Cognitive Impairment Caregiver Reported-Health Index
The Cognitive Impairment Caregiver Reported-Health Index: Development and Validation of a Caregiver-Reported Outcome Measure
Christina Shupe, MPH1; Jamison Seabury, BS1,2; Jennifer Weinstein, MS1; Charlotte Engebrecht, BS1; Charlotte Irwin, BS1; Preshetha Kanagaiah, BS1; Anika Varma, BS1,2; Spencer Rosero, BS1,3; Nuran Dilek, MS4; Abigail Arky, DO5; John Heatwole6, Elizabeth Santos, MD, MPH7; Chad Heatwole, MD, MS-CI1,4
- Christina Shupe, MPH University of Rochester Center for Health + Technology, Rochester NY, USA
- Jamison Seabury, BS University of Rochester Center for Health + Technology, Rochester NY, USA; University of Rochester School of Medicine and Dentistry, Rochester NY, USA
- Jennifer Weinstein, MS University of Rochester Center for Health + Technology, Rochester NY, USA
- Charlotte Engebrecht, BS University of Rochester Center for Health + Technology, Rochester NY, USA
- Charlotte Irwin, BS University of Rochester Center for Health + Technology, Rochester NY, USA
- Preshetha Kanagaiah, BS University of Rochester Center for Health + Technology, Rochester NY, USA
- Anika Varma, BS University of Rochester Center for Health + Technology, Rochester NY, USA; University of Rochester School of Medicine and Dentistry, Rochester NY, USA
- Spencer Rosero, BS University of Rochester Center for Health + Technology, Rochester NY, USA; University of Utah Spencer Fox Eccles School of Medicine, Salt Lake City UT, USA
- Nuran Dilek, MS University of Rochester Department of Neurology, Rochester NY, USA
- Abigail Arky, DO Trinity Health of New England, Hartford, CT, USA
- John Heatwole Cornell University College of Agriculture and Life Sciences, Ithaca, NY, USA
- Elizabeth Santos, MD, MPH University of Rochester Medical Center, Memory Care Program, Rochester NY, USA
- Chad Heatwole, MD, MS-CI1 University of Rochester Center for Health + Technology, Rochester NY, USA; University of Rochester Department of Neurology, Rochester NY, USA
OPEN ACCESS
PUBLISHED: 30 June 2025
CITATION: Shupe, C., Seabury, J., et al., 2025. The Cognitive Impairment Caregiver Reported-Health Index: Development and Validation of a Caregiver-Reported Outcome Measure. Medical Research Archives, [online] 13(6). https://doi.org/10.18103/mra.v13i6.6653
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i6.6653
ISSN 2375-1924
Abstract
Background: There is a need for valid, disease-specific, caregiver-reported outcome measures that reliably assesses the symptomatic health of individuals across the Alzheimer’s disease continuum from mild cognitive impairment to dementia.
Aims: This research describes the development and validation of the Cognitive Impairment Caregiver Reported-Health Index (CICR-HI), a disease-specific, caregiver-reported outcome measure designed according to United States Food and Drug Administration (FDA) guidance to assess the point-in-time symptomatic burden of individuals with Alzheimer’s disease, mild cognitive impairment, and dementia.
Methods: Previous work identified the symptoms of greatest importance to individuals with cognitive impairment through caregiver qualitative interviews and a cross-sectional study. We selected symptomatic questions for potential inclusion in the CICR-HI based on their high importance to individuals with cognitive impairment. We conducted beta testing and test-retest reliability testing of the CICR-HI with caregivers of individuals with cognitive impairment to determine the usability and reliability of the instrument, its subscales, and its individual questions. Lastly, we determined the known groups validity and internal consistency of the final instrument.
Results: The validated CICR-HI measures eight symptomatic domains that are shown to be highly relevant to individuals with cognitive impairment. Beta testing with 12 caregivers revealed that the CICR-HI was highly relevant and easy to use. Test-retest reliability with 30 caregivers demonstrated a high intra-class correlation (0.801). Lastly, known groups analysis demonstrated that the CICR-HI is able to statistically distinguish between groups of individuals believed to have different levels of disease severity based upon demographic or clinical characteristics.
Conclusion: The CICR-HI is a regulatory-grade, disease-specific, caregiver-reported outcome measure capable of measuring disease burden across eight symptomatic domains in individuals with cognitive impairment. This instrument was developed and validated according to published FDA guidelines for use in clinical trials to serially measure how individuals with cognitive impairment feel and function. The CICR-HI adds to existing clinical trial infrastructure and provides a mechanism to quantify changes in multifaceted disease burden in cognitive impairment over time or in response to therapeutic intervention.
Keywords: Alzheimer’s disease, mild cognitive impairment, dementia, outcome measure
Introduction
Alzheimer’s disease is a multifactorial, neurodegenerative disorder that progresses along a clinical continuum from pre-symptomatic disease to mild cognitive impairment to dementia. Alzheimer’s disease has a heterogeneous symptomology demonstrated by varied cognitive presentations, functional deficits, and rates of progression. Cognitive impairment is one of the leading causes of disability and morbidity in older adults in the world. In 2023, an estimated 416 million people worldwide were living with preclinical AD, mild cognitive impairment, or dementia. A projected worldwide growth in the elderly population is anticipated to triple the number of dementia cases by 2050, causing further strain on healthcare resources and caregiving social support systems.
