Combination Therapy for T2DM: Dapagliflozin and Vildagliptin
DApagliflozin 10 mg and Vildagliptin 100 mg SR use in Cardiology Practice: A real world Clinical Insight (DAa-ViNCI Study)
JC Mohan¹, Mahesh K Shah², Smit Shrivastava³, Sona Warrier⁴
- Chairman and HOD, Department of Cardiology, Jaipur Golden Hospital, Sector 3, Rohini, Delhi-62
- Interventional Cardiologist, MK’s Heart Care Clinic, Mumbai
- Professor & Head, Cardiology at Advanced Cardiac Institute, Medical College Raipur
- Scientific Services, USV Pvt Ltd, Mumbai
OPEN ACCESS
PUBLISHED: 31 May 2025
CITATION: Mohan, J.C., et al., 2025. DApagliflozin 10 mg and Vildagliptin 100 mg SR use in Cardiology Practice A real world Clinical Insight (DAa-ViNCI Study). Medical Research Archives, [online] 13(5).
https://doi.org/10.18103/mra.v13i6.4449
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i6.4449
ISSN 2375-1924
ABSTRACT
Background: Diabetes mellitus is a global epidemic characterized by persistently high blood sugar levels. The combination of dipeptidyl peptidase-4 inhibitors (DPP-4i) and sodium-glucose cotransporter-2 inhibitors (SGLT2i) is potentially effective in managing type 2 diabetes mellitus (T2DM). The present study analyzed the efficacy, safety, and practice patterns of the fixed-dose combination (FDC) of vildagliptin 100 mg and dapagliflozin 10 mg in cardiology practice.
Methods: This retrospective, non-randomized, non-comparative, multi-center study was conducted at 200 sites across India from June 2023 to March 2024. Patients of either sex, ≥18 years of age, diagnosed with T2DM, and who received the FDC of vildagliptin 100 mg SR and dapagliflozin 10 mg for the treatment of their condition were included. Comprehensive patient information collected from the medical records were analyzed.
Results: A total of 2,199 patients were included, with a mean age of 55.04 years. Approximately 63% of the patients were male. Hypertension (74.81%) was the most common comorbidity, followed by dyslipidemia (38.52%). The FDC of vildagliptin 100 mg SR and dapagliflozin 10 mg reduced the mean glycated hemoglobin (7.7% vs 5.8%), fasting plasma glucose (142.4 mg/dL vs 90.2 mg/dL), postprandial glucose (205.64 mg/dL vs 123.38 mg/dL) levels from baseline to 3 months. The mean systolic blood pressure decreased from 141.35 mmHg to 131.59 mmHg, and the mean diastolic blood pressure decreased from 89.65 mmHg to 84.42 mmHg following FDC therapy. Glycemic goals were achieved in 78.08% of patients. Adverse events were reported in 0.36% of patients.
Conclusion: The FDC of vildagliptin and dapagliflozin demonstrated substantial efficacy in managing T2DM and was well-tolerated, with a low incidence of adverse events.
Keywords: Adverse events, comorbidities, efficacy, fixed-dose combination, glycemic control, tolerability, type 2 diabetes mellitus.
Introduction
Diabetes mellitus is a global epidemic characterized by persistently high blood sugar levels.¹ The global prevalence of diabetes was 536.6 million (10.5%) in 2021, and it is projected to reach 783.2 million (12.2%) by 2045.²
Type 2 diabetes mellitus (T2DM) is associated with several physiological abnormalities collectively known as the “ominous octet.” These include increased hepatic glucose synthesis, decreased insulin sensitivity, increased insulin secretion, increased lipolysis, increased glucagon secretion, increased renal glucose reabsorption, and dysregulated neurotransmitter modulation.³ However, diabetes is no longer a standalone metabolic disorder—it is intricately linked with cardiovascular (CV) complications from the very onset. Patients with diabetes often present with early CV risk factors such as hypertension and dyslipidemia, setting the stage for progressive vascular damage. The traditional approach of addressing cardiovascular risks only after overt complications emerge is no longer viable. Instead, a paradigm shift towards early, comprehensive intervention is crucial.⁴
Managing diabetes with a focus on early CV risks can significantly alter disease trajectories. Evidence suggests that timely glycemic control combined with CV risk reduction strategies can prevent major adverse cardiovascular events (MACE) and improve long-term outcomes.⁴ The role of novel therapeutic options, such as Fixed-Dose Combinations (FDCs), that address both hyperglycemia and CV risk factors, is becoming increasingly important in this strategy.
