his follow up visit, he was again symptomatic with shortness of breath and dizziness. Thyroid function tests improved with a TSH of 15, T3 of 3 and T4 of 13pmol/L. However, the repeated echocardiography showed re-accumulation of the pericardial fluid (6cm) with partial collapse of both right atrium and right ventricle that necessitates a second pericardiocentesis. Re-analysis of the fluid gave the same results. The thyroxin replacement continued with clinical, laboratory and echocardiography parameter improvement. After six months of therapy there was no more pericardial effusion and the left ventricular thickness and left atrial size were back to normal values as well as the diastolic function that was noted to improve earlier during management (Table 1).
Figure 1:
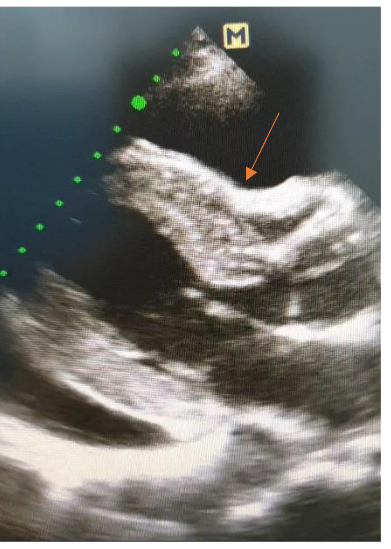
Trans-thoracic echocardiography still image showing pericardial effusion, right ventricular collapse and increased left ventricular walls thickness
Figure 2:
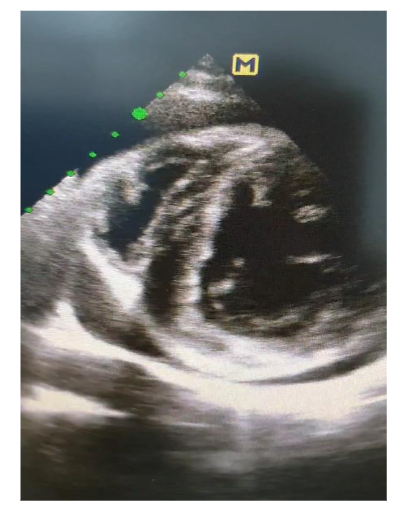
https://youtu.be/CWeoJF4BWu8
Trans-thoracic echocardiography video showing cardiac tamponade, reduced stroke volume, dilated left atrium and increased wall thickness
Table 1: Timeline table of the cardiac measurements and function with the change of TSH levels
| Time | TSH | Septal LV wall thickness at end diastole | Posterior LV wall thickness at end diastole | PE size | Diastolic function | LA size | Clinical HF |
|---|---|---|---|---|---|---|---|
| Day 1 | 59 | 33mm | 18mm | 6.5 cm | Grade 2 | 5 cm | YES |
| Day 60 | 15 | 22mm | 15mm | 6 cm | Grade 1 | 4.7 cm | YES |
| Day 90 | 2.04 | 18mm | 12mm | 4.5 cm | Grade 1 | 4.3 cm | No |
| Day 180 | 2 | 12mm | 10mm | <1 cm | Grade 1 | 2.7 cm | No |
Discussion
Hypothyroidism is a clinical syndrome resulting from thyroid hormones deficiency, which in turn results in slowing down of the metabolism, causing multi-organ dysfunction. Hypothyroidism affects 2% of adult women and 0.1-0.2% of adult men. Thyroid hormones have direct cellular functions on almost all tissues of the body⁵.
EFFECTS OF HYPOTHYROIDISM ON THE CARDIOVASCULAR SYSTEM:
Thyroid Hormone Actions at a cellular level:
T4 and T3 are synthesized by the thyroid gland in response to TSH. The thyroid gland primarily secretes T4 (85%), which is converted to T3 by 5-monodeiodin in the liver, kidney, and skeletal muscles⁶. The heart relies mainly on serum T3 and not T4, because it lacks myocyte intracellular deiodinase activity. T3 acts through binding to thyroid hormone nuclear receptors (TR). These receptor proteins mediate the induction of transcription by binding to thyroid hormone response elements (TRE). TR receptors induce transcription, and in the absence of T3 they repress transcription⁶. Negatively regulated cardiac genes such as myosin heavy chain and phospholamban are induced in the absence of T3 and inhibited in the presence of T3⁷. The myosin heavy chain genes encode the 2 contractile protein
isoforms of the thick filament in the cardiac myocyte. The sarcoplasmic reticulum Ca2-ATPase and its inhibitor, phospholamban, regulate intracellular calcium cycling. They are largely in charge of the cardiac contractile function and diastolic relaxation⁸.
The-adrenergic receptors and sodium potassium ATPase are also under T3 regulation. Thyroid hormone also has extranuclear nongenomic effects on the cardiac myocyte and on the systemic vessels. These effects can occur rapidly and do not need TRE-mediated transcription⁹.
Thyroid hormones affect the renin-angiotensin-aldosterone system. Renin is synthesized in the liver in response to T3. Thus, in a hypothyroidism, diastolic blood pressure increases, pulse pressure narrows, and renin levels decrease. This results in a sodium sensitive diastolic hypertension¹⁰.
