COVID-19 Symptoms in Smokers vs. Non-Smokers: A Study
Clinical Presentation and Associated Factors to SARS-CoV-2 infection: A Cross-sectional and Comparative Study Between Smokers and Non-Smokers Population
Osvaldo Erik Sanchez-Hernandez¹, Daniel Lopez-Hernandez², Leticia Brito-Aranda³, Alejandra Hernandez-Cervuelos¹, Abraham Espinoza-Perdomo³, Karina Ayala-Lopez⁴, Maria Luisa Lucero Saldivar-Gonzalez⁵, Guadalupe Jacqueline Flores-Morales³, Edgar Cruz-Aviles⁶, Alberto Vazquez-Sanchez⁷, Maria Clara Hernandez-Almazan⁸, Perla Veronica Salinas-Palacios⁹, Luis Beltran-Lagunes³, Edgar Esteban Torres-García⁷, Liliana Grisel Liceaga-Perez¹⁰
- Área Académica de Medicina. Instituto de Ciencias de la Salud, Universidad Autónoma del Estado de Hidalgo, Hidalgo, México.
- Clínica de Medicina Familiar “División del Norte”, Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado, Cuidad de México, México.
- Centro de Investigación y de Educación Continua, S.C. Estado de México, México.
- Centro Médico “Dr. Ignacio Chávez”, Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado de Sonora, Sonora, México.
- Clínica de Medicina Familiar “Milpa Alta”, Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado, Cuidad de México, México.
- Clínica de Medicina Familiar “Ermita”, Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado, Cuidad de México, México.
- Subdirección de Prevención y Protección a la Salud, Dirección Médica, Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado, Cuidad de México, México.
- Clínica de Medicina Familiar “Aragón”, Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado, Cuidad de México, México.
- Centro Médico Nacional “20 de noviembre”, Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado, Cuidad de México, México.
- Clínica de Medicina Familiar “Guerrero”, Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado, Cuidad de México, México.
OPEN ACCESS
PUBLISHED: 30 November 2025
CITATION: Sanchez-Hernandez, OE., Lopez-Hernandez, D., et al., 2025. Clinical Presentation and Associated Factors to SARS-CoV-2 infection: A Cross-sectional and Comparative Study Between Smokers and Non-Smokers Population. Medical Research Archives, [online] 13(11). https://doi.org/10.18103/mra.v13i11.7113
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i11.7113
ISSN 2375-1924
ABSTRACT
Introduction: Characterising the symptom profile of COVID-19 in smokers compared to non-smokers enables the identification of distinct clinical patterns and potential risk factors. This, in turn, supports more accurate risk stratification and the formulation of targeted public health strategies, particularly within primary care settings, where early detection and timely intervention are essential.
Material and Methods: A population-based, cross-sectional comparative study was conducted using national surveillance data from 475 healthcare units across Mexico. The analysis focused on patients tested for SARS-CoV-2, stratified by smoking status (smokers and non-smokers). Variables included age, clinical symptoms, comorbidities, test results, and vaccination status. Associations between symptoms, comorbidities, and COVID-19 positivity were assessed using multivariate logistic regression models in both groups.
Results: The study included two groups: a total of 48,314 smokers (12.5%; 95% CI 12.4-12.6) and 338,900 non-smokers patients (87.5%; 95% CI 87.4-87.6), resulting in a total study population of 387,214 patients. From this population, we identified 193,607 patients with COVID-19 (21,777 smokers versus 171,830 non-smokers patients). Smokers exhibited a higher prevalence of all reported symptoms in comparison to non-smokers: headache (62% vs 60.3%), cough (57.7% vs 56.6%), myalgia (42.9% vs 39.7%) and odynophagia (40.4% vs 36.9), with the exception of fever (41.3% vs 43.4%). Signs and symptoms positively associated with a higher likelihood of COVID-19 in both smokers and non-smokers included fever, cough, dyspnoea, chest pain, chills, myalgia, arthralgia, malaise, rhinorrhoea, polypnoea, and cyanosis. In contrast, type 2 diabetes, obesity, and hypertension were linked to an increased risk of COVID-19 only among non-smokers. Age showed an inverse relationship with COVID-19 risk in both groups. Notably, being female was the only factor consistently associated with a higher risk of infection across both populations.
Conclusion: The observed profile among smokers suggests a more symptomatic or severe clinical presentation. The lack of association among obesity, hypertension, and diabetes with COVID-19 in this group suggests a possible existence of distinct biological or behavioural mechanisms influencing disease susceptibility and progression.
