COVID-19 Vaccine-Induced Myopericarditis: Insights & Management
COVID-19 Vaccine-Induced Subclinical Myopericarditis: Pathophysiology, Diagnosis, and Clinical Management
Peter A. McCullough, MD, MPH¹*, M. Nathaniel Mead, MSc, PhD¹, Nicolas Hulscher, MPH¹
- 16320 LBJ Freeway, Suite 221, Dallas, Texas 75240
OPEN ACCESS
PUBLISHED: 30 November 2025
CITATION:McCullough, PA., Mead, MN., Hulscher, N., 2025. COVID-19 Vaccine-Induced Subclinical Myopericarditis: Pathophysiology, Diagnosis, and Clinical Management. Medical Research Archives, [online] 13(11).
https://doi.org/10.18103/mra.v13i11.7078
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i11.7078
ISSN 2375-1924
ABSTRACT
The currently approved COVID-19 mRNA boosters carry a warning for acute, clinically apparent, vaccine-induced myocarditis. This serious condition has resulted in hospitalization and, in well-documented cases, fatalities. However, there is growing concern that long-lasting synthetic mRNA and persistent production of SARS-CoV-2 Spike protein may accumulate in the heart and cause cardiotoxicity over time. Additionally, subtler cardiovascular symptoms, which may not require immediate hospitalization, can also develop. Such symptoms include atypical or pleuritic chest pain, palpitations, intermittent arrhythmias, labile blood pressure with hyper- and hypotension and effort intolerance. Areas of inflammation found at autopsy too small to be detectable by cardiac magnetic resonance imaging have been associated with sudden death. Alarmingly, the initial presentation can include cardiac arrest with no premonitory symptoms. A thorough evaluation, including history of SARS-CoV-2 infections, the number and type of mRNA COVID-19 vaccines received, quantitative Spike protein antibody levels, ECG, imaging, and laboratory tests, form the cornerstone of initial assessment. Clinical and preclinical observations suggest combined oral administration of nattokinase, bromelain, and curcumin may support detoxification of the heart and cardiovascular system from Spike protein. The addition of colchicine and other targeted therapies may be essential in reducing myocardial and systemic vascular inflammation. These approaches hold promise to risk mitigation of sudden cardiac death in immunized individuals affected by subclinical COVID-19 vaccine induced myopericarditis.
Keywords
COVID-19, vaccine, myocarditis, myopericarditis, Spike protein, mRNA, cardiovascular health
Introduction
The COVID-19 pandemic has driven rapid development and deployment of genetic vaccines, notably the synthetic modified mRNA products, BNT162b2 and mRNA-1273, manufactured by Pfizer-BioNtech and Moderna, respectively. Significant controversy regarding COVID-19 mRNA vaccine safety has arisen based largely on postmarketing studies, autopsy studies, and careful reanalysis of the registrational trials that led to emergency use authorization of the products. Nevertheless, it is estimated that 81% of Americans took one or more doses. Among the myriad of safety concerns, subclinical myopericarditis following mRNA vaccination has emerged as an area of ongoing clinical research. Subclinical myopericarditis refers to inflammation of the myocardium (heart muscle) and pericardium (the sac surrounding the heart) detected primarily through laboratory or imaging findings, without overt or pronounced clinical symptoms. Epidemiologists and cardiologists alike have voiced concern that subclinical vaccine-induced myopericarditis could explain changes in cardiac arrest rates at a population level. Most cases of myocarditis are more accurately referred to as myopericarditis, as cardiac MRI often reveals concurrent pericardial involvement, such as thickening or effusion, alongside myocardial inflammation. Symptoms may include chest pain, palpitations, or shortness of breath, but in subclinical cases, individuals remain asymptomatic, and the condition is usually detected through elevated biomarkers, electrocardiogram (ECG) changes, or imaging studies such as cardiac MRI.
In this paper, we synopsize key aspects of COVID-19 mRNA vaccine-induced subclinical myocarditis in terms of etiopathogenesis, epidemiology, diagnostic challenges, clinical care issues, policy implications, and future research directives. We outline a comprehensive evaluation protocol involving medical history, Spike protein antibody levels, ECG, imaging, and laboratory tests to assess vaccine-induced myopericarditis and related conditions. Our empirically supported approach to mitigating myocardial and vascular inflammation is aimed at reducing the elevated rates of sudden cardiac death observed in mRNA-vaccinated individuals with no other identifiable cause.