In patients with T2DM, fixed-dose combinations (FDC) have proven to be more effective than their individual components.⁵ When taking oral antidiabetic medications in combination, 58% of participants in an Indian study chose FDC.⁶ The combination of dipeptidyl peptidase-4 (DPP-4) and sodium-glucose cotransporter-2 (SGLT2) inhibitors is potentially effective.⁷ Sodium-glucose cotransporter-2 inhibitors lower blood sugar levels by increasing glucose excretion through urine, without impacting insulin secretion or action. Dipeptidyl peptidase-4 inhibitors improve glucose control by inhibiting the degradation of active incretin hormones, which boosts insulin secretion and reduces glucagon levels.⁸ Together, these medications address at least six of the eight elements of the “ominous octet”.
Among DPP-4 inhibitors, vildagliptin has been extensively researched for its clinical benefits. Its efficacy is noted for its minimal risk of hypoglycemia, lack of weight gain, and no increased risk of cardiovascular events. Both clinical trials and real-world evidence suggest that vildagliptin has a safety and tolerability profile comparable to that of a placebo.⁹
Dapagliflozin, a highly potent and reversible SGLT2 inhibitor, is the main transporter for glucose absorption in the gut. It effectively controls blood sugar levels and reduces blood pressure and body weight, whether used alone or in combination with other antihyperglycemic drugs. Dapagliflozin has attracted attention for its benefits in patients with established atherosclerotic cardiovascular disease or multiple cardiovascular risk factors, offering glycemic control, cardioprotection, and potentially renal protection, while generally maintaining a favorable tolerability profile.¹⁰,¹¹
The complementary mechanisms of action of these two drug classes render them suitable treatment options for combination therapy with any glucose-lowering agents, including insulin.¹² However, there is a scarcity of data evaluating the efficacy, safety, and tolerability of the combination of vildagliptin and dapagliflozin in patients with T2DM within the Indian settings. Therefore, this real-world study was conducted to analyze the efficacy, safety, and practice patterns of the FDC of vildagliptin 100 mg and dapagliflozin 10 mg in cardiology practice.
Materials and methods
STUDY DESIGN AND ETHICAL CONSIDERATION
This retrospective, non-randomized, non-comparative, multi-center study was conducted at 200 sites across India from June 2023 to March 2024. The study was
conducted in compliance with the principles of the Declaration of Helsinki. The study protocol received approval from the ACEAS Independent Ethics Committee (Registration no. ECR/281/Indt/GJ/2017).
INCLUSION CRITERIA AND EXCLUSION CRITERIA
Patients of either sex, above 18 years of age, diagnosed with T2DM, and who received the FDC of vildagliptin 100 mg and dapagliflozin 10 mg for the treatment of their condition were included in the study. Additionally, the inclusion criteria required that the treating physician agreed to provide information regarding the participant’s treatment. Patients with incomplete data files or with any condition not suitable for inclusion, as indicated by the investigator’s discretion were excluded from the study.
SAMPLE SIZE
The estimated sample size was approximately 4,000 patients with T2DM.
DATA COLLECTION
Comprehensive patient information, including demographic characteristics, treatment duration, comorbidities, concomitant medications, and adverse events was collected from medical records authenticated by the treating physicians. The collected data were then entered into the case report forms and analyzed.
STUDY ENDPOINTS
The endpoint of the study was to determine the number and percentage of patients receiving the FDC of vildagliptin 100 mg and dapagliflozin 10 mg; the number and percentage of patients of various age groups receiving the FDC of vildagliptin 100 mg and dapagliflozin 10 mg; the duration of FDC of vildagliptin 100 mg and dapagliflozin 10 mg therapy; the duration of comorbidities; compliance to FDC of vildagliptin 100 mg and dapagliflozin 10 mg therapy; adverse events reported in last 1-year related to FDC therapy; efficacy and safety parameters including quality of life (adherence and lack of side effects).
STATISTICAL ANALYSIS
Data were analyzed using the Statistical Package for Social Sciences (SPSS) software, version 28. Descriptive analysis was used to present the study outcomes. Continuous variables were described as mean and standard deviation (SD), whereas categorical variables were described as numbers and percentages.