The United States Food and Drug Administration (FDA) approvals of anti-amyloid therapies for the treatment of AD were met with controversy in the global AD community, despite the growing need for therapeutic advancement. The ensuing debate on the world stage challenged the use of clinicostatistical measures of cognition and function in isolation as primary outcomes and the absence of data regarding the patient’s and caregiver’s perspective on their symptoms, their ability to perform daily tasks, and the impact of the disease on their lives.
Following these approvals, the FDA, the European Medicines Agency (EMA), the National Institute on Aging (NIA) and the National Institute of Neurological Disorders and Stroke (NINDS) have all acknowledged the imperative need for the use of patient-centered outcomes to determine patient-centered, clinically meaningful changes for clinical trials of AD disease-modifying treatments in Alzheimer’s disease. The utilization of patient insight to quantify cognitive, functional, and emotional health is critical for a robust assessment of the effectiveness of future AD interventions. Individuals with more severe disease-related disruptions to memory and cognition may not be able to reliably self-report the impact of disease and treatment. The intensive, proximal role of a caregiver in the life of an individual with cognitive and functional deficits renders them a valuable resource for both assessing observable symptomatic burden and quantifying changes in health over time.
This study documents the development and validation of the Cognitive Impairment Caregiver Reported-Health Index (CICR-HI), a caregiver-reported outcome measure designed to quantify from the caregiver perspective how individuals with AD, dementia, or mild cognitive impairment (MCI) feel and function in response to therapeutic intervention. In prior work, we determined the extent and importance of symptoms experienced by individuals with AD, dementia, and MCI through interviews and large-scale surveys with patients and caregivers. This caregiver-reported outcome measure is designed in alignment with FDA guidance to sensitively assess the multifactorial point-in-time disease burden of symptoms determined to have the greatest impact on the lives of individuals with cognitive impairment. Our work seeks to bolster existing clinical trial infrastructure by facilitating patient-meaningful therapeutic assessments in a population unable to reliably self-report.
Methods
PARTICIPANT SELECTION AND ELIGIBILITY
Study eligibility required that a participant be: (1) 18 years of age or older; (2) a caregiver for an individual diagnosed with AD, MCI, or dementia; (3) English speaking; and, (4) residing in the United States. Exclusion criteria restricted individuals from participating in this study if they did not meet all study inclusion criteria. Study participants were recruited from the Alzheimer’s Prevention Registry (Banner Health Institute), ResearchMatch.com, the University of Rochester (UR) AD-Care, Research, and Education (AD-CARE) program, UR Memory Care Program, and UR Clinical Translational Science Institute (CTSI) Registry. Participants provided consent by phone or online after receiving a study information letter detailing study activities and their rights as a voluntary participant. Participant information and responses were stored and protected according to United States Health Insurance Portability and Accountability Act (HIPAA) guidelines. All study activities conform to the Declaration of Helsinki and were approved by the University of Rochester’s Research Subjects Review Board (RSRB).
QUESTION SELECTION & CONTENT VALIDITY
In prior work, we utilized qualitative interviews and a large cross-sectional study of patients and caregivers to identify the symptoms with the greatest impact on individuals with cognitive impairment. Semi-structured qualitative interviews were conducted with 15 caregivers (Appendix 1), who provided 1,097 quotes regarding the symptomatic burden experienced by individuals with cognitive impairment for whom they care. Data obtained from these interviews were used to design a cross-sectional survey asking about 99 potential symptoms of importance. This online survey was accessed on Research Electronic Data Capture (REDCap), a secure, HIPAA-compliant, web-based platform. This survey was completed by 329 eligible caregivers to determine the most prevalent and impactful symptoms among individuals with cognitive impairment.
Data from the cross-sectional survey was used to calculate each symptom’s population impact scores (PIP), which describes the relationship between a symptom and its impact on the individual’s life. The PIP is a metric represented on a scale from 0 – 4, calculated as the product of the symptom’s prevalence in individuals with cognitive impairment and its average impact. Symptom questions with a PIP less than 1.0 were excluded from the first version of the CICR-HI, as this indicated less relevance to individuals with cognitive impairment. Questions were also removed if they were: (1) redundant; (2) potentially offensive to future participants; (3) unlikely to be affected by a therapeutic intervention; (4) inapplicable to a broad population of individuals with cognitive impairment; (5) above an eighth grade reading level; or, (6) not directly observable by a caregiver. This methodology has been previously documented and used by our laboratory to generate instruments for over 30 different diseases. Symptom questions that met all selection criteria were grouped into subscales, or symptomatic themes, based on the domain of patient health that they represented.
BETA INTERVIEWS – INSTRUMENT RELEVANCE & USABILITY
The first version of the CICR-HI was evaluated by caregiver participants through beta interviews. Following the online completion of the CICR-HI, caregivers participated in semi-structured interviews, providing feedback on the content, relevance, and usability of the instrument. Participants addressed if they believed that the instrument was capable of measuring how a patient feels and functions and if they were able to complete the instrument without experiencing user burden. Interview questions also inquired about the comprehension, usability, response processes, recall strategies (or timeframe of responses), and relevance of the first version of the CICR-HI. Caregivers provided feedback on any symptom questions that should be reworded for clarity, or additional symptoms they felt were missing from the first version of the CICR-HI. Participant interviews were conducted from January 2022 to September 2022. The interviews were audio-recorded, transcribed, and analyzed. Using the feedback from these interviews, modifications were made to create the second version of the CICR-HI.