Etiology and Pathogenesis
The pathogenesis of myopericarditis is typically associated with viral infections, autoimmune conditions, or rare reactions to medications and vaccines. In the context of COVID-19 vaccination, the mRNA-based vaccines have been most commonly associated with cases of vaccine-induced myopericarditis. The exact mechanisms remain under investigation but are hypothesized to involve immune-mediated processes triggered by the mRNA vaccine’s stimulation of the immune system to produce Spike protein antigens within the myocardium as well as in the systemic circulation. Several studies provide strong biological plausibility for a causal relationship between the COVID-19 mRNA vaccinations and myocarditis. Upon autopsy of mRNA vaccinees, Krauson et al. isolated synthetic mRNA (Pfizer/Moderna) in the human heart, strongly indicating cardiac involvement. Baumeier and colleagues found vaccine Spike protein in the hearts of young men suffering from vaccine myopericarditis. Yonker et al. described unbound circulating Spike protein in blood of young persons hospitalized with COVID-19 vaccine-induced myopericarditis, while a vaccinated asymptomatic control group had appropriately neutralized Spike with anti-Spike antibodies. Thus, the inflammation of myopericarditis appears to be secondary to locally present vaccine mRNA and Spike protein. The time course for myocardial accumulation of these factors is unknown but conceivably could occur over several years with chronic Spike protein circulation in the blood, myocardial perfusion, and deposition into the coronary arteries, capillary endothelium and the myocardial tissue.
Epidemiology, Risks, and Outcomes
Myopericarditis following COVID-19 mRNA vaccination is well-documented, occurring in all age groups for both genders. The majority of cases, however, have occurred in young males (
The incidence of subclinical myopericarditis is challenging to estimate, as it requires sensitive screening and is not associated with symptoms that would prompt clinical evaluation. Chiu et al found among 763 students a rate of 17.1% had at least one cardiac symptom after the second vaccine dose, mostly chest pain and palpitations. Three prospective cohort studies have evaluated the incidence of subclinical myopericarditis after injection of synthetic mRNA. Mansanguan et al. reported an incidence rate of 2.3%, Buergin et al. reported an incidence rate of 2.8%, and a Pfizer-sponsored study yielded an estimated incidence rate of 1.0%, though that study omitted daily cardiac troponin measurements. It is reasonable to surmise that approximately 1-3% of COVID-19 mRNA vaccine recipients suffer some degree of myocardial injury per dose, thereby representing a profound concern for the short- and long-term health of younger vaccinees.
Population-level studies, such as cardiac MRI screening in selected cohorts, have identified a higher incidence of subclinical myocarditis than previously recognized, again mostly in adolescent and young adult males. Nonetheless, most observational studies are ineffectual in this determination because they do not involve careful clinical examinations, imaging, and measurements of cardiac biomarkers in all subjects before and after vaccine administration. Without prospective monitoring of cardiac function, subclinical myopericarditis cases following COVID-19 vaccinations are grossly underreported in large cohort and surveillance studies.
Despite the mild outward or even asymptomatic appearance of subclinical myocarditis, many cases can involve severe cardiac fibrosis (scarring), leading to permanent heart muscle damage and a lifelong risk of fatal arrhythmias. This damage may eventually progress to congestive heart failure and premature death. Physical stress, such as vigorous exercise, may trigger cardiac arrest in younger adults with either clinical or subclinical myocarditis due to an adrenaline surge, potentially contributing to fatal outcomes.
Clinical Presentation of Subclinical Myopericarditis
Unlike overt myopericarditis, subclinical cases may lack obvious symptoms.
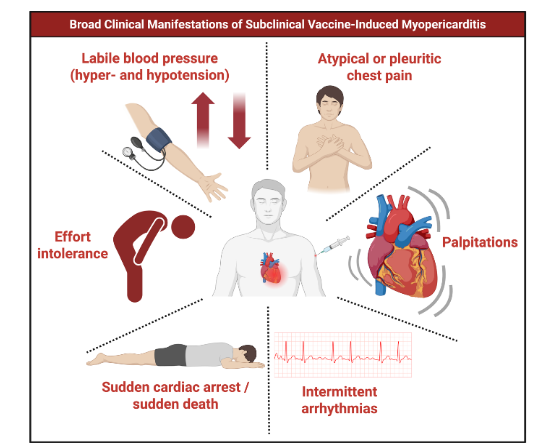
Subclinical myopericarditis may be suspected, identified, and supported by the following: 1) new symptoms arising shortly after mRNA vaccination, 2) labile blood pressure and heart rate (postural orthostatic tachycardia syndrome, or POTS), 3) elevated cardiac biomarkers (e.g., troponin I or T, BNP, galectin-3, d-dimer), 4) markedly elevated quantitative antibodies to Spike antigen, 5) presence of circulatory Spike protein, 6) presence of persistent vaccine mRNA in blood or tissue, 7) abnormal electrocardiogram (ECG) findings (e.g., ST-segment changes, arrhythmias), 8) imaging abnormalities (e.g., late gadolinium enhancement on cardiac MRI). Any combination of these factors, particularly when observed in the context of recent mRNA vaccination, could aid in the detection of subclinical myopericarditis. In certain cases, however, the initial manifestation may be catastrophic, with sudden cardiac arrest or sudden death representing the most severe end of the clinical continuum of vaccine-induced subclinical myopericarditis. This possibility underscores the importance of proactive screening and surveillance to detect subclinical cases early, before they progress to life-threatening outcomes.
