Antifungal Susceptibility of Candida Species: Colorimetric Method
Determination of Antifungal Susceptibility of Candida Species Isolated from Clinical Samples using Colorimetric Method
Iskender Karalti 1, Günay Tülay Colakoglu 2, Zuhal Tekkanat Tazegun 3, Zehra Kipritci 4, Yeşim Gürol 5, Gülden Celik 4
- Central Laboratory of Educational Therapeutic Clinic, Azerbaijan Medical University, Baku, Azerbaijan.
- Department of Biology, Faculty of Science, Marmara University Istanbul, Türkiye.
- Medical Microbiology, Laboratory Burdur State Hospital, Burdur, Türkiye.
- Department of Medical Microbiology, Medical Faculty, Bahcesehir University, Istanbul Türkiye.
- Medical Microbiology Laboratory, Beykoz State Hospital, Istanbul, Türkiye.
OPEN ACCESS
PUBLISHED 30 November 2025
CITATION Karalti, I., Tülay, GT., et al., 2025. Determination of Antifungal Susceptibility of Candida Species Isolated from Clinical Samples using Colorimetric Method. Medical Research Archives, [online] 13(11). https://doi.org/10.18103/mra.v13i11.7131
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i11.7131
ISSN 2375-1924
ABSTRACT
It was aimed to determine the antifungal susceptibility of Candida species isolated from patients by the colorimetric method, one of the standard modifications. Therefore ‘Sensititre YeastOne’ using an indicator alamar blue standard commercial kit was used.
The samples from patients applied to Yeditepe University Hospital Medical Microbiology Laboratory between years 2009-2011 were included to the study. 149 strains were isolated from 136 samples. These strains were isolated from abscess, mouth swab, bronchoalveolar lavage (BAL), sputum, urine and urine catheter, blood culture, catheter, tracheal aspirate, sterile body fluid, vaginal swab and wound swab samples. Candida, Candida glabrata, Candida parapsilosis, Candida tropicalis, Candida dubliniensis, Candida krusei and, Candida incons./norveqen. of genus Candida were isolated.
Antifungal susceptibility tests were applied for seven antifungals including fluconazole, caspofungin, voriconazole, amphotericin B, posaconazole, itraconazole and 5-flusitosine. There was no resistant strain to caspofungin although the most resistant group was found in azole group antifungals. The resistance rates for fluconazole, voriconazole, itraconazole, 5-flusitosine, respectively were as 18.8%, 13.4%, 37.6%, 2.7%. For amphotericin B and posaconazole minimum inhibition concentration (MIC) values because of in vivo-in vitro discrepancy, were excluded from the evaluation. It was performed by E TEST method, because of high fluconazole resistance rate and the same resistance rate was observed. Because of easy evaluation, this test is found as a practical test for antifungal susceptibility.
Keywords: Candida, Colorimetric, Antifungal, Sensititre YeastOne.
Introduction
The Candida species are present at the normal flora of skin, mucosa and gastrointestinal system. They are colonized at the mucosae immediately after the birth and pose a risk for the endogenous injection. The increase seen at the diagnostic and therapeutic interventional procedures which have been applied to the patients along with the development of modern medicine, the transplantation applications, the use of broad spectrum antibiotic and frequent incidence of immunosuppression have led to increase on the rate of Candida albicans and non albicans infections 1. Along with the increase observed at the Candida infections, the resistance against the antifungals used has been also gradually increasing 2. The Candida 3 and Aspergillus 4 infections are most frequently seen among the fungal infections and the mortality of these infections occur at high level 5. It is thought that the transplantation patients, neutropenic cases, allergic reactions and the increase at the numbers of patients who receive corticosteroid treatment at high doses and other immunosuppressive patient groups may lead to the increase of mortality 4.
At Candida infections, Candida albicans is the specie which is most frequently seen especially in the invasive candidiasis. Besides that, Candida glabrata, Candida tropicalis, Candida parapsilosis and Candida kruse are also frequently isolated from the clinical samples 6. Candida species may be also identified with various biochemical tests and commercial kits. The success ratio of commercial kits is very high 7.
