Diabetic Neuro-Ischaemic Foot Syndrome: Key Interactions
Diabetic neuro-ischaemic foot syndrome and the key underlying interactions between chronic arterial disease and peripheral neuropathy
Vlad Adrian Alexandrescu MD, PhD 1, Arnaud Kerzmann MD 1, Vincent Tchana-Sato MD, PhD 1.
- Cardiovascular and Thoracic Surgery Department, CHU Sart-Tilman University Hospital Center, Liège, Belgium.
OPEN ACCESS
PUBLISHED: 28 February 2025
CITATION: Alexandrescu, VA., Kerzmann, A., et al., 2025. Diabetic neuro-ischaemic foot syndrome and the key underlying interactions between chronic arterial disease and peripheral neuropathy. Medical Research Archives, [online] 13(2). https://doi.org/10.18103/mra.v13i2.6364
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i2.6364
ISSN 2375-1924
ABSTRACT
Chronic limb-threatening ischaemia and diabetic peripheral neuropathy are common complications of poorly controlled hyperglycaemia. Numerous clinical evidences suggest that peripheral arterial disease and diabetic peripheral neuropathy may be interconnected pathological entities with bidirectional interactions that respond to hyperglycaemia. The deleterious effects of hyperglycaemia manifest on a systemic scale as well as at the neuro-ischaemic limb level. Persistent hyperglycaemia-related injuries are induced by either direct cellular toxicity or indirect endothelial macrocirculatory damage, leading to cellular dysfunction, increased hypoxia, chronic inflammation, and cellular loss. This paper discusses some of the interactions between chronic limb-threatening ischaemia and diabetic peripheral neuropathy that constitute a complex bidirectional interplay. The scope of this editorial was to explore in depth these possible interactions, as encountered in daily clinical practice, from subclinical to clinical morphological and pathophysiological manifestations. It is also aimed to emphasize the important role played by these two entangled entities, as intricate risk factors for tissue and limb loss, linked to the harmful effects of persistent hyperglicaemia in diabetic, neuro-ischemic limbs. Each significant clinical interaction (or groups of reciprocal interplay) is discussed in association with practical recommendations that are developed in distinct paragraphs, for better clinical applicability.
Keywords:
- chronic limb-threatening ischemia
- diabetic neuropathy
- wound healing
- diabetic foot
- chronic hyperglycaemia
- peripheral angioplasty
- microangiopathy
- arterial calcification
- inferior limb salvage
Introduction
The incidence of diabetes mellitus has dramatically increased in recent decades, with a worldwide incidence of approximately 9.8% in 2019. A recent review revealed that 66% of patients with type 1 DM and 59% of those with type 2 DM showed evidence of diabetic peripheral neuropathy (DPN), a common chronic complication of diabetes mellitus, at comparable time intervals. Painful DPN has been reported in approximately 20%–40% of patients with diabetes mellitus and represents a severe form of DPN that is associated with a high rate of mortality (42%) at five years. DPN is often accompanied by multilevel peripheral arterial disease which is a vascular complication of diabetes mellitus. Peripheral arterial disease and DPN are particularly prevalent in patients with long-term or poorly controlled hyperglycaemia. Chronic limb-threatening ischaemia (CLTI) is a severe form of peripheral arterial disease that is associated with an aggressive macro- and microcirculatory arterial occlusive disease and a mortality rate of 20% within 6 months. Recently, clinical evidence has suggested that CLTI and DPN may represent interconnected evolving pathological entities that are correlated to other similar microvascular systemic manifestations of hyperglycaemia.
The scope of this editorial was to explore the current interactions evinced between CLTI and DPN that are encountered in daily clinical practice from subclinical, to current clinical morphological and pathophysiological manifestations. It is also aimed to emphasize the important role played by these two entangled entities, as intricate risk factors for tissue and limb loss, inside the harmful effects that persistent hyperglycaemia enhances in diabetic, neuro-ischemic limbs.
Material and Methods
PUBLICATION SCREENING
A tandem Medline database collection adding unrestricted online records exploration was performed for all types of publications related to DPN and CLTI aetiological, morphological, and pathophysiological interactions, in diabetic neuro-ischemic feet (Figure 1). Data collection mainly focused on the last decade publications. Seventeen keywords were employed, including: “chronic hyperglycaemia”, “metabolic syndrome”, “chronic limb-threatening ischemia”, “diabetic peripheral neuropathy”, “diabetic autonomic neuropathy”, “diabetic neuro-ischemic foot”, “microangiopathy”, “arterial calcification”, “medial arterial sclerosis”, “inferior limb salvage”, “peripheral angioplasty” etc, without restriction on the paper language. Conference papers, duplicate studies, podcasts, experimental research, monographs, and papers holding poorly or indistinctly analyzed neuro-vascular interactions were excluded.
