Acute Kidney Injury in Congenital Heart Surgery: Insights
Newer insights into acute kidney injury following congenital cardiac surgery using staging criteria & biomarker analysis
Gananjay G. Salve, M.Ch.,¹ Madhavi Putta, M.B.B.S.,² Darshan D.S., M.Ch.,¹ Nidhi G. Manvi, DNB.,³ Veeresh Manvi, FNB.,³ Danish A.K. Memon, D.M.,³ Sonali Bijjargi, M.D.,² Abhishek Prabhu, M.Ch.,¹ Parishwanath Patil, M.Ch.,¹ Sharanagouda Patil, M.D.,⁴ Mahantesh V. Patil, M.D.²
- Department of Cardiovascular & Thoracic Surgery, Jawaharlal Nehru Medical College, KLES Academy of Higher Education & Research, Belgaum, 590010, Karnataka, India
- Division of Paediatric Nephrology, Jawaharlal Nehru Medical College, KLES Academy of Higher Education & Research, Belgaum, 590010, Karnataka, India
- Department of Paediatric Cardiology, Jawaharlal Nehru Medical College, KLES Academy of Higher Education & Research, Belgaum, 590010, Karnataka, India
- Department of Cardiac Anaesthesia, Jawaharlal Nehru Medical College, KLES Academy of Higher Education & Research, Belgaum, 590010, Karnataka, India
OPEN ACCESS
PUBLISHED: 28 February 2025
CITATION: Salve, G.G., et al., 2025. Newer insights into acute kidney injury following congenital cardiac surgery using staging criteria & biomarker analysis. Medical Research Archives, [online] 13(2).
https://doi.org/10.18103/mra.v13i3.6321
COPYRIGHT: © 2025 European Society of Medicine.This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i3.6321
ISSN 2375-1924
ABSTRACT
Objective: To assess the efficacy of staging criteria (Renal Angina Index-RAI & Acute Kidney Injury Network-AKIN), and biomarker Cystatin-C in predicting acute kidney injury (AKI) following congenital cardiac surgery.
Methods: Study included 52 consecutive patients undergoing congenital heart surgery at our hospital between March-May 2023. AKIN criteria, RAI score and serum Cystatin-C level were assessed within 24–48 hours of surgery. Chi-square/ Fisher’s exact test, and Student’s t’ test analysed variable association. Receiver operating characteristic (ROC) curve determined the predictive value for RAI and Cystatin-C.
Results: The mean age was 2.42 ± 2.6 years, 24 (46%) were male. RAI was positive (score ≥ 8) in 25 (48.08%) patients postoperatively. 92% with AKI by RAI had Stage 1 AKIN and 8% had Stage 2 & 3 AKIN. AKI by RAI and AKIN staging correlated with each other (p = 0.227). Preoperative creatinine level predicted AKI by RAI (p = 0.024).
Serum Cystatin-C levels were significantly associated with AKI (n=4) (p = 0.001). The Area under the curve for RAI for predicting AKI was 0.81 (0.57–1) (p = 0.046) and the cut-off value for predicting AKI was > 9, with a diagnostic accuracy of 87.8%. The Area under the curve for Cystatin-C for predicting AKI was 0.93 (0.85–1) (p = 0.004) and the cut-off value for predicting AKI was > 1.3 mg/dl, with a diagnostic accuracy of 95.1%.
Conclusions: Preoperative creatinine and postoperative Cystatin-C predict postoperative AKI. RAI and AKIN criteria are comparable in predicting AKI. Cystatin-C has a higher diagnostic accuracy for AKI than RAI.
Keywords: Acute kidney injury; Congenital cardiac surgery; Renal Angina Index; Acute Kidney Injury Network; Cystatin C
Glossary of Abbreviations:
AKI – Acute Kidney Injury
AKIN – Acute Kidney Injury Network
CPB – Cardiopulmonary Bypass
GFR – Glomerular Filtration Rate
KDIGO – Kidney Disease- Improving Global Outcomes
ICU – Intensive Care Unit
RACHS – Risk-Adjusted Classification for Congenital Heart Surgery
RAI – Renal Angina Index
ROC – Receiver Operating Characteristic
Introduction
Congenital heart disease is the most common congenital anomaly, occurring with an incidence of 1% of all live births, and is the most common cause of newborn death among birth abnormalities.¹ Acute kidney injury (AKI) is a common complication of congenital cardiac surgery that affects 20–65% of high-risk kids.²⁻⁵ Even in individuals with very slight changes in creatinine, AKI is linked to longer periods of mechanical ventilation, inotropic support, and ICU admission, as well as higher mortality. The fluid overload caused by AKI is, additionally, related to worse clinical outcomes and even death.
Understanding the epidemiology of acute kidney injury with the modifiable and non-modifiable risk factors, recognizing the illness earlier, and assessing the incidence and severity of acute kidney injury would prevent short-term and long-term adverse outcomes in children undergoing cardiac surgery for congenital heart diseases.⁶⁻⁹
Renal angina index (RAI), which is derived from variations in renal function, is used to stratify critically ill children who are at high risk of AKI. It is believed that the RAI may be used as a biomarker to identify early indicators of persistent AKI.¹⁰ The RAI is an idea that seems appealing, but it hasn’t been proven to work or validated in non-Western populations like Asian nations.¹¹,¹²
The National Institute of Health and the American Society of Nephrology called for the development of more efficient diagnostic tools for acute kidney injury. Three procedures could be used to identify high-risk individuals to prevent cardiac surgery-associated acute kidney injury: biomarker analysis, clinical risk factor assessment, and identification of preoperative, intraoperative, and postoperative risk factors. Additionally, following the treatment guidelines provided by Kidney Disease: Improving Global Outcomes (KDIGO) may lower the risk of AKI.¹³⁻¹⁵
In this study, we aimed to compare Acute Kidney Injury Network (AKIN) criteria¹⁶ and Renal Angina Index (RAI)¹⁰ for early prediction of severity of acute kidney injury in congenital heart disease patients undergoing cardiac surgery. We also aimed to ascertain the role of serum Cystatin C level and various risk factors in predicting acute kidney injury among these children.
Patients & Methods
ETHICS STATEMENT
The JNMC Institutional Ethics Committee of KLES Academy of Higher Education and Research approved this study on 27.09.2022 and individual patient consent was obtained (Ref. No. MDC/JNMCIEC/100).
PATIENTS
This is a cross-sectional study that included 52 consecutive patients, aged 1 month to 12 years, with congenital heart defects presented to our hospital and operated on for congenital heart surgery, between March 2023 and May 2023. All children with congenital heart defects planned for surgical repair were included except for those with chronic renal failure and acquired heart disease. Also, patients >12 years of age were excluded from the study.
