Cranial Nerve Vibration for Migraine and Trigeminal Pain
Modulation of Multiple Cranial and Cervical Nerve Sensory Fields for Migraine and Trigeminal Neuropathic Pain
Ronald M. Harper,1 Rebecca K. Harper,1 Danielle Snodgrass,2 Sergey Shaboyan,1 Greta Eggenberger,1 and Eberhardt K. Sauerland3*
- Department of Neurobiology,
David Geffen School of Medicine at UCLA; University of California at Los Angeles, Los Angeles, CA 90095, USA; - Audiology Clinic, David Geffen School of Medicine at UCLA; University of California at Los Angeles, Los Angeles, CA 90095, USA;
- Department of Anatomy, University of Nevada at Reno, Reno, Nevada, 89577, USA
OPEN ACCESS
PUBLISHED: 28 February 2026
CITATION: Harper RM, Harper, RK, et al 2026. Modulation of Multiple Cranial and Cervical Nervesensory Fields for Migraine and Trigeminal Neuropathic Pain. Medical Research Archives, [online] 14(2).
https://doi.org/10.18103/mra.v14i2.7186
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
DOI https://doi.org/10.18103/mra.v14i2.7186
ISSN 2375-1924
Abstract
The effectiveness of mechanical vibration to sensory fields of cranial nerves CN 5, 7, 8, 9, and 10 and cervical nerves C2 and C3 to mitigate migraine and trigeminal neuropathy pain was assessed. Vibratory devices were fitted within silicon impressions of the auditory meatus in 72 subjects (47 females, 25 males; 15 with trigeminal neuropathy pain, 57 with migraine pain). After initial pain level self-reports, oxygen saturation, heart and respiratory rates, and beat-by-beat blood pressure were collected during a 10-minute control period, followed by 10-minute low- and 20-minute high-amplitude 128 Hz mechanical vibration, and a second 10-minute control period and pain assessment. Migraine and trigeminal pain declined by a mean average of 3.54 (p<0.00001) self-report levels. Visual and vestibular aura were reduced or abolished. Heart rates declined due to vibratory stimulation, and partially recovered post-vibration, but remained lower than at trial onset. In a subset of 20 subjects, successive trials reduced the frequency and intensity of pain attacks over time. Non-invasive mechanical stimulation of cranial and cervical nerves can suppress pain and ameliorate deleterious aura, possibly through action of force sensing Piezo1 and Piezo2 ion channels.
Keywords
Piezo channels, Vibration, Pain, Heart Rate, Breathing, Neuromodulation, Xerostomia.
Introduction
A substantial need exists for non-pharmacologic interventions for pain arising from migraine, trigeminal, and cervical sources, and for management of autonomic, visual, and vestibular dysfunctions that often accompany such attacks. Although significant progress in pharmacologic-based interventions has emerged, including anesthetic blockade of occipital nerves or the sphenopalatine ganglia, and medications directed to neurotransmitter action, many of these pharmacologic interventions have significant drawbacks, including development of major unwanted cognitive or physical symptoms, intolerance to the agents, developmental or teratogenic effects, or ineffectiveness with even short-term use. A number of non-pharmacologic means have been developed; some are relatively invasive, and include direct electrical stimulation of the vagus nerve (CN10) through implanted electrodes; see for review. Other means are less invasive, and involve external electrical stimulation of cutaneous sensory fields served by the trigeminal, cervical/occipital, or remote sensory cervical and upper arm nerves. Non-invasive external stimulation of CN10 via electrical stimulation applied to the auricle has been effective for drug-resistant migraine or cluster headache; the supraorbital region served by the trigeminal nerve has also been useful, as well as transcranial magnetic stimulation. The efficacy of these approaches has been well-described.
Although invasive or external electrical stimulation to cranial nerves mediating migraine pain offers several advantages for intervention, including cost, ease of delivery, and precise control over stimulus parameters, its use leads to several concerns: electrical stimuli applied to the cutaneous surface can be unpleasant, and direct stimulation of nerves or to the skin pose issues of potential injury following long-term use. Invasive stimulation procedures carry significant cost and risk issues, including surgery-related infection as well as potential mechanical injury to the target nerves or to nearby tracts, and functional consequences from stimulation of autonomic efferent fibers. Electrical stimulation to vagal fibers of the neck, applied to the cutaneous surface, for example, provides a large field effect with the potential to reach fibers of the sympathetic tract proximal to CN 10, with potential influences on the cardiovascular system. Similarly, multiple efferent fibers of the vagus may be activated, carrying fibers to structures distal to the stimulation site with the possibility of unexpected consequences.
A promising means to intervene for migraine and trigeminal pain, as well as relief from deleterious physiological phenomena accompanying pain attacks, is offered by manipulating Piezo1 and Piezo2 proteins that sense force transduction and modulate action of a range of activities, including pain, temperature, and cardiovascular functions; for review, see. Mechanical stimuli, delivered by vibratory sources, offer the potential to deliver non-invasive and innocuous activation via force transduction of ion channels that could modulate the perception of migraine and trigeminal pain, as well as the significant visual, vestibular, breathing, and cardiovascular disturbances that can accompany the primary concern of pain.
Chronic oral or other trigeminal neuropathy pain is common, and may be diffuse, as with migraine. These presentations may have poorly defined triggers, and develop from dental procedures, or by neural injury from such interventions as radiation to cervical or oral regions. The pain origin may be difficult to localize or be referred to sites outside initial injury targets; thus, burning sensations can arise from tissue overlying the anterior mandible or tongue from molar interventions (burning mouth syndrome). Typically, pain arises from branches of the 5th (trigeminal), cranial nerve, since that nerve innervates the facial surface, a large portion of the skull, the dura, blood vessels of the brain, major portions of the oral cavity, the nasal cavities, and the orbit. Moreover, there is a need to address sensory fields of oral tissue served by CN 7, as well as posterior areas of the oral cavity which may involve CN 9 and CN 10, and in some cases of cervical pain, discomfort arising from the neck and shoulder, supplied by C2 and C3. The first three cervical nerves also provide innervation to the dura, a particular source of concern for migraine pain. Migraineurs can exhibit autonomic or motor disturbances reflected in sympathetic and parasympathetic pattern changes to challenges, disturbed respiratory-cardiac variability interactions, or impaired breathing; interventions for migraine or trigeminal pain must consider management of accompanying autonomic dysfunctions, which include nausea and vomiting, changes in blood pressure and heart rate, and reduced salivation which frequently accompanies pain from oral sources. Visual and vestibular dysfunctions also appear, and include narrowing or total occlusion of a visual field, perceptual distortions with intruding patterns, or fogginess of vision. Vestibular disturbances often include dizziness or ataxia; some of the signs are related to hypotension accompanying the primary condition, but can be independent of cardiovascular signs. Although a primary focus for migraine interventions is to reduce pain, the ancillary symptoms also require attention, and point to the widespread nature of afflicted brain regions and the multiplicity of disruptive afferent sources. Thus, the autonomic sequelae accompanying migraine attacks may benefit by modulating parasympathetic fibers accompanying or within cranial nerves 5, 7, 9, and 10, or by triggering central interactions of parasympathetic and sympathetic systems. Addressing disrupted autonomic functions during pain attacks may require concurrent masking or interfering activation of those autonomic fibers.