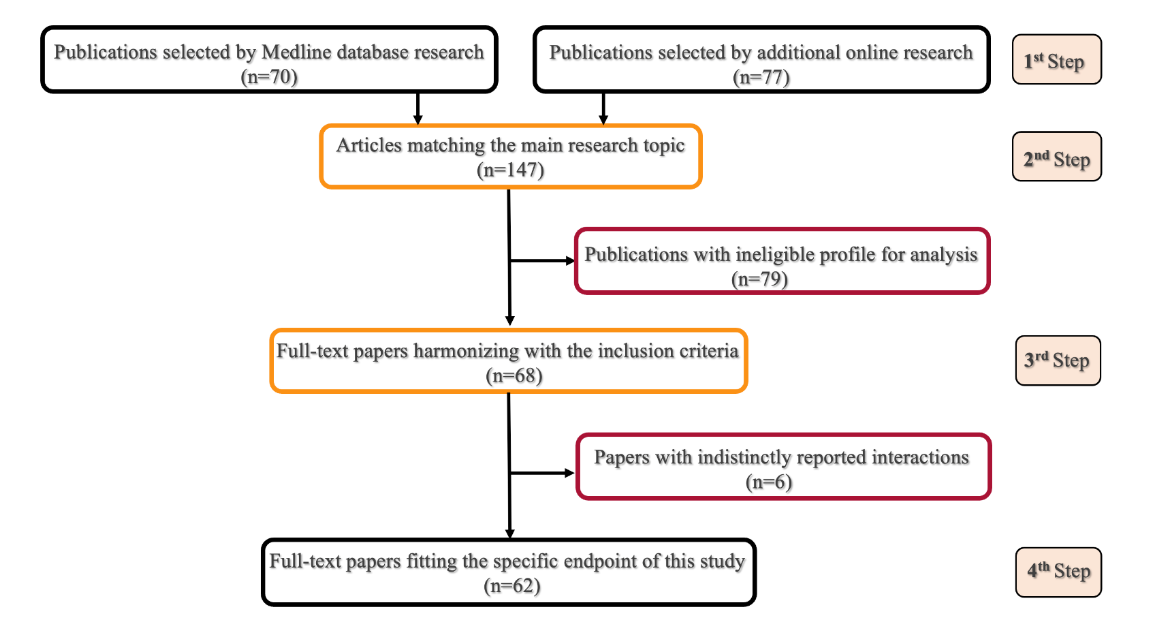
After identification of all publications matching with the key words of this research (1st step in data collection), the non-duplicate papers fitting with the main endpoint were regrouped (2nd selection step). Succeeding the removal of irrelevant publications selected upon title, abstract, and design, and following the withdrawal of the conference papers, experimental research, podcasts, and monographs, the next phase (3rd step) allowed a complementary articles selection, that harmonizes with the inclusion criteria of this research. Succeeding an additional retrieval of papers that indistinctly reported CLTI/DPN interactions, the 4th step was achieved by specific analysis of full-text papers fitting the characteristic end-point of this work.
DATA COLLECTION
Because of the significant heterogeneity in papers profile, study design, and research protocols in the selected studies, several variations in defining and interpreting the reciprocal CLTI/DPN interactions and their clinical implications were observed. However, after incorporation of all data, concrete mention of the bilateral interactions revealed limited, while the evidence-based level was low.
RESEARCH ENDPOINT
The main endpoint of this research was the assessment of all potential reciprocal interactions described between CLTI vs. DPN, that are enhanced by persistent hyperglycaemia in diabetic neuro-ischemic limbs.
Taxonomic Description
Based on the recent taxonomy used in the literature, these conditions can be described as follows: Diabetic neuropathy is commonly defined as the presence of neuropathic symptoms and/or signs of central or peripheral nerve dysfunction after ruling out other systemic causes in individuals with diabetes. The specific definition of DPN (according to the Toronto neuropathy expert group recommendations) is ‘a symmetrical, neural fibre length-dependent sensorimotor polyneuropathy’ following a ‘glove-and-stocking’ limb distribution. Painful DPN can be defined as ‘neuropathic pain appearing as a direct consequence of abnormalities in the peripheral somatosensory neural system in individuals with diabetes mellitus.
CLTI is usually described as an ‘end-stage form of peripheral arterial disease’ that unilaterally affects the ischaemic inferior limb (by rest pain, or ischaemic wounds). It is associated with symptoms persisting for more than two weeks and specific haemodynamic findings (segmental pressures in the affected limb).