Thyroid hormones affect endothelial functions through thyroid hormone receptor (THR)-α1 and THR-β. Activation of THR-β induces angiogenesis by initiating the protein kinase pathway. Activation of THR-α1 increases coronary and pulmonary blood flow and decreases resistance in mice models. The effect is probably due to the increase in the production of nitric oxide in endothelial and vascular smooth muscle cells¹⁰.
Inflammatory markers, like C-reactive protein (CRP), tumor necrosis factor alpha (TNF-α), interleukin 6 (IL6) and pro-fibrotic transforming growth factor beta 1 (TGF-β1), were elevated in hypothyroid rats. Cardiac stress markers, brain natriuretic peptide (BNP) and cardiac troponin T (Tn T)¹¹ were increased as well.
Expressions of cardiac remodeling genes and the development of fibrosis were induced in hypothyroid rats and led to a decline in cardiac function with chamber dilation seen in echocardiography¹¹. Rapid correction of hypothyroidism improved cardiac function and decreased cardiac remodeling markers. However, this further increased the levels of inflammatory and fibrotic markers in the plasma and heart and led to myocardial cellular infiltration and injury¹¹.
2. Atherosclerosis:
Atherosclerosis and coronary artery disease, are the result of dyslipidemia and diastolic hypertension¹². This explains that hypothyroid patients also have increased risk of stroke¹³. The cardiovascular changes in hypothyroidism namely the blood pressure, lipid profile, cardiac contractility, and increased SVR are caused by decreased thyroid hormone action on the heart, liver, and peripheral vasculature. They are reversible after thyroid hormone replacement¹⁴. Treatment with levothyroxine in those with overt thyroid dysfunction has been shown to improve LDL cholesterol, total cholesterol, triglycerides, hypertension, diastolic dysfunction, heart rate, and to delay progression of atherosclerosis¹⁵.
3. Left ventricular increased thickness and or hypertrophy:
Cardiac hypertrophy is reported in both humans and experimental animals diagnosed with hyperthyroidism¹⁶. The reason is primarily due to the increased cardiac performance to cope for the hemodynamic load caused by hyperdynamic circulation seen in hyperthyroidism¹⁵. However, in hypothyroidism the dynamic situation is the opposite. The heart beats slower, but against an increased vascular and pulmonary resistance. Thus, the presence of increased myocardial thickness needs to have a different and or an additional etiology and composition than just myocardial hypertrophy. Both increased interventricular septum (IVS) thickness⁵, and increased left ventricular posterior wall (LVPW) thickness¹⁷ were reported. Cianciulli et al, reported a case in 2021 of hypothyroidism leading to septal asymmetric hypertrophy and a “cherry on the top” strain pattern similar to the pattern in hypertrophic cardiomyopathy and cardiac amyloid. However, unlike them, it recovered after thyroid hormone replacement therapy¹⁸. Autopsy findings in another study, found acid mucopolysaccharide deposition and resulting edema in the myocardium of a hypothyroid patient after sudden cardiac death¹⁹.
4. Cardiac Magnetic Resonance in hypothyroidism and new promising evidence:
Recent evidence showed that native T1 mapping (MOLLI sequence without contrast administration) in CMR has a significantly increased values in the myocardium of hypothyroid patients. This suggests the presence of edema and diffuse fibrosis. These effects were not seen using T2 or late gadolinium enhancement (LGE) (Figure 3). It is a non-invasive way to prove the consistency and explain the diastolic and systolic dysfunction linked to hypothyroidism. They also found significantly reduced peak filling rate (PFR) and prolonged peak filling time (PFT) in hypothyroid patients as well as reduced stroke volume and cardiac index²⁰.
Figure 3:
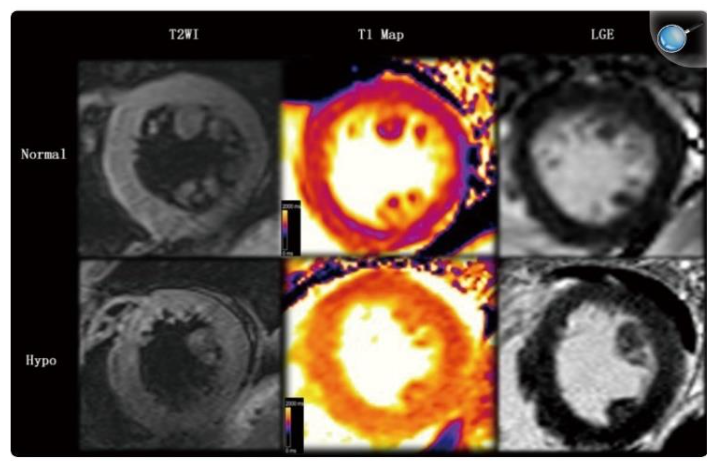
Borrowed from Gao et al. (2016)²⁰
Cardiac MRI comparing a patient with hypothyroidism (bottom row) and a control patient (upper row). T1 signals (middle column) unlike T2 and LGE showed increased values in the hypothyroid patient. T1 time in the hypothyroid patient is 1301ms (orange range of the color scale) while in the control it is 1040ms (purple range of the color scale)
5. Arrhythmia:
Prolongation of the QT interval in hypothyroid patients is well documented in several studies. This predisposes the patients to ventricular irritability that may rarely proceed to a reversible, Torsade de Pointes¹⁵.