DEFINITION OF AKI
The Kidney Disease: Improving Global Outcomes (KDIGO) criteria was used to diagnose and stage cardiac surgery-associated acute kidney injury.¹⁷
• A rise in serum creatinine level by ≥ 0.3 mg/dl (≥26.5 μmol/l) within 48 hours of surgery.
• A rise in serum creatinine level to ≥1.5 times the baseline levels, which is known or presumed to have occurred within the prior 7 days.
• Urine volume <0.5 ml/kg/hr for 6 hr.
METHODS
Informed and written consent was obtained from each of the children’s parent/guardian. Subjects were recruited according to the inclusion criteria. A detailed history was followed by a physical examination of the patient. Infants and children who have been diagnosed with acute kidney injury following congenital heart surgery were assessed with AKIN criteria and Renal Angina Index (RAI) within 24 hours to 48 hours of surgery.
RAI¹⁰ is calculated by assessing risk strata and injury strata. The risk strata include admission to intensive care unit (Score 1), solid organ or stem cell transplantation (score 3), and mechanical ventilation and/or vasoactive support (score 5). The injury strata for RAI include an increase in serum creatinine level above the baseline and the percentage of fluid overload. Decrease or no change in serum creatinine with < 5% fluid overload has score 1; >1x – 1.49x times increase in creatinine with 5–10% fluid overload has score 2; 1.5x – 1.99x times increase in creatinine with 10–15% fluid overload has score 4; and ≥ 2x times increase in creatinine with >15% fluid overload has score 8.
AKIN¹⁶ includes serum creatinine criteria and urine output criteria. Stage 1 involves increase in serum creatinine of ≥ 0.3 mg/dl or an increase to ≥ 150 to 200% from baseline, and urine output < 0.5 ml/kg/hr for 6 hours. Stage 2 involves an increase in serum creatinine to >200 to 300% from baseline, and urine output < 0.5 ml/kg/hr for 12 hours. Stage 3 involves an increase in serum creatinine to > 300% from baseline or serum creatinine of ≥ 4.0 mg/dl with an acute increase of at least 0.5 mg/dl, and urine output < 0.3 ml/kg/hr for 24 hours or anuria for 12 hours.
In secondary analysis, serum Cystatin C levels were measured in these patients within 24 hours of cardiac surgery to assess its predictive value and correlate it with that of RAI.
STATISTICAL ANALYSIS
Data was entered in a Microsoft Excel sheet and analyzed using Statistical Package of Social Science (SPSS) software version 21. Numerical variables were represented in the mean and standard deviation. Categorical variables were represented in frequencies and percentages. Renal Angina Index (RAI) was categorized with a cut-off of 8 signifying more than 8 suggesting of acute kidney injury. The association between the RA index scoring and AKIN staging was analyzed using the Chi-square test. Fisher’s exact test was used when more than 20% of the cell values had an expected value of less than 5.
When a categorical variable was associated with RA index and AKIN staging, the chi-square test was used to test the significance. Fisher’s exact test was used when more than 20% of the cell values had an expected value of less than 5. When a continuous variable was associated with RA index and AKIN staging, the variable was represented by mean (± standard deviation) and the significance of the difference between the means was tested by the Student’s t test.
The validity of the RA index and Cystatin C in predicting AKI was represented as sensitivity, specificity, and positive and negative predictive values. The cut-off value was determined using the receiver operating characteristic (ROC) curve. p-value less than 0.05 was considered statistically significant.
Results
The minimum required sample size was determined by the following formula–
n = Z² P (100 − P) / d²
n is the sample size required
Z is the required value corresponding to the level of confidence
P is the percentage occurrence of the condition (prevalence)
d is the percentage maximum error
Considering that about 15% of children will have acute kidney injury in congenital heart disease children post cardiac surgery at our department (considering the frequency of complex congenital heart surgery), at a 95% confidence level and 10% maximum error, the sample size was given by,
n = 1.96² × 15(100 − 15) / 10²
= 48.98
DEMOGRAPHIC CHARACTERISTICS
There were 52 patients included in this study, with a mean age of 2.42 ± 2.6 years. 24 (46%) were male children and 28 (54%) were female children. Common congenital heart defects present in these patients were ventricular septal defect 20 (38.46%), atrial septal defect 15 (28.85%), patent ductus arteriosus 6 (11.54%), tetralogy of Fallot 4 (7.69%), and total anomalous pulmonary venous connection 3 (5.77%). Figure 1 describes the spectrum of diagnoses in this study group.
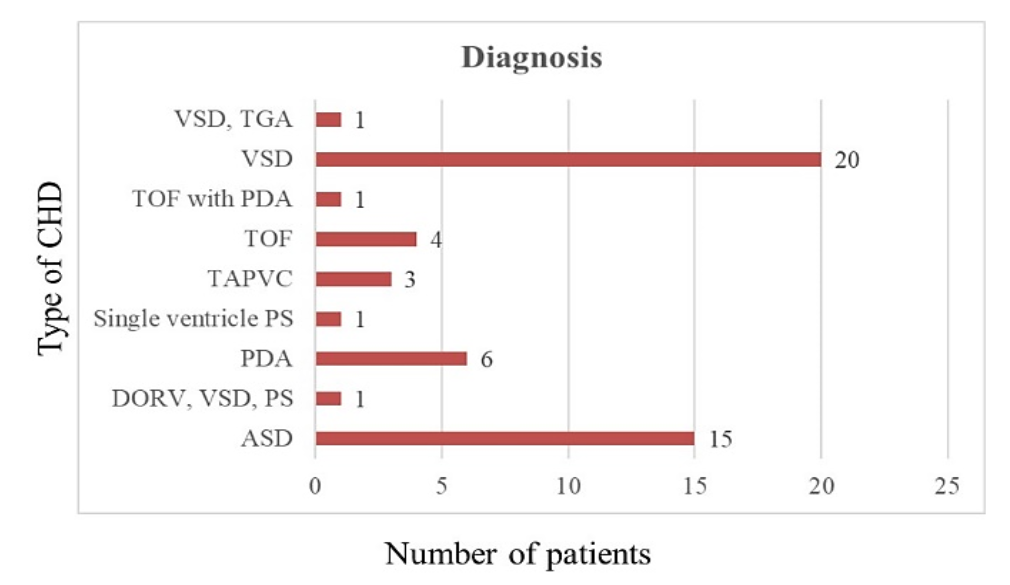 Figure 1 – A bar diagram showing the spectrum of congenital heart diseases with their frequency of occurrence.