Aetiology And Pathophysiology
The overall effects of uncontrolled hyperglycaemia are vast and incompletely understood. Hyperglycaemia is associated with direct cellular toxicity at different organ levels and indirect harm to the same structures through irreversible and interconnected microcirculatory hypoxic degradation.
DIRECT HYPERGLYCAEMIC HARM TO PERIPHERAL NEURAL STRUCTURES
Although hyperglycaemia appears to be the primary cause of DPN in type 1 diabetes mellitus, dyslipidaemia and metabolic syndrome also play important aetiologic roles in type 2 diabetes mellitus. Persistent hyperglycaemia, dyslipidaemia, and altered insulin activation can exert direct toxicity on neural tissue through abnormal macrophage activation, typical stress-induced damage of the endoplasmic reticulum, and persistent hypoxic mitochondrial dysfunction related to the accumulation of abnormal quantities of reactive oxygen species and altered functional signalling. Diabetic peripheral neuropathy is the clinical expression of several deleterious mechanisms showing direct neural toxicity, that harm via the (a) sorbitol pathway, (b) formation of advanced glycation end products, (c) protein kinase C, and (d) hexosamine pathways. Because of these mechanisms, peripheral neurons cease to function normally, may undergo irreversible demyelination, and eventually die.
Several additional risk factors have been suggested, such as (1) possible autoimmune disorders and related glial cell activation, (2) eventual alterations in the functioning of axonal sodium and calcium channels, and (3) central pain perception as a result of increased thalamic perfusion and irregular descending neural pathway control. Myelin plays a crucial role in rapid nerve conduction and interneural junction control. Diabetic peripheral neuropathy is characterised by a specific gradual distal-to-proximal process of unfolding demyelination that corresponds to direct cellular, and/or indirect endothelial microcirculatory damage.
Finally, persistent progression of the aforementioned deleterious hyperglycaemic pathways leads to direct neural destruction, worsening of painful DPN, and regional limb denervation.
INDIRECT HYPERGLYCAEMIA-INDUCED INJURES IN DIABETIC PERIPHERAL NEUROPATHY
Increased reactive oxygen species production and the consequent mitochondrial impairment can be similarly triggered via hyperglycaemic capillary and vasa nervorum damage, owing to the indirect or microvascular-enhanced deleterious primum movens that lead to DPN. In this pathogenic pathway, the initial process activated by persistent hyperglycaemia can damage numerous endothelial cells along the entire neuro-ischaemic inferior limb by various mechanisms. This vast microcirculatory hurdle evolves from subclinical forms to manifest as regional limb denervation. In this process, it affects the kilometric endothelial network of arteriolar and capillary nourishing tissue branches from the skin to the deep tissue. Owing to this distinct microcirculatory affliction, diabetes mellitus is known to induce an increased prevalence of major cardiovascular events. Notably, the neuronal network, which is affected the earliest and shows the most severe injury, is the most fragile biological structure exposed to chronic ischaemia.
DIRECT VS. INDIRECT MACRO AND MICROVASCULAR RESPONSES TO HYPERGLYCAEMIA
The difference between the effects of macro- and microcirculation is useful for understanding the possible detrimental effects of diabetes mellitus on systemic circulation. Although reports on this topic are limited, a contemporary six-level stratification of the inferior limb arterial vasculature proposed the following simple dichotomic stratification based on histologic criteria: the arterial macrocirculatory levels (1–4) from the iliac vessels to the foot arches and down to the large foot arterial collaterals (all featuring a common 5-layer arterial wall structure), distinct from the microcirculatory levels (5 and 6) consisting of arterioles and capillaries with a diameter of approximately 5–100 µm and arterial wall structures with less than five layers. This vast ubiquitarian microvascular system also functions as a performant regulatory functional unit in all irrigated tissues, including peri-arterial vasa vasorum-specific and peri-neural vasa nervorum-specific ramifications.
The capillary endothelial cells and their basement membranes play a fundamental role in ensuring correct irrigation for all types of tissue, including neural, vascular, sensory, and muscular tissue. Therefore, morphological damage or functional collapse of this endothelial barrier at the macrovascular level (levels 1–4) or at the microvascular level (levels 5 and 6) may enhance or exacerbate various forms of ischaemic tissue damage.