6. Pericardial effusion:
Severe hypothyroidism can cause pericardial and pleural effusion. This can be small or huge. It usually accumulates slowly and rarely causes cardiac tamponade. Though the mechanism is unclear, increased capillary permeability and reduced lymphatic drainage from the pleural and pericardial space have been suggested¹⁰. Most of the pericardial and pleural changes and effusion secondary to hypothyroidism are reversible with thyroxin replacement. The effusion is well known to be protein rich¹⁵.
7. Diastolic dysfunction:
Diastolic dysfunction in some studies were found in 27% of patients⁵, while systolic dysfunction is seen in 7% of patients²¹. The decreased cardiac contractility associated with hypothyroidism results from the reduced expression of the sarcoplasmic reticulum Ca2-ATPase, and the increased expression of its inhibitor, phospholamban²² ²³. In hypothyroidism, these two proteins function in intracellular calcium cycling and thus regulate the diastolic function, particularly slowing the isovolumic relaxation phase of diastolic function. Isovolumic relaxation time increases proportionately to the severity of
hypothyroidism. It is not affected by pulse rate or blood pressure and is mainly affected by the thyroid hormone¹⁵. Values can range between the normal 50–90 ms and up to 150 ms.
8. Dyslipidaemia:
90% of patients with hypothyroidism had hypercholesterolemia. High total, LDL, VLDL and triglycerides and low HDL are all noted in hypothyroidism, with elevated triglycerides being in almost all patients⁵. The hypercholesterolemia in hypothyroidism results from reduced catabolism of lipoproteins, and decreased lipoprotein receptors and activity in the liver²⁴. A hypothyroid state results in decreased hepatic LDL receptors and reduced activity of the enzyme cholesterol-α-monooxygenase, resulting in decreased LDL clearance. Also noted are elevations in both C-reactive protein and homocysteine¹⁰.
Females with subclinical and overt hypothyroidism had significantly higher total and LDL cholesterol. Females with total cholesterol greater than 7 mmol/L had about a 7 times higher risk of hypothyroidism²⁵. The catabolism of cholesterol into bile is mediated by the enzyme cholesterol 7-hydroxylase¹⁵. This liver enzyme is negatively regulated by T3 and may add to the decreased catabolism and increased serum cholesterol seen in hypothyroidism²⁶. The increased lipid levels in subclinical hypothyroidism as well as in overt disease are potentially associated with increased cardiovascular risk²⁷. Treatment with thyroid hormone replacement reverses the risk²⁶.
9. Electrocardiogram:
ECG mainly shows bradycardia, low voltage and electrical alternans especially in the presence of pericardial effusion⁵.
10. Echocardiography:
Echocardiography can be normal, with increased LV mass, pericardial effusion, and systolic or diastolic dysfunction⁵.
11. Hypothyroidism and Heart Failure:
Review of multiple cross-sectional studies showed that 30% of patients with congestive cardiac failure have low T3 levels. The decrease in serum T3 is proportional to the severity of the heart disease²⁸ ²⁹. Low T3 syndrome is defined as a low serum T3 together with normal serum T4 and TSH. The syndrome results from defected hepatic conversion of T4 to T3, by 5-monodeiodination. The cardiac myocyte has no deiodinase activity and relies on the plasma T3¹⁵. Heart failure is found to be associated with iodine induced hypothyroidism diagnosed within 60 days of exposure to iodine containing contrast media³⁰. Females were affected more than males.
There are several possible mechanisms accounting for the adverse prognosis of hypothyroidism on HF patients. The link between hypothyroidism and pulmonary hypertension is well reported³¹ ³², which accordingly increases mortality of HF patients³³. Hypothyroidism can significantly decrease preload, increase afterload, and thus reduce stroke volume and cardiac output³⁴. Anemia secondary to hypothyroidism can lead to reduced exercise tolerance and worsening HF. Hypothyroidism can also lead to altered lipid metabolism, elevated C-reactive protein, and increased prevalence of aortic atherosclerosis, which can all increase the prevalence of myocardial infarction and mortality in HF patients³⁵.
Both subclinical hypothyroidism and subclinical hyperthyroidism are associated with adverse prognosis in HF patients. Subclinical hypothyroidism can increase the risk of both all-cause mortality and cardiac death and/or hospitalization in HF patients³⁵.
Hypothyroidism caused severe, progressive systolic dysfunction and increased chamber diameter/wall thickness ratio in rats. It also resulted in impaired myocardial blood flow and dramatic loss of arterioles. This highlights two important mechanisms by which hypothyroidism may lead to heart failure³⁶.
12. Hypertension:
In hypothyroidism, endothelial dysfunction and impaired vasomotor relaxation led to increased SVR³⁷. These effects caused diastolic hypertension in 30% of patients, and replacement therapy restored endothelial vasorelaxation and thus blood pressure to normal in most patients³⁸.