Keywords:
Comorbidity; COVID-19; primary care; smoking
Introduction
The COVID-19 pandemic has highlighted the critical importance of understanding the role of modifiable risk factors in disease severity and outcomes among communicable diseases and their relationship with non-communicable diseases. Among these, tobacco use has emerged as a significant determinant, not only in increasing susceptibility to SARS-CoV-2 infection but also in exacerbating the severity of clinical outcomes. Hamer et al., in 2020, reported that smoking was independently associated with an increased risk of being hospitalised due to COVID-19 among adults residing in the United Kingdom (UK). The study utilised data from the UK Biobank, a large prospective cohort comprising 387,109 participants (mean age 56.4 ± 8.8 years; 55.1% female). Multiple studies have shown that smokers are more likely to contract COVID-19 (Odds Ratio, OR=1.42; Confidence Interval 95%, CI95%=1.12-1.79) and experience worse disease progression (disease severity: current smoking OR=1.58; CI95%=1.16-2.15, p=0.004 and former smoking OR=2.48; CI95%=1.64-3.77, p<0.001), including higher rates of hospitalisation (OR=2.31; 95%CI=1.94-2.74), intensive care unit (ICU) admission, and mortality (current smoking OR=1.35; CI95%=1.12-1.62, p=0.002; and former smoking OR=2.58; CI95%=2.15-3.09, p<0.001). The pro-inflammatory and immunosuppressive effects of tobacco, coupled with pre-existing respiratory conditions and cardiovascular and metabolic comorbidities commonly seen in smokers, contribute to a heightened vulnerability to severe COVID-19. However, debates are still ongoing as to the effect of cigarette smoking on vulnerability to COVID-19, due to several observational studies reporting that smoking may reduce the risk of SARS-CoV-2 infection and COVID-19, which has sparked significant controversy within the scientific community. Besides, initial reports noted a low number of active smokers among hospitalised COVID-19 patients, prompting speculation about a potential “smoker’s paradox,” whereby smoking might offer some form of protection against infection or severe illness. However, such conclusions have been met with widespread scepticism, as they are largely based on observational studies with notable methodological flaws. These include biased patient selection and incomplete or inaccurate recording of smoking status. Furthermore, underreporting of tobacco use—particularly in clinical settings—and the presence of confounding factors such as socioeconomic status and comorbidities further complicate the interpretation of these findings. Given the high prevalence of smokers and former smokers in clinical practice, particularly in primary care, research into the interaction between smoking and COVID-19 remains essential. From a public health perspective, this association represents a substantial burden. Smoking-related vulnerability to COVID-19 strains healthcare resources across all levels of care. Furthermore, at the primary care level, clinicians must be vigilant in identifying smokers as high-risk individuals, providing targeted preventive counselling and early intervention. At the secondary and tertiary levels, the increased likelihood of complications among smokers requires greater resource allocation, including ICU beds, ventilatory support, and prolonged hospital stays. For the society, the pandemic has amplified health inequalities already linked to tobacco use, disproportionately affecting socioeconomically disadvantaged populations where smoking prevalence is higher. This underscores the need for governments to reinforce tobacco control policies, integrating them into public health strategies. Therefore, policy interventions such as smoking cessation programmes, taxation, public awareness campaigns, and regulatory restrictions on tobacco advertising become not only tools for chronic disease prevention but also for mitigating the impact of infectious diseases like COVID-19. In this context, addressing the intersection between smoking and COVID-19 is not merely a clinical or epidemiological concern—it is a societal imperative that requires coordinated action across healthcare systems, governmental policy, and community engagement to reduce harm, save lives, and build more resilient public health infrastructures. Therefore, grasping the clinical presentation of COVID-19 in smokers can enhance clinical decision-making and aid in the formulation of targeted public health strategies.
Material and Methods
STUDY DESIGN, SETTING AND PARTICIPANTS
This study was designed as a population-based, cross-sectional, comparative, and analytical investigation, utilising a previously published secondary dataset comprising patients in Mexico who underwent nasopharyngeal testing for SARS-CoV-2 at various healthcare facilities. Consequently, subjects were classified as suspected cases of viral respiratory infection through the national epidemiological surveillance system. The original research, which followed a case–control design, utilised a risk set sampling method to enhance the internal validity of the analysis. In this framework, control subjects were drawn from the population at risk at the time each case was identified, ensuring temporal comparability and avoiding selection bias. This approach permitted a robust estimation of rate ratios using odds ratios, even in the absence of the rare disease assumption. Importantly, unlike conventional cumulative or survivor sampling strategies, risk set sampling recognises that some individuals in the control group may later develop the disease—a critical consideration when working with dynamic and continuously updated health registries. Hence, for the current analysis, we conducted a cross-sectional comparison using the same dataset, focusing specifically on two subgroups: smokers and non-smokers. This design enabled us to explore differences in clinical presentation, comorbidities, and health outcomes related to SARS-CoV-2 infection between these two groups, without employing a longitudinal or time-to-event analytical framework.
DATABASE AND DATA SOURCES
The data used in this study comes from the National Epidemiological Surveillance System for Viral Respiratory Diseases, which collects information from 475 Viral Respiratory Disease Monitoring Health Units (Unidades de Salud Monitoras de Enfermedad Respiratoria Viral, USMER), as well as additional Non-USMER units that have been adapted nationwide for COVID-19 screening. Both types of units—USMER and Non-USMER—follow standardised protocols to screen individuals exhibiting respiratory symptoms, recording clinical and epidemiological data through the SISVER platform. SARS-CoV-2 diagnostic testing via RT-PCR is routinely conducted for all patients with severe symptoms. Conversely, patients with mild symptoms are tested selectively: USMER units perform testing in at least 10% of these cases, while non-USMER units test according to their operational capacities. Confirmed cases are classified as positive, negative, or pending for SARS-CoV-2 and are subject to validation across various administrative levels before inclusion in the dataset. This study specifically examined two subgroups—smokers and non-smoker patients—to facilitate a comparative assessment within this population-based dataset.
STUDY VARIABLES
All the information was consolidated into a national database that contains SARS-CoV-2 test results, along with sociodemographic data, clinical features, and details related to the healthcare facilities involved. Sociodemographic variables included: patient’s age, sex, and nationality (classified as either Mexican or non-Mexican). Incidentally, clinical data encompassed pre-existing comorbidities, as well as the signs and symptoms present at the time of evaluation. Comorbidities were documented as binary variables (present or absent) and included: diabetes, chronic obstructive pulmonary disease (COPD), asthma, immunosuppression, hypertension, HIV/AIDS, cardiovascular disease, obesity, chronic kidney disease, and other unspecified conditions. Additionally, the total number of comorbidities was also documented and the smoking status was classified as either smoker or non-smoker. Besides, signs and symptoms were recorded as binary variables and included fever, cough, odynophagia, dyspnoea, irritability, diarrhoea, chest pain, chills, headache, myalgia, arthralgia, malaise, rhinorrhoea, polypnoea, vomiting, abdominal pain, conjunctivitis, cyanosis, and the sudden onset of symptoms. On the other hand, Additional information included whether the patient had been vaccinated. All the data was collected using a standardized Respiratory Triage Form completed by the attending physician. For laboratory-confirmed COVID-19 cases, additional information was collected regarding admission to the intensive care unit (ICU), the need for intubation, and mortality. The data related to healthcare facilities indicated whether the unit belonged to the USMER or Non-USMER surveillance networks, as well as the type of facility where the patient received care.
