Taste Masking Challenges in Pediatric Pharmaceuticals
Taste Masking Challenge of 155 Active Pharmaceutical Ingredients
David A. Tisi, Jeffrey H. Worthington1
- Senopsys LLC, Woburn, MA 01801, USA
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Tisi, DA. and Worthington, JH., 2024. Taste Masking Challenge of 155 Active Pharmaceutical Ingredients. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5890
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5890
ISSN 2375-1924
Abstract
Regulations in the US and EU require and incentivize the development of palatable, age-appropriate medicines for children. However, the taste masking challenge of new drug actives is generally unknown, making development of palatable drug products extremely difficult. To develop palatable drug products, formulation scientists first need to determine if the drug active is bitter, has an offensive aroma (malodor) or is burning as each perception requires a different formulation approach. This retrospective compilation of results of 155 taste assessment studies reveals diversity in aversive flavor attributes of active ingredients. Bitterness was the primary taste masking challenge for 65% of the drug actives. Aversive aromas (e.g., solvent, fishy, oxidized oil) were the second most common challenge, impacting 8% of drug actives. Approximately 5% had trigeminal irritation, and a smaller subset were sour or salty (4%). None were found to be sweet. Of note, 14% of drug actives were “bland” in flavor, with no measured aversive attributes. Complicating development, most actives (>90%) were found to have multiple aversive flavor attributes. These findings highlight the need to determine the aversive attributes early in clinical development (Phase 1) to guide dosage form selection and formulation design.
Keywords: Active pharmaceutical ingredient, sensory analysis, taste, flavor, taste masking, aversive flavor attribute, flavor profile
Introduction
Flavor is a combination of taste, aroma, mouthfeel and texture. Many Active Pharmaceutical Ingredients (APIs) have an aversive flavor element that can lead to dose rejection or poor compliance, particularly in pediatric populations. Tablets and capsules are not suitable for children who cannot swallow them or adults with dysphagia and therefore other forms are required. However, many alternative pediatric dosage forms (e.g., liquid, multipartculate, orally disintegrating tablet) have comparatively long oral residence times that increase the perception of aversive flavor attributes, frequently making them unacceptable. In addition to ensuring that medicines are safe and effective for children, the US Food and Drug Administration (FDA) and European Medicines Agency (EMA) require drug developers to address the creation of palatable, age-appropriate dosage forms as part of required Pediatric Study Plan and Pediatric Investigation Plan, respectively.
The American Association of Pharmaceutical Scientists (AAPS) Pediatric Formulations Task Force published a report on pediatric medicines that advanced a decision framework for guiding the development of palatable pediatric dosage forms. The recommended first step is to identify and quantify the aversive flavor attributes of the API to assess the taste masking challenge and provide taste masking technology guidance, which includes five general approaches – signal interruption, flavor system, alternate API form, complexation, and encapsulation.
Aversive attributes are measured via sensory analysis, which is the scientific discipline used to “evoke, measure, analyze, and interpret those responses to products that are perceived by the senses of sight, smell, touch, taste, and hearing”. Sensory analysis entails the use of human subjects (in-vivo) to measure perceptions of flavor. In-vitro models (e.g., in-silico/e-tongue, cultured cell-based biosensors, and animal surrogates) are under investigation in pre-clinical research but in the absence of correlation to human data, they are of limited utility in predicting flavor issues or guiding the development of palatable formulations.
Sensory analysis methods are broadly divided into two broad classes – analytical and affective:
- Analytical methods are used to quantitatively measure the attributes of products and guide formulation development. Analytical sensory testing methods utilize trained assessors to identify the perceived attributes – taste, aroma, mouthfeel and texture – and rate the perceived intensity of each attribute using a measurement scale that is established with chemical reference standards.
- Affective methods measure human response to products and include measures of hedonics (liking) and preference and always use untrained respondents. Affective methods do not characterize or quantify flavor attributes and untrained subjects frequently confuse flavor attributes. Thus, these methods may be used as a surrogate measure of acceptability but cannot be effectively used to guide formulation development.
Published research literature of sensory data includes a single drug active or at best a small number of related actives. The data described herein presents a retrospective analysis of the flavor attributes of 155 drug actives and reveals the diversity, complexity and difficulty of developing palatable, age-appropriate drug products.