Figure 1 – A bar diagram showing the spectrum of congenital heart diseases with their frequency of occurrence.
ASD Atrial septal defect; DORV Double outlet right ventricle; VSD Ventricular septal defect; PDA Patent ductus arteriosus; PS Pulmonary stenosis; TAPVC Total anomalous pulmonary venous connection; TGA Transposition of the great arteries; TOF Tetralogy of Fallot.
The mean pre-operative urea was 22.05 (± 8.22) mg/dl, ranging from 10 to 52 mg/dl, the mean pre-operative creatinine was 0.33 (± 0.11) mg/dl, ranging from 0.14 to 0.68 mg/dl and the mean pre-operative estimated glomerular filtration rate (eGFR) was 98.62 (± 30.56) ml/min ranging from 38 to 188.8 ml/min. Among the subjects, 18 (34.62%) received oral diuretics preoperatively as a part of treatment for congestive heart failure.
Among the patients who were subjected to cardiopulmonary bypass for surgical repair (45; 86.5%), the mean aortic cross-clamp time was 52.19 (± 28.45) min, ranging from 18 to 165 min, and the mean bypass time was 80.15 (± 32.84) min, ranging from 36 to 207 min.
The mean postoperative urea was 23.92 (± 6.6) mg/dl, ranging from 16 to 41 mg/dl, the mean postoperative creatinine was 0.38 (± 0.19) mg/dl, ranging from 0 to 1.5 mg/dl, and the mean postoperative eGFR was 87.12 (± 22.11) ml/min ranging from 37.5 to 132.1 ml/min. The mean values of postoperative urine output at 24 hours, 48 hours and 72 hours have been depicted in Figure 2. Among the subjects, 18 (34.62%) had 1 day, 20 (38.46%) had 2 days, 9(17.31%) had 3 days, 3 (5.77%) had 4 days and 2 (3.85%) had 5 days of mechanical ventilation postoperatively. All patients received 48 hours of antibiotics, and variable duration of inotropes, diuretics, and non-steroidal anti-inflammatory drugs postoperatively.
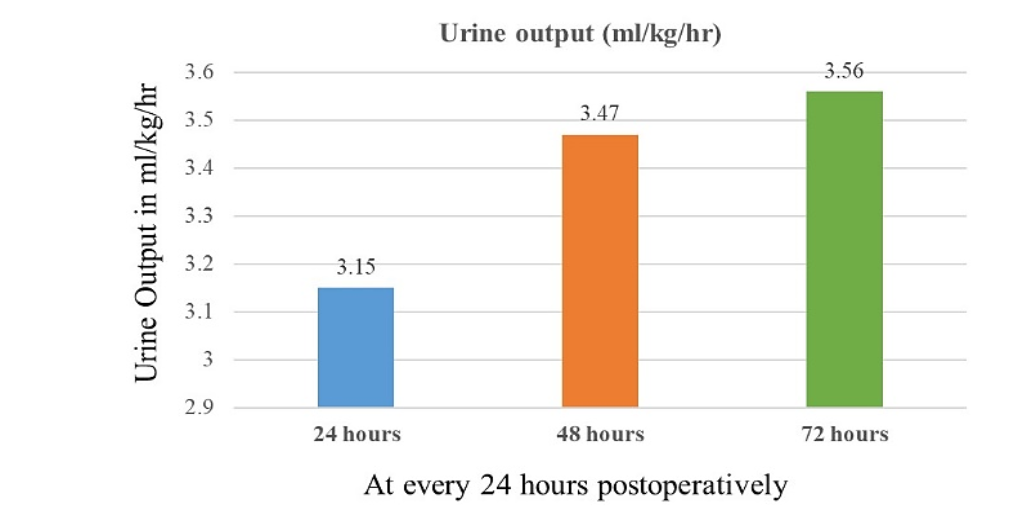
Figure 2 – A bar diagram showing the mean urine output in ml/kg/hour postoperatively at 24-hour intervals.
RENAL ANGINA INDEX & AKIN STAGING
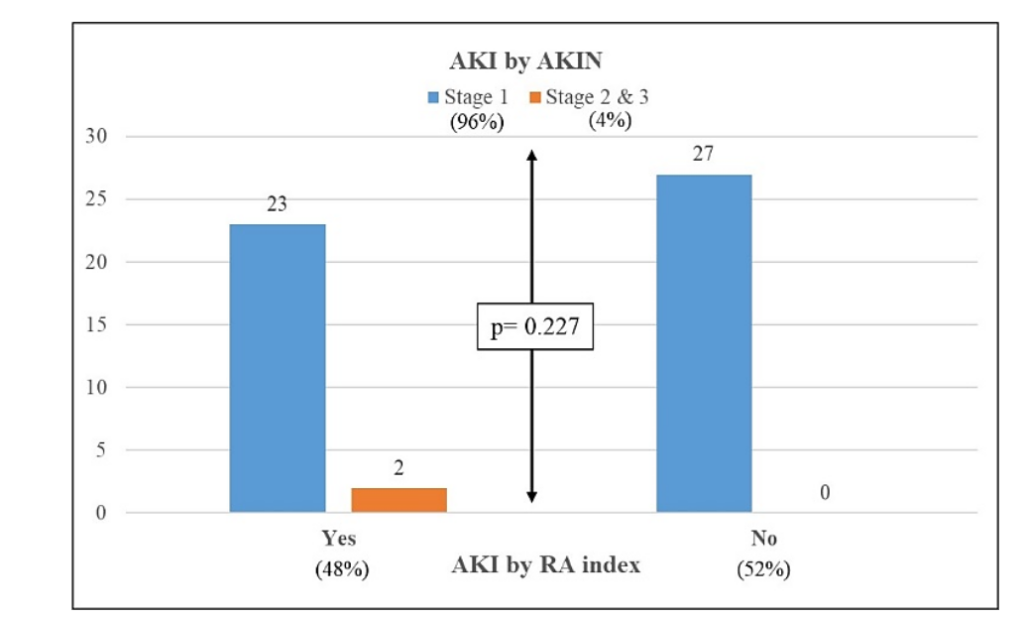
Renal Angina Index (RAI)¹⁰ was positive (score ≥ 8) in 25 (48.08%) patients postoperatively. Among the subjects, 27 (51.92%) had RA index score of 6, 22 (42.31%) had score of 12 and 3 (5.77%) had score of 24 out of 40. After considering AKIN staging,¹⁶ 50 (96.15%) patients had Stage 1 injury, 1 (1.92%) had Stage 2 injury and 1 (1.92%) had Stage 3 injury.