Diagnostic Tools and Criteria
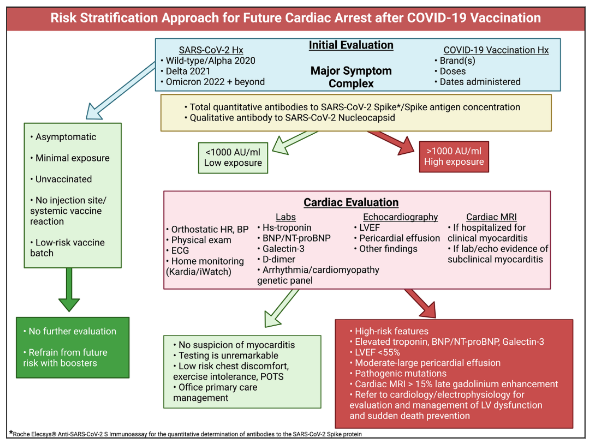
Diagnosis of and risk stratification for subclinical myopericarditis typically depends on a combination of laboratory and imaging findings: 1) laboratory Tests: high-sensitivity troponin, BNP, galectin-3, d-dimer assays may reveal mild to moderate elevations, suggestive of myocardial injury, 2) electrocardiography: non-specific changes such as mild to moderate ST-segment or T-wave abnormalities may be present, 3) cardiac imaging: echocardiographic evidence of ventricular dysfunction and or pericardial effusion and cardiac MRI evidence of myocardial inflammation, with findings such as late gadolinium enhancement and edema. Large pericardial effusions warrant serologic testing for the antinuclear antibody and may require drainage.
Because these patients lack overt severe symptoms, diagnosis is often incidental or made in the context of research studies or screening protocols. However, recent evidence suggests that a more comprehensive risk stratification approach is warranted. A Spike Protein exposure history: Each SARS-CoV-2 infection or mRNA vaccination represents a cumulative exposure. A higher number of exposures correlates with greater risk of persistent Spike protein in circulation and myocardium. Anti-Spike antibody concentration is prognostic and available in quantitative antibody assays (e.g., Roche Elecsys) serving as proxies for prior Spike exposure. Low titers (<1000 U/mL) suggest lower risk, whereas persistently high titers (>1000 U/mL, sometimes >25,000 U/mL for years) indicate heightened risk. Genetic testing is prudent to understand predisposition in high-risk patients or resuscitated cardiac arrest cases; genetic screening for ion channelopathies (e.g., SCN5A mutations) may identify inherited susceptibility. Cardiac MRI with contrast demonstrating LGE indicates fibrosis. In other cardiomyopathies, late gadolinium enhancement ≥15% of the left ventricle has been predictive of sudden cardiac arrest and may serve as a high-risk marker here as well. Autopsy studies have indicated, however, that small patches of inflammation too small to be detected by cardiac MRI can be the presumed source of fatal re-entrant arrhythmias without the antecedent development of left ventricular dysfunction. Thus a normal cardiac MRI does not rule out the presence of potentially lethal subclinical COVID-19 vaccine myopericarditis.
Risk modifiers include vaccine batch-to-batch variability, younger age, male sex, cumulative doses, and shorter intervals between doses should be incorporated into patient evaluation. Collectively, this expanded diagnostic and risk stratification framework allows clinicians to identify patients with silent subclinical myopericarditis who might otherwise appear healthy, thereby facilitating early intervention and potentially preventing sudden cardiac arrest.
Clinical Course and Prognosis
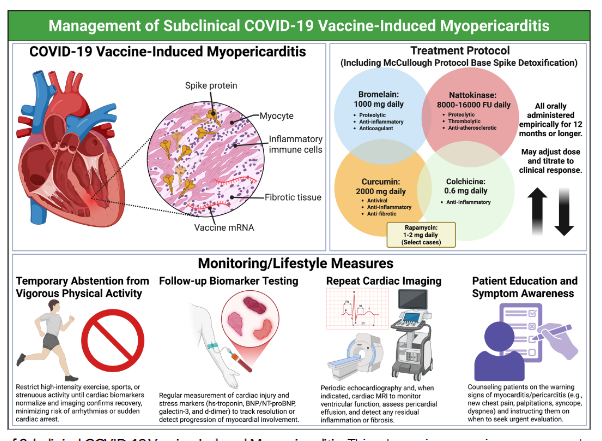
Most reported cases of vaccine-induced subclinical myopericarditis are self-limited, with normalization of biomarkers and imaging findings within weeks to months. It is critical to understand the importance of long-lasting tissue Spike deposition in this disease. Resolution of this issue is potentially greatly assisted with Spike protein detoxification combined with colchicine over the course of a year or more. The proposed detoxification includes nattokinase 8000-16000 FU, bromelain 500-1000 mg, and curcumin 500-1000 mg daily (McCullough Protocol Base Spike Detoxification).™ Severe outcomes, such as persistent cardiac dysfunction or arrhythmias, are common and managed in accordance with current guidelines for subclinical cases.
