Characterization of Cannabis-Derived Extracellular Vesicles
Characterization of cannabis plant-derived extracellular vesicles for biomedical applications
Ivy N. Thweatt¹²³, Sandani V.T. Wijerathne¹², Bernard B. Efa¹², Chioma C. Ezeuko¹², Ayodeji O. Ipinmoroti¹², Rachana Pandit¹², Junhuan Xu²³, Kelvin A. Fluker²³, Olufemi Ajayi²³ and Qiana L. Matthews¹²
- Microbiology Program, College of Science, Technology, Engineering and Mathematics, Alabama State University, Montgomery, AL 36104, USA
- Department of Biological Sciences, College of Science, Technology, Engineering and Mathematics, Alabama State University, Montgomery, AL 36104, USA
- Industrial Hemp & Specialty Crops Program, Alabama State University, Montgomery, AL 36104, USA
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Thweatt, I.N., Wijerathne, SVT., et al., 2024. Characterization of cannabis plant-derived extracellular vesicles for biomedical applications. Medical Research Archives, [online] 12(10).
https://doi.org/10.18103/mra.v12i10.5405
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5405
ISSN 2375-1924
Abstract
The scientific interest in cannabis plants is continuously growing, with heightened interest in properties of plant-derived extracellular vesicles. This manuscript focuses on isolating and characterizing vesicles originating from cannabis plants. Establishing the most appropriate and efficient isolation procedure for plant vesicles remains a challenge due to vast differences in the physio-structural characteristics of different plant cultivars within the same species, and different species within the same genera. In this study, we employed a crude but standard isolation procedure for the extraction of apoplastic wash fluid, which is known to contain plant-derived extracellular vesicles. This method includes a detailed stepwise process of plant-derived extracellular vesicles extraction from two (2) cultivars of cannabis plants, namely: Citrus and BaOx. Approximately, 150 leaves were collected from each plant strain. In order to collect plant-derived extracellular vesicles pellets, apoplastic wash fluid was extracted from the plants via negative pressure permeabilization and infiltration followed by high-speed differential ultracentrifugation. Apoplastic wash fluid fractions were collected for Citrus and BaOx fractions, P100 and P40. Particle tracking analysis of plant-derived extracellular vesicles revealed particle sizes ranging from 60 to 160 nanometers. Both cultivar fractions yielded high levels of plant-derived extracellular vesicles, and contained enriched plant-derived extracellular vesicles RNA levels. Our results suggest that the cannabis apoplastic wash fluid fractions (P100 and P40) yielded plant-derived extracellular vesicles. In total, the results provide a guide for the selection and optimization of cannabis-derived extracellular vesicles. Subsequently, these cannabis plant-derived vesicles can be used for further biomedical uses.
Keywords
Plant-derived extracellular vesicles; Cannabis; Cannabidiol; Cultivar; Apoplastic wash fluid; Extracellular vesicles
1. Introduction
Hemp, Cannabis sativa L., is one of the oldest cultivated plants. The 2014 and 2018 Farm Bills legalized hemp production in the United States. In recent years, there has been a resurgence of interest in cannabis use, largely fueled by its valuable byproducts. This renewed focus on cannabis and its beneficial derivatives has ignited significant enthusiasm across diverse fields such as medicine, agriculture, and pest management. Cannabis, a controversial plant with complicated pharmacological qualities that is now at the center of a growing industry, presents both distinct regulatory and economic obstacles as well as medicinal and economic prospects. Indoor cannabis cultivation allows growers to control environmental conditions such as temperature, humidity, air circulation, and sunlight for both vegetative and flowering developmental stages. Cultivation in greenhouses also makes it possible to modify the abiotic factors for the ideal agricultural environment. The highest chances of success, higher yields, better quality, and higher earnings all depend on managing environmental dangers. Cannabis has had a variety of medical uses, including reducing anxiety and depression, specifically, cannabidiol (CBD) has been investigated for its use to decrease anxiety and aggression in cats and dogs. In addition, CBD-based products have been used for inflammation and pain reduction in companion animals. The use of CBD products is regulated differently throughout the world. For medical uses in humans, CBD is used to treat anxiety neurological disorders, gynecological pain, and to alleviate pain related to chronic non-malignant pain as well as malignant pain management. Cannabis is such a versatile plant that it has become necessary to evaluate its plant-derived materials and cargo, namely, extracellular vesicles (EVs). With increasing knowledge of plant EVs, it has become important to investigate these vesicles for cell-to-cell communication.