92% of the patients with AKI by RA index had Stage 1 AKIN and 8% had Stage 2 & 3 AKIN. All the subjects with an RA index less than 8 had Stage 1 AKIN. The association between AKI by RA index and AKIN staging was not statistically significant (p = 0.227) which suggests that AKI by RA index and AKIN staging had no difference and correlated with each other. (Table 1).
SIGNIFICANT PREDICTORS FOR AKI
Considering AKIN Staging, postoperative urea and creatinine levels were significantly high among stage 2 & 3 subjects compared to stage 1 subjects. eGFR had no statistically significant difference between AKIN stages. Urine output was significantly higher in stage 1 subjects at 24 hours, 48 hours and 72 hours compared to stage 2 & 3 subjects. (Table 2)Table 1 – Association of RA Index & AKIN Staging
| AKI by RA index | AKIN Staging Stage 1 | Stage 2 & 3 | Total | Fisher exact p-value |
|---|---|---|---|---|
| Yes | 23 (92%) | 2 (8%) | 25 (100%) | |
| No | 27 (100%) | 0 (0%) | 27 (100%) | 0.227 |
| Total | 50 (96.15%) | 2 (3.84%) | 52 (100%) |
AKI Acute Kidney Injury; AKIN Acute Kidney Injury Network; RA Renal Angina
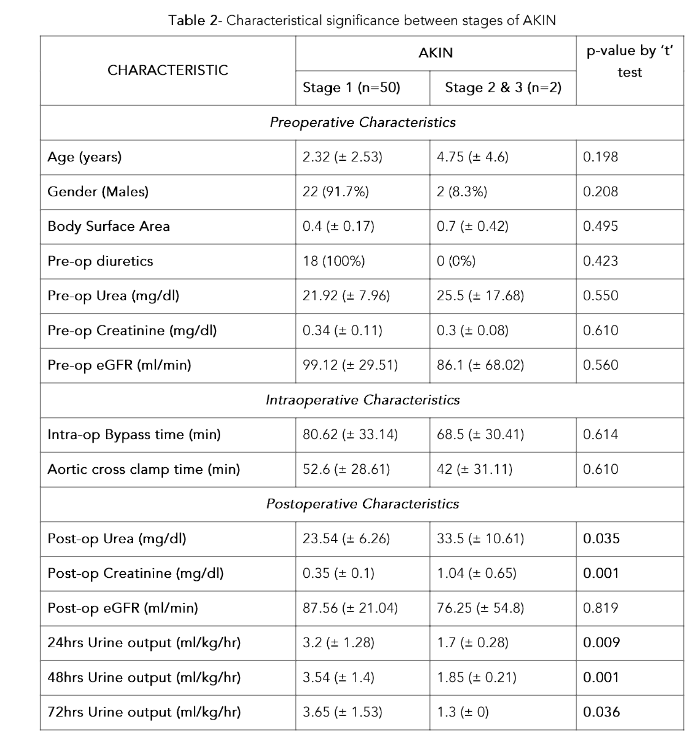
AKIN Acute Kidney Injury Network; eGFR estimated Glomerular Filtration Rate; Intra-op Intraoperative; Post-op Postoperative; Pre-op Preoperative
Preoperative creatinine level was significantly high among subjects with AKI by RAI scoring (Table 3). Also, preoperative use of diuretics did not predict occurrence of AKI (Fisher exact p value 0.31). None of the other characteristics showed significant differences among the positive and negative RA index groups.
Table 3 – Characteristical significance between positive and negative RA Index groups
| CHARACTERISTIC | RA index ≥ 8 (n=25) | RA index < 8 (n=27) | p value by ‘t’ test (“ – Chi sq. test) |
|---|
Preoperative Characteristics
Age (years) – 2.82 (± 3) | 1.65 (± 1.37) | 0.061
Gender (Males) – 14 (58.3%) | 10 (41.7%) | 0.143ʷ
Body Surface Area – 0.42 (± 0.21) | 0.39 (± 0.13) | 0.569
Pre-op diuretics – 14 (77.8%) | 4 (22.2%) | 0.100ʷ
Pre-op Urea (mg/dl) – 22.64 (± 8.73) | 20.94 (± 7.25) | 0.484
Pre-op Creatinine (mg/dl) – 0.31 (± 0.08) | 0.38 (± 0.14) | 0.024
Pre-op eGFR (ml/min) – 102.85 (± 31.59) | 90.64 (± 27.61) | 0.173
Intraoperative Characteristics
Intra-op Bypass time (min) – 70.97 (± 22.2) | 97.5 (± 42.33) | 0.051
Aortic cross clamp time (min) – 44.56 (± 20.26) | 66.61 (± 35.98) | 0.057
Postoperative Characteristics
Post-op Urea (mg/dl) – 24.4 (± 6.64) | 23.02 (± 6.63) | 0.479
Post-op Creatinine (mg/dl) – 0.39 (± 0.22) | 0.37 (± 0.09) | 0.794
Post-op eGFR (ml/min) – 88.56 (± 23.4) | 84.41 (± 19.79) | 0.525
24hrs Urine output (ml/kg/hr) – 3.12 (± 1.3) | 3.2 (± 1.3) | 0.829
48hrs Urine output (ml/kg/hr) – 3.56 (± 1.39) | 3.31 (± 1.47) | 0.549
72hrs Urine output (ml/kg/hr) – 3.47 (± 1.44) | 3.73 (± 1.81) | 0.582
eGFR estimated Glomerular Filtration Rate; Intra-op Intraoperative; Post-op Postoperative; Pre-op Preoperative; RA Renal Angina
CORRELATION OF CYSTATIN C LEVELS AND RA INDEX IN AKI
Serum Cystatin C levels (n = 41) had a mean value of 0.73 ± 0.32 mg/dl within 24 hours postoperatively. Cystatin C was higher in 4 patients with AKI who had the highest RAI score, implying a significant association between Cystatin C and AKI (p = 0.001).
The Area under the curve for RA index (Figure 3) for predicting AKI was 0.81 (0.57–1), which was significant (p = 0.046). The cut-off of RA index for predicting AKI was > 9 in our study, which had a sensitivity of 50%, specificity of 91.8%, positive predictive value of 40% and negative predictive value of 94.4%, with a diagnostic accuracy of 87.8%.
