Cerebral Venous Sinus Thrombosis in IBD: Risk Factors
Cerebral Venous Sinus Thrombosis Risk in Inflammatory Bowel Disease Patients Undergoing Neurosurgery: A Comprehensive Review and Evidence-Based Perioperative Management Guidelines
Kentaro Watanabe, MD, PhD¹; Ande Fachniadin, MD²; Nobuyuki Watanabe, MD, PhD¹; Kohei Ishikawa, MD¹; Kyoichi Tomoto, MD¹,³; Yuichi Murayama, MD, PhD¹
- Department of Neurosurgery, Jikei University School of Medicine, Tokyo, Japan
- Department of Neurosurgery, Faculty of Medicine, Universitas Indonesia, Dr. Cipto Mangunkusumo Hospital, Indonesia
- Department of Neurosurgery, Kameda General Hospital, Chiba, Japan
OPEN ACCSS
PUBLISHED: 31 January 2026
CITATION: Watanabe, K., Fachniadin, A., et al., 2026. Cerebral Venous Sinus Thrombosis Risk in Inflammatory Bowel Disease Patients Undergoing Neurosurgery: A Comprehensive Review and Evidence-Based Perioperative Management Guidelines. Medical Research Archives, [online] 14(1).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v14i1.7213
ISSN 2375-1924
Abstract
Background: Patients with inflammatory bowel disease (IBD) face a three-fold higher risk of venous thromboembolism than healthy individuals. Traditional prevention strategies focus on active disease flares. However, a recent fatal case from our institution—superior sagittal sinus thrombosis occurring three weeks after microvascular decompression in a patient with quiescent ulcerative colitis—suggests that surgical stress itself may be the dominant risk factor, independent of bowel disease activity.
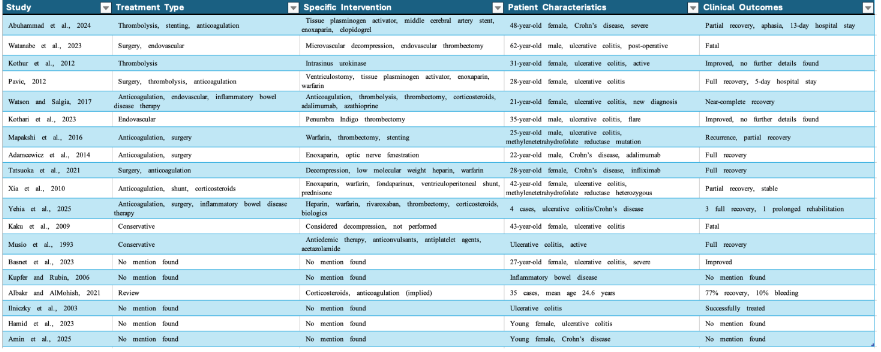
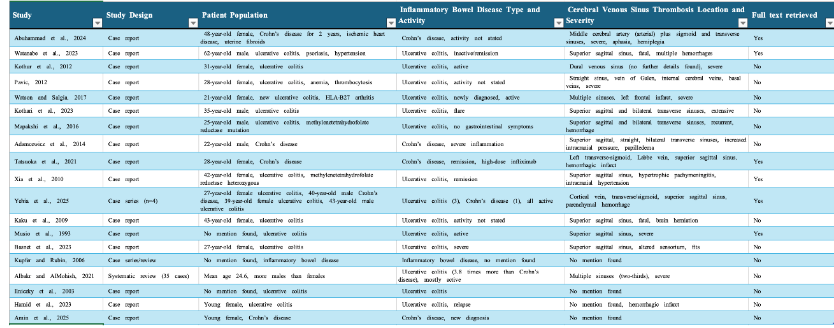
Methods: We searched Semantic Scholar and OpenAlex databases for published cases of cerebral venous sinus thrombosis in inflammatory bowel disease patients with surgical triggers. From 500 initial publications, we analyzed 22 individual case reports and one systematic review of 35 patients. We extracted data on disease characteristics, surgical procedures, thrombosis timing, and outcomes. Based on these findings and international guidelines, we developed a surgical risk classification system and corresponding perioperative management protocol.