Results
A total of 2,199 patients were included in the study, with a mean age of 55.04 years. The majority of the patients (62.62%) were male. Among the study population, 62.94% of patients were in the 40–60 years age group, followed by 27.47% in the 60–80 years age group. The mean systolic blood pressure (SBP) of the patients was 139.31 mmHg, and the mean diastolic blood pressure (DBP) was 88.15 mmHg. The mean (SD) duration of treatment with the FDC of vildagliptin 100 mg and dapagliflozin 10 mg was 8.07 (3.0) months. Hypertension (74.81%) was the most common comorbidity, followed by dyslipidemia (38.52%), coronary artery disease (CAD, 14.60%), and stroke/transient ischemic attack (TIA, 5.23%). The mean (SD) durations of hypertension and dyslipidemia were 44.85 (39.04) and 40.49 (35.00) months, respectively (Table 1).
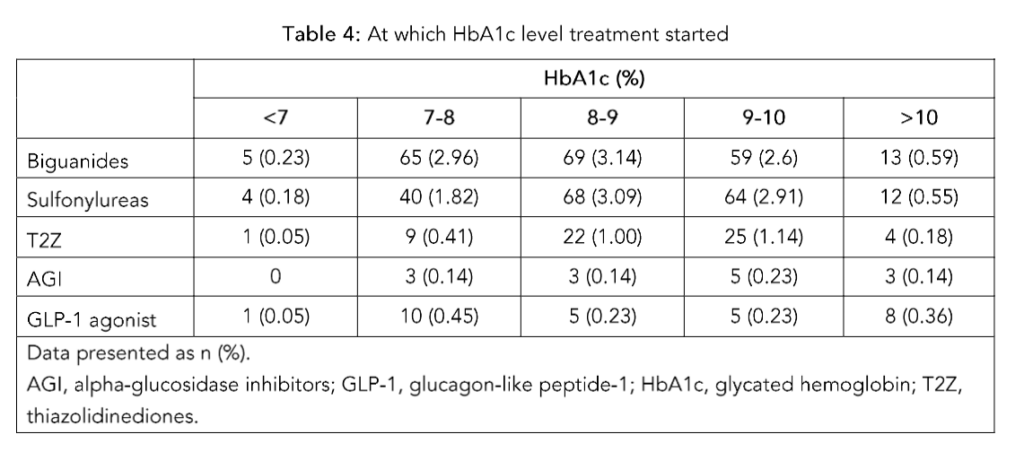
Glycemic parameters of the patients from baseline to 3 months are presented in table 2. The mean (SD) HbA1c level decreased from 7.7% (3.1) at baseline to 5.8% (2.88) at 3 months. Similarly, the mean (SD) fasting plasma glucose (FPG) level decreased from 142.4 (88.4) mg/dL at baseline to 90.2 (62.44) mg/dL at 3 months; the mean (SD) postprandial glucose (PPG) decreased from 205.64 (128.0) mg/dL at baseline to 123.38 (89.15) mg/dL at 3 months.
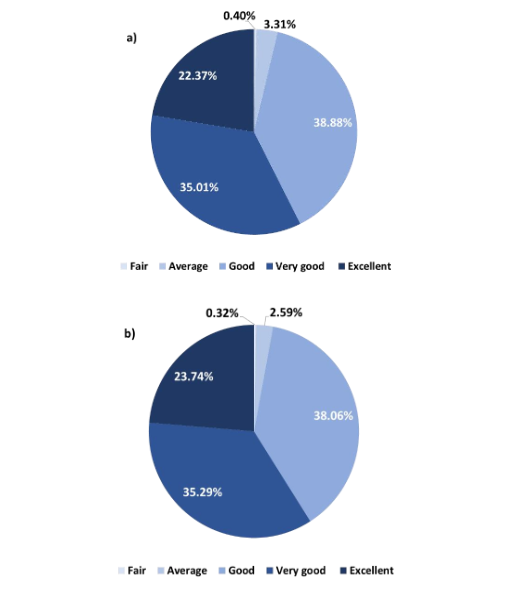
Physician’s global assessments are illustrated in Figure 1a and 1b. About 38.88% of physicians reported that the FDC of vildagliptin 100 mg and dapagliflozin 10 mg showed good tolerability, while 35.01% reported very good tolerability. Additionally, 38.06% of physicians observed good efficacy, while 35.29% reported very good efficacy.