The principal cranial nerves classically implicated in mediating migraine pain are those of the 5th cranial nerve system; however, CN 7, 9, and 10 also provide sensory innervation to portions of the oral cavity, and the dura is supplied by spinal nerves C1, C2 and C3, as well as CN 5. Those cranial and cervical nerves could be used to mask or interfere with pain signals of CN 5; the cranial and cervical nerves use the descending nucleus of CN 5 for mediating cranial pain, providing a means to disrupt adjacent activity by proximity of integrative neurons of the descending nucleus for the major cranial nerves. Since migraine and trigeminal pain can develop from multiple, varying sources within the cranium, and neural processes involving autonomic control are widespread, interventions may require interfering with multiple cranial and cervical nerves that trigger or mediate central pain processing and autonomic regulation.
The use of mechanical force transduction to activate Piezo1 and Piezo2 channels for pain suppression would be optimized if the sensory fields serving the regulating cranial structures were relatively localized for ease of intervention delivery, and superficial for ready access. Activation of the multiple cranial nerves mediating pain perception is made difficult in that sensory fields for cranial nerves 7, 8, 9, and 10 are usually considered not superficial, require surgical intervention for access, and are sited in diverse locations. However, the auditory meatus and external pinna contain sensory fields for cranial nerves 5, 7, 8, 9, and 10, as well as cervical nerves C2 and C3, all of which serve significant roles in migraine or trigeminal pain or accompanying vestibular signs and can contribute to conditioning central nervous system processes. That proximal position of sensory fields allows regional surface stimulation to activate sensory fields of multiple cranial and cervical nerves that project to a common sensory nucleus mediating pain, the descending nucleus of the trigeminal system, which also receives pain fibers for cranial nerves 7, 9, and 10, and spinal nerves C2 and C3. Sensory activation by large fiber stimulation, such as that elicited by mechanical vibration, has long been proposed as playing a significant role in gating pain. The description of Piezo1 and Piezo2 channels extends the role of mechanical force deformation for both pain and multiple other autonomic actions on a range of fiber types. Somatosensory vibration at an adequate frequency and amplitude provides a non-invasive means to elicit activation of the sensory processes for both migraine and trigeminal pain as well as accompanying dysfunctions in vision, vestibular and autonomic dysfunctions. We evaluated the effectiveness of such vibratory intervention in migraine and trigeminal neuropathy subjects with acute presentation of pain. Since the intervention activated sensory nerves which influence both cardiovascular and breathing patterns, those activities were also assessed. Longer-term changes over months with repeated trials were examined in a subset of subjects, and anecdotal reports of even longer periods were documented.
Methods
SUBJECTS
The studies were approved by the Institutional Review Board of the University of California at Los Angeles (IRB# 13-001592), and written informed consent was obtained from each subject. Between the years 2013 through 2025, seventy-two subjects, aged 14-76 years, diagnosed with moderate-to-severe migraine (57 subjects) or trigeminal neuropathy pain (15 subjects) by a UCLA headache pain physician in the UCLA Department of Neurology, oral pain dentist in the UCLA School of Dentistry Oral Facial Pain Clinic, or Department of Neurology, Mayo Clinic, were recruited; recruitment notices were distributed in the UCLA Center for Health Sciences and in the UCLA School of Dentistry Pain Clinic. Final decisions regarding eligibility were made through review of medical records and subjects’ medical history by the Director of the UCLA Autonomic Control Laboratory. Subjects were in otherwise good health, with representation by sex approaching the ratio in the general population, i.e., 25 males and 47 females. Subjects endured migraine attacks over a 2-30 year period, and had undergone treatment by multiple physicians (often, 5-10) with little relief, and were on a range of medications for pain interventions. Subjects were instructed not to change medications during any part of the trial. Three subjects had failed neurosurgical interventions. Most subjects, having failed multiple medications, surgical intercessions, or other manipulations, considered the study a last resort for relief.
SUBJECT EXCLUSIONS
No subjects were pregnant (an exclusion criterion). One potential subject found initial exposure to the vibration disturbing and did not continue the study; another three recruitments had impressions made, but did not appear for the vibration trial for headache intervention. An additional recruitment, after four days of not demonstrating a migraine attack during trials shortly before evaluation (a requirement for study entry), revealed that she had begun (and continued) a cannabidiol oil intervention three days prior to the study. These recruitments were excluded from analysis and are not included in the subject count. No compensation other than incidental transportation, parking, or meal costs was offered to subjects.
DEVICE
Targeted sensory fields of CN 5, 7, 9, and 10 and cervical nerves C2 and C3 are shown in panel A of Figure 1. The cartoon of panel B of Figure 1 indicates that projections from those nerves reach the descending nucleus of CN5, the principal integrative area for pain of the head and neck. Fibers also pass rostrally to the periaqueductal gray and ventral thalamus and to insular and cingulate cortices (not shown). Cranial nerves 8 (auditory and vestibular fibers) and 9 (fibers to carotid baroreceptors) also have sensory fields near the auditory meatus, and serve roles for blood pressure regulation, an important aspect for modulation of migraine. Auditory meatus fitting was done by an accredited audiologist from the UCLA Head and Neck Surgery Audiology Clinic. Each subject was fitted bilaterally with custom earpieces created with silicone impressions of the auditory canal which extended proximally to the tympanic membrane. The earpieces were constructed using conventional impression procedures as commonly employed in the construction of hearing aids. These silicon devices held an embedded metal bar cemented to a surface circular neodymium disc magnet to which a vibrating motor was later magnetically coupled (Figure 1C, 1D). The devices, when activated, stimulated mechanosensory receptors sensing vibration for CN 5, 7, 8, 9, and 10 in the auditory meatus via the metal bar, and cervical nerves C2 and C3, in the auricle via local cartridge conduction. The custom impressions for the earpieces required special care to safeguard against injury to the tympanic membrane, while at the same time being sufficiently deep to provide mechanical vibration to the sensory field of CN 9 which extends from the external portion of the tympanic membrane and continues to the middle ear. A compressible foam dam, attached to fiber threads for later ease of removal, was inserted just lateral to the tympanic membrane after an initial inspection of the auditory canal for wax, growths, or lesions. A conduit in the silicon impression, formed by the threads from the dam, provided a tunnel for air release to guard against negative pressure on the tympanic membrane with impression removal. The magnets and metal bars were added to the impressions after hardening of the silicon and removal from the auditory canal. Those subjects with trigeminal neuropathy had additional flat vibratory pieces applied over the infraorbital and mental nerve exits on the face unilaterally. A power supply contained two low-voltage (1.5 v AA) batteries for the vibration motors.
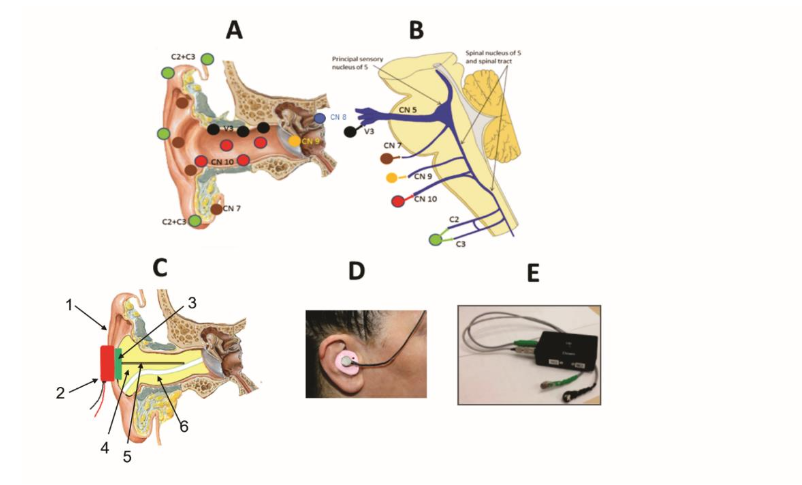
DEVICE SAFETY
The device was evaluated for sound levels at 128 Hz using a Tenma sound level meter Model 72-935. At 1 cm distance, at the “high” level, the meter showed an increase in sound pressure level 7 db above room background noise of 40 db with device onset; The “low” level was 4 db below that value. With direct physical contact with the vibratory device, i.e., direct physical contact of the device against the microphone of the sound level meter, the measure increased to 76 db, i.e., 36 db above 40 db background, while “low level” was 72 db. Those levels are substantially lower than values generally recognized for injury from sustained sound exposure, and are commonly exceeded by commercial auditory entertainment earpieces.