OUTCOMES, SUBJECT’S SELECTION, AND STATISTICAL ANALYSIS
The primary outcome variable was a diagnosis of COVID-19, defined by laboratory-confirmed SARS-CoV-2 infection and documented as a dichotomous variable (positive or negative). A census sampling method was applied to include all eligible smoker and non-smoker patients registered in the national database during the study period. The sample included patients aged 10 years and over, based on data from the 2022 National Health and Nutrition Survey (ENSANUT-2022), which reports that 4.6% of adolescents are smokers. Additionally, the World Health Organization (WHO) defines adolescence as the life stage between childhood and adulthood, encompassing individuals aged 10 to 19 years. Consequently, both study groups—smokers and non-smoker patients—consist of individuals aged 10 years and above. This approach ensured that the full available population of interest was included in each study arm. Categorical variables were described using absolute frequencies and percentages, while quantitative variables were summarised as mean, standard deviation (SD), interquartile range (IQR), minimum and maximum values, and range. All estimates were reported with their respective 95% confidence intervals (95% CI). Comparisons of categorical variables between groups were performed using Yates’ corrected chi-square test, while quantitative variables were analysed with the Median Test as independent samples. The analysis was conducted across two parallel groups: one composed of smokers and the other of non-smokers. Hence, both groups were analysed separately to identify clinical and epidemiological differences related to COVID-19 diagnosis. In order to assess the association between COVID-19 diagnosis (positive/negative) and the independent variables (e.g., age, comorbidities, signs and symptoms), a series of logistic regression models were applied within each study group. These models included continuous variables such as age, and dichotomous variables including presence of comorbidities and specific clinical symptoms. Multivariate logistic regression analyses were performed. Besides, the multivariate logistic regression models were constructed to identify independent predictors of a positive COVID-19 diagnosis while adjusting for potential confounding factors. Odds ratios (ORs) with corresponding 95% confidence intervals (CI95%) were calculated to quantify the strength and direction of associations. An OR >1 indicated an increased likelihood of COVID-19 diagnosis, whereas an OR <1 indicated a decreased likelihood. Variables included in the multivariate logistic regression models were: Age: years (numerical variable). Sex: males=0, females=1. Fever: presence=1, absence=0. Cough: presence=1, absence=0. Odynophagia: presence=1, absence=0. Dyspnoea: presence=1, absence=0. Irritability: presence=1, absence=0. Diarrhoea: presence=1, absence=0. Chest pain: presence=1, absence=0. Chills: presence=1, absence=0. Headache: presence=1, absence=0. Myalgia: presence=1, absence=0. Arthralgia: presence=1, absence=0. Malaise: presence=1, absence=0. Rhinorrhoea: presence=1, absence=0. Polypnea: presence=1, absence=0. Vomiting: presence=1, absence=0. Abdominal pain: presence=1, absence=0. Conjunctivitis: presence=1, absence=0. Cyanosis: presence=1, absence=0. SOS: presence=1, absence=0. Type 2 Diabetes: presence=1, absence=0. COPD: presence=1, absence=0. Asthma: presence=1, absence=0. Immunosuppressed: presence=1, absence=0. Hypertension: presence=1, absence=0. HIV-AIDS: presence=1, absence=0. CVD: presence=1, absence=0. Obesity: presence=1, absence=0. CKD: presence=1, absence=0. Numbers of factors: numerical variable. Vaccinated: presence=1, absence=0. Finally, all statistical tests were two-tailed, and a p-value < 0.05 was considered statistically significant.
ETHICAL CONSIDERATIONS
This study was conducted according to good clinical practices, as defined by Mexican law, and the Helsinki Declaration for research using human beings. The database designed used anonymized dataset of patients that is publicly available and accessible to anyone through the Mexican Health Ministry. The principles that emerge from the United Nations General Assembly, 1989, were used. A principle of legality and loyalty (the information was obtained in a lawful manner), a principle of accuracy (the relevance of the data was verified), a principle of purpose (the database is specific, a legitimate and a public before its creation), a principle of non-discrimination and a principle of security.
Results
CHARACTERISTICS OF THE STUDY POPULATION
The study included two groups: a total of 48,314 smokers (12.5%; 95% CI 12.4-12.6) and 338,900 non-smoker patients (87.5%; 95% CI 87.4-87.6), resulting in a total study population of 387,214 patients. From this population, we identified 193,607 patients with COVID-19 (50%, CI95% 49.8-50.2) (smokers 21,777; 45.1%, CI95% 44.6-45.5 versus non-smoker patients 171,830; 50.7%, CI95% 50.5-50.9). The mean age was 44.12 years old (SD = 16.19), with a range of 110 years old (minimum = 10 years; maximum = 120 years). The median age was 43 years old (IQR = 31–55 years). When stratified by the variable smoking, the median age (41 years old, IQR = 31–53, range = 90 years old; minimum = 10 years old, maximum = 100 years old) among smoker patients was significantly lower (p<0.001, Median Test for independent groups), compared to non-smoker patients (median age=44 years old; IQR = 32–56, range = 110 years old; minimum = 10 years old, maximum = 120 years).
