Chronic Metabolic Syndrome and Cardiac Rhythm in Rats
Chronic Metabolic Syndrome develops a frailty of cardiac rhythm similar to aging in rats
Alondra Albarado-Ibañez¹, Julian Torres-Jacome², Marcia Hiriart²
- Laboratorio de Fisiopatología Cardiovascular. Centro de Investigación en Fisicoquímica de Materiales, Instituto de Ciencias, Benemérita Universidad Autónoma de Puebla
- Neuroscience Division, Cognitive Neuroscience Department, Instituto de Fisiología Celular, Universidad Nacional Autónoma de México UNAM.
OPEN ACCESS
PUBLISHED: 30 October 2024
CITATION: Ibañez, A.A., et al., 2024. Chronic Metabolic Syndrome develops a frailty of cardiac rhythm similar to aging in rats. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5939
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI
https://doi.org/10.18103/mra.v12i10.5939
ISSN 2375-1924
Abstract
Heart rhythm decreases and alterations depend on aging and correlate with lifestyle and metabolic alterations like obesity, dyslipidemia, and chronic inflammation; these alterations may also produce ventricular hypertrophy and atrial fibrillation, increasing lethal arrhythmias. The high sucrose diet in young adult Wistar rats produces metabolic syndrome from the eighth week that continues twenty-six weeks later. In this work, we analyzed the changes presented by the heart during six months of metabolic syndrome.
After this period, the rats were anesthetized, and the electrocardiogram was recorded. We observed that metabolic syndrome produced bradycardia and arrhythmias. The electrocardiogram showed an 18% decrease in the heart rate in rats with metabolic syndrome and a decreased ability to regulate heart rate variability using the p-p interval Poincare graph. The electrical activity recorded in the sinus node showed alteration in the morphology and propagation of the action potential, therefore, a dysfunction in the pacemaker and supraventricular arrhythmias. This data correlated with the increase in the collagen and lipid area in the pacemaker that produced unexcitable segments, which induced lethal arrhythmias by alterations in electrical activity and premature aging and frailty, like those described in humans. We proposed that the changes in electrical and morphological alterations in the sinus node that are associated with metabolic syndrome are the cause of cardiovascular frailty and premature aging of the heart.
Keywords
- Chronic Metabolic Syndrome
- Cardiac Rhythm
- Aging
- Rats
Introduction
The urgency of further research on the relationship between aging, metabolism, and cardiovascular health is evident. Aging is a significant factor in dysfunction and slow deterioration in cardiac rhythm related to cardiac hypertrophy, atrial fibrillation, and heart failure(1). A growing body of evidence has linked alterations in metabolism with cardiac aging and dysfunction of the cardiovascular system. Many alterations may develop in response to lifestyle, like cardiac inflammatory processes, dyslipidemia, insulin resistance, and high fasting glucose(2).
Metabolic syndrome (MetS) is a cluster of signs that significantly elevate the risk of developing diabetes mellitus, cardiovascular disease, and certain types of cancer. The diagnosis of MetS requires the presence of at least three signs, including central obesity, dyslipidemia, insulin resistance, and hypertension. Its incidence, caused by a sedentary lifestyle and poor diet(3).
The development of MetS is multifactorial, influenced by genetic predisposition and environmental factors, such as a nutrient-rich diet and low physical activity(4). If patients with MetS have dyslipidemias and an inflammatory process for ten years, they develop an atherosclerotic cardiovascular disease (ASCVD) risk score of more than 7.5 %(5). On the other hand, human aging is associated with increased lipid accumulation in non-adipose tissues, insulin resistance, hardening extracellular matrix, and chronic inflammatory process(2,6).
The heart rhythm (HR) correlates to the pacemaker electricity in the sinus node; the current spreads through the atrial upper chambers, causing them to contract and squeeze blood into the lower chambers ventricular(7). In progressive aging, HR that originates in the sinus node(8) declines, causing an increased incidence of arrhythmias (sudden death)(9).