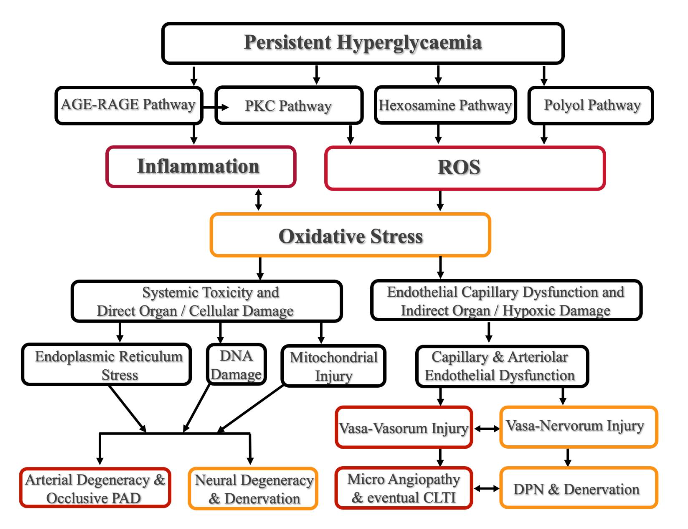
Several deleterious metabolic pathways induced by chronic hyperglycaemia (HG) were described, and are schematically depicted such as: Advanced Glycation End-product (AGE) adding Receptor for Advanced Glycation End-product (RAGE) pathway, Protein Kinase C (PKC) Pathway, Hexosamine Pathway, and Polyol Pathway. All these pathological courses converge to producing chronic inflammation and high-level reactive oxygen species (ROS), that inflict deleterious oxidative stress to tissues. As a consequence, the oxidative stress induces its harmful effect either by direct cellular damage, or by indirect hypoxic tissue injury (mediated by capillary and arteriolar endothelial decay), and dysfunction. The direct tissue toxicity of HG (the left side of the diagram) leads to cellular Endoplasmic Reticulum damage, to DNA and mitochondrial alteration, to cellular and organ dysfunction and decay, and finally to cellular loss. Alike all targeted tissues by HG, the peripheral arterial and neural systems express irreversible structural and functional degeneracy. These changes lead finally to the peripheral arterial disease (PAD) onset, and a parallel neural degeneracy manifested as diabetic peripheral neuropathy (DPN); this latest can finally conduct to limb denervation.
A concomitant indirect HG affliction (the right side of the diagram) starts with a vast endothelial capillary and arteriolar dysfunction, that also includes the tiny vasa-vasorum and vasa nervorum vessels, responsible for nourishing the peripheral arterial and neural structures. Their injury and decay induce additional tissue hypoxia, that worsens the already installed direct HG organ toxicity. In more advanced stages of the diabetic neuro-ischemic limbs, the microvascular decay joins the macrovascular (PAD) occlusive lesions, eventually leading to chronic limb-threatening ischemia (CLTI) and the jeopardy for tissue, or inferior limb loss.
LARGE ARTERIAL STRUCTURES AND VASA VASORUM
Large arterial structures that provide both oxygen and nutrient supply are capable of storing and transporting glucose directly from the bloodstream (independent of individual insulin levels) and indirectly via a vasa vasorum network of arterioles and capillaries that nourish and support the essential metabolic needs of the arterial wall structures. Therefore, vasa vasorum damage can trigger large-scale morphological and functional pathological phenomena in the host arteries. The macrovascular effects of hyperglycaemia (levels 1–4) are currently associated with other recognised deleterious cardiovascular risk factors (hypertension, high body mass index [BMI], dyslipidaemia, and chronic inflammation).
In limbs affected by peripheral arterial disease and CLTI, diabetes mellitus appears to directly inflict a characteristically more aggressive (specific long stenoses and occlusions in the tibial and pedal arteries) and more distally situated (downstream of the foot) arterial atherosclerotic occlusive disease. Specific microcirculatory damage (levels 5 and 6) to the endothelial cells encountered in patients with diabetic CLTI leads to reduced blood flow to the peripheral tissue, increased capillary permeability, progressive imbalance between arteriogenesis and angiogenesis, progressive thickening of the capillary basement membrane, and the medial layer of the arteriolar wall. All these pathological changes indicate the development of a chronic, hypoxic, and pro-inflammatory status that further increases local injury to the endothelial cells. This pro-thrombotic state equally occurs because of the growing levels of procoagulant circulating factors, and decreased endothelial nitric oxide production. This adverse environment ultimately leads to arteriolar and capillary microthrombosis and collapse, accelerating the loss of small arterial collaterals and increasing tissue ischaemia; accelerating hypoxia frequently is accompanied by sepsis, eventually leading to loss of the regional tissue or the entire limb.