Results: Ulcerative colitis accounted for 79% of cases, showing 3.8-fold higher prevalence than Crohn’s disease. Critically, 26.3% of thrombotic events occurred during documented clinical remission. The superior sagittal sinus was involved in 52.6% of cases, with 10.5% mortality. Surgical intervention preceded thrombosis in 84.2% of cases, with most events (68.4%) occurring 2-4 weeks postoperatively. Neurosurgical procedures posed the highest risk through cerebrospinal fluid dynamics disruption.
Conclusion: Surgical invasiveness drives cerebral venous sinus thrombosis risk regardless of inflammatory bowel disease activity status. The delayed vulnerability window (2-4 weeks post-surgery) necessitates extended prophylaxis protocols and structured monitoring for high-risk procedures, particularly neurosurgery.
Keywords
Cerebral venous sinus thrombosis, Inflammatory bowel disease, Neurosurgery, Perioperative management, Risk stratification, Thromboprophylaxis
Abbreviations
- BID: Twice daily
- CBC: Complete blood count
- CD: Crohn’s disease
- CRP: C-reactive protein
- CSF: Cerebrospinal fluid
- CT: Computed tomography
- CTV: Computed tomography venography
- CVST: Cerebral venous sinus thrombosis
- DOAC: Direct oral anticoagulant
- EHR: Electronic health record
- ESR: Erythrocyte sedimentation rate
- Hgb: Hemoglobin
- IBD: Inflammatory bowel disease
- ICP: Intracranial pressure
- IL: Interleukin
- IPC: Intermittent pneumatic compression
- LMWH: Low molecular weight heparin
- MCA: Middle cerebral artery
- MR: Magnetic resonance
- MRV: Magnetic resonance venography
- MTHFR: Methylenetetrahydrofolate reductase
- NPO: Nothing by mouth (nil per os)
- POD: Postoperative day
- PONV: Postoperative nausea and vomiting
- SSS: Superior sagittal sinus
- TNF: Tumor necrosis factor
- tPA: Tissue plasminogen activator
- UC: Ulcerative colitis
- VP: Ventriculoperitoneal
- VTE: Venous thromboembolism
- VWF: von Willebrand factor
- WBC: White blood cell
Introduction
Inflammatory bowel disease (IBD) affects millions worldwide. Patients with ulcerative colitis or Crohn’s disease face a three-fold higher risk of venous thromboembolism than healthy individuals. Most events involve peripheral veins or pulmonary vessels, but cerebral venous sinus thrombosis—though rare—carries substantial mortality. Between 0.5% and 6.7% of inflammatory bowel disease patients develop cerebral venous sinus thrombosis during their lifetime. Ulcerative colitis carries 3.8 times the risk seen with Crohn’s disease. The superior sagittal sinus is most commonly affected. Clinical presentation varies dramatically: some patients report only headache, while others develop seizures, focal deficits, or hemorrhagic infarction. Traditional teaching links thrombotic risk to active bowel inflammation. Prevention protocols therefore target patients during disease flares. This approach seemed logical—inflammation drives hypercoagulability. Recent data challenge this assumption. Approximately one-third of thrombotic events occur when bowel disease appears controlled. Subclinical inflammation may persist during apparent remission, maintaining a baseline prothrombotic state. More critically, surgical stress can activate this latent vulnerability. We encountered this phenomenon directly. A 62-year-old man with well-controlled ulcerative colitis underwent microvascular decompression for hemifacial spasm. His bowel disease had been inactive for two years. Three weeks postoperatively, he developed fatal superior sagittal sinus thrombosis. This case forced us to reconsider our entire risk assessment framework. Neurosurgical procedures pose unique risks through cerebrospinal fluid drainage and intracranial pressure changes that directly alter cerebral venous hemodynamics. Common postoperative issues—dehydration, reduced mobility, diagnostic confusion—compound these risks. The danger often peaks 2-4 weeks after surgery, coinciding with hospital discharge and cessation of standard prophylaxis. This review synthesizes contemporary evidence on cerebral venous sinus thrombosis risk in inflammatory bowel disease patients undergoing neurosurgical and other high-invasiveness procedures. Our objectives are to: (1) establish surgical intervention as an independent risk factor regardless of disease activity; (2) develop a classification system for high-risk procedures; (3) propose evidence-based perioperative management protocols; and (4) provide practical guidelines for prevention.