Heart rate variability (HRV) is a tool for measuring cardiac function, and changes in this parameter predict cardiovascular disease mortality and/or morbidity(10). Hypertension, obesity, family history, and work stress can alter the HRV(11,12). As mentioned earlier, the decline in intrinsic heart rate (HR) is age-dependent due to a depression in individual pacemaker myocyte excitability(13).
The current research proposes MetS-induced electrical-mechanical alterations in both chamber types. Moreover, changes in heart rhythm by modifications in the extracellular matrix in the sinus node. This study proposes that MetS produces electrical and morphological alterations in the sinus node associated with changes in the extracellular matrix, inducing cardiovascular frailty and aging.
Methods
Metabolic Syndrome Chronic Model
Young adult Wistar rat males (250–280 g) were kept in a 12 h light/dark cycle, fed with standard rat chow composed of laboratory rodent diet 501 with 28.5% protein, 13% fat, and 58% carbohydrate for fifty-two weeks. Tap water was provided to all the control group or twenty-six weeks. After the treatment, we anesthetized the rats with an intraperitoneal sodium phenobarbital injection (40 mg/kg). We measured changes in the rats’ metabolism, body weight (kg), abdominal circumference (cm), and body length (cm). Finally, the animals were euthanized by cervical dislocation, and samples of the heart, blood, peripancreatic, and epididymis fat were obtained(14). All methods used in this study were previously approved by the Animal Care Committee of the Instituto de Fisiología Celular, Universidad Nacional Autónoma de México (CICUAL MHU-188-22). We took care to ensure the well-being of the animals, performing all procedures according to the “International Guiding Principles for Biomedical Research Involving Animals” Council for International Organization of Medical Science 2010.
Heart Rhythm
The heart rhythm was measured with an electrocardiogram (EKG). The rats were anesthetized with ketamine/xylazine (0.03/0.01 mg Kg–1, IP), and bipolar EKGs were recorded using subcutaneous
High sugar diet induced the heart rate variability in MetS (n=15, grey; black control, n=15).
needle electrodes and following the EKG lead I configuration. The EKG signal was 700X amplified and acquired at 10 KHz for 10 minutes. All rats were continuously monitored to guarantee the right ventilation and temperature. The data was stored in a personal computer and analyzed using Clamp Fit (Molecular Devices).
Analysis of Pacemaker Variability
The variation in heart rate was evaluated with nonlinear methods using the Poincare plots, using the R-R intervals frequency domains. Five-minute EKGs were analyzed SD1 and SD2 as standard deviation(15) and the Poincare index as a measure of heart rate variability SD1/SD2(16). The SD1, SD2, and SD1/SD2 values of control and MetS rats were determined, and the T-student with a significance of 0.05 was used to report significant differences.
Pacemaker Rhythm
The heart was taken off in a Langendorff, the SA node (pacemakers) was dissected, and the action potentials (AP) of the pacemaker were recorded using the conventional microelectrode conventional technique(17). The electrical activity obtained from the SA node was characterized by maximal amplitude, maximal depolarization rate, and AP duration at 10, 20, 30, 60, and 90% of repolarization. Also, the AP-AP intervals were measured using the variability SA node, and SD1, SD2, and index SD1/SD2 were analyzed. All these parameters were compared between control and MetS rats for analysis using the variance with a confidence interval of 0.05 to determine if they were different.
The Extracellular Matrix of the Sinoatrial Node
At the end of the twenty-sixth week of treatment, animals were euthanized. The SA node was dissected, and the electrical activity was recorded. Then, it was embedded in Tissue-Tek and frozen, and the node was cut coronally with 5 μm thick slices stained with Van-Gieson and red Oil. The staining area was measured and observed for aging, with increased collagen and adipocyte area and frailty with collagen associated with changes in electrical activity in rats control and MetS(17).