Extracellular vesicles are a class of vesicles that are released by several organisms including plants. Plant-derived extracellular vesicles (PDEVs) were isolated and functionally characterized to show that they include biomolecules such as, lipids, proteins, metabolites, and short RNAs (sRNA). PDEVs are tiny membrane-enclosed structures secreted by plants and are used for intercellular communication. They play a crucial role in orchestrating molecular interactions, particularly in the context of plant-plant, plant-host microbe, or vector interactions, especially during the activation of defense responses. In this study, we established and optimized PDEV isolation protocols for CBD-derived cultivars (Citrus and BaOx) fractions, P100 and P40. Results from this study could allow for improved cultivation of cannabis and generating cannabis-PDEVs. This work serves as a brief method report.
2. Methods
PDEV ISOLATION
Leaves of Citrus and BaOx cannabis plants (5 to 6 months old) were obtained and subjected to saturation in the buffer solution to obtain the apoplastic wash fluid (AWF). All samples were initially weighed to determine the dry biomass before the extraction process commenced. Subsequently, each leaf was immersed in deionized water for a duration of 5 minutes (min). Following the initial wash, each sample was dried and then placed in a 60 mL syringe, which was subsequently filled to the brim with an infiltration buffer solution. The buffer solution comprised 20 mM MES, 2 mmol CaCl, and 0.1 M NaCl with a pH level of 6. To permeabilize the leaves, a 60 mL syringe top (29.2 mm, diameter) was securely sealed, and pressure was applied by intermittently pulling back on the pump for a period of 20 min to create negative pressure or until it was evident that most of the leaves were sufficiently moist, indicating the buffer solution had permeated the vessels of the leaves. Following the second rinse with the buffer solution, the samples were dried, and their wet biomass was determined. Subsequently, each sample was carefully wrapped in plastic wrap and placed into a 50 mL conical tube. These tubes were then subjected to centrifugation at 1000 x g in a tabletop centrifuge for a duration of 10 min. Following this step, the supernatant was carefully extracted and transferred to a new conical tube for a subsequent round of centrifugation at 2000 x g for 10 min. After this step, the pellet was discarded. Next, a 0.45 μm filter was used to filter the buffer, then the media was collected and placed into another tube, which was then subjected to ultracentrifugation at 10,000 x g for 30 min, then the pellet was discarded. Following this, the remaining buffer was once again centrifuged, and the infiltration buffer was spun at a speed of 100,000 x g for a duration of one hour. After one hour of centrifugation, the buffer was discarded, and the resulting pellet was carefully collected. For the P40 fraction the same process was conducted with the exemption that the ultracentrifugation was performed at 40,000 x g for one hour.
NTA TRACKING ANALYSIS
The concentration (particles per mL) and size distribution (nm) of isolated PDEVs were analyzed using nanoparticle tracking analysis (NTA). NTA analyzes the size of the particle in fluids based on the rate of Brownian motion to dynamic light scattering (DLS). The diluted EV samples (1:75 in microbial cell culture grade water) were injected into the machine sample chamber of the ZetaViewR Particle Tracking Analyzer, and the mean values (concentration and size) of particles (mean standard deviation of the mean values) were recorded and analyzed in 11 separate locations for each sample using the ZetaViewR Analyze (version 8.50.14 SP7) software. NTA was used to determine the size and concentration of PDEVs in the P100 (N = 4) and P40 (N=3) samples.
PROTEIN QUANTITATION
Protein quantitation was determined in the PDEVs via the bicinchoninic acid assay. In brief, five microliters of standards (0, 0.2, 0.4, 0.8, and 1.6 µg/µL) of bovine serum albumin and EV samples were added in triplicate in a 96-well tissue culture plate. Subsequently, 25 µg of protein reagent A and 200 µL of protein reagent B were added to each well, and the plate was wrapped with aluminum foil and placed on a shaker for 10 min. The absorbance was recorded at 595 nm, and a standard curve was plotted to determine the exact concentration of total proteins in the isolated EVs.
