Cost-Effectiveness of Gene Therapy in Hemophilia B
Cost-effectiveness of gene therapy with etranacogene dezaparvovec versus factor IX prophylaxis in men with hemophilia B in Brazil
Juliana Alvares-Teodoro¹, Ana Clara Silva Mendes¹, Ricardo Mesquita Camelo¹², Augusto Afonso Guerra Junior¹, Francisco de Assis Acurcio¹
- Faculdade de Farmácia, CCATES, Universidade Federal de Minas Gerais, Belo Horizonte, MG, Brasil.
- Faculdade de Medicina, Universidade Federal de Minas Gerais, Belo Horizonte, MG, Brasil.
OPEN ACCESS
PUBLISHED: 31 January 2025
CITATION: Alvares-Teodoro, J., Mendes, ACS., et al., 2024. Cost-effectiveness of gene therapy with etranacogene dezaparvovec versus factor IX prophylaxis in men with hemophilia B in Brazil. Medical Research Archives, [online] 13(1).
https://doi.org/10.18103/mra.v13i1.6167
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i1.6167
ISSN 2375-1924
ABSTRACT
Background: The high costs and uncertainties surrounding the effectiveness and safety of new health technologies necessitate continuous evaluation to balance access and sustainability within Brazil’s Unified Health System.
Aims: Cost-effectiveness analysis comparing etranacogene dezaparvovec versus factor IX prophylaxis in people with hemophilia B from the Brazilian healthcare system perspective.
Methods: A cost-effectiveness analysis was conducted using TreeAge Pro software. A decision tree model compared the costs and effectiveness of etranacogene dezaparvovec versus factor IX prophylaxis in preventing bleeding in adult male with severe or moderately severe hemophilia B over a three-year horizon. A deterministic sensitivity analysis identified variables impacting incremental cost-effectiveness ratio.
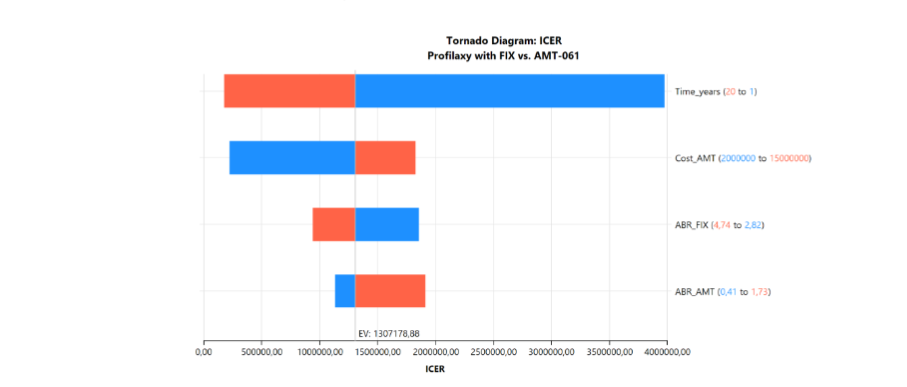
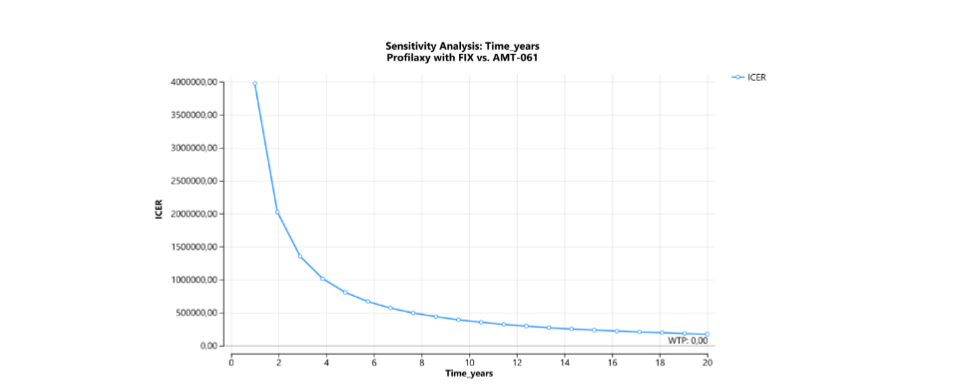
Results: etranacogene dezaparvovec incurred a cost of USD 2,335,747 (BRL 10,810,993) versus USD 46,451 (BRL 232,256) for plasma-derived factor IX prophylaxis, with an incremental cost of USD 2,115,747 (BRL 10,578,737). The effectiveness (number of bleeding events) was 2.86 for etranacogene dezaparvovec and 10.95 for factor IX prophylaxis, with etranacogene dezaparvovec avoiding 8.09 additional bleeding events. The incremental cost-effectiveness ratio was USD 261,436 (BRL 1,307,179) per bleeding event avoided. Sensitivity analysis revealed treatment duration as the most impactful variable on incremental cost-effectiveness ratio, contributing to 80% of model uncertainty.