Management Strategies
For subclinical cases, management is largely conservative, as patients are asymptomatic and recover spontaneously. Monitoring may include follow-up biomarker testing and repeat cardiac imaging to confirm resolution. Recommendations may include: 1) temporary abstention from vigorous physical activity until biomarkers and imaging normalize and a reasonable period of detoxification has been performed, 2) Spike detoxification with oral nattokinase, bromelain, and curcumin, 3) colchicine, 4) rapamycin (select cases). Pharmacologic therapy is generally reserved for those with symptoms or evidence of significant inflammation or cardiac dysfunction.
Implications for Vaccine Policy
Recognition of subclinical myopericarditis has informed COVID-19 vaccine policy, particularly for young males with warnings in US Food and Drug Administration package inserts for Pfizer and Moderna mRNA vaccines. Some countries have recommended alternative dosing intervals, use of specific vaccine brands, or additional monitoring in higher-risk groups. Others have called for the complete market removal of COVID-19 vaccines to fully avoid any more cardiac injury on a population level.
Comparative Risks
The risk of myopericarditis from the mRNA inoculations is substantially higher and may be more severe than purported infection-associated cases. COVID-19 vaccination, not SARS-CoV-2 infection, remains the main culprit in serious illness, hospitalization, and death after injection. Historically, no other group of vaccine products has represented such a clear, large-scale risk. An analysis of data from the Vaccine Adverse Event Reporting System (VAERS) indicated that myocarditis reports following the 2021 COVID-19 vaccine rollout from recipients were statistically at a 223 times greater volume than the average for all vaccines over the prior 30 years.
Research Gaps and Future Directions
Ongoing research aims to clarify: 1) true cumulative incidence of subclinical myopericarditis through population screening studies, 2) mechanisms underlying synthetic mRNA and Spike damage to the myocardium and the immune response leading to further cardiac involvement, 3) long-term outcomes of subclinical and mild clinical cases including heart failure and sudden death, 4) clinical, serological, and genetic markers of susceptibility. Continued surveillance and reporting will help refine vaccine recommendations and ensure optimal safety.
Patient and Public Communication
Clear communication honoring the precautionary principle is essential to maintain vaccine safety and ensure individuals are aware of the potential risks, including sudden cardiac death. Health professionals should be vigilant in cases of subclinical myopericarditis, while encouraging Spike detoxification as a safe and easily implemented health care measure, at least until more research is completed.
Limitations
We recognize that large, prospective, blinded, triple-dummy, placebo-controlled trials will be required to make regulatory claims concerning one or more components of Spike detoxification. To our knowledge, no such trials have been proposed and budgeted let alone organized and started enrollment. As years continue to pass in the pandemic there is a greater need for empirical management with close clinical observations. Such a conclusive trial from funding to publication could take 5 to 20 years.
Conclusion
COVID-19 vaccine-induced subclinical myopericarditis may be more common than has been officially recognized, primarily occurring among younger males following mRNA vaccination. However, the true prevalence and incidence of subclinical cases is unknown due to lack of routine cardiac screening for functional complications post-vaccination in the general population. Persistent SARS-CoV-2 Spike protein accumulation in the heart may lead to subtle cardiovascular symptoms, cardiac arrest, or sudden death. Most cases resolve with Spike protein detoxification alone with some requiring colchicine. The theoretical benefits of vaccination continue to be far outweighed by the risks of subclinical vaccine myopericarditis. Ongoing research will further elucidate the mechanisms, true incidence, and long-term outcomes of this phenomenon, supporting safe and effective public health strategies and timely medical care.
Disclosures
NH is an employee of the McCullough Foundation, NMM has received payments from the McCullough Foundation, PM is the President of the McCullough Foundation without compensation, PM is the Chief Scientific Officer of The Wellness Company (salary support, minor equity interest) which markets the Ultimate Spike Detox formulated in part from McCullough Protocol Base Spike Detoxification™ a combination of nattokinase, bromelain, curcumin, selenium, turmeric, black seed extract, dandelion extract, and black pepper fruit extract. The Wellness Company did not have input or play any role in writing this manuscript.
Conflict of Interest Statement: None.
Funding Statement: None.
Acknowledgements: None.