13. Heart Block:
Hypothyroidism is well-known to affect the chronotropic property of the cardiac conducting system through functional mechanisms. Cases of bradycardia and all degrees of atrioventricular (AV) block were reported with hypothyroidism. However, the response to treatment with thyroxin replacement have variable results. Few cases did recover whether due to primary hypothyroidism³⁹, hypothyroidism secondary to amiodarone treatment⁴⁰, or subclinical hypothyroidism⁴¹. A theory of hypothyroid induced oedema with recovery after thyroid hormone replacements was one of the explanations in patients with myxoedema⁴². However, most of the cases continued to have AV block and needed permanent pacemakers (PPM) despite hormone replacement and normalization of the lab results⁴³. These facts and data are against the general understanding that the condition is entirely reversible by treatment. It highlights the possibility that there is permanent damage caused by the lack of thyroxin, or that the heart block is not related to thyroxin deficiency from the start, especially that the incidence of heart block in hypothyroid patients is closer to that in the general population⁴³.
14. Amiodarone:
Amiodarone contains about 37% iodine by weight, and a 200-mg dose exposes the patient to about 300 times the recommended daily allowance. Because of its high iodine content, amiodarone can cause either hypothyroidism (5% to 25% of treated patients) or hyperthyroidism (2% to 10% of treated patients)¹⁰. Amiodarone also inhibits the conversion of T4 to T3 by inhibition of 5-deiodinase activity, leading to amiodarone-induced hypothyroidism. The risk increases in the presence of preexisting thyroid disease⁴⁴ ⁴⁵.
In general, patients treated with amiodarone should have thyroid function testing (specifically TSH) throughout therapy¹⁵. If hypothyroidism developed with a persistent rise in TSH, the patient should have thyroxine replacement⁴⁶. Thyroxin replacement does not impair the antiarrhythmic effect, as the antiarrhythmic effects is independent of the effect of Amiodarone on thyroid hormone metabolism⁴⁷.
15. Anemia:
Serum erythropoietin levels are low in hypothyroidism and it explain the normochromic, normocytic anemia found in almost 35% of those patients¹⁵.
16. Glucose metabolism:
Hypothyroidism is associated with decreased insulin sensitivity due to downregulation of glucose transporters and direct effects on insulin secretion and clearance¹⁰.
17. Hemodynamics:
Thyroid hormones have a variety of effects on the cardiovascular system that can greatly impact cardiac function. Bradycardia and smaller stroke volume are together the reason for reduced cardiac output while diminished heart sounds and cardiomegaly are mainly the result of pericardial effusion⁵. In hypothyroidism, cardiac output may decrease by 30–50%¹⁵. The arterial compliance is reduced, which leads to increased SVR. Impaired endothelium-dependent vasodilatation due to reduction in nitric oxide is seen in subclinical hypothyroidism as well¹⁵. Hypothyroidism is often associated with high diastolic blood pressure, and because the cardiac output is low, the pulse pressure is narrowed. The increase in diastolic pressure occurs with low serum renin levels and is sodium sensitive¹⁵.
18. Subclinical Hypothyroidism:
Subclinical hypothyroidism (SCH) is defined as a serum TSH concentration above the upper limit of the reference range (0.45–4.49 mIU/L) and serum free thyroxine (FT4) concentration within its reference range. This means TSH (4.5 to 6.9–19.9 mIU/L) and normal T4⁴⁸.
Several studies found that subclinical hypothyroidism is associated with an increased incidence and mortality of coronary heart disease⁴⁹, systolic and diastolic cardiac dysfunction, especially with TSH
levels greater than 10 mIU/L⁵⁰ ⁵¹. Atherosclerosis and myocardial infarction risk are increased in women with subclinical hypothyroidism⁵². Subclinical hypothyroidism is associated with an increased risk of congestive heart failure in older patients with a TSH level of 7.0 mIU/L or more⁵³. A recent study confirms the cardiovascular benefits of therapy⁵⁴. In a study of women with subclinical hypothyroidism, 18 months of treatment resulted in normalization of systolic and diastolic blood pressure and of total and LDL cholesterol as well as decreased carotid intima thickness⁵⁵.
19. Carotid Intimal Medial Thickness:
Carotid intima-media thickness (C-IMT) is a generally agreed on measure of subclinical atherosclerotic. It is used to assess the efficacy of diseases and interventions that increase or reduce atherosclerosis⁵⁶. C-IMT is a parameter of prophylaxis of cardiovascular disease in many guidelines. Progression of atherosclerosis is diagnosed when the value of C-IMT is over the threshold of 0.9 mm. The prospective risk of myocardial infarction increases from 10% to 15%, and stroke risk from 13% to 15% with a 0.1 mm increase in C-IMT⁵⁷.
A meta-analysis showed that high blood pressure is an independent risk factor for C-IMT⁵⁸. A second one found that SCH is a cause of higher systolic and diastolic blood pressures⁵⁹. Female sex, advanced age, and high cholesterol levels sub analysis significantly influenced the association between SCH and C-IMT in a third meta-analysis⁶⁰. Interestingly, low lymphocyte-monocyte ratio, which is an inflammatory marker, is associated with increased hsCRP and C-IMT in subclinical hypothyroidism patients⁶¹.
