Cutaneous Manifestations of Non-Neoplastic Bowel Diseases
Cutaneous Manifestations Associated with Non-Neoplastic Bowel Diseases
Alexia Vignau BS1, Clara Milikowski MD2
- University of Miami Miller School of Medicine
- University of Miami Miller School of Medicine, Department of Pathology and Laboratory Medicine
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Vignau, A., and Milikowski, C., 2024. Cutaneous Manifestations Associated with Non-Neoplastic Bowel Diseases. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5869
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5869
ISSN 2375-1924
ABSTRACT
There are several non-neoplastic bowel diseases, which include inflammatory bowel disease and celiac disease, both of which have been shown to have associated extraintestinal manifestations many of which are cutaneous. The implications of dermatologic manifestations are vast, as earlier identification can spur expedited testing and diagnosis of associated gastrointestinal disease as well as being a marker for gastrointestinal flare ups. It has been estimated that approximately 25.8% of cases of extra-intestinal manifestations precede the diagnosis of inflammatory bowel disease. This is especially useful as skin diseases may be the first easily recognizable feature of an underlying gastrointestinal condition. In the case of inflammatory bowel disease, lack of timely intervention either medically or surgically can lead to progression and worsening of symptoms, which if left untreated could lead to chronic and severe disease as well as malignancy. Similarly, in celiac disease, earlier diagnosis and treatment/ lifestyle modifications can not only improve the quality of life, but also decrease risks of long-term complications of other systemic extra-intestinal manifestations including neuropathies, infertility, osteoporosis, and malignancy. In this report, inflammatory bowel disease and celiac disease will be investigated through the review of literature, and identification of illustrative cases from pathology files, to elucidate the extraintestinal manifestations focusing on cutaneous manifestations.
Keywords: Cutaneous Extraintestinal Manifestations, Celiac Disease, Crohn’s Disease, Ulcerative Colitis, Dermatitis Herpetiformis
INTRODUCTION
The dysregulation of the gastrointestinal tract immune system in Crohn’s Disease (CD), Ulcerative Colitis (UC) and Celiac Disease (CeD) is correlated with several extra-intestinal manifestations (EIMs). Cutaneous EIMs associated with inflammatory bowel disease (IBD) include erythema nodosum, pyoderma gangrenosum, Sweet syndrome, and aphthous stomatitis and CeD is primarily associated with dermatitis herpetiformis. Manifestations on the skin bear the unique opportunity to trigger further work-up for gastrointestinal (GI) diseases if they appear before the non-neoplastic disease is discovered and may be identified without need for invasive procedures. Additionally, the presence of these cutaneous manifestations may also be an indicator of GI disease prognosis, and early recognition may aid in earlier and more effective disease management. Although not fully clarified, the links between these cutaneous diseases and gastrointestinal disease can shine light on disease pathophysiology and ultimately provide insight into treatment of comorbid illnesses. This association is further highlighted by the development of IBD or cutaneous EIMs secondary to the treatment with Biologics. Here, we explore what is known of the overlap between non-neoplastic bowel disease and their cutaneous manifestations and their etiologies.
INFLAMMATORY BOWEL DISEASE
Inflammatory bowel disease (IBD) includes Crohn’s disease (CD) and ulcerative colitis (UC) and is defined by chronic gastroenteric tissue inflammation. Crohn’s disease is a subtype of IBD characterized by chronic discontinuous lesions of inflammation that can involve the entire gastrointestinal tract. CD most commonly involves the terminal ileum, cecum and perianal areas. Meanwhile, ulcerative colitis, the other subtype of IBD in which the chronic relapsing and remitting inflammation is typically restricted to the mucosal surface of the colon in a continuous fashion most often starting in the rectum. The etiology of IBD is still largely uncertain, but studies indicate multifactorial causes including genetic predisposition, environmental exposure, immune dysfunction, and interaction with the patient’s intestinal microbiome (dysbiosis). Distribution is worldwide with IBD being more common in the west, with UC more prevalent worldwide than Crohn’s disease. In the context of associated Extra Intestinal Manifestations (EIMs), there is overlap between cutaneous manifestations associated with CD and UC, and their presence on the skin can serve to trigger further investigation of either possible underlying IBD diagnosis. Up to 50% of patients with CD experience EIMs prior to diagnosis, including cutaneous manifestations such as erythema nodosum, pyoderma gangrenosum, Sweet syndrome, and aphthous stomatitis. The significance of cutaneous presentations leading to early diagnoses of IBD and the potential to avoid ramifications with early treatment is important. Colorectal carcinoma is a long-term complication in IBD. For UC, the extent of involvement as well as duration of the disease are the two most important risk factors. Those with pancolitis who have had the disease for 8-10 years have the highest risk and those with left sided colitis only have the same risk as the general population, however only after 15-20 years. Data is inconclusive for patients with CD with some studies showing similar rates to UC.
