Post-Weaning Diarrhea: Scoring Systems for Treatment
Adapted faecal clinical scoring systems for post-weaning diarrhea due to enterotoxigenic Escherichia coli and swine dysentery due to Brachyspira hyodysenteriae
Frédéric Vangroenweghe 1,2 and James Mark Hammer 3
- Elanco Animal Health Benelux, BU Swine & Ruminants, 2018 Antwerpen, Belgium
- Unit of Porcine Health Management, Faculty of Veterinary Medicine, Department of Internal Medicine–Reproduction–Population; Medicine, Ghent University, 9820 Merelbeke, Belgium
- Elanco Animal Health US, BU Swine, Greenfield, IN 46140, USA
OPEN ACCESS
PUBLISHED: 30 October 2024
CITATION: Vangroenweghe, F. and Hammer, J.M., 2024. Adapted faecal clinical scoring systems for post-weaning diarrhoea due to enterotoxigenic Escherichia coli and swine dysentery due to Brachyspira hyodysenteriae. Medical Research Archives, [online] 12(10).
https://doi.org/10.18103/mra.v12i10.5875
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5875
ISSN 2375-1924
ABSTRACT
Swine production globally still has major challenges with swine intestinal disease, which might be provoked by post-weaning diarrhoea or growing pig diarrhoea. The major etiological agent of post-weaning diarrhea is enterotoxigenic Escherichia coli, characterized by the presence of both fimbriae (adhesins, mainly F4 (previously known as K88) and F18) and the presence of enterotoxins (thermostable toxin a and b; thermolabile toxin). Piglets affected with post-weaning diarrhea have mild to severe watery diarrhea, dehydration, and retarded growth which may lead to mortality. In contrast, growing pig diarrhea has, besides several viral and parasitic causes, three major bacterial causes, namely Lawsonia intracellularis, Brachyspira species or Salmonella species. Swine dysentery is provoked by Brachyspira hyodysenteriae and leads to mild to severe diarrhea which can be associated with addition of mucus, fibrin, necrotic material, or blood into the faeces. Ileitis caused by Lawsonia intracellularis causes mild to severe diarrhea without melena. Depending on the clinical presentation, low mortality with sudden death may be observed. In contrast, Salmonella is ubiquitous and clinical signs may be different depending on serotype or caused by dysbiosis. Both under field and experimental conditions, the assessment of severity of swine intestinal disease remains a challenge. Many studies only report presence or absence of diarrhea without further differentiation of specific severity ranging from mild to severe faecal appearance. Therefore, a need for standardized scoring to evaluate and analyze results of treatment and prevention is crucial. Based on three published field studies, we discuss the importance of a standardized faecal clinical scoring system adapted to a specific pathogenic challenge, namely enterotoxigenic Escherichia coli or Brachyspira hyodysenteriae. For enterotoxigenic Escherichia coli, a post-weaning diarrhea faecal clinical scoring based on a score grid from 0 to 4 is applied, to differentiate between normal, pasty, mild, moderate, and severe diarrhea. For Brachyspira hyodysenteriae, Lawsonia intracellularis and Salmonella species a more differentiated approach is necessary considering the different possible additions (mucus, blood, fibrin, necrotic material). The swine dysentery faecal clinical scoring system, therefore, includes 3 sub criteria phase-consistency, colour, and additions, which are scored separately and subsequently combined into a total faecal score. Using both scoring systems, several statistical analyses can be performed ranging from kinetics over time, area under the curve, days to maximum score, and in case of individual piglet faecal clinical scoring number of piglets with diarrhea on a particular observation day can be assessed. These analyses are used to compare different intervention strategies in 3 field experiments. They are vaccination against enterotoxigenic Escherichia coli using an oral live avirulent Escherichia coli F4/F18 vaccine in the prevention of post-weaning diarrhea due to enterotoxigenic Escherichia coli, inclusion of a β-mannanase enzyme to degrade β-mannans in the diet, and therapeutic treatment with an oral Zn-chelate product to treat clinical signs of swine dysentery. In conclusion, adapted faecal clinical scoring systems for different swine intestinal diseases or phases of production can be an important tool in both field and experimental studies to objectively assess the severity of swine intestinal disease and make an evidence-based evaluation of clinical effects following therapeutic or preventive interventions.
Keywords: faecal clinical score, score grid, enterotoxigenic Escherichia coli, Brachyspira hyodysenteriae, evaluation
1. Introduction
Swine production globally still encounters major challenges related to swine intestinal disease, which might be provoked by post-weaning diarrhea (PWD)¹–³ or growing pig diarrhea⁴–¹⁰. Both disease conditions are provoked by specific intestinal pathogens and remain a major reason for reduced pig performance and increased antimicrobial use to control or eliminate the pathogen. Post-weaning diarrhea in pigs is a worldwide economically important disease¹¹, characterized by increased mortality, weight loss, retarded growth, increased treatment costs, higher use of antibiotics, and pig weight variation²,³,¹¹,¹⁵. Enterotoxigenic E. coli (ETEC) is regarded as the most important cause of PWD. The ETEC pathotype is typically characterized by the presence of fimbrial adhesins, which mediate attachment to porcine intestinal enterocytes, and enterotoxins, which disrupt fluid homeostasis in the small intestine. This results in mild to severe diarrhea within a few days post-weaning, associated with clinical signs of dehydration, loss of body condition (= disappearance of muscle volume) and mortality¹⁶. The adhesive fimbriae most commonly occurring in ETEC from pigs with PWD are F4 (K88) and F18⁹. Other fimbriae such as F5 (K99), F6 (987P) and F41 rarely occur in E. coli isolates from PWD¹⁶,²⁰. In contrast, growing pig diarrhea has three major bacterial causes, namely Lawsonia intracellularis, Brachyspira species or Salmonella species, besides many viral or parasitic etiologies. Swine dysentery (SD) is provoked by Brachyspira hyodysenteriae – a β-hemolytic Gram-negative oxygen-tolerant anaerobic spirochete – and leads to mild to severe, characterized by bloody to mucoid, diarrhea, which is associated with mucus, fibrin, necrotic material, or blood in the faeces²⁴. Clinical signs usually start with loss of appetite and mild, yellow to grey colored diarrhea, further progressing to watery diarrhea with blood, mucus, fibrin, and pseudo-membranes⁶. This results in economic damage due to growth losses, mortality, increased variation in pig weight, and decreased feed conversion at farm level. Ileitis caused by L. intracellularis causes mild to severe diarrhea with or without melena. Depending on the clinical presentation, a chronic form (porcine proliferative adenomatosis) with low mortality or an acute form (porcine hemorrhagic enteritis) with sudden death may be observed. A subclinical form is also described causing weight loss and feed efficiency losses but without diarrhea, so it is only mentioned for completeness. In contrast, Salmonella is ubiquitous and clinical signs may be different depending on serotype or caused by dysbiosis.
