CIRS Protocol: Evidence-Based Treatment for Biotoxins
The CIRS Protocol: A Sequential, Evidence-Based Treatment for Biotoxin-Associated Chronic Inflammatory Response Syndrome
Eric Dorninger, ND, LAc1
- Roots and Branches Integrative Healthcare, Louisville, Colorado
OPEN ACCESS
PUBLISHED: 31 August 2025
CITATION Dorninger, E., 2025. The CIRS Protocol: A Sequential, Evidence-Based Treatment for Biotoxin-Associated Chronic Inflammatory Response Syndrome. Medical Research Archives, [online] 13(8). https://doi.org/10.18103/mra.v13i8.6770
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i8.6770
ISSN 2375-1924
ABSTRACT
Chronic Inflammatory Response Syndrome (CIRS) is a complex, multisystem illness driven by sustained innate immune activation following exposure to biotoxins such as mold, mycotoxins, Actinobacteria, and bacterial endotoxins in water-damaged buildings (WDB), as well as tick-borne and marine toxins. The CIRS Protocol is a sequential 12-step therapeutic framework designed to diagnose and resolve CIRS through a combination of environmental, pharmacologic, and biochemical interventions.
This paper outlines the clinical application of the protocol, beginning with Step 1 complete removal from biotoxin exposure which requires advanced environmental assessment using qPCR, LAL assays, and other validated building clearance criteria. Step 2 involves removal of internal biotoxin reservoirs using bile acid sequestrants (cholestyramine/Welchol), following immune system priming with high-dose EPA/DHA fish oil. Subsequent steps address eradication of Multiple Antibiotic Resistant Coagulase Negative Staphylococci (MARCoNS), correction of gliadin antibodies, and restoration of depleted androgens particularly dehydroepiandrosterone (DHEA) often suppressed in CIRS due to Hypothalamic-Pituitary-Adrenal (HPA) axis dysregulation.
The protocol progresses to targeted correction of disrupted biomarkers including Antidiuretic Hormone (ADH)/osmolality, Matrix Metalloproteinase-9 (MMP-9), Vascular Endothelial Growth Factor (VEGF), Complement 3a (C3a), Complement 4a (C4a), and Transforming Growth Factor beta-1 (TGFβ-1), each linked to specific symptoms and tissue-level dysfunction. Evidence-based use of adjunctive agents such as fish oil, losartan, and vasoactive intestinal peptide (VIP) is discussed in depth, with rationale grounded in peer-reviewed clinical and mechanistic data.
When implemented in the prescribed sequence with precision, the CIRS Protocol results in measurable biomarker normalization and clinical resolution in most compliant patients, restoring neuroendocrine-immune homeostasis and function.
Despite evidence from peer-reviewed studies and human clinical trials, the CIRS Protocol remains outside of mainstream medical practice, while unvalidated treatments proliferate leading to poor outcomes and resource exhaustion for patients. This review aims to provide a clear, detailed, and referenced guide to the CIRS Protocol to support provider education and address the growing need for competent care in biotoxin-related illness.
Keywords
CIRS, biotoxins, treatment protocol, chronic inflammatory response syndrome, environmental assessment, immune modulation
Introduction
This review of the CIRS Protocol is intended to explain the details and standards of each step and support a cohesive, consolidated, and referenced learning experience for healthcare providers. In turn, it supports the training of the next generation of providers needed to address the crisis of this illness.
The CIRS Protocol is a 12-step sequential treatment developed by Dr. Ritchie Shoemaker for resolving Chronic Inflammatory Response Syndrome. This treatment has been shown to heal most patients afflicted with CIRS when applied rigorously and executed authentically. The most complex step by remove Patient from Biotoxin Environmental Professional (IEP) to diagnose the sources of biotoxins in the indoor environment and produce a detailed report with instructions for remediation professionals.
The goal of this publication, beginning with Part A and Part B, is to bring certainty to diagnosis and treatment to patients sickened by exposure to the interior environment of water-damaged buildings. A quick review of alternative methods of diagnosis and treatment of these patients shows a diversity of opinions rarely supported by published data. A vocal group of practitioners advocate using antifungal drugs, an idea condemned by the CDC, even though CIRS is not caused by fungal infections. Control studies supporting measurement of mycotoxins in urine show no difference between cases and controls, even if such control groups were used.
Our methods are transparent: we reviewed the published literature in Part A (see McMahon) to support our contention that diagnosis is well-the treatment approach used widely since 2006 (see SAIIE) with ongoing publication of results of transcriptomics that show salutary effects on gene activity that follow use of the CIRS Protocol. We present detailed procedures for each of the sequential steps in the protocol with discussion of possible problems that may require additional steps to follow. We base our recommendations on published, peer-reviewed papers.
The 12-step protocol is presented as a therapeutic pyramid for three reasons: 1. The most fundamental steps are visually represented as the largest. 2. When the steps especially first three are applied correctly, some downstream abnormalities self-correct (e.g., inflammatory cytokines and neuroregulatory peptides can stabilize). 3. If steps are skipped or performed out of sequence, symptom resolution and overall clinical success are significantly reduced.
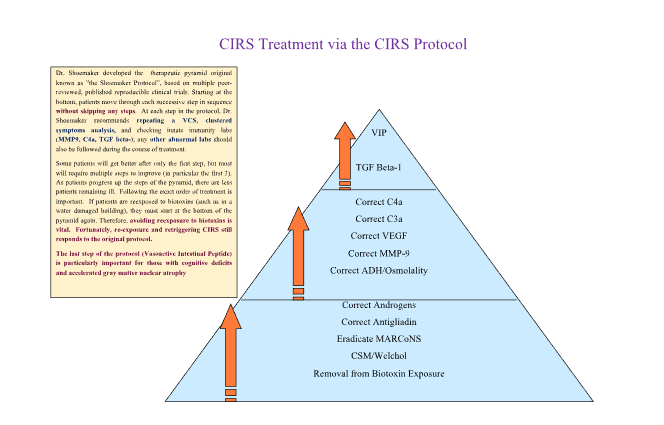
Step 1: Remove Patient from Biotoxin Exposure
Biotoxins are ionophores or amphipaths/amphiphiles with hydrophilic and hydrophobic properties. Biotoxins can move easily from cell to cell due to their size (as small as 1.4 angstroms) and their ability to share electrons. Biotoxins store in adipose and nerve tissue, where they interact with cellular receptors including Toll-like receptor (e.g., TLR2, TLR4), mannose, dectin and C-type lectin receptors, triggering innate immune activation.