CUTANEOUS EIMS OF IBD
Extraintestinal manifestations are common in patients with IBD, and can involve the skin, hepatobiliary tract, skeletal system and eyes. EIMs are defined as inflammatory processes outside of the gastrointestinal tract caused by the same drivers that produce inflammation in the gastrointestinal tract. The pathogenesis is not completely understood but may represent an immune response at an extraintestinal site due to shared epitopes. Prevalence rates for EIMS range from 6-47%. Among all of the EIMs, the most common are the mucocutaneous manifestations which are present in up to 22%-75% of patients with CD and 5-11% of UC. Up to 40% of all patients have one or several EIMS suggesting that one EIM increases susceptibility of having another. Cutaneous manifestations were seen more commonly in females and younger patients with both CD and UC. Patients taking antibiotics, steroids, immunomodulators and anti-TNF antibodies in both CD and UC and in patients with UC taking calcineurin inhibitors were found to have more cutaneous EIMs than those not taking them. Some of the most common skin lesions associated with IBD include erythema nodosum, pyoderma gangrenosum, Sweet syndrome and aphthous stomatitis, as well as immune and inflammatory conditions such as psoriasis, atopic dermatitis and hidradenitis suppurativa and treatment induced skin lesions. Most EIMs manifest after a diagnosis of IBD has been established, however up to 25.8% occur before a diagnosis is established and in some cases by greater than 2 years. The cutaneous manifestations have been divided into several subcategories. Specific EIMs have the same histologic findings as the underlying IBD, are seen only in CD and not really considered an EIM. Specific manifestations include continuous or mucocutaneous CD appearing in the perianal and oral regions and are considered an extension of the gastrointestinal inflammation and “metastatic CD” which is not contiguous to the gastrointestinal tract. Reactive EIMs are those which share pathophysiologic mechanisms with IBD but do not share histologic characteristics. These may be due to cross reactivity of antigens in the gastrointestinal tract and skin. Diseases seen in this group includes pyoderma gangrenosum and Sweet’s syndrome and other extremely rare diseases such as bowel associated dermatosis-arthritis syndrome; pyodermatitis-pyostomatitis vegetans; synovitis, acne, pustulosis, hyperostosis, osteitis syndrome (SAPHO); pyogenic arthritis; and pyoderma gangrenosum and acne syndrome. Associated EIMs are those that are frequently seen in IBD but the pathogenic mechanism is unknown. The underlying causes are the chronic inflammatory state and expression of HLA-DR2 or HLA-B27. These include aphthous stomatitis, erythema nodosum, psoriasis, and epidermolysis bullosa acquisita. Treatment-induced EIMs are temporally related to treatment, disappear after the therapy has been stopped and are not associated with the underlying IBD activity.
SPECIFIC SKIN MANIFESTATIONS
Continuous mucocutaneous manifestations occur in continuity with the intestine and are characterized by fissures, fistulae, abscesses and ulcers in the oral or perianal area with perianal disease being the first manifestation in 36% of CD patients. Oral lesions such as aphthous stomatitis are seen in 8-9% of CD patients and are represented by angular cheilitis, lip ulcers, lip and tongue fissures, gingival nodules, gingivitis and cobblestone oral mucosa. Oral lesions typically parallel disease activity.
Metastatic CD presents as ulcerating nodules, abscesses, fistulae and violaceous plaques anywhere on the body but predominantly on the anterior abdominal wall, submammary area, intertriginous zones, extremities, and rarely genitalia and face. Histology is similar to the underlying CD with noncaseating granulomas. The lesions respond to therapy for the underlying disease and do not parallel disease activity.
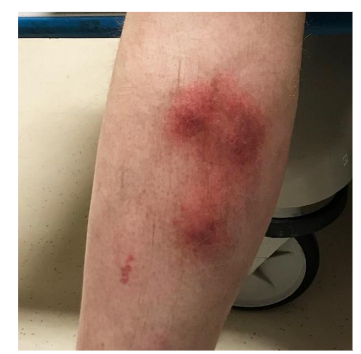
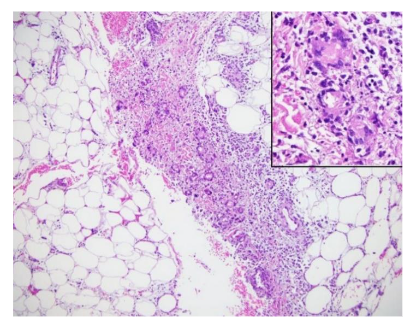
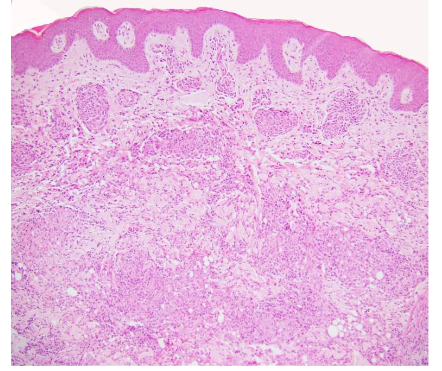
Erythema nodosum is the most common cutaneous EIM in IBD seen in 3%-15% of patients with up to 15% in CD and 10% in UC patients. It is more common in CD, adult females and equally in males and females in the pediatric population. Typically correlates with disease activity however, it precedes onset of IBD in 15% of cases. Erythema nodosum presents as painful, erythematous, subcutaneous nodules most commonly on the extensor surfaces of extremities but also the face, torso and upper extremity. Typically, it presents bilaterally and symmetrically with lesion size varying from one to five cm in diameter. Frequently, it is associated with systemic symptoms including fever, myalgia, arthralgia, headache, hepatosplenomegaly and pleuritis. It is the most common cause of septal panniculitis, or inflammation of the subcutaneous fat. Histopathology of erythema nodosum is a septal panniculitis, characterized by neutrophilic and lymphohistiocytic infiltrates involving septae of the subcutaneous adipose tissue. This inflammation is typically seen with absence of true vasculitis, though hemorrhage can be seen in small vessels. Older lesions may display lobule effacement and septal fibrosis with lymphohistiocytic and multinucleated giant cell infiltration with minimal neutrophils. Miescher’s radial granulomas may also be present and are made up of small aggregates of histiocytes around a central star-shaped cleft.