ORCID
1.Peter A. McCullough – 0000-0002-0997-6355
2.M. Nathaniel Mead – 0009-0003-3574-4675
3.Nicolas Hulscher – 0009-0008-0677-7386
References
- Fraiman J, Erviti J, Jones M, Greenland S, Whelan P, Kaplan RM, Doshi P. Serious adverse events of special interest following mRNA COVID-19 vaccination in randomized trials in adults. Vaccine. 2022;40(40):5798-5805. doi: 10.1016/j.vaccine.2022.08.036
- Michels C, Perrier D, Kunadhasan J, Clark E, Gehrett J, Gehrett B, Kwiatek K, et al. Forensic analysis of the 38 subject deaths in the 6-Month Interim Report of the Pfizer/BioNTech BNT162b2 mRNA Vaccine Clinical Trial. IJVTPR 2023; 3(1): 973-1009. doi: https://doi.org/10.56098/ijvtpr.v3i1.85
- Gøtzsche PC, Demasi M. Serious harms of the COVID-19 vaccines: a systematic review. medRxiv Preprint. 2022. doi.org: 10.1101/2022.12.06.22283145
- Pfizer, Inc. Appendix 2.2 Cumulative and Interval Summary Tabulation of Serious and Non-serious Adverse Reactions From Post-marketing Data Sources (BNT162B2). New York, NY. 2022. Accessed: December 9, 2024: https://www.globalresearch.ca/wp-content/uploads/2023/05/pfizer-report.pdf
- Horowitz D. Confidential Pfizer document shows the company observed 1.6 million adverse events covering nearly every organ system. 2023. Accessed: December 9, 2024: https://www.conservativereview.com/horowitz-confidential-pfizer-document-shows-the-company-observed-1-6-million-adverse-events-covering-nearly-every-organ-system-2661316948.html
- Hulscher N, Hodkinson R, Makis W, McCullough PA. Autopsy findings in cases of fatal COVID-19 vaccine-induced myocarditis. ESC Heart Fail. 2024 Jan 14. doi: 10.1002/ehf2.14680. Epub ahead of print
- USA Facts. US Coronavirus vaccine tracker. Latest update on 10 May 2025. Accessed on 7 Sept. 2025. https://usafacts.org/visualizations/covid-vaccine-tracker-states/
- Mead MN, Seneff S, Wolfinger R, Rose J, Denhaerynck K, Kirsch S, McCullough PA. COVID-19 Modified mRNA “Vaccines” Part 1: Lessons Learned from Clinical Trials, Mass Vaccination, and the Bio-Pharmaceutical Complex. Vaccine Theory Prac & Res. 2024;3(1):1112-1178. doi:10.56098/fdrasy50. https://doi.org/10.56098/fdrasy50.
- Mead MN, Seneff S, Rose J, Wolfinger R, Hulscher N, McCullough PA. COVID-19 Modified mRNA “Vaccines” Part 2: Lessons Learned from Clinical Trials, Mass Vaccination, and the Bio-Pharmaceutical Complex. Vaccine Theory Prac & Res. 2024; 3(2), 1275-1344. https://doi.org/10.56098/w66wjg87.
- Mead MN, Rose J, Makis W, Milhoan K, Hulscher N, McCullough PA. Myocarditis after SARS-CoV-2 infection and COVID-19 vaccination: Epidemiology, outcomes, and new perspectives. Int J Cardiovasc Res Innov. 2025; 3(1):1-43. https://doi.org/10.61577/ijcri.2025.100001
- Koizumi T, Ono M. Cardiac Multiple Micro-Scars: An Autopsy Study. JACC Case Rep. 2025;30(5):103083. doi: 10.1016/j.jaccas.2024.103083.
- Maruyama T, Uesako H. Lessons Learnt from Case Series of Out-of-hospital Cardiac Arrest and Unexpected Death after COVID-19 Vaccination. Intern Med. 2023;62(22):3267-3275. doi: 10.2169/internalmedicine.2298-23.
- Hulscher N, Cook MJ, Stricker RB, McCullough PA. Excess Cardiopulmonary Arrest and Mortality after COVID-19 Vaccination in King County, Washington. J Emerg Med OA, 2024; 2(1), 01-11. https://www.opastpublishers.com/open-access-articles/excess-cardiopulmonary-arrest-and-mortality-after-covid19-vaccination-in-king-county-washington.pdf
- Choi S, Lee S, Seo JW, Kim MJ, Jeon YH, Park JH, Lee JK, Yeo NS. Myocarditis-induced Sudden Death after BNT162b2 mRNA COVID-19 Vaccination in Korea: Case Report Focusing on Histopathological Findings. J Korean Med Sci. 2021;36(40):e286. doi: 10.3346/jkms.2021.36.e286.
- Gill JR, Tashjian R, Duncanson E. Autopsy Histopathologic Cardiac Findings in 2 Adolescents Following the Second COVID-19 Vaccine Dose. Arch Pathol Lab Med. 2022;146(8):925-929. doi: 10.5858/arpa.2021-0435-SA.
