Colorectal Cancer Detection Act: Impact on Health Equity
The Colorectal Cancer Detection Act of 2023: Benefits and Concerns from a Health Policy Perspective
Michael J Farber1, Sreejan Saha1, Karthik Madhira1, Steven W. Tseng1, Jonathan Kim1, Sonika Rathi1, Arthur A. Klein2, Joerg R. Leheste1
- Department of Biomedical Sciences, New York Institute of Technology College of Osteopathic Medicine (NYITCOM), Old Westbury, New York, 11568, USA
- Office of the Dean, NYITCOM, Old Westbury, New York, 11568, USA
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Farber, MJ., Saha, S., et al., 2024. The Colorectal Cancer Detection Act of 2023: Benefits and Concerns from a Health Policy Perspective. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5754
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v12i10.5754
ISSN 2375-1924
ABSTRACT
Colorectal cancer (CRC) is a major public health concern in the United States, accounting for the second highest number of cancer-related deaths after lung and bronchus cancer. Individuals with CRC having spread to other parts of the body face a dismal prognosis, with a 5-year survival rate of only 15%. However, early detection at a localized stage significantly improves the survival rate to 91%. Notably, African Americans are disproportionately affected by CRC, owing to lower compliance with screening measures. The COVID-19 pandemic exacerbated the situation further, resulting in approximately 9.4 million missed cancer screenings and an additional 4,500 annual deaths in the United States. The aim of this work is to bring attention to this health discrepancy with the explicit goal of introducing the Epi proColon blood-based CRC screening test as a feasible alternative to the current standard of care to close the existing racial gap through increased compliance. For this reason, the United States Congress has already suggested Medicare coverage for the Epi proColon blood-based CRC screening test in several bills and resolutions.
Keywords: colorectal cancer (CRC), cancer-related deaths, health disparities, screening compliance, Epi proColon, public health, legislative action, COVID-19 impact, missed cancer screenings, health policy, Medicare coverage, gastroenterology, public health
Introduction
Colorectal cancer (CRC) is a leading cause of cancer in the United States, with over 50,000 deaths expected annually. Individuals with CRC that has advanced to lymph nodes and other organs have a 5-year survival rate of 72% (regional stage), but this drops to 15% when the cancer spreads to distant bodily areas (distal stage). However, when detected early, the 5-year survival rate (localized stage) climbs to 91%, illustrating the importance of early detection and equity among various population. In times when the spiraling costs of American healthcare have become unsustainable, it is crucial to know that CRC care is the second most expensive, after only breast cancer, with an estimated cost of $24.3 billion annually. Regarding equity of care, it is concerning that African Americans have the highest CRC incidence and mortality rates, with 41.9 and 16.8 per 100,000, respectively, compared to 37 and 12.9 in Caucasians. Furthermore, men are more likely than women to develop CRC. In addition to being at a higher risk for CRC, African Americans’ low screening compliance rates exacerbate the problem. Notably, African Americans with a family history of CRC had lower screening rates than Caucasians without a history of the disease. Finally, significantly lower screening rates result in CRC diagnosis at a later stage, thereby increasing the mortality disparity between racial groups.
Currently, colonoscopy, sigmoidoscopy, computed tomography colonography, fecal immunochemical test (FIT), and stool DNA-FIT are considered the benchmark screening methods. The United States Preventive Services Task Force (USPSTF) issued new colorectal cancer (CRC) screening guidelines in 2021. Patients without a family history of colorectal cancer (CRC) or inflammatory bowel disease (IBD) should adhere to these guidelines. They are as follows: colonoscopy every ten years, sigmoidoscopy every five years, computed tomography colonography every five years, FIT test once a year, and stool DNA-FIT analysis every one to three years. As a preventative care screening technique that enables the removal of precancerous lesions, colonoscopy remains the gold standard for colorectal cancer screening.