| Parameters | N=2199 |
|---|---|
| Age (years) | 55.04 (10.60) |
| Age groups (years) | |
| <40 | 137 (6.23) |
| 40-60 | 1384 (62.47) |
| >60 | 678 (30.80) |
| Gender, n (%) | |
| Male | 1377 (62.62) |
| Female | 822 (37.38) |
| Weight (kg) | 75.53 (11.21) |
| BMI (kg/m²) | 29.43 (4.20) |
| SBF (mmHg) | 139.51 (21.23) |
| Duration of T2DM (years) | 8.8 (4.25) |
| Duration of treatment with the FDC of vildagliptin 100 mg and dapagliflozin 10 mg | 8.07 (3.80) |
Glycemic parameters from baseline to 3 months
| HbA1c (%) | N=2189 | Cholesterol | N=2187 | PPG (mg/dL) | N=2174 |
|---|---|---|---|---|---|
| Baseline | 7.7 (0.13) | 75.7 (7.83) | 142.4 (8.44) | 138.74 (16.23) | |
| 1st month | 11.04 (8.62) | 110.84 (26.07) | 31.94 (1.16) | 151.97 (16.81) | |
| 3rd month | 9.5 (1.25) | 90.2 (62.44) | 135.19 (12.71) | 116.95 (127.11) |
Only eight (0.36%) patients reported adverse events, with urinary tract infection (UTI) affecting three patients (0.13%). Other adverse events included hypotension, photosensitivity, fungal phimosis, moderate hypoglycemia, and hypersensitivity, each occurring in one patient (0.05%). Of the 1,315 patients (59.8%) who experienced weight loss, 708 (32.2%) lost 0–2 kg, 508 (23.1%) lost 2–4 kg, and 99 (4.5%) lost more than 4 kg.
A total of 1,717 (78.08%) patients achieved their glycemic goal with the FDC of vildagliptin 100 mg and dapagliflozin 10 mg (Table 3).
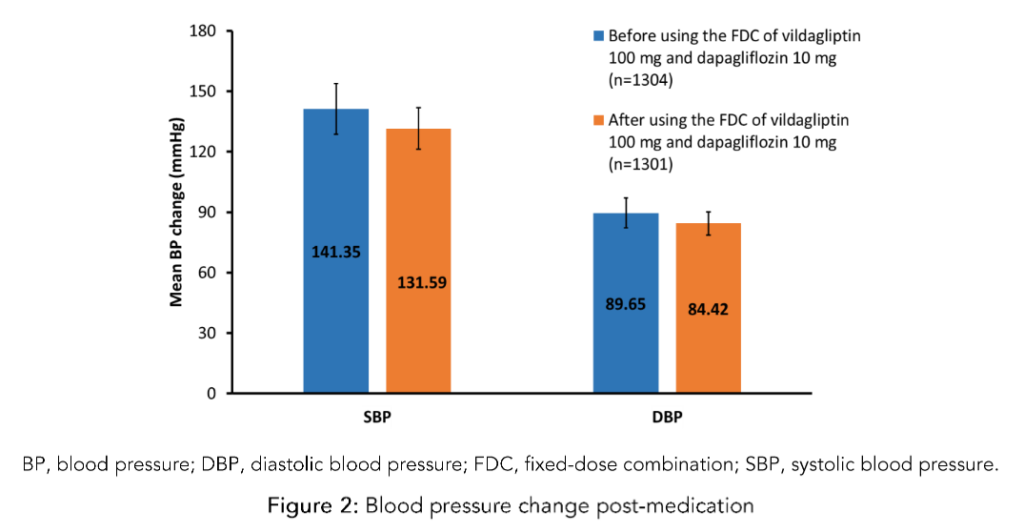
The blood pressure change following the FDC of vildagliptin 100 mg and dapagliflozin 10 mg therapy is represented in Figure 2. The mean (SD) SBP decreased from 141.35 (12.60) mmHg to 131.59 (10.38) mmHg, and the mean (SD) DBP decreased from 89.65 (7.37) mmHg to 84.42 (5.78) mmHg after the FDC therapy.