- Hulscher N, Alexander PE, Amerling R, Gessling H, Hodkinson R, Makis W, Risch HA, et al. A systematic review of autopsy findings in deaths after COVID-19 vaccination. Science, Public Health Policy and the Law. 2024; v5.2019-2024 https://publichealthpolicyjournal.com/a-systematic-review-of-autopsy-findings-in-deaths-after-covid-19-vaccination/
- Caforio AL, Pankuweit S, Arbustini E, Basso C, Gimeno-Blanes J, Felix SB, Fu M, et al.; European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013;34(33):2636-48, 2648a-2648d. doi: 10.1093/eurheartj/eht210
- Lurz P, Eitel I, Klieme B, Luecke C, de Waha S, Desch S, Fuernau G, Klingel K, et al. The potential additional diagnostic value of assessing for pericardial effusion on cardiac magnetic resonance imaging in patients with suspected myocarditis. Eur Heart J Cardiovasc Imaging. 2014;15(6):643-50. doi: 10.1093/ehjci/jet267
- Marschner CA, Shaw KE, Tijmes FS, Fronza M, Khullar S, Seidman MA, Thavendiranathan P, et al. Myocarditis Following COVID-19 Vaccination. Cardiol Clin. 2022;40(3):375-388. doi: 10.1016/j.ccl.2022.05.002
- Spotts PH, Zhou F. Myocarditis and Pericarditis. Prim Care. 2024;51(1):111-124. doi: 10.1016/j.pop.2023.07.006
- Gao J, Feng L, Li Y, Lowe S, Guo Z, Bentley R, Xie C, et al. A Systematic Review and Meta-analysis of the Association Between SARS-CoV-2 Vaccination and Myocarditis or Pericarditis. Am J Prev Med. 2023;64(2):275-284. doi: 10.1016/j.amepre.2022.09.002
- Yasmin F, Najeeb H, Naeem U, Moeed A, Atif AR, Asghar MS, Nimri N, et al. Adverse events following COVID-19 mRNA vaccines: A systematic review of cardiovascular complication, thrombosis, and thrombocytopenia. Immun Inflamm Dis. 2023 Mar;11(3):e807. doi: 10.1002/iid3.807
- Polykretis P, Donzelli A, Lindsay JC, Wiseman D, Kyriakopoulos AM, Mörz M, Bellavite P, Fukushima M, Seneff S, McCullough PA. Autoimmune inflammatory reactions triggered by the COVID-19 genetic vaccines in terminally differentiated tissues. Autoimmunity. 2023;56(1):2259123. doi: 10.1080/08916934.2023.2259123.
- Krauson AJ, Casimero FVC, Siddiquee Z, Stone JR. Duration of SARS-CoV-2 mRNA vaccine persistence and factors associated with cardiac involvement in recently vaccinated patients. NPJ Vaccines. 2023;8(1):141. doi: 10.1038/s41541-023-00742-7.
- Baumeier C, Aleshcheva G, Harms D, Gross U, Hamm C, Assmus B, Westenfeld R, et al. Intramyocardial Inflammation after COVID-19 Vaccination: An Endomyocardial Biopsy-Proven Case Series. Int J Mol Sci. 2022;23(13):6940. doi: 10.3390/ijms23136940.
- Yonker LM, Swank Z, Bartsch YC, Burns MD, Kane A, Boribong BP, Davis JP, et al. Circulating Spike Protein Detected in Post-COVID-19 mRNA Vaccine Myocarditis. Circulation. 2023;147(11):867-876. doi: 10.1161/CIRCULATIONAHA.122.061025.
- Pillay J, Gaudet L, Wingert A, Bialy L, Mackie AS, Paterson DI, Hartling L. Incidence, risk factors, natural history, and hypothesised mechanisms of myocarditis and pericarditis following Covid-19 vaccination: living evidence syntheses and review. BMJ. 2022;378:e069445. doi: 10.1136/bmj-2021-069445
- Karlstad Ø, Hovi P, Husby A, Härkänen T, Selmer RM, Pihlström N, Hansen JV, et al. SARS-CoV-2 Vaccination and Myocarditis in a Nordic Cohort Study of 23 Million Residents. JAMA Cardiol. 2022;7(6):600-612. doi: 10.1001/jamacardio.2022.0583
- Chiu SN, Chen YS, Hsu CC, Hua YC, Tseng WC, Lu CW, Lin MT, Chen CA, Wu MH, Chen YT, Chien TH, Tseng CL, Wang JK. Changes of ECG parameters after BNT162b2 vaccine in the senior high school students. Eur J Pediatr. 2023 Mar;182(3):1155-1162. doi: 10.1007/s00431-022-04786-0. Epub 2023 Jan 5. PMID: 36602621; PMCID: PMC9813456.
- Mansanguan S, Charunwatthana P, Piyaphanee W, Dechkhajorn W, Poolcharoen A, Mansanguan C. Cardiovascular Manifestation of the BNT162b2 mRNA COVID-19 Vaccine in Adolescents. Trop Med Infect Dis. 2022;7(8):196. doi: 10.3390/tropicalmed7080196.
- Buergin N, Lopez-Ayala P, Hirsiger JR, Mueller P, Median D, Glarner N, Rumora K, Herrmann T, Koechlin L, Haaf P, Rentsch K, Battegay M, Banderet F, Berger CT, Mueller C. Sex-specific differences in myocardial injury incidence after COVID-19 mRNA-1273 booster vaccination. Eur J Heart Fail. 2023;25(10):1871-1881. doi: 10.1002/ejhf.2978
- Albertson TE, Hansen C, Bihari S, Gayed J, Xu X, Simón-Campos JA, Dever ME, et al. C4591031, C4591007 Clinical Trial Groups. Serum Troponin I Assessments in 5- to 30-Year-Olds After BNT162b2 Vaccination. Infect Dis Ther. 2024;13(4):699-714. doi: 10.1007/s40121-024-00927-0.