Discussion: International studies suggest that etranacogene dezaparvovec is cost-effective and dominant in multiple contexts, particularly when compared to extended half-life recombinant factor IX (rFIX-EHL), offering greater quality-adjusted life years for people with hemophilia B at lower long-term costs. However, a lack of robust data underscores the need for ongoing monitoring and careful evaluation before incorporation by the health system. It should also be noted that high pricing of new treatments like etranacogene dezaparvovec poses challenges for Brazil’s Unified Health System sustainability and equitable access.
Introduction
Hemophilia B (HB) is a hereditary coagulopathy caused by mutations in the F9 gene located on the X chromosome, which encodes coagulation factor IX (FIX)¹. Globally, an estimated 37,385 individuals live with hemophilia B, including 2,277 in Brazil². Reduced residual plasma FIX activity elevates the risk of spontaneous bleeding, particularly hemarthrosis, which can lead to hemophilic arthropathy, reduced mobility, and impaired quality of life³⁴.
Standard treatment for hemophilia B involves intravenous replacement of exogenous FIX to prevent or manage bleeding episodes. Prophylaxis is the globally recommended approach to mitigate bleeds and associated complications⁴. In Brazil, prophylaxis was incorporated into the SUS in 2012, with FIX procured by the Ministry of Health and distributed free of charge through Hemophilia Treatment Centers⁸. However, lifelong infusion requirements hinder treatment adherence⁹ and FIX pharmacokinetic variability results in different degrees of protection between doses¹⁰. Expenses related to FIX concentrates and its administration account for over 99% of the total prophylactic treatment costs for people with hemophilia B. In Brazil, plasma-derived FIX (pdFIX) is the product provided by the SUS¹¹.
Gene therapy aims to achieve stable endogenous FIX activity through a single dose¹² and has been under investigation for over two decades¹³. Recently, two technologies have been approved for clinical use¹⁴¹⁵. The first employs an adeno-associated virus serotype 5 (AAV5) vector containing a wild-type human F9 cassette (AMT-060)¹⁶ resulting in stable endogenous FIX expression and reducing bleeding episodes for up to five years¹⁷. AAV5 targets hepatocytes, where the transgene is inserted, enhancing plasma FIX activity¹⁸. Building on this, an AAV5 vector with a mutated F9-Padua cassette was developed to improve thrombin generation¹⁹. Consequently, etranacogene dezaparvovec (AMT-061) was created, an AAV5 vector carrying the F9-Padua gene variant²⁰²¹²². Since this technology had not been registered or priced in Brazil at the time of analysis, this study estimated its value based on another gene therapy available in the country, onasemnogene abeparvovec, used for treating spinal muscular atrophy²³.
The phase 3 HOPE-B study evaluated the efficacy and safety of etranacogene dezaparvovec compared to FIX prophylaxis in 54 adult people with hemophilia B with residual plasma FIX activity ≤2%²⁴. Between 7 and 18 months post-treatment, annualized bleeding rates decreased by 64% (total bleeds), 77% (treated bleeds), 78% (treated or untreated hemarthroses), and 79% (treated hemarthroses). However, uncertainties remain regarding FIX expression durability¹⁷²⁴ and long-term side effects. In the short-medium term, 42% developed drug-induced hepatitis, with 38% requiring corticosteroids²¹.