To characterise this multifactorial neurovascular background, O’Neal et al proposed the original concept of global vascular decay as end-artery occlusive disease, a diabetic neuro-ischaemic condition. According to this theory, ‘a few millimetres of skin, up to the entire foot, may depend on one single, still permeable, nourishing artery’. Various primary glucose transporters are located in various types of cells and organs, and these transporters represent specific barriers to the hyperglycaemic hurdle. However, not all of these transporters function similarly; they act via different molecular pathways and across different timelines to antagonise elevated glycaemic levels. The specificity of these transporters may explain why specific target tissues, such as the kidney, retina, and peripheral neural structures (including those involved in complex microcirculatory diabetic foot syndrome), are more likely to develop intracellular hyperglycaemic toxicity secondary to poor blood glucose regulation. Consequently, these ‘primary target tissues’ seem to be at a higher risk for developing diabetes mellitus microangiopathy and the related complications.
Therefore, microvascular harm in long-term diabetes mellitus progressively leads to additional multiorgan dysfunction beyond the aforementioned first-line targets. This form of systemic hyperglycaemic microcirculatory aggression includes microvascular harm to the brain (lacunar stroke, cognitive and behaviour impairment), as well as macro- and microvascular damage at the cardiac level (diabetic cardiomyopathy), injury to the entire circulatory system (as a result of vasa vasorum dysfunction), parallel lung damage (the ‘narrowing alveolar space’ syndrome), microcirculatory effects on the skin (independent diabetic dermopathy), and adjoining independent muscular and osteo-articular damage (commonly observed in diabetic foot syndrome).
Interactions Between Diabetic Peripheral Neuropathy and Peripheral Arterial Disease
ANATOMICAL AND FUNCTIONAL SIMILARITIES
Like any other territory of the human body, the inferior limb shows a characteristic and reproducible anatomical distribution of neurovascular bundles, which was first described by Taylor et al about three decades ago. Consistent with the topographic distribution of vessels (following the foot ‘angiosomes’ and correspondent ‘venosomes’) and parallel to the skin-sensitive neural partition (the ‘dermatomes’ of the foot), Taylor et al documented reproducible similarities between the peripheral nerves and adjoined ramifications for neighbouring vessels. This replicable neurovascular scaffolding assumes the location of topographic neurovascular bundles for correct tissue functioning.
PRECLINICAL NEUROVASCULAR INTERACTIONS
The anatomical and functional integrity of neurovascular bundles, which serve as topographic ‘pacing hubs’ for tissue irrigation, is essential for balanced, and reciprocally enhanced activity. Consequently, an incipient bidirectional interplay between nascent DPN and peripheral arterial disease (at the subclinical level) can be suspected in apparently asymptomatic patients if they show specific risk factors and concomitant retinal or renal microvascular dysfunction. Asymptomatic DPN in the limbs has been reported to occur in up to 50% of patients with diabetes mellitus, whereas peripheral arterial disease occurs in approximately 29% of patients with diabetes mellitus.
Embryonic neuropathy and arteriopathy can mutually sustain a bilateral evolution, starting at the neurovascular bundles level (through a reciprocal increase in regional tissue hypoxia); this is particularly evident as the haemoglobin A1c level consistently exceeds >7%, despite the potentially misleading absence of any initiatory clinical symptoms. Accurate diagnosis of these incipient forms of DPN or peripheral arterial disease at this evolutionary stage is difficult. While certain diabetic patients with CLTI may be asymptomatic, approximately 50% of those with CLTI have no previous history of peripheral arterial disease.
PRACTICAL POINT
For early identification of any early form of DPN or peripheral arterial disease, thorough multidisciplinary and timely surveillance is essential in patients with such indeterminate conditions. This approach is particularly useful for patients with indicative risk factors (genetic data and/or parallel retinal or renal clinical manifestations).
EARLY CLINICALLY IDENTIFIABLE NEUROVASCULAR INTERACTIONS
Approximately 80% of patients with diabetic CLTI exhibit various forms of DPN, and approximately 37% of these patients exhibit concomitant latent or obvious features of peripheral arterial disease. Certain preliminary and clinical reciprocal influences between DPN and peripheral arterial disease have been described as being mediated by neighbouring inflammatory and hypoxic stimuli, as both entities may together affect adjacent neurovascular bundles and specific foot territories. This incipient phenomenon is not singular; it appears to induce global multiorgan mitochondrial endothelial cell dysfunction, including neighbouring vasa vasorum- and vasa nervorum-specific ramifications.