DNA AND RNA QUANTITATION
Five µg of proteins was used to determine the total DNA and RNA contents of the PDEVs. The total DNA and RNA of the isolated PDEVs were extracted using TRIzol Reagent (Invitrogen) for total DNA and RNA precipitation. Prior to the TRIzol extraction, 5 µg of PDEVs were pretreated with 1 unit (U) of RNase-free DNAase I and 1 U of micrococcal nuclease (MNase) (Thermo Scientific) for DNA and RNA extraction, respectively. For total DNA, samples of the PDEVs were incubated with RNase-free DNase I at 37 °C in a water bath for 30 min followed by 50 mM EDTA treatment at 65 °C for 10 min and proceeded to DNA isolation using the TRIzol extraction method. For RNA extraction, the PDEVs were treated with 1% Triton-X-100 on ice for 30 min before Mnase treatment. Total levels of DNA and RNA derived from the PDEVs were quantitated using the NanoDrop One (Thermo Scientific).
3. Results
For these experiments, we employed a crude but standard isolation procedure for the extraction of AWF, which is known to contain PDEVs. All samples were processed equally as described above (Figure 1A). The AWF was subjected to centrifugation as described in the methods section. The mean size of the Citrus PDEVs (P100, P40) ranged between 150 to 170 nm, whereas the BaOx PDEVs (P100, P40) ranged in size from 160 to 60 nm (Figure 1B-C). The concentration of PDEVs particles/mL in Citrus and BaOx cultivars (total, 3.8 x107 particles, 6.7 x107 particles) had similar concentration trends with respect to the P100 and P40 fractions, however, there were slightly more PDEVs found in the BaOx P40 fraction (4.9 x107 particle/mL) (Figure 1C). Both P100 and P40 samples of PDEVs were subjected to DNA, RNA, and protein analyses. Both Citrus and BaOx P40 fractions had higher contents of DNA as compared to the P100 fractions (Figure 1D). The DNA concentration of both cultivars ranged from 50 ng/µL to 200 ng/µL (P100, P40), whereas the RNA concentration of both cultivars was substantially higher at 1000 ng/µL to 1500 ng/µL (Figure 1D). The protein levels of both cultivars were examined, with Citrus and BaOx being comparable within the P100 fraction (0.2 µg/mL); however, the protein levels in the P40 fraction of BaOx were increased relative to the Citrus cultivar (Figure 1D).
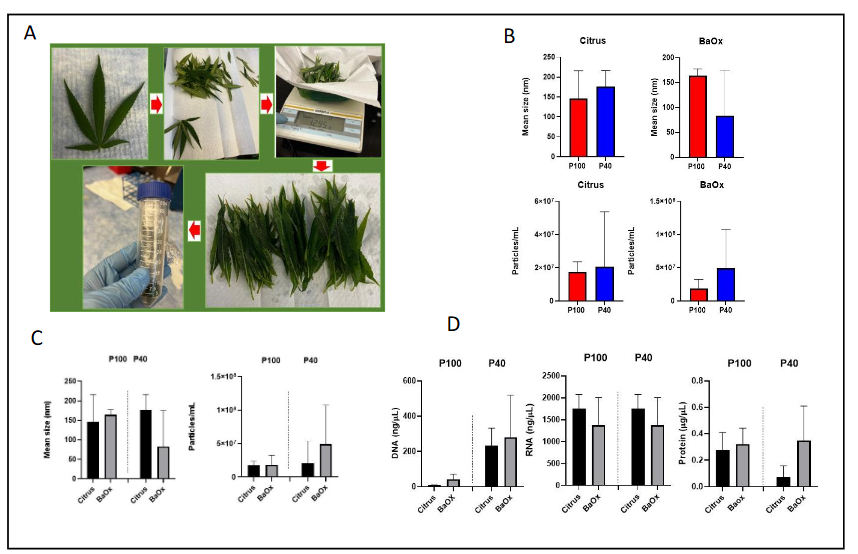
4. Discussion
Cannabidiol use and byproducts are important for animal and human health. A recent study demonstrated that CBD is a strong inhibitor of the release of EVs in cancer cell lines such as breast cancer adenocarcinoma (MDA-MB-231), prostate cancer (PC3) and hepatocellular carcinoma (HEPG2). This author’s work demonstrated that CBD sensitized cancer cells to chemotherapy. This study demonstrated that the anti-cancer effects are due partly to EV biogenesis, implying that CBD could be a therapeutic agent for EV-mediated pathological effects. Another study showed that exposure of glioblastoma cells to CBD resulted in the production of EVs with reduced levels of pro-oncogenic miR-21 and increased levels of anti-oncogenic miR-126 as compared to controls. Along these same lines, we demonstrated that alcohol administration demonstrated that alcohol significantly influences HeLa cell viability and exosome biogenesis and composition in comparison to controls. Similarly, we found that cocaine affects exosome biogenesis and composition in BV2, microglial cells. We found that cocaine exposure modulated the expression of exosomal proteins, such as heat shock proteins and Rab GTPases, suggesting the protein composition and formation of microglial-derived exosomes were regulated by cocaine.