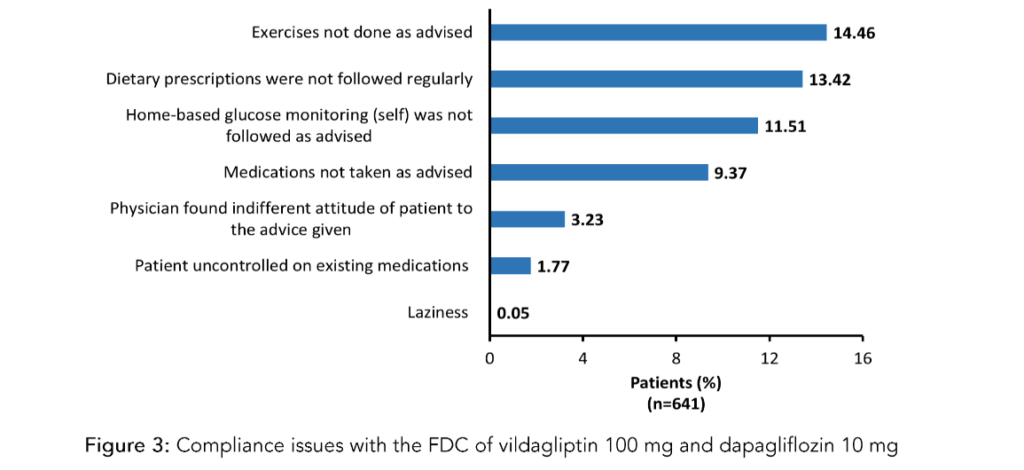
The compliance issues with the FDC of vildagliptin 100 mg and dapagliflozin 10 mg are presented in Figure 3. Issues with treatment adherence were noted in 641 (29.15%) patients, mainly due to lack of exercise (14.46%), followed by dietary non-compliance (13.42%), and failure to follow home-based glucose monitoring as advised (11.51%).

Figure 3: Compliance issues with the FDC of vildagliptin 100 mg and dapagliflozin 10 mg
Discussion
Managing T2DM requires navigating a complex landscape to achieve optimal glycemic control. Effective management often involves combination therapy, which uses antidiabetic drugs with complementary mechanisms of action. This approach addresses multiple pathophysiological defects and can lead to an additive reduction in HbA1c levels.¹³ However, there is a scarcity of real-world data from India evaluating the efficacy, safety, and tolerability of the combination of vildagliptin and dapagliflozin in patients with T2DM.
The present study revealed that most patients with T2DM in India, visiting cardiologists, fall within the age group of 40–60 years, highlighting a predominance of T2DM among middle-aged Indian adults. This finding aligns with a survey in which 90% of clinicians also reported that most T2DM patients in India are aged 40–60 years.¹⁴ However, global evidence suggests a rising trend of early-onset T2DM, particularly among adolescents and young adults in countries with low-middle and middle sociodemographic indexes.¹⁵ Recent studies emphasize the growing prevalence of T2DM among young Indian adults aged 20–40 years.¹⁶–¹⁹
The present study identified hypertension as the most common comorbidity among patients with T2DM, followed by dyslipidemia, CAD, and stroke/transient ischemic attack. This finding is consistent with the expert opinion on the use of vildagliptin in Indian patients with diabetes mellitus, where most clinicians reported hypertension (71.4%) as the prevalent comorbidity, followed by dyslipidemia (22.1%), ischemic heart disease (3.7%), and hypothyroidism (2%).²⁰
In this study, most patients achieved their glycemic goals using the FDC of vildagliptin 100 mg and dapagliflozin 10 mg. This suggests that this combination might be an effective treatment strategy for glycemic control in T2DM patients, offering a better chance of achieving and maintaining the desired HbA1c levels, a critical indicator of long-term blood glucose control. These findings are supported by an expert opinion-based consensus, where over 80% of clinical experts concurred that the vildagliptin–dapagliflozin FDC is an appealing treatment option for a broad range of Indian T2DM patients. The consensus underscored the potential of this combination therapy to enhance diabetes management outcomes in the Indian population.²¹ The current study highlights a notable decrease in mean HbA1c levels from baseline to 3 months, reinforcing the effectiveness of various combination therapies for T2DM. In a randomized clinical trial, the combination of saxagliptin and dapagliflozin achieved an HbA1c level of <7% in 41% of patients by week 24, demonstrating substantial glycemic control.²² Fixed-dose combinations of dapagliflozin with linagliptin or vildagliptin produced even more pronounced reductions in HbA1c over 16 weeks.²³ Several studies also corroborate that the combination of DPP-4 and SGLT2 inhibitor showed significant changes in HbA1c levels from baseline.²²,²⁶
In the present study, the majority of physicians reported that the FDC of vildagliptin 100 mg and dapagliflozin 10 mg demonstrated good efficacy and tolerability. This aligns with phase III trial findings, which indicated that the combination had no severe adverse effects and was well tolerated.²³ The majority of experts (82%) view the vildagliptin–dapagliflozin FDC as an appealing treatment option for T2DM due to the complementary mechanisms of action of DPP-4 inhibitors and SGLT2 inhibitors.²¹
Conclusion
The management of diabetes must evolve beyond glycemic control to encompass the broader spectrum of cardiovascular risk. Early intervention in patients with diabetes and associated risk factors like hypertension and dyslipidemia is not just beneficial—it is imperative. Delaying treatment allows silent yet progressive vascular damage to take hold, increasing the likelihood of severe complications such as heart attacks and strokes.