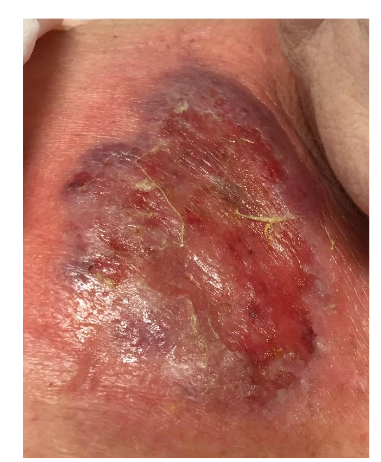
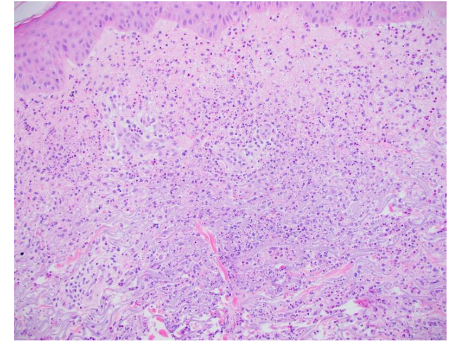
Pyoderma gangrenosum (PG) is the second most common cutaneous EIM associated with IBD, however, it is only present in 0.4-2% of patients, 1-2% of patients have UC. It is seen more commonly in CD, females, blacks of African descent and those with a family history of UC. In < 15% of patients, it occurs before the diagnosis of IBD, in some cases it parallels disease activity and in other cases, it follows an independent course. PG is a type of non-infectious neutrophilic dermatosis, similar to Sweet’s syndrome, that is associated with IBD in up to 30% of cases. Clinical presentation includes single or multiple painful erythematous lesions which can quickly develop into a blistered/necrotic ulcer with a violaceous and undermined border, most often manifesting on the legs or peristomal location. It may be accompanied by myalgia, arthralgia, fever and malaise. There are several variants of PG with the subtypes most associated with IBD being ulcerative, bullous, pustular and peristomal. It has been classified as a form of pathergy, which implies a significant skin injury after only a minor trauma. Microscopic findings for PG will differ depending on the variant but classically shows ulceration of the epidermis and dermis, along with a marked neutrophilic infiltrate undermining the epidermis, neutrophilic pustules and abscess formation. In general, PG is a very difficult diagnosis to make and is considered a diagnosis of exclusion after bacterial, fungal and mycobacterial causes of ulceration have been ruled out. Due to this, it is paramount when performing a biopsy for evaluation that the active border of the ulcer be included and that the sample penetrate into the subcutaneous tissues.
Sweet Syndrome, also known as acute febrile neutrophilic dermatosis, is an uncommon inflammatory condition associated with malignancy, infection, and inflammatory bowel disease. It is the 3rd most common disorder associated with IBD after malignancy and infection. It presents simultaneously with the diagnosis of IBD in 28% of cases, after the diagnosis in 52% and may precede the diagnosis in up to 20% of cases. It is more common in females and more prevalent in CD. Involving the face, arms and trunk, it appears as painful, erythematous papules, plaques or nodules and is associated with fever and neutrophilia. Histopathology of Sweet syndrome typically shows a predominance of mature neutrophils in the superficial dermis, though the infiltrate can also be present in the overlying epidermis and underlying adipose tissue.
