Active Metabolites in Stingless Bee Nest Materials
Active metabolites of stingless bee nest materials used for meliponitherapy: Bibliometrics, impact of biodiversity in conservation, and emerging microbiome
Patricia Vit, MSc, PhD1*, Vassya Bankova, MSc, PhD2, Gina Meccia, MSc3, David S Nogueira, MSc, PhD4, Christopher Mduda, MSc, PhD5, Megan T Halcroft, PhD6, Zhengwei Wang, MSc, PhD7, Enrique Moreno8, Qibi Wang, MSc9, Amelia Nicolas, MSc, PhD10, María Araque, MD, PhD11, and Jason E Stajich, PhD12
- Apitherapy and Bioactivity (APIBA), Food Science Department, Faculty of Pharmacy and Bioanalysis, Universidad de los Andes, Mérida 5101, Venezuela
- Institute of Organic Chemistry with Centre of Phytochemistry, Bulgarian Academy of Sciences, 1113 Sofia Bulgaria.
- Apitherapy and Bioactivity (APIBA), Research Institute, Faculty of Pharmacy and Bioanalysis, Universidad de los Andes, Mérida 5101, Venezuela
- Instituto Federal de Educação, Ciencia e Technologia do Amazonas, Cacaochiera, São Gabriel da Cachoeira, 69750-000, Amazonas, Brazil.
- Department of Crop Science and Beekeeping Technology, University of Dar es Salaam Dar es Salaam, Tanzania.
- Beec, Business, 13 Walkerty War Road, Hampton, NSW, 2790, Australia.
- CAS Key Laboratory of Tropical Forest Ecology, Xishuangbanna Tropical Botanical Garden, Chinese Academy of Sciences, Kunming 650000, China
- Smithsonian Tropical Research Institute, Balsa, Arnam, Republic of Panama
- School of Ecology and Environment, Yunnan University, Kunming 650500, China
- Central Bicol State University of Agriculture, Camarines Sur, Bicol, Philippines
- Laboratory of Microbiology, Department of Microbiology and Parasitology, Faculty of Pharmacy and Bioanalysis, Universidad de Los Andes, Mérida 5101, Venezuela.
- Institute for Integrative Genome Biology; Department of Microbiology and Plant Pathology, University of California Riverside, Riverside, CA 92521, United States of America.
OPEN ACCESS
PUBLISHED: 30 November 2025
CITATION: Vit, P., Bankova, V., et al., 2025. Active metabolites of stingless bees nest materials used for meliponitherapy: Bibliometrics, impact of biodiversity in conservation, and emerging microbiome. Medical Research Archives, [online] 13(11).
https://doi.org/10.18103/mra.v13i11.7096
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i11.7096
ISSN 2375-1924
Abstract
Stingless bees (Hymenoptera: Apidae: Apinae: Meliponini) collect biotic and abiotic resources from nature to be transformed into nest materials with diverse functions such as structural, immune, defense, and nutritional. The 605 species of stingless bees collecting natural resources processed with associated microbiota are a spectacular biodiversity forming pot-honey, pot-pollen, cerumen and propolis valued in meliponitherapy. The bioactive metabolites have botanical, entomological, and microbial origins. Only pot-honey has been regulated since 2014 in Bahia, Brazil and further national standards. Forecasts of climate change affect stingless bee distribution, their productivity, and may influence the diversity of active metabolites in the nest. A sequence of researches serving meliponitherapy illustrated the ancient use of pot-honey eye drops to the latest cerumen components, reducing oxidative stress, and recent synergism with antibiotics to overcome antimicrobial resistance. Besides the chemical composition, the antioxidant and antimicrobial activities are fundamental added values, supporting a medicinal approach for both nutritional and pharmaceutical applications. Increased aliphatic organic acid contents in fermented pot-honey is not a defect, but a microbial biotransformation to preserve their wet honey with active metabolites. Characterizing the microbiome of stingless bees and their nest materials assists in identifying potential active biomolecules of microbial origin. Authenticity and chemical variability were discussed for quality control. Bibliometrics complemented this review for medicinal stingless bees (2004–2023) and stingless bees in climate change (2010–2023). Neotropical biodiversity of stingless bees was evidenced with the 259 stingless bee species richness in Brazil, 95 of them used in meliponiculture. Nest materials of 64 stingless bee taxa in 14 countries were reviewed for their flavonoid and polyphenol contents, and 13 biological activities. Conservation of stingless bees’ biodiversity has been addressed in the face of climate change and the chemical pool represented for meliponitherapy. Active metabolites from the stingless bee nest are not envisaged to be extracted but to be used in their original matrices: pot-honey, pot-pollen, cerumen, or propolis. A synthesis of most active metabolites could be an option for pharmaceutical developments to reproduce a bioactive chemical repertoire of stingless bees in nature, with a role on Sustainable Development Goals SDG2 food security and SDG3 good health and well-being.
Keywords: antimicrobial resistance, biological activity, cerumen, climate change, flavonoids and polyphenols, medicinal, meliponitherapy, microbiome, pot-honey, pot-pollen, propolis, stingless bee.
1. Introduction
As the largest Neotropical country, Brazil has been leading conservation policies to protect biodiversity, and meliponine conservation is one example of that. The first official standard for stingless bee honey was approved in the state of Bahia for Melipona honey. Promoting nutraceutical uses of stingless bee pot-honey, pot-pollen, cerumen, and propolis, considers rational exploitation protecting natural resources for a sustainable meliponine industry. The diversity of active metabolites for meliponitherapy rely on the bee flora; the bees harvesting and transporting resources to the nest; colony and microbial processing; and chemical transformations.
The global decrease in rainforests has adverse impact on biodiversity of stingless bees and plants used as nesting sites, nest materials, and food sources for their colonies. Particularly, Brazilian deforestation in the Neotropical region, and the greatest 2020 deforestation rate of the Brazilian Amazon in a decade need our attention. Rocha et al. (2020) analyzed the loss of functional diversity. Knowing the vital role of honeydew, nectar, pollen, oil, and resin use of plant resources by stingless bee colonies, any altered tree landscape would affect bee density and performance. Having diverse requirements and food preferences, different stingless bee species would adapt better than others to the deforestation stress in a dynamic forest community, causing shifts in abundance and diversity. Bees and forests have a vital synergism. Therefore, distinctive conservational objectives are linked to the utilization of stingless bee products. The Atlantic Forest is the second largest tropical rainforest in the American continent after the Amazon, home of stingless bees, their nest products, and communities exploiting these natural resources managed for meliponiculture.

Stingless bee keepers harvest pot-honey, pot-pollen, cerumen, and propolis from stingless bee nests for their unique sensory attributes and beneficial medicinal values. Stingless bee studies have become a hotspot in the international bee product research, with a progressive expansion to characterize their entomological biodiversity, and to propose therapeutic applications.
Main topics of the Apitherapy section in the Apimondia Congress held in Santiago de Chile, September 2023, were:
- Scientific-based evidence supporting the nutritional, physiological, and health claims of bee products,
- Preclinical research – safety, pharmacology, and toxicology of bee products. Guidelines for medical applications,
- Clinical trials in apitherapy – doses, interactions, side effects (human and veterinary medicine).
- Update on the use of apitherapy in infectious diseases, and
- Regulatory issues and clinical ethics related to the integration of apitherapy as TCM in healthcare systems.
The first topic on bee products, particularly stingless bee nest materials, was reviewed because diverse nest materials have different chemical composition and added values on bioactive properties for pharmaceutical design. Their attributes caused by the botanical origin is an investigation initiated with Apis mellifera, and further variations caused by the entomological origin were mandatory for the meliponine biodiversity—Engel et al. (2023) recognize 605 stingless bee species—scientific attention was more recently addressed to the microbial origin of.
The support of traditional knowledge, such as to Scaptotrigona mexicana pot-honey by ethanolic fermentation was demonstrated with Tetragonisca angustula pot-honey in Venezuela, and recent studies exploring the meliponine rich microbiome producing pot-honey metabolites of microbial origin quantified by targeted 1H-NMR.
Another approach to differentiate nest materials is using non-invasive techniques to study volatile organic compounds (VOCs) with diverse chemical structures, ecological roles, and origins. For example, a diversity of 95 VOCs in Tetragonisca angustula cerumen types and propolis was detected, identified by HS-SPME/GC-MS, and grouped in chemical classes: 1. Acids (11), 2. Alcohols (16), 3. Aldehydes (7), 4. Esters (16), 5. Ketones (8), 6. Monoterpenes (17), 7. Oxides (5), 8. Sesquiterpenes (11), and 9. Others (4) by Betta et al. (2024). Their transformations are fascinating, waiting for suggested biochemical and microbial pathways. The acetic acid accumulated in the cerumen of empty honey pots, was esterified into methyl acetate in the entrance tube, and five acetates in the Tetragonisca angustula honey pots.
Microbial metabolites of stingless bee nest materials were reviewed by Vit (2024) for alcohols (ethanol, glycerol, isoamylic), aliphatic organic acids (AOA) (acetic, gluconic, lactic, oxalic, succinic, tartaric), amino acids (phenylalanine, proline, pyroglutamic acid), antibiotics (meliponamycin A, B, recently discovered as microbial metabolites in Melipona scutellaris), diphenylether (asterric acid), polyketide pigments (monascin), phenolic acids (3-phenyllactic acid), polyols (2,3-butanediol), statins (lovastatin), steroids (ergosterol), sugars (dihydroxyacetone, maltose, raffinose, trehalulose, turanose), surfactants (suspected sophorolipids), and vitamins (ascorbic acid). Surprisingly, a honey authenticity test revealed suspected microbial associations with the Scaptotrigona vitorum pot-honey from Ecuador.
These studies are beneficial for multifactorial medicinal stingless bee science and applications in integrative medicine as a good source of bioactive natural compounds with therapeutic and nutritional value. Salomon et al. observed significant reduction of cotton pellet-induced granuloma weights at all doses tested (27.34%, 35.53% and 47.53% granuloma inhibition) in Wistar rats treated daily with injected intravenously Tetragonisca fiebrigi honey (1000 mg/kg b.w.) for a week. In contrast, significant reduction in hind paw edema (44.44%) was achieved with Tetragonisca fiebrigi honey oral administration, causing analgesic responses in the three models used (acetic acid, formalin, tail immersion). Antioxidant activity, melissopalynological, physicochemical, phenolics, and sugars HPLC assessments of the honey were provided.
The tropical uses of pot-honey, pot-pollen, cerumen and propolis in meliponitherapy are traditional knowledge/constitute traditional knowledge gaining scientific interest for their biological activities and the most studied flavonoids and polyphenols active biomolecules. The scientific literature was tabulated for that approach, and additionally complemented with bibliometrics on medicinal stingless bees, and a further evaluation of stingless bees and climate change to understand the impact on biodiversity conservation. Protection of stingless bee and associated microbiome biodiversity is considered vital for chemical diversity of nest materials used in meliponitherapy.
This wide-ranging review integrates the chemical, biological, ecological, and therapeutic dimensions of stingless bee nest materials within the framework of meliponitherapy. It encompasses a detailed exploration of the diversity of active metabolites originating from botanical, entomological, and microbial sources found in pot-honey, pot-pollen, cerumen, and propolis of stingless bees. The review includes a bibliometric analysis of global research on medicinal stingless bees and their relationship with climate change, highlighting publication trends, collaborative networks, and research impact. It further synthesizes current evidence on the antioxidant, antimicrobial, and pharmacological activities of these bioactive compounds and their implications for nutraceutical and pharmaceutical development. It also assesses the importance of biodiversity conservation and related microbiomes, as well as their promising medical applications. The primary objective is to establish a science-based framework to promote the rational and sustainable use of these natural products for pharmaceutical and nutraceutical applications, considering the potential effects of environmental changes on their chemical composition and availability. Our aim is to provide a sound background on the science supporting meliponitherapy, to propel discoveries on microbial biomolecules as potential medicinal natural resources for health, alone or combined with drugs.
2. Bibliometrics
Science mapping from the citation network and a tool to visualize bibliometric networks such as Bibliometrix are needed to describe structures of research. This section has two bibliometric reviews on stingless bees, one on their medicinal uses (107 documents) in the period 2004 to 2023, and another on climate change (25 documents) in the period 2010 to 2023. The annual growth rate of both datasets is compared in Figure 2. There is a tendency to increase the number of documents with time. The annual growth of medicinal stingless bees recorded with the first paper in 2004, escalated in 2012 and 2013, with a sudden drop followed by a second peak in 2018, and a further drop with a steady growth in the last three years up to 15 documents in 2023. The annual growth of stingless bees and climate change shows the first paper in 2010, the second in 2015, a yearly paper from 2017 to 2019, growing interest reaching a peak of 7 papers in 2022. However, production decreased to 3 documents in 2023, that may recover in two weeks before the end of the year.
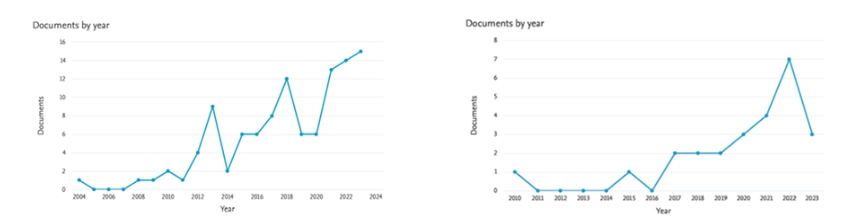
2.1 METHODOLOGY AND RESULTS OF THE BIBLIOMETRIC REVIEW ON MEDICINAL STINGLESS BEES
The Scopus database was used to review the scientific literature on medicinal stingless bees. Other words were used in the query string but the number of retrieved documents was lower; for example, using apitherapy only 16 documents, and 37 documents using pharmaceutical. Therefore, we selected medicinal for the search done on the 1st December 2023.
TITLE-ABS-KEY ( stingless bee AND medicinal )
The main information of bibliometric descriptors is presented in Table 1 including publications from the first retrieved document since 2004 to 2023, almost two decades. The 107 documents of the dataset were published in four languages: English (100), Portuguese (3), Spanish (3), and Chinese (2). Note that the addition gives 108 documents, possibly one was bilingual or considered bilingual by the dataset.
| Main information of the dataset bibliometric descriptors | Counts of all documents |
|---|---|
| Timespan | 2004:2023 |
| Sources (Journals, Books, etc) | 80 |
| Documents | 107 |
| Annual Growth Rate % | 15.32 |
| Document Average Age | 4.84 |
| Average Citations per Document | 18.63 |
| References | 5419 |
| Document Contents | |
| Keywords Plus (ID) | 869 |
| Author’s Keywords (DE) | 327 |
| Authors | 489 |
| Authors of Single-Authored Documents | 6 |
| Authors Collaboration | |
| Single-Authored Documents | 6 |
| Multi-Authored Documents | 101 |
| Co-Authors per Document | 4.97 |
| International Co-Authorships % | 24.3 |
| Document Types | |
| Article | 73 |
| Book | 1 |
| Book chapter | 12 |
| Conference paper | 4 |
| Editorial | 1 |
| Note | 1 |
| Review | 15 |
| No. of languages | 4 |
| English | 100 |
| Portuguese | 3 |
| Spanish | 3 |
| Chinese | 2 |
The first document of this dataset was Quality standards for medicinal uses of Meliponinae honey in Guatemala, Mexico and Venezuela, which was cited 132 times since 2004. The International Honey Commission was not supporting the proposal of medicinal stingless bee honey, but honey eye drops were sold in pharmacies before we started our research, and more recently online pharmacies offer the familiar eye drops, pot-honey, supplements with pot-honey, pot-pollen, and stingless bee propolis. The Vit et al. article was published the same year of the Apidologie special number for European honey, with the seminal article Main European unifloral honeys: Descriptive sheets by Livia Persano-Oddo and Roberto Piro on more than 35,000 unifloral, multifloral and honeydew honeys. The next document on this topic had to wait until 2008, Composition and antioxidant activity of Tetragonula carbonaria honey from Australia, cited as Trigona carbonaria at that time. Medicinal animals as therapeutic alternative in a semi-arid region of Northeastern Brazil, Properties of honey from Tetragonisca angustula fiebrigi and Plebeia wittmanni of Argentina and Antimicrobial activity of honey from the stingless bee Tetragonula carbonaria determined by agar diffusion, agar dilution, broth microdilution and time-kill methodology. The annual growth of medicinal research of stingless bees is plotted in Figure 2. The topic took flight in 2012 with four documents and a prolific peak in 2013 with the book Pot-honey. A legacy of stingless bees, and a further peak of productivity with the second book Pot-pollen in stingless bee melittology. A drop of productivity was observed, the following year of each book publication, and a possible effect of the COVID-19 pandemic in 2020. A spectacular recovery in 2021 shows the inner motivation of authors, with a steady growth in 2022 and 2023, a merit of the multidisciplinary teams of worldwide experts with scientific interest embracing medicinal research of tropical stingless bees.
2.1.1 Most productive authors
The top ten authors in Table 2 are from Argentina, Australia, Brazil, Malaysia, and Venezuela. Hilgert publishes on ethnomedicinal uses of stingless bees, Brooks is microbiologist of cerumen and propolis, Mustafa and Ahmad are veterinarians with papers in multiple subjects, and Vit is a biologist interested in quality control of pot-honey, pot-pollen, and propolis.
| Ranking | NP | Stingless bee medicinal research Author | Affiliation, city | Country |
|---|---|---|---|---|
| 1 | 4 | Hilgert, N.I. | Instituto de Biología Subtropical, CONICET, Facultad de Ciencias Forestales, Universidad Nacional de Misiones, Puerto Iguazú | Argentina |
| 2 | 3 | Brooks, P.R. | Faculty of Science, Health, Education and Engineering, University of the Sunshine Coast, Maroochydore | Australia |
| 3 | 3 | Mustafa, M.Z. | Hospital Universiti Sains Malaysia, Kubang Kerian 16150, Kelantan | Malaysia |
| 4 | 3 | Vit, P. | Food Science Department, Faculty of Pharmacy and Bioanalysis, Universidad de Los Andes, Mérida | Venezuela |
| 5 | 2 | Ahmad, H. | Department of Veterinary Preclinical Sciences, Faculty of Veterinary Medicine, Universiti Putra Malaysia, Serdang | Malaysia |
| 6 | 2 | Al Hatamleh, M.A.I. | Department of Immunology, School of Medical Sciences, Universiti Sains Malaysia, Kubang Kerian | Malaysia |
| 7 | 2 | Balestieri, J.P.B. | Research group on Biotechnology and Bioprospecting Applied to Metabolism (GEBBAM), Federal University of Grande Dourados, Dourados | Brazil |
| 8 | 2 | Beux, M.R. | Department of Food Engineering, Federal University of Parana (UFPR), Curitiba | Brazil |
| 9 | 2 | Campos, J.F. | Research Group on Biotechnology and Bioprospecting Applied to Metabolism (GEBBAM), Federal University of Grande Dourados, Dourados | Brazil |
| 10 | 2 | Carollo, C.A. | Laboratory of Natural Products and Mass Spectrometry, Federal University of Mato Grosso do Sul, Cidade Universitária, Campo Grande | Brazil |
2.1.2 Impact factor of top ten authors of stingless bee medicinal documents (2004–2023)
| Top ten authors | h-index | g-index | m-index | TC | NP | PY start |
|---|---|---|---|---|---|---|
| Mustafa MZ | 4 | 4 | 0.667 | 134 | 4 | 2018 |
| Brooks PR | 3 | 3 | 0.231 | 85 | 3 | 2011 |
| Vit P | 3 | 3 | 0.150 | 236 | 3 | 2004 |
| Ahmad H | 2 | 2 | 0.667 | 31 | 2 | 2021 |
| Al-Hatamleh MAI | 2 | 2 | 0.500 | 73 | 2 | 2020 |
| Balestieri JBP | 2 | 2 | 0.286 | 57 | 2 | 2017 |
| Campos JF | 2 | 2 | 0.286 | 57 | 2 | 2017 |
| Carollo CA | 2 | 2 | 0.286 | 57 | 2 | 2017 |
| Dos Santos CM | 2 | 2 | 0.286 | 57 | 2 | 2017 |
| Dos Santos EL | 2 | 2 | 0.286 | 57 | 2 | 2017 |
The most productive institutions worldwide in Table 4 show the Universiti Putra Malaysia in the top position with 7 publications, Instituto Tecnológico de Mérida with 6, Universidad de Los Andes, School of Medical Science, Universiti Sains Malaysia, and Universidad Nacional de Misiones with 5, as the top five institutions.
| Ranking | NP | Stingless bee medicinal research | Institution | Country |
|---|---|---|---|---|
| 1 | 7 | Universiti Putra Malaysia | Malaysia | |
| 2 | 6 | Instituto Tecnológico de Mérida | Mexico | |
| 3 | 5 | Universidad de Los Andes | Venezuela | |
| 4 | 5 | School of Medical Science, Universiti Sains Malaysia | Malaysia | |
| 5 | 5 | Universidad Nacional de Misiones | Argentina | |
| 6 | 5 | Universiti Sains Malaysia | Malaysia | |
| 7 | 5 | Universiti Sultan Zainal Abidin | Malaysia | |
| 8 | 3 | Universiti Kebangsaan Malaysia | Malaysia | |
| 9 | 3 | Universiti Teknologi Malaysia | Malaysia | |
| 10 | 3 | Universidad Nacional Autónoma de México | Mexico |
The top ten countries engaging with stingless bee medicinal research have 4 to 29 publications in the period (2004-2023), with Malaysia at the top (29), Brazil (19), Mexico (14), Australia and India (7), as the top five.
| Ranking | NP | Stingless bee medicinal research | Country |
|---|---|---|---|
| 1 | 29 | Malaysia | Malaysia |
| 2 | 19 | Brazil | Brazil |
| 3 | 14 | Mexico | Mexico |
| 4 | 7 | Australia | Australia |
| 5 | 7 | India | India |
| 6 | 6 | Argentina | Argentina |
| 7 | 5 | Venezuela | Venezuela |
| 8 | 4 | Indonesia | Indonesia |
| 9 | 4 | Kenya | Kenya |
| 10 | 4 | United Kingdom | United Kingdom |
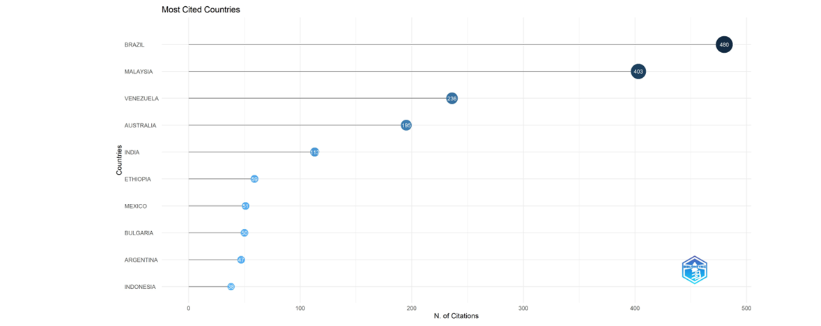
The most globally cited countries in the Bibliometrix plot were Brazil (480), Malaysia (403), Venezuela (236), Australia (195), India (113), Ethiopia (59), Mexico (51), Bulgaria (50), Argentina (47), and Indonesia (38).