AUTONOMIC AND RESPIRATORY MONITORING
Several of the cranial and cervical nerves activated by the intervention have the potential to modify cardiovascular or respiratory activity, especially CN 5 (via accompanying autonomic fibers and somatic afferent fibers of upper airway muscles), CN 8 (via vestibular afferents affecting blood pressure), CN 9 (via projections to the carotid baroreceptors), and CN 10 (via accompanying and intrinsic autonomic fibers and fibers to the smooth muscles of the upper airway), as well as C3 (components of the phrenic nerve), thus mandating monitoring of both autonomic and breathing patterns for subject safety. Prior to the intervention trials, subjects were instrumented with a pressure band to assess thoracic and abdominal wall movements, three ECG electrodes on the anterior thoracic wall surrounding the heart, and a pulse oximeter on the left index finger. Signals were collected by a SOMNOtouch RESPTM device (SOMNOmedics, Coral Gables, FL). Beat-by-beat systolic, mean, and diastolic blood pressure values were later determined from pulse transit times using the ECG and pulse oximeter signals. Conventional arm cuff blood pressure measurements were collected on the subject prior to, and at the end of data collection, to calibrate the SOMNOmedics blood pressure values. All data were subsequently transferred from the device to a laptop computer for analysis (DominoLight; SOMNOmedics).
The subjects began a trial on experiencing a migraine headache or oral pain attack and were normally accompanied by a caretaker or partner for transport; trials were carried out in a quiet, dimmed UCLA laboratory, typically over a 1-hr period. In one case, logistics allowed only a demonstration trial in a subject visiting from the United Kingdom, and that subject subsequently used the device in an attack at home, and then, prophylactically.
PAIN SCALE
The Numerical Rating Score for Pain (NRS) was completed by the subject at onset of the first trial and at the end of the experimental session; the subjects rated pain on a 0-10 scale, with 0 being no pain, and 10, a pain level sufficiently high that the subject may go unconscious.
STUDY SEQUENCE
Following completion of an initial pain rating, application of recording electrodes, and beat-by-beat blood pressure calibration, the earpieces were inserted into both ear canals, and the subject queried on comfort of the device without vibration. Following affirmation of comfort, a 10-minute control period with no vibration, but concurrent physiological recording, was acquired. A 128-Hz sinusoidal mechanical vibratory signal was then applied for 10 minutes, initially at low amplitude (1.5 volts to vibrator), and for 20 minutes at increased amplitude (3.0 volts to vibrator). A second control period was collected with continued physiological monitoring after vibration offset for an additional 10 minutes. At the end of the last control period, the pain scale was again administered, blood pressure again calibrated with an arm cuff measure, and the physiological electrodes and the earpieces removed. The subject was questioned on general pain perceptions, change in salivation, any other affective aspects, sensory alterations, such as visual or vestibular alterations, and preferences on whether they would like to use the device again. After the initial laboratory trials, a subset of 42 subjects returned for 3-4 subsequent laboratory interventions. Twenty of these subjects requested devices for prophylactic home use. These subjects were queried on outcomes of such use, and values for this subgroup were tabulated and anecdotal comments noted.
Results
Seventy-two subjects with migraine or trigeminal neuropathic pain were studied. The subjects included those with classic migraine symptoms, with pain represented in multiple sites of the head, sometimes accompanied by cervical pain from trauma, and trigeminal neuropathy patients, with a range of oral pain symptoms, including burning mouth syndrome, chronic pain from failed dental procedures, and pain originating from radiation injury to cranial nerves following oncology treatment. Pain from the posterior pharynx, presumably arising from CN 9 and CN 10, also appeared. In five cases, the symptoms included primary headache pain, but stemmed from cervical injury from whiplash or trauma, and were accompanied by significant neck and shoulder pain. Most subjects had endured pain for at least 2 years, occasionally for 5-25 years, and had sought treatment from multiple medical institutions, undergone numerous procedures, and were attended by several physicians. Most considered participation in the study as a “last resort” for intervention following prior treatments that included a wide range of pain, anti-depressive, anti-seizure, or narcotic medications. The subjects indicated that their medications were ineffective, or could not be tolerated because of nausea, visceral injury by ulceration, other allergic reactions, or interference with cognitive, arousal or motor functions. Four subjects had undergone neurosurgical procedures, including sphenopalatine lesioning, occipital and cervical nerve blocks and ablation, and nerve decompression which resulted in no relief, or worsening effects from these surgeries.
RESPONSES TO AMPLITUDE CHANGES
Interventions began with low amplitude levels and were switched to a higher level after 10 minutes. In two cases, the subject preferred low amplitude settings, and requested that the amplitude be returned from the high level to low. The remaining subjects reported the high amplitude levels to be more effective.
Twenty-one of the 72 subjects did not meet the criterion of pain reduction of >1 self-rated levels; half of those 21 reported a decline of 1 level. Seventy-one percent of subjects found relief of at least 2 self-reported pain levels. Two subjects joined the study after experiencing a migraine attack, but had a level 0 pain on arrival, and had no change by trial termination, but a delay in appearance of subsequent attacks. In the most-severe cases, the pain reduction at the end of the first trial was substantial (e.g., on a pain scale of 0 to 10, where a rating of 10 is so severe that the subject is at risk to become unconscious, 10 to 0 (no pain) occurred on two subjects, 10 to 2 on two subjects, 10 to 6 on one subject, 9 to 0 on three subjects (one was mute from pain at level 9), 9 to 3 on one subject, and 8 to 1, and 8 to 2.5 on additional subjects (however, one subject with level 8 pain declined by only I level, which was considered no change).
PHYSIOLOGICAL PHENOMENA
The physiological monitoring and subject reporting demonstrated a number of ancillary physiological changes of the intervention in addition to reduction of migraine pain, including lowering of heart rate, reduction of dizziness from body motion, abolition of visual field loss and distortions of visual field, reduction of vestibular-induced visual alterations, more-regular and deep breathing, and, in approximately a third of subjects, onset of quiet sleep, typically within 12 minutes after onset of vibration; half of those subjects continued into rapid eye movement sleep after another 5-7 minutes, as indicated by twitches, eye movements, and reports of dreaming. These sleep data have been described elsewhere in abstract form.