When our immune response genes, known as Human Leukocyte Antigen (HLA), cannot clear a biotoxin via a coordinated antigen/antibody response, inflammatory, innate immune responses multiple body systems creating multiple body symptoms. Chronic exposure to antigenic material and an ineffective antibody response leads to unchecked, innate immune responses that are the driver of CIRS; molecular hypometabolism (MHM), downstream neuroendocrine dysregulation/failure and eventual cerebral multinuclear atrophy are the consequences of CIRS.
Peer-reviewed published sources of biotoxin illness include seafood toxins (e.g., ciguatera), cyanobacteria (algal blooms), Lyme tick and co-infection biotoxins (BbTox / BB0755), recluse spider venom, fungi/molds found in water-damaged buildings (WDB), mycotoxin and bacteria (including Actinobacteria, e.g. Corynebacterium tuberculostearicum – CT and Cutibacterium acnes (formerly Propionibacterium acnes – PA) and inhaled bacterial endotoxin (lipopolysaccharides (LPS) from the outer wall of gram-negative bacteria from fecal material and/or sewer gas). For the purposes of this paper, we will be focusing on the biotoxin notorious in the water-damaged and/or high humidity damp buildings.
Screening a patient for CIRS as an underlying cause of their symptom picture requires a clinician to execute three basic evaluations to augment CIRS in their differential diagnosis: 1) Interviewing for biotoxin exposure history past or present (see Table 1), 2) Cluster analysis questionnaire (administered by the provider or sincerely trained staff), 3) Visual Contrast Sensitivity test (VCS).
The BASE (Building Assessment Survey Evaluation) study conducted by the EPA estimates that 85% of the buildings experienced water damage in the past, and 45% were experiencing current leaks. Building data was collected from 100 randomly selected buildings in 37 cities in 25 states. Ownership of the BASE buildings was primarily split between private and commercial (n=41) and government (n=44), with the remaining 15 buildings classified as academic (schools). Most of the BASE study buildings were classified as being in urban surroundings (73%), whereas twenty-three percent were in suburban surroundings and 4% were in rural surroundings.
When water damage exposure is revealed during the clinical interview and initial screenings are failed (cluster analysis and visual contrast sensitivity (VCS)), confirmatory laboratory testing for CIRS follows. This marks the beginning of the process to identify potential building-related biotoxin sources.
Ideally, the evaluation of a home or other occupied structure should be conducted by an Indoor Environmental Professional (IEP) trained in biotoxin-related assessments, including the detection of WDB molds, mycotoxins, fungal cell wall fragments (e.g., beta-glucans), Actinobacteria, and endotoxins. Unfortunately, most IEPs lack specific training in biotoxin exposure and often rely on limited or inadequate objective assessment methods, making it difficult to successfully complete the building essential biotoxins in a building envelope to ensure the statistical likelihood a CIRS patient will heal in a building: 1) WDB molds (qPCR testing known as ERMI/HERTSMI-2), Actinobacteria (NGS) 3) Endotoxin (kinetic LAL assay), 4) beta glucans (direct measuring assay for (1→3)-ß-D-glucans).
Sampling is done predominantly through analysis of dust collection and reported values are used to direct further investigation of the building envelope should have parenthesis.
Step 2: Cholestyramine / Welchol
Once exposure to a water-damaged building or biotoxin source has been eliminated, the next critical step in treating Chronic Inflammatory Response Syndrome is the removal of biotoxins from the body. This is most effectively accomplished using cholestyramine (CSM) and/or Welchol, a non-absorbable anion exchange resin that is FDA-approved for lowering cholesterol but used off-label for biotoxin binding. Cholestyramine, Welchol, colesevelam, and Questran are all drug names for the different effective bile acid sequestrants for treating CIRS.
CSM remains within the gastrointestinal tract, where it binds cholesterol, bile acids, and bile salts. Once bound, these compounds, and any attached biotoxins, are prevented from re-entering the enterohepatic circulation (via the portal vein) and are excreted in the stool.
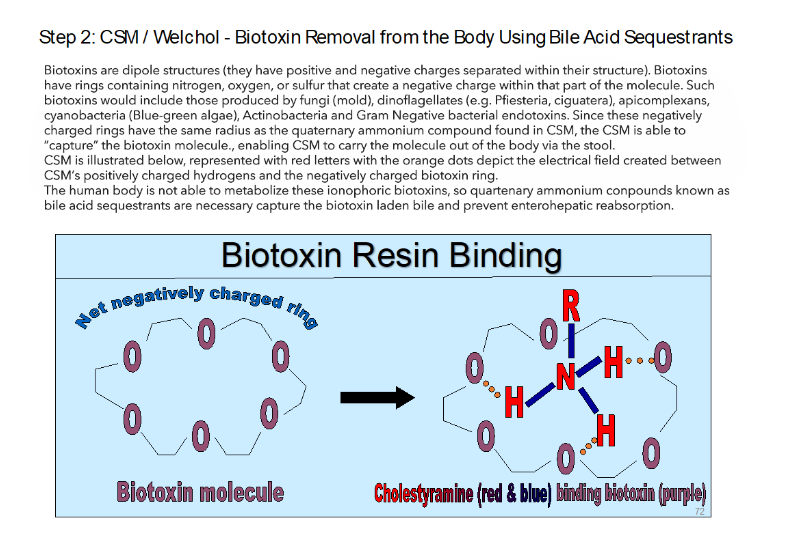
CSM is a quaternary ammonium compound in the prescription class known as bile acid sequestrants, with nitrogen-containing side chains that carry a net positive charge. This positive charge enables CSM to attract and bind negatively charged molecules, including biotoxins.
Many biotoxins are dipolar structures, possessing both positively and negatively charged regions. These molecules often contain aromatic rings with nitrogen, oxygen, or sulfur atoms that carry a negative charge. Biotoxins in this category include those produced by molds (fungi), dinoflagellates (e.g., Pfiesteria, ciguatera), apicomplexans, cyanobacteria (blue-green algae), Actinobacteria, and gram-negative bacteria (endotoxins). Because the negatively charged rings in these molecules are of equivalent size to the quaternary ammonium and hold onto them through electrostatic attraction, preventing their reabsorption.
In CIRS, the body lacks the capacity to adequately metabolize and eliminate these biotoxins. Therefore, physical removal, via binding and excretion, is currently the only known and published effective clinical approach. Current natural binders in the zeitgeist of CIRS and mold illness do not share the same net charge and organic chemistry as quaternary ammonia and have failed to reduce CIRS inflammatory cytokines with pre/post testing (C4a, MMP-9, TGFβ-1 remaining elevated).