Results
High Sucrose Diet Induced the Development of Chronic Metabolic Syndrome
The animal model with a high sucrose (HSD) diet presented MetS from week ten(14). The rats after twenty-eight weeks with HSD showed chronic MetS with central obesity 30% higher (616 ± 23 g vs. controls 475 ± 12 g), a body-mass index of 37 compared to 28 in controls, and a HOMA-IR of 0.8 vs. 0.5 of controls.
MetS rats had fasting hyperglycemia compared to controls (15.6 mM vs. 8.7 mM), and also dyslipidemia 83.5 mg/dl vs. 47 mg/dl in controls, see Figure 1. The fat tissue increased in MetS rats; the weight of epididymal fat was 8.9 ± 1 g vs. 3.2 ± 0.3 g in the control group, and for peripancreatic fat 2 ± 0.39 g vs 0.4 ± 0.07 g in controls.
The Chronic Metabolic Syndrome Induced Frailty in Heart Rate
The mean heart rate decreased in Mets rats to 188 beats/minute (bpm), compared to controls 237 bpm. However, the p/p intervals showed that the heart rhythm increased by 22% (282 ± 39 bpm) in MetS rats and 14% (253 ± 50 bpm) in control rats (Figure 2). Also, the MetS hearts produced supraventricular arrhythmias as the p-waveform raised, and there is an increase in P-R interval and ventricular arrhythmias, as high and reverse R-waveform, high T-waveform in the EKG, see Figure 2B. Additionally, as shown in Figure 2A, the Poincare plot of p-p intervals of EKG exhibited an imbalance in the modulation of the sympathetic system in the MetS group was SD1 2.8 and the parasympathetic system was SD2 6.1, while for controls were 1.8 and 3.6, respectively.and the rate variability was SD1 = 70, SD2 = 144 in control rats and SD1 = 172, SD2 = 376 in rats with MetS like that observed in aging (see Figure 2B).
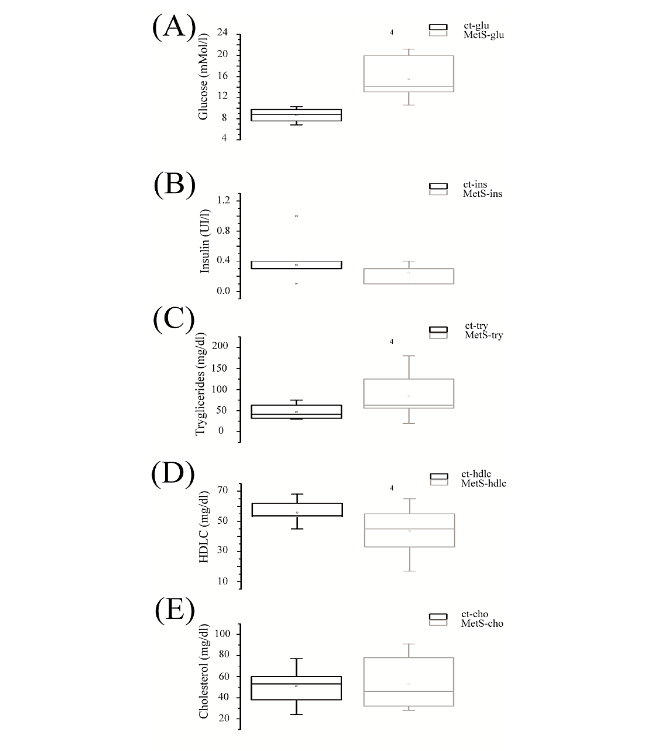
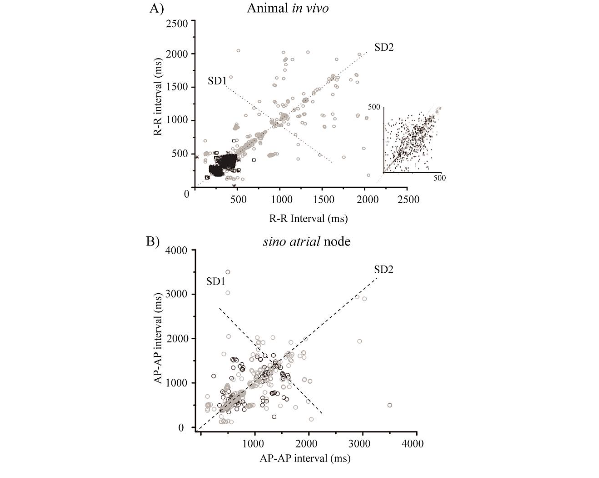
Figure 2. Metabolic syndrome changes the variability of the heart rhythm like aging.