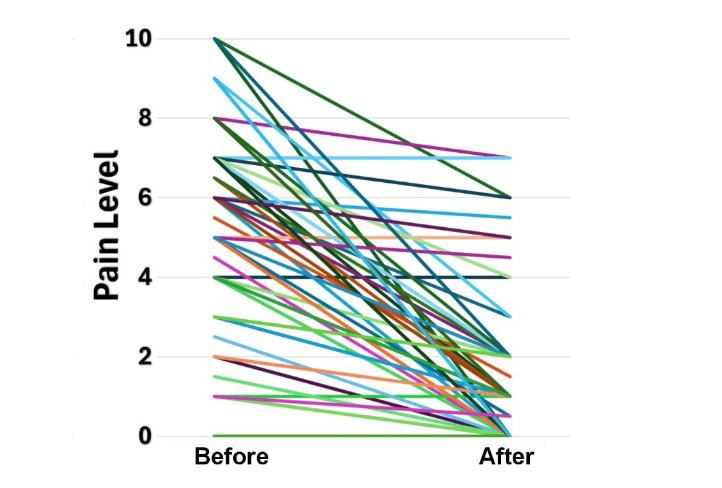
PAIN DECLINE
Individual declines in pain levels are shown in Figure 2. The mean pain level before intervention was 5.31, variance 6.35; after intervention, 1.77, variance 4.31, i.e., an average decline in pain level of 3.54. These values were significant, t Stat=10.85, two-tailed t test, p<.00001, df = 71. Several of the patients had other symptoms that often accompany migraine or trigeminal neuropathy; the most common symptom (7 subjects) was xerostomia (dry mouth syndrome), from reduced saliva flow; all reported improved salivation, with three trigeminal neuropathy subjects reporting improved salivation for a week or more following single trials. Total loss of visual fields and distortion of visual fields (speckles within fields, curving of linear shapes, partial loss of fields) during attacks were rectified in five subjects. Three subjects exhibited orthostatic hypotension, with one confined to a wheelchair; the intervention normalized blood pressure and resolved the mobility condition. One subject with migraine following traumatic brain injury was unable to breathe deeply and exhibited right-side lower limb motor weakness; both symptoms resolved with the intervention. Vestibular and vision disturbances during attacks in were resolved during trials, and symptoms continued to regress over several months to complete resolution.
PAIN LEVELS AFTER REPEATED LABORATORY TRIALS
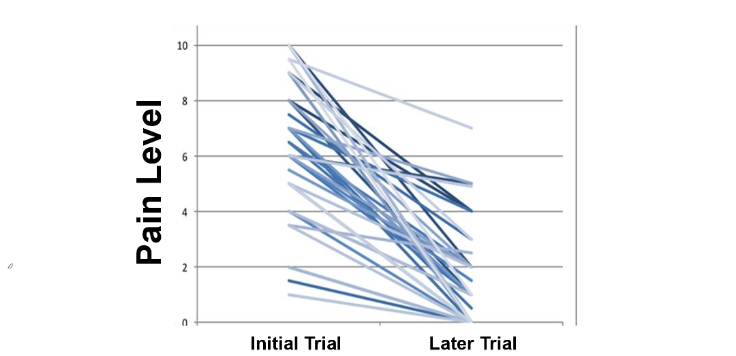
A subset of 42 subjects returned for 3-4 subsequent laboratory trials during migraine or trigeminal neuropathy attacks and showed consistent declines in perceived pain levels at onset of these later attacks relative to the initial trials. The changes in pain levels from the initial trial to the last trial are shown in Figure 3.
LONG-TERM EVALUATION
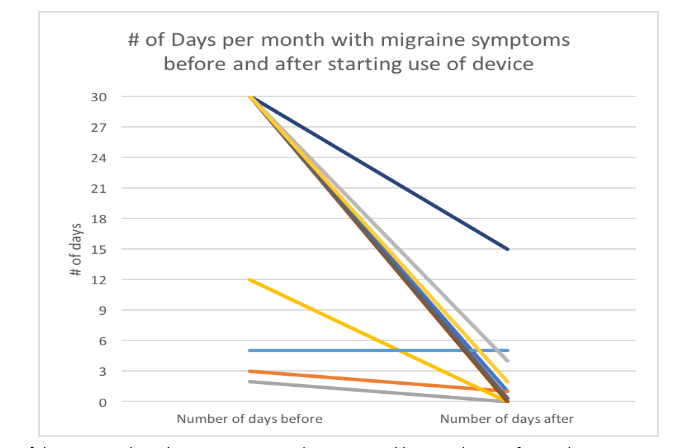
Because timing of the occurrence of migraine varied substantially between subjects, and because individuals within the subject population were often mobile, it was not possible to recall subjects at fixed intervals for follow-up interventions. However, several subjects returned repeatedly for interventions when experiencing attacks, with return sessions varying in number and time between sessions, and these sessions were documented. The 20 subjects who used the device at least once a week at home showed a decline in the number of attacks/month (Figure 4). Follow-up anecdotal reports from one subject indicated a 6-year suppression of migraine attacks with weekly twenty-minute interventions; a second subject reported complete suppression with no refresher trials over a 5-year period following the initial series of interventions.
CASE CHARACTERISTICS
The presentation of individual cases varied substantially. Some cases presented with pain levels so intense they were mute, others with modest or very low levels. The source or nature of pain also varied substantially, with accompanying features that differed between cases. Some migraine cases exhibited localized pain confined to regional areas of the head and was often lateralized. Other cases showed more-generalized pain distribution. A substantial number of cases reported migraine episodes beginning in childhood and continuing with ineffective management into adulthood. Trigeminal neuropathy cases most often showed chronic pain from unidentified sources, occasionally after oral surgery or radiation treatment for oncology. Burning mouth syndrome was common in trigeminal neuropathy cases, and xerostomia was often present in those subjects; recovery of saliva typically occurred within a single trial, and that recovery would last for a week or more.
HEART RATE
Heart rate declined during the course of the trial, and then partially recovered during the baseline period at the end of the recording (p <.001), but remained lower than values at trial onset. Technical issues related to heart rate acquisition limited the subject n to 33 migraine subjects.
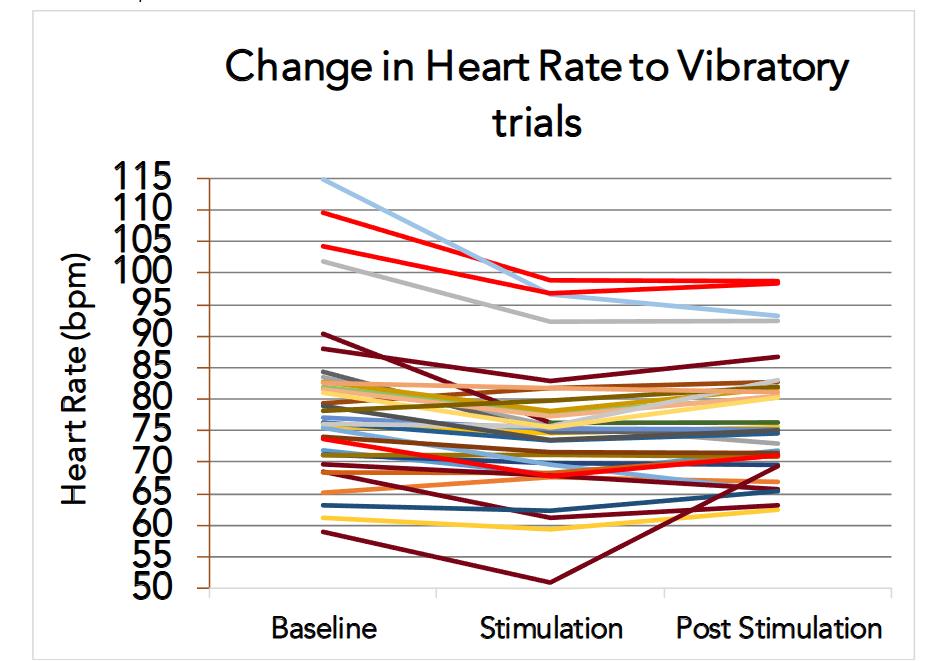
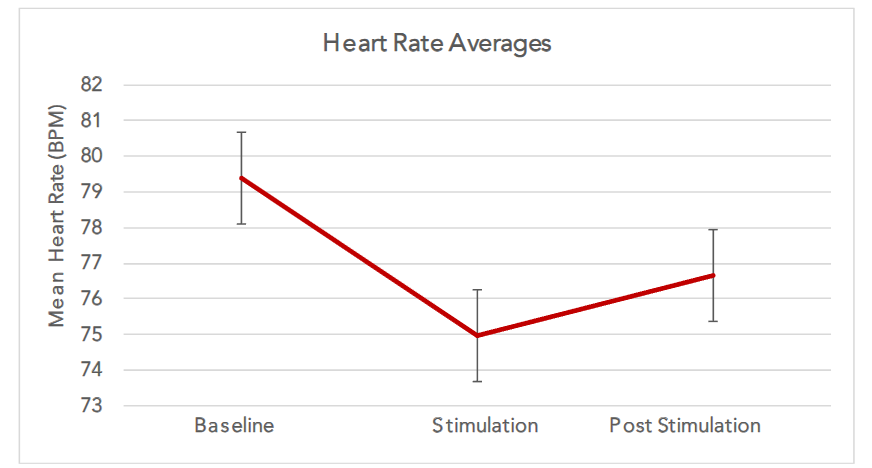
LONG-TERM TISSUE CONDITIONS
No subjects reported changes in ear canal conditions, such as pain or infection, even after prolonged use; one subject who used the devices daily for 6 months, and 2-3 times weekly for another 5 months, was evaluated by a professional audiologist, and no indications of altered auditory sensitivity, inflammation or other sign of tissue change were present.