Biotoxins are transported to the liver, which secretes them into bile using organic anion transporters (OATP). During digestion, bile enters the small intestine through the sphincter of Oddi. Under normal conditions, biotoxins in bile are reabsorbed alongside cholesterol and bile acids (enterohepatic recirculation). However, when CSM/Welchol is present in the intestinal tract after bile release, CSM binds these biotoxins, preventing their reabsorption.
Once bound to CSM, the toxins are sequestered and removed from the body through the feces.
Regular administration of CSM, timed to follow bile secretion can support recovery in CIRS patients by downregulating innate cytokine storms via capture and removal of biotoxins.
Step 3: Eradicate MARCoNS
Multiple Antibiotic Resistant Coagulase Negative Staphylococci (MARCoNS) is a biofilm forming, toxin producing Staph bacteria (typically Staph epidermidis) that colonizes the nasopharynx of approximately 80% of confirmed adult CIRS cases. In clinical data, only 1-2% of healthy controls showed nasopharyngeal MARCoNS on Api-Staph swab (Shoemaker 2006). Api-Staph isolate technique is essential for processing a nasopharyngeal swab when investigating MARCoNS.
MARCoNS culture and sensitivity show resistance to penicillin and at least one other class of antibiotics. CoNS (Coagulase Negative Staph) is a neutral colonizing bacterium that does not require treatment. MARCoNS colonization can participate in fatigue and neurological phenomenon (peripheral neuropathy, palpitations, headache) and needs to be in the differential of unexplained neurological symptoms.
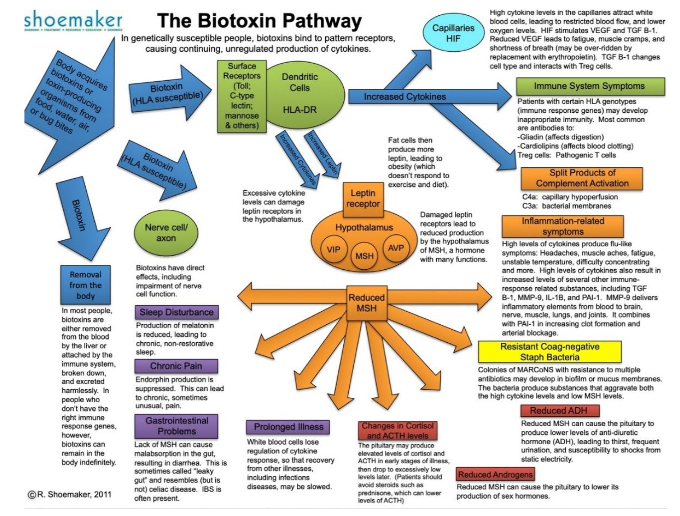
Vulnerability to MARCoNS colonization can be seen clearly on the Biotoxin Pathway first published in 2004 to show the chronological pathology of CIRS.

The pathology reveals best practices for eradication of MARCoNS:
- Remove Patient from biotoxin exposure (building clearance tests with HERTSMI-2 <10, Endotoxin <100, Beta-glucan <4450, CT and PA <10,000).
- Prime and modulate the immune system with 4200 mg of fish oil for at least 10-14 days before beginning bile acid sequestrant.
- Begin Welchol/Cholestyramine (for at least 4 weeks) to ensure internal biotoxin reservoirs are depleted before applying biofilm rupturing antimicrobial nasal sprays.
Many patients fail eradication of MARCoNS due to veering from the sequence of the protocol. Attempting to eradicate MARCoNS while still in biotoxin exposure is the major cause of failed MARCoNS treatment. If building clearance tests support healthy buildings other valid concerns for MARCoNS treatment failure include 1) partner has MARCoNS, 2) the dog has MARCoNS, and 3) MARCoNS has penetrated the dental bone. MARCoNS resides in the nares of canines and deep nasopharyngeal penetration is not necessary for culture.
The original nasal spray published for addressing MARCoNS is known as BEG spray (Bactroban (mupirocin) 0.2%, edetate disodium (EDTA) 1%, gentamicin 0.5%) and is administered at two sprays each nostril 3-4 times per day. Patients should be reminded how important it is to stay consistent with their spray. Retest MARCoNS swab is conducted at 8 weeks postnasal spray to ensure eradication.
Non-published data shows promise for stand-alone EDTA (1%), EDTA/Silver, NSB Formula 1 (biofilm rupturing/antimicrobial herbal formula), and a natural combination formula including EDTA/silver/xylitol/grape seed extract. It is rare to fail Patient clearance tests above.
Step 4: Correct Anti-Gliadin Antibodies
Early datasets revealed higher rates of autoimmune serum markers in biotoxin patients versus healthy controls. Most notable were elevated levels of pANCA (the autoantibody with specificity for ulcerative colitis), ACLA (anti-cardiolipin antibodies implicated as a possible cause of miscarriage) and anti-gliadin antibodies. If serum IgA or IgG gliadin is detected in patients, tissue transglutaminase needs to be evaluated for potential celiac disease.
If celiac disease is discovered, long term gluten removal is recommended to avoid complications of untreated celiac. If celiac negative, but gluten sensitive (i.e., gliadin IgA or IgG positive, TTG negative) removal of gluten is recommended during the remaining steps of the protocol with retesting gliadin antibodies post VIP and determining if gluten challenge (reingestion) is tolerated. Some patients feel profound improvements with gluten removal (cognitive function, mood improvement, metabolism improvement, skin improvement, GI symptom resolution) and are encouraged to continue gluten free lifestyle if symptom improvements/resolution are self-evident.
Reestablishment of immune tolerance to gliadin (if not celiac) can occur with the execution of the CIRS protocol. The normalization of autoantibodies is a regular clinical occurrence for a compliant CIRS patient as re-establishment of neuroendocrine immune modulating peptides MSH and VIP repair T-regulatory cell function and proper immune system balance.
Step 5: Correct Androgens
Dehydroepiandrosterone (DHEA): Androgens are anti-inflammatory, immune modulating, anti-aging anabolic hormones. Anabolic hormones regenerate, rejuvenate and restore tissues. It is common for anabolic hormones to be depleted during CIRS. Serum DHEA-sulfate can fall to low levels resulting in a catabolic physiological state.
DHEA (Dehydroepiandrosterone) was first discovered in 1934. In healthy physiology, DHEA is the most abundantly produced steroid. DHEA is partially metabolized into testosterone and estrogen (with minimal conversion to testosterone in males, and noticeable conversion to serum testosterone in females). Both genders experience increases in free testosterone when serum DHEA-sulfate levels reach 350-430 mcg/dL in females, 400-500 mcg/dL in males. Circulating DHEA levels (measured as DHEA-sulfate) peak around age 25 and decline progressively thereafter.