Recurrent aphthous stomatitis (RAS), is a form of chronic oral mucosal inflammation and ulceration. It is present in 7-10% with IBD, most commonly in CD but recurrence is more frequent in UC. It may be an extension of CD into the oral mucosa or a consequence of nutritional deficiency. RAS may manifest prior to appearance of intestinal inflammatory symptoms. Typically, RAS develops after a prodrome of paresthesia at the site of the lesion, followed by the development of a macule and then a papule which eventually becomes necrotic and ulcerates leaving a round ulcer coated with a fibromembranous slough and ring of erythema. The disease manifests as recurrent single or multiple painful erosions and ulcers involving cheeks, tongue and lips and occasionally gingiva and palate. Prior to ulceration of the lesion, mononuclear cells aggregate beneath the basal cell layer. As the lesion progresses to more advanced stages, neutrophils dominate the ulcer center.
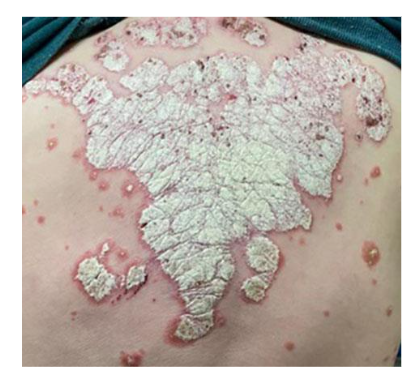
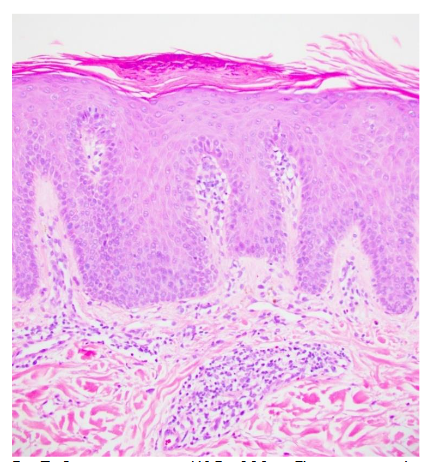
Psoriasis is the most common dermatologic condition in patients with IBD, more common in CD than UC, affecting 7-11% of patients. Psoriasis patients have a high risk of developing IBD. Genome wide studies have identified 11 genes of predisposition common in psoriasis and CD. First degree relatives of patients with CD will have psoriasis in 10% vs 3% of controls. The onset precedes IBD, and the course is independent of activity. Anti-TNF therapy may be a trigger for psoriasis. In fact, immune-related adverse events (IrAEs) may be seen in up to 22% of patients being treated with anti-TNF, a biologic used for the treatment of IBD, with eczematous or psoriasiform eruptions included. Clinically, patients with psoriasis present most commonly with the chronic plaque type which is characterized by well-circumscribed erythematous plaques covered with silvery scales. Histologically, fully developed lesions are characterized by regular acanthosis of the rete ridges with bulbous or “clubbing” tips sometimes with fusion, mitotic figures may be present 1-2 layers above the basal layer, and tortuous capillaries are present in edematous elongated dermal papillae with overlying thinning of suprapapillary epidermal plates. In addition, there is hyperkeratosis with confluent horizontal parakeratosis and hypogranulosis of the underlying superficial epidermis. 75% of cases have collections of neutrophils within the parakeratotic areas, known as Monro microabscesses and less commonly within the spinous layer, known as spongiform pustules of Kogoj. The dermis shows a superficial perivascular lymphocytic infiltrate with rare neutrophils.
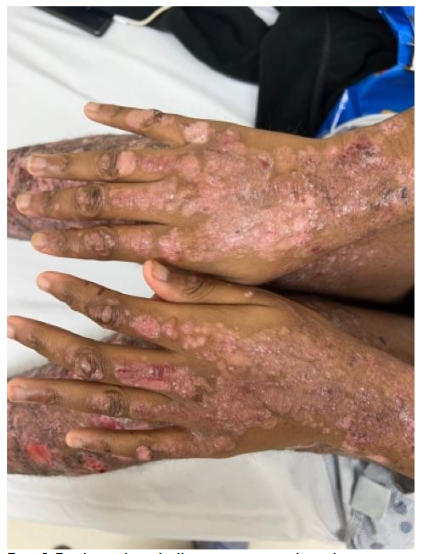
Epidermolysis bullosa acquisita (EBA) is a rare autoimmune, subepidermal blistering disease with antibodies to type VII collagen. 30% of patients with EBA have IBD. It is believed to be secondary to the chronic intestinal inflammatory process with production of antibodies to type VII collagen and epitope spreading secondarily involving the skin. Classic noninflammatory EBA presents with trauma induced erosions, vesicles and bullae affecting the hands, elbows, knees and feet and associated with subsequent hyperpigmentation or scarring. Bullous pemphigoid-like EBA presents with a vesicobullous eruption with erythema and pruritis involving the torso, intertriginous areas and extremities. Classically, histologically, there is a cell poor subepidermal blister. In the bullous pemphigoid-like variant bullae are not fragile and there is no subsequent scarring. By histology there is a subepidermal blister with a perivascular lymphocytic infiltrate with eosinophils and eosinophils may involve the blister cavity. On skin immunofluorescence there is a linear deposition with IgG and C3 which on salt split skin, is deposited on the dermal side. Similar findings are seen by immunohistochemistry for type VII collagen associated with CD. The lesions generally appear after the diagnosis of CD.