Discussion
The primary focus of reducing acute migraine pain and pain of trigeminal neuropathy with short-term non-invasive vibratory intervention appears to be attainable; the intervention appears to be effective for a wide range of both trigeminal neuropathy and migraine pain. The outcomes can extend for long time periods with a minimal number of refresher trials. The intervention also appears to have several ancillary benefits for deleterious sensory and physiological symptoms or auras which often accompany migraine or trigeminal neuropathy attacks. This study was a proof-of-concept and will require additional investigation to establish the duration of pain suppression for different pathologies, optimization of stimulation parameters, and the persistence of effects on ancillary symptoms that accompany pain attacks.
The need for migraine and trigeminal neuropathy interventions is substantial. Headache disorders are highly prevalent globally, with an estimated global prevalence of 52%, and of migraine, 14%. In the U.S., there were 27.9 million self-reported migraine cases in 1999, with a nearly 3-fold higher incidence in females. Approximately 3.2 million Americans suffer from chronic migraine, a form of the condition in which headache occurs 15 or more days a month, with the condition lasting 4 hours or more for a period of 3 consecutive months. The condition is often debilitating, requiring withdrawal from social contact and/or precipitating stimuli for days at a time. The widespread prevalence of these conditions, and the diverse expression of symptoms mandate aggressive searches for multiple procedures for intervention, and especially for non-pharmacological approaches that avoid the consequences of medication use.
The implications of this non-invasive, low cost, non-electrical treatment mode for migraine and trigeminal neuropathy pain are significant. A simple mechanical vibrating device with a vibrating element similar to those found in other personal hygiene products, placed in the external ear canal and on a portion of the surrounding auricle, and driven by a small external battery-powered power supply and stimulator, constitutes the principal intervention. The potential for injury is low, since the active stimulus is mechanical vibration, not electrical, and unlikely to induce tissue changes associated with electrical activation, or to interfere with implanted devices, such as pacemakers. Spread of current is always a concern with electrical stimulation, particularly if the application is percutaneous and has the potential to reach multiple, disparate nerve bundles, e.g., proximal vagal and sympathetic nerves in the neck. Moreover, the mechanical vibration is of low amplitude, and not likely to cause hearing injury, since the vibrator sound level is low (much lower than typical earpiece music levels), reaching a maximal 76 db measured on direct placement on a hard surface of a recording microphone, a value significantly lower than levels recognized for inducing hearing injury, and the frequency of vibration is below frequency values necessary for optimal hearing. The limited exposure needed to achieve an effective response (<30 minutes) contributes to a reduced concern for injury. No pharmacological intervention is required, and the subject can readily remove the device in the event of discomfort. The device may be used safely to intervene with acute episodes of migraine, and the evidence, albeit from a subset of the entire cohort examined, suggests that repeated device use leads to subsequent reduction in the number and severity of migraine attacks. In multiple cases, attacks have been completely resolved; in a small number of subjects “refresher” trials, typically one 20 min trial/week, are required to avoid attacks. We speculate that the lessoning of attacks suggest that a “conditioning” takes place, with the central nervous system “relearning” appropriate responses to migraine or trigeminal pain triggers so that the frequency and severity of pain episodes are reduced.
ADVANTAGE OF ACTIVATION OF MULTIPLE CRANIAL AND CERVICAL NERVES
A number of studies have shown that noninvasive activation of cranial and cervical nerves, typically through electrical means, can reduce migraine pain. Those studies usually focus on single cranial systems, often directed singly at trigeminal, vagal, or cervical fibers. Logistic issues usually restrict activation of multiple systems, with stimulating electrodes typically placed on the facial surface, pinna of the ear, cervical or occipital regions, or the upper arm (cervical innervation), with the diverse sites making concurrent placement difficult. An advantage of the system described here is that multiple cranial nerves, 5, 7, 8, 9, and 10 and cervical nerves C2 and C3, with sensory fields all located within reach of the vibration source are activated in parallel, greatly simplifying logistics of electrode placement. Simultaneous activation of trigeminal, vagal, and cervical sensory systems likely contributed to the unexpected success in lowering pain and abolition of successive attacks in migraine subjects. The processes underlying these outcomes are unclear, but may reflect the possibility that the multiple-system approach can address dysfunctions in more than one system if an affected system is unknown. It may also be the case that combined activation of several sensory systems underlying pain may be more effective if summed activation to those afferent fibers is used; the fibers all have common projections to the descending nucleus of CN 5.
SELECTION OF VIBRATORY FREQUENCY
The process for reducing pain was a fixed-frequency vibratory signal, 128 Hz, a pulse rate commonly used to elicit reflexes in neurologic testing, delivered to a set of cranial and cervical nerves which have as a common integrative site, the descending or spinal nucleus of CN 5. Although the stimulus frequency was chosen for reasons of classical neurologic use in reflex testing, and because initial trials were so effective, optimal frequencies were not evaluated and could well differ from that used here. That aspect should be a particular issue when considering the mechanisms of action for pain reduction. Activation of Piezo1 and Piezo2 channels likely are the principal mediators of the outcomes in pain suppression, and those activities appear to be more sensitive to lower frequencies; the responsiveness of those channels has been mapped over 1-50 Hz. Subharmonics of the vibratory frequency used here may be a more effective stimulus; that possibility was not explored for logistic reasons, and because of the relatively high degree of success with the 128 Hz selection. However, the possibility exists that a lower vibratory frequency may contribute to an even greater proportion of successful intervention than found here.
Although the processes underlying disruption of migraine pain likely lie with activation of Piezo1 and Piezo 2 processes, the initial impetus for the studies emerged with the description of pain reduction on body surfaces served by spinal nerves using occlusion masking principles of stimulating large, faster-conducting nerves to overcome pain mediated by slower-conducting small fibers, a mechanism advanced by Melzack and Wall. This occlusion principle has been widely assumed to be operative for percutaneous electrical stimulation for pain in nerves from the spine. Implementation of pressure and vibratory receptors by the mechanical movements used here preferentially activate fast-conducting fibers and offer an advantage over electrical stimulation. Electrical stimulation, although exciting multiple sensory receptors including those for vibration, will preferentially activate receptors for noxious sensation, not the fast-conducting receptors especially useful for gating and suppressing pain in the Melzack and Wall model.