DHEA levels in 70-year-old individuals are approximately 20% of DHEA levels in young adults. In humans, serum levels of both DHEA and DHEA-sulfate change significantly across the lifespan. Unlike DHEA, cortisol levels are preserved with age and elevate with severe injury and sepsis.
DHEA goes through sulfonation via the sulfotransferase family 2A member 1 (SULT2A1) to DHEA-S (DHEA-Sulfate) partially in the adrenal cortex and during first-pass metabolism in the liver. Serum DHEA-sulfate is several folds higher than serum DHEA.
DHEA-sulfate serves as a storage depot and is readily converted by steroid sulfatase (STS) to DHEA in the endoplasmic reticulum. Only 5% of DHEA in males with normal testicular function is converted to testosterone. In premenopausal females, 40-75% of the circulating testosterone comes from DHEA-S.
DHEA modulates and dampens glucocorticoids (e.g., cortisol). DHEA provides counter-regulation of cortisol and balances cortisol’s immunosuppressant effects. When chronic inflammation or trauma drives glucocorticoids, an intra-adrenal shift toward biosynthesis of cortisol and shunt away from DHEA occurs.
This shunt in cortisol to DHEA-S ratio is associated with multiple poor outcomes in trauma victims. This phenomenon is exacerbated in patients with pre-established adrenopause and is associated with suppressed neutrophil function and increased risk of infection.
Upregulated cortisol secretion has been reported adverse effects on hippocampal neuron survival and can potentiate β-amyloid toxicity, whereas DHEA and DHEA-sulfate counteract the inflammatory effects of excess cortisol and provide neuroprotection.
Patients showed impaired hippocampal perfusion on SPECT scan relative to controls. Cortisol/DHEA ratio (C/Dr) dove-tailed hippocampal perfusion concerns with mean cortisol levels significantly elevated, and DHEA-s lowered in the patients versus controls.
The inverse relationship of increasing basal serum cortisol level and brain atrophy, worsening cognitive performance can be offset with protection of intact DHEA-sulfate. There are two critical clinical approaches to successfully loading DHEA:
- Start with a low dose and ramp up.
- Ensure clearance pathways of hormone metabolites are supported and functional to avoid the potential side effects of DHEA supplementation.
When taking a history, evaluate for past or current adverse experiences with taking any sex hormones (Oral Contraceptive Pills, bioidenticals, Depo-Provera, prior use of DHEA or testosterone, recreational anabolic use). If patient has a medical history of adverse experiences with hormones, evaluate liver clearance pathways (lipids, bilirubin, liver enzymes, homocysteine) and consider abdominal imaging. Start with 10 mg of DHEA from a reputable company and increase by 5-10 mg every week until 25-75 mg of DHEA is taken as a daily maintenance dose.
If initial serum levels of DHEA-sulfate are in the double digits, expect 35-75 mg of DHEA to be the necessary dose to reach 350-430 mcg/dL in females, 400-500 mcg/dL in males. In patient with higher initial levels (e.g., DHEA-sulfate of 200 mcg/dL), 25 mg maintenance dose will usually reach therapeutic levels. Clinical experience shows us that overshooting the therapeutic range and pulling back improves patient outcomes better than being overly concerned with overshoot, but ensure provider is rechecking levels. When supporting DHEA, expect total and free testosterone to increase in females, but only free testosterone to increase in males.
The main contraindication for application of DHEA is history of female reproductive cancers. Although a recent large scale clinical trials demonstrated less breast cancer incidence in post-menopausal women using testosterone alone or testosterone in combination with estrogen, specific studies on DHEA and reproductive cancers have not been done yet. For women with reproductive cancer history experience, we recommend 500 mg of black cohosh in the morning and midday as a potent neuroendocrine immune modulator that can address menopausal concerns while statistically reducing the likelihood of breast cancer reoccurrence.
Step 6: Correct ADH / Osmolality
Although the discovery and development of the CIRS protocol followed objective biomarkers after every single step (one variable change), establishment and reproducibility of the protocol allows the practitioner and patient to work through the first five steps of the protocol and then pause for reevaluation of biotoxin biomarkers. VCS (if initial fail) and cluster analysis can be repeated throughout the protocol as cost-effective tracking tools, but post “correction and changes” (Step 5) is a good time to reevaluate objective serum biomarkers.
Fortunately, many of the second phase of the protocol (steps 6-12) usually autocorrect due to the removal from biotoxin exposure, application of bile acid sequestrants and eradication of MARCoNS.
When symptoms of frequent/urgent urination, excessive thirst, cramps, static shocks (due to sodium excretion on the skin), bedwetting, hypovolemic headache, hypotension and/or symptoms of postural orthostatic tachycardia syndrome (POTS) persist, you will need to correct anti-diuretic hormone (ADH), a neuroendocrine immune hormone regulating fluid homeostasis and water retention. In coordination with aldosterone (enhancing sodium retention), ADH creates optimal pressure, hydration and osmolality of cells, blood volume and tissue space.
Osmotically active solute (e.g., salt water) exerts osmotic pressure causing it to draw free water to itself. Serum osmolality is the concentration of osmotically active solute. The higher the concentration of solute (osmolality), the stronger the chemical draw it has on free water. Free water can pass across a cell membrane between extracellular fluid and intracellular fluid via concentration gradients. Hence, serum osmolality reflects intracellular osmolality.
Cells in the hypothalamus of the brain contain osmoreceptors that respond to levels of serum osmolality. When there is high serum osmolality (i.e., dehydration), these osmoreceptor cells shrink and trigger the release of ADH from the posterior pituitary to signal reabsorption of free water in the kidneys. Subsequently, serum osmolality is restored to normal levels, as cells are hydrated. When serum osmolality is low (i.e., overhydration), the osmoreceptors swell and block the release of ADH; this leads to the loss of free water into the urine.
Frequent urination is the most common CIRS symptom related to ADH/osmolality issues. For an adult drinking 64 ounces of water daily, urinating 4-6 times per day is normal, 6-10 times is frequent, and 10-20 times is extremely frequent. Occasionally, CIRS cases will urinate more than 20 times per day and will highly likely require treatment with desmopressin. See treatment for Low ADH / osmolality dysregulation in Figure 4.