A) Poincare plot of ECG RR, evidenced a decreased frequency and increased heart rate variability in MetS (n = 15, grey; black control, n = 15); the insert is the same plot the MetS present beeline in SD2; the insert showed the same behavior that RR Poincare plot in this interval time.
B) Poincare plot of firing SA node AP-AP, decrease rate and increase SD1 and SD2 twins time (n = 15, grey; black control, n = 15).
THE CHRONIC METABOLIC SYNDROME PRODUCED “PREMATURE AGING” IN ELECTRICAL ACTIVITY IN PACEMAKER
As the MetS produced sinus bradycardia, we studied the electrical activity of the SA node. The intrinsic heart rate was recorded and measured in the isolated preparation of the sinus node (the four AP were recorded in all tissue). As shown in Figure 3, the spontaneous cycle length of ASN was 350 ± 120 ms (n = 15) in controls and 571 ± 60 ms (n = 15) in MetS animals.
MetS induced the frailty in the pacemaker(9); function in the SN mapping was recorded from the endocardial surface, the four anticipated electrical activities: AP type I in blue, AP type II in blue, AP type III in green, and type IV in yellow. The MetS showed new APs; the first “train firing” magenta record, this pattern was observed in all nodal tissue sites. The second AP not firing “quiescent,” yellow record was characterized along the border of the crista terminalis and in the superior cava vein, see Figure 3.
In the sinus node, AP types I and III showed an increase of 11% in amplitude and 310% in the diastolic depolarization (DD) slope (see Figure 3). We also measured AP type II and IV duration at 60% (APD60%) and 90% (APD90%). The data from both measures illustrated a reduction of 25% and 13%, respectively (Figure 3).
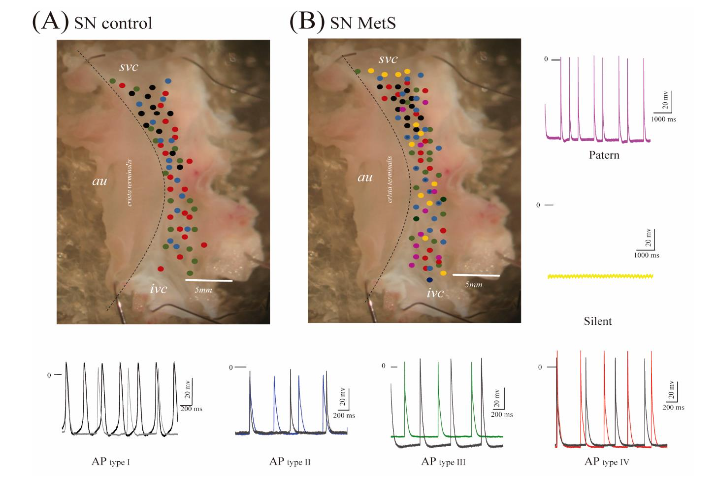 Figure 3. The electrical activity sinus.
Figure 3. The electrical activity sinus.
(A) Sinus Node control mapping.
(B) Sinus node MetS mapping with two new AP. The central activity or Action Potentials AP type I shows changes in 60 and 90% of APD by obesity and insulin resistance, which is black recorded. The AP type II (blue) shows changes in the slope of phase 4, 10, and 30% APD in MetS. AP type III green shows changes in the slope of phase 4 and amplitude, and AP type IV red shows a rise in amplitude. In addition, the pacemaker with MetS shows two other electrical activities: pattern or magenta and yellow or silent.