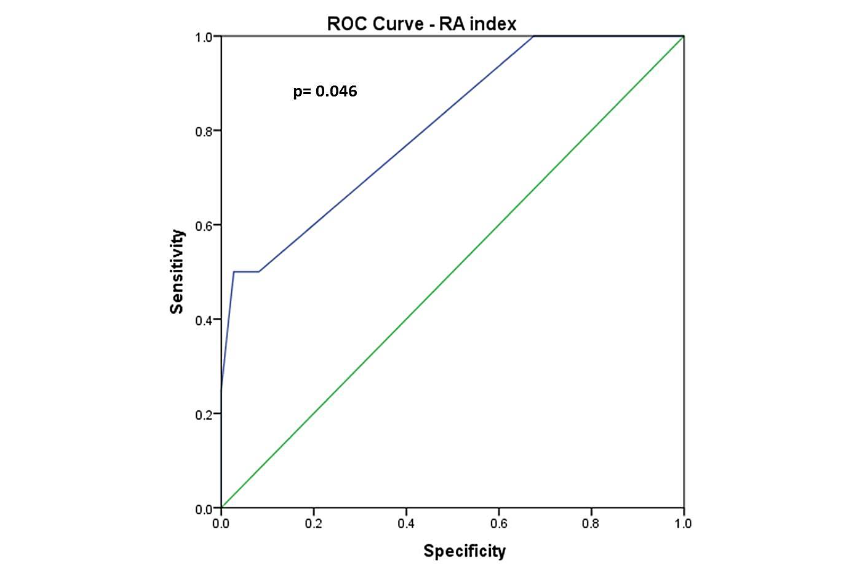
Figure 3 ROC Curve – RA index
Figure 3 – The area under the Receiver Operating Characteristic (ROC) curve for the RA index for predicting AKI was 0.81 (0.57–1), which was significant (p = 0.046). The cut-off of the RA index for predicting AKI was > 9 in our study, which had a sensitivity of 50%, specificity of 91.8%, positive predictive value of 40%, and negative predictive value of 94.4%, with a diagnostic accuracy of 87.8%.
The Area under the curve for Cystatin C (Figure 4) for predicting AKI was 0.93 (0.85–1), which was significant (p = 0.004). The cut-off for Cystatin C for predicting AKI in our study was > 1.3 mg/dl, which had a sensitivity of 50%, specificity of 100%, positive predictive value of 100% and negative predictive value of 94.8%, with a diagnostic accuracy of 95.1%.
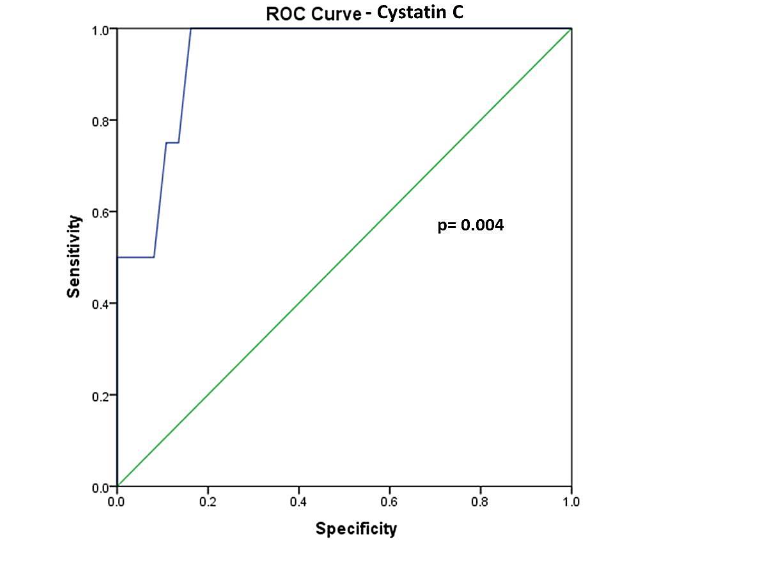
Figure 4: ROC Curve – Cystatin C
Figure 4 – The area under the Receiver Operating Characteristic (ROC) curve for Cystatin C for predicting AKI was 0.93 (0.85–1), which was significant (p = 0.004). The cut-off for Cystatin C for predicting AKI in our study was > 1.3 mg/dl, which had a sensitivity of 50%, specificity of 100%, positive predictive value of 100%, and negative predictive value of 94.8%, with a diagnostic accuracy of 95.1%.
Discussion
Acute Kidney Injury (AKI) is a common complication in children undergoing congenital heart surgery and is associated with prolonged ventilatory support, prolonged intensive care unit stay, higher morbidity and higher mortality.¹,²¹ The KDIGO group has laid down standard definition of AKI and its staging according to the severity.¹⁷
The primary mechanisms through which AKI develops when associated with cardiac surgery include renal ischemia, reperfusion injury, inflammation, and cardiopulmonary bypass induced haemolysis. The resulting inflammatory state, mediated by increased cytokines and chemokines, further damages renal tissue by recruiting neutrophils, lymphocytes, and macrophages into the renal parenchyma. This process can result in permanent renal damage due to the consequent healing and fibrosis.²²
The gold standards for diagnosing AKI are serum creatinine level and hourly urine output monitoring. Urine output following congenital cardiac surgery is significantly manipulated by the use of intravenous diuretics, with an intention to avoid decreasing output, which is further thought to lead to renal dysfunction. With this understanding, we aimed to determine the correlation of staging criteria dependent on the urine output (AKIN),¹⁶ and the one independent of urine output (RAI),¹⁰ in predicting postoperative AKI in these patients. We found that prediction of AKI by RAI and AKIN had no significant difference and they correlated with each other.
By the definition of RAI,¹⁰ the risk strata component of RAI includes the ICU admission and mechanical ventilation/ vasoactive support factors. These will be always positive for patients who have undergone congenital heart surgery. This means that the minimum RAI score in this subgroup of paediatric population undergoing congenital heart surgery will be 6.¹⁰ This was noticed in all 27 patients (51.92%) without AKI in our study, as all had an RAI score of 6. The injury strata of RAI scoring will then determine whether their scores exceed the cut-off levels. The major determinant of injury strata in RAI scoring is serum creatinine level, which is consistently measured for predicting and prognosticating postoperative renal injury in these patients. The cut off score of the RA index for predicting AKI was > 9 in our study.