In Table 6, the ranking of stingless bee medicinal research top ten sources selected by authors, hosted from 2 to 6 documents each. The most productive sources were the book Pot-Honey. A Legacy of Stingless Bees, followed by the Journal of Apicultural Research, the book Stingless Bee’s Honey from Yucatan: Culture, Traditional Uses and Nutraceutical Potential, Journal of Ethnobiology, and Ethnomedicine, and Estudios de Cultura Maya, as the top five sources. Journals h-index varied between 8 and 231, 2/8 journals are Quartile 1. The maximum impact score was 5.57 for the International Journal of Molecular Sciences ranked in the 10th position; note that alphabetical order is applied for the same number of publications. The metrics used for the journals are not available for the books in Resurchify, but the h-index was used as an impact factor in the following Bibliometrix plot.
| Ranking | NP | Stingless bee medicinal research | Sources (h-index, Quartile, impact score) | Publisher, country |
|---|---|---|---|---|
| 1 | 6 | Pot-Honey: A Legacy of Stingless Bees | Springer, New York, United States | |
| 2 | 5 | Journal of Apicultural Research | (h66, Q2, 2.08) Taylor and Francis Ltd., United Kingdom | |
| 3 | 4 | Stingless Bee’s Honey From Yucatan: Culture, Traditional Uses AND Nutraceutical Potential | ||
| 4 | 3 | Journal of Ethnobiology AND Ethnomedicine | (h 84, Q1, 4.27) BioMed Central Ltd., UK | |
| 5 | 2 | Estudios DE Cultura Maya | (h 8, Q2, 0.20) UNAM, Instituto de Investigaciones Filologicas, Mexico | |
| 6 | 2 | Ethnobiology AND Conservation | (h 18, Q2, 1.54) Universidade Federal Rural de Pernambuco, Brazil | |
| 7 | 2 | Food Research | (h15, Q3, 1.03) Malaysia | |
| 8 | 2 | Indian Journal OF Traditional Knowledge | (h40, Q2, 0.92) National Institute of Science Communication and Information Resources (NISCAIR), India | |
| 9 | 2 | Interciencia | (h 39, Q3, 0.40) Interciencia Association, Venezuela | |
| 10 | 2 | International Journal OF Molecular Sciences | (h 230, Q1, 5.57) Multidisciplinary Digital Publishing Institute (MDPI), Switzerland |
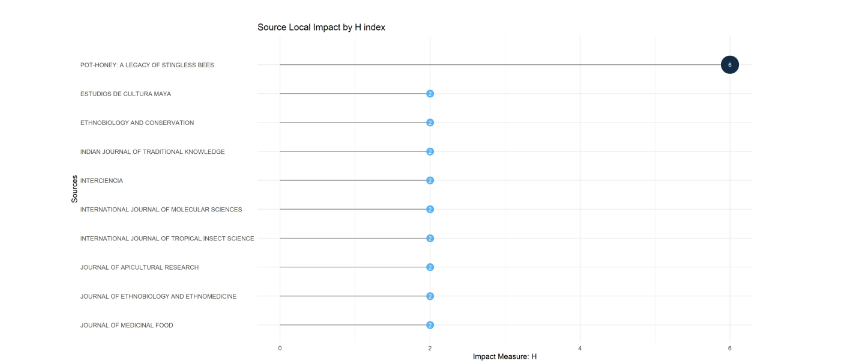
In the Bibliometrix plot, the local impact of medicinal stingless bee sources by h-index shows the highest impact (6) for the book Pot-Honey. A Legacy of Stingless Bees detached from the nine journal’s impacts (2) of the dataset. Table 6 corresponds with the most relevant sources Bibliometrix plot, not shown here. Note that the top ten Scopus-ranked sources by number of publications in Table 6 differ from the Bibliometrix-ranked sources by h-index in Figure 4.
Bibliometrix produced the most locally cited sources of top ten authors and number of articles. In Table 7, authors are considered sources. Local citations measure how many times an author (or a document) included in the dataset collection have been also cited by other authors in the dataset collection. Further Scopus search was done for each author to know the topic (stingless bee AND medicinal), total number of publication (NP), and total number of citations (NC). Six of these ten authors have zero topical publications, and therefore zero citation in this topic. The number of articles cited by other authors of the dataset (local collection) were lower than the topical citations in the Scopus database for Vit (89/234), Bankova (25/51), and Alves (19/21). Biluca had more citations from other authors in the dataset, than in the Scopus database (32/12), as well as other authors with zero topical citations.
| Sources | Document cited by another document of the dataset | NP | Scopus database (topical/total) | NC | Scopus database (topical/total) |
|---|---|---|---|---|---|
| Vit P | 89 | 3/66 | 234/1 698 | ||
| Biluca FC | 32 | 1/26 | 12/741 | ||
| Bogdanov S | 26 | 0/43 | 0/5 518 | ||
| Michener CD | 26 | 0/86 | 0/1 975 | ||
| Bankova V | 25 | 1/204 | 51/12 | ||
| Roubik DW | 24 | 0/138 | 0/6 297 | ||
| Crane E | 20 | 0/34 | 0/326 | ||
| Alves RRN | 19 | 1/266 | 21/7 917 | ||
| Kek S P | 17 | 0/6 | 0/365 | ||
| Cortopassi-Laurino M | 16 | 0/4 | 0/250 |
In Table 8, the funding sponsors for the stingless bee medicinal research are from Argentina (1), Australia (1), Austria (1), Brazil (2), Bulgaria (1), El Salvador (1), Malaysia (3). Up to 8 projects received support from two Brazilian funding agencies, from two Malaysian Ministry (7) and University (4), and the National Council of Science and Technology from El Salvador (3), as the top five funding sponsors.
| Ranking | NP | Stingless bee medicinal research | Funding sponsor | Country |
|---|---|---|---|---|
| 1 | 8 | Conselho Nacional de Desenvolvimento Científico e Tecnológico | Brazil | |
| 2 | 8 | Coordenação de Aperfeiçoamento de Pessoal de Nível Superior | Brazil | |
| 3 | 7 | Ministry of Higher Education, Malaysia | Malaysia | |
| 4 | 4 | Universiti Kebangsaan Malaysia | Malaysia | |
| 5 | 3 | Consejo Nacional de Ciencia y Tecnología | El Salvador | |
| 6 | 3 | Consejo Nacional de Investigaciones Científicas y Técnicas | Argentina | |
| 7 | 3 | Universiti Sains Malaysia | Malaysia | |
| 8 | 1 | Australia and New Zealand Banking Group Limited | Australia | |
| 9 | 1 | Austrian Development Agency | Austria | |
| 10 | 1 | Bulgarian Academy of Science | Bulgarian |
The Scopus database covers 4 broad supergroup areas (health sciences, life sciences, physical sciences, and social sciences) categorized into 27 subject areas that are automatically scrutinized in the left side menu by number of publications, and plotted as percentages of documents of a pie chart in the analyzed results report. In Table 9, the top ten studied subject areas on stingless bee medicinal research (2004–2023) had the following top five: Agricultural and Biological Sciences (30.6%), Biochemistry, Genetics and Molecular Biology (10.9%), Medicine (10.4%), Environmental Science (7.8%), and Social Sciences (5.7%).
| Ranking | NP | % | Stingless bee medicinal research | Subject area |
|---|---|---|---|---|
| 1 | 59 | 30.6 | Agricultural and Biological Sciences | |
| 2 | 21 | 10.9 | Biochemistry, Genetics and Molecular Biology | |
| 3 | 20 | 10.4 | Medicine | |
| 4 | 15 | 7.8 | Environmental Science | |
| 5 | 11 | 5.7 | Social Sciences | |
| 6 | 10 | 5.2 | Chemistry | |
| 7 | 10 | 5.2 | Pharmacology, Toxicology and Pharmaceutics | |
| 8 | 8 | 4.1 | Chemical Engineering | |
| 9 | 5 | 2.6 | Earth and Planetary Sciences | |
| 10 | 5 | 2.6 | Engineering |
2.1.2.1 Author’s keywords
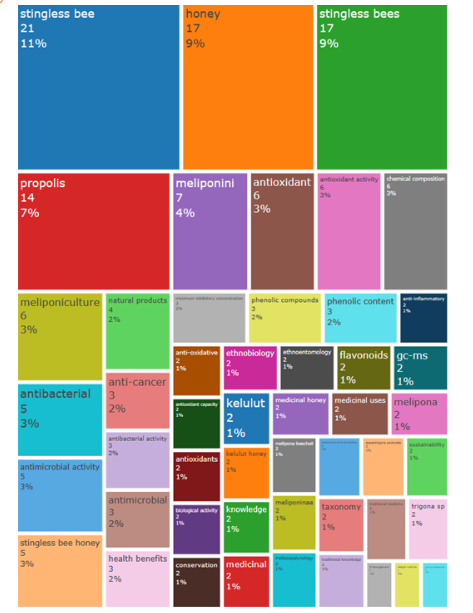
Compared with a word cloud, the tree map is structured in fields with visualized descending order of frequent keywords, both representations use bright colors. The frequencies of author’s keywords, and their percentages in the Figure 5 tree map are visible: stingless bee (21, 11%), honey (17, 9%), stingless bees (17, 9%), propolis (14, 7%), meliponini (7, 4%), antioxidant (6, 3%), antioxidant activity (6, 3%), chemical composition (6, 3%), meliponiculture (6, 3%), antibacterial (5, 3%), antimicrobial activity (5, 3%), stingless bee honey (5, 3%), natural products.
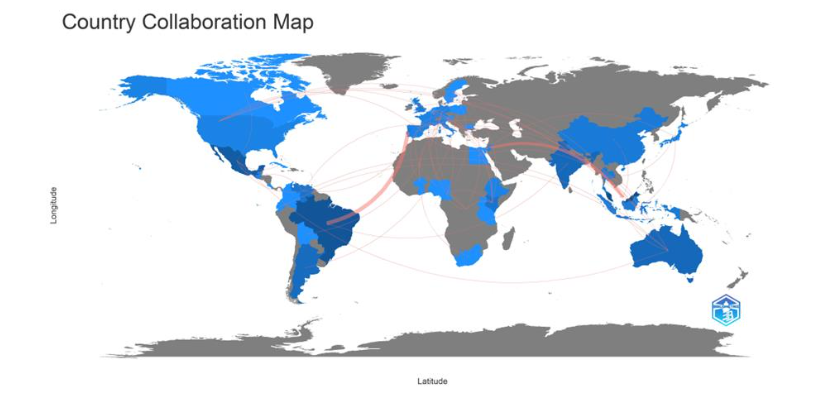
2.1.2.2 Country collaborative map
The collaboration between countries sharing publications on medicinal stingless bee research in the period 2004 to 2023 was visualized in Figure 6 using red connectors in a worldwide map. The frequencies of collaboration between two countries are available in the corresponding Excel file. The highest collaborative frequency was between Brazil and Portugal with 3 documents, and for Jordan and Malaysia with two documents.
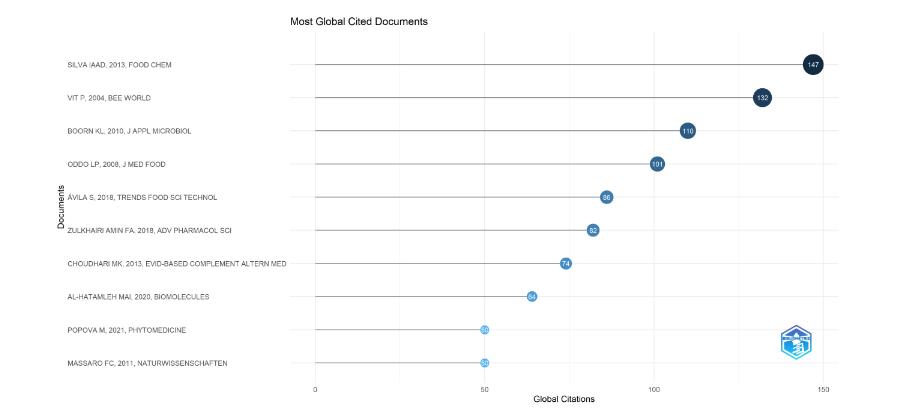
2.1.2.3 Most globally cited documents
The plot of most globally cited documents in Figure 7, shows the top ten documents cited from 147 to 50 times in publications of medicinal stingless bees (2004–2023), silva iaai 2013 food chem (147 citations), vit p 2004 bee world (132), boorn kl 2010 j appl microbiol (110), oddo lp 2008 j med food (101), avila s 2018 trends food sci technol (89), zulkhairi amin fa 2018 adv pharmacol sci (82), choudhari mk evid-based complement altern med 2013 (74), al-hatamaleh mai 2020 biomolecules (64), popova m 2021 phytomedicine (50), and massaro cf 2011 naturwissenschaften (50). The journal types were on food (2), medicine (2), a combined medicinal food (1), bees (1), biomolecules (1), microbiology (1), natural science (1), and pharmacy (1). All these documents were distributed in two clusters of the conceptual structure by factorial analysis in the next plot.
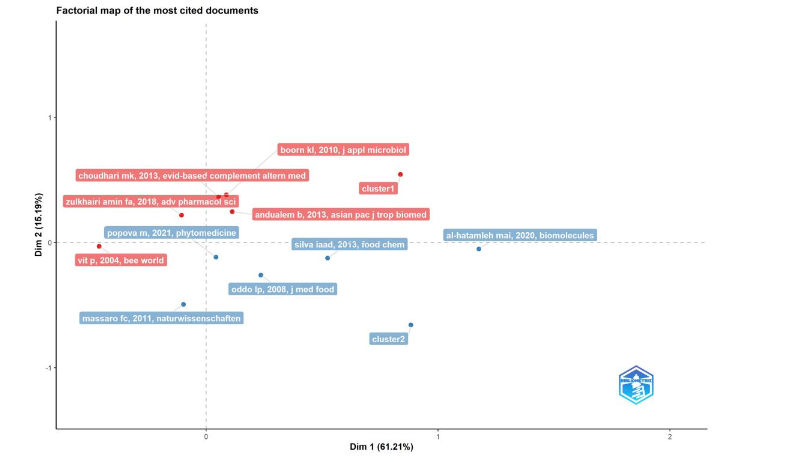
2.1.2.4 The conceptual structure for most cited articles
Concepts are embedded in a network of associations and contexts, having partial meaning based on links formed between them. Bibliometrix uses factorial analysis of correspondence analysis (CA) as a graphical method to compare variables. Scientific researchers use a conceptual framework to understand a problem and develop the analytical approach, a roadmap to conceptualize an outline that connects different ideas, concepts, and theories within a scientific field.
In this plot of research impact, the most cited medicinal stingless bee documents of the dataset (2004–2023) plotted in Figure 7 generated two clusters like principal component analysis (PCA) in a CA factorial map, clustering bipartite network of terms extracted from closeness of keyword, title or abstract fields. Factorial analysis is a data reduction technique. CA is used to represent the rows and columns of a two-way dimensional space, with Dim 1 explaining 61.21% of the variations, and Dim 2 explaining 15.81%. A red cluster 1 for boorn kl 2010, choudhari mk 2013, zulkhairi amin fa 2018, and ualem b 2013, and vit p 2004 was separated by the second dimension in the upper quadrants except vit p 2004. A blue cluster 2 for popova m 2021, silva iaai 2013, al-hatamleh mai 2020, oddo lp 2008, and massaro cf 2011. This cluster was separated by Dim 1 in the right quadrant except massaro cf 2011 located to the left, all of these documents were separated by Dim 2 in the lower quadrants.
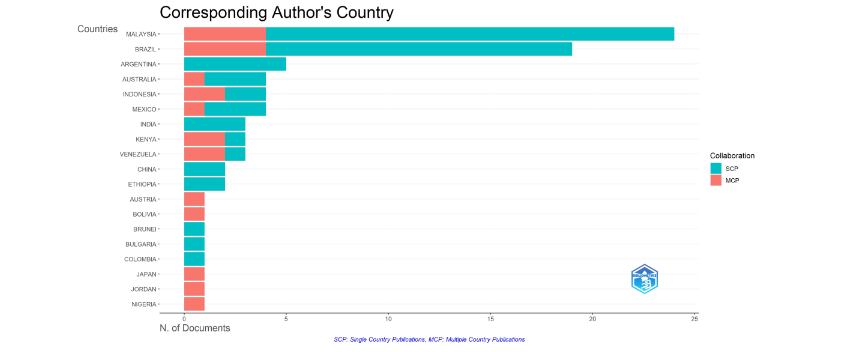
2.1.2.5 Simple and multiple country publications
Publications based on two categories of corresponding author simple country publication (SCP) and multiple country publication (MCP), are represented with bars of two colors for 19 countries in Figure 9 Bibliometrix plot. Argentina, India, China, Ethiopia, Brunei, Bulgaria, and Colombia publications of the dataset were intra-country SCP. Malaysia (24) and Brazil (19) published major number of documents with most SCP. Australia and Mexico also had more SCP than MCP, and Indonesia was balanced SCP-MCP in four documents. Kenya and Venezuela had more MCP than SCP in three documents. Austria, Bolivia, Japan, Jordan, and Nigeria had one document MCP, indicating compulsory multiple country interaction.
2.2 METHODOLOGY AND RESULTS OF THE BIBLIOMETRIC REVIEW ON STINGLESS BEES IN CLIMATE CHANGE
A second bibliometric review focused for a search of stingless bees in climate change. After reviewing the retrieved documents, we found that three of them only used climate change in the abstract, in general sentences such as: “Ecosystem services provided by such communities may be more greatly affected by environmental changes (anthropogenic activities and climate change) than are services provided by communities with greater functional redundancy”; “Used in folk medicine as antiseptic, antioxidant and antimicrobial agent, the composition is due to bee species, climate changes, local flora, and soil type”; and “Basic ecological knowledge is essential to inform agricultural management policies and to foresee preventable food scarcity problems, especially in view of climate change scenarios that predict drastic alterations in plant geographical distributions”. The dataset was retrieved with the Scopus database in the “TITLE-ABS-KEY” field query string the 1st December 2023. The operator AND was used for stingless bee AND climate change, with the operator AND NOT for guild, chayote and geopropolis, as follows.
TITLE-ABS-KEY ( stingless bee AND climate change AND NOT guild AND NOT chayote AND NOT geopropolis )
The first and most cited document of stingless bees and climate change was by Batalha-Filho et al., it was used to explain the Pleistocene estimated divergence time of Melipona quadrifasciata subspecies in Brazil, in a period of climatic and geomorphological changes in the Neotropics, causing subspecies distribution of Melipona quadrifasciata quadrifasciata to the south, and Melipona quadrifasciata anthidioides to the north. The second document was on the same species of stingless bee, a key pollinator of the Atlantic flora in Brazil, and approach to mitigate the effect of climate change in habitat fragmentation by identifying key conservation areas and strategies after a forecast to year 2080.
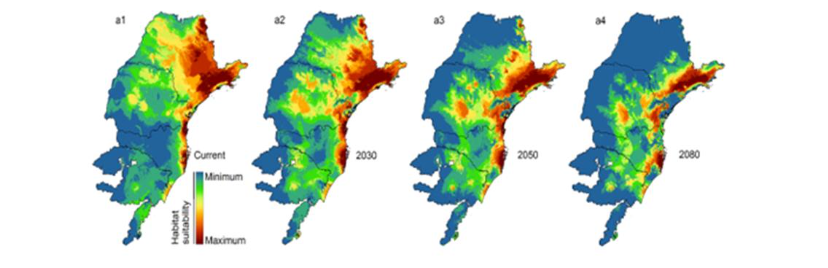
The main information of bibliometric descriptors is presented in Figure 11.1 including publications from the first retrieved document since 2010 to date in 2023, almost two decades. The 25 documents of the dataset were published in English. Three documents were excluded with the AND NOT operator because although climate change was in the abstracts, the investigations were not about climate change.
A total of 19 sources were used to disseminate the research on stingless bees and climate change. The metrics and publishers of the most prolific journals were Apidologie (5 documents, h-index 96, Quartile 1, impact factor 2.41, Springer Science + Business Media, United States), PLoS ONE (2 documents, h-index 404, Quartile 1, impact factor 3.75, Public Library of Science, United States), and Regional Environmental Change (2 documents, h-index 82, Quartile 2, impact factor 4.30, Springer Verlag, Germany).
The types of retrieved documents were 15 articles, one book chapter, one conference papers, and 4 reviews, all published in English. The first and most cited document was published by Batalha-Filho et al. in the journal Apidologie. Leading countries were Brazil (12), United States (4), Indonesia (3); Australia, Colombia, Kenya, and Thailand with 3 documents each, and one document for Argentina, Botswana, and China. The top seven authors published three documents each, and two the last three authors. Universidade de São Paulo led with 5 publications, five institutions with three, and two publications for the last in the top ten list. The top five Scopus subject areas of research on stingless bees and climate change were Agricultural and Biological Sciences (44.4% of the documents), Environmental Sciences (19.4%), Biochemistry, Genetics and Molecular Biology (13.9%), Multidisciplinary (8.3%), Earth and Planetary Sciences (5.6%), followed by Chemistry (2.8%), Computer Science (2.8%), and Engineering (2.8%).

Research for publications was sponsored by ten top agencies: Coordenação de Aperfeiçoamento de Pessoal de Nível Superior CAPES (Brazil), Conselho Nacional de Desenvolvimento Científico e Tecnológico CNPq (Brazil), Australian Research Council (Australia), Fundacao de Amparo a Pesquisa do Estado de Sao Paulo FAPESP (Brazil), National Science Foundation (United States), University of Kansas (United States), Bayer CropScience (Germany), Biotechnology and Biological Sciences Research Council (United Kingdom), Department of Atomic Energy, Government of India (India), and Direktion fur Entwicklung und Zusammenarbeit (Switzerland).
Correlations and classifications of authors’ keywords were investigated with multivariate graphical tools. Metrics of scientific literature and other Bibliometrix plots, such as topical dendrograms by Hierarchical Cluster Analysis (HCA), word clouds, country collaboration maps, most global cited documents, co-authors networks, and corresponding authors country publications were illustrated in Figure 11.