Antifungal susceptibility tests allow for the use of antifungal drug which is the most appropriate one for the factor detected. The macro and microdilution methods certified by CLSI (Clinical Laboratory Standards Institute) and The European Committee on Antimicrobial Susceptibility Testing (EUCAST) are used for the antifungal susceptibility. These methods most frequently seen in microdilution methods is the detection of MIC (minimum inhibitor concentration) value which is done with RPMI 1640 (2% glucose) agar. In addition to that, the conformity of microdilution based colorimetric methods were determined with reference methods and the use was increased due to the ease of implementation 8. In the recent years, the use of susceptibility kit which is the commercial type of colorimetric method has increased. Its use and evaluation is practical as this method is based on the principle of color change 2. Also, the result of susceptibility may simultaneously be obtained for 7 unit antifungals.
Material and Methods
Material: In this paper, the samples of patients who applied to Yeditepe University Hospital Microbiology Laboratory between the dates of December 2009 and May 2011 and the strains grew on these samples were used. The strains used in our study were isolated from abscess, mouth swab, bronchoalveolar lavage (BAL), sputum, urine and urine catheter, blood culture, catheter, tracheal aspirate, sterile body fluid, vaginal swab and wound swab samples.
Method: The Candida strains were produced from the clinical samples with the microbiological culture method and their identifications were made with the routine methods. The germ tube test was primarily applied to the strains and the pigmentation statuses were reviewed in the ChromAgar Candida medium. The strains of which germ tube test was genitive and the strains which do not appear like C. albicans in the ChromAgar Candida medium were identified with API ID 32C, Biomerieux (France) kit.
Then, the antifungal susceptibility tests of Candida strains isolated. For the antifungal susceptibility tests, there are two methods certified by CLSI (Clinical Laboratory Standards Institute, USA). These are macro and microdilution tests. Some methods which were created with the modifications of these reference methods were developed in order to meet the requirements of clinical laboratories in this regard 8. In our study, it was aimed to determine the antifungal susceptibilities of Candida species isolated by using the colorimetric method which is one of these new standard modifications. With this aim, standard commercial Sensititre YeastOne kit at which alamar blue is used as colorimetric indicator was used in determination of MIC.
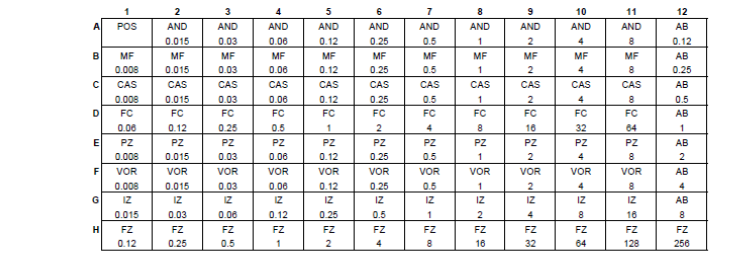
The susceptibility antifungal is a colorimetric microdilution test and used for the assigned of antifungal susceptibility of Candida and Cryptococcus species. It is a method at which qualitative and quantitative Minimum Inhibitor Concentration (MIC) is assigned in a dried plaque format.
Taken MIC results of resistant strains were also performed with E TEST (Biomerieux, Germany) method as the susceptibility ratio of Fluconazole was determined as high.
Implementation and evaluation of susceptibility test: Primarily, Candida colonies were suspended in the way that will be 0.5 McFarland in the distilled water and distributed into the plaques in the way that the final concentration will be 1.5-8×103 CFU/ml. The YeastOne plaque was covered with a guard band and incubated at 35˚C for 24-25 hours for Candida species. For control, 10 µl Sabouraud Dextrose Agar was cultured from YeastOne Broth suspension. 15-80 colony reproduction was waited. The control tests were also implemented with various standard strains (Candida albicans (ATCC 90028), Candida parapsilosis (ATCC 22019), Candida glabrata (KUEN 1573), Candida tropicalis (KUEN 1025) and Candida krusei (ATCC 6258)). In the evaluation, the value belonging the first well which did not give positive color was accepted as the minimum inhibitor 9.