Materials and Methods
The flavor attributes of 155 APIs were measured in separate programs to identify and quantify the taste masking challenge, inform dosage form selection and guide development of palatable formulations. For most programs the objective was to develop a palatable, age-appropriate, easy-to-swallow dosage form as an alternative to solid oral tablets or capsules. In a few cases, a high dose loading, or the physio-chemical properties of the API necessitated a dosage form other than a traditional oral solid. A list of the intended dosage forms is shown in Table 1.
| Intended Dosage Forms |
|---|
| 3d Printed Tablets |
| Chewable Tablets |
| Dry Syrups |
| Films |
| Gummies |
| Nasal Sprays |
| Nebulized Liquids |
| Minitablets |
| Multipartculates |
| Oro-Adhesive Tablets |
| Orally Disintegrating Tablets |
| Solutions |
| Suspensions |
Target indications for the 155 APIs represented a wide range of common and rare (orphan) diseases and disorders afflicting both children and adults (Table 2). Most APIs were investigational compounds and a few represented new dosage forms of approved prescription medicines – 505(b)(2) applications. Monographed over-the-counter actives and dietary supplements were not included. The specific APIs cannot be disclosed due to confidentiality.
| Target Indications |
|---|
| Autoimmune Disease |
| Rheumatoid Arthritis |
| Alopecia Areata |
| Psoriasis |
| Eosinophilic Esophagitis |
| Cancers |
| Myeloid Leukemia |
| Non-Hodgkin Lymphoma |
| Multiple Myeloma |
| Myelofibrosis |
| Metastatic Breast Cancer |
| Neuroblastoma |
| Lung Cancer |
| Endometrial Cancer |
| Cardiovascular |
| Hypertension |
| Gastrointestinal Disorders |
| Irritable Bowel Syndrome |
| Crohn’s Disease |
| Ulcerative Colitis |
| Gastroesophageal Reflux |
| Genetic Disorders |
| Cystic Fibrosis |
| Rett Syndrome |
| Celiac Disease |
| Pyruvate Kinase Deficiency |
| Sickle Cell Disease |
| Hereditary Angioedema |
| Infectious Diseases |
| HIV |
| Hepatitis B/C |
| Cytomegalovirus |
| Respiratory Syncytial Virus |
| Enterovirus |
| Coronavirus |
| Mental Health Conditions |
| Psychosis |
| Schizophrenia |
| Major Depressive Disorder |
| Bipolar Disorder |
| Panic Disorder |
| Substance Abuse Disorder |
| Neurological Disorders |
| Migraine |
| Epilepsy |
| Multiple Sclerosis |
| Amyotrophic Lateral Sclerosis |
| Attention Deficit Hyperactivity Disorder |
| Respiratory |
| Asthma |
| COPD |
| Allergic Rhinitis |
| Sleep Disorders |
| Narcolepsy |
| Insomnia |
| Other |
| Chronic Kidney Disease |
| Anemia |
| Hyperkalemia |
| Precocious Puberty |
| Lipid Disorder |
| Sexual Dysfunction |
Flavor is a concentration effect – 5mL of a liquid drug product tastes the same as 10mL. Accordingly, flavor attributes were measured at the highest expected clinical strength for the intended patient population to represent the worst-case taste masking challenge. Additional clinical strengths were included when known. Alternatively, a descending concentration series was evaluated to determine the maximum palatable strength. Drug actives were assessed as aqueous preparations or dry powders depending on the intended dosage form. Those intended for dosing as liquids (solutions, suspension, or in liquid vehicles) were assessed as aqueous solutions or suspensions. Actives intended for solid dosing (ODT’s, dry syrups, films, and gummies) were assessed as dry powders.
Samples were evaluated using the Flavor Profile Method of sensory analysis, an internationally recognized and ISO/ASTM approved open-source method. Flavor Profile uses highly trained adult assessors (“panelists”) to identify the flavor attributes in the order in which they are perceived and to measure the intensity of each attribute. Separate measurements are made for the initial flavor (T=0) and at specified time intervals in the aftertaste, typically 1, 3, 5, 10, 15, 20, 25, and 30 minutes.
The measured attributes include all oral flavor attributes – taste, aroma, mouthfeel, and texture.
“Taste” refers to those sensations perceived through the stimulation of the receptor cells located in the taste buds on the epithelium of the tongue and oral cavity. Five distinct tastes, known as basic tastes, are perceived in the oral cavity – sweet, sour, salty, bitter and umami (savory). Bitterness or intense sour and salty tastes are known to be aversive.