While available screening techniques are effective in detecting CRC, they also present barriers especially for minority communities, exacerbating disparities in colorectal cancer incidence and mortality. Two inherent barriers to obtaining a colonoscopy are the procedure’s high cost and invasive nature. Currently, Medicare covers all screening colonoscopies, which cost $597 for inpatient procedures and $997 for outpatient procedures. However, 11% of black Americans are uninsured, versus 6% of white Americans. These numbers underscore a severe systemic failure of the U.S. American healthcare system and the urgent need to develop cost-effective and user-friendly screening methods resolving current inequities. Another set of barriers along the socioeconomic framework are the complexities associated with a full colonoscopy screening, which frequently necessitates prior arrangements to avoid scheduling conflicts with work or other essential activities such as childcare. Families that are financially struggling may not have the ability or means to factor in time-consuming screening appointments. The FIT and Stool DNA-FIT are presenting another barrier to screening. Despite their non-invasive nature, patients’ aversion to stool handling significantly reduces adherence to them. Given all of the limitations of current screening procedures, there is an obvious need for non-invasive and patient-friendly screening techniques.
The aim of this work is to draw attention to the critical issue of colorectal cancer (CRC) in the United States, emphasizing the importance of early detection and equitable access to screening. In this regard we are focusing on the significant disparities in CRC incidence and mortality rates among different racial groups, particularly African Americans, and underscores the need for cost-effective, non-invasive screening methods to overcome existing barriers. By examining current screening guidelines, the economic burden of CRC care, and the impact of the COVID-19 pandemic on screening rates, the purpose of this review centers on patient advocacy for policy changes that promote accessible and equitable CRC screening, ultimately aiming to reduce mortality rates and healthcare costs associated with this prevalent cancer.
In this context, the Epi ProColon test detects active colorectal cancer using a blood-based SEPTIN-9 (SEPT9) gene methylation assay that has been FDA-approved. This test’s sensitivity was determined to be 72%, and its specificity was 90%. Epi ProColon was chosen based on patient preferences for non-invasive colorectal screening methods, as well as its overall convenience and reduced time requirements. The COVID-19 pandemic had a detrimental effect on CRC screenings. It has been estimated that 3.8 million colorectal screenings were missed in 2020, leading to an additional 10,000 projected deaths. There has also been an increase in emergency room visits where patients are diagnosed with CRC in later stages. This new situation calls for a change in support of a new screening method able to overcome stigma and limitations. CRC has been debated by lawmakers since 2016 with the introduction of the Donald Payne Sr. Colorectal Cancer Detection Act of 2016 (H.R.6275) to the 114th U.S. Congress. The bill had not been signed into law and has been reintroduced into subsequent Congresses, including the current 118th by various sponsors. Donald M. Payne, Sr. was a United States congressman from 1989 to 2012 is an important historical figure. He was the first African American congressman to represent New Jersey; known for his efforts to promote human rights around the world. Rep. Payne, Sr. passed away from CRC in March 2012 and was survived by his son, congressman Donald M. Payne, Jr. Rep. Donald M. Payne Jr. (D-NJ) and Rep. Mark Green (R-TN) founded the CRC caucus with the goal of honoring Rep. Payne Sr’s legacy through pushing for increased screening opportunities and ultimately a cure for CRC.