Plant-derived vesicles are becoming increasingly important in biomedical and other manufacturing industries. Cho and colleagues performed an extensive analysis of several types of plants and determined that the PDEVs as a naturally bioactive substance, has different effects from the conventional plant extracts. CBD use and misuse can be monitored by vesicle evaluation. Ganesh et al. monitored Cannabis Use Disorder in young adults by evaluating cannabis related neuropathy after collection of neuro-derived EVs in biofluids. Studies similar to theirs have spurred our interest in CBD-PDEVs. We concluded that it is possible to extract PDEVs from two CBD cultivar APW fractions. Having the ability to precisely evaluate PDEVs from varying cultivar fractions, we can potentially engineer these plants to have the highest chances of success, higher yields, better quality, and higher earnings. We performed this study based on our previously reported and modified protocols. We believe that there might be some variations between plant species etc. However, these findings might be important to a researcher attempting to save time on sample extraction or who might not have access to an ultracentrifugation with the highest speed specifications.
In addition, PDEVs have been recently utilized for preclinical vaccine therapies. Specifically, PDEVs may represent a platform for the delivery of RNA-based vaccines, exploiting their natural membrane envelope to protect and deliver nucleic acids. These PDEVs share similarities to human EVs and can be administered orally and/or intranasally. As it relates to our study, we were able to obtain high yields of PDEVs with relatively few plants/leaf numbers. Again, these findings might be useful to the biomedical industry for therapeutic purposes and vaccine development. There are few studies focusing on CBD-derived EVs, making our study of field-wide importance. Tajik et. al. recently published that extracellular vesicles of cannabis with high CBD content induce anticancer properties in human hepatocellular carcinoma. This study evaluated two chemotypes of cannabis with varying levels of D-9-tetrahydrocannabinol (THC) and CBD. In this study, EVs were isolated from each chemotype via differential centrifugation followed by ultracentrifugation on a sucrose gradient. High-performance liquid chromatography analysis illustrated the lack of THC in EVs derived from both plants. Therefore, two types of EVs were classified according to their CBD content into high-(H.C-EVs) and low-CBD EVs (L.C-EVs).
The C. sativa plants from which we isolated the EVs contain CBD, and the level of THC in it is <0.3%. Herein, we evaluated additional CBD cultivars, Citrus and BaOx, to isolate and evaluate PDEVs P100 and P40 fractions. The findings in this study were impactful in light of the limited data on CBD-derived PDEVs, with our previous study being one of very few studies in the literature. Citrus and BaOx plants ages 4-5 months were evaluated. Both the P100 and P40 fractions were enriched with PDEVs, we observed that the BaOx cultivars were infested with the two-spotted spider mites Tetranychus urticae (Koch), whereas, the Citrus variety was not (data not shown). We speculated that the total BaOx particles/mL were higher than those of the Citrus particles/mL due to the spider mite infestation. The BaOx total particles of PDEVs were 1.7 times more than that of the Citrus PDEVs. BaOx and Citrus cultivars had similar trends of DNA and protein levels for both the AWF fractions, P100 and P40. The impact of parasite infestation will need to be further investigated with more plants and varying pest infestation levels/stages.
Cannabis is a very versatile plant, frequently grown in greenhouses. Insect infestation is one of the biggest issues with greenhouse farming; numerous losses are seen as a result. The two-spotted spider mite Tetranychus urticae (Koch), a tiny arachnid that is a major pest in greenhouse production. The two-spotted spider mites with piercing-sucking mouthparts feed on plant sap beneath the leaves of hemp plants. The spider mites drain the hemp plant’s chlorophyll, preventing photosynthesis. Weak webbing and “stippling,” which are areas of discoloration on the surface of the leaves, are indicators of the spider mites. The two-spotted spider mites can discolor entire leaves leading to the death of the plant. Integrated pest management strategies are valuable tools for reducing the reliance on pesticides while still controlling pest populations. In the greenhouse environments, infestation of some sort is always possible. Naturally occurring cultivars maybe more susceptible to this or another pest.