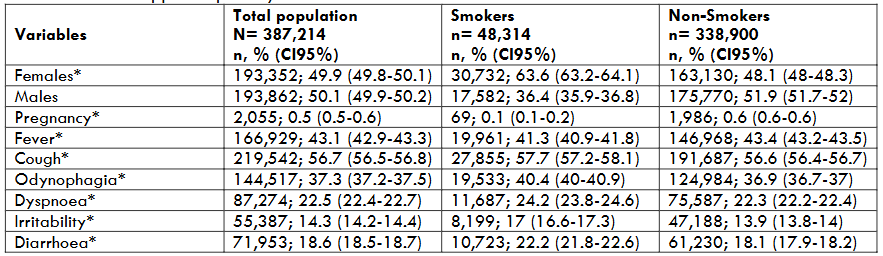
Table 1 presents a comparison of sociodemographic characteristics and the signs and symptoms observed in smokers and non-smokers with upper respiratory tract infections. The proportion of women was significantly higher in the non-smoker group compared to smokers, and conversely, the proportion of men was greater among smokers. In the total population, the sample exhibited an almost equal sex distribution. Conversely, the proportion of pregnant individuals was relatively low. The most frequently reported symptoms were headache, cough, fever, myalgia, and odynophagia. Other commonly observed symptoms included arthralgia, malaise, chills, rhinorrhoea, dyspnoea and chest pain. Additionally, symptoms such as diarrhoea, irritability, conjunctivitis, and abdominal pain were also reported in a considerable proportion of cases. Less frequently reported symptoms included polypnea, vomiting, and cyanosis. The sudden onset of symptoms was reported by 28.3% of the participants.
DESCRIPTION OF THE SMOKERS POPULATION
The prevalence of pregnancy among female smokers was notably low. Among the smoker patients in the study, a higher prevalence of all symptoms was observed compared to non-smokers. Clinically, this suggests a more intense or symptomatic clinical profile associated with tobacco use. Systemic and respiratory symptoms were especially prominent in this group. The most frequently reported symptom was headache, followed by cough, myalgia, fever, and odynophagia. Additionally, other common symptoms included malaise, chills, and rhinorrhoea. Respiratory symptoms such as dyspnoea, polypnea, and chest pain were most prevalent compared to the non-smoking group. Although gastrointestinal symptoms such as diarrhoea and abdominal pain were reported in a moderate proportion of patients, vomiting was reported in a low proportion of cases. Other symptoms included irritability, reported in 17% of patients, and conjunctivitis, observed in 14%. Cyanosis was less common, occurring in less than 5% of cases.
DESCRIPTION OF THE NON-SMOKERS POPULATION
In contrast to the smoker group, the prevalence of pregnancy among non-smokers was notably higher. Clinically, non-smoker patients also exhibited a wide range of symptoms characteristic of upper respiratory tract infections. However, the overall frequency of these symptoms was slightly lower than in smokers, with the exception of fever, which was more commonly reported among non-smokers. The most commonly reported symptoms were headache, cough, fever, myalgia, odynophagia, and arthralgia; moreover, over 20% of patients also experienced malaise, chills, rhinorrhoea, dyspnoea, and chest pain. Similarly, symptoms including diarrhoea, irritability, and conjunctivitis were present in at least 10% of patients. The symptoms reported in less than 10% of non-smoking patients were abdominal pain, polypnea, vomiting, and cyanosis, indicating that these manifestations were relatively uncommon within this population.
IDENTIFICATION OF SIGNS, SYMPTOMS, AND ASSOCIATED FACTORS RELATED TO COVID-19
Regarding Smokers and Non-Smoker Patients, the multivariate logistic regression analysis identified several clinical symptoms and comorbidities as factors associated with COVID-19. A similar pattern of factors associated with either increased or decreased probability of COVID-19 was observed in both groups, along with variables showing no association. Among the non-associated factors identified in both populations were odynophagia, diarrhoea, vomiting, chronic obstructive pulmonary disease (COPD), and chronic kidney disease (CKD). Notably, in the smoker group alone, obesity, type 2 diabetes, hypertension, and the total number of comorbidities also showed no significant association with COVID-19. The only factor consistently associated with a higher risk of COVID-19 across both groups was being female.
Furthermore, signs and symptoms that were positively associated with an increased likelihood of COVID-19 in both smokers and non-smokers included: fever, cough, dyspnoea, chest pain, chills, myalgia, arthralgia, malaise, rhinorrhoea, polypnoea, and cyanosis. Conversely, type 2 diabetes, obesity, and hypertension increased the risk of COVID-19 exclusively in non-smoking patients. Apart from that, age has an inverse association with COVID-19 risk in both groups. Symptoms and conditions associated with a lower probability of COVID-19 included irritability, headache, abdominal pain, and conjunctivitis. Similarly, the sudden onset of symptoms, as well as a history of asthma, immunosuppression, HIV/AIDS, or cardiovascular disease, were all associated with a reduced likelihood of COVID-19. Vaccination was identified as a protective factor in both populations. Lastly, the number of comorbidities showed an inverse association with COVID-19 risk, but only among non-smokers.