Although the Melzack and Wall masking principles may play a role here, with the synchronous vibration activating more rapidly conducting fibers of pressure and vibration to overshadow input from smaller pain fibers, a possible scenario is that the vibratory stimuli overwhelm integrative processing within the descending nucleus of CN 5. The possibility that the principal mechanism is one of disrupting integrative organization is strengthened by findings from individual subjects. In some subjects, pain, relieved by the device, extended from the mandibular region to areas of the medial lower limb down to the foot. Pain from such a distal region may not be mediated by the descending nucleus of CN 5, and suggests perception that is organized by higher central nervous system structures, including the periaqueductal gray and insular cortex. The insular cortex, in particular, serves major integrative roles for sensory information as well as pain, and may serve as an interactive site for the vibratory somatosensory information with pain perception.
One process that should be considered as an operative mechanism is based on a principle that a painful stimulus applied to a remote site can suppress an initial pain in another site. The principle has been summarized to a popular phrase “pain inhibits pain”, and is termed “diffuse noxious inhibitory control” (DNIC) in animals, or in humans, “conditioned pain modulation.” That process forms a forceful intrinsic analgesic mechanism capable of completely suppressing pain signals. The remote site of “pain” inhibiting migraine pain in this case may be the sensory vibratory stimulation to the descending nucleus of CN5, where cranial nerves 5, 7, 9, 10, and C2 and C3 carry pain signals. The vibratory signal is not “painful,” but may be misinterpreted as noxious. The subnucleus reticularis dorsalis appears to play a significant role in mediating pain suppression. It is unclear whether a similar process operates to suppress visual, vestibular, and visceral symptoms that can accompany migraine.
“LEARNING” OR ADAPTATION OF PAIN PERCEPTION.
A significant finding was that repeated interventions resulted in fewer episodes of pain, and reduction of severity of events over time even without changes in medication, or in most cases, reduction or cessation of medication. The beneficial aspects were not confined to one-time acute interventions, but exerted additive effects, which typically resulted in abolition of attacks after successive trials. The complete cessation of attacks was accomplished without further pharmacological intervention, and was of particular benefit for those subjects who presented with extreme levels of pain. Although attacks were typically suppressed long-term after 5-10 sessions, in some cases, 20 minute refresher trials were required when an aura of an attack appeared.
The decline in heart rate following vibratory stimulation likely follows from activation of parasympathetic components of the vagus, as well as autonomic fibers accompanying the trigeminal system. The responsivity of vascular components to vibration should be expected in view of the extreme sensitivity of Piezo1 channels to red blood cell volume displacement, as well as the role of both Piezo1 and Piezo2 channels in sensing blood pressure changes. In addition, parasympathetic fibers accompany CN 7 and CN 9, which are essential for salivation. Activation of the parasympathetic fibers resulted in exaggerated salivation, which was of significant benefit to the trigeminal neuropathy subjects who showed xerostomia. Restoration of salivation, like the vascular changes, appeared rapidly, and was sustained for periods of more than a week following a trial with the benefits for food intake and other positive attributes of improved saliva supply. Although the primary outcomes focused on sensory aspects of pain reduction, motor signs also showed alterations. The trials resulted in slowing and deepening of respiratory efforts, allowing one subject with brain trauma to take otherwise not possible deep breaths following initial injury with accompanying migraines. That restoration likely reflected the role that Piezo2 channels serve in mediating neuronal sensing of airway stretch and modulation of respiratory phase switching.
The heart rate decline was similar for both low and high-amplitude stimulation periods (data not shown), with rates rising slightly after stimulation offset. The slowing heart rate was accompanied by multiple subject statements at trial termination of the experience as “relaxing” “like meditation,” or “sleep-inducing.” The findings of sleep induction, particularly the short-latency onset of rapid eye movement sleep, were unexpected, but reflect the pronounced effect of the combined stimulation on the central nervous system.
The characteristics of pain reduction and number of events required to attain the absence of pain attacks were variable, just as the presentations of migraine were mixed, but the findings of successive interventions resulting in declines in pain intensity and number of attacks suggest that some form of reorganization of neural integration of pain takes place. It is unclear whether these processes were of a “learned” nature, or whether some other form of adaptation occurred. Differentiation of the precise mechanisms would require an evaluation of the patterns normally accepted to term a process as “learned,” i.e., show characteristics of extinction and restoration after reinforcement; such studies were outside the proof-of-concept evaluation here.
Limitations
This study was designed to evaluate whether mechanical vibration can reduce migraine and trigeminal neuropathy pain on acute presentations of attack. Thus, the effectiveness for any specific presentation of migraine or oral pain was not the objective, and the cases examined represented a wide range of distress. The disparities in presentations likely contributed to a lack of effectiveness in a number of cases. Despite the variation in types of pain, the overall effectiveness for both migraine and trigeminal neuropathy cases was unexpected. The study was not designed to follow long-term effectiveness of the intervention, and, except for limited subsets of subjects (numbering about 42 and 20, respectively), reports of longer-term benefits were principally anecdotal. However, a large number of follow-up testimonials from subjects who had endured years of pain attacks unmanaged by conventional pharmacologic, surgical, or behavioral interventions, but found relief to the vibratory trials, often permanently, suggest that vibratory stimulation of afferent input to multiple cranial and cervical nerves may be a useful approach. For those subjects arriving with pain levels of 8-10 and finding relief after the 30-minute trial, the intervention represented a major release. At the same time, many of the parameters of the study, including vibratory frequency, waveform, and intensity, pattern of vibratory input, duration of trials, and timing of intervention relative to onset of attacks, were arbitrary, and perhaps not optimal for particular classes of migraine or trigeminal pain. Those aspects perhaps could be targets for future studies.
Conclusion
We used two amplitude levels of a fixed-frequency mechanical vibration to a set of cranial and cervical nerves to achieve significant acute trigeminal and migraine pain reduction and accompanying physiological dysfunctions associated with the attacks in a large proportion of the subjects (approximately 70%), without further pharmacologic intervention. At the same time, the intervention resolved or lessened a range of secretory, visual, vestibular, and mobility issues accompanying the migraine attacks and secretory issues with trigeminal neuralgia. In a subset of patients who repeated trials, abolition of attacks was achieved. However, the device intervention only moderately affected pain in a small number of subjects and was ineffective or only modestly effective in 21 subjects (defined as a change of perceived pain level of 1 unit or less). It is unclear whether alterations in stimulation amplitude or frequency, changes in burst-pause patterning of the vibration, tapering of the amplitude of signal, or pairing of the vibration with different levels of the pain would alter the outcome of those poorly-responding cases. All of those aspects need to be further investigated. The approach suggests that mechanical vibration to the cutaneous sensory fields of a set of cranial and cervical nerves can ameliorate migraine and trigeminal pain and accompanying vestibular, visual, visceral and other physiological signs, and do so in a fashion that indicates an adaptation that minimizes future attacks. The nature of the stimulation, mechanical displacement, suggests that actions mediated through Piezo1 and Piezo2 channels are the active elements underlying the pain reduction and autonomic alterations. The responses to acute attacks appear to be repeatable; the long term suppression remains anecdotal, and needs to be evaluated.
Conflict of Interest Statement:
The University of California filed a patent application for the device naming Ronald M. Harper and E.K. Sauerland as inventors. PCT Patent Application No. PCT/US14/66191, filed November 18, 2014. Device, System and Method for Reducing Headache Pain. Published May 21, 2015, Issued: 14 August, 2018. No commercial entity has contributed to the study or has developed the device for use.