These infectious intestinal diseases are currently controlled using antimicrobials combined with preventive management strategies such as oral supplementation of acids though the waterline, and adapted feeding strategies (dietary fiber, low protein levels, prebiotics, probiotics). In many parts of the world, the emergence of antimicrobial resistance towards E. coli strains isolated from cases of PWD²¹–²⁵ or B. hyodysenteriae strains isolated from cases of SD urges the need for alternative control strategies²⁶,³⁴.
Both under field and experimental conditions, the assessment of severity of swine intestinal disease remains a challenge. Many studies only report presence or absence of diarrhea without further differentiation of specific severity ranging from mild to severe faecal appearance. Other experimental studies collected faecal scores based on different faecal clinical scoring grids varying between a 3-(normal, soft, and watery³⁵), over 4-(normal, soft, mild, and severe³⁶) to 5-level scale (dry-pelleted, formed, moist cow-dung, diarrhea, and watery diarrhea³⁷,³⁹). Therefore, a need for standardized scoring grids to evaluate and analyze results of treatment and prevention is crucial both in PWD and SD⁴⁰,⁴⁴.
The objective is to demonstrate based on three different published field examples how a standardized faecal clinical scoring grid adapted to the specific clinical disease can help to evaluate, analyze, and interpret the results obtained from therapeutic or preventive interventions under field conditions.
2. Materials and Methods
2.1. EXPERIMENT 1 – EVALUATION OF FAECAL CLINICAL SCORES DURING THE POST-WEANING PHASE FOLLOWING AN ORAL LIVE AVIRULENT ESCHERICHIA COLI F4 VACCINATION
2.1.1. EXPERIMENTAL FARM DESCRIPTION
The field trial was performed on a conventional farrow-to-finish pig farm with 600 DanBred sows in Flanders (Belgium). The farm was managed in a 4-week batch-management system (with alternate weaning) with 120 sows per production batch. Piglets were weaned at 23 days of age and housed in specifically equipped post-weaning facilities, where they were raised for 7 weeks (50 days post-weaning). The post-weaning facility was equipped with 40 pens, which could each house 16 post-weaned piglets. Dry feeders with two waterers, one on each side, were located at the pen division, thus feeding two pens with a total of 32 piglets. The pens were further equipped with fully slatted plastic floors and were heated with hot water tubes on the side walls near the air inlet. Ventilation was performed through 3 ventilation tubes and fresh air entered the compartment directly from the outside.
2.1.2. ENTEROTOXIGENIC ESCHERICHIA COLI DIAGNOSIS AND CHARACTERIZATION AT FARM
The farm was selected following ETEC diagnostics during the post-weaning period. A confirmatory diagnostic sampling was performed consisting of untreated piglets (n = 10) with typical clinical signs of PWD, such as watery faeces, thin potbellied, and signs of dehydration, were sampled using rectal swabs (Sterile Transport Swab Amies with Charcoal medium; Copan Italia S.p.A., Brescia, Italy). All sampled piglets were between 3- and 5-days post-weaning. The diagnostic samples were sent to the laboratory (IZSLER, Brescia, Italy) under cooled conditions for further processing.
Specimen were processed using standard procedures for isolation and characterization of intestinal E. coli. Briefly, samples were plated on selective media and on tryptose soy agar medium supplemented with 5% of defibrinated ovine blood and incubated aerobically overnight at 37°C. Hemolytic activity was evaluated and single coliform colonies were further characterized.
DNA samples were prepared from one up to five hemolytic and/or non-hemolytic E. coli colonies and used to perform a multiplex PCR for the detection of fimbrial and toxin genes, including those encoding for F4 (K88), F5 (K99), F6 (987P), F18, F41, LT, STa, STb, and Stx2e, but not discriminating between F4ab, F4ac and F4ad.
All collected samples were positive for F4 in combination with STa, STb, and LT. No other virulence factors could be detected.
2.1.3. VACCINATION WITH A LIVE ORAL AVIRULENT ESCHERICHIA COLI F4 VACCINE
To vaccinate piglets at least 7 days before the clinical signs to mount sufficient protective local immunity in the gut³⁵,³⁶, piglets were vaccinated at 18 days of age (5 days prior to weaning), during the suckling period. The live avirulent E. coli F4 vaccine has a rapid onset of immunity (7 days) and a duration of immunity of 21 days post-vaccination³⁶, which covers the most critical period of PWD¹. An efficacy trial using an experimental E. coli F4 challenge at 7 days post-vaccination showed reduction of the severity and duration of PWD and reduction in faecal shedding of pathogenic F4-ETEC³⁶.
Sows were randomly assigned to Vaccine (Coliprotec® F4; Elanco, Greenfield, IN) or Control group based on their parity and sow number. Parities were equally distributed to both treatment groups. Piglets from sows assigned to the Vaccine group were vaccinated orally through drenching with 2 ml of a live avirulent E. coli F4 vaccine (Coliprotec® F4; Elanco, Greenfield, IN) at 18 days of age. Piglets from sows in the Control group were not treated nor vaccinated. No antibiotics were administered to any piglets from 15 days of age onwards, to omit interference with the development of protective local immunity by the E. coli F4 vaccine during the 7 days following vaccination.
2.1.4. EXPERIMENTAL DESIGN
At weaning, piglets were further divided by treatment groups. The E. coli vaccinated piglets were randomly assigned to three groups with different feeding strategies. The unvaccinated Control piglets were randomly assigned to two groups with different feed supplemented preventive measures against PWD due to E. coli. Each treatment group consisted of 128 piglets divided over 8 pens with 16 piglets each. Sexes were distributed equally within and between different treatment groups. The treatment groups were randomly allocated to the different pens within the compartment to evenly distribute all treatments for potential interaction with specific climatic subzones within the compartment (outer walls, air inlet, central part).
Details on the experimental design in relation to feeding strategies and preventive measures are given in Table 1. Piglet treatment identification was blinded to both farmer and veterinarian involved in trial follow-up by letter codes (A, B, C, D, and E).
Table 1.