Table 2: Multivariate Logistic Regression Analysis of Clinical and Demographic Factors associated to COVID-19 in Non-Smoker population.
| Variables | Beta | OR | CI 95% | P value |
|---|---|---|---|---|
| Age | -0.005 | 0.995 | 0.995 0.996 | <0.001 |
| Sex (1) | 0.023 | 1.023 | 1.008 1.038 | 0.002 |
| Fever (1) | 0.700 | 2.014 | 1.981 2.047 | <0.001 |
| Cough (1) | 0.463 | 1.589 | 1.563 1.615 | <0.001 |
| Odynophagia (1) | 0.001 | 1.001 | 0.984 1.018 | 0.926 |
| Dyspnoea (1) | 0.264 | 1.302 | 1.276 1.328 | <0.001 |
| Irritability (1) | -0.131 | 0.877 | 0.857 0.897 | <0.001 |
| Diarrhoea (1) | -0.010 | 0.990 | 0.970 1.011 | 0.343 |
| Chest pain (1) | 0.078 | 1.081 | 1.060 1.103 | <0.001 |
| Chills (1) | 0.153 | 1.165 | 1.143 1.187 | <0.001 |
| Headache (1) | -0.065 | 0.937 | 0.921 0.953 | <0.001 |
| Myalgia (1) | 0.241 | 1.273 | 1.244 1.301 | <0.001 |
| Arthralgia (1) | 0.126 | 1.134 | 1.108 1.160 | <0.001 |
| Malaise (1) | 0.276 | 1.318 | 1.295 1.342 | <0.001 |
| Rhinorrhoea (1) | 0.138 | 1.148 | 1.127 1.169 | <0.001 |
| Polypnea (1) | 0.091 | 1.095 | 1.061 1.129 | <0.001 |
| Vomiting (1) | -0.034 | 0.966 | 0.934 1.000 | 0.049 |
| Abdominal pain (1) | -0.192 | 0.826 | 0.804 0.848 | <0.001 |
| Conjunctivitis (1) | -0.130 | 0.878 | 0.856 0.901 | <0.001 |
| Cyanosis (1) | 0.137 | 1.147 | 1.094 1.203 | <0.001 |
| SOS (1) | -0.120 | 0.887 | 0.872 0.902 | <0.001 |
Table 3: Multivariate Logistic Regression Analysis of Clinical and Demographic Factors associated to COVID-19 in Smoker Population.
| Variables | Beta | OR | CI 95% | P value |
|---|---|---|---|---|
| Age | -0.005 | 0.995 | 0.993 0.996 | <0.001 |
| Sex (1) | 0.059 | 1.061 | 1.019 1.104 | 0.004 |
| Fever (1) | 0.735 | 2.085 | 1.997 2.178 | <0.001 |
| Cough (1) | 0.385 | 1.469 | 1.406 1.535 | <0.001 |
| Odynophagia (1) | 0.034 | 1.034 | 0.990 1.080 | 0.129 |
| Dyspnoea (1) | 0.206 | 1.228 | 1.169 1.291 | <0.001 |
| Irritability (1) | -0.142 | 0.868 | 0.822 0.916 | <0.001 |
| Diarrhoea (1) | -0.022 | 0.978 | 0.931 1.028 | 0.379 |
| Chest pain (1) | 0.064 | 1.066 | 1.015 1.120 | 0.011 |
| Chills (1) | 0.203 | 1.225 | 1.168 1.286 | <0.001 |
| Headache (1) | -0.075 | 0.928 | 0.886 0.971 | 0.001 |
| Myalgia (1) | 0.256 | 1.291 | 1.219 1.368 | <0.001 |
| Arthralgia (1) | 0.123 | 1.131 | 1.067 1.198 | <0.001 |
| Malaise (1) | 0.301 | 1.351 | 1.291 1.414 | <0.001 |
| Rhinorrhoea (1) | 0.178 | 1.195 | 1.142 1.250 | <0.001 |
| Polypnea (1) | 0.099 | 1.104 | 1.026 1.188 | 0.008 |
| Vomiting (1) | -0.069 | 0.934 | 0.858 1.016 | 0.111 |
| Abdominal pain (1) | -0.211 | 0.810 | 0.760 0.863 | <0.001 |
| Conjunctivitis (1) | -0.146 | 0.865 | 0.816 0.916 | <0.001 |
| Cyanosis (1) | 0.301 | 1.351 | 1.206 1.512 | <0.001 |
| SOS (1) | -0.114 | 0.893 | 0.854 0.933 | <0.001 |
| Type 2 Diabetes (1) | 0.151 | 1.163 | 0.979 1.381 | 0.085 |
| COPD (1) | -0.101 | 0.904 | 0.728 1.121 | 0.358 |
| Asthma (1) | -0.319 | 0.727 | 0.591 0.895 | 0.003 |
| Immunosuppressed (1) | -0.309 | 0.734 | 0.569 0.948 | 0.018 |
| Hypertension (1) | -0.069 | 0.933 | 0.787 1.107 | 0.427 |
| HIV-AIDS (1) | -0.336 | 0.715 | 0.531 0.962 | 0.027 |
| CVD (1) | -0.212 | 0.809 | 0.655 0.999 | 0.049 |
| Obesity (1) | 0.082 | 1.086 | 0.920 1.280 | 0.330 |
| CKD (1) | 0.067 | 1.070 | 0.842 1.359 | 0.582 |
| Numbers of factors | 0.091 | 1.095 | 0.935 1.282 | 0.258 |
| Vaccinated (1) | -0.131 | 0.877 | 0.836 0.919 | <0.001 |
Discussion
This study provides a comprehensive analysis of the clinical characteristics and associated factors of COVID-19 among patients with upper respiratory tract infections, with a specific focus on differences between smokers and non-smokers. We observed key highlights in the clinical presentation of COVID-19 between smokers and non-smoker patients. Compared with the 40 international studies summarised in the systematic review and meta-analysis reported by Umnuaypornlert et al., our analysis introduces an important perspective by focusing on a younger population and the primary care context in low-and middle-income countries. The mean or median age of participants in most reviewed studies ranged from 19 to 74 years, with many studies focusing on hospitalised, high-risk, or elderly populations, particularly in countries such as the USA, China, Italy, and Spain. In contrast, our study population had a notably younger age distribution, with inclusion starting from 10 years of age, reflecting a broader, community-based sample that included both adolescents and adults.