By adopting a proactive, multifaceted approach that integrates advanced therapeutic options—such as Fixed-Dose Combinations (FDCs) targeting both glycemia and cardiovascular health—clinicians can significantly improve patient outcomes. The FDC of vildagliptin 100 mg and dapagliflozin 10 mg demonstrated substantial efficacy in managing T2DM, with significant improvements in glycemic control and blood pressure. The combination was well tolerated with a low incidence of adverse events. These findings support the utility of the FDC of vildagliptin 100 mg and dapagliflozin 10 mg in real-world clinical practice in the Indian context.
Conflict of interest
There are no conflicts of interest. Dr. Sandeep K. K. Dutta is an employee of USV Pvt Ltd.
Acknowledgments
The medical writing support was provided by Dr. Sanjeev Kumar (Scientific Services, USV Pvt Ltd, Mumbai) and the medical team of Abigloshesin Clinpharm Private Limited. The authors acknowledge Abigloshesin Clinpharm Private Limited for their assistance in conducting the real-world study. The authors acknowledge Mr. Vikram M., Mr. Alireza Doctor and Ms. Monal Patil from USV Pvt Ltd for their assistance in carrying out the study.
Funding
This project has been funded by USV Pvt Ltd.
Authorship
All authors take responsibility for the integrity of the work as a whole and have given their approval for this version to be published. The contents published herein represent the views and opinions of USV Ltd. and/or its affiliates. The established authors have no intention of offering educational, academic, or research purposes and are not intended as a substitute for professional medical advice, diagnostic or treatment.
References
1. Goyal R, Singhal M, Jialal I. Type 2 Diabetes. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK513253/
2. Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045 [published correction appears in Diabetes Res Clin Pract. 2023;204:110945.
3. Defronzo RA. Banting Lecture. From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes. 2009;58(4):773-95.
4. Hinnen D, Kruger D, Magwire M. Type 2 diabetes and cardiovascular disease: risk reduction and early intervention. Postgraduate medicine. 2023; 135(1):2-12.
5. Chadha M, Das AK, Deb P, Gangopadhyay KK, Joshi S, Kesavadev J, et al. Expert Opinion: Optimum Clinical Approach to Combination-Use of SGLT2i + DPP4i in the Indian Diabetes Setting. Diabetes Ther. 2022;13(5):1097-1114.
6. Alam MS, Aqil M, Qadry AK, Kapu P, Pillai KK. Utilization pattern of oral hypoglycaemic agents for type 2 diabetes mellitus patients attending out-patient department at a University Hospital in New Delhi. Pharmacol Pharm. 2014;5:636–45.
7. Scheen AJ. DPP-4 inhibitor plus SGLT-2 inhibitor as combination therapy for type 2 diabetes: from rationale to clinical aspects. Expert Opin Drug Metab Toxicol. 2016;12(12):1407-17.
8. Min SH, Yoon JH, Moon SJ, Hahn S, Cho YM. Combination of sodium-glucose cotransporter 2 inhibitor and dipeptidyl peptidase-4 inhibitor in type 2 diabetes: a systematic review with meta-analysis. Sci Rep. 2018;8(1):4466.
9. Mathieu C, Kozlovski P, Paldánius PM, Foley JE, Modgill V, Evans M, et al. Clinical Safety and Tolerability of Vildagliptin – Insights from Randomised Trials, Observational Studies and Post-marketing Surveillance. Eur Endocrinol. 2017;13(2):68-72.
10. Dhillon S. Dapagliflozin: A Review in Type 2 Diabetes. Drugs. 2019;79(10):1135-46.
11. Phrommintikul A, Wongcharoen W, Kumfu S, Jaiwongkam T, Gunaparn S, Chattipakorn S, et al. Effects of dapagliflozin vs vildagliptin on cardiometabolic parameters in diabetic patients with coronary artery disease: a randomised study. Br J Clin Pharmacol. 2019;85(6):1337-47.