Funding Statement:
This research was supported in part by the Fidelity Charitable Nancy Adams and Scott Schoen Fund and the Kraig and Linda Kupiec Charitable Giving Fund. These sources did not influence the study design or execution.
Acknowledgements:
Dr. Frisca Yan Go and Dr. Robert Merrill from UCLA (both now deceased) made significant contributions to the design and to patient selection of this study, as did Dr. Joanna Jen (now at Columbia University). A number of UCLA undergraduate researchers provided invaluable analytic assistance to the findings, including C.R. White, V. Lubera, L.C. Feulner, and M. Rhoads.
Abbreviations:
ANOVA=Analysis of Variance, CN = Cranial Nerve, C1, C2, C3= Cervical nerves 1, 2, and 3
References:
- Atraszkiewicz D, Ünal E, Bassett P, Morell-Ducos F, Bahra A. Greater occipital nerve block for the treatment of migraine: An umbrella review, systematic review, and meta-analysis. Cephalalgia. 2025 Dec; 45(12):3331024251398390. doi: 10.1177/03331024251398390.
- Unal HA, Basarı A, Celiker OS, Cakar Turhan KS, Asik I, Ozgencil GE. Comparison of Greater Occipital Nerve Blockade and Sphenopalatine Ganglion Blockade in Patients with Episodic Migraine. J Clin Med. 2024 May 21;13(11):3027. doi: 10.3390/jcm13113027.
- Gaitatzis A, Sander JW. The long-term safety of antiepileptic drugs. CNS Drugs. 2013 Jun;27(6):435-55. doi: 10.1007/s40263-013-0063-0.
- Mauskop A. Vagus nerve stimulation relieves chronic refractory migraine and cluster headaches. Cephalalgia. 2005 Feb;25(2):82-6. doi: 10.1111/j.1468-2982.2005.00611.x.
- Yuan H, Silberstein SD. Vagus Nerve and Vagus Nerve Stimulation, a Comprehensive Review: Part I. Headache. 2016 Jan;56(1):71-8. doi: 10.1111/head.12647.
- Meng FG, Zhang JG, Schoenen J, Vandersmissen B, Herroelen L, Vandenheede M, Gerard P, Magis D. Migraine prevention with a supraorbital transcutaneous stimulator: a randomized controlled trial. Neurology. 2013 Sep 17;81(12):1102. doi: 0.1212/01.wnl.0000435063.25330.55.
- Schoenen J, Vandersmissen B, Jeangette S, Herroelen L, Vandenheede M, Gérard P, Magis D. Migraine prevention with a supraorbital transcutaneous stimulator. Neurology 2013;80(8):697-704. doi: 10.1212/WNL.0b013e3182825055.
- Reed KL, Black SB, Banta CJ 2nd, Will KR. Combined occipital and supraorbital neurostimulation for the treatment of chronic migraine headaches: initial experience. Cephalalgia. 2010 Mar;30(3):260-71. doi: 10.1111/j.1468-2982.2009.01996.x.
- Dodick DW, Silberstein SD, Reed KL, Deer TR, Slavin KV, Huh B, Sharan AD, Narouze S, Mogilner AY, Trentman TL, Ordia J, Vaisman J, Goldstein J, Mekhail N. Safety and efficacy of peripheral nerve stimulation of the occipital nerves for the management of chronic migraine: long-term results from a randomized, multicenter, double-blinded, controlled study. Cephalalgia. 2015 Apr;35(4):344-58. doi: 10.1177/0333102414543331.
- Yarnitsky D, Volokh L, Ironi A, Weller B, Shor M, Shifrin A, Granovsky Y. Nonpainful remote electrical stimulation alleviates episodic migraine pain. Neurology. 2017 Mar 28;88(13):1250-1255. doi: 10.1212/WNL.0000000000003760.
- Yarnitsky D, Dodick DW, Grosberg BM, Burstein R, Ironi A, Harris D, Lin T, Silberstein SD. Remote Electrical Neuromodulation (REN) Relieves Acute Migraine: A Randomized, Double-Blind, Placebo-Controlled, Multicenter Trial. Headache. 2019 Sep;59(8):1240-1252. doi: 10.1111/head.13551.
- Straube A, Ellrich J, Eren O, Blum B, Ruscheweyh R. Treatment of chronic migraine with transcutaneous stimulation of the auricular branch of the vagal nerve (auricular t-VNS): a randomized, monocentric clinical trial. J Headache Pain. 2015;16:543. doi: 10.1186/s10194-015-0543-3.
- Kim AY, Marduy A, de Melo PS, Gianlorenco AC, Kim CK, Choi H, Song JJ, Fregni F. Safety of transcutaneous auricular vagus nerve stimulation (taVNS): a systematic review and meta-analysis. Sci Rep. 2022 Dec 21;12(1):22055. doi: 10.1038/s41598-022-25864-1.
- Goadsby PJ, Grosberg BM, Mauskop A, Cady R, Simmons KA. Effect of noninvasive vagus nerve stimulation on acute migraine: an open-label pilot study. Cephalalgia. 2014 Oct;34(12):986-93. doi: 10.1177/0333102414524494.
- Silberstein SD, Yuan H, Najib U, Ailani J, Morais AL, Mathew PG, Liebler E, Tassorelli C, Diener HC. Non-invasive vagus nerve stimulation for primary headache: A clinical update. Cephalalgia. 2020 Oct;40(12):1370-1384. doi: 10.1177/0333102420941864.
- Di Fiore P, Bussone G, Galli A, Didier H, Peccarisi C, D’Amico D, Frediani F. Transcutaneous supraorbital neurostimulation for the prevention of chronic migraine: a prospective, open-label preliminary trial. Neurol Sci. 2017 May;38(Suppl 1):201-206. doi: 10.1007/s10072-017-2916-7.
- Magis D. Neuromodulation in migraine: state of the art and perspectives. Expert Rev Med Devices. 2015 May;12(3):329-39. doi: 10.1586/17434440.2015.1005606.
- Puledda F, Goadsby PJ. An Update on Non-Pharmacological Neuromodulation for the Acute and Preventive Treatment of Migraine. Headache. 2017 Apr;57(4):685-691. doi: 10.1111/head.13069.
- Starling AJ, Tepper SJ, Marmura MJ, Shamim EA, Robbins MS, Hindiyeh N, Charles AC, Goadsby PJ, Lipton RB, Silberstein SD, Gelfand AA, Chiacchierini RP, Dodick DW. A multicenter, prospective, single arm, open label, observational study of sTMS for migraine prevention (ESPOUSE Study). Cephalalgia. 2018 May;38(6):1038-1048. doi: 10.1177/0333102418762525.
- Lloyd J, Biloshytska M, Andreou AP, Lambru G. Noninvasive Neuromodulation in Headache: An Update. Neurol India. 2021 Mar-Apr;69(12 Suppl 1):S183-S193. doi: 10.4103/0028-3886.315998.
- Villar-Martinez MD, Goadsby PJ. Non-invasive neuromodulation of the cervical vagus nerve in rare primary headaches. Front Pain Res (Lausanne). 2023 Mar 13;4:1062892. doi: 10.3389/fpain.2023.1062892.
- Cocores AN, Smirnoff L, Greco G, Herrera R, Monteith TS. Update on Neuromodulation for Migraine and Other Primary Headache Disorders: Recent Advances and New Indications. Curr Pain Headache Rep. 2025 Feb 15;29(1):47. doi: 10.1007/s11916-024-01314-7.
- Coste B, Mathur J, Schmidt M, Earley TJ, Ranade S, Petrus MJ, Dubin AE, Patapoutian A. Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science. 2010 Oct 1;330(6000):55-60. doi: 10.1126/science.1193270.