TEST-RETEST RELIABILITY
Caregiver participants longitudinally completed the CICR-HI at baseline and 14 days later. A 14-day interval between baseline and retest minimizes the likelihood of symptom progression while reducing baseline recall. The instrument was administered to caregivers electronically using REDCap. Survey administration was conducted in December 2022. Test-retest reliability was quantified using a weighted kappa (WK) value for each symptom question. Less reliable questions with a WK value less than 0.40 were considered for removal from the survey.
INSTRUMENT FORMAT, SCORING, AND INTERNAL CONSISTENCY
The CICR-HI consists of symptomatic subscales representing unique domains of health for individuals with cognitive impairment as reported by their caregivers. For each symptom question, the caregiver is asked “How much does the following impact his/her life?” Reponses are rated on a 6-point Likert Scale. We generated a short form of the CICR-HI that included one representative question from each subscale in the CICR-HI to be used as a potential surrogate to the full form. The total score, short form, and each subscale are scored between 0 and 100, where 0 indicates no disease burden and 100 indicates maximum disease burden. Questions within subscales are weighted based on their relative importance to patients (as determined by the cross-sectional study), in order to generate a total score. We determined the internal consistency of the CICR-HI and each of its subscales using Cronbach’s alpha values.
KNOWN GROUPS ANALYSIS
We conducted known groups analysis for predefined patient subgroups thought to have higher or lower disease burden including: patient age (above versus equal to and below mean 80.1 years); patient sex (male versus female); patient education (grade school, high school, or technical degree versus college, masters, or doctorate degree); patient employment status (employed full-time, part-time, retired, stay-at-home parent, or other versus on disability or not working and not on disability); patient change in employment status due to disease (yes versus no); patient marital status (married or registered partner versus single, widowed, divorced, separated, or other); patient diagnosis (AD versus MCI, AD versus non-AD dementias, and non-AD dementias versus MCI); patient living situation (resides in community versus resides in assisted, independent, or skilled-nursing living facility, or other); patient fit of ability (no symptoms or significant disability, slight disability, or moderate disability versus moderately-severe or severe disability); years since onset of thinking problems (above versus equal to or below mean 7.5 years); patient receiving genetic testing (yes versus no); APOE_4 copies (one copy versus 2 copies); and, seizures (experiences versus does not experience seizures).
We also conducted known groups analysis for predefined subgroups based on caregiver characteristics including: caregiver age (above versus equal to or below mean 65.2 years); caregiver sex (male versus female); caregiver relationship to patient (son or daughter versus spouse or partner); and, the use of a home health aide (yes versus no). The Benjamini-Hochberg analysis was applied to the known groups analysis with a false discovery rate of 0.05 and 180 test statistics. In accordance with this statistical methodology, the p values obtained from each known groups comparison were sorted by size from smallest to largest, with the largest value of i such that p(i) , 0.05, i/180 was determined. The null hypothesis associated with the p values p(1), …, p(i) were rejected, resulting in 128 discoveries.
Results
QUESTION SELECTION AND CONTENT VALIDITY
During our initial study, we interviewed 15 caregivers and identified 173 potential symptoms of importance to individuals with cognitive impairment. Our subsequent cross-sectional study evaluated the prevalence and relative importance of 99 symptomatic areas in a cohort of 329 caregiver participants. Based on the cross-sectional survey responses of these initial 99 symptom questions, we eliminated 18 questions due to low population impact (PIP < 1.0), 2 questions due to redundancy, 13 questions for being non-responsive to future therapies, 2 for vague wording, and one for being deemed not observable by a caregiver. Three questions were also removed due to statistical redundancy. Following question removal, the first version of the CICR-HI consisted of 60 symptom questions representing 8 symptomatic themes. An overview of the question selection process is provided in Figure 1. The demographics of all study participants (caregivers) and the individuals they represented are provided in Appendix 1.
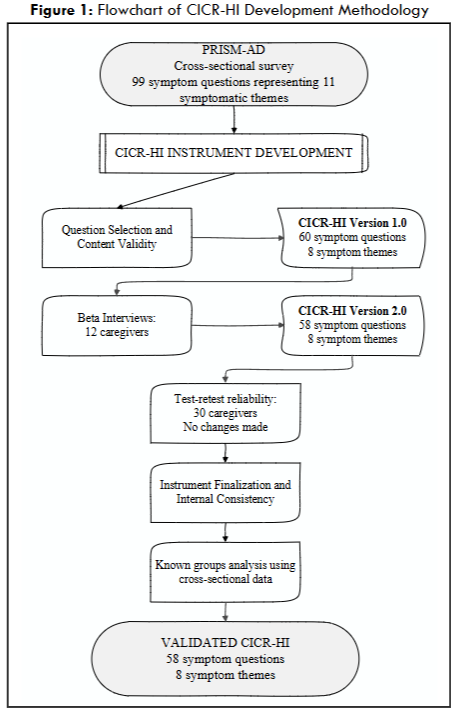
BETA INTERVIEWS – INSTRUMENT RELEVANCE AND USABILITY
Twelve caregivers of individuals with cognitive impairment participated in beta interviews and provided feedback regarding the first version of the CICR-HI (Appendix 1 provides details regarding the demographic features of these participants). Caregivers found the CICR-HI to be an appropriate way to assess disease severity and capture how the individual for whom they care feels and functions. Based on caregiver feedback, six symptom questions were reworded to enhance clarity, and two symptom questions were deleted due to redundancy. Upon completion of these revisions, the second version of the CICR-HI was developed, consisting of 58 questions representing eight symptomatic themes (8 subscales).