- Klement RJ, Walach H. Commentary: raised c-troponin levels as a sign of myocardial injury after COVID-19 vaccination in healthy individuals are worrying. Egypt Heart J. 2024;76(1):16. doi: 10.1186/s43044-024-00441-1.
- Warren J, Cheng D, Crawford N, Jones B, Ng RL, Alafaci A, Stub D, Lew P, Taylor AJ. Improved diagnosis of COVID-19 vaccine-associated myocarditis with cardiac scarring identified by cardiac magnetic resonance imaging. Open Heart. 2025;12(2):e003333. doi: 10.1136/openhrt-2025-003333.
- Barmada A, Klein J, Ramaswamy A, Brodsky NN, Jaycox JR, Sheikha H, Jones KM, Habet V, et al. Cytokinopathy with aberrant cytotoxic lymphocytes and profibrotic myeloid response in SARS-CoV-2 mRNA vaccine-associated myocarditis. Sci Immunol. 2023;8(83):eadh3455. doi: 10.1126/sciimmunol.adh3455.
- Yu CK, Tsao S, Ng CW, Chua GT, Chan KL, Shi J, Chan YY, Ip P, Kwan MY, Cheung YF. Cardiovascular Assessment up to One Year After COVID-19 Vaccine-Associated Myocarditis. Circulation. 2023;148(5):436-439. doi: 10.1161/CIRCULATIONAHA.123.064772
- Brociek E, Tymińska A, Giordani AS, Caforio ALP, Wojnicz R, Grabowski M, Ozierański K. Myocarditis: Etiology, Pathogenesis, and Their Implications in Clinical Practice. Biology (Basel). 2023;12(6):874. doi: 10.3390/biology12060874.
- Cadegiani FA. Catecholamines Are the Key Trigger of COVID-19 mRNA Vaccine-Induced Myocarditis: A Compelling Hypothesis Supported by Epidemiological, Anatomopathological, Molecular, and Physiological Findings. Cureus. 2022;14(8):e27883. doi: 10.7759/cureus.27883
- Alami A, Villeneuve PJ, Farrell PJ, Mattison D, Farhat N, Haddad N, Wilson K, et al. Myocarditis and Pericarditis Post-mRNA COVID-19 Vaccination: Insights from a Pharmacovigilance Perspective. J Clin Med. 2023;12(15):4971. doi: 10.3390/jcm12154971
- Piccirillo F, Watanabe M, Di Sciascio G. Diagnosis, treatment and predictors of prognosis of myocarditis. A narrative review. Cardiovasc Pathol. 2021;54:107362. doi: 10.1016/j.carpath.2021.107362
- McCullough PA, Hulscher N. Risk stratification for future cardiac arrest after COVID-19 vaccination. World J Cardiol. 2025;17(2):103909. doi: 10.4330/wjc.v17.i2.103909.
- Chen YS, Wu YW, Chiang CH, Lin HH. Acute Pericarditis with High Anti-Nuclear Antibody Titers Following BNT162b2 mRNA COVID-19 Vaccination. Acta Cardiol Sin. 2022 Nov;38(6):784-787. doi: 10.6515/ACS.202211_38(6).20220522A. PMID: 36440237; PMCID: PMC9692212.
- Kusunoki H, Ekawa K, Ekawa M, Kato N, Yamasaki K, Motone M, Shimizu H. Trends in Antibody Titers after SARS-CoV-2 Vaccination: Insights from Self-Paid Tests at a General Internal Medicine Clinic. Medicines (Basel). 2023 Apr 20;10(4):27. doi: 10.3390/medicines10040027. PMID: 37103782; PMCID: PMC10142734.
- Kushima H, Ishii H, Kinoshita Y, Koide Y, Komiya Y, Kato J, Umehara M, Sakata M, Miyazaki M, Ikuta M. The Limited Effect of a History of COVID-19 on Antibody Titers and Adverse Reactions Following BNT162b2 Vaccination: A Single-Center Prospective Study. J Clin Med. 2022 Sep 14;11(18):5388. doi: 10.3390/jcm11185388.
- Kusunoki H, Ohkusa M, Iida R, Saito A, Kawahara M, Ekawa K, Kato N, Yamasaki K, Motone M, Shimizu H. Longitudinal Changes in IgG-Type SARS-CoV-2 Antibody Titers after COVID-19 Vaccination and a Prominent Increase in Antibody Titers When Infected after Vaccination. Vaccines (Basel). 2023 Apr 17;11(4):860. doi: 10.3390/vaccines11040860. PMID: 37112772; PMCID: PMC10145145.
- Mink S, Drexel H, Leiherer A, Cadamuro J, Hitzl W, Frick M, Reimann P, Saely CH, Fraunberger P. Interplay of statin treatment and anti-SARS-CoV-2 antibody levels in the outcome of COVID-19: a prospective, propensity score matched cohort study. Atherosclerosis. 2025 Sep;408:120449. doi: 10.1016/j.atherosclerosis.2025.120449. Epub 2025 Jul 19. PMID: 40712415.