The metrics for 25 documents in the scientific literature of stingless bees and climate change (2010:2023) cited 1863 references, showed an average citation per document of 13.4%, used 89 author’s keywords, had an average author number of 6.52, and had 32.00% international co-authorship. Two clusters were produced in the topical dendrogram by HCA after factorial analysis of author’s keywords. The red cluster grouped traditional knowledge, traditional and recent beekeeping, propolis, medicinal honey, bumble bees, honey hunters, honeybees, pests, and social bees. The large blue cluster was visualized with two smaller clusters. The left blue cluster was mainly based on thermal biology, tetragonula, critical thermal maxima, austroplebeia, chill coma, from 30 and further branches with keywords from other papers; for example, single nucleotide polymorphism, local adaptation, isolation by resistance; and environmental associations, gene flow, species distribution modelling, hybridization, climate niche, habitat suitability.
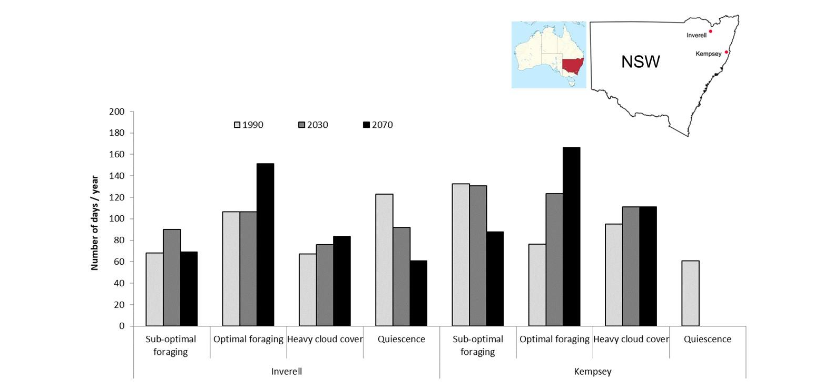
The country collaborative map showed red line interactions with at least two papers shared between Colombia and the United States. A word cloud plot visualized higher frequencies of author’s keywords with larger letters in central positions, stingless bees (7) was the most frequent, then, climate change (6), pollination (4), honey bees (3), stingless bee (3), sustainability (3), conservation (2), deforestation (2), ecological niche modeling (2), meliponiculture (2), meliponini (2), pests (2), melipona (2), social bees (2), and one for the remaining 35 keywords of the plot, anthropogenic change, biodiversity loss and biotic interaction among others. The most global cited documents were ten documents cited from 77 times (batalha-filho 2010) to 14 (gonzalez2021), in five different journals Apidologie, Food Chemistry, Evol Appl, Perspect Ecol Conserv, PLoS ONE, and one book chapter Asian Beekeeping in the 21st Century.
3. Scanning medicinal stingless bee resources, research, and efforts for wellness
3.1 SIGNIFICANCE OF THE ENTOMOLOGICAL BIODIVERSITY OF STINGLESS BEES IN APITHERAPY
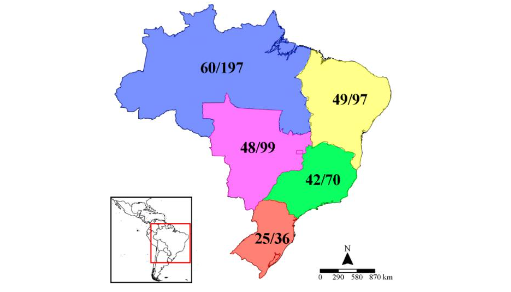
3.1.1 Richness of stingless bees in Brazil as resources for meliponiculture
The conservation of stingless bee biodiversity is of paramount importance for this natural resource with medicinal traditional uses. Brazil has a great wealth of stingless bees, which makes this country stand out from the rest of the Neotropical region, since the last survey carried out by Nogueira, counted a total of 259 species, distributed in the five regions of this country. The Amazon region with 197 of these species, 128 of which occur in the Amazonas state. Although there are many species in Brazil, the most familiar for management are used for honey production, the vast majority belonging to the genus Melipona Illiger, as M. seminigra, M. interrupta, M. flavolineata, M. subnitida, M. scutellaris and M. quadrifasciata. Species from other genera that also deserve to be highlighted in terms of productivity for Brazil are Tetragonisca angustula, Tetragona clavipes, and species of Scaptotrigona.

In a proportion of 95 managed stingless bees over the total of 259 species of stingless bees in Brazil, around half of these species are used for meliponiculture in the Northeast and Central-West regions, and a little more than half in the Southeast and South regions. On the contrary, the richest North region uses a third of these resources, with the remaining 137 species of stingless bees unexploited or not selected for meliponiculture, or which do not have data about nesting.
3.1.2 Managed stingless bee species in Brazil
Good practices of sustainable stingless bee keeping do not represent a risk for the stingless bee biodiversity conservation, on the contrary, more colonies are kept, and divided to increase productivity. However, feral nest hunting is a common practice in rural areas; moving colonies from their natural substrates to stingless bee hives. On the other hand, pot-honey hunting is more destructive, its intensity may affect the natural populations of stingless bees, but is the traditional method especially for non-domesticated underground species. For example, developing conventional stingless bee farming is an opportunity to increase availability of pot-honey in demand for ethnomedicinal use in Baringo County, Kenya. Some Brazilian stingless bee species used in meliponiculture are illustrated with the images and size of the Brazilian Association of Bee Studies.
| No. | Stingless bee species | Brazilian states |
|---|---|---|
| 1 | Trigonisca pediculana | CE, MA, PB, PA, BA, PI, AM, RO, RR, PE, MG |
| 2 | Trigonisca duckei | AM, PA, CE, MA, MT, RR |
| 3 | Trigona pallens | AC, AM, AP, PA, RO, RR, TO, MA, GO, CE, DF, MT, PI |
| 4 | Trigona cilipes | AC, AM, AP, PA, RO, MT, RR, MA, GO |
| 5 | Tetragonisca weyrauchi | AC, RO, MT |
| 6 | Tetragonisca fiebrigi | MS, RS, SP, PR, SC, MT |
| 7 | Tetragonisca angustula | AM, AP, PA, RR, BA, CE, MA, PB, PE, GO, MS, MT, ES, MG, RJ, SP, PR, RS, SC, TO, DF, AC, PI, RO |
| 8 | Tetragona quadrangula | GO, MA, MG, MT, PA, TO |
| 9 | Tetragona kaieteurensis | AM, PA, RR |
| 10 | Tetragona goettei | AC, AM, PA, MT, RO, MA, RR |
| 11 | Tetragona essequiboensis | AM, RO |
| 12 | Tetragona clavipes (syn. T. elongata) | AC, AM, RO, AP, RR, PA, MA, PI, MT, TO, BA, GO, DF, MG, MS, ES, RJ, SP, PR, SC, RS |
| 13 | Schwarziana quadripunctata | BA, GO, ES, MG, RJ, SP, PR, RS, SC, DF |
| 14 | Scaura longula | AM, AP, PA, MA, GO, MT, MG, SP, AC, BA, MS, RO, RR |
| 15 | Scaura latitarsis (syn. Scaura tenuis) | AC, AM, MT, PA, RO, RR |
| 16 | Scaptotrigona xanthotricha | BA, ES, MG, SP, SC, PR, RJ |
| 17 | Scaptotrigona tubiba | SP, MG |
| 18 | Scaptotrigona tricolorata | RO, MT, AM |
| 19 | Scaptotrigona postica | PA |
| 20 | Scaptotrigona polysticta | AC, PA, RO, TO, MA, GO, MT, MG, SP, AM, DF, PI |
| 21 | Scaptotrigona depilis | MS, MG, SP, |
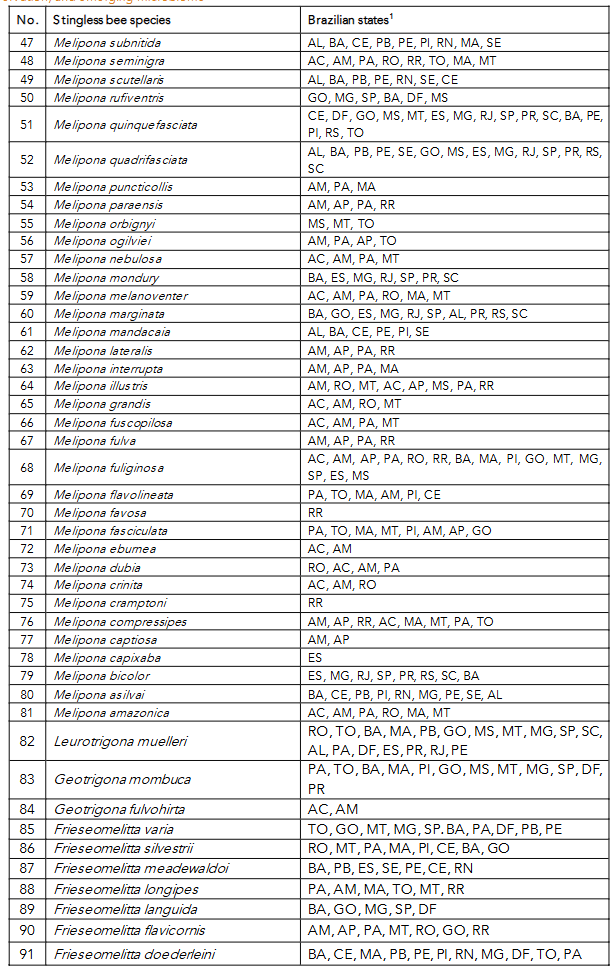
92 — Friesella schrottkyi — ES, MG, SP, PR
93 — Duckeola ghilianii — AM, AP, PA, MT, RO
94 — Cephalotrigona femorata — AM, PA, RO, MA, AC, AP, MT, TO
95 — Cephalotrigona capitata — AP, PA, CE, MT, ES, MG, SP, PR, SC, BA, RJ, MS, AL, GO, RO
¹North region: Acre (AC), Amazonas (AM), Amapá (AP), Pará (PA), Rondônia (RO), Roraima (RR) and Tocantins (TO). Midwest region: Distrito Federal (DF), Goiás (GO), Mato Grosso (MT) and Mato Grosso do Sul (MS). Southeast region: Espírito Santo (ES), Minas Gerais (MG), Rio de Janeiro (RJ) and São Paulo (SP). Northeast region: Alagoas (AL), Bahia (BA), Ceará (CE), Maranhão (MA), Paraíba (PB), Pernambuco (PE), Piauí (PI), Rio Grande do Norte (RN) and Sergipe (SE). South region: Paraná (PR), Santa Catarina (SC) and Rio Grande do Sul (RS).
Source: BRASIL¹
Some stingless bee species like Scaptotrigona postica (PA), Plebeia wittmanni (RS), Melipona tumupasae (AC), Melipona favosa (RR), Melipona cramptoni (RR), and Melipona capixaba (ES) were managed only in one state, in contrast with Tetragonisca angustula and Tetragona clavipes widely selected for meliponiculture in 24 and 21 of the 26 Brazilian states respectively.
The map of managed stingless bees was used as a geospatial product and a basis for assessing the risk of extinction of Brazilian fauna species². It was prepared through long discussions and by expert teams, and although there is no consensus on some taxonomic identifications and occurrence records, see the occurrence of the same species in Nogueira³¹; it shows promise in trying to organize the biodiversity to prevent the illegal transport of species outside the political boundaries of their occurrence, as is the case in states (Table 15). The illegal transport of nests to places where they do not naturally occur can cause a series of problems such as the transmission of diseases, increased competition for resources with native species, and genetic modification of wild and managed populations, which can compromise the permanence and maintenance of both native species of this new location, as well as species that came from a different location³⁴. In the long term, these environmental imbalances may harm both bee biodiversity and local stingless beekeepers, as species extinction may occur, especially when there is hybridization between species due to the disturbance of ecological barriers promoted by human action³⁵,³⁶. The dangers of interbreeding are well-known threats to wildlife³⁷, both for bees and extinction of rare plant species³⁶.
Direct and online interviews were focused on bees and beekeepers of 25 indigenous provinces who understand the importance of traditional stingless bee keeping in and cultural practices. Data of 272 beekeepers revealed 19 species of stingless bees are reared, mostly Tetragonula laeviceps, and climate change was one of the obstacles besides pesticides, demanding a strategy for stingless bee keeping and bee conservation to adapt and mitigate environmental changes on climate and land-use³⁸.
2.3 FLORAL AND EXTRAFLORAL NECTAR, FLORAL POLLEN, RESIN, GUM AND LATEX PLANT NATURAL RESOURCES
Tropical stingless bees nest, feed and interact with tropical plants. Tropical bee flora is represented by biodiverse Fabaceae, Asteraceae, Rubiaceae, Malvaceae, Lamiaceae, Euphorbiaceae, Arecaceae, Poaceae, Apocynaceae, and Melastomataceae as the most visited of 221 plant families³⁹. Stingless bee preferences of available tropical resources, pollen create enormous combinations of pot-honey, pot-pollen, cerumen and propolis variables, explained by natural history⁴⁰ and investigated as a healing matrix. For example, secondary metabolites like flavonoids originate from the foraged plants, and having luteolin derivatives as active phytochemicals in ocular cataract models⁴¹ have a significance for the nature of the stingless bee material, the biomolecular richness, the relationship with the environmental resources, and its biodiverse conservation.
Standard terminology for palynology is used for morphological descriptions of pollen grains⁴² and the major pollen grains in the pollen spectrum⁴³. Pollen identifications at plant family, genus and even species, are assigned after comparisons with pollen atlases and pollen reference collections. The taxonomic status of botanical taxa is systematically updated to avoid synonyms.Garden database available online⁴⁴. Extrafloral nectar causes poor pollen spectra of honey because it contains less pollen than floral nectar.
Plant resins sometimes comprise gums, latex and resin exudates from different parts of plants: bark like Dalbergia ecastaphyllum Fabaceae, and Schinus terebinthifolius Anacardiaceae; buds like Populus spp. Salicaceae; flowers like Clusia major and Clusia minor, Clusiaceae, and Dalechampia spp. Euphorbiaceae; fruits like Corymbia torelliana Myrtaceae, and Coussapoa asperifolia Cecropiaceae; and whole plant like Artocarpus heterophyllus Moraceae, Merremia umbellata Convolvulaceae. Anacardiaceae and Fabaceae are two plant resin source families for stingless bees in Brazil, China, Colombia, and India; Euphorbiaceae is common in Brazil, Colombia, and India; and Clusiaceae is a Neotropical source in Brazil, Colombia, and Venezuela⁴⁵.
Plant resins use has an evolutionary meaning of sociality in stingless bees⁴⁶. Diverse plant resin–based functions such as social immunity, cuticular hydrocarbon chemical profiles, defense, and microbial communities are associated with stingless bees⁴⁰,⁴⁶. Cerumen is a vital material in stingless bee nest architecture, composed by admixtures of beeswax and plant resins. For this reason, stingless bee foragers prioritize resin collection and reduce pollen foraging after hive splitting, as observed for the Australian Tetragonula carbonaria⁴⁷.
In Fig. 14, acetolyzed pollen grains used to identify the Coffea arabica unifloral Tetragonisca angustula honey from Costa Rica are illustrated⁴⁸. Taxa were identified as nectariferous sources, and polleniferous, considered contaminants of honey because they do not secrete nectar, the raw material transformed into honey.
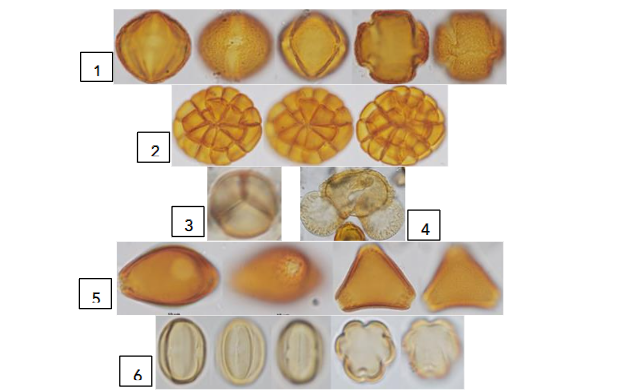
Fig. 14 Pollen grains of a unifloral pot-honey with nectariferous and polleniferous taxa. Nectariferous 1. Coffea arabica, Rubiaceae produces nectar. A frequency of 54.3% of the pollen spectrum featured this as unifloral coffee honey⁴⁸. Polleniferous 2. Polyads of Inga sp. Fabaceae–Caesalpinioideae, 3. Mimosa sp., Fabaceae–Caesalpinioideae, and single grains or monads of 4. Pinus sp. Pinaceae, 5. Paullinia sp. Sapindaceae, and 6. Miconia sp. Melastomataceae. Pollen from nectarless plants is considered contaminant pollen in melissopalynology. Photos: © E. Moreno After: Moreno et al.⁴⁸.
A unifloral honey has 45% pollen of one taxa, with exceptions for over-represented and under-represented pollen types⁴⁹. Floral pollen, Apis mellifera bee-bread and stingless bee pot-pollen are obviously pollen grains. A recent controversy has raised for a traditional palynological analysis of propolis, the pollen landed on plant resins, latex or gums, collected and processed into propolis or bee glue. Layek et al.⁵⁰ found that the pollen spectra of Tetragonula iridipennis from India was not accurate to identify the floral and non-floral sources of cerumen and propolis, because this nest material is not seasonal like floral nectar and floral pollen when in bloom.
3.3 DEMONSTRATED BIOMOLECULES, BIOLOGICAL ACTIVITIES, AND PUTATIVE THERAPEUTIC PROPERTIES OF POT-HONEY, POT-POLLEN, CERUMEN AND PROPOLIS OF THE STINGLESS BEE NEST
Flavonoids and polyphenols — and biological activities — antimicrobial and antioxidant — added values to the medicinal uses of stingless bee products, expanding with the putative therapeutic actions that would deserve more bioassays and clinical trials to support apitherapy. The stingless bee species were carefully retrieved for each study. It is recommended to inform the species in the abstracts. The corresponding entomological authority, institution, and collection where the entomological specimens are deposited, is mandatory in the materials and methods. Continuous updating of names arises with research, and this fact also deserves the attention of melittologists. Valid names should be informed as suggested by M. Engel (P. Vit, personal communication). For example, Axestotrigona ferruginea (cited as Meliponula ferruginea by Popova et al.⁵¹)
It is not our aim to provide ranges of concentrations and IC₅₀ for this table, but to summarize the chronological input for studies on biomolecules and biological activities, projecting therapeutic properties, showing the first and the last publications in each category. Timespan years and (number of publications/number of nest materials) varied as follows for the alphabetical order used in Table 11:
Biomolecules Flavonoids 1993–2023 (16/2); Polyphenols 1993–2023 (16/1);
Biological activities Anti-atherogenic 2019–2022 (3/2); Anticancer 2013–2020 (9/4); Anticataract 1997–2008 (4/1); Antihyperglycemic 2015–2023 (8/2); Antimicrobial 2013–2023 (10/4); Antioxidant 2006–2023 (22/4); Anti-inflammatory 2011–2023 (15/4); Antinociceptive 2014–2022 (3/2); Antiprofilactive 2016–2018 (2/3); Chemopreventive 2016 (2/3), Hypocholesterolemic 2021 (1/3); and Modulator of gut microbiota 2019–2022 (3/1).
Table 11. Selected stingless bees from some studies on active biomolecules and biological activities, first and last publications
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| Flavonoids | Frieseomelitta varia, Melipona compressipes, Melipona favosa, Paratrigona anduzei, Scaptotrigona depilis | Venezuela | 1993 | – | – | – | Tomás-Barberán et al.⁵² |
| Geotrigona sp., Tetragonisca fiebrigi | Ecuador | 2023 | – | – | Ferreirat et al.⁵³ | – | |
| Polyphenols | Frieseomelitta varia, Melipona compressipes, Melipona favosa, Paratrigona anduzei, Scaptotrigona depilis | Venezuela | 1993 | – | – | – | Tomás-Barberán et al.⁵² |
| Geotrigona sp., Tetragonisca fiebrigi | Ecuador | 2023 | – | – | Ferreirat et al.⁵³ | – | |
| Biological activities | Anti-atherogenic | ||||||
| Heterotrigona itama | Malaysia | 2019 | – | Othman et al.⁵⁴ | – | – | |
| Heterotrigona itama | Malaysia | 2022 | – | Zakaria et al.⁵⁵ | – | – | |
| Anticancer | Tetragonula spp. | India | 2013 | – | – | – | Choudhari et al.⁵⁶ |
| Heterotrigona itama | Malaysia | 2020 | – | Mahmood et al.⁵⁷ | – | – | |
| Anticataract | Melipona favosa | Venezuela | 2002 | – | Vits⁵⁸ | – | – |
| Commercial flavonoids present in honey¹ | Wales, UK | 2008 | – | – | – | – | |
| Antihyperglycemic | Leipidotrigona ventralis, Leipidotrigona | Thailand | 2015 | – | – | – | Vongsak et al.⁵⁹ |
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| (continuation from previous row) | terminata, Tetragonula pagdeni | ||||||
| Anti-inflammatory | Heterotrigona itama | Malaysia | 2023 | – | Cheng et al.⁶⁰ | – | – |
| Tetragonula sapiens | Indonesia | 2023 | – | – | – | Farida et al.⁶¹ | |
| Tetragonula carbonaria | Australia | 2011 | – | – | Massaro et al.²⁴ | – | |
| Tetragonula biroi | Indonesia | 2023 | – | – | – | Arung et al.⁶² | |
| Heterotrigona itama, Tetragonula reepeni, Tetragonula testaceitarsis, Tetragonula fuscobalteata, Tetragonula iridipennis, Tetragonula pagdeni | Indonesia | 2023 | – | Naibaho et al.⁶³ | – | – | |
| Melipona seminigra | Brazil | 2013 | – | da Silva et al.⁶⁴ | – | – | |
| Antimicrobial | Axestotrigona ferruginea, Axestotrigona togoensis, Melipelbea beccarii, Hypotrigona gribodoi, Dactylurina schmidti, Plebeina armata | Tanzania | 2023 | – | Mduda et al.⁶⁵ | – | – |
| Antinociceptive | Melipona subnitida | Brazil | 2014 | – | – | – | Silva et al.⁶⁶ |
| Tetragonisca fiebrigi | Argentina | 2022 | – | Salomon et al.⁶⁷ | – | – | |
| Tetragonula carbonaria | Australia | 2013 | – | – | – | Vit et al.⁶⁷ | |
| Antiproliferative | Melipona fasciculata, Melipona rufiventris, Melipona scutellaris, Melipona subnitida, Scaptotrigona polysticta | Brazil | – | – | – | – | |
| Frieseomelitta nigra, Melipona beecheii, Melipona fasciata, Melipona solani, Scaptotrigona hellwegeri, Scaptotrigona mexicana | Mexico | – | – | – | – | ||
| Melipona favosa | Venezuela | – | – | – | – | ||
| Antioxidant | Geniotrigona thoracica, Heterotrigona itama | Malaysia | 2018 | – | Ismail et al.⁶⁸ | Ismail et al.⁶⁸ | – |
| Melipona subnitida | Brazil | 2006 | – | – | Silveira et al.⁶⁹ | – | |
| Geotrigona sp., Tetragonisca fiebrigi | Ecuador | 2023 | – | – | Ferreirat et al.⁵³ | – |
Table 12. Active biomolecules and biological activities of pot-honey, pot-pollen, cerumen, and propolis.