Few researchers argue that serum creatinine and urine output are late and insensitive predictors of AKI and they correlate poorly with the onset and progression of AKI.²³,²⁴ This is supported by the fact that congenital heart surgery, especially after employing cardiopulmonary bypass, induces a cascade of inflammatory processes which can affect these gold standard markers. In addition, serum creatinine level is influenced by several non-renal variants like age, gender, muscle mass, and growth.²⁵ Studies on newer biomarker analysis were aimed to overcome these shortcomings to predict the development of AKI early, and more accurately.²⁶,²⁷
Cystatin C is a 13 Kilo-Dalton proteinase inhibitor, belonging to the cystatin superfamily of cysteine protease inhibitors. Cystatin C is a key player in the intracellular catabolism of proteins and peptides.²⁸ Serum Cystatin C is an easily measurable biomarker of renal function as it is less influenced by the above-mentioned non-renal variants and is exclusively eliminated by glomerular filtration.²⁹ Thus, in this study, we also aimed to determine the correlation between RAI scoring and serum Cystatin C levels in patients who have undergone congenital heart surgery, to compare the gold standard criteria (RAI) and the commonly used biomarker (Cystatin C). Our analysis revealed that the cut off value of Cystatin C for predicting AKI was > 1.3 mg/dl, with a better diagnostic accuracy (95.1%) as compared to that of RAI score (87.8%).
In a multicenter prospective study conducted by Simon Li et al., 42% of children aged 1 month to 18 years experienced acute renal injury within 3 days following their cardiac surgery. Prolonged mechanical ventilation and an extended hospital stay were independently linked to the development of AKI. Age and cardiopulmonary bypass duration were found to be the major determinants for development of AKI.³⁰
In a retrospective chart review by Bilal Aoun et al., among 150 children and infants who underwent cardiac surgery, 14 patients (9.3%) developed AKI using the KDIGO criteria. They observed that AKI was more likely to occur in children with anemia, lactic acidosis, and presence of cyanotic heart disease. ³¹
Sidharth Kumar Sethi, et al., studied the incidence, risk factors, severity and outcomes of acute kidney injury among children undergoing cardiac surgery. They observed that 20 patients – (9.6%) (14 of them were infants) had AKI. Age <1 year, cardiopulmonary bypass time, prolonged ventilator requirement, pump failure, sepsis and hematological complications were identified as independent risk factors for any degree of acute kidney injury.³²
In a single Centre observational study conducted by Georgios Kornelis et al., they analyzed the incidence, risk factors, severity and outcomes of acute kidney injury among children undergoing cardiac surgery. They observed that 19.3% of the patients developed AKI, amongst which the in-hospital mortality rate was 21.4%. Postoperative AKI was linked to younger age, lower weight, longer cardiopulmonary bypass time, preoperative mechanical ventilation, and cardiac diagnosis.³³
Scott J. Aydin et al., conducted a review among 458 children aged less than 18 years. They studied the incidence, risk factors, severity and outcomes of acute kidney injury among children undergoing cardiac surgery. They observed that the 234 individuals (51%) had AKI. The development of AKI was linked to younger age, greater RACHS-1 (risk-adjusted classification for congenital heart surgery) category, longer cardiopulmonary bypass time, higher preoperative blood urea nitrogen and lower preoperative serum creatinine levels.³⁴
In a recent systematic review performed by Cavalcante CTMB et al., they analyzed the capacity to predict AKI and poor outcomes of five biomarkers: Cystatin C, Neutrophil gelatinase-associated lipocalin, Interleukin-18, Kidney injury molecule-1, and Liver fatty acid-binding protein. They concluded with a need for further meta-analyses with the availability of additional studies.³⁵
In our study, 48% of the patients developed variable degree of AKI following congenital heart surgery. Preoperative creatinine level was the only significant predictor of occurrence of AKI after surgery (Table 3). Early prediction of AKI is essential and may improve outcomes in kids undergoing congenital heart surgery. This is a vulnerable population that has a multifactorial impact on kidney function following surgery due to variations in their age, cardiac diagnosis, cardiac physiology, and nature of cardiac repair. This makes it more complex to predict the renal outcomes. We compared the urine output dependent (AKIN) and independent (RAI) staging criteria, which revealed no significant difference, and they correlated with each other. We also analyzed the correlation of creatinine-dependent staging criteria (RAI) against the biomarker Cystatin C. They both predicted AKI with Cystatin C having a higher diagnostic accuracy (Figure 5).
Renal angina index is simple to calculate by assessing the risk strata and injury strata components.¹⁰ In our study, RAI value cut-off of >9 had a diagnostic accuracy of 88%. RAI would be more specific if the cut-off values were increased. Further studies can focus on the incorporation of biomarkers and other risk factors into RAI, which would add to the validity of early prediction of the development of AKI and its prognosis. Further studies with increased sample size of patients undergoing congenital heart surgery, including patients in other settings such as primary and secondary intensive care, will represent the true usefulness of RAI for early prediction of the development of AKI and its prognosis.
Limitations
Our study has some limitations. First, this is a single institutional study with a smaller cohort which limits generalizability. Preoperative levels of Cystatin C were not measured, limiting comparison to postoperative levels. Six patients with PDA were operated on without cardiopulmonary bypass (CPB), so CPB was not a uniform intraoperative factor, even though CPB time was not a significant predictor of AKI in our study.
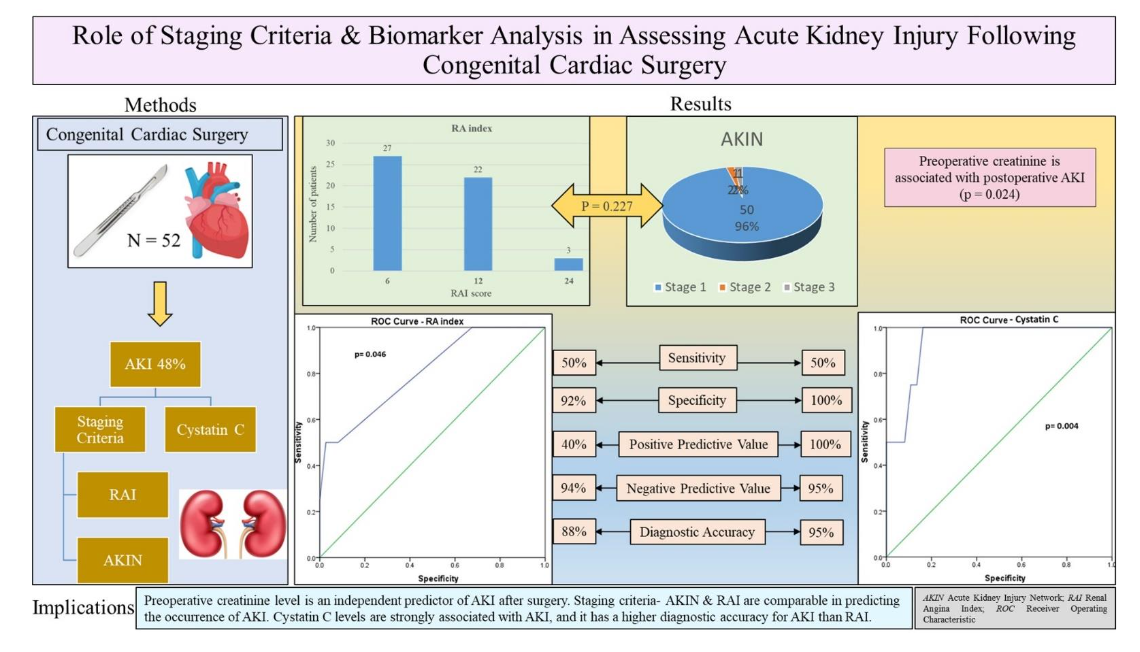
Figure 5 Role of Staging Criteria & Biomarker Analysis in Assessing Acute Kidney Injury Following Congenital Cardiac Surgery
Figure 5 – Graphical representation of the study design and outcomes involving staging criteria & biomarker analysis in assessing acute kidney injury (AKI) following congenital cardiac surgery.