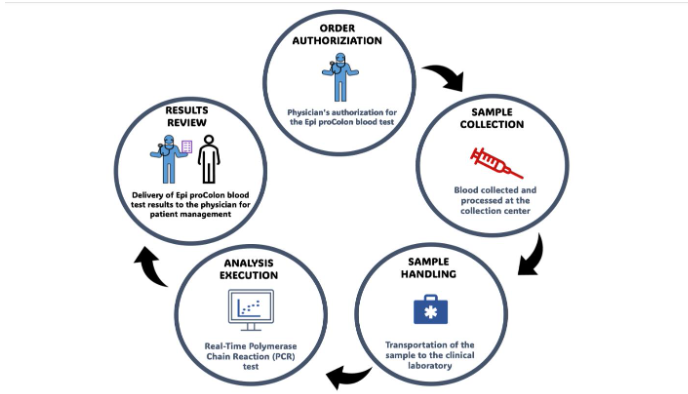
Review
REVIEW PURPOSE
Following the aim of drawing attention to a significant issue in health inequity, the purpose of this study is to determine the potential impact of the Epi proColon blood-based CRC screening test, which has been proposed for Medicare coverage, eliminate co-insurance requirements, and foster awareness education and early detection by various bills submitted to the United States Congress.: Nancy Gardner Sewell Medicare Multi-Cancer Early Detection Screening Coverage Act [H.R.2407; submitted 03-30-2023]; Medicare Multi-Cancer Early Detection Screening Coverage Act [S.2085; submitted 06-21-2023], Colorectal Cancer Act [S.892; submitted 03-21-2023], Colorectal Cancer Payment Fairness Act [H.R.3382; S.1894; respectively submitted 05-19-2023 & 06-08-2023], Colorectal Cancer Early Detection Act [H.R.7714; submitted 03-22-2024]. Examining the anticipated effects of this legislation on CRC-related mortality, especially in the African American community, can shed light on its potential effectiveness in promoting early detection and improving survival rates. This study intends to provide insights into the viability and efficacy of policy changes, as well as possible means of addressing differences in CRC outcomes between various populations particularly as a function of the recent COVID-19 pandemic and lapsed CRC screening.
REVIEW STRATEGY AND METHODOLOGY
This scoping review with health policy recommendation is based on the systematic analysis and interpretation of articles associated with the National Library of Medicine (PubMed), Google Scholar, Scopus (Elsevier), and other resources vetted by the principal investigator (PI), such as select foundations (e.g., Kaiser Family Foundation, Pew Research Center), government agencies (e.g., Library of Congress, U.S. Department of Justice, U.S. Food and Drug Administration, Centers for Disease Control and Prevention), and news agencies (e.g., CNN, The New York Times, The Wall Street Journal, Global Newswire). Article searches were conducted using the following search terms: “Colorectal cancer (CRC), CRC mortality, CRC screening methods, CRC screening standard of care, CRC cancer disparities, cancer-related deaths, health disparities, screening compliance, Epi proColon blood test, public health, legislative action, COVID-19 impact, missed cancer screenings, health policy, and Medicare coverage.” Articles and reports were chosen and assessed based on their overall relevance to the research topic, credibility, and significance. This project was conducted over a 12-month period.
THE COLORECTAL CANCER DETECTION ACT
In recent years, the U.S. Congress has increased its focus on colorectal cancer (CRC) and the health disparities associated with it. A significant development is the Colorectal Cancer Detection Act of 2023 [S.892, submitted 03-21-2023], introduced by Senator Martin Heinrich from New Mexico. This bill aims to amend Title XVIII of the Social Security Act to provide Medicare coverage for FDA-approved blood-based CRC screening tests, such as the Epi proColon. By doing so, it seeks to enhance screening participation, particularly in under-screened communities of color, and address the declines in colorectal cancer screening caused by the COVID-19 pandemic. The proposed legislation ensures that these tests fit within current screening guidelines, allowing reimbursement only when a patient is due for screening. Passing this bill would make qualifying blood-based tests accessible and reimbursable under Medicare.
PRINCIPAL STAKEHOLDERS SUPPORTING S.892
Primary supporters of this measure include patient advocacy groups that want to increase screening participation, particularly in communities of increased CRC risk, and address the decreases in colorectal cancer screening caused by the COVID-19 pandemic. Manufacturers of blood-based CRC screening technologies favor this bill because it will boost the use of their technology and thus revenue. Hospital systems embrace this bill because it not only gives them access to a larger variety of screening instruments at no cost, but it also improves patient outcomes. Primary care doctors, gastroenterologists, and oncologists, who are frequently responsible for CRC prevention and treatment in their patient populations, support this bill because it will make it easier to test for CRC in patients. This is especially true for those who are hesitant to undergo more intrusive treatments like stool collection and colonoscopy screening. Furthermore, doctors may screen any consenting patient without charge, as Medicare covers screening for all eligible patients. As a result, Medicare coverage for blood-based CRC preventative screening instruments, such as Epi proColon, will benefit both at-risk and general patient populations by increasing CRC awareness and prevention through the provision of a free, noninvasive screening approach.