Step 7: Correct MMP-9
MMP-9 (Matrix Metallopeptidase 9) is one of the three original inflammatory cytokines that initially defined the chronic inflammation seen in CIRS. When elevated, MMP-9 will break down matrix barriers (including the blood-brain barrier) and allow inflammatory cytokines to penetrate multiple body systems (e.g., brain, nerves, muscles, lungs, and joints). Elevated MMP-9 participates in cardiovascular disease as well via PAI-1 binding to lipoprotein(a) and increasing the amount of oxidized LDL cholesterol delivered to the subintimal space. Elevated MMP-9 is a sum marker of global cytokine effects in CIRS and can bring the headache and fatigue. Although removal from biotoxin exposure and taking bile acid sequestrants (Step 1 and 2) often reduce MMP-9, fish oil (EPA+DHA) is often needed to normalize serum MMP-9 to healthy control levels of <332.
Reducing MMP-9 can require three additional approaches if not normalizing with earnest completion of Steps 1 and 2 of the protocol.
- Low amylose diet (reducing and removing branched chain carbohydrate amylose can aid in MMP-9 normalization).
- 4200 mg of EPA / DHA fish oil in two divided doses for ten days (fish oil has already been implemented pre-Step 2 for better tolerance of Welchol/CSM and eradication of MARCoNS).
- Resveratrol at 1000 mg BID can be used to help lower MMP-9.
A note on fish oil and arrhythmia: Although meta-analysis suggests a net positive effect of fish oil on heart health, the research is murky when it comes to fish oil and arrhythmia.
Step 8: Correct VEGF
Vascular endothelial growth factor (VEGF) is a hormone responsible for cellular perfusion of oxygen. VEGF helps oxygen leave the capillary bed and penetrate the cells so that aerobic respiration (creation of energy in the presence of adequate oxygen) can occur. When two pyruvates from a split glucose molecule enter the mitochondria to be sensed in the presence of oxygen, the net effect is ~33-36 ATP without lactic acid production.
In contrast, the patient with low VEGF secondary to CIRS will experience exercise intolerance and the mailbox. This lactic threshold can arise from inadequate perfusion of the cells (despite normal is one of many pathways in CIRS (pulmonary hypertension and molecular hypometabolism are others) that shunt effective aerobic metabolism to ineffective anaerobic metabolism participating in fatigue and exercise intolerance.
Pathological anaerobic metabolism is seen with an elevated anion gap and low VEGF, pulmonary hypotension and molecular hypometabolism needs to be added to the differential diagnosis of metabolic acidosis.
Treatment of low VEGF includes high dose of fish oil (~2.8 grams EPA + 1.4 grams DHA) and a slow and steady increase in physical activity. The one minute of mild aerobic activity (e.g., walking) daily increasing by one-minute increments daily until a 15-minute walk is achieved (15 days to get there). Then layer one-minute daily increases of calisthenics (e.g., toe touches) until a 15-minute walk followed by 15-minute calisthenics is accomplished (30 days to get there). Finally, layer in one-minute daily increase of resistance training (1-2 pound weights or pushups at capacity – knee or traditional) daily until 15-minute walks, 15-minute calisthenics and 15-minute resistance training is accomplished. If successful with this titrated approach, most CIRS patients can return to movement that inspires with minimal setbacks.
Many patients struggle with this titrated ramp up in exercise due to pining for pre-CIRS fitness days. Compassion and education are essential for supporting this slow and steady approach, so they overdo physical activity (joy, comradery, endorphins can be experienced) and crash the following days (excessive soreness, need for naps, cramping, inability to get out of bed).
Step 9: Correct Complement 3a
C3a is a split product of complement. C3a can cause anaphylaxis through immune responses which cause smooth muscle constriction, capillary hypoperfusion, increased vascular permeability, and WBC release of oxidants, leukotrienes, and enzymes. C3a can be elevated in acute Lyme disease and Lyme disease must be ruled out if a CIRS patient has high C3a.

Treatment for elevated C3a is high dose statins (40-80 mg). Statins have unique anti-inflammatory properties (the likely reason they show mild benefit for arteriosclerosis stabilization). A review of clinical trials on statins showed reduction in T cell activation, macrophage infiltration, and vascular wall inflammation.
Clinical data has shown statins ability to normalize C3a. High dose simvastatin (Zocor) at 80 mg is no longer available to use with new patients, due to increased risk of rhabdomyolysis. However, the following statins are still available for high dose dispensing at 80 mg/day (some of these require divided dosing): pravastatin (Pravachol), atorvastatin (Lipitor), fluvastatin (Lescol), rosuvastatin (Crestor), and lovastatin (Mevacor/Altoprev).
We do not recommend utilizing fat soluble statin (e.g. Atorvastatin/Lipitor) for lowering C3a as a recent study in the Journal of Nuclear Medicine showed statistically significant conversion (over double the risk) from mild cognitive impairment (MCI) to dementia in 65-year-olds with low to moderate cholesterol levels followed with brain MRI. Water-soluble statin and non-statin taking participants did not show these cognitive concerns. 20 mg of water-soluble rosuvastatin (Crestor) twice a day for lowering C3a is a good option and can be tapered and discontinued when normal C3a levels are reached.
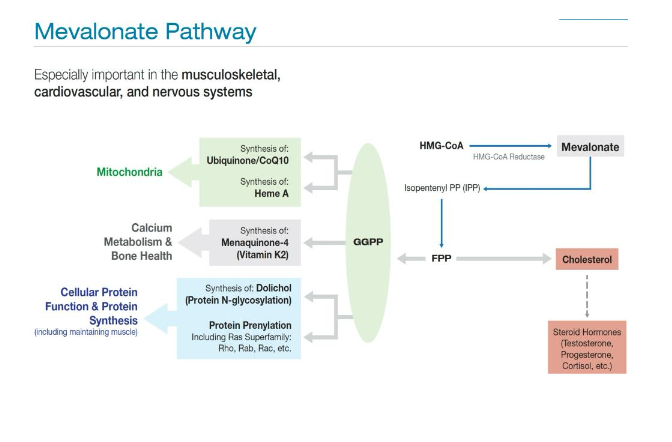
When using a statin (even transiently) one must offset the negative side effects HMG-CoA reductase inhibition and subsequent blockage of the mevalonate pathway. The original C3a reducing protocol recommended treating with CoQ10 150 mg daily for 10 days pre-statin, then introduce the statin while continuing ongoing CoQ10.
Blocking HMG-CoA reductase also inhibits the production of geranyl geraniol (GG) and can disrupt muscle, bone and mitochondrial health. This disruption is evident with the classic statin intolerance symptoms of myalgia, fatigue and for some rhabdomyolysis.