Schematic description of experimental trial set-up including study groups with a comprehensive description and feeding strategies differences (weaning starter, starter and grow starter; blocks with the same F* code have identical compositions), addition of ZnO (3,000 ppm), supplementary nutraceuticals (MCFAs, organic acids and additional fibers), and vaccination with a live oral avirulent E. coli F4 vaccine.
| Treatment description | A | B | C | D | E |
|---|---|---|---|---|---|
| Normal + ZnO | Normal + nutraceuticals | Normal start / High 2nd & 3rd phase | High 3-phases | High 3-phases + nutraceuticals | |
| Weaning starter | 3 kg F1* | 3 kg F1* | 3 kg F1* | 3 kg F2** | 3 kg F2** |
| Starter | 5 kg F3* | 5 kg F3* | 5 kg F4** | 5 kg F4** | 5 kg F4** |
| Grow starter | … kg F5* | … kg F5* | … kg F6** | … kg F6** | … kg F6** |
| ZnO (14d) | 3,000 ppm | 0 | 0 | 0 | 0 |
| Nutraceuticals | 0 | 2 kg / tonne | 0 | 0 | 2 kg / tonne |
| E. coli F4 vaccine | no | no | yes | yes | yes |
* F1, F3 and F5 are diets with the normal energy (2421, 2389, and 2349 kcal NE/kg, respectively) and protein (16.7, 17.2, and 17.0 %, respectively) levels
** F2, F4 and F6 are diets with the increased energy (2530, 2447, and 2405 kcal NE/kg, respectively) and protein (19.0,18.9, and 18.6 %, respectively) levels.
2.1.5. FEEDING STRATEGIES
Feeding strategies were based on previous results³⁷, showing that a 3-phase feeding strategy has resulted in the most optimal production results. Additionally, different combinations of normal or high energy and protein levels in the 3-phase approach were evaluated together with E. coli F4 vaccination and addition of 2 kg per tonne of extra protective nutritional supplements, i.e., nutraceuticals, consisting of a combination of MCFAs, organic acids and additional fibers. Unvaccinated Control groups were also fed the 3-phase feeding strategy. One unvaccinated group (A) was designed to resemble the current field situation at that moment of the trial with addition of 3,000 ppm ZnO to the feed during the first 14 days post-weaning.
representing a positive control group, whereas the other unvaccinated group (B) was formulated with addition of 2 kg per tonne of neutraceutical supplement as a negative control group.
2.1.6. PEN FAECAL CLINICAL SCORE
Piglet faecal consistency was scored daily from d0 to d21 using a pen faecal clinical score (FCS) as described in Table 2. FCS was performed by the same person throughout the entire duration of the study (0-21 days). For pen FCS, one score per pen was attributed daily in the morning at 9 am. For analysis, area under the curve (AUC) and days to maximum score were calculated per pen. Clinical assessment of piglets with diarrhea was performed based on appearance of fluid watery stools in the anal and perineal region. The number of piglets per pen with these clinical signs was counted daily from d0 to d21 and reported as total number of piglets with diarrhea per treatment group over the entire observation period (0-21 days).
2.1.7. STATISTICAL ANALYSIS
For the ordinal outcomes of pen FCS, effect of treatment was assessed using pairwise comparison using Wilcoxon rank sum test. The P-values were adjusted with the Bonferroni method for multiple comparison. All tests were performed at the nominal level of 5%.
Table 2.
Comprehensive description of the pen faecal clinical score (FCS) with its interpretation and clinical aspect of the faecal clinical score.
| Score | Interpretation | Clinical aspect |
|---|---|---|
| 0 | Normal | Normal faecal consistency |
| 1 | Pasty | Soft pasty consistency |
| 2 | Mild | Presence of fluid, but more particles than fluid |
| 3 | Moderate | More fluid than particles |
| 4 | Severe | Fluid watery faeces |
2.2. EXPERIMENT 2 – EVALUATION OF FAECAL CLINICAL SCORES DURING THE POST-WEANING PHASE FOLLOWING ADAPTED FEED FORMULATIONS INCLUDING A ß-MANNANASE ENZYME
2.2.1. EXPERIMENTAL FARM DESCRIPTION
The field trial was performed on a conventional farrow-to-finish pig farm with 600 DanBred sows in Flanders (Belgium), managed in a 4-week batch-management system with 120 sows per production batch. The farm was managed on all-in/all-out basis in all production phases.
Piglets (DanBred x German Piétrain) were weaned at 21 days of age and housed in a specifically equipped post-weaning facility, where they were raised for 7 weeks (49 days). The post-weaning facility was equipped with 32 pens of 14 piglets, allocated in 8 rows of 4 pens, separated by 4 inspection aisles. Every two pens were equipped with a dry feeder with a waterer on each side, located at the pen partition, so each feeder fed 28 piglets. The pens were equipped with fully slatted plastic floors. Heating was provided by hot water tubes on the ceiling near the air inlet, and ventilation was performed through 3 ventilation tubes and fresh air entered through a perforated ceiling air inlet system.
2.2.2. DESCRIPTION OF CLINICAL PROBLEMS OF POST-WEANING DIARRHEA
The farm had a history of post-weaning diarrhea (PWD), recurrently diagnosed as enterotoxigenic E. coli (ETEC) F4, which expressed enterotoxins STa, STb, and LT. Clinical signs of PWD were characterized by watery yellowish diarrhea from 3 days post-weaning (dpw) onwards and was controlled through preventive vaccination with a live oral avirulent E. coli F4 and F18 vaccine (Coliprotec® F4/F18; Elanco, Greenfield, IN) at 5 days pre-weaning. Since onset of immunity by this vaccine is 7 days, the piglets were vaccinated timely in relation to the onset of clinical signs of PWD. Without E. coli vaccination, at least 10 days of antimicrobial treatment would have been necessary, and mortality would increase to 4-5%, as observed in previous trials with a non-vaccinated control group³⁷,³⁸. The E. coli vaccination had been administered as a standard preventive measure for several years prior to the trial. The challenging F4-ETEC infection pressure on this farm combined with the E. coli F4/F18 vaccine has resulted in a stable clinical situation related to PWD, although F4-ETEC might still be diagnosed post-weaning. Since label claims of the E. coli vaccine stated a reduction in F4-ETEC and F18-ETEC pathogen excretion following vaccination, it is not usual to diagnose F4-ETEC following vaccination³⁵,³⁶. No other enteric pathogens were detected that might cause PWD.
2.2.3. EXPERIMENTAL DESIGN
2.2.3.1. TREATMENT GROUPS AND FEEDING REGIMEN
Two experimental treatments were used, where the Control group received the standard diets and Enzyme treated group received the adapted nursery diets. A 3-phase feeding program with two basal diets was used, a common commercial feeding program, and a similar, adapted program with 300 g per tonne of heat-tolerant endo-1,4-β-mannanase (Hemicell HT Dry; Elanco, Greenfield, IN), with expensive protein sources partially replaced by extruded SBM in phase 1 and fully replaced by dehulled SBM in phase 2, and the enzyme was formulated to provide 65 kcal/kg NE in phase 3. The enzyme was added to the mixer with other minor ingredients during production of the diets at the feed mill. The diets used for the two treatments had similar nutrient content per kg in all three phases: digestible lysine content of 12.1 g/kg in phase 1, 11.8 g/kg in phase 2, and 11.8 g/kg in phase 3. Identical NE content of both Control and Enzyme treated group of 2,440 kcal/kg in phase 1 and 2,425 kcal/kg in phase 2 were used, while in phase 3, the NE content was 2,399 kcal/kg in the Control diet and 2,334 kcal/kg in Enzyme treated diet for a calculated NE reduction of 65 kcal/kg.