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| Flavonoids | Frieseomelitta varia, Melipona compressipes, Melipona favosa, Paratrigona anduzei, Scaptotrigona depilis | Venezuela | 1993 | – | – | – | Tomás-Barberán et al.⁵² |
| Melipona subnitida | Brazil | 2006 | – | – | Silva et al.⁶⁹ | – | |
| Melipona spp. | Venezuela | 2011 | Truchado et al.¹³⁷ | – | – | – | |
| Tetragonisca angustula | Venezuela | 2013 | Pérez-Pérez et al.¹³⁸ | – | Pérez-Pérez et al.¹³⁸ | Pérez-Pérez et al.¹³⁸ | |
| Melipona seminigra | Brazil | 2013 | da Silva et al.⁶⁴ | – | – | – | |
| Melipona quadrifasciata, Tetragonula clypearis, Scaptotrigona spp. | Brazil | 2017 | – | – | Pazin et al.¹³⁹ | – | |
| Melipona subnitida | Brazil | 2018 | Tukistah et al.¹⁴⁰ | – | – | de Souza et al.⁶⁷ | |
| Geniotrigona thoracica, Heterotrigona itama | Malaysia | 2018 | – | – | – |
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| Heterotrigona erythrogastra, Tetrigona apicalis, Heterotrigona itama, Geniotrigona thoracica | Malaysia | 2019 | – | – | – | Asem et al.⁸⁰ | |
| Tetragonula biroi | Philippines | 2019 | – | Belina-Aldemita et al.¹⁴² | – | – | |
| Melipona quadrifasciata, Melipona asilvai, Melipona subnitida, Melipona scutellaris | Brazil | 2019 | – | Oliveira et al.¹⁴³ | – | – | |
| Scaptotrigona bipunctata, Melipona marginata, Tetragonisca angustula, Trigona hypogea, Melipona quadrifasciata, Tetragona clavipes | Brazil | 2020 | – | Biluca et al.¹⁴⁴ | – | – | |
| Heterotrigona itama | Malaysia | 2020 | – | Majid et al.¹⁴⁵ | – | – | |
| Melipona seminigra | Brazil | 2021 | – | – | – | Rebelo et al.⁷³ | |
| Tetrigona apicalis, Tetrigona binghami, Heterotrigona fimbriata | Malaysia | 2021 | – | – | – | Syed Salleh et al.⁸² | |
| Geotrigona sp., Tetragonisca fiebrigi | Ecuador | 2023 | – | – | Ferreirat et al.⁵³ | – | |
| Tetragonisca fiebrigi | Brazil | – | – | – | – | – |
Polyphenols
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| Polyphenols | Frieseomelitta varia, Melipona compressipes, Melipona favosa, Paratrigona anduzei, Scaptotrigona depilis | Venezuela | 1993 | – | – | – | Tomás-Barberán et al.⁵² |
| Tetragonula carbonaria | Australia | 2011 | – | – | Massaro et al.²⁴ | – | |
| Tetragonisca angustula | Venezuela | 2013 | Pérez-Pérez et al.¹³⁸ | – | Pérez-Pérez et al.¹³⁸ | Pérez-Pérez et al.¹³⁸ | |
| Melipona seminigra | Brazil | 2013 | – | da Silva et al.⁶⁴ | – | – | |
| Melipona fasciculata | Brazil | 2014 | – | – | – | Dutra et al.¹⁰⁶ | |
| Melipona quadrifasciata, Tetragona clavipes, Scaptotrigona spp. | Brazil | 2017 | – | – | Pazin et al.¹³⁹ | – | |
| Melipona subnitida | Brazil | 2018 | – | – | de Souza et al.¹⁴⁰ | – | |
| Geniotrigona thoracica, Heterotrigona itama, Heterotrigona erythrogastra | Malaysia | 2018 | – | Tukishta et al.¹⁴¹ | – | – | |
| Tetrigona apicalis, Heterotrigona itama, Geniotrigona thoracica | Malaysia | 2019 | – | – | – | Asem et al.⁸⁰ | |
| Melipona quadrifasciata, Melipona asilvai | Brazil | 2019 | – | Oliveira et al.¹⁴³ | – | – |
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| Melipona subnitida, Melipona scutellaris | Brazil | 2020 | – | Biluca et al.¹⁴⁴ | – | – | |
| Scaptotrigona bipunctata, Melipona marginata, Tetragonisca angustula, Trigona hypogea, Melipona quadrifasciata, Tetragona clavipes | Brazil | 2020 | – | Biluca et al.¹⁴⁴ | – | – | |
| Heterotrigona itama | Malaysia | 2020 | – | Majid et al.¹⁴⁵ | – | – | |
| Melipona seminigra | Brazil | 2021 | – | – | – | Rebelo et al.⁷³ | |
| Tetrigona apicalis, Tetrigona binghami, Homotrigona fimbriata | Malaysia | 2021 | – | – | – | Syed Salleh et al.⁸² | |
| Geotrigona sp., Tetragonisca fiebrigi | Ecuador | 2023 | – | – | Ferreira et al.⁵³ | – | |
| Tetragonula laeviceps | Thailand | 2023 | – | – | Iesa et al.¹⁴⁷ | – |
Biological activities
Anti-atherogenic
| Stingless bee taxa | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|
| Heterotrigona itama | Malaysia | 2019 | – | Othman et al.⁵⁴ | – | – |
| Geniotrigona thoracica | Malaysia | 2020 | – | Mohd Suib et al.¹⁴⁸ | – | – |
| Heterotrigona itama | Malaysia | 2022 | – | Zakaria et al.⁵⁵ | – | – |
Anticancer
| Stingless bee taxa | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|
| Tetragonula spp. | India | 2013 | – | – | – | Choudhari et al.⁵⁶ |
| Homotrigona apicalis, Tetragonula fuscibasis, Tetragonula fuscobalteata, Wallacetrigona incisa | Indonesia | 2014 | – | Kustiawan et al.¹⁴⁹ | – | Kustiawan et al.¹⁴⁹ |
| Tetragonula laeviceps | Thailand | 2015 | – | – | – | Nugitrangson et al.¹⁵⁰ |
| Lepidotrigona terminata | Malaysia | 2016 | – | Omar et al.⁷¹ | – | – |
| Melipona orbignyi | Brazil | 2017 | – | – | – | dos Santos et al.¹⁵¹ |
| Heterotrigona itama | Malaysia | 2019 | – | Ahmad et al.¹⁵² | – | – |
| Tetragonula biroi | Philippines | 2019 | – | – | – | Desamero et al.¹⁵³ |
| Heterotrigona itama | Malaysia | 2020 | – | Mahmood et al.⁵⁷ | – | – |
| Homotrigona fimbriata, Heterotrigona itama, Heterotrigona bakeri, Tetragonula sarawakensis, Tetragonula testaceitarsis, Tetragonula fuscobalteata, Tetragonula laeviceps | Indonesia | 2021 | – | Arung et al.¹⁵⁴ | – | Arung et al.¹⁵⁴ |
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|
| Anticataract | Melipona favosa, Tetragonisca angustula, Commercial luteolin derivatives present in honey¹ | Venezuela | 1997 | Vit¹⁵⁵ | – | – | – |
| Commercial luteolin derivatives present in honey¹ | Wales, UK | 2001 | – | – | – | – |
| Melipona favosa | Venezuela | 2002 | Vit⁵⁸ | – | – | – |
| Melipona favosa, Scaptotrigona mexicana, Tetragonisca angustula Commercial luteolin derivatives present in honey | Brazil, Mexico, Venezuela | 2004 | Vit et al.⁹ | – | – | – |
| Commercial flavonoids present in honey² | Wales, UK | 2008 | – | – | – | – |
| Lepidotrigona ventralis, Lepidotrigona terminata, Tetragonula pagdeni | Thailand | 2015 | – | – | – | Vongsak et al.⁵⁹ |
| Geniotrigona thoracica | Malaysia | 2017 | – | Abdul Aziz et al.¹⁵⁶ | – | – |
| Antihyperglycemic | Heterotrigona itama, Trigona apicalis | Malaysia | 2018 | – | Na et al.¹⁵⁷ | – | – |
| Tetragonula sapiens | Indonesia | 2019 | – | Pujiarahayu et al.¹⁵⁸ | – | – |
| Tetragonula biroi, Tetragonula leytebesis | Indonesia | 2019 | – | Rahmawati et al.¹⁴⁷ | – | – |
| Heterotrigona itama | Malaysia | 2020 | – | Ali et al.¹⁶⁰ | – | – |
| Heterotrigona itama | Malaysia | 2023 | – | Cheng et al.⁶⁰ | – | – |
| Tetragonula sapiens | Indonesia | 2023 | – | – | – | Farida et al.⁶¹ |
| Anti-inflammatory | Tetragonula carbonaria | Australia | 2011 | – | – | Massaro et al.²⁴ | – |
| Tetragonisca fiebrigi | Brazil | 2015 | – | – | – | Campos et al.¹⁶¹ |
| Tetragonula carbonaria | Australia | 2016 | – | – | – | Hamilton et al.¹⁶² |
| Melipona orbignyi | Brazil | 2017 | – | – | – | dos Santos et al.¹⁵¹ |
| Tetragonula carbonaria | Australia | 2017 | – | – | – | Hamilton et al.¹⁶³ |
| Melipona fasciculata | Brazil | 2019 | – | – | Lopes et al.¹⁶⁴ | – |
| Scaptotrigona bipunctata, Melipona marginata, Tetragonisca angustula, Trigona hypogea, Melipona quadrifasciata, Tetragona clavipes | Brazil | 2020 | – | Biluca et al.¹⁴⁴ | – | – |
| Tetragonula spp. | Malaysia | 2020 | – | – | Badrulhisham et al.¹⁶⁵ | – |
| Melipona fasciculata | Brazil | 2020 | – | – | – |
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| Antimicrobial | Heterotrigona itama | Malaysia | 2020 | – | – | – | Zhang et al.¹⁶⁷ |
| Heterotrigona itama | Malaysia | 2021 | – | Ooi et al.¹⁶⁸ | – | – | |
| Tetragonula carbonaria | Australia | 2022 | – | – | Hamilton et al.¹⁶⁹ | – | |
| Tetragonisca fiebrigi | Argentina | 2022 | – | Salomon et al.¹⁶⁷ | – | – | |
| Heterotrigona itama, Tetragona binghami | Malaysia | 2022 | – | Wu et al.¹⁷⁰ | – | – | |
| Tetragonula biroi | Indonesia | 2023 | – | – | – | Arung et al.¹⁵⁴ | |
| Heterotrigona itama, Tetragonula reepeni, Tetragonula testaceitarsis, Tetragonula fuscobalteata, Tetragonula iridipennis, Tetragonula pagdeni | Indonesia | 2023 | – | Naibaho et al.⁶³ | – | – | |
| Geotrigona sp. | Ecuador | 2023 | – | – | Ferreira et al.⁵³ | – | |
| Tetragonisca fiebrigi | Brazil | – | – | – | – | – | |
| Melipona seminigra | Brazil | 2013 | da Silva et al.⁶⁴ | – | – | – | |
| Tetragonisca fiebrigi | Brazil | 2015 | – | – | – | Campos et al.¹⁵⁹ | |
| Melipona quadrifasciata, Tetragonisca angustula | Brazil | 2017 | – | – | – | dos Santos et al.¹⁵¹ | |
| Geniotrigona thoracica, Heterotrigona itama, Heterotrigona erythrogastra | Malaysia | 2018 | – | Tukishta et al.¹⁴¹ | – | – | |
| Geniotrigona thoracica, Heterotrigona itama, Tetragona binghami | Brunei | 2020 | – | – | Abdullah et al.¹⁷¹ | – | |
| Axestotrigona ferruginea | Tanzania | 2021 | – | Popova et al.⁵¹ | – | Popova et al.⁵¹ | |
| Tetragonisca fiebrigi | Argentina | 2022 | – | Dallagnol et al.¹⁷² | – | – | |
| Heterotrigona itama, Tetragonula binghami | Malaysia | 2022 | – | Wu et al.¹⁷⁰ | – | – | |
| Axestotrigona ferruginea, Axestotrigona togoensis, Meliplebeia beccarii, Hypotrigona gribodoi, Dactylurina schmidti, Plebeina armata | Tanzania | 2023 | – | Mduda et al.⁶⁵ | – | – | |
| Heterotrigona itama, Tetragonula reepeni, Tetragonula pagdeni, Tetragonula iridipennis, Tetragonula fuscobalteata, Tetragonula testaceitarsis | Indonesia | 2023 | – | Naibaho et al.¹⁷³ | – | – |
Antinociceptive
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| Antinociceptive | Melipona subnitida | Brazil | 2014 | – | – | – | Silva et al.⁶⁶ |
| Melipona fasciculata | Brazil | 2019 | – | – | – | Lopes et al.¹⁶⁴ |
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| Antioxidant | Tetragonisca fiebrigi | Argentina | 2022 | Salomon et al.¹⁶ | – | – | – |
| Melipona subnitida | Brazil | 2006 | – | Silva et al.⁶⁵ | – | – | |
| Tetragonisca angustula | Venezuela | 2007 | Pérez-Pérez et al.¹¹⁰ | – | – | – | |
| Melipona sp., Tetragonisca sp. | Venezuela | 2007 | Rodríguez-Malaver et al.¹⁷⁴ | – | – | – | |
| Melipona seminigra | Brazil | 2013 | da Silva et al.⁶⁴ | – | – | – | |
| Tetragonisca angustula | Venezuela | 2013 | Pérez-Pérez et al.¹³⁸ | – | Pérez-Pérez et al.¹³⁸ | – | |
| Melipona fasciculata | Brazil | 2014 | – | – | Dutra et al.¹⁰⁶ | – | |
| Tetragonula carbonaria | Australia | 2016 | – | – | Hamilton et al.¹⁶² | – | |
| Geniotrigona thoracica, Heterotrigona itama, Tetrigona apicalis | Malaysia | 2016 | – | Nurdianah et al.¹⁷³ | – | – | |
| Tetragonula carbonaria | Australia | 2017 | – | – | Hamilton et al.¹⁶³ | – | |
| Tetrigona apicalis, Heterotrigona itama, Geniotrigona thoracica | Malaysia | 2019 | – | Hanif Fadzilah et al.¹⁷⁵ | – | – | |
| Melipona subnitida, Tetragona clavipes, Scaptotrigona spp. | Brazil | 2017 | – | – | Pazin et al.¹³⁹ | – | |
| Melipona quadrifasciata, Tetragonisca angustula | Brazil | 2019 | – | – | dos Santos et al.¹⁵¹ | – | |
| Geniotrigona thoracica, Heterotrigona itama, Heterotrigona erythrogastra | Malaysia | 2018 | – | Tukisita et al.¹⁴¹ | – | – | |
| Tetrigona apicalis, Heterotrigona itama, Geniotrigona thoracica | Malaysia | 2019 | – | – | – | Asem et al.⁸⁰ | |
| Tetragonula biroi | Philippines | 2019 | – | Belina-Aldemita et al.¹⁴² | – | – | |
| Melipona compressipes | Brazil | 2019 | – | – | Carneiro et al.¹³⁷ | – | |
| Melipona quadrifasciata, Melipona asilvai, Melipona subnitida, Melipona scutellaris | Brazil | 2019 | – | Oliveira et al.¹⁴³ | – | – | |
| Geniotrigona thoracica, Heterotrigona itama, Tetragonula binghami | Brunei | 2020 | – | – | Abdullah et al.¹⁷¹ | – | |
| Scaptotrigona bipunctata, Melipona marginata, Tetragonisca angustula, Trigona hypogea, Melipona quadrifasciata, Tetragona clavipes | Brazil | 2020 | – | Biluca et al.¹⁴⁴ | – | – | |
| Heterotrigona itama | Malaysia | 2020 | – | Majid et al.¹⁴⁵ | – | – |
Table 12 – Antiproliferative, Chemopreventive, Hypocholesterolemic, and Modulator of Gut Microbiota)
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| (continued) | Tetrigona apicalis, Tetrigona binghami, Homotrigona fimbriata | Malaysia | 2021 | – | – | – | Syed Salleh et al.⁸² |
| Axestotrigona ferruginea, Axestotrigona togoensis, Meliplebeia beccarii, Meliponula bocandei, Liotrigona spp., Plebeina armata | Kenya | 2022 | Mokaya et al.¹⁷⁸ | – | – | – | |
| Heterotrigona itama, Tetragonula binghami | Malaysia | 2022 | Wu et al.¹⁷⁹ | – | – | – | |
| Geotrigona sp., Tetragonisca fiebrigi | Ecuador | 2023 | – | – | Ferreira et al.⁵³ | – | |
| Tetragonula laeviceps | Thailand | 2023 | – | – | Iesa et al.¹⁴⁷ | – | |
| Axestotrigona ferruginea, Axestotrigona togoensis, Meliplebeia beccarii, Hypotrigona gribodoi, Dactylurina schmidti, Plebeina armata | Tanzania | 2023 | Mduda et al.⁷⁰ | – | – | – | |
| Tetragonula carbonaria | Australia | 2013 | Vit et al.⁶⁷ | – | – | – | |
| Melipona fasciculata, Melipona rufiventris, Melipona scutellaris, Melipona subnitida, Scaptotrigona polysticta, Frieseomelitta nigra, Melipona beecheii, Melipona fasciata, Melipona solani, Scaptotrigona hellwegeri, Scaptotrigona mexicana | Brazil / Mexico | – | – | – | – | – | |
| Melipona favosa | Venezuela | – | – | – | – | – |
Chemopreventive
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| Chemopreventive | Lepidotrigona terminata | Malaysia | 2016 | – | – | Omar et al.⁷¹ | – |
| Geniotrigona thoracica, Heterotrigona itama | Malaysia | 2018 | – | Ismail et al.⁶⁸ | – | Ismail et al.⁶⁸ | |
| Lepidotrigona terminata | Malaysia | 2016 | – | – | Omar et al.⁷¹ | – | |
| Tetragonula spp. | Malaysia | 2016 | – | – | Yazan et al.⁷² | – |
Hypocholesterolemic
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| Hypocholesterolemic | Melipona seminigra | Brazil | 2021 | – | – | – | Rebelo et al.⁷³ |
Modulator of gut microbiota
| Active Biomolecules | Stingless bee species | Country | Year | Pot-honey | Pot-pollen | Cerumen | Propolis |
|---|---|---|---|---|---|---|---|
| Modulator of gut microbiota | Heterotrigona itama | Malaysia | 2019 | – | – | – | Zulkahiri Amin et al.⁷⁴ |
| Heterotrigona itama | Malaysia | 2020 | – | – | – | Mohamad et al.¹⁷⁹ | |
| Tetragonula sarawakensis, Heterotrigona itama, Tetragonula testaceitarsis, Tetragonula minangkabau, Geniotrigona thoracica, Tetragonula binghami | Indonesia | 2022 | – | – | – | Melia et al.⁷⁵ |
Text below the table
The list of stingless bee taxa of this revision was tabulated in Table 13 with their Neotropical, Afrotropical, Indo-Malaysian, and Australian geographical distribution. Pot-honey, pot-pollen, cerumen and propolis from 31 Neotropical (Argentina, Brazil, Ecuador, Mexico, and Venezuela), 9 Afrotropical (Kenya and Tanzania), 22 Indo-Malaysian (Brunei, India, Indonesia, Malaysia, Philippines, and Thailand), and 1 Australian stingless bee taxa were studied for their flavonoid.
Most biological activities were studied for materials of stingless bee species from Indo-Malaysian Heterotrigona itama (9) and Geniotrigona thoracica (7), the Neotropical Tetragonisca angustula (6) and Tetragonisca fiebrigi (6), and the unique medicinal Australian bee Tetragonula carbonaria (5) in our search.