The evaluation of antifungals as susceptible or resistant were made by taking the values suggested by the kit into basis. When there is no reproduction in the plaque, the blue color occurs and this is the indicator of susceptibility of antifungals.

| Name of Antifungal | Susceptible | Medium Susceptible | Resistant |
|---|---|---|---|
| Caspofungin | ≤ 2 | > 2 | |
| Fluconazole | ≤ 8 | ≥16 | |
| 5-Flusitosine | ≤4 | 8 – 16 | ≥32 |
| Itraconazole | ≤0.12 | ≥ 1 | |
| Voriconazole | ≤1 | >4 |
Implementation and evaluation of E Test: 28 unit strains which were detected as resistant with susceptibility were studied with E Test method. For this method, each strain which was taken through sterile swab was spread on RPMI 1640 agar surface with 2% glucose. Following the dehydration of plaques, fluconazole (0.016-256 µg/ml) E Test strips were placed into the central of plaques. They were taken into evaluation at the end of 24 hours’ incubation at 37°C 9.
Results
In our study, total 136 samples were studied and 149 units Candida strains were isolated. The species isolated were identified as phenotype. In accordance with the results obtained, 54.4% of strains isolated were identified as Candida albicans and 45.6% were identified as Non-albicans Candida.
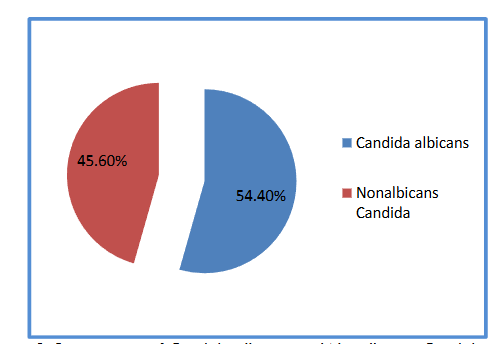
Then, advanced identification was implemented at Non-albicans Candida strains and Candida parapsilosis (15.4%), Candida tropicalis (14.1%) and Candida glabrata (12.1%) at the rate close to each other were isolated. Candida krusei (2.0), Candida dubliniensis (1.3%) and Candida incons./norveqen (0.7) in very small quantities were identified.
| Name of species | Quantity (%) |
|---|---|
| Candida albicans | 81 (54.4) |
| Candida parapsilosis | 23 (15.4) |
| Candida tropicalis | 21 (14.1) |
| Candida glabrata | 18 (12.1) |
| Candida krusei | 3 (2.0) |
| Candida dubliniensis | 2 (1.3) |
| Candida incons./norveqen. | 1 (0.7) |
The antifungal susceptibility tests were performed against 7 different antifungals (Fluconazole, Caspofungin, Voriconazole, Amphotericin B, Posaconazole, Itraconazole and 5-Flusitosine) with Sensitire YeastOne kit based on the colorimetric method. Any strain resistant to caspofungin was not determined among the echinocandins. The highest resistance ratio was determined against the itraconazole (37.6%) among the azole group antifungals. The fluconazole (18.8%) and voriconazole (13.4%) followed this. The resistance against 5-flusitosine included into the pyrimidine group was observed at 2.7% of the strains isolated.
| Antifungal | Susceptible | Medium Susceptible | Resistant |
|---|---|---|---|
| Fluconazole | 121 (81.2%) | 28 (18.8%) | |
| Voriconazole | 129 (86.6%) | 20 (13.4%) | |
| Itraconazole | 93 (62.4%) | 56 (37.6%) | |
| 5-Flusitosin | 141 (94.6%) | 3 (2.0%) | 4 (2.7%) |
| Caspofungin | 149 (100%) | 0 (0.0%) |
In terms of resistance ratios, it was determined that the highest resistance was against fluconazole in C. albicans.