In the tradition of first do no harm, we recommend increasing the priming and ongoing dose of CoQ10 Ubiquinone to 200 mg with 150 mg trans-geranylgeraniol once a day and doubling the daily dose of CoQ10 and GG if symptoms of myalgia, fatigue, or exercise intolerance are present.
In cell line studies, GG had broader protective effects against cytotoxicity of statins than exogenous ubiquinone and of inflammasome gene expression and programmed cell death related to mitochondrial dysfunction.
In an 8 week, randomized, placebo-controlled dose escalation trial using 150-300 mg/d, GG showed a statistically significant increase in total and free testosterone levels in men with suboptimal levels (total T < 700 ng/dL).
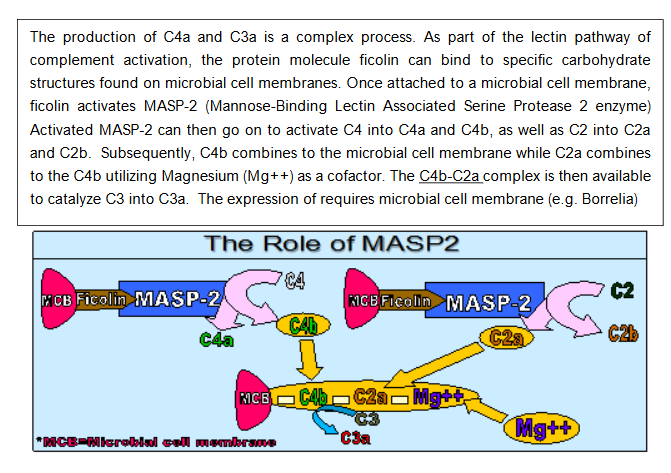
Step 10: Correct Complement 4a
C4a is a split product of complement activation through either the classical or lectin complement activation pathway. In the lectin pathway, once MASP-2 (mannose-binding lectin associated serine protease 2 enzyme) is activated, it can trigger conversion of C4 into C4a and C4b. Like C3a, C4a is an anaphylatoxin and can activate mast cells and basophils, increase smooth muscle contraction and vascular permeability, and cause release of chemotactic factors. C4a causes capillary hypoperfusion and lead to cellular hypoxia, with resultant mitochondrial dysfunction. When C4a is elevated in isolation of C3a, you are most likely dealing with exposure to a water damaged building, whereas C4a and C3a elevations can be tick born illness (e.g., Lyme) or tick-borne illness with concomitant exposure to WDB.
C4a levels will usually normalize after the patient is out of exposure, taken adequate bile acid sequestrants and has documented successful eradication of MARCoNS.
Exposure is evaluated with clearance tests for their living and working environments as previously discussed. If the first three steps of the CIRS protocol are executed and elevated C4a persists, VIP (vasoactive intestinal peptide) nasal spray can be applied (see Step 12).
Step 11: Correct Transforming Growth Factor Beta-1
Elevations of transforming growth factor beta-1 (TGFβ-1) are seen in various disorders. TGFβ-1 is an important immune modulator but in certain environments (e.g., biotoxin exposure) TGFβ-1 becomes a potent inflammatory cytokine that promotes tissue destruction and participates in fibrotic remodeling. If you have fibrosis, dysregulated and elevated levels of TGFβ-1 are participating in the pathology of fibrotic change.
Pulmonary fibrosis, liver cirrhosis, nasal polyps and/or polycystic kidney disease all suffer the consequence of imbalanced TGFβ-1.
It is imperative to evaluate any fibrotic disorder for CIRS as an underlying or participating cause. When TGFβ-1 levels are high, CD4+ CD25+ T regulatory cells are hydrolyzed. The hydrolysis of this cell types precipitates the release of pathogenic effector T cells, which then induce more TGFβ-1 production. This vicious cycle of perpetuating unregulated cytokines is a hallmark of CIRS and supports the creation of multi-system, multi-symptom illness.
Like C4a, if the first three steps of the CIRS protocol are executed, elevated TGFβ-1 usually normalize. If not responding with execution of previous steps, two treatment options are available to address elevated TGFβ-1.
The first option is using the specific angiotensin II receptor blocker (ARB), Cozaar (losartan). Losartan has been shown to work due to a degradation product called EXP 3179, which can significantly lower TGFβ-1 with minimal effects on blood pressure. Although similar in many ways to Cozaar, the ARB Benicar (olmesartan, medoxomil) lacks EXP 3179, making it ineffective in lowering TGFβ-1.
Despite being classified as a blood pressure lowering medicine, Losartan has mild to moderate effects on lowering blood pressure, but major benefit at preventing heart attack and stroke most likely due to its anti-inflammatory, immune modulating effects. Patients with normal to elevated blood pressure start on 25 mg / day and increase to 25 mg twice a day (if repeat TGFβ-1 is not responding).
For patients with lower blood pressure, 12.5 mg twice a day is a better starting point. If low blood pressure is an existing concern, VIP (vasoactive intestinal peptide) can be implemented to modulate TGFβ-1.
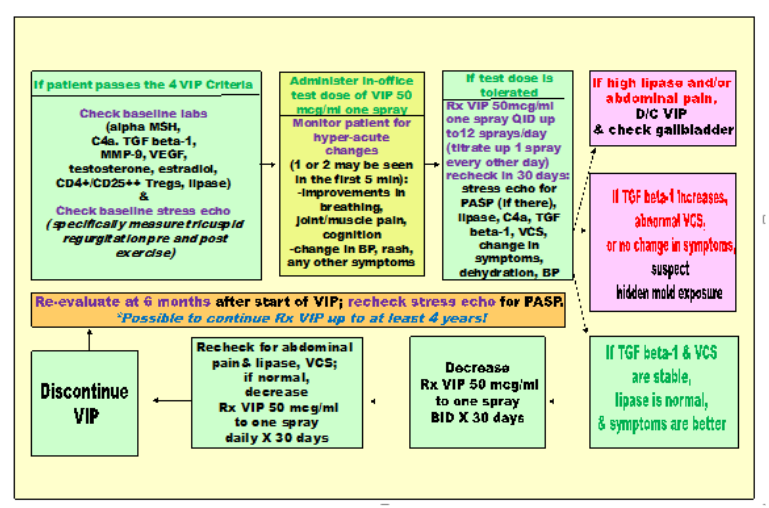
Step 12: Vasoactive Intestinal Peptide (VIP)
Vasoactive intestinal peptide (VIP) is a 28 amino acid regulatory neuropeptide with many beneficial physiologic effects. VIP is a neuroimmune modulator that can down-regulate cytokine levels and have a global restorative effect on the multisystem, multi-symptom experience of CIRS. In an open label trial with 20 refractory CIRS patients (completion of the first 11 steps), VIP brought resolution to previously non-responsive symptoms.