The 3-phase feeding program was offered as following: piglets were fed phase 1 feeds from days 1-15, phase 2 feeds from days 16-27 and phase 3 diets from days 28-49.
Substitution of expensive protein sources and reduction of 65 kcal/kg NE resulted in a substantial reduction of feed costs. The reduction of expensive protein sources in phase 1 had a substantial impact and reduced the feed cost by € 29.00 per tonne, whereas the substitutions in phase 2 and energy reduction in phase 3 had a slightly smaller impact of € 6.77 and € 4.45 per tonne, respectively.
2.2.3.2. STUDY ANIMALS
Two batches of 448 newly weaned piglets were allocated to treatment by weight and sex. Castrated males and females were penned separately. The same number of castrated male and female pigs were allocated to both treatment groups. All piglets were ear tagged with individual identification numbers. Pen represented the experimental unit. Sixteen pens of 14 piglets were included per treatment in each batch for a total of 32 replicates per treatment. The piglets at each feeder were randomly assigned to one of both treatment groups: Control or Enzyme treated.
2.2.3.3. ASSESSMENT OF FAECAL CLINICAL SCORE
Pigs were evaluated daily, and any unusual observations were recorded, including but not limited to altered behavior and disease. Diarrhea scores were assessed for each pen by scoring five droppings per pen based on the faecal clinical score (FCS) shown in Table 2³⁷,³⁸. The scoring was done by the same observer on days 0 to 11 post-weaning and on day 49 (end of trial). The scoring was done by first counting all droppings with a score 4, then score 3, subsequently 2 and 1, and finally score 0, until a total of 5 droppings had been recorded. FCS during the observation period were expressed as area under Table 3.
Faecal quality scoring grid for assessment of individual faecal quality. Sum (total faecal score, TFS) of faecal characteristics A, B and C was used for statistical analysis.
| Parameters | Score | Description |
|---|---|---|
| A. Faecal shape and consistency | 1 | Hard, dry, can be shaped by fingers |
| 2 | Normal, soft, but not watery | |
| 3 | Pasty, soft | |
| 4 | Watery | |
| B. Faecal colour | 0 | Normal dark green, brown, feed colour |
| 1 | Light brown to yellow | |
| 2 | Grey | |
| 3 | Dark brown to black | |
| C. Faecal additions | 0 | Normal, no additions |
| 1 | Slimy or foamy | |
| 2 | Bloody or fibrinous exudate |
3. Results
3.1. EXPERIMENT 1 – EVALUATION OF FAECAL CLINICAL SCORES DURING THE POST-WEANING PHASE FOLLOWING ORAL LIVE AVIRULENT ESCHERICHIA COLI F4 VACCINATION
Pen FCS was collected daily for each individual pen from d 0 to 21 days post-weaning. Pen FCS, expressed as AUC, was significantly higher (P ≤ 0.05) in group B (nutraceuticals) as compared to group C (low/high/high energy and protein), whereas pen FCS in all other groups was not significantly different (P > 0.05) from each other. Overall, pen FCS was comparable between group A (ZnO; gold standard), and the E. coli vaccinated groups (C, D, and E) (Table 4). Although numerical differences in days to maximum score occurred among treatment groups, no significant differences (P > 0.05) were observed (Table 4).
The number of piglets with clinical signs of diarrhea was significantly higher (n = 151; P ≤ 0.05) in group B (nutraceuticals) and group D (high energy and protein) as compared to group A (ZnO) and two of the E. coli vaccinated groups (C; low/high/high energy and protein, and E; high energy and protein + nutraceuticals).
Table 4.
Area under the curve (AUC) of pen faecal clinical score, time to maximum score (mean ± SEM) and total number of piglets with diarrhea during the first 21 days post-weaning. Pen FCS was scored daily on a score from 0 (= normal) to 4 (= watery diarrhea). Different treatment groups differed in diets composition (normal or high energy and protein levels) and preventive approach towards post-weaning diarrhea (ZnO, nutraceutical or vaccination against E. coli F4). Different row superscript letters indicate statistically significant differences (P < 0.05).
| A | B | C | D | E | |
|---|---|---|---|---|---|
| Pen FCS (AUC0-21) | 36.4 ± 9.7ᵃ | 72.2 ± 10.1ᵃᵇ | 27.1 ± 9.6ᵃᶜ | 44.3 ± 12.6ᵃ | 39.3 ± 7.8ᵃ |
| Days to maximum score | 7.00 ± 1.67 | 7.25 ± 1.10 | 9.62 ± 2.09 | 6.25 ± 1.01 | 9.25 ± 0.90 |
| # Piglets with diarrhea (0-21 d) | 49ᵃ | 151ᵇ | 41ᵃ | 99ᶜ | 69ᵃ |
3.2. EXPERIMENT 2 – EVALUATION OF FAECAL CLINICAL SCORES DURING THE POST-WEANING PHASE FOLLOWING ADAPTED FEED FORMULATIONS INCLUDING A ß-MANNANASE ENZYME
Pen FCS was collected daily for each individual pen from 0 to 11 dpw. Daily average pen FCS (mean ± SEM) is given in Figure 1. Pen FCS, expressed as AUC₀₋₁₁, was not significantly higher (P > 0.05) in the Control group as compared to the Enzyme treated group (Table 5). Although some numerical differences in time to maximum score, expressed as dpw, occurred between the treatments, no significant differences in the time to maximum score (P > 0.05) were observed (Table 5).
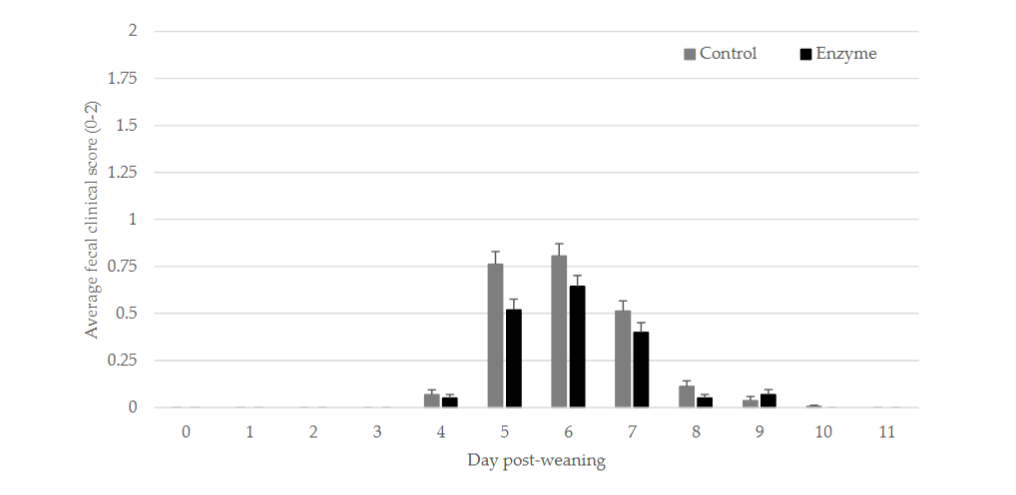
Figure 1.