PRINCIPAL STAKEHOLDERS OPPOSING S.892
The chief opponents of The Colorectal Cancer Detection Act of 2023 are Medicare and rival CRC screening manufacturers. S892 mandates that Medicare fund all blood-based CRC preventative screening technologies. As a result, if a practitioner and patient agree that blood-based screening is appropriate for the patient, Medicare is required to pay for the test. Because CRC is the second greatest cause of mortality in the U.S., this law will most likely redirect a significant amount of Medicare funding intended for other covered healthcare needs to CRC screening. As a result, Medicare representatives will most likely oppose this bill because it will necessitate budget restructuring, as previously stated, for a new screening method that is still being tested by many companies; however, it will most likely be favored by providers because it is the least invasive of the available screening methods today. Manufacturers of non-blood-based colorectal cancer screening instruments, such as FIT, will most likely oppose the passage of this bill because it will reduce their revenue. Because blood screening biomarkers can be added to any other standard screening, such as a lipid panel or CBC, clinicians are more likely to prescribe blood-based screens than stool or other screening modalities, especially if they are funded by Medicare.
PREDICTED INTENDED CONSEQUENCES
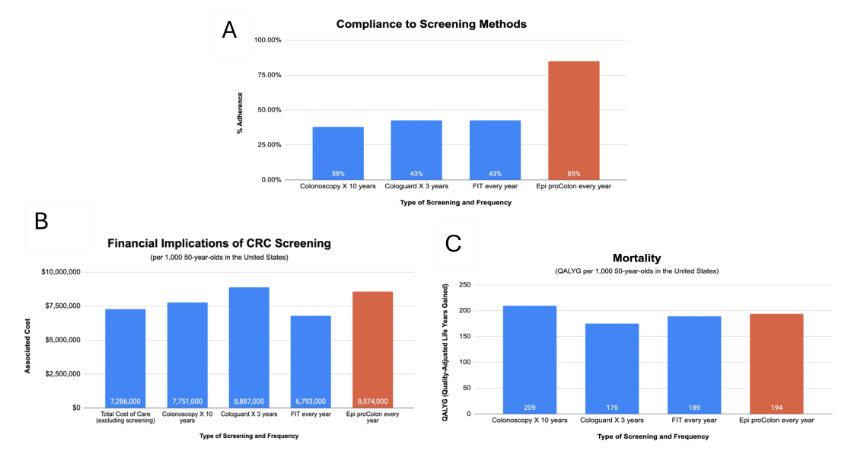
If signed into law, S.892 would provide Medicare coverage for FDA-approved blood-based CRC screening tests, such as Epi proColon, and amend title XVIII of the Social Security Act to increase participation in CRC screening in under-screened communities of color, to offset the COVID-19 pandemic-driven declines in CRC screening. Compared to other colorectal detection tools, such as stool-based tests such as the fecal immunochemical test (FIT) and Cologuard, Epi proColon is a blood-based plasma SEPT9 gene methylation assay that was approved by the US FDA in 2016 for use with average-risk individuals over 50 years of age. There are numerous advantages to the Epi proColon test but the most important one is its superior compliance rate. This feature plays an important role in the early detection of CRSs during regular screening because early detection and diagnosis of CRC in early stages, resulting to a greater 5-year survival rate of 91%. In terms of health economics, the epi proColon is the most cost-effective CRC screening test, with the most potential to enhance survival, as evaluated by quality adjusted life year gained (QALYG) per thousand persons. This assessment considers its lower cost of roughly $200 compared to Cologuard, which costs $500. This is critical for the uninsured since it makes screening more accessible. Thus, providing the Epi proColon under the Medicare program will relieve inequities in healthcare based on socioeconomic level and race, especially for the African American community, which has the highest incidence and mortality rate of colorectal cancer.