THE MetS PRODUCES FIBROSIS IN THE SINUS NODE
The sinus node of rats with MetS showed an increased collagen in the extracellular matrix. The sinus node stained with Van Gieson exhibited a stained area of 0.145 μm² in control tissue and 0.251 μm² in MetS (Figure 4). Furthermore, the extracellular matrix was remodeled with adipocytes in sites where it should be myocytes. The oil-red area staining in sinus node control was 0.05 μm², and MetS was 0.31 μm² (see Figure 4).
Discussion
During human aging, a trilogy of heart malfunction manifests as left ventricular hypertrophy, heart failure, and atrial fibrillation(5); this trilogy dramatically increases in MetS. Heart failure and left ventricular hypertrophy are associated with high blood pressure and obesity(18). However, atrial fibrillation is present in 4% of the healthy volunteers over 60 years of age, excluding clinical coronary artery disease, without identifiable cause. Our observations of MetS rats, which exhibited higher weight, hyperinsulinemia, dyslipidemia, insulin resistance, and alteration in fasting glucose despite the same average growth as controls, have significant implications for our understanding of MetS. We found that MetS reduced the heart rhythm, serving as a sign of premature aging and frailty.
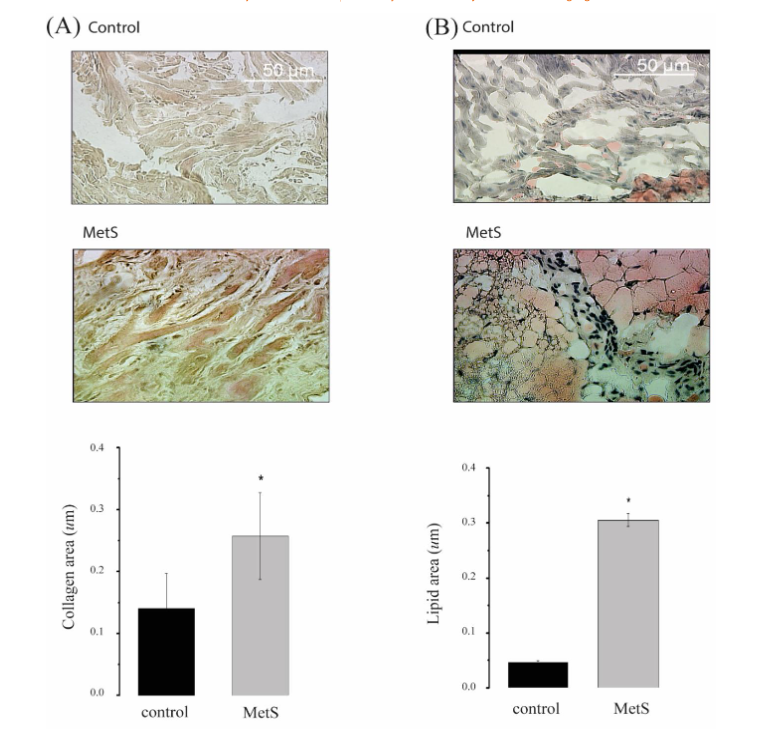
Figure 4. The MetS produces fibrosis and frailty in the sinus node.
(A) The high collagen expression in sinus-node tissue in MetS rats with insulin resistance.
The obesity in the model produced (B) high lipid ectopic in the sinus node of rats with MetS. Mean ± SE * P ≤ 0.05.