TEST-RETEST RELIABILITY
Thirty caregivers participated in test-retest reliability of the second version of the CICR-HI (Appendix 1 provides details regarding the demographic features of these participants). All symptom questions demonstrated an acceptable level of reliability, with a weighted kappa greater than 0.40. Intraclass Correlation Coefficient (ICC) values for the total instrument and each of the subscales ranged from 0.633 to 0.813 (Table 1).
| CICR-HI Subscale | Questions included in Final Subscale (n) | Mean Participant Scores | Internal Consistency (Cronbach’s alpha) | Test-Retest Reliability (Intraclass Correlation Coefficient) | Baseline | 2 Week Retest |
|---|---|---|---|---|---|---|
| Memory | 11 | 58.2 | 0.96 | 0.813 | 54.2 | |
| Cognition | 13 | 63.4 | 0.97 | 0.717 | 54.4 | |
| Fatigue | 2 | 42.6 | 0.86 | 0.684 | 45.8 | |
| Sleep or Daytime Sleepiness | 5 | 34.8 | 0.89 | 0.711 | 37.5 | |
| Social Satisfaction | 5 | 42.1 | 0.93 | 0.774 | 38.5 | |
| Communication | 5 | 43.3 | 0.85 | 0.633 | 43.4 | |
| Emotional Health | 7 | 55.3 | 0.93 | 0.746 | 50.3 | |
| Activity Participation | 10 | 45.4 | 0.94 | 0.672 | 38.7 | |
| Total Form | 50 | 49.1 | 0.98 | 0.801 | 46.0 | |
| Short Form | 8 | 51.9 | 0.90 | 0.723 | 51.0 |
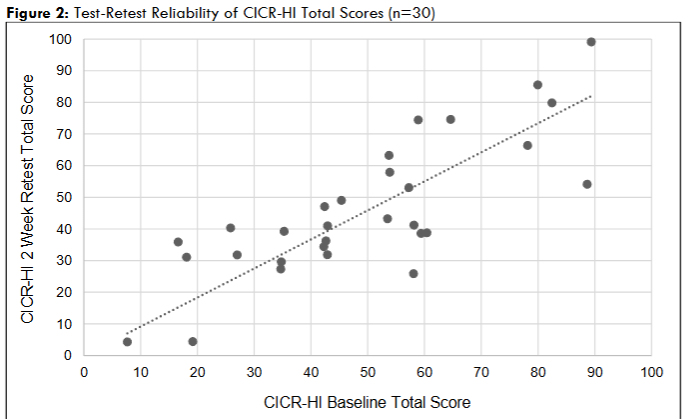
Caregivers took an average of 6.26 and 5.49 minutes to complete the baseline and 14-day time point survey, respectively. The CICR-HI total score demonstrated no floor or ceiling effects (Figure 2). No modifications to the CICR-HI were required secondary to its performance during this phase of development.
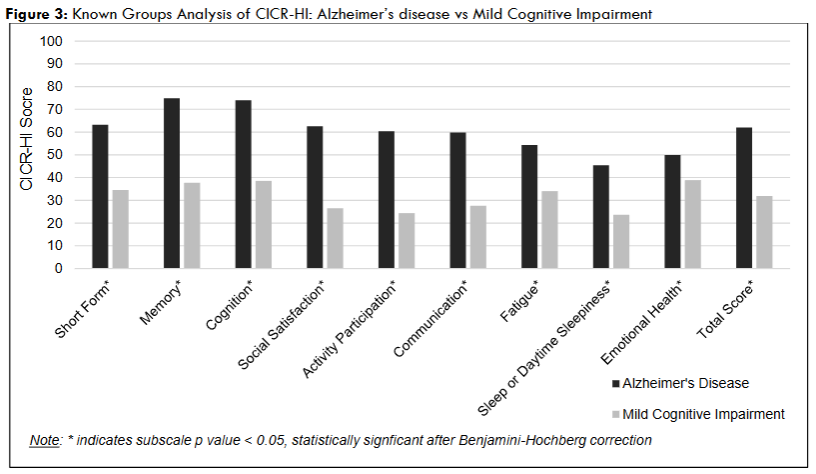
KNOWN GROUPS ANALYSIS
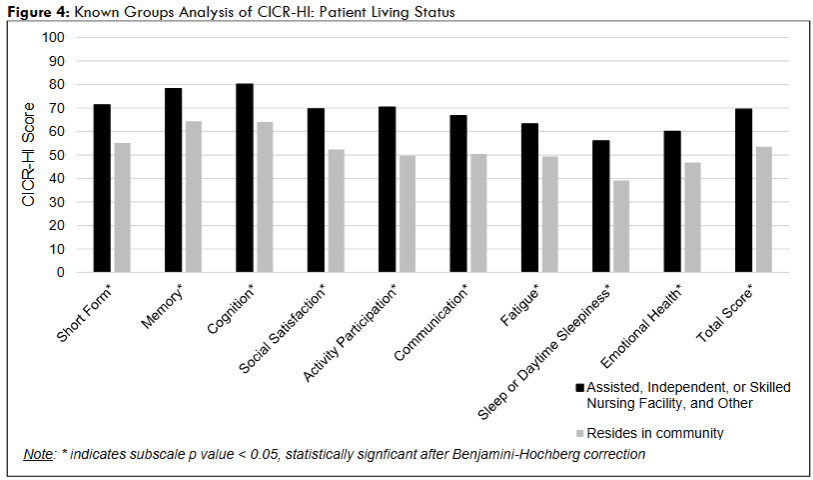
Cross-sectional data from 329 participants was used to determine the known groups validity of the CICR-HI and each of its subscales. After correcting for multiple comparisons, we found significant differences among predefined subgroups (Appendix 2). A higher CICR-HI total score, indicating higher disease burden, was associated with: patient age (above 80.1 years); sex (female); highest education received (grade school, high school, or technical school versus college or graduate school); marital status (not married or in a registered partnership); fit of ability (moderate to severe disability versus no symptoms, slight disability, or moderate disability); living situation (resides in assisted, independent, or skilled-nursing living facility versus in the community); years since onset of thinking problems (above the 7.5 years); diagnosis (AD or non-AD dementia compared to MCI); APOE_4 copies (two copies versus one copy); and the presence of seizures.