Consistent with this approach, the Cancer Moonshot was started in 2016 as part of the 21st Century Cures Act, with the goal of accelerating scientific discovery in cancer, increasing collaboration, and improving cancer data sharing. On September 12th, 2022, President Joe Biden emphasized the importance of the Cancer Moonshot by noting the remarkable progress that has been done since 2016. The National Cancer Institute (NCI) has financed more than 70 programs and 250 research projects. Furthermore, he provided an update on his new Cancer Moonshot Initiatives, such as identifying effective blood-based screening tests for early diagnosis of one or more cancers, which the NCI will conduct. As a result, in addition to the previously indicated benefits, passing the Colorectal Cancer Detection Act of 2023 aligns with President Joe Biden’s updated Cancer Moonshot objectives.
POTENTIALLY UNINTENDED CONSEQUENCES
As with any public health policy changes, there are other consequences to consider. Improved coverage of the Epi proColon test provides major benefits, particularly by allowing for early diagnosis of CRC in populations, potentially leading to significant improvements in outcomes for Black communities. However, passing this law will have unforeseen repercussions and subsequent solutions may need to follow. One key concern is that expanded medical coverage with Epi proColon may lead to a decrease in compliance with the gold standard colonoscopy screening procedure. In other words, those who are screened with Epi proColon may be less likely to have colonoscopy screening due to inconvenience, perceived redundancy, and cost. Considering the Epi proColon test has the lowest specificity and sensitivity of any known test, this could mean that it decreases the accuracy of CRC identification overall if disjointed from the gold standard. The impact on healthcare economics is significant. Individuals with false-negative results will remain undiagnosed, leading to higher costs for late-stage colon cancer care. Those with false positives will need costly colonoscopies. Both scenarios increase inconvenience for patients, who must take time off work for a colonoscopy or face the severe consequences of late-stage colorectal cancer. Additionally, the shortcomings of the Epi proColon test might discourage patients from opting for stool testing, which is a more accurate screening method. Another factor to examine is the financial ramifications of approving Epi proColon. This test costs more than its existing early screening cousin, FIT. As a result, the approval of Epi proColon could increase the overall economic burden on the healthcare system. As a result, a thorough review of its clearance includes determining the risk/benefit ratio of its application in the modern healthcare system. When compared to other CRC screening procedures, the Epi proColon test is not the most cost-effective option. According to certain research, the adoption of Epi proColon would be more costly than most other screening methods, with the exception of the Cologuard test. An important factor behind this is that the optimal screening interval has not been determined yet. This raises the question of whether Epi proColon will boost the accessibility and feasibility of screening sufficiently to justify the expense burden. How will insurers, healthcare institutions, and lobbyists evaluate these higher costs in terms of cost reduction? Furthermore, the question of whether accessibility will genuinely increase, particularly for communities at risk of a lack of CRC screening, remains. The argument behind this theory is that disparities in access to screening technologies may be compounded by differences in insurance coverage. In other words, passage of S.892 into law will benefit those who already have adequate insurance coverage. On the contrary, those who do not have insurance coverage, such as those who are at a disadvantage in screening, may not profit significantly from the passage of this legislation. Although the goal of this legislation is to promote accessibility and early detection of CRC, the unintended consequence may be to further stratify populations due to disparities in health care access. Finally, the data used to access the Epi proColon test was primarily model-based. This is because this test has not yet been approved by Medicare or other insurance providers, thus it cannot be utilized commercially. More funding and clinical trials are required to properly analyze how this test performs in comparison to other current tests. The Epi proColon is not a one-size-fits-all, straightforward solution to the unfortunate disparities that exist in early CRC diagnosis in today’s healthcare system. The debate over the possibility of unexpected consequences can go on for quite some time. The main conclusion is that, while Epi proColon has the potential to boost the feasibility and accessibility of early CRC detection, there are significant alternative consequences that are complicated in nature.