L-thyroxine replacement for one year led to decreased dyslipidemia and improved subclinical carotid atherosclerosis⁶². L-T4 treatment of SCH patients can reduce C-IMT, possibly due to the reduction in all lipid profile parameters, systolic and diastolic blood pressure, lipoprotein(a), and flow-mediated dilatation. This effect was observed in meta-analysis after long-term (>6 months) treatment⁶³.
20. The Skin:
Hypothyroidism is more prominent in females, and skin changes are seen more in hypothyroid females as well. Hypothyroidism can affect the skin in three different ways. Direct skin changes due to lack of normal levels of thyroid hormones, auto-immune skin changes associated with auto-immune hypothyroidism, and skin manifestations of other systems or diseases caused by hypothyroidism.
Xerosis and hair loss are the most common skin manifestations⁶⁴, followed by puffy oedema of the face, hands, and feet, seen more in the younger population². Direct thyroid hormone effects on the skin are carried through thyroid receptors (TR)⁶⁵. These TR are distributed in almost every component of the skin, including hair follicles, muscle cells, Schwann cells, sebaceous gland cells, the epidermal keratocytes, skin fibroblasts and vascular endothelial cells⁶⁶.
Facial puffiness, periorbital oedema, and non-pitting oedema of hands and feet are due to tissue infiltration with muco-polysaccharides mainly in the papillary dermis around the vessels as well as increase in tissue sodium concentration and water retention⁶⁷. The carotenemia is caused by carotene deposition in the dermis, this is more seen in the palms, feet, and nasolabial folds as a yellow tinge. The skin manifestation on the epidermis in hypothyroidism are dry palms and soles, and rough scaly skin that is seen histologically as thinning and hyperkeratosis⁶⁶. Cold skin is caused by reduced cutaneous blood flow confirmed by nail fold capillaroscopy and laser doppler⁶⁸ ⁶⁹. The capillary constriction is a reflex to reduction in the body temperature.
Thyroid dermopathy or the formally called pre-tibial myxoedema are raised waxy yellow brown lesions. It is due to infiltration of hyaluronic acid. The skin is non-pitting and treatment is with local steroids. Hyaluronic acid is the main glycosaminoglycan that is deposited in the dermis of hypothyroid patients with myxoedema⁷⁰. It can swell to one thousand times its dry weight when hydrated. Mucin deposition involves the skin, tongue, myocardium, and kidneys. Increased transcapillary albumin loss and impaired lymphatic drainage leads to extravascular accumulation and increases the oedema⁷¹.
The skin manifestations of other systems affected by lack of thyroid hormones are, pallor, cold intolerance, entrapments syndromes, drooping of the upper eye lids, and purpura⁶⁶. Direct skin changes in the sweat gland leads to decreased sweating and dry skin. Changes to the hair and nails include dry brittle nails, alopecia, dry brittle coarse hair, and loss of hair in the lateral third of the eye brows.
Associated skin manifestations in autoimmune diseases include dermopathy, acropathy, pruritic, vitiligo, pernicious anaemia, eczema, bullous disorders, and others⁷². The effect of hypothyroidism before and after treatment was proved on skin biopsies to reveal changes within 4 weeks of management with thyroid hormone replacement⁶⁶.
Conclusion
Hypothyroidism whether overt or subclinical has adverse cardiovascular effects. One of these effects is increased tissue thickness. This is seen in the skin, the left ventricular walls, and the carotid intima. The possible pathophysiology of increased thickness is one or a combination of tissue oedema, hypertrophy and substance infiltration with further water and salt entrapment. Fortunately, all forms of increased thickness showed response to treatment with thyroid hormone replacement. Further studies with tissue biopsy of the myocardial walls are needed to confirm the tissue consistency.
Disclosures:
None
Funding:
None
Conflict of interest:
None
Data availability:
Data is available on request from the corresponding Author
Acknowledgement:
Yousif Mutaz Yousif, Conestoga College, Canada for his contribution in image editing
Author contribution:
Author 1:
The corresponding author is the primary physician of the patient. She managed and followed up the case. She wrote the manuscript for the case report and literature review.
Author 2:
Data collection, literature collection, analysis, and referencing.
Author 3:
Data collection, literature collection, and manuscript layout.
Author 4:
Data and literature collection
References
(1) Canaris GJ, Manowitz NR, Mayor G, et al. The Colorado thyroid disease prevalence study. Arch Intern Med. 2000; 160:526–530.
(2) Jamwal A, Sharma A, Rather PA. original article. Cutaneous Manifestations of Hypothyroidism: Prospective Hospital Based Clinical Study. J Adv Med Dent Scie 2013;1(2):5-12.
(3) Kahaly GJ, Dillmann WH. Thyroid hormone action in the heart. Endocrine Rev. 2005; 26:704–728.