Notably, this initial unidirectional pathological pathway gradually associates with other parallel (inflammatory, autoimmune, metabolic, and genetic) influences and progressively evolves into a bidirectional manifestation of the interactions between DPN and CLTI. These mutual connections can be succinctly described as follows: a) Starting with CLTI, increasing microcirculatory tissue hypoxia and aggravation (demyelination) of parallel DPN can be expected. This eventually leads to painful DPN vs. complete territorial denervation. b) Conversely, starting with aggressive forms of DPN or painful DPN these can lead to abnormal tissue repair, loss of local vasculogenesis, and parallel inflammation and dysfunction of neurotrophic tissue growth factor secretion. A related reduction in dermal and muscular neural fibres is observed at this stage. This phenomenon also parallels irreversible structural changes in the arteriolar and capillary walls that lead to local microcirculatory thrombosis, and a critical loss of foot collaterals (the “end-artery disease theory); this escalating condition then promotes CLTI and the destruction of the surrounding vascular tissue, eventually resulting in unavoidable tissue necrosis and limb loss.
PRACTICAL POINT
This type of interaction can explain why certain presentations of diabetic CLTI may show atypical manifestations associated with synchronous DPN damage in the limbs and vice-versa. It can also highlight the importance of tandem assessment of CLTI and DPN (in addition to all other parallel risk factors) by a multidisciplinary approach. Although CLTI and DPN manifest separately, they are two inseparable facets of the same coin.
NEURAL DYSFUNCTION AND CUTANEOUS MICROCIRCULATORY SHUNTING
Microcirculatory changes in CLTI induce a persistent decline in arteriolar and capillary flow, jeopardising the vasa nervorum vessels. This leads to ancillary DPN decay that precipitates sympathetic denervation, increases local vasodilatation, and increases microcirculatory arteriovenous shunting at cutaneous and muscular levels. This abnormal arteriovenous shunting further triggers a local ‘steal phenomenon’ of the oxygenated blood and abnormal microvascular irrigation to the skin. Thus, the worsening sympathetic DPN further strengthens the CLTI-induced hypoxic microvascular destruction.
PRACTICAL POINT
Even angiographically proven correct revascularisation of the CLTI foot may not yield regular and complete tissue regeneration, unless the inherently associated microangiopathy, DPN, and possible cutaneous steal phenomenon are identified and controlled. These specific pathological conditions should be treated together with other current risk factors (sepsis, local pressure, inflammation, etc.) using a multidisciplinary approach.
TIBIAL AND PEDAL ARTERIAL CALCIFICATIONS AND AUTONOMIC DIABETIC PERIPHERAL NEUROPATHY
Medial arterial sclerosis and medial arterial calcification were first reported by Mönckeberg et al in 1903. Their implications in complex diabetic foot pathology have only been thoroughly considered in the last two decades. A parallel form of microvascular small-artery disease was recently individualised to be associated with medial arterial sclerosis and medial arterial calcification, and was proven to play an important role for worse clinical outcomes in patients with CLTI. In the multifactorial background of diabetic foot syndrome, which is associated with the bidirectional interplay of DPN and CLTI, small-artery disease and medial arterial calcification are currently considered analogous manifestations of the same microvascular progressive disease, which worsens the outcomes of CLTI. Recently, Lanzer et al observed that the more extended the below-the-knee calcification, the higher the risk of major amputation. Although the precise pathophysiologies of medial arterial sclerosis and medial arterial calcification are not fully known, a possible interrelationship between salient tibial-pedal calcification and DPN (often type 2 diabetes mellitus) was previously reported by Edmonds et al in 1982. Specifically, these bulky below-the-knee calcific deposits were found in patients with DPN and specific sympathetic fibre damage. Medial arterial calcifications, which present with higher calcium levels and a reduced fibro-lipid structure, have been equally described in below-the-knee arteries in patients with diabetes and in those with renal disease. Most medial arterial calcifications are associated with long chronic arterial occlusions, increased wall stiffening, and higher mortality rates. Notably, the same type of medial arterial calcification lesions in below-the-knee arteries were also reported in patients following therapeutic lumbar sympathectomy. These interesting findings indicate the possibility of an analogous mechanism underlying the interaction between sympathetic denervation and abundant below-the-knee arterial calcifications, which remains to be elucidated.
PRACTICAL POINT
Patients with sympathetic DPN may show extended tibial and pedal artery calcifications and chronic occlusions, and their interventionists should be aware that these patients are more likely to require highly challenging revascularisation procedures, which are associated with lower patency rates and higher technical failure rates at two years. Below-the-knee angioplasty trials should equally consider accurate DPN and calcification assessment, while providing patency and limb salvage data in diabetic patients.
Conclusion
Adequate macro- and microvascular flow restoration and follow-up assessments to achieve appropriate limb preservation in diabetic neuro-ischemic limbs require careful examination of all potential ongoing risk factors, including complex and reciprocal neurovascular interplay.
Conflict of Interest:
The authors have no conflicts of interest to declare.