Table 13. Stingless bee taxa of the geographical region (Neotropical, Afrotropical, Indo-Malaysian, Australian) used in medicinal stingless bee research of Table 12.
| No. | Geographical Region / Stingless Bee Taxa | Country | Biomolecules and Biological Activity |
|---|---|---|---|
| Neotropical | |||
| 1 | Frieseomelitta nigra | Mexico | Antiproliferative |
| 2 | Frieseomelitta varia | Brazil | Flavonoids, Polyphenols |
| 3 | Geotrigona sp. | Ecuador | Flavonoids, Polyphenols, Antioxidant |
| 4 | Melipona asilvai | Brazil | Flavonoids, Polyphenols, Antioxidant |
| 5 | Melipona beecheii | Mexico | Antiproliferative |
| 6 | Melipona compressipes | Brazil | Flavonoids, Polyphenols, Antioxidant |
| 7 | Melipona fasciculata | Mexico | Antiproliferative |
| 8 | Melipona fasciculata | Brazil | Polyphenols, Anti-inflammatory, Antinociceptive, Antioxidant, Antiproliferative |
| 9 | Melipona favosa | Venezuela | Flavonoids, Anticataract, Antiproliferative, Chemopreventive |
| 10 | Melipona marginata | Brazil | Flavonoids, Polyphenols, Antioxidant |
| 11 | Melipona orbignyi | Brazil | Anticancer, Anti-inflammatory |
| 12 | Melipona quadrifasciata | Brazil | Flavonoids, Polyphenols, Anti-inflammatory, Antimicrobial, Antioxidant |
| 13 | Melipona rufiventris | Brazil | Antiproliferative |
| 14 | Melipona scutellaris | Brazil | Flavonoids, Polyphenols, Antioxidant |
| 15 | Melipona seminigra | Brazil | Flavonoids, Polyphenols, Antimicrobial, Antioxidant, Antiproliferative, Hypocholesterolemic |
| 16 | Melipona solani | Mexico | Antiproliferative |
| 17 | Melipona subnitida | Brazil | Flavonoids, Polyphenols, Antinociceptive, Antioxidant, Antiproliferative |
| 18 | Melipona sp. | – | Flavonoids |
| 19 | Paratrigona anduzei | Brazil | Flavonoids, Polyphenols |
| 20 | Scaptotrigona bipunctata | Brazil | Flavonoids, Polyphenols, Anti-inflammatory, Antioxidant |
| 21 | Scaptotrigona depilis | Brazil | Flavonoids, Polyphenols |
| 22 | Scaptotrigona hellwegeri | Mexico | Antiproliferative |
| 23 | Scaptotrigona mexicana | Mexico | Anticataract, Antiproliferative |
| 24 | Scaptotrigona polysticta | Brazil | Antiproliferative |
| 25 | Scaptotrigona spp. | Brazil | Flavonoids, Polyphenols, Antioxidant |
| 26 | Tetragona clavipes | Brazil | Flavonoids, Polyphenols, Anti-inflammatory, Antioxidant |
| 27 | Tetragonisca angustula | Brazil | Flavonoids, Polyphenols, Anticataract, Anti-inflammatory, Antimicrobial, Antioxidant |
| Venezuela | Flavonoids, Polyphenols, Anticataract, Antimicrobial, Antioxidant | ||
| 28 | Tetragonisca fiebrigi | Brazil | Flavonoids, Polyphenols, Anti-inflammatory, Antimicrobial, Antinociceptive, Antioxidant |
| No. | Geographical Region / Stingless Bee Taxa | Country | Biomolecules and Biological Activity |
|---|---|---|---|
| 30 | Tetragonisca sp. | Venezuela | Antioxidant |
| 31 | Trigona hypogea | Brazil | Flavonoids, Polyphenols, Anti-inflammatory, Antioxidant |
| 5 countries |
Afrotropical
| No. | Stingless Bee Taxa | Country | Biomolecules and Biological Activity |
|---|---|---|---|
| 1 | Axestotrigona ferruginea | Tanzania | Antimicrobial, Antioxidant |
| Kenya | Antioxidant | ||
| 2 | Axestotrigona togoensis | Tanzania | Antimicrobial, Antioxidant |
| Kenya | Antioxidant | ||
| 3 | Dactylurina schmidti | Tanzania | Antimicrobial, Antioxidant |
| 4 | Hypotrigona gribodoi | Tanzania | Antimicrobial, Antioxidant |
| 5 | Liotrigona sp. | Kenya | Antioxidant |
| 6 | Meliplebeia beccarii | Tanzania | Antimicrobial, Antioxidant |
| Kenya | Antioxidant | ||
| 7 | Meliponula bocandei | Kenya | Antioxidant |
| 8 | Meliplebeia lendliana | Kenya | Antioxidant |
| 9 | Plebeina armata | Tanzania | Antimicrobial, Antioxidant |
| | | 2 countries | Antioxidant |
Indo-Malaysian
| No. | Stingless Bee Taxa | Country | Biomolecules and Biological Activity |
|---|---|---|---|
| 1 | Geniotrigona thoracica | Malaysia | Flavonoids, Polyphenols, Anti-atherogenic, Antihyperglycemic, Antimicrobial, Antioxidant, Antiproliferative |
| Brunei | Antimicrobial, Antioxidant | ||
| Indonesia | Modulator of gut microbiota | ||
| 2 | Heterotrigona bakeri | Indonesia | Anticancer |
| 3 | Heterotrigona erythrogastra | Malaysia | Polyphenols, Antimicrobial |
| 4 | Heterotrigona itama | Malaysia | Flavonoids, Polyphenols, Anti-atherogenic, Antihyperglycemic, Anti-inflammatory, Antimicrobial, Antioxidant, Antiproliferative, Modulator of gut microbiota |
| Indonesia | Anticancer, Anti-inflammatory, Antimicrobial, Modulator of gut microbiota | ||
| Brunei | Antimicrobial, Antioxidant | ||
| 5 | Homotrigona apicalis | Indonesia | Anticancer |
| 6 | Homotrigona fimbriata | Indonesia | Flavonoids, Polyphenols, Anticancer, Antioxidant |
| 7 | Lepidotrigona terminata | Malaysia | Anticancer, Antiproliferative, Chemopreventive |
| Thailand | Antihyperglycemic, Antiproliferative, Chemopreventive | ||
| 8 | Lepidotrigona ventralis | Thailand | Antihyperglycemic |
| 9 | Tetragonula fuscibasis | Indonesia | Anticancer |
| 10 | Tetragonula biroi | Philippines | Flavonoids, Anticancer, Antioxidant |
| No. | Geographical Region / Stingless Bee Taxa | Country | Biomolecules and Biological Activity |
|---|---|---|---|
| 11 | Tetragonula fuscobalteata | Indonesia | Antihyperglycemic, Anti-inflammatory |
| 12 | Tetragonula iridipennis | Indonesia | Anticancer, Anti-inflammatory, Antimicrobial |
| 13 | Tetragonula laeviceps | Thailand | Polyphenols, Anticancer, Antioxidant |
| Indonesia | Antihyperglycemic | ||
| 14 | Tetragonula minangkabau | Indonesia | Modulator of gut microbiota |
| 15 | Tetragonula pagdeni | Thailand | Antihyperglycemic, Anti-inflammatory, Antimicrobial |
| 16 | Tetragonula reepeni | Indonesia | Anti-inflammatory, Antimicrobial |
| 17 | Tetragonula sapiens | Indonesia | Antihyperglycemic |
| 18 | Tetragonula sarawakensis | Indonesia | Anticancer, Modulator of gut microbiota |
| 19 | Tetragonula testaceitarsis | Indonesia | Anticancer, Anti-inflammatory, Antimicrobial, Modulator of gut microbiota |
| 20 | Tetragonula spp. | India | Anticancer |
| Malaysia | Anti-inflammatory, Chemopreventive | ||
| 21 | Tetrigona apicalis | Malaysia | Flavonoids, Polyphenols, Antihyperglycemic, Antioxidant |
| 22 | Tetragona binghami | Malaysia | Flavonoids, Polyphenols, Anti-inflammatory, Antioxidant |
| Brunei | Antimicrobial, Antioxidant | ||
| Indonesia | Modulator of gut microbiota | ||
| 23 | Wallacetrigona incisa | Indonesia | Anticancer |
| 23 taxa | 6 countries |
Australian
| No. | Stingless Bee Taxa | Country | Biomolecules and Biological Activity |
|---|---|---|---|
| 1 | Tetragonula carbonaria | Australia | Polyphenols, Anti-inflammatory, Antioxidant, Antiproliferative, Chemopreventive |
3.4 QUALITY CONTROL OF STINGLESS BEE PRODUCTS. KEY BIOMOLECULES, METHODS AND TECHNIQUES
The developing interest towards use of stingless bee products to support human health makes the issue of their standardization increasingly important and urgent. It is well known that stingless bee honey does not comply with the requirements for honeybee honey in the CODEX Alimentarius⁷⁷, which have been created for Apis mellifera honey. The growing amount of data on the characteristics of stingless bee honey led to the need of creation of specific quality standard for pot-honey. In general, stingless bee honeys do not meet the CODEX Standard for honey moisture, free acidity, and total fructose plus glucose levels. In addition, it has been suggested to apply the presence and amount of a rare reducing sugar, trehalulose, as a marker of authenticity of pot-honey⁷⁸.
However, to produce a universal standard and quality parameters, further information from studies of the chemical composition of stingless bee honey — such as organic acids and polyphenol profiles of pot-honey from stingless bee species from different geographical regions is required.
Targeted ¹H-NMR is adequate to compare sugars, amino acids, aliphatic organic acids, HMF, ethanol, and botanical markers of pot-honey produced by diverse entomological origins¹².
Concerning stingless bee propolis, the chemical variability is much greater than that observed with A. mellifera propolis. This has been a serious hindrance in the case of Apis mellifera propolis which now, after decades of intense research, comes to an at least partial solution. The stingless bees’ propolis poses a much more difficult problem, due to its greater chemical diversity. It is interesting to note that several molecules with significant bioactivities have been found in pot-propolis (gallic acid, alpha-mangostine, propolin A), some of them new chemical entities, such as sulawesin A, mammeoin cinnamoyl ester, etc.⁷⁹ (Fig. 15).
Fig. 15 Important bioactive compounds in stingless bee propolis
The antimicrobial and antioxidant activities of stingless bee propolis are usually reported to depend on its total phenolic content, e.g. Asem et al.⁸⁰. However, it is necessary to remember that different stingless bee propolis often contain different phenolic compounds and equal number of observed gallic acid equivalents could correspond to a very different real concentration of phenolics. For this reason, the chemical type of the propolis samples should be observed by LC-MS⁸¹ or GC-MS analyses⁸². As in the case of pot-honey or stingless bee honey, pot-pollen or stingless bee pollen, and cerumen, many more studies have to be conducted on the chemistry and biological activity of stingless bee propolis before any quality control criteria could be formulated.
3.5 INFLUENCE OF THE MELIPONA GENUS BAHIA STATE HONEY NORM FROM BRAZIL (2014), AND FURTHER POT-HONEY NORMS IN MEDICINAL USES OF POT-HONEY
In their seminal paper, Gonnet et al.⁸³ evidenced the higher moisture and free acidity of Melipona honey compared to Apis mellifera honey. The first proposal for stingless bee honey standards included honey of the Melipona from Guatemala, Mexico, and Venezuela.⁹ The first Brazilian stingless bee honey norm was created for Melipona honey in the State of Bahia.² Forthcoming Brazilian State standards were established in Amazonas⁸⁴, Paraná⁸⁵, Espírito Santo⁸⁶, and Santa Santa Catarina⁸⁷. The Philippine honey norm⁸⁸ included pot-honey in the last revision, but the most important State National standard was created in 2017 for Kelulut — Malaysian name given to all stingless bees— honey in Malaysia⁸⁹, and the second was for the Argentine stingless bee Tetragonisca fiebrigi known with the ethnic name Yate’i in 2019⁹⁰.
Concomitant with the scientific research on medicinal properties of stingless bee products, regulated pot-honey facilitates administrative procedures to launch pharmaceutical products. The diverse state norms in Brazil have promoted presentations of pot-honey, pot-pollen, and propolis of pharmaceutical quality, available online⁹¹.
The facts of standardized pot-materials of the stingless bee nest are promising:
- More quality products will be produced and will command fair market prices,
- Regulated products are safe for human and animal health,
- Support of apitherapy by providing quality raw materials,
- Best stingless bee-keeping practices should be strictly followed in order to produce quality products, and especially
- Stingless bee apiaries should backup the volume of marketed stingless bee products, to prevent falsifications.
Potential categorizations of stingless bee materials may include antioxidant activity grading, as suggested for Czech honey using μmoles equivalents Trolox/100 g honey in a scale of:
- pro-oxidant (<1),
- very low (0–50),
- low (51–100),
- moderately low (101–150),
- medium (151–200),
- moderate high (201–250),
- high (251–300),
- very high (>300)
by Vit et al.⁹².
A more recent idea of categorization arises after the trehalulose discovery in pot-honey by Fletcher et al., 2020³⁹ and chemical marker analysis⁷⁸ could provide useful gradings for entomological origins.
This unique sugar needs extensive research and the following trend reveals useful for trehalulose concentrations (g/100g honey) graded for preliminary stingless bee species:
- (10.0–39.9) Heterotrigona itama, Tetragonula carbonaria, Tetragonula hockingsi;
- (40.0–49.9) Geniotrigona thoracica, Heterotrigona itama, and
- (50.0–59.9) Geniotrigona thoracica.
Our Quality of the beehive for apitherapy in the VII National Congress of Pharmaceutical Science 2009⁹⁴ has focused to Quality of the stingless bee nest for apitherapy in the Apimondia 2023 Central Symposium for Apitherapy, consistent with good practices of stingless bee keeping to achieve great quality of stingless bee products for direct use or in pharmaceutical preparations⁹⁵. Vit and Simova⁹⁶ reviewed the aliphatic organic acids (AOA) in honey and proposed updated reference values for Apis mellifera and stingless bee honeys.
3.6 INTEGRATIVE APPLICATIONS OF STINGLESS BEE NEST METABOLITES TO OVERCOME ANTIBIOTIC RESISTANCE
Antimicrobial resistance (AMR) has become a global health and socioeconomic issue requiring urgent attention⁹⁷. In 2019, an estimated 1.27 million deaths worldwide were attributable to antibiotic-resistant bacterial infections⁹⁸. The World Health Organization (WHO) estimated that by 2050, diseases caused by multidrug-resistant bacteria could lead to 10 million deaths annually, surpassing cancer as the leading cause of death⁹⁹.
In 2024, the WHO updated its list of bacterial pathogens considered a priority due to limited treatment options and their significant global health impact. At the top of this list are highly virulent bacteria, including carbapenem-resistant Acinetobacter baumannii, carbapenem-resistant Enterobacter, and third-generation cephalosporin-resistant Enterobacterales¹⁰⁰. Infections caused by these pathogens are difficult to prevent, highly transmissible, and associated with a high mortality rate, making them a major public health threat⁹⁹–¹⁰¹.
The global rise of multidrug-resistant pathogens represents one of the greatest challenges to modern medicine, prompting the search for new therapeutic strategies beyond conventional antibiotics¹⁰². In this context, stingless bee nest derivatives, such as pot-honey, pot-pollen, cerumen, and propolis, are emerging as promising sources of bioactive compounds for developing effective therapies against resistant microorganisms¹⁰³.
Stingless bees produce a wide variety of bioactive substances, including antimicrobial peptides, polyphenols, flavonoids, and enzymes that synergistically inhibit bacterial growth and biofilm formation, modulate immune responses, promote tissue repair, and reduce inflammation¹⁰⁴. Propolis, in particular, has been extensively studied for its high content of phenolic compounds and flavonoids with potent antioxidant and antimicrobial effects, capable of inhibiting both Gram-positive and Gram-negative bacteria, including multidrug-resistant strains¹⁰⁵. These compounds act through multiple mechanisms: altering bacterial cell membranes, inhibiting protein synthesis, preventing biofilm formation, and enhancing antibiotic efficacy through proven synergies¹⁰⁶.
Similarly, pot-honey exhibits antimicrobial activity due to the presence of hydrogen peroxide and other compounds that act synergistically with flavonoids and phenolic acids to destabilize bacterial membranes and prevent biofilm formation as key factors in the persistence of chronic resistant infections¹⁰⁴.
Beyond the intrinsic antimicrobial properties of stingless bee nest metabolites, numerous studies emphasize the synergistic interactions between bee products and conventional antibiotics¹⁰³,¹⁰⁷,¹⁰⁸. This synergy not only enhances antimicrobial efficacy and reduces the required antibiotic dosage but also helps prevent the emergence of new resistances and mitigates potential side or toxic effects¹⁰².
Araque and Vit¹⁰⁹ evaluated the synergistic potential of ethanolic extracts of Tetragonisca angustula pot-pollen combined with amikacin and meropenem—two frontline antibiotics used against resistant infections. Their results demonstrated strong synergistic interactions in 75% of the bacterial isolates tested, including clinically significant multidrug-resistant strains such as Klebsiella pneumoniae, Pseudomonas aeruginosa, and Acinetobacter baumannii. This synergy was evidenced by up to a threefold reduction in the minimal inhibitory concentrations (MICs) of the antibiotics, indicating a potential adjuvant role for pot-pollen in restoring antibiotic susceptibility and reducing therapeutic dosages.
Complementing these findings, Vit et al.¹¹⁰ examined the viable compounds in pot-pollenand explored their bioactivity and synergism with antibiotics. In addition to antimicrobial and anti-inflammatory properties, they reported innovative applications of these volatiles in enhancing food flavors, potentially improving the acceptability and clinical application of pot-pollen-derived products. Their bibliometric analysis revealed increasing scientific interest and strong evidence supporting the anti-AMR potential of pot-pollen as a multifunctional natural resource.
From a critical perspective, despite the favorable consensus, the variability in chemical composition due to botanical origin, geographical location, and technical processing presents a challenge in translating findings into standardized and regulated therapeutic applications¹¹¹. Moreover, most studies remain preclinical or in vitro, highlighting the need for robust clinical trials to confirm efficacy and safety in human populations. Nonetheless, advances in the chemical characterization and functional analysis of these natural compounds are paving the way for their inclusion in innovative formulations such as nanoemulsions, gels, and other controlled-release systems¹⁰⁷–¹¹².
The therapeutic promise of stingless bee nest materials extends beyond their capacity to combat bacterial resistance. Their metabolic and cellular protective effects may enhance overall host health and offer potential for managing metabolic and inflammatory diseases that are increasingly prevalent. This integrative perspective underscores the value of stingless bee nest materials as complementary agents in therapeutic management, where responsible and evidence-based utilization could play a pivotal role in the post-antibiotic era.
3.7 GENOMIC AND METAGENOMIC INSIGHTS INTO THE MEDICINAL POTENTIAL OF STINGLESS BEE NEST MATERIALS
Antimicrobial agents biosynthesized for the survival of microbial communities in honey contribute to its well-known antimicrobial properties¹¹³. Similarly, interactions among microbial populations inhabiting stingless bee nest materials may explain the frequently reported antimicrobial activities of pot-honey¹¹⁴, pot-pollen¹¹⁵, cerumen¹¹⁶, and propolis¹¹⁷. The wide variety of bioactive molecules of microbial origin has been comprehensively reviewed by Vit (2024)¹¹⁸ and Alves et al. (2024)¹¹⁹.
Metabolites generated by the stingless bee nest microbiome represent a vast reservoir of largely unexplored natural compounds that play critical ecological roles and hold strong potential for biotechnological and medicinal applications, including drug discovery. Developing such applications requires understanding the complex microbiota associated with stingless bees to identify and isolate specific bacterial and fungal strains and their metabolites as potential therapeutic agents or dietary supplements. As shown in bumble bees, microbiome assembly and maintenance are dynamic across the lifespan of social bees¹²⁰, and likely also in stingless bees and their nest materials, a factor that must be considered when harvesting high-quality materials.
Current research on stingless bee gut microbiomes remains at an exploratory stage. The core microbiome of Apis mellifera differs markedly from that of the few stingless bee species studied in Australia¹²¹, Brazil¹²²,¹²³, Mexico¹²⁴, and Thailand¹²⁵. Emerging evidence suggests that host-specific and geographical factors influence microbiome composition in stingless bee nest materials, representing an untapped source of bioactive compounds that may contribute both to colony health and to human health applications.
Biotransformations of phytochemicals such as flavonoids and polyphenols by gut microbes involve two-way interactions:
- microbial degradation reduces molecule size, while
- phenolics modulate microbial populations¹²⁶.
Investigating how microbes transform compounds, communicate with each other within communities, interact with the host, and analyzing their microbial products provides deeper insight into host–microbe interactions and their effects on health and disease. Knowing the microbiome helps finding potential active biomolecules by using advanced metabolomics to identify and quantify the chemicals microbes produce. Exploring their bioactive properties reveals molecules that can act as therapeutics, drug targets, or function as biomarkers. By deciphering microbiome-mediated transformations of host-derived molecules and xenobiotics, researchers can uncover novel enzymes and biochemical pathways. Understanding metabolic fingerprints of the microbiome connects microbial communities or a unique microbe from food storage, architectural materials of the nest, or the stingless bee itself with a particular function or a set of roles.depilis, a Brazilian stingless bee, three microbial populations coordinate during pupal metamorphosis: one produces the steroid ergosterol, two modulate its availability¹²⁷, and the symbiotic yeast Zygosaccharomyces sp. promotes metamorphic development¹²⁸. Such examples illustrate the intricate interplay between stingless bees and their symbionts. Our particular interest lies in exploring how these associated microbes may contribute to human health by:
- Modulating host physiology,
- Influencing disease dynamics,
- Interacting with immune responses, and
- Enabling discovery of novel drugs and therapeutic agents.
Genomics and metagenomics are two technologies with complementary approaches to this pursuit. Metagenomics characterizes microbial communities and their functional genes within complex nest environments, while genomics focuses on individual organisms and their genetic composition. Genomics supports applications in drug discovery, whereas metagenomics advances environmental microbiome research and natural product exploration. To fully realize the therapeutic potential of stingless bee nest materials, metagenomics and genomics are needed in tandem.
Instead of morphological identifications of pollen grains by melissopalynology⁴⁹, DNA metabarcoding was proposed to characterize the botanical origin of honey¹³⁰ but a multi-kingdom resolution is now represented by a major high throughput technology of whole genome shotgun sequencing (WGS) known as shotgun metagenomics¹³¹ covering all DNA instead of primers, unlocking potential applications. A comprehensive view of the entire community of organisms and their genes is achieved by sequencing all DNA from a sample using this powerful tool for studying complex materials, fragmenting all the DNA, sequencing it, and using bioinformatics to identify the organisms present, their functional genes, and metabolic pathways, regardless of whether they are animals, bacteria, fungi, plants or viruses¹³²,¹³³. Amplicon sequencing and shotgun metagenomics are two platforms used in epidemiological research. Larger microbiome datasets characterize pooled amplicon/shotgun data compared to pure shotgun metagenomic. Usyk et al.¹³⁴ harmonized this pooling approach to leverage the exponential amplicon sequencing data produced over two decades.
3.8 FRAUD CONTROL OF STINGLESS BEE PRODUCTS
Stingless bee products have lower yields than Apis mellifera because their colonies and nests are smaller, with variable size according to the stingless bee species and environmental factors. Productivity varies with years and some colonies are more propolizers than others. In Table 14, colony size and nest type of some stingless bee species from Brazil³³.
Table 14. Colony size of selected stingless bee species
| Stingless bee species (Maximum flight distance) | Bee size (mm) | Colony size Average No. workers (min–max) | Type of nest |
|---|---|---|---|
| Cephalotrigona capitata | |||
| [1.7 km] | 9.5 | 1250 (1000–1500) | • Living tree cavities |
| • Horizontal brood combs with involucrum | |||
| • Rather large storage pots, with pot-honey and pot-pollen in different sections | |||
| • Permanent deposit of detritus | |||
| • Entrance decorated with plant resins and a hardened cerumen landing platform | |||
| Frieseomelitta varia | |||
| [1.4 km] | 5.5 | 1200 (800–1600) | • Tree cavities |
| • Horizontal brood combs without involucrum | |||
| • Small spheroidal honey pots, larger pollen-pots slightly elongated | |||
| • Entrance size of worker head | |||
| Geotrigona mombuca | |||
| [unknown] | 5.0 | 2500 (2000–3000) | • Underground |
| • Cylindrical storage pots, draining gallery at nest bottom, permanent deposits of detritus | |||
| • Underground channel connects the nest with the entrance in the surface |
Due to the lower yields of pot-honey, pot-pollen and propolis, stingless bee products are known to have higher costs than Apis mellifera, and thus pot-honey has been imitated, mixed with honeybee products, sugars and syrups used in the bee fraud industry (P. Vit, personal observation). Pot-honey yields of Melipona scutellaris are 2–15 kg/year (R.M.O. Alves personal observation), Scaptotrigona spp 3 kg/year, and Tetragonisca angustula 1 kg/year¹⁴⁰ at about 100–200 USD/kg. Pot-pollen yields of Melipona scutellaris are 4–5 kg/year, Scaptotrigona spp. 5–6 kg/year, and Tetragonisca angustula 3–4 kg/year with a value from 32 to 257 USD/kg¹⁴¹.