Average faecal clinical score (mean ± SEM) from 0 to 11 dpw. Five relevant faecal droppings were scored daily per pen during the observation period. Piglets in the Control group were fed a standard diet, whereas piglets in the Enzyme treated group were fed an adapted diet with more soybean meal in phase 1 and 2, and a decreased level of 65 kcal/kg net energy in phase 3. No significant differences between groups were observed.
Table 5.
Area under the curve (expressed in AUC₀₋₁₁) of pen faecal clinical score, days to maximal FCS (expressed in dpw; mean ± SEM) for piglets during the first 11 dpw are given. Piglets in the Control group were fed a standard diet, whereas piglets in the Enzyme treated group were fed an adapted diet with more soybean meal in phase 1 and 2, and a decrease level of 65 kcal/kg net energy in phase 3. Different row superscript letters indicate statistically significant differences (P < 0.05).
| Control | Enzyme | |
|---|---|---|
| Pen FCS¹ (AUC₀₋₁₁) | 14.40 ± 0.215ᵃ | 13.65 ± 0.134ᵃ |
| Days to maximum score (dpw) | 7.06 ± 0.100ᵃ | 6.25 ± 0.077ᵃ |
¹Pen FCS was scored daily on a score from 0 (= normal) to 2 (= watery diarrhea).
AUC₀₋₁₁: area under the curve 0-11 dpw
dpw; days post-weaning
3.3. EXPERIMENT 3 – EVALUATION OF FAECAL CLINICAL SCORE DURING THE POST-WEANING AND GROW-FINISHING PHASE FOLLOWING TREATMENT OF SWINE DYSENTERY WITH A ZN-CHELATE PRODUCT
The total faecal score (TFS) of the individually monitored pigs was comparable between ID-treated pigs and control pigs on SD0 with an average score of 3.16 and ranging from 0 to 7 for both treatment groups (t-test, P > 0.05). ID-treated pigs had a lower TFS as compared to the control pigs (P > 0.05; Figure 2) from SD2 to SD6. Overall, at SD6, 25 of the 60 ID-treated pigs showed a TFS of zero compared with 20 out of the 57 control pigs.
At SD14, 8 days after the end of the ID treatment, TFS in ID-treated animals (TFS 0.39) remained significantly lower (P < 0.01) as compared to control pigs (TFS 1.23). This difference was mainly due to faecal consistency and faecal colour scores. Over the entire study, ID-treated pigs showed an average daily improvement of their TFS of 0.74, whereas TFS in control pigs only had an average daily improvement of 0.38. Moreover, an increase in TFS was observed in control pigs at SD14 (Figure 2). At SD14, the number of ID-treated pigs with a TFS of zero was 39 out of 60, while in control pigs, a TFS of zero was at 20 of 60.
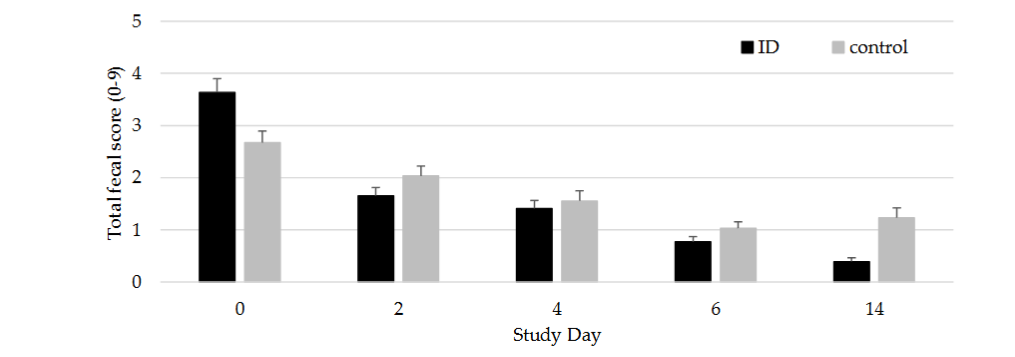
Figure 2.
Total faecal score (average ± SEM) of ID-treated and control pigs per study day from SD 0 to 14. Pigs were treated with ID from SD 0 to 6. Significant differences (P < 0.05) are indicated with asterisk*.
4. Discussion
The objective is to demonstrate based on three different published field examples how a standardized faecal clinical scoring grid can help to evaluate, analyze, and interpret the results obtained from therapeutic or preventive interventions under field conditions.
In the first field experience with a live oral avirulent E. coli F4 vaccine to protect against PWD (C, D, and E), both a negative control group (nutraceutical approach; B) and a positive control (ZnO 3,000 ppm for 14 days; A) were included in the evaluation. The nutraceutical approach (B) resulted in a significant increase (P < 0.05) in the total number of piglets with diarrhea as compared to the E. coli F4 vaccinated groups and the positive control group. Pen FCS in the nutraceutical group (B) was also significantly higher than in the E. coli F4 vaccinated group with the low/high energy and protein diet (C). Pen FCS in the nutraceutical group was only numerically higher as compared to positive control (A) and the E. coli F4 vaccinated groups with high energy and protein (D), and high energy and protein with additional nutraceuticals (E). No difference in the days to maximum score could be observed between all treatment groups, indicating that independent of the clinical severity of PWD, the timepoint with maximal loose stools was quite similar in all study groups.
Indeed, intestinal pathogens have many different mechanisms to interact with their host, which makes complete inhibition of their pathogenesis through specific feed additives or combination of these additives rather challenging⁴⁰,⁴¹. Incomplete control may explain the results obtained for total number of piglets with diarrhea and pen FCS being inconsistent. However, when combining the nutraceutical supplementation with an E. coli F4 vaccination in a high energy and protein diet, pen FCS improved and total number of piglets with clinical signs of PWD decreased. This indicates that under conditions of additional protection with an E. coli F4 vaccine, the nutraceuticals could support general gut health to improve the supplemented piglets’ clinical condition. Recently, vaccination with a live oral avirulent E. coli F4 or E. coli F4 and F18 vaccine has demonstrated efficacy against PWD due to F4-ETEC, and F4-ETEC and F18-ETEC³⁶. Immunization against the F4-ETEC and F18-ETEC pathogens resulted in decreased severity and duration of PWD clinical signs but faecal shedding of F4-ETEC and F18-ETEC may still occur³⁵,³⁶. Our results are in line with these observations, indicating that feeding strategy with an increased content of energy and protein had no negative impact on results induced by immunization with an E. coli F4 vaccine under field conditions. This implies that farms suffering from PWD due to F4-ETEC can still explore alternative or feeding strategies that result in higher piglet performances when piglets are concurrently protected through immunization with an E. coli F4 vaccine. Immunization may mitigate the increased risk of feeding higher energy and protein diets currently thought to induce PWD.