5. Conclusions
Our experiment highlights that PDEVs can be obtained from an infected cultivar. Further downstream applications may or may not be impacted by infestation. In conclusion, cannabis-derived vesicles and other plant vesicles are becoming a more useful tool as biomedical and/or cosmetic tools. Ultimately, usage of PVEDs could be just as important as the usage of the plant extracts, themselves.
Acknowledgments
This work was funded by the National Science Foundation (IOS-1900377), Center for Disease Control and Prevention (NU38OT000279), and the US Department of Education, Office of Postsecondary Education, and Institutional Services (Title III, part B, HBCU Program).
Conflicts of interests
There are no conflicts to declare.
Authors’ contribution
INT, SVTW, BBE, CCE, AOI, RP, KAF all contributed to the experimentation. INT, JX, OA, and QLM all contributed to the experimental design and the writing of the experiments.
References
- Agriculture Improvement Act of 2018.
- Agriculture Act of 2014.
- Fike J. Industrial Hemp: Renewed Opportunities for an Ancient Crop. Critical Reviews in Plant Sciences. 2016;35(5-6):406-424.
- Fordjour E, Manful CF, Sey AA, et al. Cannabis: a multifaceted plant with endless potentials. Frontiers in pharmacology. 2023;14:1200269. doi:10.3389/fphar.2023.1200269
- Small E. Evolution and Classification of Cannabis sativa (Marijuana, Hemp) in Relation to Human Utilization. Botanical Review. 08/19 2015;81:189-294. doi:10.1007/s12229-015-9157-3
- Bridgeman MB, Abazia DT. Medicinal Cannabis: History, Pharmacology, And Implications for the Acute Care Setting. P & T : a peer-reviewed journal for formulary management. Mar 2017;42(3):180-188.
- Hillig K. Genetic Evidence for Speciation in Cannabis (Cannabaceae). Genetic Resources and Crop Evolution. 01/03 2005;52:161-180. doi:10.1007/s10722-003-4452-y
- Corsato Alvarenga I, Panickar KS, Hess H, McGrath S. Scientific Validation of Cannabidiol for Management of Dog and Cat Diseases. Annual review of animal biosciences. Feb 15 2023;11:227-246. doi:10.1146/annurev-animal-081122-070236
- Yu CHJ, Rupasinghe HPV. Cannabidiol-based natural health products for companion animals: Recent advances in the management of anxiety, pain, and inflammation. Research in veterinary science. Nov 2021;140:38-46. doi:10.1016/j.rvsc.2021.08.001
- Hameed M, Prasad S, Jain E, et al. Medical Cannabis for Chronic Nonmalignant Pain Management. Current pain and headache reports. Apr 2023;27(4):57-63. doi:10.1007/s11916-023-01101-w
- Hill KP, Palastro MD. Medical cannabis for the treatment of chronic pain and other disorders: misconceptions and facts. Polish archives of internal medicine. Nov 30 2017;127(11):785-789. doi:10.20452/pamw.4123
- Patel AD. Cannabinoids in Neurologic Illnesses. Neurologic clinics. Feb 2021;39(1):231-241. doi:10.1016/j.ncl.2020.09.012
- Armour M, Sinclair J, Cheng J, et al. Endometriosis and Cannabis Consumption During the COVID-19 Pandemic: An International Cross-Sectional Survey. Cannabis and cannabinoid research. Aug 2022;7(4):473-481. doi:10.1089/can.2021.0162
- Moreno-Rius J. The Cerebellum, THC, and Cannabis Addiction: Findings from Animal and Human Studies. Cerebellum (London, England). Jun 2019;18(3):593-604. doi:10.1007/s12311-018-0993-7
- Suharta S, Barlian A, Hidajah AC, et al. Plant-derived exosome-like nanoparticles: A concise review on its extraction methods, content, bioactivities, and potential as functional food ingredient. Journal of food science. Jul 2021;86(7):2838-2850. doi:10.1111/1750-3841.15787
- Urzì O, Raimondo S, Alessandro R. Extracellular Vesicles from Plants: Current Knowledge and Open Questions. International journal of molecular sciences. May 20 2021;22(10) doi:10.3390/ijms22105366
- Huang Y, Wang S, Cai Q, Jin H. Effective methods for isolation and purification of extracellular vesicles from plants. Journal of integrative plant biology. Dec 2021;63(12):2020-2030. doi:10.1111/jipb.13181