(4) Biondi B, Palmieri EA, Lombardi G, et al. Effects of thyroid hormone on cardiac function: the relative importance of heart rate, loading conditions, and myocardial contractility in the regulation of cardiac performance in human hyperthyroidism. J Clin Endocrinol Metab. 2002; 87:968–974.
(5) Ramesh K, Nayak B, Balaji Prasad Nayak. A study of cardiovascular involvement in Hypothyroidism. IAIM. 2016;3(5):74-80. https://www.iaimjournal.com/storage/2016/05/iaim_2016_0305_12.pdf
(6) Brent GA, Epstein FH. The molecular basis of thyroid hormone action. N Engl J Med. 1994; 331(13):847-853.
doi: https://doi.org/10.1056/nejm199409293311306
(7) Danzi S, Dubon P, Klein I. Effect of serum T3 on the regulation of cardiac gene expression: role of histone acetylation. Am J Physiol Heart Circ Physiol. 2005; 289:1506-1511.
(8) Mintz G, Pizzarello R, Klein I. Enhanced left ventricular diastolic function in hyperthyroidism: noninvasive assessment and response to treatment. J Clin Endocrinol Metab. 1991; 73:146–150.
(9) Hiroi Y, Kim H-H, Ying H, et al. Rapid nongenomic actions of thyroid hormone. Proc Natl Acad Sci U SA. 2006; 103:14104–14109.
(10) Udovcic M, Pena RH, Patham B, et al. Hypothyroidism and the Heart. Methodist DeBakey Cardiovascular Journal. 2017;13(2):55-59. doi: https://doi.org/10.14797/mdcj-13-2-55
(11) Hajje G, Saliba Y, Itani T, et al. Hypothyroidism and its rapid correction alter cardiac remodeling. PLoS One. 2014 Oct 15;9(10): e109753. doi: 10.1 371/journal.pone.0109753. PMID: 25333636; PMCID: PMC4198123.
(12) Cappola AR, Ladenson PW. Hypothyroidism and atherosclerosis. J Clin Endocrinol Metab. 2003; 88:2438–2444.
(13) Qureshi AI, Suri FK, Nasar A, et al. Free thyroxine index and risk of stroke: results from the National Health and Nutrition Examination Survey Follow-up Study. Med Sci Monit. 2006; 12:CR501–CR506.
(14) Palmieri EA, Fazio S, Lombardi G, et al. Subclinical hypothyroidism and cardiovascular risk: a reason to treat. Treat Endocrinol. 2004; 3:233–244.
(15) Klein I, Danzi S. Thyroid Disease, and the Heart. Circulation. 2007;116(15):1725-1735.
doi: https://doi.org/10.1161/circulationaha.106.678326
(16) Dorr M, Wolff B, Robinson DM, et al. The association of thyroid function with cardiac mass and left ventricular hypertrophy. J Clin Endocrinol Metab. 2005; 90:673–677.
(17) Rawat B, Satyal A. An echocardiographic study of cardiac changes in hypothyroidism and the response to treatment. Kathmandu University Medical Journal, 2003; 2(7): 182-187.
(18) Cianciulli TF, Morita LA, Saccheri MC, et al. Hypothyroid cardiomyopathy: A reversible phenocopy of hypertrophic cardiomyopathy. Echocardiography. 2021 Sep;38(9):1673-1677. doi: 10.1111/echo.15 183. Epub 2021 Sep 12. PMID: 34510529.
(19) Pastor Quirante FA, Pastor-Pérez FJ, Manzano-Fernández S, et al. Un expected autopsy findings after sudden cardiac death: Cardiovascular myxoedema and endocardial fibroelastosis. International Journal of Cardiology. 2015; 182:281-283. doi: https://doi.org/10.1016/j.ijcard.2014.12.156
(20) Gao x, Liu M, Qu A, et al. Native Magnetic Resonance T1 mapping identifies Diffuse Myocardial Injury in Hypothyroidism. 2016, Mar. PLOS one. 10;11(3): e0151266. Doi: 10.1371/journal. pone. 0151266
(21) Forfar JC, Muir AL, Toff AD. Left ventricular function in hypothyroidism. Br Heart J., 1982; 48: 278-284.
(22) Klein I, Ojamaa K. Thyroid hormone and the cardiovascular system. N Engl J Med. 2001; 344: 501–509.
(23) Dillmann WH. Cellular action of thyroid hormone on the heart. Thyroid. 2002; 12:447–452.
(24) Scarbottolo L, Trezze E, Roma P et al. Experimental hypothyroidism modulates the expression of the low-density lipoprotein receptor by the liver. Atherosclerosis, 1986; 59:329–33.
(25) Mayer O, Šimon J, Filipovský J, et al. Hypothyroidism in coronary heart disease and its relation to selected risk factors. Vascular Health and Risk Management. 2006;2(4):499-506. doi: https://doi.org/10.2147/vhrm.2006.2.4.499
(26) Rush J, Danzi S, Klein I. Role of thyroid disease in the development of statin-induced myopathy. The Endocrinologist. 2006; 16:279–285.
(27) Hak AE, Pols HA, Visser TJ, et al. Subclinical hypothyroidism is an independent risk factor for atherosclerosis and myocardial infarction in elderly women: the Rotterdam Study. Ann Intern Med. 2000; 132:270–278.