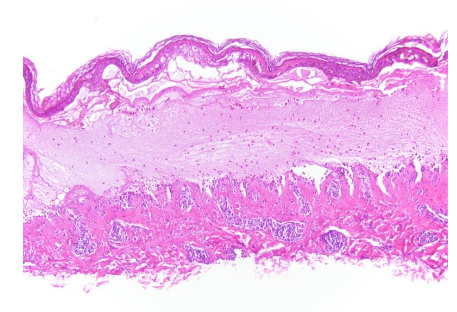
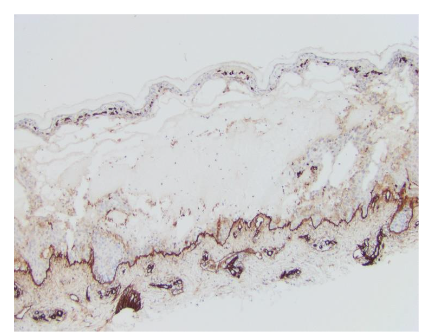
In terms of predisposition for cutaneous manifestations for IBD, genetic factors are under ongoing study. Associations of EIMS in 70% of parent-child pairs and 84% of sibling pairs as well as high rates of family history of IBD in patients presenting with EIMs are suggestive of genetic factors. One gene has already been identified: TRAF3IP2. Despite this, the pathophysiology of EIMs, like IBD, is not clearly understood. However, at least for the lesions that share similar pathophysiology and are not the result of therapy or IBD complications such as malabsorption, treatment with anti-TNF antibodies has had efficacy toward both treating the IBD and EIMs. Patients with IBD are also at increased risk of cutaneous malignancies compared to the general population. Mechanisms include chronic inflammation, immune dysfunction, cellular damage and many are secondary to use of immunosuppressive agents, most notably thiopurines and TNF-alpha inhibitors. A 4-7 fold increased risk of skin cancer has been reported in patients taking immunosuppressants, specifically immune modulators and biologics. Skin cancers included squamous cell carcinoma, basal cell carcinoma, and melanoma. Up to 34% reported having a first degree relative with skin cancer and others reported severe sunburns. Shared genetic risk variants have been discovered involving IBD and skin cancer.
CELIAC DISEASE
Celiac disease (CeD), also known as gluten sensitive enteropathy and celiac sprue, is an autoimmune condition which develops in genetically susceptible individuals and is triggered by an environmental agent- gluten. CeD affects all ages from infants to late adulthood. Classical presentation includes diarrhea, bloating, steatorrhea, and abdominal pain. Others have findings attributed to malabsorption which include failure to thrive, short stature, infertility, neurological symptoms, anemias, especially iron deficiency and other EIMs including dermatitis herpetiformis (DH) and aphthous stomatitis.
CUTANEOUS EIMS OF CELIAC DISEASE
Cutaneous EIMs such as dermatitis herpetiformis (DH), urticaria, atopic dermatitis, psoriasis, aphthous stomatitis, and rosacea are associated with celiac disease. In 2019, Abenavoli et al did a PubMed/Medline database search for associations between CeD and skin disorders. There were 7923 papers of which 100 were chosen based on Oxford Centre for Medicine 2011 criteria and consensus by the group. They found reports of twenty-four inflammatory skin conditions as possibly being associated with CeD. Some conditions shared HLA haplotypes, antibody profiles, cytokine production pathways, similar immunofluorescence patterns and many improved with a gluten free diet. Of these, they only considered psoriasis and most specifically DH as having a strong association.
The best-known cutaneous manifestation associated with CeD is dermatitis herpetiformis, also known as Duhring-Broque Disease. It is present in > 85% of those with CeD and improves with a gluten free diet. Both DH and CeD hold a well characterized association with HLA DQ2 and DQ8. These genes are present in 85% of patients with DH vs 25% of controls. First degree relatives with DH have an increased risk of DH and CeD. As with CeD, there is an association with autoimmune thyroiditis, type 1 diabetes mellitus and the risk of lymphoma. Patients with DH do not always have gastrointestinal symptoms, but in duodenal biopsy, 75% of patients diagnosed with DH had varying degrees of villous blunting, which is characteristic of CeD, emphasizing the marked correlation between the two phenotypes. After gluten ingestion, gliadin is absorbed and crosslinks with tissue transglutaminase in the intestine. Ultimately, antibodies form against tissue transglutaminase. Via epitope spreading, these antibodies become capable of binding to epidermal transglutaminase. These IgA antibodies deposit in the papillary dermis due to their greater affinity for epidermal transglutaminase than tissue transglutaminase. This causes inflammation at the site of IgA aggregation and recruitment of neutrophils, leading to microabscesses.