Conclusion
To determine whether to approve the blood-based colorectal cancer (CRC) screening tool Epi proColon, this health policy research analysis weighed the benefits and drawbacks considering The Colorectal Cancer Detection Act of 2023. Overall, the Epi ProColon’s high compliance and user-friendliness set it apart from competing testing methods. A key factor in its exceptional QALYG is its excellent compliance. It will not address important factors that contribute to screening discrepancies, like insurance access, and it is still more costly than the FIT. People who are eligible for colorectal cancer screening would greatly benefit from this bill’s passage since it would provide doctors more leeway to screen their patients. One possible negative aspect is that patients can mistakenly believe that the less precise Epi ProColon can stand in for a more precise screening procedure, which is not always the case. Those in the medical and pharmaceutical industries that benefit from non-traditional screening procedures would also be hurt by this. Rates are on the rise, particularly among younger patients, highlighting the need to address the problem of access to CRC screening.
References
- Centers for Medicare & Medicaid Services. National Coverage Analysis (NCA) for Colorectal Cancer Screening – Blood-Based Biomarker Tests (CAG-00454N). (2021). Accessed: 7-25-2024: https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx.
- Grützmann R., Molnar B., Pilarsky C., et al.: Sensitive detection of colorectal cancer in peripheral blood. PLOS ONE. 2008, 3:1-8. 10.1371/journal.pone.0003759
- American Cancer Society. Survival Rates for Colorectal Cancer. (2024). Accessed: 7-25-2024: https://www.cancer.org/cancer/types/colon-rectal-cancer/detection-diagnosis-staging/survival-rates.html.
- CDC. National Center for Chronic Disease Prevention and Health Promotion (NCCDPHP). Health and Economic Benefits of Colorectal Cancer Interventions.. (2024). Accessed: 7-25-2024: https://www.cdc.gov/nccdphp/priorities/colorectal-cancer.html.
- Carethers, J.M.: Racial and ethnic disparities in colorectal cancer incidence and mortality. Advances in Cancer Research, 151. 2021, 151:197-229. 10.1016/bs.acr.2021.02.007
- Gangireddy V. G. R., & Swathi M. D.: Gender disparities in the incidence of colorectal cancer in the era of screening colonoscopy. American Journal of Gastroenterology. 2018, 176:99-101.
- Laiyemo A. O., Thompson N., Williams C. D., et al.: Race and colorectal cancer screening compliance among persons with a family history of cancer. World Journal of Gastrointestinal Endoscopy. 2015, 7:1300-1305. 10.4253/wjge.v7.i18.1300
- Siegel R., DeSantis C., & Jemal A.: Colorectal cancer statistics. CA: A Cancer Journal for Clinicians. 2014, 64:104-117.
- Medicare.gov. Preventive & screening services. Colorectal cancer screenings. (2024). Accessed: 7-26-2024: https://www.medicare.gov/coverage/preventive-screening-services.
- KFF. Health Coverage by Race and Ethnicity, 2010-2022. Latoya Hill, Samantha Artiga, and Anthony Damico. (2024). Accessed: 7-26-2024: https://www.kff.org/racial-equity-and-health-policy/issue-brief/health-coverage-by-race-and-ethnicity/.
- Grego S., Welling C. M., Miller G. H., et al.: A hands-free stool sampling system for monitoring intestinal health and disease. Scientific Reports. 2022, 12:14803-14809. 10.1038/s41598-022-14803-9
- Adler A., Geiger S., Keil A., et al.: Improving compliance to colorectal cancer screening using blood and stool based tests in patients refusing screening colonoscopy in Germany. BMC Gastroenterology. 2014, 14:1-8. 10.1186/1471-230X-14-183
- Chen R. C., Haynes K., Du S., et al.: Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncology. 2021, 7:878-884. 10.1001/jamaoncol.2021.0884
- Mazidimoradi A., Hadavandsiri F., Momenimovahed Z., et al.: Impact of the COVID-19 pandemic on colorectal cancer diagnosis and treatment: A systematic review. Journal of Gastrointestinal Cancer. 2023, 54:1-11. 10.1007/s12029-021-00752-5
- CONGRESS.GOV. H.R.6275 – Donald Payne Sr. Colorectal Cancer Detection Act of 2016 114th Congress (2015-2016). (2016). Accessed: 7-25-2024: https://www.congress.gov/bill/114th-congress/house-bill/6275.