Additionally, a higher CICR-HI total score, indicating higher disease burden, was associated with: caregiver age (older than 65.2 years); caregiver relationship to the individual (child versus spouse); and use of a home health aide. There was no difference in CICR-HI total score based on individual with cognitive impairment’s employment status, employment status change due to their disease, if the individual received genetic testing, or the caregiver’s sex. Of note, higher scores were observed across CICR-HI total form, short form, and subscales based upon the individual diagnosis (AD and non-AD dementias, compared to MCI).
INSTRUMENT FINALIZATION AND INTERNAL CONSISTENCY
The final version of the CICR-HI consists of 58 symptomatic questions representing eight symptomatic subscales that address the following areas of disease burden: Memory function; Cognition; Fatigue; Sleep or daytime sleepiness; Social satisfaction; Communication; Emotional health; and, Activity participation. The internal consistency of the final CICR-HI and each is its subscales is provided in Table 1, with the CICR-HI full form having a Cronbach’s alpha score of 0.98.
Discussion
The CICR-HI is a caregiver-reported outcome measure that comprehensively measures the physical, mental, social, and disease-specific health of individuals with cognitive impairment through the caregiver perspective. This instrument, developed according to published FDA guidelines for use in clinical trials, prioritizes domains of health identified in prior research as being the most important to individuals with cognitive impairment. The CICR-HI was assessed by caregivers as being a comprehensive and appropriate measure of a patient’s health and function while being easy to complete and understand. On average, the CICR-HI was completed in approximately six minutes, with the short form being completed in less than one minute. Importantly, the instrument measures the symptomatic concepts that are most important to individuals with cognitive impairment, provides a reliable mechanism to quantify their multifactorial disease burden over time, and is capable of differentiating between patients with greater or lower levels of symptomatic burden.
Known groups analysis of individuals across the Alzheimer’s continuum provides insight into differences in multidimensional symptomatic burden. When comparing AD patients to patients with non-AD dementias, similar scores were observed for the CICR-HI total, short form, and subscale scores, however, AD patients reported a significantly higher symptomatic burden in the CICR-HI Memory function subscale. This is consistent with previous findings that patients with AD experience memory deficits that are unique from those experienced by non-AD dementia patients, including severe and focused impairment related to verbal memory, recognition, and attention deficits.
Caregivers who were children of the patient reported significantly higher CICR-HI total, short form, and subscale scores when compared to spousal caregivers. We suspect that this difference is due to relationship-based reporting perspective and, more importantly, differences in patient age between these two groups. Specifically, child caregivers reported on behalf of older patients compared to spousal caregivers. Among primary caregivers who were the children of the patient, the average age of the patient was 85.9 years old. In contrast, for the primary caregiver who were spouses or partners to the patient with cognitive impairment, the average patient age was 74.6 years.
The CICR-HI addresses gaps in clinical trial readiness by including subscales that measure symptomatic concepts that are infrequently measured by other disease-specific instruments. The CICR-HI includes Fatigue and Sleep and daytime sleepiness subscales, domains shown to be highly relevant in Alzheimer’s disease, which are not assessed in existing instruments such as the Dementia Severity Scale and Dementia Quality of Life Instrument – Proxy Version (DEMQOL-Proxy). Since each symptomatic domain contained in the CICR-HI is independently validated, the CICR-HI enables testing of therapeutic interventions in targeted symptomatic issues. As such, the CICR-HI is relevant for the assessment of specific symptom management therapies and multi-symptomatic disease-modifying therapies.
Individuals with cognitive impairment increasingly rely on their family and caregivers as their condition progresses. Previous literature indicates that caregivers are ideally suited to observe the health of their loved one and are capable of reporting on observable symptoms such as fatigue, mobility, and activity participation, all of which have been identified by individuals with cognitive impairment as being symptoms of great importance. As an individual with cognitive impairment progresses, their ability to read and reliably complete a patient-reported outcome measure may decline. As such, the CICR-HI provides a much-needed option to measure small but meaningful changes in a patient’s health through the direct observation of those who are engaged and optimally positioned to make this determination.
In order to advance therapeutic development for individuals with cognitive impairment, there is a need for an outcome measure capable of tracking disease burden throughout the progression of their disease. Disease progression from MCI to dementia can occur in as few as 3-5 years for 10-15% of the Alzheimer’s population. The CICR-HI will satisfy a key recommendation by the FDA with its ability to comprehensively measure disease burden early and consistently throughout disease progression.