| Name of Antifungal | C. albicans | C. parapsilosis | C. glabrata | C. tropicalis |
|---|---|---|---|---|
| Fluconazole | 14.8% | 4.3% | 27.8% | 33.3% |
| Voriconazole | 9.9% | 4.3% | 0.0% | 28.6% |
| Itraconazole | 22.2% | 8.7% | 77.8% | 81.0% |
| 5-Flusitosine | 1.2% | 0.0% | 5.6% | 19.0% |
| Caspofungin | 0.0% | 0.0% | 0.0% | 0.0% |
The resistance ratio of fluconazole was determined as 18.8% with E Test. MIC50 and MIC90 values determined in terms of species are given in the following table.
| Fluconazole | Voriconazole | Posaconazole | Itraconazole | 5-Flusitosine | Amphotericin B | Caspofungin | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
|
|
|
Discussion
Candida species may show resistance against the antifungal drugs with various mechanisms. Today, there are antifungal drugs which have various mechanisms of action although they are not too much. Selecting the right antifungal is very significant in order to increase the success of treatment. This is because Candida species have natural resistance against some antifungals. For example; some of C. glabrata species have natural resistance against the azole group antifungal drugs. In order that, antifungal susceptibility tests are required to be conducted 10.
In the recent years, it has been observed that C. albicans strains show resistance against the azoles as primarily fluconazole 11. It was determined that C. albicans strains which were isolated in our study were mostly resistant against the azole group antifungals and a strain which was resistant against an azole group antifungal were determined as resistant against other antifungals in almost every group. It is thought that the repetitive use of azoles has led to the resistance increasing against the azoles and the candidiasis which fail to respond the systemic antifungal treatment 10. C. albicans strains were determined as high susceptible against the antifungals outside the azole group. This case is also stated in similar studies (12). As this specie is Candida specie which is most frequently isolated among the clinical samples, knowing the antifungal susceptibilities carries significant. This is seen especially in the patients who are in intensive care units and women as infection factor 13-14. It is the primary one of the fungal species which lead to opportunistic infections in the patients of which immune systems have been suppressed. C. albicans specie 7 which is the main factor of superficial or deep acute or chronic fungal infections, respiratory tract infections or systemic fungal infections were frequently isolated in our study.
C. parapsilosis is the primary one of nosocomial infections which are frequently isolated in the blood samples. It is frequently seen not only in oncology and transplantation patients but also in the patients who are in the intensive care units. It is put forth that various surgical interventions and long hospitalization durations lead to this 15. The increase of C. parapsilosis infections has drawn attraction in the recent years 16. In our study, the specie which is most isolated is C. parapsilosis following C. albicans. Also, 18 of 23 strains isolated were isolated from blood culture and the patients at whom this specie was isolated were the intensive care patients who had to hospitalized in the hospitality for long time or the patients who received oncology treatment. In various studies like our study, it is stated that this specie is most isolated among Non-albicans Candida strains in Europe and Latin America 17-19. In C. parapsilosis strains, susceptible results at high ratios were taken. In a study which was conducted in Spain in 2011, the antifungal susceptibilities of C. parapsilosis strains were studies and it was stated that susceptibility at high ratios were obtained for voriconazole, amphotericin B and anidulafungin 15.
In terms of antifungal susceptibility results of C. glabrata strains, it was seen that its resistance ratio was high against the azole group antifungals. Fluconazole resistant in C. glabrata strains were seen in elder patients. While resistance against the azoles have increased in the recent years, the dose increase in treatment sometimes give effective results (20). In our study, fluconazole resistance was seen at five strains in this specie and these strains were isolated from the patients who were above 65 years.
C. glabrata is the second most frequent candidemia factor in USA. But C. tropicalis or C. parapsilosis take the second place in European countries. C. glabrata is frequently seen in the patients at whom fluconazole is used at prophylaxis and old people are seen at an ever-increasing frequency and the infections depending on this specie progress more seriously and fatal. In Turkey, as European countries, C. parapsilosis and C. tropicalis are the species which make most frequent candidemia following C. albicans. C. tropicalis candidemia are seen in the patients who rather have neutropenia and mucositis and especially the patients who have not received fluconazole prophylaxis 21.