Untargeted ¹H-NMR studies of sugar profiles of stingless bee honey and chemometrics are particularly useful to detect adulterations¹⁶². For certain entomological origins, the recently discovered trehalulose⁹³ is a potential sugar for genuine pot-honey authenticity⁷⁸. Targeted ¹H NMR revealed distinctive composition of pot-honey at genus level¹². A simple palynological screening would detect absence of pollen spectra in sugar syrups manufactured to fake honey¹². However, sophistication requires diverse bioanalytical techniques for pot-honey fraud control. For the expert, a simple sensory evaluation uncovers the adulteration, but consumers are not always acquainted with stingless bee nest materials as meliponicultors or a community with stingless bee apiaries or meliponaries. A honey authenticity test based on interface emulsion produced after shaking a honey dilution with diethyl ether, differentiated genuine from fake honey¹⁸⁴, and recently provided a further interpretation on suspected microbial associations with the Ecuadorian Scaptotrigona vitorum producing biosurfactants in pot-honey¹².
Conclusions
The biotic materials processed to form pot-honey and pot-pollen have botanical, entomological, and microbial origins, along with plant resins that are essential for producing cerumen, a vital component of stingless bee nests. These nest materials contain diverse compounds and active metabolites that play crucial roles in their biological activities. Their antimicrobial and antioxidant properties contribute significantly to the added value of stingless bee nest materials, supporting their medicinal potential for both nutritional and pharmaceutical applications.
Research on the metabolites present in stingless bee nest materials and their ecological roles provides valuable insights into the complex relationships between stingless bees and their environment. Understanding the chemical composition of these materials may aid conservation efforts targeting both the pollinators and the ecosystems they sustain. The loss of stingless bee biodiversity would also mean the loss of chemical diversity in the bioactive metabolites within their nests, and, consequently, the loss of valuable natural healing molecules.
Our bibliometric analyses evaluated global scientific research on medicinal stingless bees (2004–2023) and stingless bees in climate change (2010–2023), using Bibliometrix to visualize datasets retrieved from the Scopus database. The medicinal dataset included 107 documents, showing greater research interest compared with the climate change dataset, which contained 25 documents. Top researchers, institutions, sponsors were identified. Hilgert N.I. (Argentina) was the most prolific author in medicinal stingless bee research for her contributions in ethnomedicine, while Martins C.F. (Brazil) led climate change–related studies on habitat loss and pollinator protection, each of them contributing four publications in their respective areas.
Malaysia, Brazil, and Mexico were the top three countries contributing with 62 of 107 documents for medicinal stingless bee research; and for stingless bees in climate change, whereas Brazil, the United States, and Indonesia accounted for 22 of 25 documents on stingless bees and climate change. The most prominent publication sources used to disseminate stingless bee research were the book Pot-Honey: A Legacy of Stingless Bees for the medicinal dataset, and the journal Apidologie for climate change research. Initiatives promoting pollinator protection within agricultural frameworks benefit both stingless bee conservation and the use of active metabolites of their nests in melipotherapy.
The role of microbiomes in metabolite transformation, the authenticity and chemical variability of nest materials, and the relevance of stingless bee biodiversity conservation areemphasized as interconnected drivers of medicinal potential and ecosystem sustainability. In bridging ethnomedicine, microbiology, chemistry, and environmental science, this paper provides a multidisciplinary framework that supports meliponitherapy as a promising field contributing to food security, health, and the Sustainable Development Goals (SDG 2 and SDG 3).
Proposals for Future
Bibliometric reviews on the medicinal uses of stingless bee products for the human body systems need to be periodically updated, and medicinal research funded. Studies on medicinal properties should involve interactive collaboration among entomologists, chemists specializing in stingless bee products, melissopalynologists, and sensory scientists to establish standardized characterizations of raw materials and extracts used by experts in health sciences, experimental biology, and molecular biology. This multidisciplinary approach, which should also include statisticians, can provide a strong foundation for elucidating mechanisms of action over time.
Medicinal and climate change approaches interact over the long term. Brazil, which harbors the greatest stingless bee biodiversity for a country (259 of 605 recognized species worldwide), manages 95 species across its five regions, 12 of which are featured in the A.B.E.L.H.A. catalog, with sizes ranging from 3.5 to 10.5 mm. It is important to note that research on stingless bee nest materials remains ongoing. Understanding the specific active metabolites and their potential applications for human health continues to evolve framed in the Sustainable Development Goals SDG2 food security and SDG3 good health and well-being.
Stingless bees yield increasing amounts of medicinal pot-honey, pot-pollen, cerumen, and propolis, and their derivative products are being progressively developed. The conservation of natural ecosystems and the establishment of stingless bee–friendly managed environments are vital strategies for biodiversity preservation and sustainable meliponiculture, particularly in anticipation of shifting geographical distributions caused by climate change.
Reviews focusing on chemical classes are valuable for developing databases, updating bioanalytical methods, and revising reported units in the scientific literature. For instance, After reviewing aliphatic organic acids in honey and pot-honey, Vit and Simova⁹⁶ provided updated reference concentration values to replace the underestimated 0.5% AOA content commonly cited in scientific literature. Access to public databases that collect, verify, publish, and maintain DNA sequences globally¹³⁵ will be essential for advancing the study of stingless bee nest materials through expert curation, facilitating data exchange, and understanding. Moreover, targeting outer membrane components to enhance antibiotic permeability in Gram-negative bacteria could represent an innovative strategy to curb antimicrobial resistance, similar to recent insights into fungal capsule structures for approaches on novel disease control¹³⁶.
Conflict of Interest Statement:
The authors have no conflicts of interest to declare.
Funding Statement:
None.
Acknowledgements:
To stingless bee keepers, conservationists, and meliponitherapists of the world. To our families for their unconditional support. To Dr. Cristina Mateescu, President of the Apitherapy Commission, for her invitation to the 48th Apimondia Congress September 4–8, 2023 held in Santiago de Chile, and the Chilean hospitality. To Prof. Breno Freitas from Universidade Federal de Ceará, Fortaleza, Brazil for the A.B.E.L.H.A. link. To our academic institutions for their constant support on stingless bee medicinal research. To stingless bee keepers of the world for their commitment with science, and our families for their steadfast encouragement. To native tropical cultures using meliponitherapy following ancestral knowledge. To modern laboratories interested in stingless bee nest materials to advance in the knowledge of their medicinal applications. To the United Nations University–Biotechnology for Latin America and the Caribbean UNU-BIOLAC for the Research Fellowship at the University of California Riverside to P. Vit, and benefactor Edward RH McDowell Jr.
References:
- BRASIL. (2021). Ministerio do Meio Ambiente, Instituto Chico Mendes de Conservacao da Biodiversidade. Portaria n. 665 de 3 de novembro de 2021. Diario Oficial (da) Republica Federativa do Brasil, Poder Executivo, Brasilia, DF 3 de novembro de 2021. secao 1, p. 121.
- ADAB. (2014). Agência de Defesa Agropecuária da Bahia. Portaria ADAB n° 207 de 21/11/2014. Regulamento Técnico de Identidade e Qualidade do Mel de Abelha Social sem Ferrão, do Gênero Melipona. Bahia, Brazil, pp. 1–4.
- Kim, D-H, Sexton, JO, Townshend JR. (2015). Accelerated deforestation in the humid tropics from the 1990s to the 2000s. Geophysical Research Letters 42, 3495–3501. https://doi.org/10.1002/2014GL062777
- Matricardi EAT, Skole DL, Costa OB, Pedlowski MA, Samek JH, Miguel EP. (2020). Long-term forest degradation surpasses deforestation in the Brazilian Amazon. Science 369, 1378–1382. https://doi.org/10.1126/science.abb3021
- Silva Jr CHL, Pessôa ACOM, Carvalho NS, Reis JBC, Anderson LO, Aragão LEOCE. (2021). The Brazilian Amazon deforestation rate in 2020 is the greatest of the decade. Nature Ecology & Evolution 5, 144–145. https://doi.org/10.1038/s41559-020-01368-x
- Rocha-Santos L, Mayfield MM, Lopes AV, Pessoa MS, Talora DC, Faria D, Cazetta E. (2020). The loss of functional diversity: A detrimental influence of landscape-scale deforestation on tree reproductive traits. Journal of Ecology 108, 212–223. https://doi.org/10.1111/1365-2745.13232
- Toledo-Hernández E, Peña-Chora G, Hernández-Velázquez VM, Toribio-Jiménez J, Romero-Ramírez V. (2022). The stingless bees (Hymenoptera: Apidae: Meliponini): a review of the current threats to their survival. Apidologie 53, 8. https://doi.org/10.1007/s13592-022-00913-w
- Engel IMS, Rasmussen C, Ayala R, de Oliveira FF. (2023) Stingless bee classification and biology (Hymenoptera, Apidae): a review, with an updated key to genera and subgenera. Zookeys 1172, 239–319. https://zookeys.pensoft.net/article/104944/list/1/
- Vit P, Medina M, Enriquez ME. (2004). Quality standards for medicinal uses of Meliponinae honey in Guatemala, Mexico and Venezuela, Bee World 85, 2–5. https://doi.org/10.1080/0005772X.2004.11099603
- Pérez-Pérez E, Rodríguez-Malaver J, Vit P. (2007). Efecto de la fermentación en la capacidad antioxidante de miel de Tetragonisca angustula Latreille, 1811. BioTecnología 10, 14–22.
- Echeverrigaray S, Scariot FJ, Foresti L, Schwarz LV, Rocha RKM, da Silva GP, Moreira JP, Delamare APL. (2021). Yeast biodiversity in honey produced by stingless bees raised in the highlands of southern Brazil. International Journal of Food Microbiology 347, 109200. https://doi.org/10.1016/j.ijfoodmicro.2021.109200
- Vit P, van der Meulen JJ, Pedro SRM, Esperanca I, Zakaria R, Beckh G, Maza F, Meccia G, Engel MS. (2023). Impact of genus (Geotrigona, Melipona, Scaptotrigona) in the ¹H-NMR organic profile, and authenticity test by interphase emulsion of honey processed in cerumen pots by stingless bees in Ecuador. Current Research in Food Science 6. https://doi.org/10.1016/j.crfs.2022.11.005
- Betta E, Vit P, Meccia G, Pedro SRM, Romano A, Khomenko I, Biasoli F. (2024). Volatile and sensory profile of cerumen, plant resin deposits, and propolis in tropical Tetragonula melliponaries compared. Planta Medica, pp. 1–12.
- Vit P. (2024). Metabolites from microbial cell factories in stingless bee nests. pp. 53–114. In Vit P., Bankova V, Popova M, Roubik DW (Eds.). Stingless Bee Nest Cerumen and Propolis. Springer Nature; Cham, Switzerland. Volume 2, 501 pp.
- Vit P. (2022). A honey authenticity test by interphase emulsion reveals biosurfactant activity and biotechnology in the stingless bee nest of Scaptotrigona vitorum ‘Catiana’ from Ecuador. Interciencia 47, 416–425. https://www.interciencia.net/volumen-47-2022/volumen-47-numero-10/
- Salomon V, Brockievi ZZL, Gennari G, Maldonado R, Romero CM, Vera NR. (2022). Argentine stingless bee honey: bioactive compounds and health-promoting properties. Natural Resources for Human Health 2. https://doi.org/10.53365/nrfhh/144727
- Mejia G, Mu M, Zhang Y, Kajikawa Y. (2021). Exploring topics in bibliometric research through citation networks and semantic analysis. Frontiers in Research Metrics and Analytics 6, 742311. Aria M, Cuccurullo C. (2017). Bibliometrix: An R-tool for comprehensive science mapping analysis. Journal of Informetrics 11, 959–975. https://doi.org/10.1016/j.joi.2017.08.007
- Persano-Oddo L, Heard TA, Rodriguez-Malaver A, Pérez RA, Fernández-Muiño M, Sancho M.T., Sesta G., Lusco L., Vit P. (2008). Composition and antioxidant activity of Trigona carbonaria honey from Australia. Journal of Medicinal Food 11, 789–794. https://doi.org/10.1089/jmf.2007.0724
- Persano-Oddo L, Piro R. (2004). Main European unifloral honeys: Descriptive sheets. Apidologie 35(Suppl. 1), S38-S81. https://doi.org/10.1051/apido:2004049
- Alves RRN, Oliveira MGG, Barboza RRD, Singh R, Lopez LCS. (2009). Medicinal animals as therapeutic alternative in a semi-arid region of Northeastern Brazil. Forschende Komplementarmedizin 16, 305–312. https://doi.org/10.1159/000235855
- Sgariglia MA, Vattone MA, Vattone MMS, Soberón JR, Sampietro DA. (2010). Properties of Argentine Tetragonisca angustula fiebrigi and Plebeia wittmanni of Argentina [Propriétés du miel produit par Tetragonisca angustula fiebrigi et Plebeia wittmanni en Argentine] [Die Eigenschaften von argentinischen Tetragonisca angustula fiebrigi und Plebeia wittmanni-Honigen]. Apidologie 41, 667–675. https://doi.org/10.1051/apido/2010028
- Boorn KL, Khor Y-Y, Sweetman E, Tan F, Heard TA, Hammer KA. (2010). Antimicrobial activity of honey from the stingless bee Trigona carbonaria determined by agar diffusion, agar dilution, broth microdilution and time-kill methodology. Journal of Applied Microbiology 108: 1534–1543. https://doi.org/10.1111/j.1365-2672.2009.04552.x
- Massaro FC, Brooks PR, Wallace HM, Russell FD. (2011). Cerumen of Australian stingless bees (Tetragonula carbonaria): gas chromatography-mass spectrometry fingerprints and potential anti-inflammatory properties. Naturwissenschaften 98, 329–337. https://doi.org/10.1007/s00114-011-0770-7
- Vit P, Pedro SRM, Roubik D (Eds.) (2013). Pot-Honey: A Legacy of Stingless Bees. New York, USA: Springer. 654 pp.
- Vit P, Pedro SRM, Roubik DW (Eds.). (2018). Pot-Pollen in Stingless Bee Melittology. Cham, Switzerland; Springer. 481 pp.
- Batalha-Filho H, Waldschmidt ÂM, Campos LAO, Tavares MG, Fernandes-Salomão TM. (2010). Phylogeography and historical demography of the neotropical stingless bee Melipona quadrifasciata (Hymenoptera: Apidae): Incongruence between morphology and mitochondrial DNA. Apidologie 41, 534–547. https://doi.org/10.1051/apido/2010001
- Giannini TC, Tambosi LR, Acosta AL, Jaffé R, Saraiva AM, Imperatriz-Fonseca VL, Metzger JP. (2015). Safeguarding ecosystem services: A methodological framework to buffer the joint effect of habitat configuration and climate change. PLoS ONE 10, e0129225. https://doi.org/10.1371/journal.pone.0129225
- Halcroft MT. (2013). Investigations into the biology, behaviour and phylogeny of a potential crop pollinator: the Australian stingless bee, Austroplebeia australis. School of Health and Science. PhD thesis. University of Western Sydney; Sydney, Australia. 388 pp. https://www.beebusiness.com.au/articles/Investigations%20into%20the%20biology,%20behaviour%20and%20phylogeny%20of%20a%20potential%20crop%20pollinator,%20the%20Australian%20stingless%20bee%20Austroplebeia%20australis.pdf
- Nates S, Hall MA, Gloag R, Lynch KE, Spooner-Hart RN, Cook JM, Riegler M. (2022). Heat stress survival and thermal tolerance of Australian stingless bees. Journal of Thermal Biology 117, 103671. https://doi.org/10.1016/j.jtherbio.2023.103671
- Nogueira DS. (2023). Overview of stingless bees in Brazil (Hymenoptera: Apidae: Meliponini). EntomoBrasilis 16, e1041. https://doi.org/10.12741/ebrasilis.v16.e1030
- Kiprono SJ, Mengich G, Kosgei J, Mutai C, Kimoli S. (2022). Ethnomedicinal uses of stingless bee honey among native communities of Baringo County, Kenya. Scientific African 16, e01297. https://doi.org/10.1016/j.sciaf.2022.e01297
- A.B.E.L.H.A. Associação Brasileira de Estudos das Abelhas. Fichas catalográficas das espécies relevantes para a meliponicultura. n/d; https://abelha.org.br/fichas-catalograficas-das-especies-relevantes-para-a-meliponicultura-2/
- Jaffé R. (2018). Influência do transporte de colmeias sobre a estrutura genética das populações de abelhas. pp. 39–47. In: Vilela Neto A, Menezes C (Orgs) Desafios e recomendações para o manejo e Jaffé R. (2018). Influência do transporte de colmeias sobre a estrutura genética das populações de abelhas. pp. 39–47. In: Vilela Neto A, Menezes C (Orgs) Desafios e recomendações para o manejo e transporte de polinizadores. Associação Brasileira de Estudos das Abelhas (A.B.E.L.H.A); São Paulo, Brazil. 100 pp.
- Levin DA, Francisco-Ortega J, Jansen RK. (2002). Hybridization and the extinction of rare plant species. Conservation Biology 10, 10–16. http://www.jstor.org/stable/2386938
- Resende HC, Campos LAO. (2023). Novo registro de hibridização entre as abelhas Melipona capixaba Moure & Camargo, 1994 e Melipona scutellaris Latreille, 1811. pp. 245–254. In: Resende HC, Werneck HA (Orgs.) Estudo sobre abelhas e vespas brasileiras: uma homenagem ao professor Lucio Campos. Florestal, MG: Laboratório de Genética da Conservação de Abelhas. Universidade Federal de Viçosa. 260 pp.
- Levin DA. (2002). Hybridization and Extinction: In protecting rare species, conservationists should consider the dangers of interbreeding, which compound the more well-known threats to wildlife. American Scientist 90, 254–261. https://www.jstor.org/stable/27857661
- Buchori D, Rizali A, Priawandiputra R, Ratnhiun R, Sartami D, Pujasuti Y, Jauharina J, Pradana MG, Meilin A, Laetemia JA, Sudiarta IP, Rustam R, Nelly N, Lestari P, Syahputra E, Hasriyanti H, Watung JF, Daud IA, Hariani N, Jihadi A, Johannis M. (2022). Beekeeping and managed bee diversity in Indonesia: Perspective and preference of beekeepers. Diversity 14. 52. https://doi.org/10.3390/d14010052
- Buono FGB, Kendall L, Alves DA, Tamara ML, Heard T, Latty T, Gloag R. (2023). Stingless bee floral visitation in the global tropics and subtropics. Global Ecology and Conservation 43, e02454. https://doi.org/10.1016/j.gecco.2022.e02454
- Grüter C. (2020). Stingless bees. Their behaviour, ecology and evolution. Series: Fascinating Life Sciences. Cham, Switzerland: Springer Nature; Cham, Switzerland. 385 pp. https://doi.org/10.1007/978-3-030-60090-7
- Vit P. (2001). Stingless bee honey and the treatment of cataracts. pp. 37–40. In: Munn P, Jones R. Honey and Healing. International Bee Research Association; Cardiff, UK. 49 pp.
- Vit P. (2005). Melissopalynology Venezuela. APIBA-CDCHT, Universidad de Los Andes; Mérida, Venezuela. 205 pp.
- Punt W, Hoen PP, Blackmore, et al. (2007). Glossary of pollen and spore terminology. Review of Palaeobotany and Palynology 143, 1–81. https://doi.org/10.1016/j.revpalbo.2006.06.008
- Tropicos.org (2022) Missouri Botanical Garden. https://www.tropicos.org
- Vit P, Wang Z, Massaro CF, Ekundayo TC. (2024). Global trends on plant resin use by stingless bees (1985–2022) and Apis mellifera (1967–2022) research: A bibliometric analysis. pp. 45–74. In: Vit P, Bankova V, Popova M, Roubik DW (Eds.), Stingless Bee Nest Cerumen and Propolis. Springer Nature; Cham, Switzerland. Volume 1, 535 pp.
- Shanahan M, Spivak M. (2021). Resin use by stingless bees: a review. Insects 12, 719. https://doi.org/10.3390/insects12080719
- Newis R, Nichols J, Farrar MB, Bai SH, Wilson RS, Wallace HM. (2023). Stingless bee (Tetragonula carbonaria) foragers prioritize resin and reduce pollen foraging after hive splitting. Apidologie 54. https://doi.org/10.1007/s13592-023-01018-4
- Moreno E, Vit P, Aguilar I, Barth OM. (2023). Melissopalynology of Coffea arabica honey produced by the stingless bee Tetragonisca angustula (Latreille, 1811) from Alajuela, Costa Rica. AlMS Agriculture and Food 8, 799–824. https://doi.org/10.3934/agrfood.2024029
- Louveaux J, Maurizio A, Vorwohl G. (1978). Methods of melissopalynology. Bee World 9, 139–157. https://doi.org/10.1080/0005772X.1970.11097312
- Layek U, Das N, Kumar De S, Karmakar P. (2023). The botanical origin of cerumen and propolis of Indian stingless bees (Tetragonula iridipennis Smith): pollen spectrum does not accurately indicate latex and resin sources. Apidologie 54. https://doi.org/10.1007/s13592-023-00994-1
- Popova M, Gerginova D, Trusheva B, Simova S, Tamfu AN, Ceylan O, Clark K, Bankova V. (2021). A preliminary study of chemical profiles of honey, cerumen, and propolis of the African stingless bee Meliponula ferruginea. Foods 10, 997. https://doi.org/10.3390/foods10050997
- Tomás-Barberán FA, García-Viguera C, Vit-Olivier P, Ferreres F, Tomás-Lorente F. (1993) Phytochemical evidence for the botanical origin of tropical propolis from Venezuela. Phytochemistry 34, 191–196. Ferreira IC, Côrrea RC, Orúe SL, Leite DF, da Rocha PD, Cardoso CA, Mussury RM, Vit P, de Picolli Souza K, dos Santos EL, Campos JF. (2023). Chemical components and antioxidant activity of Geotrigona sp. and Tetragonisca fiebrigi stingless bee cerumen reduce juglone-induced oxidative stress in Caenorhabditis elegans. Antioxidants 12, 1276. https://doi.org/10.3390/antiox12061276
- Othman ZA, Wan Ghazali WS, Noordin L, Mohd. Yusof NA, Mohamed M. (2019). Phenolic compounds and the anti-atherogenic effect of bee bread in high-fat diet-induced obese rats. Antioxidants 9, 33. https://doi.org/10.3390/antiox9010033
- Zakaria Z, Othman ZA, Suleiman JB, Mustafa KM, Jalil NA, Ghazali WS, Zulkipli NM, Mohamed M, Kamaruzaman KA. (2022). Therapeutic effects of Heterotrigona itama (stingless bee) bee bread in improving hepatic lipid metabolism through the activation of the Keap1/Nrf2 signaling pathway in an obese rat model. Antioxidants 11, 2190. https://doi.org/10.3390/antiox11112190
- Choudhari MK, Haghniazar R, Rajwade JM, Paknikar KM. (2013). Anticancer activity of Indian stingless bee propolis: an in vitro study. Evidence-Based Complementary and Alternative Medicine 2013. https://doi.org/10.1155/2013/928280
- Mahmood R, Asif JA, Shahidan WN. (2020). Stingless-bee (Trigona itama) honey adversely impacts the growth of oral squamous cell carcinoma cell lines (HSC-2). European Journal of Integrative Medicine 37, 101162. https://doi.org/10.1016/j.eujim.2020.101162
- Vit P. (2002). Effect of stingless bee honey in senile induced cataracts. Apiacta 3, 1–2.