- Alzahrani FA, Khan MI, Kameli N, Alsahafi E, Riza YM. Plant-Derived Extracellular Vesicles and Their Exciting Potential as the Future of Next-Generation Drug Delivery. Biomolecules. May 15 2023;13(5) doi:10.3390/biom13050839
- Ipinmoroti AO, Turner J, Bellenger EJ, et al. Characterization of cannabis strain-plant-derived extracellular vesicles as potential biomarkers. Protoplasma. Nov 2023;260(6):1603-1606. doi:10.1007/s00709-023-01870-6
- Pandit R, Ipinmoroti AO, Crenshaw BJ, Li T, Matthews QL. Canine Coronavirus Infection Modulates the Biogenesis and Composition of Cell-Derived Extracellular Vesicles. Biomedicines. Mar 21 2023;11(3) doi:10.3390/biomedicines11030976
- Kosgodage US, Mould R, Henley AB, et al. Cannabidiol (CBD) Is a Novel Inhibitor for Exosome and Microvesicle (EMV) Release in Cancer. Frontiers in pharmacology. 2018;9:889. doi:10.3389/fphar.2018.00889
- Kosgodage US, Uysal-Onganer P, MacLatchy A, et al. Cannabidiol Affects Extracellular Vesicle Release, miR21 and miR126, and Reduces Prohibitin Protein in Glioblastoma Multiforme Cells. Translational oncology. Mar 2019;12(3):513-522. doi:10.1016/j.tranon.2018.12.004
- Kumar S, Crenshaw BJ, Williams SD, Bell CR, Matthews QL, Sims B. Cocaine-Specific Effects on Exosome Biogenesis in Microglial Cells. Neurochemical research. Apr 2021;46(4):1006-1018. doi:10.1007/s11064-021-03231-2
- Jones LB, Kumar S, Curry AJ, et al. Alcohol Exposure Impacts the Composition of HeLa-Derived Extracellular Vesicles. Biomedicines. Sep 30 2019;7(4) doi:10.3390/biomedicines7040078
- Cho JH, Hong YD, Kim D, et al. Confirmation of plant-derived exosomes as bioactive substances for skin application through comparative analysis of keratinocyte transcriptome. Applied Biological Chemistry. 2022/01/31 2022;65(1):8. doi:10.1186/s13765-022-00676-z
- Ganesh S, Lam TT, Garcia-Milian R, et al. Peripheral signature of altered synaptic integrity in young onset cannabis use disorder: A proteomic study of circulating extracellular vesicles. The world journal of biological psychiatry : the official journal of the World Federation of Societies of Biological Psychiatry. Sep-Oct 2023;24(7):603-613. doi:10.1080/15622975.2023.2197039
- Tajik T, Baghaei K, Moghadam VE, Farrokhi N, Salami SA. Extracellular vesicles of cannabis with high CBD content induce anticancer signaling in human hepatocellular carcinoma. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie. Aug 2022;152:113209. doi:10.1016/j.biopha.2022.113209
- Kalarikkal SP, Sundaram GM. Edible plant-derived exosomal microRNAs: Exploiting a cross-kingdom regulatory mechanism for targeting SARS-CoV-2. Toxicology and applied pharmacology. Mar 1 2021;414:115425. doi:10.1016/j.taap.2021.115425
- Pomatto MAC, Gai C, Negro F, et al. Oral Delivery of mRNA Vaccine by Plant-Derived Extracellular Vesicle Carriers. Cells. Jul 11 2023;12(14) doi:10.3390/cells12141826
- Pomatto MAC, Gai C, Negro F, et al. Plant-Derived Extracellular Vesicles as a Delivery Platform for RNA-Based Vaccine: Feasibility Study of an Oral and Intranasal SARS-CoV-2 Vaccine. Pharmaceutics. Mar 17 2023;15(3) doi:10.3390/pharmaceutics15030974
- Grammenos G, Kouneli V, Mavroeidis A, et al. Beneficial Insects for Biological Pest Control in Greenhouse Cannabis Production. vol 78. 2021:85-93.
- McPartland jM. A review of Cannabis diseases. Journal of the International Hemp Association 1996;3(1):19-23.
- McPartland JM. Hemp diseases and pests: management and biological control: an advanced treatise.
- Schreiner M, ; Cranshaw, W, . A Survey of the Arthropod Fauna Associated with Hemp (Cannabis sativa L.) Grown in Eastern Colorado. Research. Journal of the Kansas Entomological Society 2020;93(2):113-131.
- Hirsch KLaKAK. Two-spotted spider mites in hemp. Alabama Cooperative Extension System. ANR-2850.