“Aroma” is the perception of volatile chemical compounds perceived via the sense of smell (olfaction) through stimulation of receptor cells in the olfactory epithelium located in the upper reaches of the nasal cavity. Examples of aversive aromas include solvent, oxidized oil, fishy, sulfurous and fecal.
“Mouthfeels” are sensations that arise when chemical compounds directly stimulate free nerve endings in the trigeminal (Vth cranial) nerve. Examples of aversive mouthfeels are numbing (produced by compounds such as clove oil or parabens) and bite/burn (produced by compounds such as pepper or ethanol).
“Textures” are tactile characteristics perceived in the oral cavity when a product is deformed through mastication – chewed, swished, rolled, agitated, or swallowed. Notable aversive textural attributes include grittiness and mouthcoating.
The perceived intensity of each attribute is measured on the 7-point Flavor Profile scale that ranges from none (0) to strong (3) and is established with chemical reference standards. Attributes at a slight (1) intensity are discernable to untrained subjects (consumers and patients) and this intensity is known as the recognition threshold. Therefore, the goal for palatability is for aversive attributes (e.g., bitterness or irritation) to be below this threshold. The Flavor Profile scale and definitions are as shown in
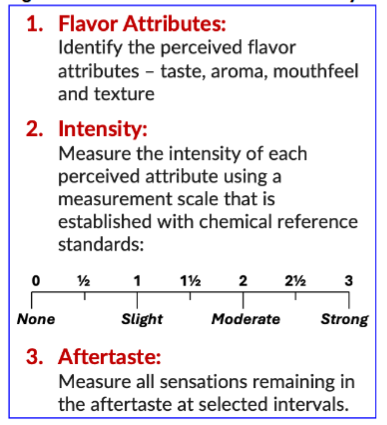
.
All studies were conducted following ICH Good Clinical Practice. Up to ten panelists were enrolled in each study. Taste assessment samples were compounded at the clinical site or supplied by sponsor as cGMP drug products.
Results
The primary aversive flavor attribute of the 155 APIs is shown in
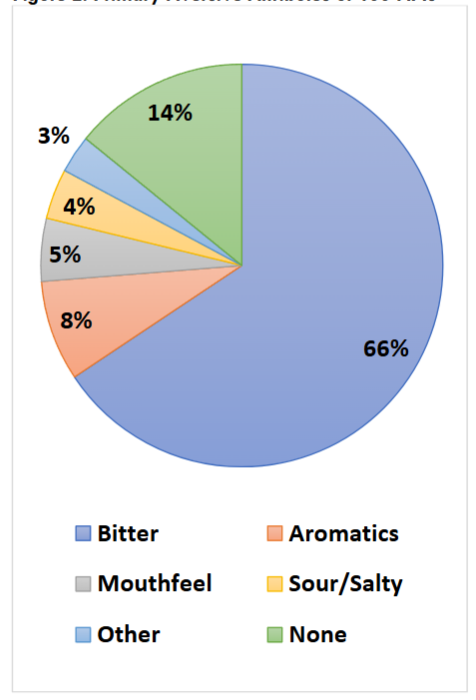
. Bitterness was the most common primary taste masking challenge, representing 65% of the APIs. Aversive aromas were the next most common challenge, impacting 8% of APIs. Some descriptors of these malodors included “phenolic” (like Band-Aids), “sulfidy” (overcooked cruciferous vegetables), “oxidized oil” (like old paint), and “solventy” (e.g., acetone, or ether-like). Approximately 5% produced mouthfeels such as trigeminal irritation (e.g., burning), and a smaller subset were sour or salty (4%). None were found to be sweet. A full 14% of actives were “bland” in flavor, with no measured aversive attributes.
Most actives were characterized by multiple aversive attributes, and the secondary challenges are shown in
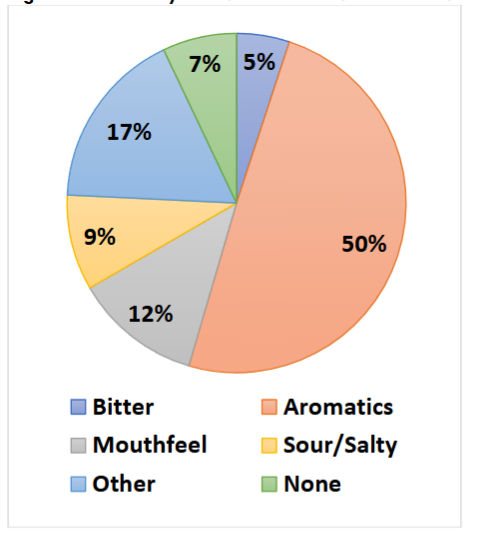
.