- Vongsak B, Kongkiatpaiboon S, Jaisamut S, Machana S, Pattarapanoich C. (2015). In vitro alpha glucosidase inhibition and free-radical scavenging activity of propolis from Thai stingless bees in mangosteen orchard. Revista Brasileira de Farmacognosia 25, 445–450. https://doi.org/10.1016/j.bjp.2015.07.004
- Cheng Z, Shafiq MZ, Zawawi N, Der Ooi J, Chan KW, Ismail N, Ishak NA, Esa NM. (2023). In vitro Investigation of antioxidant and antidiabetic properties of phenolic-rich extract from stingless bee honey (Heterotrigona itama). Malaysian Journal of Medicine & Health Sciences 19, 6–19. https://doi.org/10.47836/mjmhs.19.6.19
- Farida S, Pratami DK, Sahlan M, Mun’im A, Djamal R, Wisart W, Ayub R, Alhamid TA, Rahmawati SI, Putra MY, Bayu A. (2023). In vitro study on antidiabetic and antihypertensive activities of ethanolic extracts of propolis of Indonesian stingless bee Tetragonula sapiens. Journal of King Saud University-Science 5, 102738. https://doi.org/10.1016/j.jksus.2023.102738
- Arung ET, Kusuma IW, Paramita S, Amen Y, Kim YU, Naibaho NM, Ramadhani R, Ariyanta HA, Fariastari W, Shimizu K. (2023). Antioxidant, anti-inflammatory and anti-acne activities of stingless bee (Tetragonula biroi) propolis. Fitoterapia 164, 105375. https://doi.org/10.1016/j.fitote.2022.105375
- Naibaho NM, Fariastari W, Kusuma IW, Arung ET. (2023). Phytochemical screening, antioxidant and anti-inflammatory properties of several stingless bee pollens processed using different drying methods. Uludağ Arıcılık Dergisi 23, 153–166. https://doi.org/10.31467/uluaricilik.1286430
- da Silva GP, da Silva TM, Cantara C, Queiroz M, Magnani M, de Novais JS, Soledade LE, de Oliveira EJ, Perônico D, Souza AG, de Souza AG. (2013). Sensory, physicochemical and antioxidant analysis of stingless bee honey from Amazonas, Northern Brazil. Food Chemistry 141, 3552–3558. https://doi.org/10.1016/j.foodchem.2013.06.076
- Mduda CA, Muruke MH, Hussein JM. (2023). Antimicrobial properties of honeys produced by stingless bees (Hymenoptera, Apidae, Meliponini) from different vegetation zones of Tanzania. International Journal of Tropical Insect Science 43, 1563–1581. https://doi.org/10.1007/s42690-023-01070-y
- Silva TM, De Souza SA, Dias TL, Silva TM, Falcão RA, Moreira MS, Silva EM, Camara CA. (2014). Chemical composition, antinociceptive and free radical-scavenging activities of geopropolis from Melipona subnitida Ducke (Hymenoptera: Apidae: Meliponini). Sociobiology 61, 560–565. https://doi.org/10.13102/sociobiology.v61i4.560-565
- Vit P, Yu JO, Huq F. (2013). Use of honey in cancer prevention and therapy. 481–493 pp. In: Vit P, Pedro SRM, Roubik (Eds.), Pot-honey: A legacy of stingless bees. Springer; New York, USA. 654 pp.
- Wesnail M, Hussin NN, Mazlan SN, Hussin NH, Radzi MN. (2018). Physicochemical analysis, antioxidant and antifungal activities of honey and propolis harvested fromhttps://doi.org/10.1016/S0031-9422(00)90804-5https://doi.org/10.3389/frma.2021.742311Wesnail M, Hussin NN, Mazlan SN, Hussin NH, Radzi MN (2018). Physicochemical analysis, antioxidant and antifungal activities of honey and propolis harvested from stingless bees. In IOP Conference Series: Materials Science and Engineering 440, 012048. https://doi.org/10.1088/1757-899X/440/1/012048
- Silva TM, Camara CA, da Silva Lins AC, Barbosa-Filho JM, da Silva EM, Freitas BM, dos Santos FD. (2006). Chemical composition and free radical scavenging activity of pollen loads from stingless bee Melipona subnitida Ducke. Journal of Food Composition and Analysis 19, 507–511. https://doi.org/10.1016/j.jfca.2005.12.011
- Mduda CA, Muruke MH, Hussein JM. (2023). Antimicrobial properties of honeys produced by stingless bees (Hymenoptera, Apidae, Meliponini) from different vegetation zones of Tanzania. International Journal of Tropical Insect Science 43, 1563–1581. https://doi.org/10.1007/s42690-023-01070-y
- Omar WA, Azhar NA, Fadzilah NH, Kamal NNM. (2016). Bee pollen extract of Malaysian stingless bees enhances the effect of cisplatin on breast cancer cell lines. Asian Pacific Journal of Tropical Biomedicine 6, 265–269. http://dx.doi.org/10.1016/j.apjtb.2015.12.011
- Yazan LS, Zail MFSN, Ali RM, Zainal NA, Esa N, Sapuan S, Ong YS, Tor YS, Gopalsamy B, Ling Voon FL, Alwi SSS. (2016). Chemopreventive properties and toxicity of kelulut honey in Sprague Dawley rats induced with azoxymethane. BioMed Research International 403692. http://dx.doi.org/10.1155/2016/403692
- Rebelo KS, Cazarin CB, Iglesias AH, Stahl MA, Kristiansen K, Carvalho-Zilse GA, Grimaldi R, Reyes FG, Danneskiold-Samsøe NB, Junior MR. (2021). Nutritional composition and bioactive compounds of Melipona seminigra pot-pollen from Amazonas, Brazil. Journal of the Science of Food and Agriculture 101, 4907–4915. https://doi.org/10.1002/jsfa.11134
- Zulkhairi AFMA, Sabri S, Ismail M, Chan KW, Ismail N, Mohd Esa N, Mohd Lila MA, Zawawi N. (2020). Probiotic properties of Bacillus strains isolated from stingless bee (Heterotrigona itama) honey collected across Malaysia. International Journal of Environmental Research and Public Health 17, 278. https://doi.org/10.3390/ijerph17010278
- Melia S, Aritonang SJN, Juliyarsi I, Kunia Y, Rusdimansyah R, Heminta YO. (2022). The screening of probiotic lactic acid bacteria from honey of stingless bee from West Sumatra, Indonesia and using as starter culture. Biodiversitas Journal of Biological Diversity 23. https://doi.org/10.13057/biodiv/d231235
- Vit P, Jacob TJ. (2008). Putative anticataract properties of honey studied by the action of flavonoids on a lens culture model. Journal of Health Science 54, 196–202. https://doi.org/10.1248/jhs.54.196
- Codex Stan. Standard for Honey. (1987). CXS 12-1981 Adopted in 1981. Revised in 1987, 2001. Amended in 2019. Codex Alimentarius. FAO, WHO. International Food Standards. 1981; pp. 1–8 (World-wide standard) Rev. 1. https://www.fao.org/3/w0076e30.htm Five languages https://www.fao.org/fao-who-codexalimentarius/
- Zawawi N, Zhang J, Hungerford NL, Yates AAS, Webber DC, Farrell M, Tinggi U, Bhandari B, Fletcher MT. (2022). Unique physicochemical properties and rare reducing sugar trehalulose mandate new international regulation for stingless bee honey. Food Chemistry 373, 131566. https://doi.org/10.1016/j.foodchem.2021.131566
- Popova M, Trusheva B, Bankova V. (2021). Propolis of stingless bees: A phytochemist’s guide through the jungle of tropical biodiversity. Phytomedicine 86, 153098. https://doi.org/10.1016/j.phymed.2019.153098
- Asem N, Abdul Qapar RA, Abd Hapiz NH, Omar EA. (2020). Correlation between total phenolic and flavonoid contents (measured by spectrophotometric methods) with antioxidant activity of Malaysian stingless bee propolis extract. Journal of Apicultural Research 5, 437–442. https://doi.org/10.1080/00218839.2019.1684050
- Mohamed WAS, Ismail NZ, Muhamad M, Omar EA, Samad NA, Ooi JP, Mohamad S. Q-TOF LC-MS compounds evaluation of propolis extract derived from Malaysian stingless bees, Tetrigona apicalis, and their bioactivities in breastcancer cell, MCF7. Saudi Journal of Biological Sciences 2022; 29: 103403.
- Syed Salleh SNA, Mohd Hanapiah NA, Ahmad H, Wan Johari WL, Osman NH, Mamat MR. Determination of total phenolics, flavonoid content, antioxidant activity and GC-MS analysis of Malaysian stingless bee propolis water extracts. Scientifica 2021, 3789531. https://doi.org/10.1155/2021/3789531Gonnet M, Lavie P, Nogueira-Neto P. Étude de quelques charactéristiques des miels récoltés par certains Méliponines brésiliens. Comptes Rendus de l’Académie des Sciences Paris 1964; 258: 3107–3109. https://doi.org/10.1111/j.1096-3642.1955.tb00591.x
- ADAF. Agência de Defesa Agropecuária e Florestal do Estado do Amazonas. Portaria ADAF nº 253 de 31 de outubro de 2016. Regulamento Técnico de Identidade e Qualidade do Mel de Abelha Social Sem Ferrão para o Estado do Amazonas. Brazil. 2016; 1–9.
- ADAPAR. Agência de Defesa Agropecuária do Paraná. Portaria N° 63, de 10 de março de 2017. Regulamento Técnico de Identidade e Qualidade do Mel de Abelhas Sem Ferrão para o Estado do Paraná, Brazil. 2017; 1–9 pp.
- IDAF. Instituto de Defesa Agropecuária e Florestal do Espírito Santo. Instrução Normativa n° 01, de 17 de abril de 2019. Regulamento Técnico de Identidade e Qualidade do Mel de Abelhas Sem Ferrão para o Estado do Espírito Santo, Brazil. 2019; 1–7 pp.
- SAR. Secretaria de Estado da Agricultura e da Pesca e do Desenvolvimento Rural. Portaria SAR n° 37/2020, de 04/11/2020. Decreto Estadual n° 39. 2020. Norma Interna Regulamentadora do Mel de Abelhas Sem Ferrão no Estado de Santa Catarina, Brazil. 2020; 16–22.
- Philippine National Standard PNS/BAFS 185:2016 ICS. Bureau of Agriculture and Fisheries Standards. Honey. Department of Agriculture; Quezon City, Philippines. 2016; 10 pp.
- Department of Standards Malaysia. Kelulut (Stingless bee) honey – Specification MS 2683: 2017. Available at https://es.scribd.com/document/398215369/Kelulut-Stingless-bee-honey-Specification
- Secretaría de Regulación y Gestión Sanitaria y Secretaría de Alimentos y Bioeconomía. Miel de Tetragonisca fiebrigi (yate). Resolución Conjunta 17/2019 RESFC-2019- 17- APN-SRYGS#MSYDS 02/05/2019 Y N 29258/19 v. 02/05/2019. 2019; 1–8.
- Vit P, Aguilar I, Chuttong B, Barth OM, Cervancia C, Loscan A, Baroqa-Barbecho J, Rebelo KS, Vossler F, Thanh LN, Karmakar P, Layek U, Meccia G, Guevara P, Maza F, Rasmussen C, Simeon I, Halcroft M, Yurita Obis CL, Loayza S, Infante J, Pérez A, Nicolás AZ, Adim MZ, Carrucci MC, Nogueira DS, Ortiz-Vázquez E, Mduda C, Wang Z, Ramírez-Arriga E, Bankova V. Nutraceutical and health benefits of meliponine honey, pollen, cerumen, and propolis. Female stingless bee keeping and pharmaceutical developments. Current Research in Insect Science (submitted). 2024b.
- Vit P, Gutiérrez MG, Tièra D, Bednář M, Rodríguez-Malaver AJ. Mieles checas categorizadas según su actividad antioxidante. Acta Bioquímica Clínica Latinoamericana 2008; 42: 237–244.
- Fletcher M, Hungerford NL, Webber D, de Jesus MC, Zhang J, Stone ISJ, Zawawi N. Stingless bee honey, a novel source of trehalulose: a biologically active disaccharide with health benefits. Scientific Reports 2020; 10, 12128. https://doi.org/10.1038/s41598-020-68940-0
- Vit P. Curso Calidad de la Colmena para la Apiterapia. VII Congreso Nacional de Ciencias Farmacéuticas. Facultad de Farmacia y Bioanálisis, Universidad de Los Andes Mérida, Venezuela. 2000 https://www.apiculture.com/en/?preview=1&option=com_dropfiles&format=&task=frontfile.download&catid=29&id=1186&Itemid=1000000000000
- Vit P, Simova S. (2023). The Review on Aliphatic Organic Acids (AOA) of Honey and Pot-honey for Bee Science. E-book. APIBA, CDCHTA-ULA, Universidad de Los Andes, Mérida, Venezuela. 70 pp. http://www.saber.ula.ve/bitstream/handle/123456789/49623/review_AOA_Vit_Simova.pdf?sequence=4&isAllowed=y
- WHO. (2022). WHO Global antimicrobial resistance and use surveillance system (GLASS) report 2022. World Health Organization; Geneva, Switzerland. 71 pp. https://www.who.int/publications/i/item/9789240062702
- Antimicrobial Resistance Collaborators. (2022). Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet 399(10325), 629–655.Vit P, Simova S. (2023). The Review on Aliphatic Organic Acids (AOA) of Honey and Pot-honey for Bee Science. E-book. APIBA, CDCHTA-ULA, Universidad de Los Andes, Mérida, Venezuela. 70 pp. http://www.saber.ula.ve/bitstream/handle/123456789/49623/review_AOA_Vit_Simova.pdf?sequence=4&isAllowed=y
- WHO. (2022). WHO Global antimicrobial resistance and use surveillance system (GLASS) report 2022. World Health Organization; Geneva, Switzerland. 71 pp. https://www.who.int/publications/i/item/9789240062702
- Antimicrobial Resistance Collaborators. (2022). Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet 399(10325), 629–655.Vit P, Simova S. (2023). The Review on Aliphatic Organic Acids (AOA) of Honey and Pot-honey for Bee Science. E-book. APIBA, CDCHTA-ULA, Universidad de Los Andes, Mérida, Venezuela. 70 pp. http://www.saber.ula.ve/bitstream/handle/123456789/49623/review_AOA_Vit_Simova.pdf?sequence=4&isAllowed=y
- WHO. (2022). WHO Global antimicrobial resistance and use surveillance system (GLASS) report 2022. World Health Organization; Geneva, Switzerland. 71 pp. https://www.who.int/publications/i/item/9789240062702
- Antimicrobial Resistance Collaborators. (2022). Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet 399(10325), 629–655.Salam MA, Al-Amin MY, Salam MT, Pawar JS, Akhter N, Rabaan AA, Alqumber MAA. (2023). Antimicrobial resistance: A growing serious threat for global public health. Healthcare 11, 1946. https://doi.org/10.3390/healthcare11131946
- WHO. (2024). WHO Bacterial Priority Pathogens List, 2024: bacterial pathogens of public health importance to guide research, development and strategies to prevent and control antimicrobial resistance. Geneva: World Health Organization; 56 pp. https://www.who.int/publications/i/item/9789240093461
- Mancuso G, Midiri A, Gerace E, Biondo C. (2021). Bacterial Antibiotic resistance: the most critical pathogens. Pathogens 10, 1310. https://doi.org/10.3390/pathogens10101310
- Szweda P. (2021). Editorial for the Special Issue: “Honey Bee Products as an Alternative or Complement to Classical Antibiotics”. Antibiotics (Basel) 10, 234. https://doi.org/10.3390/antibiotics10030234
- Ratajczak M, Kaminska D, Matuszewska E, Holderna-Kedzia M, Jjorack KJ, Janyszek M. (2021). Promising antimicrobial properties of bioactive compounds from different honeybee products. Molecules 26, 4007. https://doi.org/10.3390/molecules26134007
- Vit P, Araque M, Chuttong B. (2024). A multifaceted bioactive resource of stingless bees: Unlocking the therapeutic anti-antimicrobial-resistance (anti-AMR) potential of pot-pollen. Medical Research Archives 12(10). https://doi.org/10.18103/mra.v12i10.5864
- Vit P, Ricciardelli D’Albore G, Barth OM, Peña-Vera M, Pérez-Pérez E. (2018). Characterization of pot-pollen from Southern Venezuela. pp. 361–375. In P. Vit, SRM Pedro, DW Roubik (editors), Pot-pollen in Stingless Bee Melittology. Springer Nature; Cham, Switzerland; 481 pp. https://doi.org/10.1007/978-3-319-61839-5_26
- Subarán-Mora M, Peña-Vera M, Pérez-Pérez E, Vit P. (2018). Antibacterial activity of ethanolic extracts of pot-pollen from eight meliponine species from Venezuela. pp. 391–399. In P. Vit, SRM Pedro, DW Roubik (editors), Pot-pollen in Stingless Bee Melittology. Springer Nature; Cham, Switzerland; 481 pp. https://doi.org/10.1007/978-3-319-61839-5_28
- Ogwu, MC, Izah, SC. (2025). Honey as a natural antimicrobial. Antibiotics 14, 255. https://doi.org/10.3390/antibiotics14030255
- Bava R, Puteo C, Lombardi R, Garcea G, Lupia C, Spano A, Liguori G, Palma E, Britti D, Castagna F. (2025). Antimicrobial properties of hive products and their potential applications in human and veterinary medicine. Antibiotics 14, 172. https://doi.org/10.3390/antibiotics14020172
- Araque M, Vit P. (2024). Evaluation of the potential synergistic effect of pot-pollen with amikacin and meropenem against extensively drug-resistant bacteria of clinical origin. Medical Research Archives 12(9). https://doi.org/10.18103/mra.v12i9.5924
- Vit P, Araque M, Chuttong B, Moreno E, Contreras RR, Wang Q, Wang Z, Betta E, Bankova V., (2024). Pot-Pollen volatiles, bioactivity, synergism with antibiotics, and bibliometrics overview, including stingless bee nests. Food 13, 3879. https://doi.org/10.3390/foods13233879
- Vit P, Chuttong B, P Aguilar I, Barth OM, Sarmiento Rebelo K, Vossler F, Thanh LN, Karmakar P, Layek U, Meccia G, Ramírez-Arriaga E, Maza F, Rasmussen C, Kimoli S, Halcroft M, Yurita Obis CL, Loayza S, Infante J, Pérez A, Nicolás A, Azmi WA, Marcucci MC, Silva Nogueira D, Ortiz-Vázquez E, Mduda C, Wang Z, Araque M, Magalhães Freitas B, Bankova B. (2025). Phytochemicals and nutraceutical potential of stingless bee nest biomaterials: the emerging role of female stingless bees keeping in global food security and pharmaceutical development. Frontiers in Nutrition 12. https://doi.org/10.3389/fnut.2025.1573821
- Salatino A. (2022). Perspectives for uses of propolis in therapy against infectious diseases. Molecules 27, 4594. https://doi.org/10.3390/molecules27144594
- Budzynski K. (2021). Honey as an ecological reservoir of antibacterial compounds produced by antagonistic microbial interactions in plant nectars, honey and bees. Antibiotics 10, 551. https://doi.org/10.3390/antibiotics10050551
- Vit P, Chuttong B, Ramírez-Arriaga E, Enríquez E, Wang Z, Cervancia C, Vossler F, Kimooli S, Engel MS, Contreras RR, Mduda CA, Tomás-Barberán F. (2025). Stingless bee honey: Nutraceutical properties… and urgent call for proposed global standards. Trends in Food Science & Technology 157, 104844. https://doi.org/10.1016/j.tifs.2024.104844
- Mduda CA, Vit P, Rikohe IF, Lukiko SB. (2025). Axestotrigona ferruginea pot-pollen from Tanzania: a high-value nutritional supplement with antioxidant potential. Journal of Apicultural Research 1–12. https://doi.org/10.1080/00218839.2025.2559466
- Ferreira IC, Côrrea RCD, Orúe SL, Leite DF, da Rocha PdS, Cardoso CAL, Mussury RM, Vit P, de Picoli Souza K, dos Santos EL, Campos JF. (2023). Chemical components and antioxidant activity of Geotrigona sp. and Tetragonisca fiebrigi stingless bee cerumen reduce juglone-induced oxidative stress in Caenorhabditis elegans. Antioxidants 12, 1276. https://doi.org/10.3390/antiox12061276
- Chuttong B, Lim K, Praphaiwail P, DanneK K, Maitip J, Vit P, Wu M-C., Ghosh S, Jung C, Burgett M, Hongsibsong S. (2023). Exploring the functional properties of geopropolis, geopropolis, and cerumen with a special emphasis on their antimicrobial effects. Food 12, 3909. https://doi.org/10.3390/foods12213909
- Vit P. (2024). Metabolites from microbial cell factories in stingless bee nests. pp. 53–114 pp. In P Vit, V Bankova, M Popova, DW Roubik (editors), Stingless Bee Nest Cerumen and Propolis. Springer Nature; Cham, Switzerland; Volume 2, 501 pp. https://doi.org/10.1007/978-3-031-43887-5_4
- Alves VF, Chaul LT, Bueno, GCA, Reinecke I, Silva TCG, Brito PVA, De Martins ECP. (2024). Associated bacterial microbiota of honey and related products from stingless bees as novel sources of bioactive compounds for biotechnological applications. Current Opinion in Food Science 55, 101122. https://doi.org/10.1016/j.cofs.2023.101122
- Hammer TJ, Calabria AE, Moran NA. (2023). Microbiome assembly and maintenance across the lifespan of bumble bee workers. Molecular Ecology 32(3), 724–740. https://doi.org/10.1111/mec.16769
- Tarlinton B, Massaro FC, Hauxwell C. (2023). 16S Amplicon metabarcoding of the nest materials of native Australian stingless bees. Microbiological Resources Announcement 12, e0118122. https://doi.org/10.1128/mra.01181-22
- Meireles SD, dos Santos SF, Rafael MS, da Mota AJ, da Silva CGN. (2022). Yeasts from the nests of two Amazonian stingless bees: screening and PCR-RFLP molecular analysis. Symbiosis 87, 153–163. https://doi.org/10.1007/s13199-022-00865-w
- de Paula GT, Melo WGDP, de Castro I, Menezes C, Paludo CR, Rosa CA, Pupo MT. (2023). Further evidences of an emerging stingless bee-yeast symbiosis. Frontiers in Microbiology 14, 1221724. https://doi.org/10.3389/fmicb.2023.1221724
- Ramírez-Ahuja MDL, Peña-Carrillo KI, Gómez-Govea MA, Jiménez-Martínez ML, Trujillo-Rodríguez GJ, Espinoza-Ruiz M, Guzmán Velasco A, Flores AE, González-Rojas JI, Reséndez-Pérez D, Rodríguez-Sánchez IP. (2025). Gut Microbiota Diversity in 16 Stingless Bee Species (Hymenoptera: Apidae: Meliponini). Microorganisms 13, 1645. https://doi.org/10.3390/microorganisms13071645
- Castillo DC, Sinpooc C, Phokasem P, Castillo DC, Yongsawas R, Sansupa C, Attasopa K, Suwannarachen R, Wiwawang S, Noirungsree N, Disayathaworn T. (2024). Distinct fungal communities of pollen pots of stingless bees, Lepidotrigona terminata and Tetragonula pagdeni, and their possible roles in pot maturation. Frontiers in Microbiology 14, 1367010. https://doi.org/10.3389/fmicb.2024.1367010
- Selma MV, Espín JC, Tomas-Barberan FA. (2000). Interaction between phenolics and gut microbiota: role in human health. Journal of Agricultural and Food Chemistry 57, 6485–6501. https://doi.org/10.1021/jf902170d
- Paludo CR, Menezes C, Silva EA, Vollet-Neto A, Andrade Dominguez A, Pishchany G, Khadempour L, Nascimento FS, Currie CR, Koter R, Clardy J., Pupo, M.T. (2018). Stingless bee larvae require fungal steroid to pupate. Scientific Reports 8,1122. https://doi.org/10.1038/s41598-018-19583-9
- Paludo CR, Pishchany G, Andrade-Dominguez A, Silva EA, Menezes C, Nascimento FS, Currie CR, Kolter R, Clardy J, Pupo MT. (2019). Microbial community modulates growth of symbiotic fungus required for stingless bee metamorphosis. PLoS One 14, e0217966. https://doi.org/10.1371/journal.pone.0217966
- Atanasov AG, Zotchev SB, Dirsch VM, International Natural Product Sciences Taskforce, Supuran CT. (2021). Natural products in drug discovery: Advances and opportunities. Nature Reviews Drug Discovery, Hawkins J, de Vere N, Griffith A, Ford CR, Allainguillaume J, Hegarty MJ, Baillie L, Adams-Groom B. (2015) Using DNA metabarcoding to identify the floral composition of honey: A new tool for investigating honey bee foraging preferences. PLoS ONE 10, e0134735. https://doi.org/10.1371/journal.pone.0134735
- Bovo S, Utzeri VJ, Ribani A, Cabbri R, Fontanesi L. (2020). Shotgun sequencing of honey DNA can describe honey bee derived environmental signatures and the honey bee hologenome complexity. Scientific Reports 10, 9279. https://doi.org/10.1038/s41598-020-66127-1
- Galanis A, Vardakas P, Reczko M, Harokopos V, Hatzis P, Skoulikas EMC, Pavlopoulos GA, Patalano S. (2022). Bee foraging preferences, microbiota and pathogens revealed by direct shotgun metagenomics of honey. Molecular Ecology Resources 22, 2506–2523. https://doi.org/10.1111/1755-0998.13626
- Paluoja P, Vaher M, Teder H, Krijustköv K, Salumets A, Raime K. (2025). Honey bulk DNA shotgun analysis reveals species composition and monitor honey bee pathogens. NPJ Science of Food 9, 91. https://doi.org/10.1038/s41538-025-00464-1
- Usyk M, Peters BA, Karthikeyan S, McDonald D, Sollecito CC, Vazquez-Baeza Y, Shaffer JP, Gelman MD, Talavera GA, Daviglus ML, Thyagarajan B, Knight R, Qi O, Kaplan R, Burk RD. (2023). Comprehensive evaluation of shotgun metagenomics, amplicon sequencing, and harmonization of these platforms for epidemiological studies. Cell Reports Methods 3, 100391. https://doi.org/10.1016/j.crmeth.2022.100391
- Edwards D, Stajich JE, Hansen D. (2009). DNA Sequence Databases. pp. 1–11. In D Edwards, JE Stajich, D Hansen (editors), Bioinformatics: Tools and Applications, Springer; NYC, United States. 451 pp.