Acknowledgments:
The corresponding author acknowledges the Cardio-Vascular and Thoracic Surgery Department academic team, for their courtly support in refining this paper.
References
- Schaper NC, van Netten JJ, Apelqvist J, et al. Practical guidelines on the prevention and management of diabetes-related foot disease (IWGDF 2023 update). Diab Metab Res Rev. 2024; e3657. https://doi.org/10.1002/dmrr.3657.
- Hingorani A, LaMuraglia GM, Henke P, et al. The management of the diabetic foot: a clinical practice guideline by the Society for Vascular Surgery in collaboration with the American Podiatric Medical Association and the Society for Vascular Medicine. J Vasc Surg. 2016;63(2 Suppl):3S–21S. doi: 10.1016/j.jvs.2015.10.003.
- Zaffar H. An introduction to Diabetic Neuropathy: A Review. World J Pharm Sciences. 2016; (Print): 2321-3310, (Online): 2321-3086. http://www.wjpsonline.org/
- Chang MC, Yang S. Diabetic peripheral neuropathy essentials: a narrative review. Ann Paliat Med. 2023;12(2):390-398. https://dx.doi.org/10.21037/apm-22-693.
- Mallick -Searle T, Adler JA. Update on treating painful diabetic peripheral neuropathy: a review of current US guidelines with a focus on the most recently approved management options. J Pain Res. 2024; 17:1005-1028. doi: 10.2147/JPR.S442595. eCollection 2024.
- Tesfaye S, Boulton AJM, Dyck PJ, et al. Diabetes neuropathies: update on definitions, diagnostic criteria, estimation of severity, and treatments. Diab Care. 2010;33:2285-2293. doi: 10.2337/dc10-1303.
- Jensen TS, Karlsson P, Gylfadottir SS, et al. Painful and non-painful diabetic neuropathy, diagnostic challenges and implications for future management. Brain. 2021; 144; 1632-1645. doi: 10.1093/brain/awab079.
- Alexandrescu VA, Van Overmeire L, Makrygiannis G, et al. Clinical implications of diabetic peripheral neuropathy in primary infrapopliteal angioplasty approach for neuro-ischemic foot wounds. J Endovasc Ther. 2022;30(6)920-930. doi: 10.1177/15266028221106312.
- Conte MS, (4) Bradbury AW, Kolh P, et al. Global vascular guidelines on the management of chronic limb-threatening ischemia. Eur J Vasc Endovasc Surg. 2019;58(1S):S1-S109.e33.70:662. doi: 10.1016/j.ejvs.2019.05.006.
- Uccioli L, Meloni M, Izzo V, Giurato L, Merolla S, Gandini R. Critical limb ischemia: current challenges and futures perspectives. Vasc Health Risk Manag. 2018;14:63-74.
- Rosenberg DC, Blechschmidt V, Timmerman H, Wolff A, Treede RD. Challenges of neuropathic pain: focus on diabetic neuropathy. J Neural Transm. 2020; 127(4):589-624. doi: 10.1007/s00702-020-02145-7.
- Alexandrescu VA, Kerzmann A, Boesmans E, et al. Singularities of the pedal circulation in CLTI: time for novel merging guidelines. J Crit Limb Ischem. 2023;3: E1.
- O’Neal LW. Surgical pathology of the foot and clinicopathologic correlations. The end-artery occlusive disease conception. In: Bowker JH, Pfeifer MA. Chapter 18, Levin and O’Neal’s The Diabetic Foot. 7th Ed. Mosby Elsevier, Philadelphia. 2007:367-401.
- Shang Y, Zilin C, Dongye Z, Shuangmei L. Different types of cell death in diabetic neuropathy: a focus on mechanisms and therapeutic strategies. Int J Mol Sci. 2024;25(15):8126. https://doi.org/10.3390/ijms25158126.
- Wei J, Tian J, Tang C, et al. The influence of different types of diabetes on vascular complications. J Diabetes Res. 2022;22:2022:344818. doi: 10.1155/2022/3448618. eCollection 2022.
- Mauricio D, Gratacos M, Franch-Nadal J. Diabetic microvascular disease in non-classical beds: the hidden impact beyond the retina, the kidney, and the peripheral nerves. Cardiovasc Diabetol. 2023;22:314. doi: 10.1186/s12933-023-02056-3.
- Alexandrescu VA, Pottier M, Balthazar S, Azdad K. The foot angiosomes as integrated level of lower limb arterial perfusion: amendments for chronic limb threatening ischemia presentations. J Vasc Endovasc Ther. 2019;4(1):1-7. http://vascular-endovascularsurgery.imedpub.com/
- Mansour A, Mousa M, Abdelmannan D, Tay G, Hassoun A, Alsafar H. Microvascular and macrovascular complications of type 2 diabetes mellitus: exome wide association analyses. Front Endocrinol. 2023; 14:1143067. doi: 10.3389/fendo.2023.1143067.