Approximately half of the APIs had a secondary aversive aroma, with a complete listing in Table 3 below.
| Aversive Aroma Descriptors |
|---|
| Acetate |
| Acetone |
| Burnt Polyethylene |
| Butyric Acid |
| Candle Wax |
| Ethanol |
| Ether-like |
| Fecal |
| Fishy Amine |
| Geosmin (Moldy) |
| Heptane |
| Iso-Propyl Alcohol |
| Musty |
| Oxidized Oil |
| Peroxide |
| Petroleum Ether |
| Phenolic |
| Sulfidy |
| Wet Cardboard |
| Wet Paper |
The time duration that an aversive attribute is perceptible into the aftertaste is a useful measurement of the overall intensity of the flavor of a drug active. In
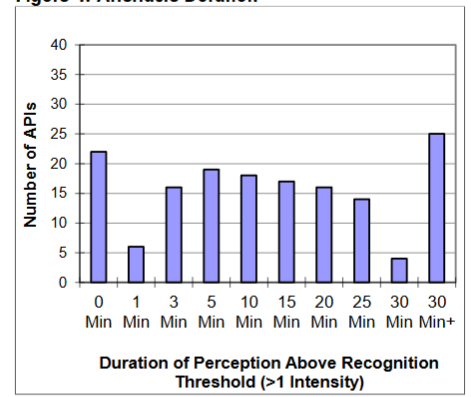
, the length of time that the primary aversive attribute is present above the recognition threshold (> 1) is plotted as a histogram for the 155 APIs across all categories (e.g. bitterness, aromatic, or mouthfeel).
The time intensity data is broken down by category in
, where the mean time-intensity profiles are plotted for APIs with a primary bitter, aromatic, mouthfeel or sour challenge. In this figure, the background shading represents the intensity above the recognition threshold (1-intensity). Amongst the common aversive attributes, bitterness lingered longest in the aftertaste, with average bitterness only declining to the recognition threshold by 30 minutes in the aftertaste. Aversive aromas and sourness had on average much shorter aftertaste profiles, dropping below a 1-intensity by 3 minutes in the aftertaste. The average mouthfeel profile was in between, with the mean profile reaching a 1-intensity by 15 minutes in the aftertaste.
Discussion
Identifying the aversive flavor attributes of a drug active is a necessary first step in developing a palatable drug product. The four components of flavor – taste, aroma, mouthfeel and texture – each have separate and distinct perception pathways that are mitigated by fundamentally different formulation approaches. For example, bitterness can be reduced via the complementary tastes of sweet, sour and salty whereas aversive aromas are reduced by blending with more patient-acceptable ones. Importantly, flavor excipients added to mask a target aversive attribute need to persist in the aftertaste as long as the attribute it is trying to cover. Accordingly, strongly bitter APIs with a short aftertaste (like caffeine in coffee or quinine in tonic water) have a lower taste masking challenge than those that linger for 5, 10, 30 minutes or longer. This time-dependent difficulty stems from the limited effectiveness of potential taste masking approaches as most flavor excipients do not linger at perceptible intensities longer than a few minutes.
For the 155 APIs, the duration of the aversive flavor attributes in the aftertaste can be used to categorize the overall difficulty of the taste masking challenge as shown in
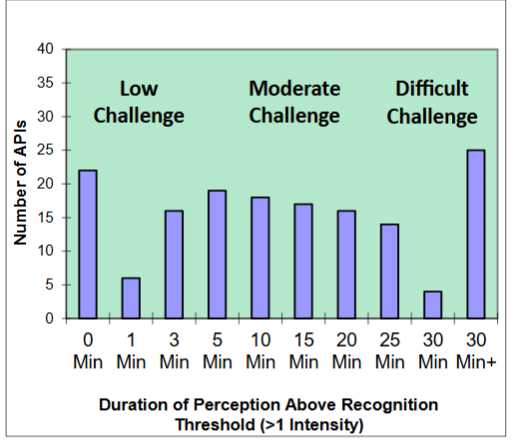
.
