Advancing Education in Transfusion Medicine Quality
FILLING A GAP IN EDUCATION AND RESEARCH IN TRANSFUSION MEDICINE
Cees Th. Smit Sibinga1, MD, PhD, FRCP Edin, FRCPath
- IQM Consulting – International Development of Quality Management in Transfusion Medicine, University of Groningen, NL
OPEN ACCESS
PUBLISHED 31 August 2025
CITATION Smit Sibinga, CT., 2025. FILLING A GAP IN EDUCATION AND RESEARCH IN TRANSFUSION MEDICINE. Medical Research Archives, [online] 13(8). https://doi.org/10.18103/mra.v13i8.6848
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i8.6848
ISSN 2375-1924
ABSTRACT
Transfusion medicine has come a long way from the antique mystical époque till today. Although Landsteiner did breaking research at the end of the 19th Century, it lasted till 1975 when the World Health Assembly in Geneva endorsed a Resolution to develop a safe and effective blood supply based on four key elements: Safety, Purity, Potency and Clinical Efficacy. Since the outbreak of the AIDS pandemic in the early 1980s WHO started to map the world for the blood systems and presence of these four key elements. Simple recommendations (Aide Mémoires) and distance learning modules (DLM) were initiated and in 2000 a Quality Management Training (QMT) course was developed. However, what was forgotten was the development of governance, legal frameworks and structured education. An attempt to close that gap was undertaken in Groningen, the Netherlands with the creation of an Academic Institute for International Development of Transfusion Medicine and 4 years later an International Consultation for the development of quality management in transfusion medicine (IQM) on a cost-free basis.
Keywords: Transfusion Medicine, quality, safety, clinical efficacy, education, governance
Introduction
Transfusion Medicine (TM) is one of the youngest medical specialties. WHO defined this specialty in 1991 as follows:
Transfusion medicine deals with that part of the health care system which undertakes the appropriate provision and use of human blood resources. It provides the essential scientific and clinical information for transfusion practice, and the link between the blood donor’s altruistic donation and the patient.
The definition learns that TM is an integral part of the health care system and provides essential information for science and clinical transfusion practices. It stretches from the altruistic (motivated and non-remunerated) donor in the community to the patient in the bedside (healthcare institute). In other words source to the consumer (outcome).
Transfusion Medicine (TM) has come a long way in its development starting in the mid-seventeenth Century at the court of Louis XIV in Paris, where the young court physician and most able Cartesian philosopher Jean Baptiste Denis from Montpellier together with the surgeon Paul Emmerez did quite some dog-to-dog transfusion experiments. When he was presented a severely ill 15 years old boy with fever and weakness due to the many blood-lettings, he decided to transfuse the boy with lambs blood, which resulted in a miraculous recovery effect! Shortly after this éclat success a second 45 years old healthy male was successfully transfused, followed by the son of the Minister of Foreign Affairs of the king of Sweden who fell seriously ill while in Paris. Denis decided to treat him with two subsequent transfusions, and with good success. The report was published in the Philosophical Transactions of the Royal Society of July 1667.
The following patient transfused by Denis was a 34 years old man Antoine Mauroy, who suffered from a tragic love affair.

He received over a period of a couple of months several calf blood transfusions, but started after the second transfusion to react with fever, pain in the lumps, increased pulse rate, sweating, and dyspnea, excreting black urine. Denis has carefully documented the event, thereby uniquely describing for the first time in medical history classical acute hemolytic transfusion reaction.

He survived, but when a few months later his mental condition again deteriorated, Denis decided to treat Antoine Mauroy with another transfusion, which unfortunately caused his death due to acute lethal hemolysis. Denis was accused of murder but during the Châtelet trial in Paris plead not guilty. However, the conservative Paris University Sorbonne forbid further blood transfusion experiments. Also in England further experiments were forbidden, followed by the anathema of the Pope of Avignon. Almost a century later the French scientist Cantwell from Paris raises his voice for a plea to revive the experiments as he stated that blood transfusion could very well be life-saving in case of severe trauma and calamities. Unfortunately he was not well received and it lasted again more than half a century until in England the progressive gynecologist and obstetrician James Blundell from London, who did his medical education in Edinburgh, showed a deep interest in the potential of blood transfusion.