Obesity is associated with diastolic dysfunction for remodeling and induces hypertrophy in the ventricular wall(8,19,20). In men’s middle age, the heart rate decreases from 16 % to 35 % on average(8). Similarly, in the rats at 40 weeks ago (around 42 years old humans), the heart rate decreased by 8% compared with younger controls 18-20 weeks ago (20 years old humans), but in animals with MetS decreased by 23%(21). Our animals with MetS did not show ventricular hypertrophy. The weight of the heart in control was 1.98 mg, MetS was 2.1 mg, and the circumference was also similar. These results suggest that MetS caused alterations in electrical activity in the sinus heart and that obesity is an etiology for frailty in rats with MetS, like in humans(22).
Chronic MetS reduced the heart rhythm associated with changes in SD1 and SD2, which indicates that the influence of the autonomic nervous system is also altered, as shown during aging. The HRV in the MetS rats increased in SD1 and SD2, and the RR intervals were regular in SD2. This last trend decreases the interval range and the ability to regulate heart rate, which can develop cardiac supra and ventricular arrhythmias. As we hypothesized, the results indicate that MetS induced alterations in electrical activity in the pacemaker of the heart. The arrhythmias were started in the pacemaker because the frequency and Poincare plot of the p-p intervals showed similar behavior in both isolated ASN and intact animal recordings. The new morphologies and changes in the sinus action potential (Figure 2) suggest a block in the electrical propagation generated pauses in the pacemaker rhythm, sinus bradycardia, and general arrhythmias(8).
The density of adipocytes in the sinoatrial node increased in MetS rats (Figure 4) suggesting a potential increase in resistance to electrical coupling in this tissue, which could lead to an electrical uncoupling between the node cells. Also, by increasing this resistance the tissue can no longer work as a syncytium and multiple pacemakers are generated, creating independent systems of oscillators that produce complex firing patterns known in neurons as phase-locking(23) and arrhythmias in MetS rats. This finding opens new avenues for future research to understand the role of adipocytes in heart function and MetS.
Conclusion
The high sucrose diet for twenty-six weeks induces fibrosis in the cellular matrix of the sino auricular node due to inflammation. These phenomena altered the cardiac rhythm, increased oscillators in the SA node produced by adipocytes, such as blockades of electrical central activity or AP type I, and the periphery of the electrical activity showed characteristics like AP type I. The electrical changes in the pacemaker produced bradycardia in rats with metabolic syndrome, heart rate variability, and frailty like aging. These implications underscore the importance of further research in this area.
Conflict of Interest:
The authors report no conflicts of interest in this work.
Acknowledgments:
We want to thank Carmen Sánchez-Soto, for her technician activity in MH lab; Sandra Daniela Rodriguez Montaño from the Histology Unit, Microscopy Unit personnel, Ana Escalante, and Francisco Pérez from the Compute Unit of the Instituto de Fisiología Celular, UNAM.
Grants:
This work was supported by DGAPA-PAPIIT IN215611 and IN228623 to MH; PROFOCIE 2016 to TJJ; and VIEP 00454 to TJJ; DGAPA-PAPIIT all from Mexico.
References
1. Christensen A, Liu J, Pike CJ. Aging Reduces Estradiol Protection Against Neural but Not Metabolic Effects of Obesity in Female 3xTg-AD Mice. Front Aging Neurosci. 2020;12:113.
2. Ajoolabady A, Pratico D, Vinciguerra M, Lip GYH, Franceschi C, Ren J. Inflammaging: mechanisms and role in the cardiac and vasculature. Trends Endocrinol Metab. 2023;34(6):373-87.
3. Islam MS. Islets of Langerhans. 2nd edition. ed. New York: Springer; 2014. pages cm p.
4. Lakatta EG. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part III: cellular and molecular clues to heart and arterial aging. Circulation. 2003;107(3):490-7.
5. Lakatta EG, Levy D. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part I: aging arteries: a “set up” for vascular disease. Circulation. 2003;107(1):139-46.
6. Wang M, Kim SH, Monticone RE, Lakatta EG. Matrix metalloproteinases promote arterial remodeling in aging, hypertension, and atherosclerosis. Hypertension. 2015;65(4):698-703.