The candidemia based on C. krusei are rather seen in the patients who have hematologic malignity, bone marrow and blood receptors and especially in the patients to whom fluconazole prophylaxis is applied.
C. dublininesis is a Candida specie which is frequently isolated from the persons infected with HIV and confused with C. albicans due to the germ tube positivity in the routine diagnosis methods. In our study, two units were isolated among these species and differentiated in ChromAgar Candida medium. Therefore, it is certain that this medium assist in the diagnosis.
Sensititre YeastOne Kit may be used for Candida, Cryptococcus, Aspergillus which reproduce easily and various yeast species which reproduce fast. It is not convenient for using detecting the antifungal susceptibilities of Histoplasma or Blastomyces of which reproduction is hard or slow and filamentous molds. In the studies which were conducted in order to determine the antifungal susceptibilities of Candida species, it was determined that the results obtained with Sensititre Yeast One kit were in conformity with the reference methods at the rate of 85% 22.
Various researchers believe that reading MIC results after 24 hours’ incubation is more convenient than reading after 48 hours’ incubation. But it is emphasized that CLSI 48 hours’ reading is more successful. But as there is not sufficient data, the time of accurate reading remains unanswered. Therefore, the time of reading should be also clearly stated in reporting. At the end of 24 hours, the reading should not be made if the control well does not completely become positive. Also, the discoloration in the wells is analyzed in accordance with the complete discoloration not the principle of turbidity. Thanks to that, the challenges in the determination of antifungal susceptibilities of candida strains which yield different MIC values and named as trailing are recovered as a result of 24 and 48 hours’ incubations. These strains are rather isolated from the blood and other sterile body fluids 9.
When the resistance ratios in our country and the resistance ratio of C. albicans (14.8%) strains against the fluconazole that we obtained in our study; we observed high rate of resistance 23-25. The view of that high resistance ratio against fluconazole may be trailing was created. But these data should be verified with more samples. The fluconazole resistance ratio was also studied with E Test method certified by FDA due to this high ratio acquired and it was determined at same level. While the resistance ratio of C. albicans strains against the fluconazole was determined as 5.5% in a study conducted by Altuncu et al. (2010), it was stated that the response which was clinically acquired was higher compared to this ratio due to in vivo-in vitro discrepancy 26.
It is indisputable that the non-albicans species showed an increase in candida infections and the resistance of these species against the antifungal drugs was high and therefore determination of factor at specie level in Candida infections and research of antifungal susceptibility is beneficial in terms of effectiveness of treatment and prevention of resistance development 27.
Due to that the diagnosis and treatment challenges are very much in candidemia and invasive candidiasis, the prevention and control of these diseases gain importance. There are three effective, easy and cheap methods in this regards; hand hygiene, catheter care and prudent and careful use of antifungals 28.
Conclusion
In conclusion, this method may be suggested due to that its evaluation as a result of the data obtained is objective. But it is required to study with more samples.
Conflicts of Interest Statement: All authors declare that they have no conflicts of interest.
Acknowledgement: This study was supported by Marmara University Scientific Research Projects Commission (BAPKO) with the project no FEN-C-DRP-080410-0090. We thank to Rector’s Office of Marmara University for your contributions.
References
- Iyer RS, Pal RB, Patel RY, Banker DD. Polymerase chain reaction based diagnosis of systemic fungal infections and sensitivity testing of the fungal isolates. Indian J. Med Microbiol., 2002; 20:132-136.
- Gürol Y, Karaltı I, Oztürk Y, Tazegün TT, Yılmaz, G. Poster: Onkoloji servisinde yatan hastalardan izole edilen Candida türlerinin kolorimetrik yöntem ile antifungal duyarlılıklarının belirlenmesi. 24. ANKEM Congress, Fethiye, May, 2009:106.
- Kuo-Wei C, Hsiu-Jung L, Yu-Hui L, Shu-Ying L. Comparison of four molecular typing methods to assess genetic relatedness of Candida albicans clinical isolates in Taiwan. J. of Med. Microbiol. 2005; 54:249–258.