The CICR-HI was developed by using the same methodology that has been used by our outcomes group to develop caregiver and patient-reported outcome measures for over 30 diseases. These outcome measures have been translated and culturally validated for use in over 30 different countries, and have been used extensively in therapeutic trials worldwide. Collectively, they have been found to detect meaningful changes in health during therapeutic evaluation. The CICR-HI addresses a potential gap in clinical trial infrastructure for those who suffer from mild, moderate, or severe cognitive impairment. While prior studies have demonstrated the sensitivity of our instruments to detect clinical change over time, additional studies will be needed to further evaluate the sensitivity of the CICR-HI and each of its subscales in response to a specific treatment and over a longer period of time.
We acknowledge several limitations with this study. The demographic characteristics of the caregiver participants throughout this study were predominately white non-Hispanic individuals in the United States. Requirements to participate also included an English-language proficiency. Due to this, the sampled cohort is likely not a perfect representation of all caregivers of individuals with cognitive impairment. Additionally, the nature of this study was remote, requiring internet or phone to access recruitment and study activities, likely limiting the inclusion of participants without access to these devices.
Conclusion
The CICR-HI is a comprehensive caregiver-reported outcome measure, developed and validated in accordance with FDA guidelines, and capable of measuring multifactorial levels of disease burden of individuals with cognitive impairment. Our study demonstrated reliability, relevance, consistency, and usability of the CICR-HI for measuring disease burden across the Alzheimer’s continuum. The CICR-HI importantly enables individuals, who are unable to self-report due to advanced stage of disease, the opportunity to participate in clinical trials and advance therapeutic development.
Conflicts of Interest
C. Shupe has no disclosures to report. J. Seabury has no disclosures to report. J. Weinstein has no disclosures to report. C. Engebrecht has no disclosures to report. C. Irwin has no disclosures to report. P. Kanagaiah has no disclosures to report. A. Varma has no disclosures to report. S. Rosero has no disclosures to report. N. Dilek has no disclosures to report. A. Arky has no disclosures to report. J. Heatwole has no disclosures to report. E. Santos has no disclosures to report. C. Heatwole receives royalties for the use of multiple disease specific instruments. He has provided consultation to Biogen Idec, Ionis Pharmaceuticals, aTyr Pharma, AMO Pharma, Acceleron Pharma, Cytokinetics, Expansion Therapeutics, Harmony Biosciences, Regeneron Pharmaceuticals, Astellas Pharmaceuticals, AveXis, Recursion Pharmaceuticals, IRIS Medicine, Inc., Takeda Pharmaceutical Company, Scholar Rock, Avidity Biosciences, Novartis Pharmaceuticals Corporation, SwanBio Therapeutics, Neurocrine Biosciences, Sanofi, Lupin Pharmaceuticals, Dyne Therapeutics, Vertex, and the Marigold Foundation. He receives grant support from the Department of Defense, Duchenne UK, Parent Project Muscular Dystrophy, Recursion Pharmaceuticals, Swan Bio Therapeutics, Sanofi, Lupin Pharmaceuticals, the National Institute of Neurological Disorders and Stroke, the Muscular Dystrophy Association, the Friedreich’s Ataxia Research Alliance, Cure Spinal Muscular Atrophy, the Amyotrophic Lateral Sclerosis Association, the University of Miami, Novartis Pharmaceuticals Corporation, and the Michael J. Fox Foundation. He is the director of the University of Rochester’s Center for Health and Technology.
Acknowledgements
This research was conducted in collaboration with the Banner Health Institute Alzheimer’s Prevention Registry. Additional information regarding the CICR-HI, our other regulatory outcome measures, and licensing opportunities, can be found at www.healthindexes.com. The authors would like to thank the participants for their generosity in sharing their time and experiences for this study.
References
- Kim CK, Lee YR, Ong L, Gold M, Kalali A, Sarkar J. Alzheimer’s Disease: Key Insights from Two Decades of Clinical Trial Failures. Journal of Alzheimer’s Disease. 2022;87(1):83-100. doi:10.3233/JAD-215699.
- Duara R, Barker W. Heterogeneity in Alzheimer’s Disease Diagnosis and Progression Rates: Implications for Therapeutic Trials. Neurotherapeutics. 2022;19(1):25. doi:10.1007/S13311-022-01185-Z.
- 2023 Alzheimer’s disease facts and figures. Alzheimer’s and Dementia. 2023;19(4):1598-1695. doi:10.1002/alz.13016.
- Gustavsson A, Norton N, Fast T, et al. Global estimates on the number of persons across the Alzheimer’s disease continuum. Alzheimer’s & Dementia. 2023;19(2):658-670. doi:10.1002/ALZ.12694.
- Langa KM. Cognitive Aging, Dementia, and the Future of an Aging Population. In: Majmundar MK, Hayward MD, eds. Future Directions for the Demography of Aging. National Academies Press; 2018. doi:10.17226/25064.
- US Food and Drug Administration. Early Alzheimer’s Disease: Developing Drugs for Treatment Guidance for Industr.; 2024. https://www.fda.gov/vaccines-blood-biologics/guidance-compliance-regulatory-information-biologics/biologics-guidances.
- Hunter P. The controversy around anti-amyloid antibodies for treating Alzheimer’s disease: The European Medical Agency’s ruling against the latest anti-amyloid drugs highlights the ongoing debate about their safety and efficacy. EMBO Rep. 2024;25(12):5231. doi:10.1038/S44319-024-00294-4.
- Saunders S, Muniz-Terrera G, Watson J, et al. Participant outcomes and preferences in Alzheimer’s disease clinical trials: The electronic Person-Specific Outcome Measure (ePSOM) development program. Alzheimer’s & Dementia: Translational Research & Clinical Interventions. 2018;4:702. doi:10.1016/J.TRCI.2018.10.013.