While many of the referenced studies identified positive associations between smoking and increased disease severity or mortality of COVID-19, the strength and consistency of these associations varied widely across settings. Some studies, such as those conducted in Kuwait and Spain, reported high odds ratios (ORs > 2.0), indicating significant risk increases. However, several other studies—including those from the USA, China, and Japan—found no significant associations (often with wide confidence intervals due to limited sample sizes or methodological differences). Our findings reveal distinct patterns in symptom prevalence and risk factors between the two groups, with smoking status appearing to influence the overall clinical presentation and susceptibility to COVID-19. Although, smokers presented a more symptomatic clinical profile (with a higher prevalence of practically all reported signs and symptoms compared to non-smokers), traditional comorbidities such as obesity, diabetes, and hypertension did not increase the risk of COVID-19 within this group, in contrast to non-smokers, where such associations were significant. Therefore, this divergence suggests the presence of differential risk profiles or potential residual confounding related to access to testing, health-seeking behaviour, or biological mechanisms. Moreover, our population-based approach offers a more granular view of mild and moderate COVID-19 cases in primary care, in contrast to most previous studies based in hospitals or intensive care units, where patients already presented with severe disease. This difference in case severity and healthcare level can explain some of the differences observed between our findings and those of other studies.
Additionally, our findings align with prior evidence suggesting that tobacco use may exacerbate respiratory conditions and amplify symptom severity, probably due to altered native and adaptive immune responses. Headache, cough, and myalgia were the most frequently reported symptoms, followed by fever and sore throat. Additionally, smokers reported higher rates of respiratory symptoms such as dyspnoea, polypnoea, and chest pain, as well as systemic manifestations like malaise and chills. Although gastrointestinal symptoms such as diarrhoea and abdominal pain were present in a moderate proportion of smokers, where vomiting was relatively uncommon. Interestingly, symptoms like irritability and conjunctivitis were reported in over 10% of smokers, while cyanosis was rare (<5%). In contrast, non-smokers showed a slightly less intense clinical profile, though still consistent with typical viral respiratory illness. Fever was more prevalent in this group compared to smokers, which may reflect differences in systemic inflammatory responses. Common symptoms included headache, cough, myalgia, arthralgia, sore throat, and malaise, while diarrhoea, irritability, and conjunctivitis were reported in over 10% of non-smokers, other symptoms such as abdominal pain, vomiting, polypnoea, and cyanosis were relatively uncommon (<10%). The most plausible mechanism underlying the potential increase in risk may involve the angiotensin-converting enzyme 2 (ACE2) receptor, which is expressed in mucosal epithelial cells and alveolar lung tissue and has been identified as a key entry point for SARS-CoV-2. Notably, ACE2 gene expression is upregulated in smokers (current and former), and COPD patients compared to individuals who have never smoked. However, several studies have reported associations between ACE2 polymorphisms and components of metabolic syndrome (MetS) in populations from China and Brazil. In a southern Chinese population, the minor alleles of ACE2 rs2074192 and rs2106809 were significantly associated with increased risk of MetS in women. These variants were also linked to obesity, diabetes, and low HDL-cholesterolemia. Interestingly, in men, the rs2074192 minor allele was associated with a reduced prevalence of MetS and a decreased risk of obesity and elevated triglyceride levels, suggesting a gender-specific effect. Hence, these findings indicate that rs2074192 and rs2106809 SNPs are independently associated with MetS and could serve as predictive biomarkers and potential molecular targets, although part of the gender heterogeneity may be explained by differences in obesity.
In Brazil, a genetic association study explored ACE and ACE2 polymorphisms. While no individual association was found for either gene alone, a significant association emerged when both polymorphisms were considered jointly in women. Female carriers of the ACE DD genotype and ACE2 G_ variants had an increased risk of developing systemic arterial hypertension, with an even stronger association in DD/GG carriers. Additionally, the D allele of the ACE gene was associated with altered lipid profiles, particularly elevated triglycerides and VLDL-cholesterol levels (p = 0.01). One limitation of these studies is that they did not account for environmental, lifestyle, or genetic differences between populations, which may influence the observed associations. This is particularly relevant given that, in our study, we did not find an association between metabolic factors, COPD, and COVID-19 among smoker patients, despite the fact that increased expression of ACE2 has been consistently reported in this population. This apparent discrepancy may suggest that, although ACE2 expression is elevated in smokers, genetic variants such as ACE2 rs2074192 and rs2106809—which have been linked to components of metabolic syndrome—might contribute differently to disease susceptibility depending on sex, ethnicity, or the presence of other environmental and lifestyle factors not captured in our analysis.
On the other hand, multivariate logistic regression analysis revealed a broadly similar pattern of COVID-19-associated factors across both populations. Several signs and symptoms—such as fever, cough, dyspnoea, chest pain, chills, myalgia, arthralgia, malaise, rhinorrhoea, polypnoea, and cyanosis—were positively associated with COVID-19 in both smokers and non-smokers. Conversely, symptoms such as irritability, headache, abdominal pain, and conjunctivitis were negatively associated with the disease. Likewise, a sudden onset of symptoms and a history of asthma, immunosuppression, HIV/AIDS, or cardiovascular disease were all associated with a lower likelihood of COVID-19. Of particular interest, certain clinical characteristics and comorbidities —including odynophagia, diarrhoea, vomiting, COPD, and CKD—showed no significant association with COVID-19 in either group. However, in smokers specifically, obesity, type 2 diabetes, hypertension, and the number of comorbidities also lacked association with COVID-19, in contrast to non-smokers, where these conditions significantly increased risk (except the number of comorbidities). These findings may reflect differential pathophysiological interactions between smoking-related chronic conditions and SARS-CoV-2 infection. Furthermore, age demonstrated an inverse association with COVID-19 risk across both groups, suggesting younger individuals in this dataset may have been more frequently diagnosed, potentially due to surveillance or reporting bias.