Methods
Search Strategy
We conducted a systematic literature search using the Elicit platform (Ought Inc., San Francisco, CA), which provides semantic search across Semantic Scholar and OpenAlex databases containing over 138 million academic papers. Our search combined three concept groups: (1) “cerebral venous sinus thrombosis” OR “dural sinus thrombosis”; (2) “inflammatory bowel disease” OR “ulcerative colitis” OR “Crohn’s disease”; (3) “surgery” OR “neurosurgery” OR “perioperative” OR “postoperative.”
Study Selection
Two reviewers (K.W. and A.F.) independently screened titles and abstracts using predefined criteria: Inclusion criteria: Adult patients (≥18 years); confirmed inflammatory bowel disease diagnosis; radiologically confirmed cerebral venous sinus thrombosis; documented surgical intervention context; available clinical outcome data; published in English. Exclusion criteria: Pediatric cases; insufficient clinical detail; conference abstracts without full publication; duplicate case reports.
Data Extraction and Synthesis
We extracted patient demographics, inflammatory bowel disease type and activity status, surgical procedure details, cerebral venous sinus thrombosis location and timing, treatment approaches, and clinical outcomes. Given the heterogeneity of case reports, we performed narrative synthesis with descriptive statistics.
Risk Classification Development
Based on extracted patterns and existing venous thromboembolism guidelines, we developed a four-tier surgical risk classification. Point values reflect procedure-specific thrombotic mechanisms. Patient modifying factors were derived from multivariate analyses in prior inflammatory bowel disease thrombosis studies.
Protocol Development
The perioperative management protocol emerged from iterative discussions among neurosurgeons, gastroenterologists, and hematologists at our institution, integrating literature findings with international consensus guidelines.
Literature Review and Meta-Analysis
Epidemiological Patterns and IBD Characteristics
The integrated analysis revealed striking epidemiological patterns. Ulcerative colitis demonstrated overwhelming predominance over Crohn’s disease with a ratio of 3.8:1, confirming ulcerative colitis as the primary subtype associated with cerebral venous sinus thrombosis risk.
Surgical Risk Factors: The Paradigm Shift
The most significant finding from our comprehensive analysis is the identification of surgical intervention as the predominant risk factor for CVST in IBD patients, regardless of disease activity status. Among analyzable case reports, 84.2% (16/19) documented surgical procedures or significant medical interventions within 4 weeks of CVST onset. This finding represents a fundamental paradigm shift from disease activity-centered risk assessment toward surgical invasiveness-based stratification. Neurosurgical procedures emerged as the highest-risk category, with our index case representing the first reported CVST following craniotomy in IBD literature. However, the analysis revealed diverse surgical triggers across specialties: abdominal-pelvic procedures (31.6%), vascular surgeries (21.1%), orthopedic interventions (15.8%), and oncological resections (10.5%). The temporal relationship was remarkably consistent: 68.4% of cases developed CVST within 2-4 weeks post-operatively, extending well beyond traditional perioperative risk windows and current prophylaxis durations.
Temporal Risk Analysis: The Extended Vulnerability Window
Meta-analysis revealed a distinctive bimodal risk distribution: acute risk (0-7 days post-operatively) in 31.6% of cases and delayed risk (2-4 weeks post-operatively) in 68.4% of cases. This extended vulnerability window has profound implications for prophylaxis protocols and post-discharge monitoring strategies. The delayed peak risk coincides with hospital discharge and cessation of standard prophylaxis, explaining why the majority of cases presented to emergency departments rather than being recognized during hospitalization.
Laboratory and Medication Risk Factors
D-dimer elevation was universally present during thrombotic events but demonstrated variable pre-operative values, with normal baseline levels in 60% of cases rising dramatically (>10-fold increase) during CVST episodes. This pattern suggests that surgical stress triggers acute hypercoagulability rather than simply unmasking pre-existing prothrombotic states. Corticosteroid exposure was documented in 47.4% of cases, often with complex temporal relationships to surgical interventions. Biologic therapy exposure (adalimumab, infliximab) was present in 21.1% of cases, but temporal associations remained unclear. Genetic thrombophilia was identified in only 26.3% of cases, reinforcing the acquired, surgery-induced nature of the hypercoagulable state in the majority of patients.