- Stoeckel LE, Fazio EM, Hardy KK, Kidwiler N, McLinden KA, Williams B. Clinically meaningful outcomes in Alzheimer’s disease and Alzheimer’s disease related dementias trials. Alzheimer’s & Dementia: Translational Research & Clinical Interventions. 2025;11(1):e70058. doi:10.1002/TRC2.70058.
- Barrios PG, González RP, Hanna SM, et al. Priority of Treatment Outcomes for Caregivers and Patients with Mild Cognitive Impairment: Preliminary Analyses. Neurol Ther. 2016;5(2):183-192. doi:10.1007/s40120-016-0049-1.
- European Medicines Agency. Revised guideline on clinical studies for Alzheimer’s disease medicines | European Medicines Agency (EMA). February 28, 2018. Accessed May 11, 2025. https://www.ema.europa.eu/en/news/revised-guideline-clinical-studies-alzheimers-disease-medicines.
- Frank L, Lenderking WR, Howard K, Cantillon M. Patient self-report for evaluating mild cognitive impairment and prodromal Alzheimer’s disease. Alzheimers Res Ther. 2011;3(6):35. doi:10.1186/ALZRT97.
- Seabury J, Weinstein J, Varma A, et al. Patient and Caregiver-Reported Impact of Symptoms in Alzheimer’s Disease, Mild Cognitive Impairment and Dementia. Neurol Clin Pract. 2025;15(2).
- Varma A, Weinstein J, Seabury J, et al. The Crohn’s Disease-Health Index: Development and Evaluation of a Novel Outcome Measure. J Clin Gastroenterol. Published online January 26, 2024. doi:10.1097/MCG.0000000000001976.
- Varma A, Weinstein J, Seabury J, et al. The Facioscapulohumeral Muscular Dystrophy-Health Index: Development and evaluation of a disease-specific outcome measure. Muscle Nerve. 2023;68(4):422-431. doi:10.1002/MUS.27951.
- Brumfield OS, Zizzi CE, Dilek N, et al. The Huntington’s Disease Health Index: Initial Evaluation of a Disease-Specific Patient Reported Outcome Measure. J Huntingtons Dis. 2022;11(2):217-226. doi:10.3233/JHD-210506.
- Varma A, Weinstein J, Seabury J, et al. The amyotrophic lateral sclerosis-health index (ALS-HI): development and evaluation of a novel outcome measure. Amyotroph Lateral Scler Frontotemporal Degener. 2023;24(5-6):514-522. doi:10.1080/21678421.2023.2204871.
- Seabury J, Rosero S, Varma A, et al. Friedreich’s Ataxia-Health Index: Development and Validation of a Novel Disease-Specific Patient-Reported Outcome Measure. Neurol Clin Pract. 2023;13(5). doi:10.1212/CPJ.0000000000200180.
- Seabury J, Varma A, Weinstein J, et al. Friedreich Ataxia Caregiver-Reported Health Index. Neurol Clin Pract. 2024;14(3). doi:10.1212/CPJ.0000000000200300.
- Heatwole C, Bode R, Johnson N, et al. Myotonic Dystrophy Health Index: initial evaluation of a disease-specific outcome measure. Muscle Nerve. 2014;49(6):906-914. doi:10.1002/MUS.24097.
- Zizzi CE, Luebbe E, Mongiovi P, et al. The Spinal Muscular Atrophy Health Index: A novel outcome for measuring how a patient feels and functions. Muscle Nerve. 2021;63(6):837-844. doi:10.1002/MUS.27223.
- Heatwole C, Bode R, Johnson NE, et al. Myotonic dystrophy health index: Correlations with clinical tests and patient function. Muscle Nerve. 2016;53(2):183-190. doi:10.1002/MUS.24725.
- Stouffer JA, Bates K, Thacker LR, Heatwole C, Johnson NE. The Limb Girdle Muscular Dystrophy Health Index (LGMD-HI). Neuromuscul Disord. 2024;39:48-53. doi:10.1016/J.NMD.2024.04.008.
- Johnson NE, Heatwole C, Creigh P, et al. The Charcot Marie Tooth Health Index: Evaluation of a Patient Reported Outcome. Ann Neurol. 2018;84(2):225. doi:10.1002/ANA.25282.
- CHeT Outcomes – Our Expertise – Center for Health + Technology (CHeT) – University of Rochester Medical Center. Accessed March 24, 2025. https://www.urmc.rochester.edu/chet/our-expertise/outcomes.
- Qualification Process for Drug Development Tools: Guidance for Industry and FDA Staff.; 2020.
- Bronnick K, Breitve MH, Rongve A, Aarsland D. Neurocognitive Deficits Distinguishing Mild Dementia with Lewy Bodies from Mild Alzheimer’s Disease are Associated with Parkinsonism. Journal of Alzheimer’s Disease. 2016;53(4):1277-1285. doi:10.3233/JAD-160294.
- Karantzoulis S, Galvin JE. Distinguishing Alzheimer’s disease from other major forms of dementia. Expert Rev Neurother. 2011;11(11):1579. doi:10.1586/ERN.11.155.
- Sokolovič L, Hofmann MJ, Mohammad N, et al. Neuropsychological differential diagnosis of Alzheimer’s disease and vascular dementia: a systematic review with meta-regressions. Front Aging Neurosci. 2023;15:1267434. doi:10.3389/FNAGI.2023.1267434.