- Pasqualotto A.C. Differences in pathogenicity and clinical syndromes due to Aspergillus fumigatus and Aspergillus flavus. Med. Mycol. 2009; 47:1-10.
- Gharizadeh B, Norberg E, Löffler J et al. Identification of medically important fungi by the Pyrosequencing technology. Mycoses. 2004; 47:29-33.
- Schabereiter-Gurtner C, Selitsch B, Rotter ML, Hirschl AM, Willinger B. Development of novel real-time PCR assays for detection and differentiation of eleven medically important Aspergillus and Candida species in clinical specimens Journal of Clinical Microbiology 2007; 45:906–914.
- Tarini NMA, Wahid MH, Fera I, Yasmon A, Djauzi S. Development of multiplex-PCR assay for rapid detection of Candida spp. Medical Journal of Indonesia 2010; 19:83-87.
- Carrillo-Muñoz AJ, Quindós G. Ruesga M et al. In vitro antifungal susceptibility testing of filamentous fungi with Sensititre Yeast One. Mycoses. 2006; 49:293-7.
- Sensititre YeastOne Antifungal Susceptibility Test Kit Prospectus. TREK DiagnosticSystems UK 2010;1-10.
- Wilke TA, Güner S, Doğanay M. Enfeksiyon Hastalıkları ve Mikrobiyolojisi. Ankara: Nobel Medical Publishing, 2008.
- Chryssanthou E. Trends in antifungal susceptibility among Swedish Candida species bloodstream isolates from 1994 to 1998 Comparison of the E-test and the Sensititre YeastOne colorimetric antifungal panel with the NCCLS M27-A reference method. Journal of Clinical Microbiolology. 2001; 39:4181–4183.
- Espinel-Ingroff A, Pfaller M. Messer AS. Multicenter comparison of the Sensititre YeastOne colorimetric antifungal panel with the National Committee for Clinical Laboratory Standards M27-A reference method for testing clinical isolates of common and emerging Candida spp., Cryptococcus spp., and other yeasts and yeast-like organisms. Journal of Clinical Microbiology. 1999; 37:591–595.
- Martina R, Wächtlerb B, Schallerc M, Wilsonb D, Hubeb B. Host–pathogen interactions and virulence-associated genes during Candida albicans oral infections. International Journal of Medical Microbiology. 2011; 301:417-422.
- Wächtler B, Wilson D, Haedicke K, Dalle F, Hube B. From attachment to damage Identified genes of Candida albicans mediate adhesion, invasion and damage during interaction with oral epithelial cells. Plos One. 2011; 23:1-14.
- de Toro M. Torres MJ, Maite R, Aznar J. Characterization of Candida parapsilosis complex isolates. Clinical Microbiolology and Infection. 2011; 17:418-424.
- Borghi E, Sciota R, Iatta R, Biassoni C, Montagna MT, Morace G. Characterization of Candida parapsilosis complex strains isolated from invasive fungal infections. European Journal of Clinical Microbiology & Infectious Diseases. 2011; 30:1-5.
- Tortorano AM, Biraghi E, Astolfi A et al. European Confederation of Medical Mycology (ECMM) prospective survey of candidaemia: report from one Italian region. Journal of Hospital Infection. 2002; 41:297–304.
- Almirante B, Rodríguez D, Cuenca-Estrella M. Epidemiology, risk factors, and prognosis of Candida parapsilosis bloodstream infections: Case–control population-based surveillance study of patients in Barcelona, Spain, from 2002 to 2003. Journal of Clinical Microbiolology. 2006; 44:4181–4183.
- Pfaller MA, Diekema DJ. Epidemiology of invasive candidiasis: a persistent public health problem. Clinical Microbiology Reviews. 2007; 20:133–163.
- Balkan II, Savaş A, Geduk A, Yemisen M, Mete B, Ozaras R. Candida glabrata perinephric abscess. The Eurasian Journal of Medicine. 2011; 43:63-65.
- Willke A. Kandidemi: Nasıl değerlendirilmeli ne yapılmalı? Turkish Journal of Infection. 2007; 21:117-122.