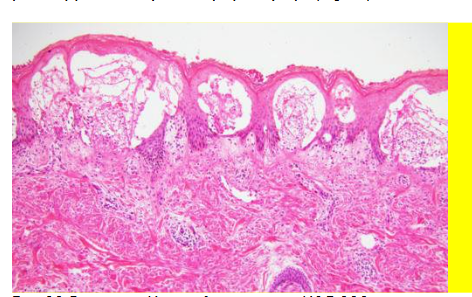
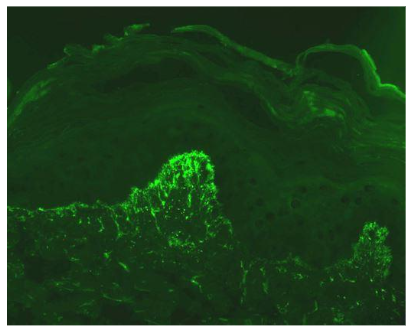
DISCUSSION
The study of the correlation between inflammatory bowel disease and the skin underscores the importance of the skin as an indicator of internal pathology. This relationship bears the potential to further pinpoint or confirm which inflammatory processes are involved in the etiology of both co-existing conditions. For instance, in CeD, the theorized epitope spreading of the IgA autoantibodies to the papillary dermis causing dermatitis herpetiformis. The benefit of the correlation of these diseases for research and ultimately treatment is further highlighted in the relationship between immunomodulating drugs and the subsequent development of similar autoimmune manifestations. For example, TNF-alpha inhibitors which are used to treat certain autoimmune skin conditions such as psoriasis as well as Crohn’s disease, have also been shown to trigger autoimmune-like manifestations of these very same diseases. These TNF-alpha-induced psoriasiform lesions manifest as scaly erythematous plaques with pustules as well as nail involvement. This reaction is referred to as a “paradoxical” reaction as TNF-alpha inhibition, particularly with infliximab, has also been used to treat psoriasis. This correlation between drug-induced versus IBD associated psoriasis in patients being treated with TNF-alpha inhibition for their Crohn’s or UC is elucidated by subsequent improvement of skin lesions after drug withdrawal, sometimes with complete resolution. In individuals who do have this reaction, it typically arises with the third or fourth administration of the biologic. Interestingly, these paradoxical effects of TNF-alpha inhibition also include de novo IBD as well, particularly CD in individuals being treated for rheumatoid arthritis with biologics. It is postulated that IBD may develop or worsen in periods of intense immunosuppression with TNF-alpha inhibition due to paradoxical pro-inflammatory effects despite anti-inflammatory effects. There are a few hypotheses as to why this is. One is that TNF-alpha inhibition triggers activation of auto-reactive T cells, which subsequently results in an autoimmune reaction and tissue destruction. It is also thought that there may be a genetic predisposition in individuals with NOD2/CARD15 mutations, which coupled with cytokine imbalance secondary to the TNF-alpha inhibition, might lead to background conditions for IBD. Other studies have observed that TNF-alpha blockade via neutralizing antibodies may inhibit regulatory T cells which modulate the reactivity of T cells to microbial antigens in the intestinal lumen, which is thought to influence IBD. Clearly, autoimmune disease of the gut presents some marked challenges for treatment, and the medications or strategies tackled to address them, be it avoidance of gluten or immunosuppression, require a fundamental understanding of the disease process. Especially when cutaneous disease or IBD is present, it is important to consider that it may also co-exist with manifestations in other systems and consider surveillance of both manifestations to ensure treatment response and success. Importantly, physicians must also consider that the treatment itself may be causing involvement of the other system, and take special account of symptom timeline and appearance, to appropriately pivot. For instance, if a psoriatic lesion appears after commencing an immunomodulating drug for IBD, the possibility that this might be secondary to the drug and not an extension of the initial autoimmune condition must be considered. This evaluation is necessary to trigger alteration of treatment or the decision to add another drug to try and address the new manifestation. This balance between suppressing the immune system and possibly incurring further autoimmunity as a byproduct, is a dance that necessitates further understanding of IBD and its cutaneous manifestations, in order to hopefully develop even more refined and targeted care.
CONCLUSION
Cutaneous manifestations of non-neoplastic inflammatory bowel disease serve as not only a harbinger of bowel pathology, relevant for clinic practice and investigation for diagnosis, they also could potentially provide insight into disease mechanism and our understanding of inflammatory autoimmune pathologies. Further investigation of EIMs in IBD and CeD as well as the underlying pathophysiological mechanisms as elucidated via histology, is needed to assist in discrimination of best treatment practices, improving diagnostics and informing of medication toxicity. Research and elucidation of correlations between EIMs and IBD is important, because not only are EIMs common in IBD patients, in up to one quarter of patients EIMs can present prior to establishment of IBD diagnosis. The existence of cutaneous EIMs can also be harbingers of disease prognosis- pyoderma gangrenosum exhibiting the most debilitating disease progression. Further clarification into EIM/IBD pathophysiology and associations would be beneficial for earlier disease treatment and identification.
CONFLICT
The authors have no conflicts of interest to declare.