As expected, supplementation of ZnO resulted in acceptable pen FCS although days to maximum FCS did not differ among treatment groups. Nevertheless, from 14 days post-weaning onwards, at removal of the ZnO from the feed, pen FCS increased (data not shown), in contrast to the other groups, where pen FCS remained stable during that specific period. In practice, this phenomenon is referred to as ‘post-ZnO diarrhea’ and sometimes even needs antimicrobial treatment. E. coli vaccinated piglets had similar pen FCS, which remains an important evaluation parameter in practice, due to lack of many other directly available data for evaluation of preventive or clinical interventions to prevent or control PWD due to E. coli.
In the second field experience with supplementation of a β-mannanase enzyme combined with an adapted feed formulation, we compared pen FCS and days to maximum score in a Control group with a normal 3-phase diet to an Enzyme group with an adapted diet including replacement of digestible proteins by SBM and reducing the NE content with 65 kcal/kg of feed. Although some differences in kinetics could be observed for pen FCS, AUC₀₋₁₁ did not differ significantly (P > 0.05) between both treatment groups. Neither did time to maximum score during the entire period from 0-11 dpw differ significantly (P > 0.05) between both treatment groups. These results indicated that feeding post-weaned piglets with an adapted diet with more SBM in the phase 1 and 2 combined with supplementation of 300 g per tonne of a β-mannanase enzyme did not deteriorate piglet intestinal health during this critical period post-weaning. Therefore, feeding post-weaned piglets cheaper diet with inclusion of a β-mannanase enzyme could be an alternative to cope with high feed prices under the current economic circumstances.
In poultry, beneficial effects of a β-mannanase enzyme on the performance of chickens challenged with Eimeria sp. and Clostridium perfringens when observed together reduced lesion scores in disease-challenged birds⁴². Therefore, we selected a field trial facility with a substantial intestinal challenge related to PWD due to E. coli, which was partly ameliorated using an oral live oral avirulent E. coli vaccine (Coliprotec® F4/F18; Elanco, Greenfield, IA). Indeed, based on the pen FCS, we observed a clinical challenge with a maximum score between 6 and 7 dpw.
In the third field experience of an innovative therapeutic treatment of SD using ID (an oral Zn-chelate product) for 6 consecutive days, we compared a Control group with a standard therapeutic approach to an ID Treatment group. Treatment with ID for 6 consecutive days significantly improved TFS, which is a cumulative score of faecal consistency, colour, and additions (mucus, foam, blood, and necrotic material). Although the level of TFS was higher at SD0 in the Treatment group as compared to the Control group, treatment with ID resulted in a more rapid decrease (-55% vs. -24% in Treatment and Control group, respectively) of TFS. The faecal quality remained acceptable following the end of the 6-day ID treatment and even continued to decrease until SD14, the end of the field experience. This observation is rather interesting, especially since the pigs were housed under challenging conditions, including a high-welfare environment, which implicates more than 80 percent solid flooring, partly (50 percent) bedded with straw, and only a very little slatted surface. These circumstances do not favor the lowering of B. hyodysenteriae-infected faeces throughout the study and might expose the pigs to moderate to high amounts of potentially contagious faecal material. Moreover, intensive observation of the pigs during the study revealed that a high percentage of pigs consumed liquid faecal material present on the solid pen floors (coprophagia). Therefore, the continuing reduction of TFS in the ID-treated group, even after the 6-day treatment period, remains a remarkable and encouraging finding.
Based on these different field experiences, we can conclude that the practical use of a differentiated faecal scoring grid with multiple levels has its advantages over a simple score indicating only presence vs. absence of diarrhea. Using a 3- to 5-level scale, depending on the situation and the specific infection, gives one the opportunity to analyze several aspects of the clinical evolution and severity of diarrhea problems in pigs at different production stages. From a kinetic point of view, AUCperiod and days to maximum score can be calculated and compared among different study groups, resulting in a more detailed documentation of clinical differences of preventive or therapeutic interventions. When including the total number of pigs with diarrhea over a defined period, the total impact of an intervention can also be assessed in a more quantitative approach. These advantages allow a more sophisticated statistical analysis to be performed to quantify the effect of preventive or therapeutic interventions on the clinical outcomes.
5. Conclusions
In conclusion, adapted faecal clinical scoring systems for different swine intestinal diseases or phases of production are an important tool in both field and experimental studies to objectively assess the severity of swine intestinal disease and make an evidence-based evaluation of clinical effects following therapeutic or preventive interventions.
Author Contributions:
Conceptualization, F.V. and J.M.H.; methodology, F.V.; formal analysis, F.V.; investigation, F.V.; data curation, F.V.; writing—original draft preparation, F.V. and J.M.H.; writing—review and editing, F.V. and J.M.H. All authors have read and agreed to the published version of the manuscript.”
Funding:
This research received no external funding
Institutional Review Board Statement:
Ethical review and approval were waived for this study, due to the lack of direct interaction with the animals while collecting faecal clinical scores during the different studies.
Data Availability Statement:
Data available upon reasonable request to the author.
Acknowledgments:
The authors greatly acknowledge the technical support by all swine farmers during the trials.
Conflicts of Interest:
The authors declare no conflict of interest.
References
1. Fairbrother, J.M.; Nadeau, É.; Gyles, C.L. Escherichia coli in postweaning diarrhea in pigs: an update on bacterial types, pathogenesis, and prevention strategies. Anim Heal Res Rev 2005, 6, 17-39.
2. Hoa, N.X.; Kalhoro, D.H.; Lu, C. Distribution of serogroups and virulence genes of E. coli strains isolated from porcine post weaning diarrhea in Thua Thien Hue province Vietnam. Tạp chí Công ngh Sinh học 2013, 11, 665-672.
3. Lyutskanov, M. Epidemiological characteristics of post-weaning diarrhea associated with toxin-producing Escherichia coli in large intensive pig farms. Trakia J Sci 2011, 9, 68-73.
4. Alvarez-Ordóñez, A.; Martinez-Lobo, F.J.; Arguello, H.; Carvajal, A.; Rubio, P. Swine dysentery: aetiology, pathogenicity, determinants of transmission and the fight against the disease. Int J Environ Res Public Health 2013, 10, 1927-1947.