On the other hand, vaccination emerged as a consistent protective factor in both populations, reaffirming its critical role in reducing susceptibility to COVID-19, irrespective of smoking status. Interestingly, the number of comorbidities was inversely associated with COVID-19 only in non-smokers, which may point to more cautious behaviour or better adherence to preventive measures in this subgroup.
From an epidemiological and public health standpoint, the inclusion of a wider age range and the analysis of primary care data allow for more representative conclusions about the broader impact of tobacco use on COVID-19 risk across the population. These insights are crucial for policy development, particularly in countries like Mexico, where the use of tobacco remains a major modifiable risk factor and access to healthcare is stratified by region and population group.
In terms of governance and public health planning, these findings suggest that universal risk communication strategies may not be sufficient, and that targeted messaging considering age, smoking status, and local epidemiological patterns may improve risk perception and preventive behaviour. Furthermore, they reinforce the importance of integrating primary care data into national COVID-19 surveillance efforts, which has often been underutilised in international comparisons.
Limitations of the study
This study has several limitations. First, as a cross-sectional study comparing smokers and non-smokers, the groups differed in size, although both were analysed using a census sampling approach within the available population. Our design restricts the ability to infer causal relationships, but allows for the identification of epidemiological associations that may serve as a basis for future longitudinal or interventional studies. Additionally, the observed differences in clinical presentation between smokers and non-smokers may reflect true biological variation, differences in gene-population background on ACE2 expression, or biases related to healthcare-seeking behaviour or data capture. Importantly, although ACE2 expression is known to be upregulated in smokers, we did not find a significant association between metabolic factors and COVID-19 in this subgroup, contrasting with genetic studies that link ACE2 polymorphisms to metabolic syndrome components. This apparent discrepancy underscores the complexity of gene-environment interactions and highlights the need for multicenter studies that integrate clinical, genetic, and environmental data to better understand the role of smoking in COVID-19 pathophysiology.
Conclusions
These findings underscore the importance of considering smoking status when evaluating clinical presentation and risk profiles for COVID-19. The differences observed between smokers and non-smokers highlight the need for tailored public health messaging and clinical management strategies, especially in populations with high tobacco use. Therefore, while both groups shared a core set of prevalent symptoms—such as headache, cough, and myalgia—smokers consistently exhibited a higher frequency of nearly all clinical manifestations, suggesting a more symptomatic or severe clinical profile. Moreover, the observed divergence in associated factors—particularly the lack of association between common comorbidities (e.g., obesity, hypertension, diabetes) and COVID-19 among smokers—points to potentially distinct biological or behavioural pathways influencing disease susceptibility and progression.
From an epidemiological perspective, these findings underscore the necessity of stratified analyses by smoking status in future surveillance and research efforts. Failure to account for such differences may obscure relevant associations or generate misleading conclusions regarding risk and symptomatology. In terms of public health, recognising that smokers may present with a broader and more intense range of symptoms is vital for early detection, triage, and targeted intervention strategies, especially in the context of respiratory viral outbreaks. Likewise, the differential risk profiles observed call for tailored preventive and therapeutic approaches that consider individual-level exposures such as tobacco use. For public policy, the evidence reinforces the urgent need to prioritise smoking cessation as a fundamental component of pandemic preparedness and chronic disease management. Public health campaigns and clinical guidelines should incorporate smoking status not only as a risk factor but also as a determinant of symptom severity. In the context of governance, these findings advocate for stronger intersectoral coordination between epidemiological intelligence, health promotion, and regulatory frameworks. Moreover, policies that integrate behavioural health with communicable disease control can enhance health system resilience, equity, and responsiveness, particularly in settings where tobacco consumption remains high. What is more, differentiating COVID-19 patterns by smoking status is not only clinically relevant but also essential for informed epidemiological modelling, effective public health action, evidence-based policymaking, and integrated health governance.
Acknowledgements
The authors would like to thank Professor Susana Ortiz Vela, Master in translation, and also express their gratitude to the Centro de Investigación y de Educación Continua S.C. for their support in translation.
Authors’ contributions
All authors contributed to conceptualization (ideas, formulation, or development of research goals and objectives), formal analysis (application of statistical, mathematical, computational, or other formal techniques to analyse or synthesize study data), writing – original draft (preparation, creation, and/or presentation of the published work, specifically writing the initial draft), writing – review and editing (preparation, creation, and/or presentation of the published work by the research group, specifically critical review, commentary, or revisions, including pre- or post-publication stages), and visualization (preparation, creation, and/or presentation of the published work, specifically data visualization/presentation).
Daniel López-Hernández, in addition to the above, contributed to project administration (responsibility for managing and coordinating the planning and execution of the research activity), investigation (development of a research process, specifically experiments or data collection/testing), methodology (development or design of methodology, creation of models), supervision (responsibility for supervision and leadership in the planning and execution of the research activity, including external mentoring), and validation (verification, whether as part of the activity or separately, of the overall replicability/reproducibility of the results/experiments and other research outcomes).
Conflict of interest
All authors declare not have interest conflict.