His interest was not only based on the personal experience with women in labor who postpartum bled to death, but also by the scientific experiments of John Leacock from Barbados. In 1816 John Henry Leacock reported systematic experiments in Edinburgh on dogs and cats that established that donor and recipient must be of the same species, and recommended inter-human transfusion. He then returned to Barbados and published nothing more. However, James Blundell, who extended Leacock’s experiments and publicized the results widely, is credited by many with introducing transfusion into clinical use, but he always gave credit to Leacock for his initial work. In fact they were the founders of modern immunology and the principle of compatibility presenting scientific evidence for species specificity. The scientific and clinical value of these observations became much later understood and practiced. Blundell decided based on his animal experiments to apply the lessons learned in human pathology. A 35 year old man with terminal stomach cancer was successfully transfused directly. Most of his work was published in The Lancet.
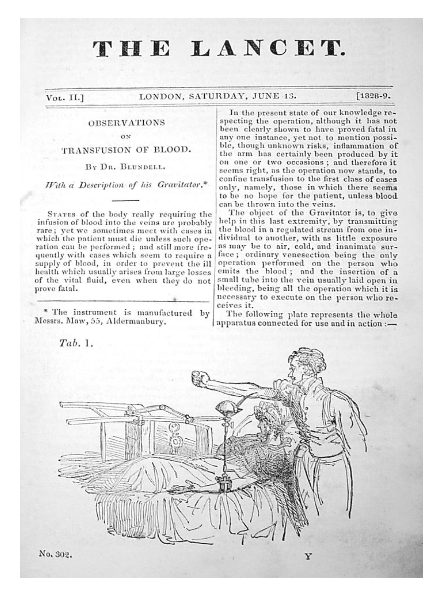
In an editorial of the 1825 Philadelphia Journal of Medicine, Physics and Science Blundell’s premiere has been debated in a footnote arguing that Dr. Philip Syng Physick did the same already in 1815. However, that practice was never published nor presented publicly. Blundell continued his work and managed to save the lives of dozens of women in labor and was frequently consulted about blood transfusion. He was indeed the first clinical specialist who deserved the classification of ‘Transfusion Medicine Specialist’.
These historical events illustrate the clinical scientific aspects of Transfusion Medicine. It lasted till the turn of the 19th Century, when Karl Landsteiner discovered the red cell antigens of the most common blood group system named ABO for which he was awarded the Nobel prize in 1930. The focus shifted from the bedside to the research laboratory. For long Transfusion Medicine was regarded the Cinderella in medicine and health sciences, unjustly regarded a laboratory/pathology science immunohematology, microbiology and virology instead of a clinical science albeit dominated by laboratory research. However, from there on different eras became recognizable:
- Blood group serology and immunohematology;
- Anticoagulation and preservation of blood, biocompatibility and separation/manufacturing of blood products/components;
- Transmissible diseases: bacterial (syphilis), viral (hepatitis), parasitic (malaria, Babeziosis), prions (Creutzfeld-Jacob);
- Community public awareness (Red Cross/Crescent), voluntary non-remunerated donors (VNRD, motivation);
- Quality management, blood safety and availability- immunohematology and transfusion transmissible infectious agents, transfusion technology;
- Patient Blood Management (PBM, rational use of blood/liberal vs restricted);
- Organization, regulatory framework, governance, leadership and stewardship, responsibilities (product liability, patient rights protection);
Scope and purpose
In these eras of development education became a paramount albeit fragmented element next to the research element, in the beginning largely laboratory focused. A literature study disclosed the historical eras, which started with patient-oriented research, knowledge and practice:
Historical eras
| Era | Focus | Key Figures |
|---|---|---|
| 1700s | patient – (treatment) oriented | Denis, Emmerez, Lewison |
| 1800s | patient – (treatment) oriented | Blundell, Virchov |
| 1900s first half | patient – (treatment) oriented | Landsteiner, Levine, Broman, Mollison |
| 1900s second half | test tube – (safety) oriented | immunohematology, micro-organisms (hepatitis B and C, HIV) |
| 1975 | Four WHO key elements | Safety, Purity, Potency and Clinical Efficacy of the blood supply |
| 1991 | WHO initiates global transfusion medicine education | trigger HIV/AIDS and a series of emerging pathogens (e.g., viruses, prions), need for uniformity, quality and safety |
| 2000s | revival (treatment) patient oriented | patient blood management (PBM) and organization, regulatory framework, education (teaching and training, knowledge and skills); Aide Mémoires, Distance Learning Material (DLM, paper and digital) focused on primary processes |
Quality management turn of the Century a Quality Management Training (QMT) course was developed and regionally implemented but not sustained (pilot Harare, Zimbabwe, 1st course in Groningen, the Netherlands); Governance, organization and structure, legal framework and operational (legal) responsibilities; WHO initiated an Academic Institute for International Development of Transfusion Medicine (IDTM) at the University of Groningen, the Netherlands focused on the still existing gap – governance, organization and structure, legal framework and operational (legal) responsibilities (product liability and patient rights protection), which forms a justified reason for separation of the manufacturing (product liability) and the clinical application functions (patient right protection).