- Ittiwut C, Mahasirimongkol S, Srisont S, Ittiwut R, Chockjamsai M, Durongkadech P, Sawaengdee W, Khunphon A, Larpadisorn K, Wattanapokayakit S, Arunotong S, Srimahachota S, Pittayawonganon C, Thammawijaya P, Sutdan D, Doungngern P, Khongphatthanayothin A, Kerr SJ, Shotelersuk V. Genetic basis of sudden death after COVID-19 vaccination in Thailand. Heart Rhythm. 2022 Nov;19(11):1874-1879. doi: 10.1016/j.hrthm.2022.07.019. Epub 2022 Aug 5. PMID: 35934244; PMCID: PMC9352648.
- Minato H, Yoshikawa A, Tsuyama S, Katayanagi K, Hachiya S, Ohta K, Myojo Y. Fatal arrythmia in a young man after COVID-19 vaccination: An autopsy report. Medicine (Baltimore). 2024 Feb 2;103(5):e37196. doi: 10.1097/MD.0000000000037196. PMID: 38306524; PMCID: PMC10843519.
- Hulscher, N., & McCullough, P. A. (2024). Delayed Fatal Pulmonary Hemorrhage Following COVID-19 Vaccination: Case Report, Batch Analysis, And Proposed Autopsy Checklist. Preprints. https://doi.org/10.20944/preprints202402.1096.v1
- Cavalli M, Eriksson N, Baron T, Yalcinkaya A, Landegren N, Brodin P, Hallberg P, Wadelius M. Genome-wide association study of myocarditis and pericarditis following COVID-19 vaccination. NPJ Vaccines. 2025;10(1):88. doi: 10.1038/s41541-025-01139-4.
- Kyriakopoulos, AM, Nigh, G, McCullough, PA, Seneff, S. Proteolytic Targets for SARS-CoV-2 Spike Protein Degradation: Hope for Systemic Detoxification. J Am Phys Surg, 2023;28(3):86–93.
- Lesgards JF, Cerdan D, Perronne C, Sabatier JM, Azalbert X, Rodgers EA, McCullough PA. Toxicity of SARS-CoV-2 Spike Protein with Respiratory Infection and Produced from COVID-19 mRNA and Adenoviral DNA Vaccines. Arch Microbiol Immunol. 2023;7: 121-138.
- Crespo-Barrios J. Vaxtherapy, a Multiphase Therapeutic Protocol Approach for Longvax, the COVID-19 Vaccine-Induced Disease: Spike Persistence as the Core Culprit and Its Downstream Effects. Diseases. 2025 Jun 30;13(7):204. doi: 10.3390/diseases13070204. PMID: 40709992; PMCID: PMC12293371.
- McCullough PA, Wynn C, Procter BC. Clinical Rationale for SARS-CoV-2 Base Spike Protein Detoxification in Post COVID-19 and Vaccine Injury Syndromes. J Am Phys Surg. 2023;28(3):90–94. https://doi.org/10.5281/zenodo.8286460
- McDonald MA, Kafil TS, Khoury M, Luk AC, Wright MK, Hawkins NM. Myocarditis and Pericarditis After mRNA COVID-19 Vaccination: 2024 Status and Management Update. Can J Cardiol. 2024;40(9):1536-1540. doi: 10.1016/j.cjca.2024.03.016.
- Hulscher N, Procter BC, Wynn C, McCullough PA. Clinical Approach to Post-acute Sequelae After COVID-19 Infection and Vaccination. Cureus. 2023;15(11):e49204. doi: 10.7759/cureus.49204.
- Hulscher N, Vickery A, McCullough PA. Resolution of Refractory COVID-19 Vaccine-Induced Myopericarditis with Adjunctive Rapamycin. Med Res Arch. 2024; 12(11). ISSN 2375-1924. https://esmed.org/MRA/mra/article/view/6099.
- Hulscher, N., Bowden MT, McCullough PA. Review of Calls for Market Removal of COVID-19 Vaccines Intensify: Risks Far Outweigh Theoretical Benefits. Science, Public Health Policy and the Law. 2025; 6:2019-2025
- Bourdon PS. Duriseti R, Gromoll HC, Dalton DK, Bardosh K, Krug AE. A Reanalysis of the FDA’s Benefit-Risk Assessment of Moderna’s mRNA-1273 COVID Vaccine: For 18–25-Year-Old Males, Risks Exceeded Benefits Relative to Hospitalizations. arXiv:2410.11811v1. 15 Oct 2024 https://arxiv.org/html/2410.11811v1
- Krug A, Stevenson J, Høeg TB. BNT162b2 Vaccine-Associated Myo/Pericarditis in Adolescents: A Stratified Risk-Benefit Analysis. Eur J Clin Invest. 2022;52(5):e13759. doi: 10.1111/eci.13759
- Rose J, Hulscher N, McCullough PA. Determinants of COVID-19 vaccine-induced myocarditis. Ther Adv Drug Saf. 2024;15:20420986241226566. doi: 10.1177/20420986241226566