The price of AMT-061, launched in 2022, was US$ 3.5 million (approximately BRL 17.5 million)¹⁵. International economic evaluations estimate costs ranging from US$ 1.8 to 2.0 million²⁵²⁶. The high cost, coupled with uncertainties regarding effectiveness and safety, underscores the need for continuous assessments to balance access to new technologies with the sustainability of the Brazil’s Unified Health System (SUS)²⁷.
So, this study aims to analyze the cost-effectiveness of etranacogene dezaparvovec compared to FIX prophylaxis in severe and moderately severe hemophilia B from the Brazilian healthcare system perspective.
Methods
COSTS
The cost calculation for prophylactic treatment was based on the dosage and acquisition price of pdFIX, as well as the cost of infusion procedures²⁸. The price per IU for the latest national pdFIX procurement (April 27, 2023) was obtained from the Banco de Preços em Saúde²⁹ (Tables 1 and 2). Infusion procedure costs were retrieved from the Departamento de Informática do Sistema Único de Saúde²⁹ (Table 1).
The costs for people with hemophilia B treatment with etranacogene dezaparvovec included the price of the drug and hepatic monitoring exams. For therapy with onasemnogene abeparvovec, a 40% reduction in its launch price was observed prior to commercialization in Brazil. Similarly, the etranacogene dezaparvovec launch price of US$ 3.5 million²⁹ was reduced, yielding a model-based medication cost of US$ 2.1 million.
The costs included hepatic monitoring tests for aspartate aminotransferase (AST) and alanine aminotransferase (ALT), performed weekly for three months following infusion²¹. For patients treated with etranacogene dezaparvovec who experienced therapeutic failure, additional costs for resuming prophylaxis with pdFIX were incorporated (Table 2).
Bleeding treatment costs were estimated based on the required amount of pdFIX concentrate. The prices of pdFIX concentrates and infusion procedures were the same as those used for pdFIX prophylaxis²⁹³⁰ (Tables 1 and 2).
EFFECTIVENESS
Effectiveness data for etranacogene dezaparvovec were derived from the phase 3 clinical trial, which included 54 people with hemophilia B, 52 of whom discontinued FIX prophylaxis after a single dose of etranacogene dezaparvovec, reflecting a 96% treatment success rate²¹. The annualized bleeding rates for treated bleeds with FIX prophylaxis (3.65) versus etranacogene dezaparvovec (0.84) were also sourced from the HOPE-B study²¹. The effectiveness of any FIX concentrate in this study was considered equivalent to that of pdFIX concentrate.
Table 1. Parameters used in the economic model
| Parameters | Prophylaxis with pdFIX | Range (min.and max.) | AMT-061 | Range (min.and max.) | References |
|---|---|---|---|---|---|
| Weight (kg) | 74.6 | 65.3–74.6 | 74.6 | 65.3–74.6 | IBGE*, 2008 |
| Gene Therapy Success Rate (%) | NA | NA | 96 | NA | Pipe SW et al., 2023 |
| Duration (years) | 3 | 0–3 | 3 | 0–3 | Pipe SW et al., 2023 |
| pdFIX dose in prophylaxis (IU/kg) | 25 | 20–30 | NA | NA | |
| Frequency of pdFIX in prophylaxis (infusions/month) | 9 | 7–10 | NA | NA | |
| ABR for Treated Bleeds | 3.7 | 2.8–4.7 | 0.8 | 0.4–1.7 | Pipe SW et al., 2023 |
| pdFIX dose in on-demand treatment (IU/kg) | 49 | 29–79 | 9 | 9–39 | Brasil. 2015 Manual de hemofilia |
| Frequency of pdFIX in On-Demand Treatment (days) | 3 | 1–7 | 3 | 1–7 | Premise |