The APIs that linger between 0 and 3 minutes (cumulatively 44 APIs) represent a low taste masking challenge. Those that linger between 5 and 25 minutes (cumulatively 84 APIs) represent a moderate challenge, and those that linger for 30 minutes or longer (29 APIs) represent a very difficult challenge.
As illustrated previously in

, the average API bitterness lingers for 30 minutes in the aftertaste, requiring a flavor masking system that includes complementary basic taste excipients (i.e., sweet, salty, sour) that also lingers for 30 minutes. By comparison, the average sour challenge only requires 3 minutes of coverage via blending with its complementary basic tastes (sweet, salty, and bitter). Excipients that impart basic tastes such as organic acids (sour), saccharides (sweet) and flavor potentiators like sodium chloride (salt) have temporal qualities (i.e., how long taste lingers into the aftertaste). This lingering in the aftertaste can be selected to match the aversive attribute of the API. In the realm of sweeteners, this often means that high intensity options with longer aftertaste duration are more useful for lingering APIs than bulk sweeteners such as sucrose with shorter persistence.
Aversive mouthfeels such as burning and numbing linger at a patient perceptible intensity for an average of 15 minutes and require masking over this time. Aversive mouthfeels may require application of an API sequestration technology as excipient approaches are of limited utility in this domain. Coating systems may introduce their own challenges (e.g. grittiness) that can be reduced through particle size reduction, if severe, or through viscosity systems, if moderate. Similarly, the mean aroma profile lingers above a recognition threshold (>1) for 5 minutes, which requires blending with identifying aromas (e.g., cherry, grape, or orange) that persist for at least 5 minutes in the aftertaste.
Conclusion
The Flavor Profile method of sensory analysis is a useful tool for identifying and quantifying the taste masking challenge of drug actives, informing dosage form selection and guiding development of palatable formulations. The taste masking challenge is a function of the modality (taste, aroma, mouthfeel, texture), intensity and duration of the aversive flavor attributes. This retrospective compilation of results of 155 taste assessment studies reveals diversity in the primary taste masking challenge and highlights that many drug actives have multiple aversive attributes – bitterness, aroma (malodor), mouthfeel (irritancy) and others, each of which need to be addressed to create a palatable drug product. The persistency or duration of aversive attributes in the aftertaste is also widely distributed and can linger above the recognition threshold for over 30 minutes, increasing the overall taste masking challenge. The resulting data can also be used to guide the selection of the most appropriate taste masking technology approach.
References
- European Medicines Agency Committee for Medicinal Products for Human use (CHMP) Formulations of choice for the paediatric population. Reference Number: EMEA/CHMP/PEG/194810/2005
- Zajicek, A.; Fossler, M.J.; Barrett, J.S.; Worthington, J.H.; Ternik, R.; Charkoftaki, G.; Lum, S.; Breitkreutz, J.; Baltezor, M.; Macheras, P.; et al. A report from the pediatric formulations task force: Perspectives on the state of child-friendly oral dosage forms. AAPS J. 2013, 15, 1072–1081.
- Thomas F. Avoiding Bitter Taste. Pharmaceutical Technology 11-02-2019, Volume 43, Issue 11 Pages: 26–29
- Stone, H., & Sidel, J. L. (2004). Sensory Evaluation Practices. Academic Press Inc., Tragon Corporation.
- Latha RS, Lakshmi PK. Electronic tongue: An analytical gustatory tool. J Adv Pharm Technol Res. 2012 Jan;3(1):3-8.
- Wang Z, Li J, Hong X, Han X, Liu B, Li X, Zhang H, Gao J, Liu N, Gao X, Zheng A. Taste Masking Study Based on an Electronic Tongue: The Formulation Design of 3D Printed Levetiracetam Instant-Dissolving Tablets. Pharm Res. 2021 May;38(5):831-842.
- Podrażka M, Bączyńska E, Kundys M, Jeleń PS, Witkowska Nery E. Electronic Tongue-A Tool for All Tastes? Biosensors (Basel). 2017 Dec 31;8(1):3. doi: 10.3390/bios8010003.
- Woertz K, Tissen C, Kleinebudde P, Breitkreutz J, Taste Sensing Systems (Electronic Tongues) for Pharmaceutical Applications, International Journal of Pharmaceutics, Volume 417, Issues 1–2, 2011, Pages 256-271
- Liu S, Zhu P, Tian Y, Chen Y, Liu Y, Wang M, Chen W, Du L, Wu C. A Taste Bud Organoid-Based Microelectrode Array Biosensor for Taste Sensing. Chemosensors. 2022; 10(6):208.
