Cytokine Levels in Dental Implant Failure: A Study
Macrophage Sensitization to TiO2: Insights from Failed and Survived Implant Cases
Patrick R. Schmidlin¹ and Philipp Sahrmann²
- Clinic of Conservative and Preventive Dentistry, Division of Periodontology and Periimplant Diseases, Center of Dental Medicine, University Zürich, Plattenstrasse 11, 8032 Zürich
- Department of Periodontology, Endodontology and Cariology, University Centre for Dental Medicine, University of Basel, Basel, Switzerland
OPEN ACCESS
PUBLISHED: 30 September 2025
CITATION: Schmidlin, P. and Sahrmann, P., 2025. Macrophage Sensitization to TiO2: Insights from Failed and Survived Implant Cases. Medical Research Archives, [online] 13(9). https://doi.org/10.18103/mra.v1
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v1
ISSN 2375-1924
ABSTRACT
Background/Objectives: To evaluate whether patients with prior implant loss without clinical symptoms of peri-implantitis exhibit elevated cytokine levels (TNF-α and IL-1β) compared to age- and gender-matched controls with healthy peri-implant conditions after 10 years, hypothesizing an association between implant loss and increased cytokine expression.
Methods: Patients were selected from a long-term randomized controlled trial, including individuals with implant loss and controls with healthy peri-implant tissues. Blood samples were exposed to titanium dioxide (TiO2) particles and analyzed for cytokine release using a macrophage stimulation test.
Results: No significant association was observed between implant loss and TNF-α levels. IL-1β levels were elevated in the implant loss group when tested with correction for non-normally distributed data. Within the limitations of this study, the results align with recent studies questioning systemic cytokine-peri-implant health relationships while contrasting with findings of heightened cytokine levels in typical peri-implantitis cases.
Conclusions: This preliminary study highlights variability in host immune responses and suggests that systemic biomarkers alone may not explain implant loss, particularly in non-inflammatory scenarios such as potential mechanical overload. Larger studies with diverse populations and integrated systemic and local inflammatory analyses are needed to better understand the underlying mechanisms of implant complications and refine diagnostic approaches.
Keywords: Dental implant, debridement, peri-implantitis, implant loss, occlusal force, implant failure
1. Introduction
Dental implants are a highly effective option for replacing missing teeth, with long-term success rates often exceeding 90% under strict clinical criteria(1-3). However, implant failure remains a clinical reality, with peri-implantitis being one of the most frequent causes(2,4). Peri-implantitis, characterized by biofilm-induced inflammation and marginal bone resorption, is widely recognized as a primary driver of implant failure(5). Additionally, other factors such as mechanical overload and material properties, including the release of titanium dioxide (TiO2) particles, are increasingly implicated in peri-implant diseases(6).
A recent randomized controlled trial investigating implants of different lengths demonstrated a tendency towards higher rates of implant failure among short implants, with failures occurring in the absence of typical peri-implant inflammation(6). Furthermore, studies have proposed that mechanical overload, often associated with short implants, may trigger bone remodeling processes that impair implant stability over time(6-8). This raises questions about the interplay between biomechanical stress, local inflammatory responses. As an additional potential factor in understanding implant failure, systemic and local immune sensitization to TiO₂ particles has been proposed as a contributor to the disintegration at the interface between titanium implants and bone (9). Given that the literature reflects this issue in a controversial manner, there remains a need to investigate possible interrelations.
Accordingly, this study builds on these insights by analyzing cytokine responses (TNF-α and IL-1β) in patients with implant loss due to assumed overload or non-inflammatory causes, compared to matched controls with stable implants. By integrating systemic cytokine analyses with clinical observations, we aim to elucidate the potential mechanisms underlying implant failure and their diagnostic implications. It was the hypothesis of the present study, that patients with implant loss would display higher cytokine values upon macrophage exposure to TiO2.
2. Materials and Methods
In the present study, patients who lost an implant as part of a long-term randomized controlled clinical trial and corresponding control subjects from the same study with healthy peri-implant tissues were recruited(7). The control subjects were matched for sex and age. All implants were lost without clinical signs of inflammation but got lost after a linear disintegration at the bone-implant interface(8).