AKIN Acute Kidney Injury Network; RAI Renal Angina Index; ROC Receiver Operating Characteristic.
Conclusions
Acute kidney injury is very common and is a major adverse outcome following congenital heart surgery. Preoperative creatinine level is an independent predictor of AKI after surgery. Staging criteria – AKIN & RAI are comparable in predicting the occurrence of AKI signifying that urine output is important, but not solely reliable. RAI scoring and Cystatin C levels are strongly associated with AKI, while Cystatin C has a higher diagnostic accuracy for AKI than RAI.
Correlation of Renal Angina Index (RAI) and Acute Kidney Injury Network (AKIN) criteria in predicting AKI following congenital heart surgery
Central Message:
Acute Kidney Injury (AKI) is common after congenital heart surgery, having an identical correlation with Renal Angina Index (RAI) score and Acute Kidney Injury Network (AKIN) criteria. Preoperative creatinine and postoperative Cystatin C levels predict its occurrence.Date and number of IRB approval:
The JNMC Institutional Ethics Committee of KLES Academy of Higher Education and Research approved this study on 27.09.2022 and individual patient consent was obtained (Ref. No. MDC/JNMCIEC/100).
Perspective Statement:
Staging criteria dependent on urine output (AKIN), and the one independent of urine output (RAI) have a similar prediction of postoperative AKI following congenital heart surgery. Preoperative serum creatinine level independently predicts the incidence of postoperative AKI. Both RAI score and serum Cystatin C levels are strongly associated with AKI, while Cystatin C has a higher diagnostic accuracy.
Conflicts of Interest:
None
Disclosures/ Funding:
None
References
1. Reller MD, Strickland MJ, Riehle-Colarusso T, Mahle WT, Correa A. Prevalence of congenital heart defects in metropolitan Atlanta, 1998-2005. J Pediatr. 2008;153:807-13. doi: 10.1016/j.jpeds.20 08.05.059.
2. Gil-Ruiz Gil-Esparza MA, Alcaraz Romero AJ, Romero Otero A, Gil Villanueva N, Sanavia Moran E, Rodriguez Sanchez de la Blanca A, et al. Prognostic relevance of early AKI according to pRIFLE criteria in children undergoing cardiac surgery. Pediatr Nephrol. 2014;29:1265–72.
doi: 10.1007/s00467-014-2757-z.
3. Sugimoto K, Toda Y, Iwasaki T, Shimizu K, Kanazawa T, Muto N, et al. Urinary albumin levels predict development of acute kidney injury after pediatric cardiac surgery: a prospective observational study. J Cardiothorac Vasc Anesth. 2016;30:64–8. doi: 10.1053/j.jvca.2015.05.194.
4. Ruf B, Bonelli V, Balling G, Horer J, Nagdyman N, Braun SL, et al. Intraoperative renal near-infrared spectroscopy indicates developing acute kidney injury in infants undergoing cardiac surgery with cardiopulmonary bypass: a case-control study. Crit Care. 2015;19:27. doi: 10.1186/s13054-015-0760-9.
5. Meersch M, Schmidt C, Van Aken H, Rossaint J, Görlich D, Stege D, et al. Validation of cell-cycle arrest biomarkers for acute kidney injury after pediatric cardiac surgery. PLoS One. 2014;9:e110 865. doi: 10.1371/journal.pone.0110865.
6. Sutherland SM, Alobaidi R, Gorga SM, Iyengar A, Morgan C, Heydari E, et al; ADQI 26 Workgroup. Epidemiology of acute kidney injury in children: a report from the 26th Acute Disease Quality Initiative (ADQI) consensus conference. Pediatr Nephrol. 2024;39:919-928. doi: 10.1007/s0 0467-023-06164-w.
7. Kellum JA, Romagnani P, Ashuntantang G, Ronco C, Zarbock A, Anders HJ. Acute kidney injury. Nat Rev Dis Primers. 2021;7:52. doi: 10.103 8/s41572-021-00284-z.
8. Gameiro J, Fonseca JA, Outerelo C, Lopes JA. Acute Kidney Injury: From Diagnosis to Prevention and Treatment Strategies. J Clin Med. 2020;9:1704. doi: 10.3390/jcm9061704.
9. Hoste EAJ, Kellum JA, Katz NM, Rosner MH, Haase M, Ronco C. Epidemiology of acute kidney injury. Contrib Nephrol. 2010;165:1-8. doi: 10.1159/000313737.
10. Goldstein SL, Chawla LS. Renal angina. Clin J Am Soc Nephrol. 2010;5:943-9. doi: 10.2215/CJ N.07201009.
11. Cruz DN, Ferrer-Nadal A, Piccinni P, Goldstein SL, Chawla LS, Alessandri E, et al; NEFROINT Investigators. Utilization of small changes in serum creatinine with clinical risk factors to assess the risk of AKI in critically ill adults. Clin J Am Soc Nephrol. 2014;9:663-72. doi: 10.2215/CJ N.05190513.
12. Chawla LS, Goldstein SL, Kellum JA, Ronco C. Renal angina: concept and development of pretest probability assessment in acute kidney injury. Crit Care. 2015;19:93. doi: 10.1186/s13054-015-0779-y.
13. Meersch M, Schmidt C, Hoffmeier A, Van Aken H, Wempe C, Gerss J, et al. Prevention of cardiac surgery-associated AKI by implementing the KDIGO guidelines in high risk patients identified by biomarkers: the PrevAKI randomized controlled trial. Intensive Care Med. 2017;43:1551-1561. doi: 10.1007/s00134-016-4670-3.