Patients with high TGF beta-1 have a myriad of possible signs and symptoms, including the following:
- Lung symptoms
- Neurological problems – Oligoclonal band (+) multiple sclerosis (MS) that responds to treatment; atypical seizures; resting tremor; Parkinson-like syndromes; transverse myelitis.
- Autoimmune disorders – Lupus, rheumatoid arthritis, dermatomyositis, chronic ulcerative colitis. If (+) ACLA, ANCA, AGA, or anti-actin antibodies, look for TGF beta-1 elevation; these autoimmune issues may abate once TGFβ-1 is lowered.
- Learning Disabilities – Neurodevelopmentally delayed children with high TGF beta-1 may have developmental improvement after treatment.
- Fibrotic Diseases – Interstitial lung, gadolinium associated fibrosis, scleroderma-like illnesses.
- Nasal & Vocal Cord Polyps.
- Increased Pulmonary Artery Systolic Pressure – This has shown to improve by lowering high TGF beta-1 in Biotoxin Illness patients.
Even more impressive is the 2017 study showing intranasal VIP safely restored volume to multiple grey matter nuclei in patients with CIRS. VIP nasal spray can restore gray matter volume as measured by pre and post brain MRI with NeuroQuant®.
The initial paper shows one spray of VIP (50 mcg per spray) in alternating nostrils 4x per day regrew the grey matter over 18 months. Subsequent unpublished data sets show a similar phenomenon with the same number of total sprays but executed in 6 months at 4 sprays, taken three times per day (i.e., 4 sprays per day for 18 months versus 12 sprays per day for 6 months).
The major contraindication for VIP is elevating fasting lipase with usage. Fasting lipase should be checked after first time usage and repeated two weeks into the spray and after every major increase in sprays. Up to 24 sprays divided into 4x/day dosing schedules have been used for severe brain atrophy with stricter monitoring of fasting lipase.
Conclusion
The treatment of Chronic Inflammatory Response Syndrome (CIRS) requires a methodical, evidence-based approach that addresses the unique immune, endocrine, and neurological disruptions caused by biotoxin exposure. The 12-step CIRS Protocol, evidence-based framework and enhanced through clinical refinement, offers a reproducible and comprehensive path to recovery. Central to its success is the execution of foundational steps, particularly removal from biotoxin exposure and elimination of internal biotoxin reservoirs before progressing to targeted biomarker correction and immune modulation.
This protocol emphasizes precision sequencing, objective biomarker tracking, and patient-specific customization, integrating both conventional and adjunctive therapies including bile acid sequestrants, fish oil, hormone restoration, antifibrotic agents like losartan, and restorative peptides such as VIP. When applied with clinical rigor and patient adherence, this protocol facilitates resolution of multisystem symptoms, reverses immune dysregulation, and in many cases, restores structural brain integrity as evidenced by NeuroQuant® imaging.
Given the rising prevalence of environmentally acquired illnesses and the complexity of chronic inflammatory conditions, the CIRS Protocol stands as a scalable, systems-based model of medicine with significant potential for improving outcomes in patients with previously unexplained, treatment-resistant illness.
Gratitude: We are grateful for the comments of David Lark, Ritchie Shoemaker MD and Scott McMahon MD.
Conflict of Interest Statement: None.
Funding Statement: None.
Acknowledgements: Special thanks to Dr. Shoemaker for relentless commitment to eradicating chronic illness, Scott McMahon MD for establishing CIRS pediatric norms, Yvonne Berry MD for help with Diagrams and Deb Waidner for helping train the next generation of providers to serve those suffering with CIRS.
References
- Donta ST. Borrelia burgdorferi 0755, a novel cytotoxin with unknown function in Lyme disease. Toxins (Basel). 2024;16(6):233.
- Shoemaker RC. Metabolism, molecular hypometabolism and inflammation: complications of proliferative physiology include metabolic acidosis, pulmonary hypertension, T reg cell deficiency, insulin resistance and neuronal injury. Trends Diabetes Metab. 2020;3.
- Consensus statement. Medically sound investigation and remediation of water-damaged buildings in cases of CIRS-WDB.
- Hudnell HK. Chronic biotoxin-associated illness: multiple-system symptoms, a vision deficit, and effective treatment. Neurotoxicol Teratol. 2005;27(5):733-743.
- Shoemaker RC, House DE. A time-series study of sick building syndrome: chronic, biotoxin-associated illness from exposure to water-damaged buildings. Neurotoxicol Teratol. 2005;27(1):29-46.
- Sekine T, Cha S, Endou H. The Mult specific organic anion transporter (OAT) family. Eur J Physiol. 2000;440:337-350.
- Yu Z, You G. Recent advances in the regulations of organic anion transporters. Pharmaceutics. 2024;16(11):1355.
- Anzai N, Kanai Y, Endou H. Organic anion transporter family: current knowledge. J Pharmacol Sci. 2006;100(5):411-414.
- Ticho AL, Malhotra P, Dudeja PK, Gill RK, Alrefai WA. Bile acid receptors and gastrointestinal functions. Liver Res. 2019;3(1):31-39.
- Kim YS, Kim JW, Ha NY, Kim J, Ryu HS. Herbal therapies in functional gastrointestinal disorders: a narrative review and clinical implication. Front Psychiatry. 2020;11:601.
- Cheema HS, Singh MP. The use of medicinal plants in digestive system related disorders: a systematic review. J Ayurvedic Herb Med. 2021:182-187.
- Fifi AC, Axelrod CH, Chakraborty P, Saps M. Herbs and spices in the treatment of functional gastrointestinal disorders: a review of clinical trials. Nutrients. 2018;10(11):1715.
- Ford AC, Talley NJ, Spiegel BM, et al. Effect of fiber, antispasmodics, and peppermint oil in the treatment of irritable bowel syndrome: systematic review and meta-analysis. BMJ. 2008;337.
- Khanna R, MacDonald JK, Levesque BG. Peppermint oil for the treatment of irritable bowel syndrome: a systematic review and meta-analysis. J Clin Gastroenterol. 2014;48(6):505-512.
- Wegener T, Wagner H. The active components and the pharmacological multi-target principle of STW 5 (Iberogast). Phytomedicine. 2006;13 Suppl 5:20-35.