ACKNOWLEDGEMENTS:
Clinical Photographs – Dr. Scott Elman, Department of Dermatology, University of Miami Miller School of Medicine
Clinical Photographs – Dr. Caresse Gamret, Jackson Health System
REFERENCES
- Greuter T, Vavricka SR. Extraintestinal manifestations in inflammatory bowel disease – epidemiology, genetics, and pathogenesis. Expert Rev Gastroenterol Hepatol. Apr 2019;13(4):307-317. doi:10.1080/17474124.2019.1574569
- Greuter T, Navarini A, Vavricka SR. Skin Manifestations of Inflammatory Bowel Disease. Clin Rev Allergy Immunol. Dec 2017;53(3):413-427. doi:10.1007/s12016-017-8617-4
- Guillo L, D’Amico F, Serrero M, et al. Assessment of extraintestinal manifestations in inflammatory bowel diseases: A systematic review and a proposed guide for clinical trials. United European Gastroenterol J. Nov 2020;8(9):1013-1030. doi:10.1177/2050640620950093
- Roth N, Biedermann L, Fournier N, et al. Occurrence of skin manifestations in patients of the Swiss Inflammatory Bowel Disease Cohort Study. PLoS One. 2019;14(1):e0210436. doi:10.1371/journal.pone.0210436
- Alvarez-Payares JC, Ramírez-Urrea S, Correa-Parra L, Salazar-Uribe D, Velásquez-López M. Mucocutaneous Manifestations of Inflammatory Bowel Disease. Cureus. Aug 2021;13(8):e17191. doi:10.7759/cureus.17191
- Rodrigo L, Beteta-Gorriti V, Alvarez N, et al. Cutaneous and Mucosal Manifestations Associated with Celiac Disease. Nutrients. Jun 21 2018;10(7)doi:10.3390/nu10070800
- Guan Q. A Comprehensive Review and Update on the Pathogenesis of Inflammatory Bowel Disease. J Immunol Res. 2019;2019:7247238. doi:10.1155/2019/7247238
- Torres J, Mehandru S, Colombel JF, Peyrin-Biroulet L. Crohn’s disease. Lancet. Apr 29 2017;389(10080):1741-1755. doi:10.1016/s0140-6736(16)31711-1
- Feuerstein JD, Cheifetz AS. Crohn Disease: Epidemiology, Diagnosis, and Management. Mayo Clin Proc. Jul 2017;92(7):1088-1103. doi:10.1016/j.mayocp.2017.04.010
- Du L, Ha C. Epidemiology and Pathogenesis of Ulcerative Colitis. Gastroenterol Clin North Am. Dec 2020;49(4):643-654. doi:10.1016/j.gtc.2020.07.005
- Ordás I, Eckmann L, Talamini M, Baumgart DC, Sandborn WJ. Ulcerative colitis. Lancet. Nov 3 2012;380(9853):1606-19. doi:10.1016/s0140-6736(12)60150-0
- Mak WY, Zhao M, Ng SC, Burisch J. The epidemiology of inflammatory bowel disease: East meets west. J Gastroenterol Hepatol. Mar 2020;35(3):380-389. doi:10.1111/jgh.14872
- Peppercorn MA, Kane SV. Clinical manifestations, diagnosis, and prognosis of crohn disease in adults. Accessed Jan, 2024. https://www.uptodate.com/contents/clinical-manifestations-diagnosis-and-prognosis-of-crohn-disease-in-adults
- Peppercorn MA, Kane SV. Clinical manifestations, diagnosis, and prognosis of ulcerative colitis in adults. UpToDate. https://www.uptodate.com/contents/clinical-manifestations-diagnosis-and-prognosis-of-ulcerative-colitis-in-adults?search=peppercorn%202022%20ulcerative%20colitis&source=search_result&selectedTitle=5%7E150&usage_type=default&display_rank=5
- Peppercorn MA, Cheifetz AS. Dermatologic and ocular manifestations of inflammatory bowel disease. Accessed Jan, 2023. https://www.uptodate.com/contents/dermatologic-and-ocular-manifestations-of-inflammatory-bowel-disease#!