Academic Institute for International Development of Transfusion Medicine
The existing knowledge and science gap was filled by the establishment of the Academic Institute for International Development of Transfusion Medicine. The original objectives of IDTM were:
- Focus on the developing world (economically restricted);
- Postgraduate Master course (management and Leadership (MMTM);
- Guidance and advice on promotion of Transfusion Medicine implementation;
- International knowledge and science resource (knowledge economy focused on LMICs);
- Active promotion of applied scientific research (scientific publications, PhD thesis, scientific publications).
To implement the academic program of IDTM into the blood supply, a second scientific initiative was developed in 2004: the non-for-profit consultancy for International Development of Quality Management in Transfusion Management (IQM Consulting) was initiated on a non-for-profit basis. This institute offers a wealth of experience and knowledge in the field of Transfusion Medicine and related Health Sciences. The emphasis is on the principles of quality culture covering a broad area of themes and documents. Such as the implementation of the integration of Transfusion Medicine in the national Health Care System but also capacity building knowledge and skills through knowledge economy, applied academic science and research, and most important an international network and reference functions.
And what else?
International Quality Management consulting provides tailor-made professional services in the field of Transfusion Medicine and related Health Sciences through custom-oriented guidance and advice, filling knowledge gaps with tailor-made education programs, initiating applied research and development programs to improve on the evidence-based quality management and operations embedded in an attractive governance environment and appealing climate. It provides access to scientific work for which WHO established and maintains accessible libraries in 4 of the 6 Regional offices and at the headquarter in Geneva:
- HQ Geneva Global Index Medicus (GIM)
- EMR Index Medicus EMR (IMEMR)
- SEAR Index Medicus SEAR (IMSEAR)
- WPR Index Medicus WPR (IMWPR)
- AR African Index Medicus
Additionally EMRO in Cairo supports annually the International Student Congress on Bio-Medical Science (ISCOMS) providing as an educational gesture the World Health Award to the winner of the best global public health abstract and presentation; a one year subscription to the Eastern Mediterranean Health Journal (EMHJ).
Conclusion
Last but certainly not least: IQM Consulting provides its guidance and advice activities according to WHO principles as developed by the WHO Blood Transfusion Safety (BTS) unit of the Department of Essential Health Technologies (EHT) in Geneva. This guidance and advice function includes an extensive and tailor-made education (teaching and training) activity, preferably in-country to avoid privileging individual fellows, and using knowledge economy (sharing) contributing to the universal availability of safe, effective and quality-assured blood and blood products.
Conflict of interest: The author declares to have no conflicts of interest.
Funding: No financial support was received.
References:
- WHO.
- Hoff HE, Guillemin R. (1983) The first experiments on transfusion in France. Hist Med Allied Sci; 18:103-124
- Denis J. Concerning a new way of curing sundry diseases by transfusion of blood. (1667) Phil Trans R Soc Lond3on 3:489-504
- Young JH. James Blundell (1790-1878) Experimental physiologist and obstetrician. (1964) Med Hist 8:159
- Leacock JH. On the transfusion of blood in extreme cases of haemorrhage. (1816) Med Chir J & Rev 3:276
- Blundell J. Observations on transfusion of blood. (1828) The Lancet 2:321-324
- Landsteiner K. Uber Agglinationserscheinungen normalem menschlichem Blutes. (1901) mWiener Klein Wochenschr 14:1132-1134
- IQM website https://www.iqmconsulting.nl/
- WHO. Global Index Medicus (GIM), accessible through
- Index Medicus for the WHO Eastern Mediterranean Region (IMEMR), accessible through www.applications.emro.who.int/library/imjournals/
- WHO SEAR. Index Medicus for the WHO South-East Asia Region (IMSEAR), accessible through www.searo.who.int/publications/library_resources/en/
- WHO WPR. Index Medicus for the WHO Western Pacific Region (IMWPR) accessible at https://www.globalindexmedicus.net/biblioteca/wprim/
- WHO AFR. Index Medicus for the WHO Africa Region (AIM) accessible at https://www.globalindexmedicus.net/biblioteca/aim/
- ISCOMS web site https://iscoms.com/
- WHO. Action framework to advance universal access to safe, effective and quality-assured blood products 2020-2923. Geneva, World Health Organization; (2020.) Licence: CC BY-NC-SA 3.0 IGO