- Yugueros SI, Peláez J, Stajich JE, Fuertes-Rabanal M, Sánchez-Vallet A, Largo-Gosens A, Mélida H. (2024). Study of fungal cell wall evolution through its monosaccharide composition: An insight into fungal species interacting with plants. The Cell Surface 11, 100127. https://doi.org/10.1016/j.tcsw.2024.100127
- Truchado P, Vit P, Ferreres F, Tomas-Barberan F. (2011). Liquid chromatography-tandem mass spectrometry analysis allows the simultaneous characterization of C-glycosyl and O-glycosyl flavonoids in stingless bee honeys. Journal of Chromatography A 1218, 7601–7607. https://doi.org/10.1016/j.chroma.2011.07.049
- Pérez-Pérez EM, Suárez E, Peña-Vera MJ, Vit P. (2013). Antioxidant activity of nest products of Tetragonisca angustula from Mérida, Venezuela. In: Vit P, Roubik DW, editors. Stingless bees process honey and pollen in cerumen pots. Mérida, Venezuela: Facultad de Farmacia y Bioanálisis, Universidad de Los Andes; pp. 1–6. http://www.saber.ula.ve/handle/123456789/35292
- Pazin WM, LM, Mónaco LM, Soares E, Miguel FG, Berretta A, Ito AS. (2017). Antioxidant activities of three stingless bee propolis and green propolis types. Journal of Apicultural Research 56, 1–6. https://doi.org/10.1080/00218839.2016.1263496
- de Souza SA, da Silva TM, da Silva EM, Camara CA, Silva TM. (2018). Characterization of geopropolis using UPLC-QTOF-MS/MS of geopropolis from the stingless bee Melipona subnitida (jandaíra). Phytochemical Analysis 29, 549–558. https://doi.org/10.1002/pca.2758
- Belina-Aldemita MD, Schreiner M, D’Amico S. (2020). Characterization of phenolic compounds and antioxidative potential of pot-pollen produced by stingless bees (Tetragonula biroi Friese) from the Philippines. Journal of Food Biochemistry 44, e13102. https://doi.org/10.1111/jfbc.13102
- Oliveira RG, Jain S, Freitas LD, Araújo ED. (2019). Phenolic compound, nutritional and antioxidant profile of pollen collected by the genus Melipona in North Eastern Brazil. Brazilian Journal of Food Technology 22. https://doi.org/10.1590/1981-6723.09178
- Biluca FC, da Silva B, Caon T, Mohr E, Vieira GN, Gonzaga LV, Vitali L, Micke G, Fett R, Dalmarco EM, Costa AC. (2020). Investigation of phenolic compounds, antioxidant and anti-inflammatory activities in stingless bee honey (Melipona quadrifasciata). Food Research International 192, 108756. https://doi.org/10.1016/j.foodres.2019.108756
- Majid M, Ellulu M, Abu Bakar MF. (2020). Melissopalynology and chemical compositionDira S, Tawiri M, Dian Hastuti R, Bankova V, Popova M, Vit P. (2020). Melissopalynology and antioxidant properties of Heterotrigona itama honey from Johor, Malaysia. Scientifica 2020. https://doi.org/10.1155/2020/2529592
- Dutra RP, Abreu BV, Cunha MS, Batista MC, Torres LM, Nascimento FR, Ribeiro MN, Guerra RN. (2014). Phenolic acids, hydrolyzable tannins, and antioxidant activity of geopropolis from the stingless bee Melipona fasciculata Smith. Journal of Agricultural and Food Chemistry 62, 2549–2557. https://doi.org/10.1021/jf404875v
- Iesa NB, Chaipoot S, Phongphisuthinant R, Wiriyacharee P, Lim BG, Sringarm K, Burgett M, Chuttong B. (2023). Effects of maltodextrin and gum arabic composition on the physical and antioxidant activities of dewaxed stingless bee cerumen. Foods 12, 3740. https://doi.org/10.3390/foods12203740
- Mohd Suib MS, Wan Omar WA, Omar EA, Mohamed R. (2021). Ethanolic extract of propolis from the Malaysian stingless bee Geniotrigona thoracica inhibits formation of THP-1 derived macrophage foam cells. Journal of Apicultural Research 60, 478–490. https://doi.org/10.1080/00218839.2020.1720125
- Kustiwan PM, Puthong S, Arung ET, Chanchoo C. (2014). In vitro cytotoxicity of Indonesian stingless bee propolis against human cancer cell lines. Asian Pacific Journal of Tropical Biomedicine 4, 549–556. https://doi.org/10.12980/APJTB.4.2014APJTB-2013-0039
- Nugitrangson P, Puthong S, Iempridee T, Pimtong W, Pornpakakul S, Chanchoo C. (2016). In vitro and in vivo characterization of the anticancer activity of Thai stingless bee (Tetragonula laeviceps) cerumen. Experimental Biology and Medicine 241, 166–176. https://doi.org/10.1177/1535370215600102
- dos Santos HF, Campos JF, Santos CM, Balestieri JB, Silva DB, Carollo CA, de Picoli Souza K, Estevinho LM, dos Santos EL. (2017). Chemical profile and antioxidant, anti-inflammatory, antimutagenic and antimicrobial activities of geopropolis from the stingless bee Melipona orbignyi. International Journal of Molecular Sciences 18, 953. https://doi.org/10.3390/ijms18050953
- Ahmad F, Seeranpan P, Mustafa MZ, Zul Faizuddin Osman ZF, Jafri Malin Abdullah JM, Idris Z. (2018). in vivo anti-apoptotic properties of Heterotrigona itama sp. honey in the induction of apoptosis in malignant glioma cells. Malaysian Journal of Medical Sciences 26, 30–39. https://doi.org/10.21315/mjms2019.26.2.4
- Desamero MJ, Kakuta S, Tang Y, Chambers JK, Uchida K, Estacio MA, Cervancia C, Kominami Y, Ushio H, Nakayama J, Nakayama H. Tumor-suppressing potential of stingless bee propolis in in vitro and in vivo models of differentiated-type gastric adenocarcinoma. Scientific Reports 9, 19635. https://doi.org/10.1038/s41598-019-55465-4
- Arung ET, Ramadhan R, Khairunnisa B, Amen Y, Matsumoto M, Nagata M, Kusuma IW, Paramita S, Sandirogang N, Takemoto N, Kim YU. (2021). Cytotoxicity effect of honey, bee pollen, and propolis from seven stingless bees in some cancer cell lines. Saudi Journal of Biological Sciences 28, 7182–7189. https://doi.org/10.1016/j.sjbs.2021.08.017
- Vit P. (1997). Cataratas y Mieles Terapéuticas. Consejo de Desarrollo Científico, Humanístico y Tecnológico, Universidad de Los Andes; Mérida, Venezuela. 167 pp.
- Abdul Aziz MS, Girinabu N, Ch’ng ES, Ain MF. (2017). Pancreatoprotective effects of Geniotrigona thoracica cerumen on nicotinamide-induced male diabetic rats. Biomedicine & Pharmacotherapy 89, 135–145. https://doi.org/10.1016/j.biopha.2017.02.026
- Nna VU, Bakar AB, Lazin MR, Mohamed M. (2018). Antioxidant, anti-inflammatory and synergistic anti-hyperglycemic effects of Malaysian propolis and metformin in streptozotocin-induced diabetic rats. Food and Chemical Toxicology 120, 305–320. https://doi.org/10.1016/j.fct.2018.07.022
- Pujirahayu N, Bhattacharjya DK, Suzuki T, Katayama T. (2019). α-Glucosidase inhibitory activity of cycloartane-type triterpenes isolated from Indonesian stingless bee propolis and their structure–activity relationship. Pharmaceuticals 12, 102. https://doi.org/10.3390/ph12030102
- Rahmawati OP, Pratami DK, Rafiudin R, Sahlan M. (2019). Alpha-glucosidase inhibitory activity of stingle bees honey from Tetragonula biroi and Tetragonula laeviceps. AIP Conference Proceedings 2092. https://doi.org/10.1063/1.5096705
- Ali H, Abu Bakar MF, Majid M, Muhammad N, Lim SY. (2020). In vitro anti-diabetic activity of stingless bee honey from different botanical origins. Food Research 4, 1421–1426. Pujirahayu N, Bhattacharjya DK, Suzuki T, Katayama T. (2019). α-Glucosidase inhibitory activity of cycloartane-type triterpenes isolated from Indonesian stingless bee propolis and their structure–activity relationship. Pharmaceuticals 12, 102. https://doi.org/10.3390/ph12030102
- Rahmawati OP, Pratami DK, Rafiudin R, Sahlan M. (2019). Alpha-glucosidase inhibitory activity of stingle bees honey from Tetragonula biroi and Tetragonula laeviceps. AIP Conference Proceedings 2092. https://doi.org/10.1063/1.5096705
- Ali H, Abu Bakar MF, Majid M, Muhammad N, Lim SY. (2020). In vitro anti-diabetic activity of stingless bee honey from different botanical origins. Food Research 4, 1421–1426. Ali H, Abu Bakar MF, Majid M, Muhammad N, Lim SY. (2020). In vitro anti-diabetic activity of stingless bee honey from different botanical origins. Food Research 4, 1421–1426. https://doi.org/10.26656/fr.2017.4(5).1470https://doi.org/10.26656/fr.2017.4(5).1470https://doi.org/10.26656/fr.2017.4(5).1470Campos JF, Santos UP, Rocha PD, Damião MJ, Balestieri JB, Cardoso CA, Paredes-Gamero EJ, Estevinho LM, de Picoli Souza K, Santos EL. (2015). Antimicrobial, antioxidant, anti-inflammatory, and cytotoxic activities of propolis from the stingless bee Tetragonisca fiebrigi (Jataí). Evidence-Based Complementary and Alternative Medicine 2015. https://doi.org/10.1155/2015/296186
- Hamilton KD. (2016). Evaluation of the anti-inflammatory, anti-oxidant and wound-healing potential of cerumen from the Australian native stingless bee, Tetragonula carbonaria. Doctoral dissertation, University of the Sunshine Coast; Sippy Downs QLD, Australia. 213 pp. https://doi.org/10.25907/00591
- Hamilton KD, Brooks PR, Ogbourne SM, Russell FD. (2017). Natural products isolated from Tetragonula carbonaria cerumen modulate free radical-scavenging and 5-lipoxygenase activities in vitro. BMC Complementary and Alternative Medicine 17, 8. https://doi.org/10.1186/s12906-017-1784-6
- Lopes AJ, Vasconcelos CC, Pereira FA, Silva RH, Queiroz PF, Fernandes CV, García JB, Ramos RM, Rocha TD, Sinnott ST. (2019). Cartagenes MD. Anti-inflammatory and antinociceptive activity of pollen extract collected by stingless bee Melipona fasciculata. International Journal of Molecular Sciences 20, 4512. https://doi.org/10.3390/ijms20184512
- Badrulhisham NS, Ab Hamid SN, Ismail MA, Yong YK, Zakuan MN, Harith HH, Saidi HI, Nurdin A. (2020). Harvested locations influence the total phenolic content, antioxidant levels, cytotoxic, and anti-inflammatory activities of stingless bee honey. Journal of Asia-Pacific Entomology 23, 950–956. https://doi.org/10.1016/j.aspen.2020.07.015
- Barboza JR, Pereira FA, Fernandes RA, Vasconcelos CC, Cartagenes MD, Oliveira Lopes AJ, Melo AC, Guimarães ID, Rocha CQ, Ribeiro MN. (2020). Cytotoxicity and pro-apoptotic, antioxidant and anti-inflammatory activities of geopropolis produced by the stingless bee Melipona fasciculata Smith. Biology 9, 292. https://doi.org/10.3390/biology9090292
- Zhang W, Cai Y, Chen X, Ji T, Sun L. Optimized extraction based on the terpenoids of Heterotrigona itama propolis and their antioxidative and anti-inflammatory activities. Journal of Food Biochemistry 44, e13296. https://doi.org/10.1111/jfbc.13296
- Ooi TC, Yaacob M, Rajab NF, Shahar S, Sharif R. (2021). The stingless bee honey protects against hydrogen peroxide-induced oxidative damage and lipopolysaccharide-induced inflammation in vitro. Saudi Journal of Biological Sciences 28, 2987–2994. https://doi.org/10.1016/j.sjbs.2021.02.039
- Hamilton KD, Czajkowski D, Kong NJ, Tran TD, Gustafson KR, Pauly G, Boyle GM, Simmons JL, Steadman R, Moseley R, Brooks PR. (2022). Antifibrotic potential of Tomentosenol A, a constituent of cerumen from the Australian native stingless bee Tetragonula carbonaria. Antioxidants 11, 1604. https://doi.org/10.3390/antiox11081604
- Wu MC, Wu CY, Klaithin K, Tiong KK, Peng CC. (2022). Effect of harvest time span on physicochemical properties, antioxidant, antimicrobial, and anti-inflammatory activities of Meliponinae honey. Journal of the Science of Food and Agriculture 102, 5750–5758. https://doi.org/10.1002/jsfa.11942
- Asma’ A, Zakaria R, Ibrahim WN, Zaini SN, Taha H, Hasni F, Usman A. (2020). Mineral contents, antioxidants, and antimicrobial activities of propolis produced by Bruneian stingless bees Geniotrigona thoracica, Heterotrigona itama, and Tetrigona binghami. Saudi Journal of Biological Sciences 27, 2902–2911. https://doi.org/10.1016/j.sjbs.2020.09.014
- Dallagnol AM, Dallagnol VC, Vignolo GM, Lopes NP, Brunetti AE. (2022). Flavonoids and phenylethylamides are pivotal factors affecting the antimicrobial properties of stingless bee honey. Journal of Agricultural and Food Chemistry 70, 12596–12603. https://doi.org/10.1021/acs.jafc.2c04120
- Naibaho NM, Salusu HD, Rudito R, Saragih B, Kusuma IW, Fariasari W, Arung ET. (2023). Sensory evaluation and antibacterial activity of bee pollen extracts isolated from several stingless bees in two drying methods. Biodiversitas Journal of Biological Diversity 24. https://doi.org/10.13057/biodiv/d240521
- Rodríguez-Malaver AJ, Pérez-Pérez EM, Vit P. (2007). Capacidad antioxidante de mieles venezolanas de los géneros Apis, Melipona y Tetragonisca, evaluada por métodos de FRAP. Revista del Instituto Nacional de Higiene Rafael Rangel 38, 13–18.Nurdianah HF, Firdaus AA, Azam OE, Adnan WW. (2016). Antioxidant activity of bee pollen ethanolic extracts from Malaysian stingless bees measured using DPPH-HPLC assay. International Food Research Journal 23, 403. https://www.ifrj.upm.edu.my/23%20(01)%202016/(59).pdf
- Harif Fadzilah N, Jaapar MF, Jajuli R, Wan Omar WA. (2017). Total phenolic content, total flavonoid and antioxidant activity of ethanolic bee pollen extracts from three species of Malaysian stingless bee. Journal of Apicultural Research 56, 130–135. https://doi.org/10.1080/00218839.2017.1287996
- Carneiro AL, Gomes AA, Alves da Silva L, Alves LB, Cardoso da Silva E, da Silva Pinto AC, Tadei WP, Pohlit AM, Simas Teixeira MF, Gomes CC, Naiff MD. (2019). Antimicrobial and larvicidal activities of stingless bee pollen from Maues, Amazonas, Brazil. Bee World 96, 98–103. https://doi.org/10.1080/0005772X.2019.1650564
- Mokaya HO, Nkoba K, Ndunza RM, Vereecken NJ. (2022). Characterization of honeys produced by sympatric species of Afrotropical stingless bees (Hymenoptera, Meliponini). Food Chemistry 366, 130597. https://doi.org/10.1016/j.foodchem.2021.130597
- Mohammad SM, Mahmud-Ab-Rashid NK, Zawawi N. (2020). Probiotic properties of bacteria isolated from bee bread of stingless bee Heterotrigona itama. Journal of Apicultural Research 60, 172–87. https://doi.org/10.1080/00218839.2020.1801152
- Alves RMO. (2013). Production and marketing of pot-honey. 541–556 pp. In: Vit P, Pedro SRM, Roubik D, editors. Pot-Honey. A Legacy of Stingless Bees. Springer; New York, USA. 654 pp.
- Alves RMO, Carvalho CAL. (2018). Pot-pollen ‘samburá’ making in Brazil and suggested legislation. 435–443 pp. In: Vit P, Pedro SRM, Roubik D, editors. Pot-Pollen in Stingless Bee Melittology. Springer Nature; Cham, Switzerland. 481 pp.
- Yong CH, Muhammad SA, Nasir FI, Mustafa MZ, Ibrahim B, Kelly SD, Cannavan A, Seow EK. (2022). Detecting adulteration of stingless bee honey using untargeted ¹H NMR metabolomics with chemometrics. Food Chemistry 368, 130808.
- Vit P. (1993). Miel de Abejas. Cuaderno Ciencia de los Alimentos No. 1. Consejo de Publicaciones ULA; Mérida, Venezuela. 97 pp.
- Vit P. (1998). A test to detect cane sugar honey. Archivos Latinoamericanos de Nutrición 48, 62–64.