14. Ostermann M, Kunst G, Baker E, Weerapolchai K, Lumlertgul N. Cardiac Surgery Associated AKI Prevention Strategies and Medical Treatment for CSA-AKI. J Clin Med. 2021;10:5285. doi: 10.3390/j cm10225285.
15. Ortega-Loubon C, Fernández-Molina M, Carrascal-Hinojal Y, Fulquet-Carreras E. Cardiac surgery-associated acute kidney injury. Ann Card Anaesth. 2016;19:687-698. doi: 10.4103/0971-978 4.191578.
16. Mehta RL, Kellum JA, Shah SV, Molitoris BA, Ronco C, Warnock DG, et al; Acute Kidney Injury Network. Acute Kidney Injury Network: report of an initiative to improve outcomes in acute kidney injury. Crit Care. 2007;11:R31. doi: 10.1186/cc5713.
17. The Kidney Diseases Improving Global Outcomes (KDIGO) Working Group. Summary of Recommendation Statements Kidney International Supplements (2012) 2, 8–12; doi:10.1038/kisup.20 12.7.
18. Pourhoseingholi MA, Vahedi M, Rahimzadeh M. Sample size calculation in medical studies. Gastroenterol Hepatol Bed Bench. 2013;6:14-7.
19. Taylor ML, Carmona F, Thiagarajan RR, Westgate L, Ferguson MA, del Nido PJ, et al. Mild postoperative acute kidney injury and outcomes after surgery for congenital heart disease. J Thorac Cardiovasc Surg. 2013;146:146–52. doi: 10.1016/j.j tcvs.2012.09.008.
20. Toth R, Breuer T, Cserep Z, Lex D, Fazekas L, Sapi E, et al. Acute kidney injury is associated with higher morbidity and resource utilization in pediatric patients undergoing heart surgery. Ann Thorac Surg. 2012;93:1984–90. doi: 10.1016/j.atho racsur.2011.10.046.
21. Toda Y, Sugimoto K. AKI after pediatric cardiac surgery for congenital heart diseases-recent developments in diagnostic criteria and early diagnosis by biomarkers. J Intensive Care. 2017; 5:49. doi: 10.1186/s40560-017-0242-z.
22. Sharma A, Chakraborty R, Sharma K, Sethi SK, Raina R. Development of acute kidney injury following pediatric cardiac surgery. Kidney Res Clin Pract. 2020;39:259-268. doi: 10.23876/j.krcp.20.053.
23. Koyner JL, Garg AX, Coca SG, Sint K, Thiessen-Philbrook H, Patel UD, et al; TRIBE-AKI Consortium. Biomarkers predict progression of acute kidney injury after cardiac surgery. J Am Soc Nephrol. 2012;23:905-14. doi: 10.1681/ASN.2011 090907.
24. Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P; Acute Dialysis Quality Initiative workgroup. Acute renal failure – definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8:R204-12. doi: 10.1186/cc2872.
25. Schwartz GJ, Work DF. Measurement and estimation of GFR in children and adolescents. Clin J Am Soc Nephrol. 2009;4:1832-43. doi: 10.221 5/CJN.01640309.
26. Koyner JL, Parikh CR. Clinical utility of biomarkers of AKI in cardiac surgery and critical illness. Clin J Am Soc Nephrol. 2013;8:1034-42. doi: 10.2215/CJN.05150512.
27. Vanmassenhove J, Vanholder R, Nagler E, Van Biesen W. Urinary and serum biomarkers for the diagnosis of acute kidney injury: an in-depth review of the literature. Nephrol Dial Transplant. 2013;28:254-73. doi: 10.1093/ndt/gfs380.
28. Zakaria M, Hassan T, Refaat A, Fathy M, Hashem MIA, Khalifa N, et al. Role of serum cystatin C in the prediction of acute kidney injury following pediatric cardiac surgeries: A single center experience. Medicine (Baltimore). 2022;101: e31938. doi: 10.1097/MD.0000000000031938.
29. Herget-Rosenthal S, Trabold S, Pietruck F, Holtmann M, Philipp T, Kribben A. Cystatin C: efficacy as screening test for reduced glomerular filtration rate. Am J Nephrol. 2000;20:97-102. doi: 10.1159/000013564.
30. Li S, Krawczeski CD, Zappitelli M, Devarajan P, Thiessen-Philbrook H, Coca SG, et al; TRIBE-AKI Consortium. Incidence, risk factors, and outcomes of acute kidney injury after pediatric cardiac surgery: a prospective multicenter study. Crit Care Med. 2011;39:1493-9. doi: 10.1097/CCM.0b013e31821 201d3.
31. Aoun B, Daher GA, Daou KN, Sanjad S, Tamim H, El Rassi I, et al. Acute Kidney Injury Post-cardiac Surgery in Infants and Children: A Single-Center Experience in a Developing Country. Front Pediatr. 2021;9:637463. doi: 10.3389/fped.2021.6 37463.
32. Sethi SK, Kumar M, Sharma R, Bazaz S, Kher V. Acute kidney injury in children after cardiopulmonary bypass: risk factors and outcome. Indian Pediatr. 2015;52:223-6. doi: 10.1007/s13312-015-0611-4.
33. Kourelis G, Kanakis M, Samanidis G, Tzannis K, Bobos D, Kousi T, et al. Acute Kidney Injury Predictors and Outcomes after Cardiac Surgery in Children with Congenital Heart Disease: An Observational Cohort Study. Diagnostics (Basel). 2022;12:2397. doi: 10.3390/diagnostics12102397.
34. Aydin SI, Seiden HS, Blaufox AD, Parnell VA, Choudhury T, Punnoose A, et al. Acute kidney injury after surgery for congenital heart disease. Ann Thorac Surg. 2012;94:1589-95. doi: 10.1016/j.athoracsur.2 012.06.050.
35. Cavalcante CTMB, Cavalcante MB, Castello Branco KMP, Chan T, Maia ICL, Pompeu RG, et al. Biomarkers of acute kidney injury in pediatric cardiac surgery. Pediatr Nephrol. 2022;37:61-78. doi: 10.1007/s00467-021-05094-9.
Most read articles by the same author(s)
- Sumiran Mahajan, D.M., Abhishek Prabhu, M.Ch., Gananjay G. Salve, M.Ch., Sweta G. Sooragonda, M.D., Jabbar Momin, M.D., Mohan D. Gan, M.Ch., Mallikarjun Karishetti, D.N.B., Risk factors and prognostic significance of acute kidney injury in coronary artery bypass graft surgery: Insights from the KDIGO criteria , Medical Research Archives: Vol 13 No 8 (2025): Vol.13, Issue 8, August 2025