(28) Schmidt-Ott UM, Ascheim DD. Thyroid hormone and heart failure. Curr Heart Fail Rep. 2006; 3:114–119.
(29) Pingitore A, Landi P, Taddei MC, et al. Triiodothyronine levels for risk stratification of patients with chronic heart failure. Am J Med. 2005; 118:132–136.
(30) Inoue K, Guo R, Lee ML, et al. Iodine- Induced Hypothyroidism and Long-Term Risks of Incident of Heart Failure. J Am Heart Assoc. 2023; 12: e030511. DOI: 10.1161/JAHA.123.030511
(31) Li JH, Safford RE, Aduen JF, et al. Pulmonary hypertension and thyroid disease. Chest. 2007; 132(3):793–7.
(32) Vakilian F, Attaran D, Shegofte M, et al. Assessment of thyroid function in idiopathic pulmonary hypertension. Res Cardiovasc Med. 2016; 5(2): e29361.
(33) Ghio S, Gavazzi A, Campana C, et al. Independent and additive prognostic value of right ventricular systolic function and pulmonary artery pressure in patients with chronic heart failure. J Am Coll Cardiol. 2001;37(1):183–8.
(34) Ripoli A, Pingitore A, Favilli B, et al. Does subclinical hypothyroidism affect cardiac pump performance? Evidence from a magnetic resonance imaging study. J Am Coll Cardiol. 2005;45(3):439–45.
(35) Yang G, Wang Y, Ma A, et al. Subclinical thyroid dysfunction is associated with adverse prognosis in heart failure patients with reduced ejection fraction. BMC Cardiovascular Disorders (2019). https://doi.org/10.1186/s12872-019-1055-x
(36) Tang YD, Kuzman JA, Said S, et al. Low thyroid function leads to cardiac atrophy with chamber dilatation, impaired myocardial blood flow, loss of arterioles, and severe systolic dysfunction. Circulation. 2005 Nov 15;112(20):3122-30. doi: 10.1161/CIRCULATIONAHA.105.572883. Epub 2005 Nov 7. PMID: 16275864.
(37) Napoli R, Biondi B, Guardasole V, et al. Impact of hyperthyroidism and its correction on vascular reactivity in humans. Circulation. 2001; 104:3076–3080.
(38) Taddei S, Caraccio N, Virdis A, et al. Impaired endothelium-dependent vasodilatation in subclinical hypothyroidism: beneficial effect of levo thyroxine therapy. J Clin Endocrinol Metab. 2003; 88:3731–3737.
(39) Schoenmakers N, de Graaff WE, Peters RH. Hypothyroidism as the cause of atrioventricular block in an elderly patient. Neth Heart J 2008; 16:57–9.
(40) Mangiardi L, Gaita F, Brun S, et al. Atrioventricular block complicating amiodarone-induced hypothyroidism in a patient with pre-excitation and rate-dependent bilateral bundle branch block. J Am Coll Cardiol 1986; 7:180–4.
(41) Nakayama Y, Ohno M, Yonemura S, et al. A case of transient 2:1 atrioventricular block, resolved by thyroxine supplementation for subclinical hypothyroidism. Pacing Clin Elec trophysiol 2006; 29:106–8.
(42) Singh JB, Starobin OE, Guerrant RL, et al. Reversible atrioventricular block in myxedema. Chest 1973; 63:582–5.
(43) Ozcan K.S, Osmonov D, Erdinler I, et al. Atrioventricular block in patients with thyroid dysfunction: Prognosis after treatment with hormone supplementation or antithyroid medication. Journal of Cardiology 60 (2012) 327–332
(44) Harjai KJ, Licata AA. Effects of amiodarone on thyroid function. Ann Intern Med. 2006; 126:63–73.
(45) Martino E, Bartalena L, Bogazzi F, et al. The effects of amiodarone on the thyroid. Endocr Rev. 2001; 22:240–254.
(46) Bogazzi F, Bartalena L, Tomisti L, et al. Glucocorticoid response in amiodarone induced thyrotoxicosis resulting from destructive thyroiditis is predicted by thyroid volume and serum free thyroid hormone concentrations. J Clin Endocrinol Metab. 2007; 92:556–562.
(47) Klein I, Ojamaa K. Thyroid hormone and the cardiovascular system. N Engl J Med. 2001; 344: 501–509.
(48) Surks MI, Ortiz E, Daniels GH, et al. Subclinical thyroid disease: scientific review and guidelines for diagnosis and management. JAMA. 2004; 291:228–238.
(49) Rodondi N, den Elzen WP, Bauer DC, et al. Subclinical hypothyroidism and the risk of coronary heart disease and mortality. JAMA. 2010; 304:1 365–1374.
(50) Rodondi N, Bauer DC, Cappola AR, et al. Subclinical thyroid dysfunction, cardiac function, and the risk of heart failure: the Cardiovascular Health Study. J Am Coll Cardiol. 2008; 52:1152–1159.
(51) Galli E, Pingitore A, Iervasi G. The role of thyroid hormone in the pathophysiology of heart failure: clinical evidence. Heart Fail Rev. 2010; 15: 155–169.