- Ohayon MM, Vecchierini MF. Daytime Sleepiness and Cognitive Impairment in the Elderly Population. Arch Intern Med. 2002;162(2):201-208. doi:10.1001/ARCHINTE.162.2.201.
- Harvey PD, Moriarty PJ, Kleinman L, et al. The Validation of a Caregiver Assessment of Dementia The Dementia Severity Scale. http://journals.lww.com/alzheimerjournal.
- Chua KC, Brown A, Little R, et al. Quality-of-life assessment in dementia: the use of DEMQOL and DEMQOL-Proxy total scores. Quality of Life Research. 2016;25(12):3107. doi:10.1007/S11136-016-1343-1.
- McPherson CJ, Addington-Hall JM. Judging the quality of care at the end of life: can proxies provide reliable information? Soc Sci Med. 2003;56(1):95-109. doi:10.1016/S0277-9536(02)00011-4.
- McGrattan AM, Pakpahan E, Siervo M, et al. Risk of conversion from mild cognitive impairment to dementia in low- and middle-income countries: A systematic review and meta-analysis. Alzheimer’s & Dementia: Translational Research & Clinical Interventions. 2022;8(1):e12267. doi:10.1002/TRC2.12267.
- Mitchell AJ, Shiri-Feshki M. Rate of progression of mild cognitive impairment to dementia – meta-analysis of 41 robust inception cohort studies. Acta Psychiatr Scand. 2009;119(4):252-265. doi:10.1111/J.1600-0447.2008.01326.X.
- Ward A, Tardiff S, Dye C, Arrighi HM. Rate of Conversion from Prodromal Alzheimer’s Disease to Alzheimer’s Dementia: A Systematic Review of the Literature. Dement Geriatr Cogn Dis Extra. 2013;3(1):320. doi:10.1159/000354370.
- Rajan KB, Weuve J, Barnes LL, McAninch EA, Wilson RS, Evans DA. Population estimate of people with clinical Alzheimer’s disease and mild cognitive impairment in the United States (2020–2060). Alzheimer’s & Dementia. 2021;17(12):1966-1975. doi:10.1002/ALZ.12362.
- Zaidman C, Proud C, Thronhoff J, et al. Six Month Results of Spinraza in Adults with Spinal Muscular Atrophy (SAS) Study. American Academy of Neurology. 2024. Accessed July 23, 2024. https://www.aan.com/MSA/Public/Events/AbstractDetails/47935.
- Heatwole C, Luebbe E, Hamel J, et al. Results from a 36-Week Open-Label Study of Recombinant Human Growth Hormone and Testosterone in Facioscapulohumeral Muscular Dystrophy (FSHD) (S48.009). Neurology. 2023;100(17_supplement_2). doi:10.1212/WNL.0000000000202958.
- Stimming EF, Claassen DO, Kayson E, et al. Safety and efficacy of valbenazine for the treatment of chorea associated with Huntington’s disease (KINECT-HD): a phase 3, randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2023;22(6):494-504. doi:10.1016/S1474-4422(23)00127-8.
- Dyne Therapeutics Announces Positive Initial Clinical Data from ACHIEVE Trial in DM1 Patients and DELIVER Trial in DMD Patients Demonstrating Promise of the FORCETM Platform in Developing Therapeutics for Rare Muscle Diseases. Published online January 3, 2024.
- Applied Therapeutics Announces Positive Results from 12-month Interim Analysis of Govorestat (AT-007) in the Ongoing INSPIRE Phase 3 Trial in Sorbitol Dehydrogenase (SORD) Deficiency. Appl Ther. Published online February 15, 2024.
APPENDIX
Appendix 1: Participant Demographic Characteristics
| Total Participants | Primary Interviews | Cross Sectional Study | Beta Interviews | Test Retest Reliability |
|---|---|---|---|---|
| 15 | 329 | 12 | 30 |
| Caregiver Demographics | Age (years) | Mean (SD) | Range | Sex, n (%) | Female | Male | Race, n (%) | American Indian/ Alaska Native | Asian | Black | White | Hawaiian/Pacific Islander | Other | Relationship to Individual with Cognitive Impairment, n (%) | Spouse | Son/Daughter | Home Health Aide | Other Relative | Other | Caregiver Ethnicity, n (%) | Hispanic/Latino | States represented, n |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Caregiver | Mean (SD) | 67.7 (11.1) | 45-85 | n (%) | 10 (66.7) | 5 (33.3) | n (%) | – | – | 2 (13.3) | 13 (86.7) | – | – | n (%) | 12 (80.0) | 2 (13.3) | 1 (6.7) | – | – | n (%) | – | 5 |
Appendix 2: Known Groups Analysis of CICR-HI Scores by Subgroups (n=329)
| Total Score | Short Form | Memory | Cognition | Social Satisfaction | Activity Participation | Communication | Fatigue | Sleep or Daytime Sleepiness | Emotional Health | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Patient Age | 80.1 years and younger | 52.4 | 53.6 | 62.7 | 64.6 | 50.0 | 47.5 | 52.7 | 45.3 | 36.4 | 48.1 |
| Older than 80.1 years | 61.9 | 64.0 | 72.2 | 70.8 | 62.3 | 61.3 | 55.8 | 59.8 | 49.4 | 51.3 | |
| p value | 0.002* | 0.0006* | 0.005* | 0.108 | 0.0014* | 0.0002* | 0.453 | <.0001* | <.0001* | 0.323 |