- CONGRESS.GOV. H.R.1765 – Donald Payne Sr. Colorectal Cancer Detection Act of 2019. 116th Congress (2019-2020). (2019). Accessed: 7-25-2024: https://www.congress.gov/bill/116th-congress/house-bill/1765.
- CONGRESS.GOV. S.2149 – Donald Payne, Sr., Colorectal Cancer Detection Act of 2021. 117th Congress (2021-2022). (2021). Accessed: 7-26-2024: https://www.congress.gov/bill/116th-congress/house-bill/1765.
- CONGRESS.GOV. S.892 – Colorectal Cancer Detection Act. 118th Congress (2023-2024). (2023). Accessed: 7-26-2024: https://www.congress.gov/bill/116th-congress/house-bill/1765.
- Biographical Directory of the United States Congress 1774 – Present. PAYNE, Donald Milford (1934-2012). (2012). Accessed: 7-26-2024: https://bioguideretro.congress.gov/Home/MemberDetails?memIndex=P000149.
- CLERK. United States House of Representatives. Office of the Tenth Congressional District of New Jersey. (2024). Accessed: 7-26-2024: https://payne.house.gov/.
- CONGRESS.GOV. S.1894 – Colorectal Cancer Payment Fairness Act. 118th Congress (2023-2024). (2023). Accessed: 7-26-2024: https://www.congress.gov/bill/118th-congress/senate-bill/1894.
- CONGRESS.GOV. H.R.7714 – Colorectal Cancer Early Detection Act 118th Congress (2023-2024). (2024). Accessed: 7-26-2024: https://www.congress.gov/bill/118th-congress/house-bill/7714.
- CONGRESS.GOV. H.R.2407 – Nancy Gardner Sewell Medicare Multi-Cancer Early Detection Screening Coverage Act. 118th Congress (2023-2024). (2024). Accessed: 7-26-2024: https://www.congress.gov/bill/118th-congress/house-bill/2407.
- CONGRESS.GOV. S.2085 – Medicare Multi-Cancer Early Detection Screening Coverage Act 118th Congress (2023-2024). (2023). Accessed: 7-26-2024: https://www.congress.gov/bill/118th-congress/senate-bill/2085.
- CONGRESS.GOV. https://www.congress.gov/bill/117th-congress/house-bill/1655. (2023). Accessed: 7-26-2024: https://www.congress.gov/bill/117th-congress/house-bill/1655.
- Song L. L., & Li Y.: Current noninvasive tests for colorectal cancer screening: An overview of colorectal cancer screening tests. World Journal of Gastrointestinal Oncology. 2016, 8:793-800. 10.4251/wjgo.v8.i11.793
- D’Andrea E., Ahnen D. J., Sussman D. A., et al.: Quantifying the impact of adherence to screening strategies on colorectal cancer incidence and mortality. Cancer Medicine. 2020, 9:2735-2743. 10.1002/cam4.2735
- Peterse E. F. P., Meester R. G. S., de Jonge L.: Comparing the cost-effectiveness of innovative colorectal cancer screening tests. JNCI: Journal of the National Cancer Institute. 221, 113:154-162. 10.1093/jnci/djaa103
- THE WHITE HOUSE. FACT SHEET: President Biden Details Cancer Moonshot Progress and New Initiatives on 60th Anniversary of President Kennedy’s Moonshot Address. (2022). Accessed: 7-26-2024: https://www.whitehouse.gov/briefing-room/statements-releases/2022/09/12/fact-sheet-president-biden-details-cancer-moo….
- Lin KW. :mSEPT9 (Epi proColon) Blood Test for Colorectal Cancer Screening. American Family Physician. 2019, 100:10-11