Full blood samples were taken from the cephalic or cubital vein of these patients and sent to a specialized laboratory (IMD Laboratory, Berlin, Germany) and stored at 7° until the courier collected the samples. Delivery took place within 24 hours overnight by a special courier in a refrigerated supply chain. In the laboratory, blood samples were exposed to TiO2 particles in a standardized macrophage stimulation test. The levels of expressed TNF-α and IL-1ß released from macrophages of the individual patient were determined with enzyme-linked immunosorbent assays (ELISA). Levels of the pro-inflammatory cytokine TNF-alpha and interleukin-1ß were measured.
The datasets were tested for normal distribution using the Kolmogorov-Smirnov and the Shapiro-Wilk test. In the case of an asymmetric distribution, inter-group differences were tested for significance using the Mann-Whitney U test. The level of significance was set at 0.05.
3. Results
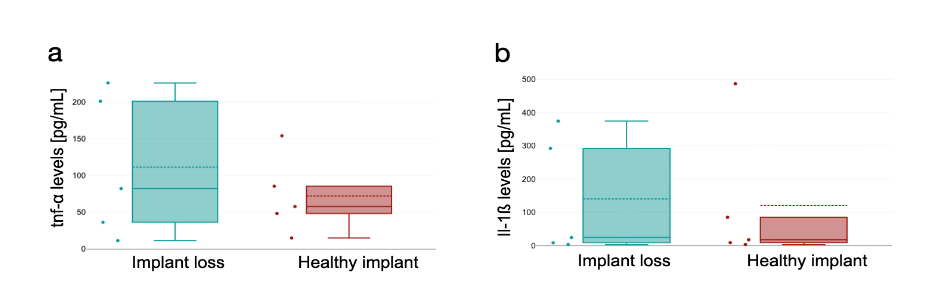
For both, the levels of TNF-α and IL-1ß, the Mann-Whitney U test found no intergroup difference (p=0.693 and 0,876, respectively). Levels for TNF-α and IL-1ß are given in Figure 1.
4. Discussion
This preliminary study evaluated whether patients with implant loss exhibited higher cytokine levels compared to those with stable implants after 10 years, reflecting peri-implant stability. No association was found between implant loss and TNF-α levels. While a tendency towards higher IL-1ß expression was observed in patients with implant loss using certain statistical tests, this difference was not significant when tested without correction. These results actually align with recent findings that challenge the link between systemic cytokine profiles and peri-implant health(10), but contrast with other studies demonstrating heightened inflammatory cytokine levels in peri-implantitis cases(11). This may reflect distinct pathophysiological and individual response profile, which may range from classical infection infestations to or more aseptic conditions, for example due to mechanical overload.
Of course, a notable limitation of evaluation is the very small sample size, which considerably limits the statistical power, rendering the postulates for normal distribution generally very unlikely(12). Thus, a stricter test for non-parametric data distribution was mandatory, and the hypothesis had to be rejected.
In addition, additional local clinical and immunological peri-implant tissue analyses are lacking, which could have provided more detailed insights into a potential inflammatory response at the implant site and respective aetiopathological conditions.
A key strength is the origin from a well-controlled and meticulously maintained long-term cohort. However, this strength may also introduce a potential bias, as the study population represents a highly compliant group with regular follow-ups, which might not fully reflect the broader population with implant complications. Additionally, a considerable part of the study participants had a history of periodontitis, even if periodontal stability was one of the inclusion criteria.
Peri-implantitis is very similar to periodontitis a multifactorial disease. With an immunologic response triggered by the presence of biofilms on non-shedding intra-oral surfaces, many risk factors are already known (uncontrolled diabetes, cigarette smoking and insufficient oral hygiene)(13,14) and others still under discussion, that might contribute to the chronic deterioration of the bone-to-implant interface.
The present results do not indicate, however, that inter-individually different differences in the expression of the cytokines tnf-a and il-1ß are associated with implant failure over the considerable time period of 10y.
Despite these important limitations, our findings underscore the need for a nuanced approach when interpreting systemic biomarkers in the context of failing and ailing implants. Future studies warrant a more specific and elaborated focus on refining diagnostic assays, incorporating larger cohorts, and exploring the interplay between systemic cytokine responses and local inflammatory pathways.
5. Conclusions
Our data offer preliminary insights in the complexity of evaluating cytokine markers in implant loss. Larger-scale studies with diverse populations and standardized assays are essential to better understand the potential link between cytokine levels and implant loss.