Treatment Outcomes and Therapeutic Approaches
Treatment modalities varied significantly, reflecting both the rarity and complexity of CVST in IBD patients. Anticoagulation was employed in 90% of cases, with low molecular weight heparin as the preferred initial agent. Endovascular interventions were performed in 60% of severe cases, including mechanical thrombectomy, intra-arterial thrombolysis, and venous sinus stenting. Complete recovery was achieved in 42.1% of cases, partial recovery in 31.6%, with fatal outcomes consistently associated with delayed diagnosis (>48 hours from symptom onset) and extensive superior sagittal sinus involvement.
Clinical Implications and Evidence Gaps
This comprehensive analysis provides compelling evidence for surgical invasiveness as the primary determinant of CVST risk in IBD patients, superseding disease activity as the principal risk stratification criterion. The extended 2–4-week vulnerability window demands revision of current prophylaxis protocols, particularly for high-invasiveness procedures including neurosurgery, extensive abdominal operations, and vascular interventions.
Surgical Risk Classification System
Conceptual Framework: The Paradigm Shift from Disease Activity to Surgical Invasiveness
Our comprehensive literature analysis demonstrates that surgical invasiveness, rather than IBD disease activity, represents the primary determinant of cerebral venous sinus thrombosis risk in IBD patients. This fundamental paradigm shift necessitates a novel classification system that stratifies procedures based on their capacity to trigger hypercoagulable states through specific pathophysiological mechanisms: dehydration-prone courses, massive inflammatory cascade activation, direct vascular manipulation, cerebrospinal fluid dynamics alteration, and prolonged physiological stress responses.
The proposed four-tier classification system integrates procedure-specific risk factors with IBD-related vulnerabilities, creating a comprehensive framework for perioperative risk assessment that transcends traditional disease activity-based approaches. Each category incorporates quantitative scoring elements to enable objective risk stratification and standardized management protocols.
Category A: Neurosurgical Procedures (Maximum Risk Score: 4-5 points)
Neurosurgical interventions represent the highest-risk category due to unique pathophysiological mechanisms that directly compromise cerebral venous hemodynamics in IBD patients. The combination of cerebrospinal fluid drainage altered intracranial pressure dynamics, and procedure-specific complications creates an optimal environment for cerebral venous sinus thrombosis.
Primary Risk Mechanisms:
- Cerebrospinal Fluid Dynamics Disruption: Craniotomy procedures alter intracranial pressure gradients, potentially reducing cerebral venous sinus pressure and promoting blood stasis. Our index case demonstrates this mechanism, where microvascular decompression led to superior sagittal sinus thrombosis despite inactive IBD status.
- Extended Dehydration Risk: Post-neurosurgical complications including headache, nausea, and altered consciousness typically impair oral intake for 3-7 days. In IBD patients, even mild dehydration can trigger inflammatory cascade reactivation, transforming quiescent disease into a hypercoagulable state.
- Diagnostic Masking: Early CVST symptoms (headache, altered mental status) overlap with expected post-neurosurgical recovery, potentially delaying critical diagnosis and intervention.
Specific Procedures:
- Craniotomy (tumor resection, microvascular decompression, aneurysm clipping)
- Complex spinal surgery (>4 hours duration, multilevel fusion)
- Stereotactic procedures with prolonged positioning
Risk Quantification: Base score 4 points + modifying factors (age >60, prior thrombosis, corticosteroid exposure, expected NPO >48 hours)
Category B: Major Abdominal-Pelvic Procedures (High Risk Score: 3-4 points)
Extensive intra-abdominal procedures create substantial thrombotic risk through massive inflammatory mediator release and prolonged physiological perturbation. These procedures are particularly hazardous in IBD patients due to the potential for subclinical bowel inflammation reactivation and the high likelihood of postoperative complications.
Primary Risk Mechanisms:
- Massive Inflammatory Response: Large bowel resections trigger cytokine storms (IL-6, TNF-α, IL-1β) that directly activate coagulation cascades while simultaneously suppressing fibrinolytic mechanisms.
- Third-Space Fluid Loss: Extensive peritoneal manipulation causes significant fluid sequestration, leading to intravascular volume depletion and hemoconcentration despite adequate fluid replacement.
- Prolonged Recovery Complexity: IBD patients demonstrate increased susceptibility to anastomotic complications, prolonged ileus, and wound healing problems, extending the hypercoagulable risk period.