- Chen P, Wang B, Cheng G, Wang P. Taste receptor cell-based biosensor for taste specific recognition based on temporal firing. Biosens Bioelectron. 2009 Sep 15;25(1):228-33.
- Cocorocchio M, Ives R, Clapham D, Andrews PL, Williams RS. Bitter Tastant Responses in the Amoeba Dictyostelium Correlate with Rat and Human Taste Assays. ALTEX. 2016;33(3):225-36.
- Soto J et. al Rats can predict aversiveness of Active Pharmaceutical Ingredients, European Journal of Pharmaceutics and Biopharmaceutics, Volume 133, 2018, Pages 77-84.
- J. Bennett, A. Coburn, A. Coupe, D. Stedman, A. Taylor, A. Tyler, Development of a Zebrafish Model for Bitter Taste Assessment, International Journal of Pharmaceutics, Volume 536, Issue 2, 2018, Pages 521-522.
- Lawless, H. T., & Heymann, H. (2010). Sensory evaluation of food: Principles and practices. Chapman & Hall.
- M. O’Mahony, M. Goldenberg, J. Stedmon, J. Alford. Confusion in the use of the taste adjectives ‘sour’ and ‘bitter’ Chemical Senses, Volume 4, Issue 4, December 1979, Pages 301–318.
- Vikas et. al. Taste Assessment Trials for Sensory Analysis of Oral Pharmaceutical Products Pak. J. Pharm. Sci., Vol.21, No.4, October 2008, pp.438-450.
- Morris JB, Tisi DA, Tan DCT, Worthington JH. Development and Palatability Assessment of Norvir® (Ritonavir) 100 mg Powder for Pediatric Population. Int J Mol Sci. 2019 Apr 6;20(7):1718.
- Strickley RG. Pediatric Oral Formulations: An Updated Review of Commercially Available Pediatric Oral Formulations Since 2007. J Pharm Sci. 2019
- Keane, P. The Flavor Profile. ASTM Manual on Descriptive Analysis Testing for Sensory Evaluation; ASTM International: West Conshohocken, PA, USA, 1992; pp. 2–15
- Hort J et. al (Ed.). Time-Dependent Measures of Perception in Sensory Evaluation. West Sussex, UK: Wiley Blackwell.
- International Council for Harmonisation (ICH). 2016. Integrated Addendum to ICH E6(R1): Guideline for Good Clinical Practice E6 (R2).
- Green BG, Lim J, Osterhoff F, Blacher K, Nachtigal D. Taste Mixture Interactions: Suppression, Additivity, and the Predominance of Sweetness. Physiol Behav. 2010 Dec 2;101(5):731-7.
- Russell S.J Keast, Paul A.S Breslin, An Overview of Binary Taste–Taste Interactions, Food Quality and Preference, Volume 14, Issue 2, 2003, Pages 111-124.
- B. DeFer, Food ingredients—Sweetener Technical Overview and Allowable Daily Intake (Adapted), The NutraSweet Company, Chicago, 2010. 22.
- Drašković, M., Medarević, D., Aleksić, I., & Parojčić, J. (2016). In Vitro And In Vivo Investigation of Taste-Masking Effectiveness of Eudragit E PO as Drug Particle Coating Agent in Orally Disintegrating Tablets. Drug Development and Industrial Pharmacy, 43(5), 723–731.
- Szejtli, J., and L. Szente. Elimination Of Bitter, Disgusting Tastes of Drugs and Foods by Cyclodextrins European Journal of Pharmaceutics and Biopharmaceutics 61.3 (2005): 115-125.
- Sriwongjanya M, Bodmeier R, Effect of Ion Exchange Resins on the Drug Release from Matrix Tablets, European Journal of Pharmaceutics and Biopharmaceutics, Volume 46, Issue 3, 1998, Pages 321-327.
- Santagiuliana M, Broers L, et. al Strategies to Compensate for Undesired Gritty Sensations in Foods, Food Quality and Preference, Volume 81, 2020.
- Imai, E., Hatae, K. And Shimada, A. (1995), Oral Perception of Grittiness: Effect of Particle Size and Concentration of The Dispersed Particles and The Dispersion Medium. Journal Of Texture Studies, 26: 561-576.