References
- Wood S, Harrison SE, Judd N, Bellis MA, Hughes K, Jones A. The impact of behavioural risk factors on communicable diseases: a systematic review of reviews. BMC Public Health. 2021;21(1):2110. Published 2021 Nov 17. doi:10.1186/s12889-021-12148-y
- Xu X, Shi Z, Zhou L, et al. Impact of COVID-19 on risks and deaths of non-communicable diseases in the Western Pacific region. Lancet Reg Health West Pac. 2023;43:100795. Published 2023 Dec 1. doi:10.1016/j.lanwpc.2023.100795
- Hamer M, Kivimäki M, Gale CR, Batty GD. Lifestyle risk factors, inflammatory mechanisms, and COVID-19 hospitalization: A community-based cohort study of 387,109 adults in UK. Brain Behav Immun. 2020;87:184-187. doi:10.1016/j.bbi.2020.05.059
- Hernández-Pérez A, García-Gómez L, Rodríguez-Llamazares S, Thirión-Romero I, Osio-Echánove J, Pérez-Padilla R. Associated risks of smoking and possible benefits of cessation in Covid-19: a rapid narrative review. Salud Publica Mex. 2021;63(2, Mar-Abr):262-267. Published 2021 Feb 26. doi:10.21149/9371
- Umnuaypornlert A, Kanchanasurakit S, Lucero-Prisno DEI, Saokaew S. Smoking and risk of negative outcomes among COVID-19 patients: A systematic review and meta-analysis. Tob Induc Dis. 2021;19:09. Published 2021 Feb 4. doi:10.18332/tid/132411
- López-Hernández D. Clinical Presentation and Factors Associated to COVID-19 Disease in Mexican Patients. CJAST. 2022,41(1):40-58.
- Puebla Neira D, Watts A, Seashore J, Polychronopoulou E, Kuo YF, Sharma G. Smoking and risk of COVID-19 hospitalization. Respir Med. 2021;182:106414. doi:10.1016/j.rmed.2021.106414
- Salehi Z, Motlagh Ghoochani BFN, Hasani Nourian Y, Jamalkandi SA, Ghanei M. The controversial effect of smoking and nicotine in SARS-CoV-2 infection. Allergy Asthma Clin Immunol. 2023;19(1):49. Published 2023 Jun 1. doi:10.1186/s13223-023-00797-0
- Haddad C, Bou Malhab S, Sacre H, Salameh P. Smoking and COVID-19: A Scoping Review. Tob Use Insights. 2021;14:1179173X21994612. Published 2021 Feb 15. doi:10.1177/1179173X21994612
- Paleiron N, Mayet A, Marbac V, et al. Impact of Tobacco Smoking on the Risk of COVID-19: A Large Scale Retrospective Cohort Study. Nicotine Tob Res. 2021;23(8):1398-1404. doi:10.1093/ntr/ntab004
- van Westen-Lagerweij NA, Meijer E, Meeuwsen EG, Chavannes NH, Willemsen MC, Croes EA. Are smokers protected against SARS-CoV-2 infection (COVID-19)? The origins of the myth. NPJ Prim Care Respir Med. 2021;31(1):10. Published 2021 Feb 26. doi:10.1038/s41533-021-00223-1
- Almazeedi S, Al-Youha S, Jamal MH, et al. Characteristics, risk factors and outcomes among the first consecutive 1096 patients diagnosed with COVID-19 in Kuwait. EClinicalMedicine. 2020;24:100448. Published 2020 Jul 4. doi:10.1016/j.eclinm.2020.100448
- Torres-Macho J, Ryan P, Valencia J, et al. The PANDEMYC Score. An Easily Applicable and Interpretable Model for Predicting Mortality Associated With COVID-19. J Clin Med. 2020;9(10):3066. Published 2020 Sep 23. doi:10.3390/jcm9103066
- Strzelak A, Ratajczak A, Adamiec A, Feleszko W. Tobacco Smoke Induces and Alters Immune Responses in the Lung Triggering Inflammation, Allergy, Asthma and Other Lung Diseases: A Mechanistic Review. Int J Environ Res Public Health. 2018;15(5):1033. Published 2018 May 21. doi:10.3390/ijerph15051033
- Arcavi L, Benowitz NL. Cigarette smoking and infection. Arch Intern Med. 2004;164(20):2206-2216. doi:10.1001/archinte.164.20.2206
- Bauer CMT, Morissette MC, Stämpfli MR. The influence of cigarette smoking on viral infections: translating bench science to impact COPD pathogenesis and acute exacerbations of COPD clinically. Chest. 2013;143(1):196-206. doi:10.1378/chest.12-0930
- Bourgonje AR, Abdulle AE, Timens W, et al. Angiotensin-converting enzyme 2 (ACE2), SARS-CoV-2 and the pathophysiology of coronavirus disease 2019 (COVID-19). J Pathol. 2020;251(3):228-248. doi:10.1002/path.5471
- Beyerstedt S, Casaro EB, Rangel ÉB. COVID-19: angiotensin-converting enzyme 2 (ACE2) expression and tissue susceptibility to SARS-CoV-2 infection. Eur J Clin Microbiol Infect Dis. 2021;40(5):905-919. doi:10.1007/s10096-020-04138-6
- Leung JM, Yang CX, Tam A, et al. ACE-2 expression in the small airway epithelia of smokers and COPD patients: implications for COVID-19. Eur Respir J. 2020;55(5):2000688. Published 2020 May 14. doi:10.1183/13993003.00688-2020
- Pan M, Yu M, Zheng S, Luo L, Zhang J, Wu J. Genetic variations in ACE2 gene associated with metabolic syndrome in southern China: a case-control study. Sci Rep. 2024;14(1):10505. Published 2024 May 7. doi:10.1038/s41598-024-61254-5
- Pinheiro DS, Santos RS, Jardim PCBV, et al. The combination of ACE I/D and ACE2 G8790A polymorphisms reveals susceptibility to hypertension: A genetic association study in Brazilian patients. PLoS One. 2019;14(8):e0221248. Published 2019 Aug 20. doi:10.1371/journal.pone.0221248