7. Gencer B, Butler J, Bauer DC, Auer R, Kalogeropoulos A, Marques-Vidal P, et al. Association of electrocardiogram abnormalities and incident heart failure events. Am Heart J. 2014;167(6):869-75 e3.
8. Jones SA. Ageing to arrhythmias: conundrums of connections in the ageing heart. J Pharm Pharmacol. 2006;58(12):1571-6.
9. Arroyo-Carmona RE, Mitre-Velasco Y, Martinez-Laguna Y, Torres-Jacome J, Albarado-Ibanez A. A maternal diet high in carbohydrates causes bradyarrhythmias and changes in heart rate variability in the offspring sex-dependent in mice. Lab Anim Res. 2024;40(1):34.
10. D’Souza A, Trussell T, Morris GM, Dobrzynski H, Boyett MR. Supraventricular Arrhythmias in Athletes: Basic Mechanisms and New Directions. Physiology (Bethesda). 2019;34(5):314-26.
11. Singh D, Vinod K, Saxena SC, Deepak KK.
Effects of RR segment duration on HRV spectrum estimation. Physiol Meas. 2004;25(3):721-35.
12. Sharma RK, Deepak KK, Bijlani RL, Rao PS. Short-term physical training alters cardiovascular autonomic response amplitude and latencies. Indian J Physiol Pharmacol. 2004;48(2):165-73.
13. Larson ED, St Clair JR, Sumner WA, Bannister RA, Proenza C. Depressed pacemaker activity of sinoatrial node myocytes contributes to the age-dependent decline in maximum heart rate. Proc Natl Acad Sci U S A. 2013;110(44):18011-6.
14. Albarado-Ibanez A, Avelino-Cruz JE, Velasco M, Torres-Jacome J, Hiriart M. Metabolic syndrome remodels electrical activity of the sinoatrial node and produces arrhythmias in rats. PLoS One. 2013;8(11):e76534.
15. Boyett MR, Honjo H, Yamamoto M, Nikmaram MR, Niwa R, Kodama I. Regional differences in effects of 4-aminopyridine within the sinoatrial node. Am J Physiol. 1998;275(4):H1158-68.
16. Arroyo-Carmona RE, Lopez-Serrano AL, Albarado-Ibanez A, Mendoza-Lucero FM, Medel-Cajica D, Lopez-Mayorga RM, et al. Heart Rate Variability as Early Biomarker for the Evaluation of Diabetes Mellitus Progress. J Diabetes Res. 2016; 2016:8483537.
17. Guerra F, Mancinelli L, Angelini L, Fortunati M, Rappelli A, Dessi-Fulgheri P, et al. The association of left ventricular hypertrophy with metabolic syndrome is dependent on body mass index in hypertensive overweight or obese patients. PLoS One. 2011;6(1):e16630.
18. Lauer MS, Larson MG, Evans JC, Levy D. Association of left ventricular dilatation and hypertrophy with chronotropic incompetence in the Framingham Heart Study. Am Heart J. 1999; 137(5):903-9.
19. Hashem MS, Kalashyan H, Choy J, Chiew SK, Shawki AH, Dawood AH, et al. Left ventricular relative wall thickness versus left ventricular mass index in non-cardioembolic stroke patients. Medicine (Baltimore). 2015;94(20):e872.
20. Movahed MR, Bates S, Strootman D, Sattur S. Obesity in adolescence is associated with left ventricular hypertrophy and hypertension. Echocardiography. 2011;28(2):150-3.
21. Sengupta P. The Laboratory Rat: Relating Its Age With Human’s. Int J Prev Med. 2013;4(6):62 4-30.
22. Bisset ES, Howlett SE. The biology of frailty in humans and animals: Understanding frailty and promoting translation. Aging Med (Milton). 2019;2 (1):27-34.
23. Budelli R, Torres J, Catsigeras E, Enrich H. Two-neurons network. I. Integrate and fire pacemaker models. Biol Cybern. 1991;66(2):95-101.