| Total number of AST and ALT tests | NA | NA | 24 | NA | Cslbehring, 2023 |
1 US$ = 5 R$ (Reais)
min.: minimum; max.: maximum; NA: Not applicable; pdFIX: Plasma-derived factor IX; ABR: Annualized bleeding rate; IU/kg: International Unit/kilogram; AST: Aspartate aminotransferase; ALT: Alanine aminotransferase; AMT-061: etranacogene dezaparvovec
- IBGE: Instituto Brasileiro de Geografia e Estatística (The Brazilian Institute of Geography and Statistics)
Table 2. Prices used in the economic model
| Parameters | Prophylaxis with pdFIX | Range (min.and max.) | AMT-061 | Range (min.and max.) | References |
|---|---|---|---|---|---|
| Price per infusion (R$) | 5.39 | NA | 5.39 | NA | Datasus**, 2023 proc. 030602002-5 |
| Price of pdFIX (R$/IU) | 0.33 | NA | 0.33 | NA | BPS***, 2023 code 0450529 |
| Price of AMT-061 (R$/dose) | NA | NA | 10.8 million | 2–15 millions | Premise |
| Price of AST and ALT tests (R$) | NA | NA | 2.01 | NA | Datasus**, 2023 |
1 US$ = 5 R$ (Reais)
min.: minimum; max.: maximum; NA: Not applicable; pdFIX: Plasma-derived factor IX; AMT-061: etranacogene dezaparvovec; IU/kg: International Unit/kilogram; AST: Aspartate aminotransferase; ALT: Alanine aminotransferase
** Datasus: Departamento de Informática do SUS (Department of Informatics of the Unified Health System) – SIGTAP: Sistema de Gerenciamento da Tabela de Procedimentos, Medicamentos, Órteses, Próteses e Materiais Especiais do SUS
*** BPS: Banco de Preços em Saúde (Health Price Database) SIASG: Sistema Integrado de Administração de Serviços Gerais
COST-EFFECTIVENESS MODEL
A cost-effectiveness analysis was conducted using TreeAge Pro, LLC software (Williamstown, USA). A decision tree model was developed to compare the costs and effectiveness of etranacogene dezaparvovec versus FIX prophylaxis in preventing bleeding episodes in people with hemophilia B with severe or moderately severe conditions over a three-year horizon from the perspective of Brazilian Ministry of Health (Figure 1).
Figure 1. Decision tree of the economic cost-effectiveness analysis model of gene therapy with AMT-061 compared to prophylaxis with plasma-derived factor IX.
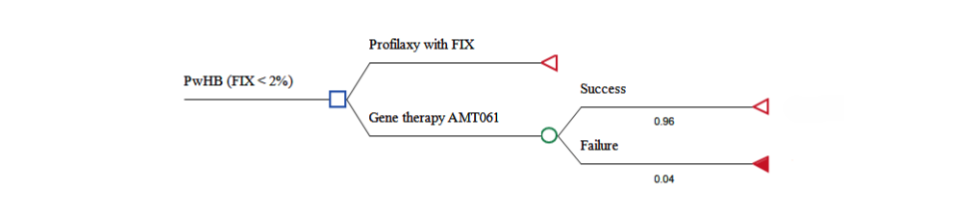
The modeled population consisted of adult male with hemophilia B with severe (residual FIX activity <1%) or moderately severe hemophilia (1% ≤ residual FIX activity ≤ 2%) undergoing regular FIX prophylaxis, as recommended by the World Federation of Hemophilia⁴. This cohort aligns with participants from the HOPE-B study²¹. The three-year time horizon was selected based on phase 2b trial data indicating a mean endogenous FIX activity of 36.9% (range: 32.3%–41.5%) three years post-treatment with etranacogene dezaparvovec²⁴. As a novel technology, data on its efficacy or effectiveness beyond this period are not yet available.
FIX prophylaxis has a variable dosage depending on the product used and the bleeding phenotype of the people with hemophilia B⁴. Plasma-derived FIX (pdFIX) has a standard half-life of approximately 17 hours, with a dosage of 20–30 International Units (IU/kg) of body weight administered twice weekly, adjustable to individual needs. This study assumed an average dosage of 25 IU/kg twice weekly for pdFIX, with 100% adherence.
For etranacogene dezaparvovec treatment, a single-dose administration was assumed. Reapplication of etranacogene dezaparvovec was