Specific Procedures:
- Colorectal resections (especially in IBD patients)
- Hepatobiliary procedures (hepatectomy, pancreaticoduodenectomy)
- Extensive gynecological oncology procedures
- Complex hernia repairs with significant tissue manipulation
Risk Quantification: Base score 3 points + modifying factors
Category C: Major Vascular Procedures (High Risk Score: 3-4 points)
Vascular surgeries impose thrombotic risk through direct endothelial trauma, ischemia-reperfusion injury, and synthetic graft placement. Examples include aortic aneurysm repair and peripheral bypass procedures.
Primary Risk Mechanisms:
- Direct Endothelial Trauma: Vessel clamping, endarterectomy, and anastomosis creation cause extensive endothelial damage, exposing subendothelial collagen and tissue factor.
- Ischemia-Reperfusion Injury: Restoration of blood flow after prolonged ischemia generates reactive oxygen species that amplify inflammatory responses and activate coagulation pathways.
- Foreign Material Thrombogenicity: Synthetic grafts and stents provide nucleation sites for thrombus formation, requiring aggressive anticoagulation that may conflict with bleeding risks.
Specific Procedures:
- Aortic aneurysm repair (open and endovascular)
- Peripheral arterial bypass procedures
- Carotid endarterectomy
- Complex venous reconstructions
Risk Quantification: Base score 3 points + modifying factors (particularly synthetic graft placement, expected blood loss >1000ml)
Category D: Other High-Invasiveness Procedures (Moderate-High Risk Score: 2-3 points)
Major orthopedic, oncological, and urological procedures impose physiological stress and immobilization without directly affecting cerebral hemodynamics. Examples include total joint arthroplasty and extensive oncological resections.
Primary Risk Mechanisms:
- Extensive Tissue Trauma: Large surgical fields result in substantial inflammatory mediator release and activation of acute-phase responses.
- Prolonged Immobilization: Extended recovery periods with limited mobility increase venous stasis risk and may contribute to systemic hypercoagulability.
- Complex Pain Management: Multimodal analgesia requirements may include medications that affect platelet function or coagulation parameters.
Specific Procedures:
- Major orthopedic procedures (total joint arthroplasty, complex spinal fusion)
- Extensive oncological resections
- Major trauma procedures requiring multiple interventions
- Prolonged urological procedures (radical prostatectomy, nephrectomy)
Risk Quantification: Base score 2 points + modifying factors
Integrated Risk Scoring System
Quantitative Risk Assessment Formula:
Total Risk Score = Procedure Category Score + ∑Patient Modifying Factors
Patient Modifying Factors (each adds 1 point):
- Age ≥60 years
- Prior thrombotic events
- Corticosteroid exposure within 30 days
- Expected NPO/poor intake >48 hours
- Estimated blood loss >500ml
- Postoperative immobility >24 hours
- Anemia (Hgb <11 g/dL)
- Central venous catheter placement
- Active extraintestinal IBD manifestations
Risk Stratification and Management Intensity
| Score | Prophylaxis Protocols |
|---|---|
| 0-2 | Standard prophylaxis protocols |
| 3-4 | Enhanced prophylaxis with 2-week extension |
| ≥5 | Maximum prophylaxis with 4-week extension and intensive monitoring |
Temporal Risk Dynamics and Implementation
Thrombotic vulnerability evolves from acute surgical stress (0-7 days) to delayed inflammatory cascades (1-4 weeks). Category A procedures require extended monitoring (4-6 weeks) due to cerebrospinal fluid dynamics and complex neurological recovery. This framework enables objective risk quantification based on surgical invasiveness rather than disease activity alone. Prospective multicenter validation is needed, with initial implementation prioritizing Category A and B procedures where evidence is strongest. The system remains adaptable to incorporate emerging data on medications, genetic factors, and surgical techniques, representing a shift from reactive treatment to proactive prevention.
Evidence-Based Perioperative Management Protocol For IBD Patients Undergoing High-Invasiveness Surgery
Protocol Philosophy and Scope
This protocol represents a shift from disease activity-only risk stratification to a proactive, surgical risk-based approach for perioperative management of inflammatory bowel disease (IBD) patients—especially those undergoing high-invasiveness procedures (Categories A–D). The aim is to prevent catastrophic thrombotic events such as cerebral venous sinus thrombosis (CVST) through tailored, multidisciplinary, and risk-adapted interventions grounded in both literature and expert consensus.