- Cho MS, Park JW, Kim J, Ko SJ. The influence of herbal medicine on serum motilin and its effect on human and animal model: a systematic review. Front Pharmacol. 2023;14:1286333.
- Poggioli R, Hirani K, Jogani VG, Ricordi C. Modulation of inflammation and immunity by omega-3 fatty acids: a possible role for prevention and to halt disease progression in autoimmune, viral, and age-related disorders. Eur Rev Med Pharmacol Sci. 2023;27(15):7380-7400.
- Gutiérrez S, Svahn SL, Johansson ME. Effects of omega-3 fatty acids on immune cells. Int J Mol Sci. 2019;20(20):5028.
- Mendivil CO. Dietary fish, fish nutrients, and immune function: a review. Front Nutr. 2021;7:617652.
- Damsgaard CT, Lauritzen L, Kjær TMR, et al. Fish oil supplementation modulates immune function in healthy infants. J Nutr. 2007;137(4):1031-1036.
- Gallai V, Sarchielli P, Trequattrini A, et al. Cytokine secretion and eicosanoid production in the peripheral blood mononuclear cells of MS patients undergoing dietary supplementation with n-3 polyunsaturated fatty acids. J Neuroimmunol. 1995;56(2):143-153.
- Marciani L, Cox E, Hoad C, et al. Effects of various food ingredients on gall bladder emptying. Eur J Clin Nutr. 2013;67:1182-1187.
- Salman A, Zaheer M, Mallick I, Hassan M. Choleretic and cholagogic effects of anti-cholelithiatic plants. J Pharmacogn Phytochem. 2021;10:1-7.
- Polymeros D, Beintaris I, Gaglia A, et al. Partially hydrolyzed guar gum accelerates colonic transit time and improves symptoms in adults with chronic constipation. Dig Dis Sci. 2014;59(9):2207-2214.
- Arnaud MJ. Mild dehydration: a risk factor of constipation? Eur J Clin Nutr. 2003;57 Suppl 2: S88-95.
- Anti M, Pignataro G, Armuzzi A, et al. Water supplementation enhances the effect of high-fiber diet on stool frequency and laxative consumption in adult patients with functional constipation. Hepatogastroenterology. 1998;45(21):727-732.
- Song BK, Kim YS, Kim HS, et al. Combined exercise improves gastrointestinal motility in psychiatric inpatients. World J Clin Cases. 2018;6(8):207-213.
- Kim YS, Song BK, Oh JS, Woo SS. Aerobic exercise improves gastrointestinal motility in psychiatric inpatients. World J Gastroenterol. 2014;20(30):10577-10584.
- Bi L, Triadafilopoulos G. Exercise and gastrointestinal function and disease: an evidence-based review of risks and benefits. Clin Gastroenterol Hepatol. 2003;1(5):345-355.
- Shafer RB, Prentiss RA, Bond JH. Gastrointestinal transit in thyroid disease. Gastroenterology. 1984;86(5 Pt 1):852-855.
- Harvey RF, Read AE. Effects of oral magnesium sulphate on colonic motility in patients with the irritable bowel syndrome. Gut. 1973;14(12):983-987.
- Bueno L, Fioramonti J, Geux E, Raissiguier Y. Gastrointestinal hypomotility in magnesium-deficient sheep. Can J Anim Sci. 2011;60(2):293-301.
- Stacewicz-Sapuntzakis M, Bowen PE, Hussain EA, et al. Chemical composition and potential health effects of prunes: a functional food? Crit Rev Food Sci Nutr. 2001;41(4):251-286.
- Wilkinson-Smith V, Dellschaft N, Ansell J, et al. Mechanisms underlying effects of kiwifruit on intestinal function shown by MRI in healthy volunteers. Aliment Pharmacol Ther. 2019;49(6):759-768.
- Shoemaker R, Hudnell K, House D, Domenico P. Association of nasal carriage of methicillin and multiple antibiotic resistant coagulase negative staphylococci species with deficiency of alpha melanocyte stimulating hormone in chronic fatigue syndrome: implication for expanded treatment options. American Society of Microbiology. 2003.
- Becker K, Heilmann C, Peters G. Coagulase-negative staphylococci. Clin Microbiol Rev. 2014;27(4):870-926.
- Heilmann C, et al. Are coagulase-negative staphylococci virulent? Clin Microbiol Infect. 2019;25(9):1071-1080.
- Le KY, Park MD, Otto M. Immune evasion mechanisms of Staphylococcus epidermidis biofilm infection. Front Microbiol. 2018;9:359.
- Qin L, Da F, Fisher EL, et al. Toxin mediates sepsis caused by methicillin-resistant Staphylococcus epidermidis. PLoS Pathog. 2017;13(2):e1006153.
- Coagulase-negative staphylococcus, nosocomial infections. Infectious Disease Advisor. February 16, 2024. Accessed July 29, 2025. https://www.infectiousdiseaseadvisor.com/ddi/coagulase-negative-staphylococcus/
- Vuong C, Otto M. Staphylococcus epidermidis infections. Microbes Infect. 2002;4(4):481-489.
- Marincola G, Liong O, Schoen C, et al. Antimicrobial resistance profiles of coagulase-negative staphylococci in community-based healthy individuals in Germany. Front Public Health. 2021;9:684456.
- Asante J, Hetsa BA, Amoako DG, et al. Multidrug-resistant coagulase-negative staphylococci isolated from bloodstream in the uMgungundlovu District of KwaZulu-Natal Province in South Africa: emerging pathogens. Antibiotics (Basel). 2021;10(2):198.
- He S, Lin J, Li Y, et al. Insights into the epidemiology of methicillin-resistant coagulase-negative staphylococci carriage in community-based drug users. J Infect Public Health. 2020;13(11):1742-1748.
- Catassi GN, Pulvirenti A, Monachesi C, et al. Diagnostic accuracy of IgA anti-transglutaminase and IgG anti-deamidated gliadin for diagnosis of celiac disease in children under two years of age: a systematic review and meta-analysis. Nutrients. 2021;14(1):7.
- Saadah OI, Alamri AM, Al-Mughales JA. Deamidated gliadin peptide and tissue transglutaminase antibodies in children with coeliac disease: a correlation study. Arab J Gastroenterol. 2020;21(3):174-178.
- Pumar M, Choo S, Rosenbaum J, et al. No-biopsy diagnosis of coeliac disease in children without anti-endomysial IgA antibody testing: combining anti-tissue transglutaminase IgA and anti-deamidated gliadin IgG antibodies. J Paediatr Child Health. 2025;61(4):628-634.