12. Sharma MD. Potential for combination of dipeptidyl peptidase-4 inhibitors and sodium-glucose co-transporter-2 inhibitors for the treatment of type 2 diabetes. Diabetes Obes Metab. 2015;17(7):616-21.
13. Abdul-Ghani M. Where does combination therapy with an SGLT2 inhibitor plus a DPP-4 inhibitor fit in the management of type 2 diabetes? Diabetes Care. 2015;38(3):373-5.
14. Akshay Bafna, Mehta A, Garg R, Jayagopal Pathiyil Balagopalan, Rajput R, Nitin Zalte, et al. Diabetes Treatment with Dapagliflozin and Its Combinations: Insights from Clinical Practice. Journal of Diabetology. 2024;15(2):183–92.
15. Xie J, Wang M, Long Z, Ning H, Li J, Cao Y, et al. Global burden of type 2 diabetes in adolescents and young adults, 1990-2019: Systematic analysis of the Global Burden of Disease Study 2019. BMJ. 2022;379:e072385.
16. Pradeepa R, Mohan V. Epidemiology of type 2 diabetes in India. Indian J Ophthalmol 2021; 69:2932–8.
17. Rajbhandari SM, Kumar KV, Selvarajan R, Murali T. High prevalence of diabetes in young people in Bangalore, India. Br J Diabetes. 2020;20:104–6.
18. India State-Level Disease Burden Initiative Diabetes Collaborators. The increasing burden of diabetes and variations among the states of India: The global burden of disease study 1990-2016. Lancet Glob Health 2018;6:e1352–62.
19. Nagarathna R, Bali P, Anand A, Srivastava V, Patil S, Sharma G, et al. Prevalence of diabetes and its determinants in the young adults indian population-call for yoga intervention. Front Endocrinol (Lausanne) 2020;11:507064.
20. Manjula S, and Krishna Kumar M. Expert Opinion on the Use of Vildagliptin in Indian Patients with Diabetes Mellitus. Asian Journal of Research and Reports in Endocrinology. 2024;7(1):11-19.
21. Agrawala RK, Akbar SS, Kaushik Biswas DN, Garg UK, Warrier S, Prasad A, et al. The Use of Vildagliptin-Dapagliflozin Fixed Dose Combination in Different Patient Profiles in Indian Settings: An Opinion-Based Consensus. International Journal of Clinical and Surgical Research & Reviews. 2023;6 (03):2010-17.
22. Rosenstock J, Hansen L, Zee P, Li Y, Cook W, Hirshberg B, et al. Dual add-on therapy in type 2 diabetes poorly controlled with metformin monotherapy: a randomized double-blind trial of saxagliptin plus dapagliflozin addition versus single addition of saxagliptin or dapagliflozin to metformin. Diabetes Care. 2015;38(3):376-83.
23. Dharmalingam M, Kumar Sharma S, Prakash V, Maiti A, Kumar R, Murthy LSS, et al. Evaluating the Efficacy, Safety, and Tolerability of Combination Therapy of Dapagliflozin and Linagliptin Over Dapagliflozin and Vildagliptin in Patients with Type 2 Diabetes Mellitus Inadequately Controlled with Metformin. Cureus. 2024;16(4):e58115.
24. Petchiappan V, Mathew E, Jose J, Fardan M, Chidambaram Y, Thangavelu S. Use of Fixed-dose Combination Therapy with Remogliflozin and Vildagliptin as an Add-on Drug in Improving the Glycemic Control of Type 2 Diabetes Mellitus: An Observational Study. Journal of Pharmacology and Pharmacotherapeutics. 2023;14(1):72-78.
25. Real-world effectiveness of dapagliflozin and sitagliptin fixed-dose combination in Indian patients with type 2 diabetes: a retrospective analysis of electronic medical records stratified by BMI. Available from:
https://www.endocrine-abstracts.org/ea/0099/ea0099ep639
26. Søfteland E, Meier JJ, Vangen B, Toorawa R, Maldonado-Lutomirsky M, Broedl UC. Empagliflozin as Add-on Therapy in Patients with Type 2 Diabetes Inadequately Controlled with Linagliptin and Metformin: A 24-Week Randomized, Double-Blind, Parallel-Group Trial. Diabetes Care. 2017;40(2): 201-09.