- Taylor GI, Gianoutsos MP, Morris SF. The neurovascular territories of the skin and muscles: anatomic study and clinical implications. Plast Reconstr Surg. 1994; 94(1): 1-36.
- Atmaca A, Ketenci A, Sahin I, et al. Expert opinion on screening, diagnosis and management of diabetic peripheral neuropathy: a multidisciplinary approach. Front Endocrinol. 2024; 17:15:1380929 doi: 10.3389/fendo.2024.1380929. eCollection 2024.
- Jörneskog G. Why critical limb ischemia criteria are not applicable to diabetic foot and what the consequences are. J Scan Surg. 2012; 101(2): 114-118. doi: 10.1177/145749691210100207.
- Csonka V, Varju C, Lendvay M. Diabetes mellitus-related musculoskeletal disorders: unveiling the cluster of diseases. Prim Care Diabetes; 2023; 17: 548-553. doi: 10.1016/j.pcd.2023.08.003.
- Li Y, Miao Y, Feng Q, et al. Mitochondrial dysfunction and onset of type 2 diabetes along with its complications: a multi-omics Mendelian randomization and colocalization study. Front Endocrinol. 2024; 15: 1401531. doi: 10.3389/fendo.2024.1401531.
- Cheng Y, Chen Y, Li K, et al. How inflammation dictates diabetic peripheral neuropathy: an enlightening review. CNS Neurosci Ther. 2024;30(4):e14477. doi: 10.1111/cns.14477.
- Fiordaliso F, Clerici G, Maggioni S, et al. Prospective study on microangiopathy in type 2 diabetic foot ulcer. Diabetologia. 2016;59(7):1542–1548. doi: 10.1007/s00125-016-3961-0.
- Alexandrescu VA, Neville RF, Kerzmann A, et al. Applicability of intentional topographic revascularization following the angiosome model in patients with chronic limb-threatening ischemia: from theory, to current clinical challenges. Clin Surg. 2024;9:3701. https://dx.doi.org/10.25107/cis-v9-id3701.
- Arora S, Pomposelli F, LoGerfo FW, et al. Cutaneous microcirculation in the neuropathic diabetic foot improves significantly but not completely after successful lower extremity revascularization. J Vasc Surg. 2002;35(3):501–505. doi: 10.1067/mva.2002.121126.
- Alexandrescu VA, Brochier S, Schoenen S, et al. Grades of below-the-ankle arterial occlusive disease following the angiosome perfusion: a new morphological assessment and correlations with the inframalleolar GVG stratification in CLTI patients. Ann Vasc Surg. 2022.1;1-20. https://doi.org/10.1016/j.avsg.2021.09.031.
- Mönckeberg JG: Über die reine Mediaverkalkung der extremitätenarterien und ihr verhalten zur arteriosklerose. Virch Arch. 1903; 169: 141-67.
- Lanzer P, Ferraresi R. Medial sclerosis-epidemiology and clinical significance. Dtsch Arztebl Int. 2023; 120(21): 365–372. doi: 10.3238/arztebl.m2023.0066.
- Ferraresi R, Ucci A, Pizzuto A, et al. A novel scoring system for small artery disease and medial arterial calcification is strongly associated with major adverse limb events in patients with chronic limb-threatening ischemia. J Endovasc Ther. 2021;28(2):194-207. doi: 10.1177/1526602820966309.
- Liu IH, Wu B, Krepkiy V, et al. Pedal arterial calcification score is associated with hemodynamic change and major amputation after infrainguinal revascularization for chronic limb-threatening ischemia. J Vasc Surg. 2022; 76: 1688–1697.
- Lanzer P, Hannan FM, Lanzer JD, et al. Medial arterial calcification, JACC state-of-the-art review. J Am Coll Cardiol. 2021;78(11): 1145–1165. doi: 10.1016/j.jacc.2021.06.049.
- Edmonds ME, Morrison N, Laws JW, Watkins PJ. Medial arterial calcification and diabetic neuropathy. Br Med J. 1982;284(6320):928–930. doi: 10.1136/bmj.284.6320.928.
- Mustapha JA, Diaz-Sandoval LJ, Saab F. Infrapopliteal calcification patterns in critical limb ischemia: diagnostic, pathologic and therapeutic implications in the search for the endovascular holy grail. J Cardiovasc Surg (Torino). 2017;58(3):383-401. doi: 10.23736/S0021-9509.17.09878-0.