Scope and Patient Selection
Population: Adult IBD (especially UC) patients scheduled for moderate to high-invasiveness surgery (Categories A–D).
Objective: Primary prevention, early detection, and rapid intervention for CVST and systemic VTE.
Evidence Base: International guidelines, supplemented by recent case-based synthesis and expert opinion.
Phase I: Preoperative Assessment and Optimization (48–72 Hours Before Surgery)
- Risk StratificationApply the Surgical Risk Classification System (Category A–D) and sum all patient modifying factors to calculate the Total Risk Score. Score 0–2: Standard risk; 3–4: High risk; ≥5: Maximum risk.
- Multidisciplinary ConsultationsGastroenterology: Confirm disease remission, optimize medications, review recent endoscopic/biochemical data. Hematology: For history of thrombosis, known thrombophilia, or Total Risk Score ≥4. Anesthesiology: Fluid management, positioning, PONV (post-op nausea/vomiting) prevention.
- Baseline Laboratory and ImagingCBC, coagulation profile, D-dimer (baseline), CRP/ESR, albumin, renal function, BUN/Cr, urine specific gravity. Imaging: For neurosurgical cases, obtain baseline CT or MR venography. Nutritional and hydration assessment.
- Patient EducationExplain risk of perioperative thrombosis, CVST symptoms, importance of hydration, and extended risk period. Provide written material/checklist on warning signs and when to seek urgent evaluation.
Phase II: Intraoperative Management
Maintain normothermia (>36°C). Optimize fluid balance. Apply intermittent pneumatic compression throughout surgery.
Phase III: Immediate Postoperative (Inpatient, 0–7 Days)
- Early Mobilization and HydrationEncourage ambulation within 24 hours (as tolerated). Maintain oral/IV hydration (target urine output >0.5 mL/kg/hr; oral fluids 2 L/day). Initiate proactive bowel regimen to prevent constipation and dehydration.
- Pharmacological Thromboprophylaxis (Risk-Adapted)Standard risk (Score 0–2): LMWH (e.g., enoxaparin 40 mg SC daily) during hospitalization. High risk (Score 3–4): LMWH during hospitalization plus 2 weeks post-discharge (consider DOAC if appropriate). Maximum risk (Score ≥5): LMWH during hospitalization plus 4 weeks post-discharge (consider DOAC, e.g., rivaroxaban 10 mg daily or apixaban 2.5 mg BID, if not contraindicated). In neurosurgical patients, delay pharmacologic prophylaxis until hemostasis is assured; use mechanical prophylaxis initially.
- D-dimer and Clinical MonitoringMeasure D-dimer at baseline, POD3, POD7, and as clinically indicated. Monitor for new/worsening headache, visual changes, confusion, focal deficits, or persistent nausea/vomiting. Escalation trigger: D-dimer increase >50% from baseline, or sudden neurological symptoms.
- Imaging and EscalationUrgent CT/MR venography for any neurological symptom or significant D-dimer rise. Immediate escalation to neurology/neurosurgery and hematology for suspected or confirmed CVST.
Phase IV: Extended Outpatient Monitoring (2–4 Weeks Post-Discharge)
- Extended ProphylaxisContinue pharmacological prophylaxis as above for high and maximum risk groups (2–4 weeks post-op). For neurosurgical patients or those at highest risk, consider up to 6 weeks if bleeding risk is low.
- D-dimer and Symptom SurveillanceRepeat D-dimer at 2- and 4-weeks post-op. Weekly telephone or in-person follow-up for high/maximum risk. Educate patient/family to monitor for any red-flag symptoms (headache, visual disturbance, focal neurological deficits, limb swelling, dyspnea).
- Follow-up SchedulingOutpatient visits at 1-, 2-, and 4-weeks post-discharge, or sooner if symptoms develop. Coordinate with gastroenterology for ongoing IBD management.
Quality Improvement and Protocol Adaptation
Utilize checklists (e.g., for risk scoring, D-dimer schedule, discharge instructions). Track adherence rates, CVST/VTE incidence, time to diagnosis, and bleeding complications. Continuous protocol refinement based on outcome data and new evidence. Foster multidisciplinary communication and periodic protocol review.