- Koç NA. Antifungal duyarlılık testleri ve klinik önemi. Ankem Journal. 2010; 26 (Annex 2):270-276.
- Demirbilek M, Timurkaynak F, Can F, Azap O, Arslan H. Hastane kaynaklı Candida türlerinde biyofilm oluşumu ve antifungal duyarlılık paternleri. Microbiological Bulletin. 2007; 41: 261-269.
- Evci C, Ener B, Göral G, Akçağlar S. Comparative evaluation of the antifungal susceptibility of Candida isolates from blood specimens: results of a study in a tertiary care hospital in Bursa, Turkey. Turk J Med Sci. 2010; 40(1):141-149.
- Atalay MA; Sav H, Demir G, Koç NA. Kan kültürlerinden izole edilen Candida türlerinin dağılımı ve amfoterisin B ve flukonazole in vitro duyarlılıkları. Medicine Journal of Selçuk. 2012; 28(3):149-151.
- Altuncu E, Bilgen E, Çerikçioğlu E, Ilki A, Ulger N, Bakır M, Akman I, Ozek E. Neonatal kandida enfeksiyonları ve etkenlerinin antifungal duyarlılıkları. Microbiological Bulletin. 2010; 44: 593-603.
- Koçoğlu E, Bayram A, Balcı I. Klinik örneklerden izole edilen kandida türleri ve antifungal duyarlılıkları. Medical Journal of Van. 2005; 12:195-200.
- Cornely OA, Sprute R, Bassetti M, Chen SC, Groll AH, Kurzai O, Lass-Flörl C, Ostrosky-Zeichner L, Rautemaa-Richardson R, Revathi G, Santolaya ME, White PL, Alastruey-Izquierdo A, Arendrup MC, Baddley J, Barac A, Ben-Ami R, Brink AJ, Grothe JH, Guinea J, Hagen F, Hochhegger B, Hoenigl M, Husain S, Jabeen K, Jensen HE, Kanj SS, Koehler P, Lehrnbecher T, Lewis RE, Meis JF, Nguyen MH, Pana ZD, Rath PM, Reinhold I, Seidel D, Takazono T, Vinh DC, Zhang SX, Afeltra J, Al-Hatmi AMS, Arastehfar A, Arikan-Akdagli S, Bongomin F, Carlesse F, Chayakulkeeree M, Chai LYA, Chamani-Tabriz L, Chiller T, Chowdhary A, Clancy CJ, Colombo AL, Cortegiani A, Corzo Leon DE, Drgona L, Dudakova A, Farooqi J, Gago S, Ilkit M, Jenks JD, Klimko N, Krause R, Kumar A, Lagrou K, Lionakis MS, Lmimouni BE, Mansour MK, Meletiadis J, Mellinghoff SC, Mer M, Mikulska M, Montravers P, Neoh CF, Ozenci V, Pagano L, Pappas P, Patterson TF, Puerta-Alcalde P, Rahimli L, Rahn S, Roilides E, Rotstein C, Ruegamer T, Sabino R, Salmanton-García J, Schwartz IS, Segal E, Sidharthan N, Singhal T, Sinko J, Soman R, Spec A, Steinmann J, Stemler J, Taj-Aldeen SJ, Talento AF, Thompson GR 3rd, Toebben C, Villanueva-Lozano H, Wahyuningsih R, Weinbergerová B, Wiederhold N, Willinger B, Woo PCY, Zhu LP. Global guideline for the diagnosis and management of candidiasis: an initiative of the ECMM in cooperation with ISHAM and ASM. Lancet Infect Dis. 2025 May; 25(5):e280-e293. doi: 10.1016/S1473-3099(24)00749-7. Epub 2025 Feb 13. Erratum in: Lancet Infect Dis. 2025 Apr;25(4):e203. doi: 10.1016/S1473-3099(25)00154-9. Erratum in: Lancet Infect Dis. 2025 May;25(5):e261. doi: 10.1016/S1473-3099(25)00252-X.