- Leung AKC, Leong KF, Lam JM. Erythema nodosum. World J Pediatr. Dec 2018;14(6):548-554. doi:10.1007/s12519-018-0191-1
- Pérez-Garza DM, Chavez-Alvarez S, Ocampo-Candiani J, Gomez-Flores M. Erythema Nodosum: A Practical Approach and Diagnostic Algorithm. Am J Clin Dermatol. May 2021;22(3):367-378. doi:10.1007/s40257-021-00592-w
- George C, Deroide F, Rustin M. Pyoderma gangrenosum – a guide to diagnosis and management Clin Med (Lond). May 2019;19(3):224-228. doi:10.7861/clinmedicine.19-3-224
- Maverakis E, Marzano AV, Le ST, et al. Pyoderma gangrenosum. Nat Rev Dis Primers. Oct 8 2020;6(1):81. doi:10.1038/s41572-020-0213-x
- Cohen PR. Sweet’s syndrome–a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. Jul 26 2007;2:34. doi:10.1186/1750-1172-2-34
- Lima CDS, Pinto RDB, Góes HFO, Salles SAN, Vilar EAG, Lima CDS. Sweet’s syndrome associated with Crohn’s disease. An Bras Dermatol. Mar-Apr 2017;92(2):263-265. doi:10.1590/abd1806-4841.20175298
- Slebioda Z, Szponar E, Kowalska A. Etiopathogenesis of recurrent aphthous stomatitis and the role of immunologic aspects: literature review. Arch Immunol Ther Exp (Warsz). Jun 2014;62(3):205-15. doi:10.1007/s00005-013-0261-y
- Akintoye SO, Greenberg MS. Recurrent aphthous stomatitis. Dent Clin North Am. Apr 2014;58(2):281-97. doi:10.1016/j.cden.2013.12.002
- Cui RZ, Bruce AJ, Rogers RS, 3rd. Recurrent aphthous stomatitis. Clin Dermatol. Jul-Aug 2016;34(4):475-81. doi:10.1016/j.clindermatol.2016.02.020
- Cottone M, Sapienza C, Macaluso FS, Cannizzaro M. Psoriasis and Inflammatory Bowel Disease. Dig Dis. 2019;37(6):451-457. doi:10.1159/000500116
- Murphy M, Kerr P, Grant-Kels JM. The histopathologic spectrum of psoriasis. Clin Dermatol. Nov-Dec 2007;25(6):524-8. doi:10.1016/j.clindermatol.2007.08.005
- Kimmel JN, Taft TH, Keefer L. Inflammatory Bowel Disease and Skin Cancer: An Assessment of Patient Risk Factors, Knowledge, and Skin Practices. J Skin Cancer. 2016;2016:4632037. doi:10.1155/2016/4632037
- Cushing KC, Du X, Chen Y, et al. Inflammatory Bowel Disease Risk Variants Are Associated with an Increased Risk of Skin Cancer. Inflamm Bowel Dis. Nov 2 2022;28(11):1667-1676. doi:10.1093/ibd/izab336
- Dev K, Rahul F, Makheja K, et al. Frequency of Cutaneous Disorders in Patients With Celiac Disease. Cureus. Sep 2021;13(9):e18148. doi:10.7759/cureus.18148
- Rodrigo L, Beteta-Gorriti V, Alvarez N, et al. Cutaneous and Mucosal Manifestations Associated with Celiac Disease. Nutrients. Jun 21 2018;10(7)doi:10.3390/nu10070800
- Abenavoli L, Dastoli S, Bennardo L, et al. The Skin in Celiac Disease Patients: The Other Side of the Coin. Medicina (Kaunas). Sep 9 2019;55(9)doi:10.3390/medicina55090578
- Thrash B, Patel M, Shah KR, Boland CR, Menter A. Cutaneous manifestations of gastrointestinal disease: part II. J Am Acad Dermatol. Feb 2013;68(2):211 e1-33; quiz 244-6. doi:10.1016/j.jaad.2012.10.036
- Hull C, Zone J, Ofori A. Dermatitis Herpetiformis. UpToDate. 2024. https://www.uptodate.com/contents/dermatitis-herpetiformis?search=hull%20dermatitis%20herpetiformis&source=search_result&selectedTitle=1~42&usage_type=default&display_rank=1
- Rose C, Brocker EB, Zillikens D. Clinical, histological and immunpathological findings in 32 patients with dermatitis herpetiformis Duhring. J Dtsch Dermatol Ges. Apr 2010;8(4):265-70, 265-71. doi:10.1111/j.1610-0387.2009.07292.x
- Garcia C, Araya M. [Dermatitis herpetiformis and celiac disease]. Rev Med Chil. Sep 2021;149(9):1330-1338. Dermatitis herpetiforme y enfermedad celiaca. doi:10.4067/S0034-98872021000901330
- Thrash B, Patel M, Shah KR, Boland CR, Menter A. Cutaneous manifestations of gastrointestinal disease: part II. J Am Acad Dermatol. Feb 2013;68(2):211.e1-33; quiz 244-6. doi:10.1016/j.jaad.2012.10.036
- Mihai IR, Burlui AM, Rezus, II, et al. Inflammatory Bowel Disease as a Paradoxical Reaction to Anti-TNF-α Treatment-A Review. Life (Basel). Aug 20 2023;13(8)doi:10.3390/life13081779
- Passarini B, Infusino SD, Barbieri E, et al. Cutaneous manifestations in inflammatory bowel diseases: eight cases of psoriasis induced by anti-tumor-necrosis-factor antibody therapy. Dermatology. 2007;215(4):295-300. doi:10.1159/000107622
- Peer FC, Miller A, Pavli P, Subramaniam K. Paradoxical psoriasiform reactions of anti-tumour necrosis factor therapy in inflammatory bowel disease patients. Intern Med J. Dec 2017;47(12):1445-1448. doi:10.1111/imj.13637