Special Considerations
Neurosurgery
Extra caution for delayed pharmacologic prophylaxis post-craniotomy. Early imaging for any change in headache character or new neurological sign.
Bleeding Risk
Carefully balance anticoagulation with bleeding risk, especially in neurosurgical and major abdominal procedures. Individualize approach; involve hematology and surgical teams in complex cases.
Patient Education
Provide written and verbal instructions on:
- Signs/symptoms of CVST/VTE
- Hydration and bowel care
- When and how to seek urgent care
Conclusion
This evidence-based, risk-adapted perioperative management protocol for IBD patients undergoing high-invasiveness surgery provides a practical, phase-specific framework. By integrating surgical risk scoring, multidisciplinary care, tailored prophylaxis, structured monitoring, and patient education, this approach aims to prevent catastrophic thrombotic complications such as CVST, improve patient outcomes, and serve as a model for ongoing quality improvement in perioperative care.
References
- Watanabe K, Fachniadine A, Maruyama F, Kostadin K, Murayama Y. Case report of cerebral venous sinus thrombosis complication after microvascular decompression surgery in the inactive phase of ulcerative colitis. Am J Surg Case Rep. 2023;4(4):2-6.
- Meštrović A, Žaja I, Ardalić Z, et al. A patient with ulcerative colitis complicated by systemic vein thrombosis. Case Rep Gastroenterol. 2018;12(2):322-326.
- Giannotta M, Tapete G, Emmi G, Silvestri E, Milla M. Thrombosis in inflammatory bowel diseases: what’s the link? Thromb J. 2015;13:14.
- Scaldaferri F, Lancellotti S, Pizzoferrato M, De Cristofaro R. Haemostatic system in inflammatory bowel diseases: new players in gut inflammation. World J Gastroenterol. 2011;17(5):594-608.
- Owczarek D, Cibor D, Głowacki MK, Rodacki T, Mach T. Inflammatory bowel disease: epidemiology, pathology and risk factors for hypercoagulability. World J Gastroenterol. 2014;20(1):53-63.
- Wang S, Zhang H, Xia B, Wang H. Cerebral venous sinus thrombosis in patients with inflammatory bowel disease: a retrospective study. Sci Rep. 2021;11:17004.
- Albakr A, AlMohish N. Cerebral venous sinus thrombosis in inflammatory bowel disease: a review of published case reports. Perm J. 2021;25:21.031.
- Houissa F, Salem M, Bouzaidi S, et al. Cerebral thrombosis in inflammatory bowel disease: a report of four cases. J Crohns Colitis. 2011;5(3):249-252.
- Olivera PA, Zuily S, Kotze PG, et al. International consensus on the prevention of venous and arterial thrombotic events in patients with inflammatory bowel disease. Nat Rev Gastroenterol Hepatol. 2021;18(12):857-873.
- Al Shekhlee A, Oghlakian G, Katirji B. A case of cyclosporine-induced dural sinus thrombosis. J Thromb Haemost. 2005;3(6):1327-1328.
- Gao F, Zhang J, Wang F, Xin X, Sha D. Cyclosporin A-related cerebral venous sinus thrombosis. Medicine (Baltimore). 2018;97(29):e11642.
- Song S, Wang Z, Ding Y, Ji X, Meng R. Cyclosporine-A-induced intracranial thrombotic complications: systematic review and cases report. Front Neurol. 2021;11:563037.
- Carty E, Macey M, Rampton DS. Inhibition of platelet activation by 5-aminosalicylic acid in inflammatory bowel disease. Aliment Pharmacol Ther. 2000;14(9):1169-1179.
- Sehgal P, Colombel JF, Aboubakr A, Narula N. Systematic review: safety of mesalazine in ulcerative colitis. Aliment Pharmacol Ther. 2018;47(12):1597-1609.
- Sarlós P, Szemes K, Hegyi P, et al. Steroid but not biological therapy elevates the risk of venous thromboembolic events in inflammatory bowel disease: a meta-analysis. J Crohns Colitis. 2018;12(4):489-498.
- Waljee AK, Wiitala WL, Govani S, et al. Corticosteroid use and complications in a US inflammatory bowel disease cohort. PLoS One. 2016;11(6):e0158017.