1. Hampson, D.J.; Lugsomya, K.; La, T.; Phillips, N.D.; Trott, D.J.; Abraham, S. Antimicrobial resistance in Brachyspira – an increasing problem for disease control. Vet Microbiol 2019, 229, 59-71.
2. Hampson, D.J. Brachyspiral colitis. In: Zimmerman JJ, Karriker LA, Ramirez A, Schartz KJ, Stevenson GW, editors. Diseases of Swine. 10th ed. Chichester: Wiley-Blackwell. 2012, 680-696.
3. Amaechi, N.; Ezeronye, O.U. Piggery environment as a source of Salmonella contamination for swine. J Anim Vet Adv 2006, 5, 102-107.
4. Barber, D.A.; Bahnson, P.B.; Isaacson, R.; Jones, C.J.; Weigel, R.M. Distribution of Salmonella in swine production ecosystems. J Food Prot 2002, 65, 1861-1868.
5. Karuppannan, A.K.; Opriessnig, T. Lawsonia intracellularis: Revisiting the Disease Ecology and Control of This Fastidious Pathogen in Pigs. Front Vet Sci 2018, 5, 181-187.
6. McOrist, S.; Gebhart, C.J. Proliferative enteropathy. In: Zimmerman JJ, Karriker LA, Ramirez A, Schartz KJ, Stevenson GW, editors. Diseases of Swine. 10th ed. Chichester: Wiley-Blackwell. 2012, 811-820.
7. Svensmark, B.; Jorsal, S.E.; Nielsen, K.; Willeberg, P. Epidemiological studies of piglet diarrhoea in intensively managed Danish sow herds. I. Pre-weaning diarrhoea. Acta Vet Scand 1989, 30, 43-53.
8. Svensmark, B.; Nielsen, K.; Willeberg, P.; Jorsal, S.E. Epidemiological studies of piglet diarrhea in intensively managed Danish sow herds. II. Post-weaning diarrhea. Acta Vet Scand 1989, 30, 55-62.
9. Tubbs, R.C.; Hurd, H.S.; Dargatz, D.; Hill, G. Preweaning morbidity and mortality in the United States swine herd. Swine Heal Prod 1993, 1, 21-28.
10. USDA. Part II. Reference of Swine Health and Health Management in the United States. USDA:APHIS:VS, CEAH, National Animal Health Monitoring System, Fort Collins, CO. 2002, vol. #N355.0202.
11. Zhang, W.; Zhao, M.; Ruesch, L.; Omot, A.; Francis, D. Prevalence of virulence genes in Escherichia coli strains recently isolated from young pigs with diarrhea in the US. Vet Microbiol 2007, 123, 145-152.
12. Fairbrother, J.M.; Gyles, C.L. Colibacillosis. In: Zimmerman JJ, Karriker LA, Ramirez A, Schartz KJ, Stevenson GW, editors. Diseases of Swine. 10th ed. Chichester: Wiley-Blackwell. 2012, p. 723-749.
13. Chen, X.; Gao, S.; Jiao, X.; Liu, X.F. Prevalence of serogroups and virulence factors of Escherichia coli strains isolated from pigs with postweaning diarrhoea in eastern China. Vet Microbiol 2004, 103, 13-20.
14. Frydendahl, K. Prevalence of serogroups and virulence genes in Escherichia coli associated with postweaning diarrhoea and edema disease in pigs and a comparison of diagnostic approaches. Vet Microbiol 2002, 85, 169-182.
15. Luppi, A.; Gibellini, M.; Gin, T.; Vangroenweghe, F.; Vandenbroucke, V.; Bauerfeind, R.; Bonilauri, P.; Labarque, G.; Hidalgo, Á. Prevalence of virulence factors in enterotoxigenic Escherichia coli isolated from pigs with post-weaning diarrhea in Europe. Porcine Health Manag 2016, 2, 20-25.
16. Vu-Khac, H.; Holoda, E.; Pilipcinec, E.; Blanco, M.; Blanco, J.E.; Mora, A.; Dahbi, G.; Lopéz, C.; González, E.A.; Blanco, J. Serotypes, virulence genes, and PFGE profiles of Escherichia coli isolated from pigs with postweaning diarrhoea in Slovakia. BMC Vet Res 2006, 2, 13-20.
17. Abraham, S.; Trott, D.J.; Jordan, D.; Gordon, D.M.; Groves, M.D.; Fairbrother, J.M.; Smith, M.G.; Zhang, R.; Chapman, T.A. Phylogenetic and molecular insights into the evolution of multidrug-resistant porcine enterotoxigenic Escherichia coli in Australia. Int J Antimicrob Agents 2014, 44, 105-111.
18. Abraham, S.; Jordan, D.; Wong, H.S.; Johnson, J.R.; Toleman, M.A.; Wakeham, D.L.; Gorden, D.M.; Turnidge, J.D.; Mollinger, J.L.; Gibson, J.S.; Trott, D.J. First detection of extended-spectrum cephalosporin- and fluoroquinoloneresistant Escherichia coli in Australian food-producing animals. J Glob Antimicrob Resist 2015, 3, 273-277.
19. Boyen, F.; Vangroenweghe, F.; Butaye, P.; De Graef, E.; Castryck, F.; Heylen, P.; Vanrobaeys, M.; Haesebrouck, F. Disk prediffusion is a reliable method for testing colistin susceptibility in porcine E. coli strains. Vet Microbiol 2010, 144, 359-362.
20. Jahanbakhsh, S.; Smith, M.G.; Kohan-Ghadr, H.R.; Letellier, A.; Abraham, S.; Trott, D.J.; Fairbrother, J.M. Dynamics of extended-spectrum cephalosporin resistance in pathogenic Escherichia coli isolated from diseased pigs in Quebec, Canada. Int J Antimicrob Agents 2016, 48, 194-202.
21. Luppi, A.; Bonilauri, P.; Dottori, M.; Gherpelli, Y.; Biasi, G.; Merialdi, G.; Maioli, G.; Martelli, P. Antimicrobial resistance of F4+ Escherichia coli isolated from swine in Italy. Transbound Emerg Dis 2013, 62, 67-71.
22. Mahu, M.; Pasmans, F.; Vranckx, K.; De Pauw, N.; Van de Maele, L.; Vyt, P.; Vandersmissen, T.; Martel, A.; Haesebrouck, F.; Boyen, F. Presence and mechanisms of acquired antimicrobial resistance in Belgian Brachyspira hyodysenteriae isolates belonging to different clonal complexes. Vet Microbiol 2017, 207, 125-132.