- Beech DJ, Kalli AC. Force Sensing by Piezo Channels in Cardiovascular Health and Disease. Arterioscler Thromb Vasc Biol. 2019 Nov;39(11):2228-2239. doi: 10.1161/ATVBAHA.119.313348.
- Akerman S, Holland PR, Hoffmann J. Pearls and pitfalls in experimental in vivo models of migraine: dural trigeminovascular nociception. Cephalalgia. 2013 Jun;33(8):577-92. doi: 10.1177/0333102412472071.
- Lv X, Wu Z, Li Y. Innervation of the cerebral dura mater. Neuroradiol J. 2014 Jun;27(3):293-8. doi: 10.15274/NRJ-2014-10052.
- Mosek A, Novak V, Opfer-Gehrking TL, Swanson JW, Low PA. Autonomic dysfunction in migraineurs. Headache. 1999 Feb;39(2):108-17. doi: 10.1046/j.1526-4610.1999.3902108.x.
- Ebinger F, Kruse M, Just U, Rating D. Cardiorespiratory regulation in migraine. Results in children and adolescents and review of the literature. Cephalalgia. 2006 Mar;26(3):295-309. doi: 10.1111/j.1468-2982.2005.01039.x.
- Blau JN, Dexter SL. Hyperventilation during migraine attacks. Br Med J. 1980 May 24;280(6226):1254. doi: 10.1136/bmj.280.6226.1254.
- Akgün H, Taşdemir S, Ulaş ÜH, Alay S, Çetiz A, Yücel M, Öz O, Odabaşı Z, Demirkaya Ş. Reduced breath holding index in patients with chronic migraine. Acta Neurol Belg. 2015 Sep;115(3):323-7. doi: 10.1007/s13760-014-0375-y.
- Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders: 2nd edition. Cephalalgia. 2004;24 Suppl 1:9-160. doi: 10.1111/j.1468-2982.2003.00824.x.
- Peuker ET, Filler TJ. The nerve supply of the human auricle. Clin Anat. 2002 Jan;15(1):35-7. doi: 10.1002/ca.1089. PMID: 11835542.
- Melzack R, Wall PD. Pain mechanisms: a new theory. Science. 1965 Nov 19;150(3699):971-9. doi: 10.1126/science.150.3699.971.
- Wall PD. The gate control theory of pain mechanisms. A re-examination and re-statement. Brain. 1978 Mar;101(1):1-18. doi: 10.1093/brain/101.1.1.
- Gesche H, Grosskurth D, Küchler G, Patzak A. Continuous blood pressure measurement by using the pulse transit time: comparison to a cuff-based method. Eur J Appl Physiol. 2012 Jan;112(1):309-15. doi: 10.1007/s00421-011-1983-3.
- Hennig A. and Patzak A. Continuous blood pressure measurement using pulse transit time. Somnologie 2013, 17(2):104-110. doi: 10.1007/s11818-013-0617-x
- Hawker GA, Mian S, Kendzerska T, French M. Measures of adult pain: Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale (SF-36 BPS), and Measure of Intermittent and Constant Osteoarthritis Pain (ICOAP). Arthritis Care Res (Hoboken). 2011 Nov;63 Suppl 11:S240-52. doi: 10.1002/acr.20543.
- Harper, R.M., White, M.R., Harper, R.K., Snodgrass, D., Yan-Go, F. , Jen, J., Taghizadeh, A., Sauerland, E.K.. Sleep induction following cranial nerve modulation in migraine subjects. American Autonomic Society, 28th International Symposium, Clearwater Beach, Florida, 2017.
- Bigal ME, Serrano D, Reed M, Lipton RB. Chronic migraine in the population: burden, diagnosis, and satisfaction with treatment. Neurology. 2008 Aug 19;71(8):559-66. doi: 10.1212/01.wnl.0000323925.29520.e7.
- Hu XH, Markson LE, Lipton RB, Stewart WF, Berger ML. Burden of migraine in the United States: disability and economic costs. Arch Intern Med. 1999 Apr 26;159(8):813-8. doi: 10.1001/archinte.159.8.813.
- Lipton RB, Stewart WF, Diamond S, Diamond ML, Reed M. Prevalence and burden of migraine in the United States: data from the American Migraine Study II. Headache. 2001 Jul-Aug;41(7):646-57. doi: 10.1046/j.1526-4610.2001.041007646.x.
- Natoli JL, Manack A, Dean B, Butler Q, Turkel CC, Stovner L, Lipton RB. Global prevalence of chronic migraine: a systematic review. Cephalalgia. 2010 May;30(5):599-609. doi: 10.1111/j.1468-2982.2009.01941.x.
- Stovner LJ, Hagen K, Linde M, Steiner TJ. The global prevalence of headache: an update, with analysis of the influences of methodological factors on prevalence estimates. J Headache Pain. 2022 Apr 12;23(1):34. doi: 10.1186/s10194-022-01402-2.
- Steiner TJ, Stovner LJ. Global epidemiology of migraine and its implications for public health and health policy. Nat Rev Neurol. 2023 Feb;19(2):109-117. doi: 10.1038/s41582-022-00763-1.
- Lewis AH, Cui AF, McDonald MF, Grandl J. Transduction of Repetitive Mechanical Stimuli by Piezo1 and Piezo2 Ion Channels. Cell Rep. 2017 Jun 20;19(12):2572-2585. doi: 10.1016/j.celrep.2017.05.079.
- Craig AD. Significance of the insula for the evolution of human awareness of feelings from the body. Ann N Y Acad Sci. 2011 Apr;1225:72-82. doi: 10.1111/j.1749-6632.2011.05990.x.
- Youssef AM, Macefield VG, Henderson LA. Pain inhibits pain; human brainstem mechanisms. Neuroimage. 2016 Jan 1;124(Pt A):54-62. doi: 10.1016/j.neuroimage.2015.08.060.
- Le Bars D, Dickenson AH, Besson JM. Diffuse noxious inhibitory controls (DNIC). I. Effects on dorsal horn convergent neurones in the rat. Pain. 1979 Jun;6(3):283-304. doi: 10.1016/0304-3959(79)90049-6.
- Le Bars D, Dickenson AH, Besson JM. Diffuse noxious inhibitory controls (DNIC). II. Lack of effect on non-convergent neurones, supraspinal involvement and theoretical implications. Pain. 1979 Jun;6(3):305-327. doi: 10.1016/0304-3959(79)90050-2.
- Bouhassira D, Villanueva L, Bing Z, le Bars D. Involvement of the subnucleus reticularis dorsalis in diffuse noxious inhibitory controls in the rat. Brain Res. 1992 Nov 13;595(2):353-7. doi: 10.1016/0006-8993(92)91071-l.
- Cahalan SM, Lukacs V, Ranade SS, Chien S, Bandell M, Patapoutian A. Piezo1 links mechanical forces to red blood cell volume. Elife. 2015 May 22;4:e07370. doi: 10.7554/eLife.07370.
- Zeng WZ, Marshall KL, Min S, Daou I, Chapleau MW, Abboud FM, Liberles SD, Patapoutian A. PIEZOs mediate neuronal sensing of blood pressure and the baroreceptor reflex. Science. 2018 Oct 26;362(6413):464-467. doi: 10.1126/science.aau6324.
- Nonomura K, Woo SH, Chang RB, Gillich A, Qiu Z, Francisco AG, Ranade SS, Liberles SD, Patapoutian A. Piezo2 senses airway stretch and mediates lung inflation-induced apnoea. Nature. 2017 Jan 12;541(7636):176-181. doi: 10.1038/nature20793.