(52) Rodondi N, Aujesky D, Vittinghoff E, et al. Subclinical hypothyroidism, and the risk of coronary heart disease: a meta-analysis. Am J Med. 2006; 119:541–551.
(53) Rodondi N, Newman AB, Vittinghoff E, et al. Subclinical hypothyroidism and the risk of heart failure, other cardiovascular events, and death. Arch Intern Med. 2005 Nov 28;165(21):2460-6. doi: 10.1001/archinte.165.21.2460. PMID: 16314541.
(54) Razvi S, Ingoe L, Keeka G, et al. The beneficial effect of L-thyroxine on cardiovascular risk factors, endothe lial function and quality of life in subclinical hypothyroidism: ran domised, crossover trial. J Clin Endocrinol Metab. 2007; 92:1715–1723.
(55) Adrees M, Gibney J, El-Saeity N, et al. Effects of 18 months of L-T4 replacement in women with subclinical hypothyroidism. Clin Endocrinol (Oxf). 2009 Aug;71(2):298-303.
(56) O’Leary DH, Polak JF. Intima-media thickness: a tool for atherosclerosis imaging and event prediction. Am J Cardiol 2002; 90:18l–21
(57) Gao N, Zhang W, Zhang YZ, et al. Carotid intima-media thickness in patients with subclinical hypothyroidism: a meta-analysis. Atherosclerosis 2013; 227:18–25.
58) Wang JG, Staessen JA, Li Y, et al. Carotid intima-media thickness, and antihypertensive treatment: a meta-analysis of randomized controlled trials. Stroke 2006; 37:1933–40.
(59) Cai Y, Ren Y, Shi J. Blood pressure levels in patients with subclinical thyroid dysfunction: a meta-analysis of cross-sectional data. Hypertens Res 2011; 34:1098–105.
(60) Isailă, O.-M.; Stoian, V.E.; Fulga, I. et al. The Relationship between Subclinical Hypothyroidism and Carotid Intima-Media Thickness as a Potential Marker of Cardiovascular Risk: A Systematic Review and a Meta-Analysis. J. Cardiovasc. Dev. Dis. 2024, 11, 98. https://doi.org/10.3390/jcdd11040098
(61) Yurtdaş M, Asoğlu R, Özaydın A. Lymphocyte to monocyte ratio may predict increased carotid intima-media thickness in patients with subclinical hypothyroidism. Clin Hemorheol Microcirc. 2021; 77(2):133-142. doi: 10.3233/CH-200820. PMID: 33074218.
(62) del Busto-Mesa A, Cabrera-Rego JO, Carrero-Fernández L, et al. Changes in arterial stiffness, carotid intima-media thickness, and epicardial fat after L-thyroxine replacement therapy in hypothyroidism. Endocrinol Nutr. 2015 Jun-Jul;62 (6):270-6. English, Spanish. doi: 10.1016/j.endon u.2015.02.005. Epub 2015 Apr 14. PMID: 25882083.
(63) Zhao T, Chen B, Zhou Y, et al. Effect of levothyroxine on the progression of carotid intima-media thickness in subclinical hypothyroidism patients: a meta-analysis. BMJ Open. 2017 Oct 22;7(10): e016053. doi: 10.1136/bmjopen-2017-016053. PMID: 29061604; PMCID: PMC5665330.
(64) Dogra A, Dua A, Singh P. Thyroid, and skin. Indian J Dermatol 2006; 51: 96-99.
(65) Billoni N, Buan B, Gautier B, et al. Thyroid hormone receptor beta-1 is expressed in the human hair follicle. British Journal of Dermatology 2000; 142:645-52.
(66) Safer JD. Thyroid hormone action on skin. Dermato-endocrinology. 2011;3(3):211-215. doi: https://doi.org/10.4161/derm.3.3.17027
(67) Saadia Z, Alzolibani AA, Al Robaee A, et al. Cutaneous manifestations of hypothyroidism amongst gynecological consultations. Int J Health Sci (Qassim) 2010; 4:168 77.
(68) Weiss M, Milman B, Rosen B, et al. Quantitation of thyroid hormone effect on skin perfusion by laser doppler flowmetry. J Clin Endocrinol Metab 1993; 76:680.
(69) Pazos-Moura CC, Moura EG, Breitenbach MMD, et al. Nailfold capillaroscopy in hypothyroidism: blood flow velocity during rest and post occlusive reactive hyperemia. Angiology 1998; 49:471.
(70) Smith TJ, Bahn RS, Gorman CA. Connective tissue, glycosaminoglycans and diseases of the thyroid. Endocrine Rev 1989; 10:366-91.
(71) Parving HH, Hansen JM, Nielsen SL, et al. Mechanisms of edema formation in myxedema: increased protein extravasation and relatively slow lymphatic drainage. N Engl J Med 1979; 301:460.
(72) Ai J, Leonhardt JM, Heymann WR. Autoimmune thyroid diseases: Etiology, pathogenesis, and dermatologic manifestations. J Am Acad Dermatol 2003; 48:641-59.