23. Duinhof, T.F.; Dierickx, C.M.; Koene, M.G.J.; van Bergen, M.A.P.; Mevius, D.J.; Veldman, K.T.; van Beers-Schreurs, H.M.G.; de Winne, R.T.J.A. Multiresistentie bij Brachyspira hyodysenteriae-isolaten op een varkensvermeerderingsbedrijf in Nederland. Tijdschr Diergeneesk 2008, 133, 604-608.
24. Hidalgo, A.; Carvajal, A.; Vester, B.; Pringle, M.; Naharro, G.; Rubio, P. Trends towards lower antimicrobial susceptibility and characterization of acquired resistance among clinical isolates of Brachyspira hyodysenteriae in Spain. Antimicrob Ag Chemother 2011, 55, 3330-3337.
25. Joerling, J., Barth, S.A.; Schlez, K.; Willems, H.; Herbst, W.; Ewers, C. Phylogenetic diversity, antimicrobial susceptibility and virulence gene profiles of Brachyspira hyodysenteriae isolates from pigs in Germany. PlosOne 2018, 13, e0190928.
26. Karlsson, M.; Aspán, A.; Landén, A.; Franklin, A. Further characterization of porcine Brachyspira hyodysenteriae isolates with decreased susceptibility to tiamulin. J Med Microbiol 2004, 53, 281-285.
27. Kirchgässner, C.; Schmitt, S.; Borgström, A.; Wittenbrink, M.M. Antimicrobial susceptibility of Brachyspira hyodysenteriae in Switzerland. Schweiz Arch Tierheilk 2016, 158, 405-410.
28. Lobová, D.; Smola, J.; Čížek, A. Decreased susceptibility to tiamulin and valnemulin among Czech isolates of Brachyspira hyodysenteriae. J Med Microbiol 2004, 53, 287-291.
29. Massacci, F.R.; De Luca, S.; Cucco, L.; Tentellini, M.; Perreten, V.; Pezzotti, G.; Magistrali, C.F. Multiresistant Brachyspira hyodysenteriae shedding by pigs during the fattening period. Vet Rec 2018. https://doi.org/10.1136/vr.104886.
30. Fonseco Pascoal, L.A.F.; Thomaz, M.C.; Watanabe, P.H.; dos Santos Ruiz, U.; Bertocco Ezequiel, J.M.; Borges Amorim, A.; Daniel, E.; Iselda Masson, G.C. Fiber sources in diets of newly weaned piglets. Rev Bras Zootec 2012, 41, 636-642.
31. Liu, G.; Guan, G.; Fang, J.; Martinez, Y.; Chen, S.; Bin, P.; Duraepandiyan, V.; Gong, T.; Tossou, M.C.B.; Al-Dhabi, N.A.; Yin, Y. Macleaya cordata extract decreased diarrhea score and enhanced intestinal barrier function in growing piglets. BioMed Res Int 2016, 1069595. https://doi.org/10.1155/2016/1069585.
32. Madec, F.; Bridoux, N.; Bounaix, S.; Cariolet, R.; Duval-Iflah, Y.; Hampson, D.J.; Jestin, A. Experimental models of porcine post-weaning colibacillosis and their relationship to post-weaning diarrhoea and digestive disorders as encountered in the field? Vet Microbiol 2000, 72, 295-310.
33. Girard, M.; Thanner, S.; Pradervand, N.; Hu, D.; Ollagnier, C.; Bee, G. Hydrolysable chestnut tannins for reduction of postweaning diarrhea: Efficacy on an experimental ETEC F4 model. PlosOne 2018, 13, e0197878.
34. Zhao, P.; Li, H.; Lei, Y.; Li, T.; Kim, S.; Kim, I. Effect of fermented medicinal plants on growth performance, nutrient digestibility, fecal noxious gas emissions, and diarrhea score in weanling pigs. J Sci Food Agric 2016, 96, 1269-1274.
35. Fairbrother, J.M.; Nadeau, E.; Bélanger, L.; Tremblay, C.-L.; Tremblay, D.; Brunelle, M.; Wolf, R.; Hellmann, K.; Hidalgo, A. Immunogenicity and protective efficacy of a single-dose live non-pathogenic Escherichia coli oral vaccine against F4-positive enterotoxigenic Escherichia coli challenge in pigs. Vaccine 2017, 35, 353-360.
36. Nadeau, E.; Fairbrother, J.M.; Zentek, J.; Bélanger, L.; Tremblay, D.; Tremblay, C.-L.; Röhe, I.; Vahjen, W.; Brunelle, M.; Hellmann, K.; Cvejíc, D.; Brunner, B.; Schneider, C.; Bauer, K.; Wolf, R.; Hidalgo, A. Efficacy of a single oral dose of a live bivalent E. coli vaccine against post-weaning diarrhea due to F4 and F18-positive enterotoxigenic E. coli. Vet J 2017, 226, 32-39.
37. Vangroenweghe, F.; Thas, O. Improved piglet performance and reduced antibiotic use following oral vaccination with a live avirulent Escherichia coli F4 vaccine against post-weaning diarrhea. J Clin Res Med 2020, 3, 1-8.
38. Vangroenweghe, F; Thas, O. Application of high energy and protein diets in combination with a live avirulent Escherichia coli F4 vaccine against post-weaning diarrhea. Vacc Res 2020, 7, 1-9.
39. Vangroenweghe, F.; Poulsen, K.; Thas, O. Supplementation of a β-mannanase enzyme reduces post-weaning diarrhea and antibiotic use in piglets on an alternative diet with additional soybean meal. Porcine Health Manag 2021, 7, 8-19.
40. Tran, T.H.T.; Everaert, N.; Bindelle, J. Review on the effects of potential prebiotics on controlling intestinal enteropathogens Salmonella and Escherichia coli in pig production. J Anim Physiol Anim Nutr (Berl) 2018, 102, 17-32.
41. Daudeling, J.-F.; Lessard, M.; Beaudoin, F.; Nadeau, E.; Bissonnette, N.; Boutin, Y.; Brousseau, J.-P.; Lauzon, K.; Fairbrother, J.M. Administration of probiotics influences F4 (K88)-positive enterotoxigenic Escherichia coli attachment and intestinal cytokine expression in weaned pigs. Vet Res 2011, 42, 69-79.
42. Vangroenweghe, F.; Allais, L.; Van Driessche, E.; van Berkel, R.; Lammers, G.; Thas, O. Evaluation of a zinc chelate on clinical swine dysentery under field conditions. Porcine Health Manag 2022, 6, 1-10.
43. Jackson, M.E.; Anderson, D.M.; Hsiao, H.Y.; Mathis, G.F.; Fodge, D.W. Beneficial effect of β-mannanase feed enzyme on performance of chicks challenged with Eimeria sp. And Clostridium perfringens. Av Dis 2003, 47, 759-763.