Author Contributions:
PSH and PSA Conceptualization; methodology; analysis; investigation; writing, original draft preparation, review and editing. Both authors have read and agreed to the published version of the manuscript.
Funding:
No external funding was available for this study.
Institutional Review Board Statement:
Patients of this study were selected from the Sahrmann P et al. Ten-Year Performance of Posterior 6-mm Implants with Single-Tooth Restorations: A Randomized Controlled Trial. J Dent Res. 2023 Aug; 102(9):1015-1021 ethics committee StV Nr. 07/13).
Informed Consent Statement:
Written informed consent was obtained from all subjects involved in the study.
Data Availability Statement:
Data supporting reported in this study can be found in the text and respective tables.
Acknowledgments:
The authors thank the co-authors of the original study for their invaluable support, which enabled this additional evaluation based on previous long-term observations of the study mentioned in this study.
Conflicts of Interest:
The authors declare no conflicts of interest.
Abbreviations:
The following abbreviations are used in this manuscript:
TNF Tumor Necrosis Factor
IL Interleukin
TiO2 Titanium Oxide
References:
- Benic GI, Bernasconi M, Jung RE, Hammerle CH. Clinical and radiographic intra-subject comparison of implants placed with or without guided bone regeneration: 15-year results. J Clin Periodontol. 2017;44(3):315-25.
- Moraschini V, Barboza E. Success of dental implants in smokers and non-smokers: a systematic review and meta-analysis. Int J Oral Maxillofac Surg. 2016;45(2):205-15.
- Moraschini V, Poubel LA, Ferreira VF, Barboza Edos S. Evaluation of survival and success rates of dental implants reported in longitudinal studies with a follow-up period of at least 10 years: a systematic review. Int J Oral Maxillofac Surg. 2015;44(3):377-88.
- Heitz-Mayfield LJ, Mombelli A. The therapy of peri-implantitis: a systematic review. Int J Oral Maxillofac Implants. 2014;29 Suppl:325-45.
- Lee CT, Huang YW, Zhu L, Weltman R. Prevalences of peri-implantitis and peri-implant mucositis: systematic review and meta-analysis. J Dent. 2017;62:1-12.
- Messous R, Henriques B, Bousbaa H, Silva FS, Teughels W, Souza JCM. Cytotoxic effects of submicron- and nano-scale titanium debris released from dental implants: an integrative review. Clin Oral Investig. 2021;25(4):1627-40.
- Sluka B, Naenni N, Jung RE, Attin T, Schmidlin PR, Sahrmann P. Changes of radiopacity around implants of different lengths: Five-year follow-up data of a randomized clinical trial. Clin Oral Implants Res. 2020;31(5):488-94.
- Sahrmann P, Naenni N, Jung RE, Hammerle CHF, Attin T, Schmidlin PR. Ten-Year Performance of Posterior 6-mm Implants with Single-Tooth Restorations: A Randomized Controlled Trial. J Dent Res. 2023;102(9):1015-21.
- Mombelli A, Hashim D, Cionca N. What is the impact of titanium particles and biocorrosion on implant survival and complications? A critical review. Clin Oral Implants Res. 2018;29 Suppl 18:37-53.
- Sahrmann P, Schoen P, Naenni N, Jung R, Attin T, Schmidlin PR. Peri-implant bone density around implants of different lengths: A 3-year follow-up of a randomized clinical trial. J Clin Periodontol. 2017;44(7):762-8.
- Sahrmann P, Tartsch J, Schmidlin PR. Comparison of clinical parameters of peri-implantitis and parameters related to tissue macrophage sensitization on TiO(2). Clin Oral Investig. 2024;28(9):499.
- Reito A, Raittio L, Helminen O. Revisiting the Sample Size and Statistical Power of Randomized Controlled Trials in Orthopaedics After 2 Decades. JBJS Rev. 2020;8(2):e0079.
- Valdemarsson S, Ljunggren S, Bramnert M, Norrhamn O, Nordstrom CH. Early postoperative growth hormone levels: high predictive value for long-term outcome after surgery for acromegaly. J Intern Med. 2000;247(6):640-50.
- Dreyer H, Grischke J, Tiede C, Eberhard J, Schweitzer A, Toikkanen SE, et al. Epidemiology and risk factors of peri-implantitis: A systematic review. J Periodontal Res. 2018;53(5):657-81.