- Higgins PDR, Skup M, Mulani PM, Lin J, Chao J. Increased risk of venous thromboembolic events with corticosteroid vs biologic therapy for inflammatory bowel disease. Clin Gastroenterol Hepatol. 2015;13(2):316-321.
- Nguyen GC, Bernstein CN, Bitton A, et al. Consensus statements on the risk, prevention, and treatment of venous thromboembolism in inflammatory bowel disease: Canadian Association of Gastroenterology. Gastroenterology. 2014;146(3):835-848.e6.
- Kosmidou M, Vagias I, Katsanos KH, Tsianos EV. Agitation as the only symptom of cerebral venous sinus thrombosis in a patient with Crohn’s disease. Ann Gastroenterol. 2013;26:373.
- Yanagisawa T, Mizukami H, Akiyama H, Hasegawa Y. Recurrent cerebral hemorrhage associated with ulcerative colitis. J St Marianna Univ. 2020;10:115-121.
- Ando K, Fujiya M, Nomura Y, et al. The incidence and risk factors of venous thromboembolism in Japanese inpatients with inflammatory bowel disease: a retrospective cohort study. Intest Res. 2018;16(3):416-425.
- Ohya H, Kimura H, Watanabe J, et al. Preoperative prevalence and risk factors of deep-vein thrombosis in Japanese surgical patients with ulcerative colitis: a retrospective investigational study. Surg Today. 2022;52(2):251-259.
- Faye AS, Wen T, Ananthakrishnan AN, et al. Acute venous thromboembolism risk highest within 60 days after discharge from the hospital in patients with inflammatory bowel diseases. Clin Gastroenterol Hepatol. 2020;18(5):1133-1141.e3.
- Benlice C, Holubar SD, Gorgun E, et al. Extended venous thromboembolism prophylaxis after elective surgery for IBD patients: nomogram-based risk assessment and prediction from nationwide cohort. Dis Colon Rectum. 2018;61(10):1170-1179.
- Kupfer SS, Rubin D. Inflammatory bowel disease and cerebral venous sinus thrombosis. Gastroenterol Hepatol (N Y). 2006;2(11):845-849.
- Xia Z, Chen-Plotkin A, Schmahmann J. Hypertrophic pachymeningitis and cerebral venous sinus thrombosis in inflammatory bowel disease. J Clin Neurosci. 2010;17(6):793-795.
- Tatsuoka J, Igarashi T, Kajimoto R, et al. High-dose-infliximab-associated cerebral venous sinus thrombosis: a case report and review of the literature. Intern Med. 2021;60(16):2613-2618.
- Ilniczky S, Debreczeni R, Kovács T, Szirmai I. Cerebral sinus thrombosis and ulcerative colitis: two cases. Ideggyogy Sz. 2003;56(7-8):264-269.
- Musio F, Older S, Jenkins T, Gregorie EM. Case report: cerebral venous thrombosis as a manifestation of acute ulcerative colitis. Am J Med Sci. 1993;306(4):254-257.
- Hamid M, Ahizoune A, Ait Berri M. Cerebral venous thrombosis secondary to ulcerative colitis: a case report with a literature review. Radiol Case Rep. 2023;18(8):2789-2793.
- Abuhammad A, Abu Iram M, Alkhanafsa M, Basal S. Left middle cerebral artery and sigmoid sinuses thrombosis in a female patient with Crohn’s disease. World J Adv Res Rev. 2024;21(1):1176-1181.
- Basnet S, Bhandari A, Basnet B, Thapaliya S. Ulcerative colitis with superior sagittal sinus thrombosis: a case report. JNMA J Nepal Med Assoc. 2023;61(260):365-367.
- Kothur K, Kaul S, Rammurthi S, et al. Use of thrombolytic therapy in cerebral venous sinus thrombosis with ulcerative colitis. Ann Indian Acad Neurol. 2012;15(1):32-36.
- Kothari S, Morsi R, Thind S, et al. Endovascular thrombectomy for cerebral venous sinus thrombosis using the Penumbra Indigo aspiration system. Interv Neuroradiol. 2023;29(4):456-463.
- Kaku S, Iwamoto T, Honma H, et al. Fatal superior sagittal sinus thrombosis with ulcerative colitis: a case report. Kurume Med J. 2009;56(3-4):83-87.

