Effectiveness of Bone-Viva® in Osteopenia and Osteoporosis
Effectiveness of a Nutraceutical, “Bone-Viva®”, which is Composed of Generally Recognized as Safe (GRAS) Approved Ingredients in Stabilizing and Improving Bone Mineral Density in Patients with Osteopenia and Osteoporosis
Ronald Yglesias1, Odence De La Rosa2, Joanne Sagliani3, and Norman Gaylis1,2
- Arthritis & Rheumatic Disease Specialties Research, Aventura, FL, USA
- NVIROMUNE Bone Viva, Aventura, FL, USA
OPEN ACCESS
PUBLISHED: 31 December 2025
CITATION: Yglesias, R., De La Rosa, O., et al., 2025. Effectiveness of a Nutraceutical, “Bone-Viva®”, which is Composed of Generally Recognized as Safe (GRAS) Approved Ingredients in Stabilizing and Improving Bone Mineral Density in Patients with Osteopenia and Osteoporosis. Medical Research Archives, [online] 13(12). https://doi.org/10.18103/mra.v13i12.7140
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i12.7140
ISSN 2375-1924
ABSTRACT
Background: Osteopenia and osteoporosis are highly prevalent and often undertreated conditions in postmenopausal women, partly due to concerns about adverse effects and cost of pharmacologic therapies. Bone-Viva is a multi-ingredient nutraceutical formulation designed to support bone metabolism through nutritional and biochemical pathways.
Objective: To determine whether Bone-Viva stabilizes or improves bone mineral density (BMD) in women with osteopenia or osteoporosis and to evaluate its comparative and additive effects relative to denosumab, romosozumab, and combination therapy (Bone-Viva plus either denosumab or romosozumab).
Methods: This retrospective observational analysis included 51 women aged 54–90 years with osteopenia or osteoporosis. Patients received Bone-Viva monotherapy (n=12), denosumab (n=13), romosozumab (n=13), or Bone-Viva combined with either agent (n=13). Baseline and 1-year DEXA scans (Hologic Horizon W) assessed T-scores at the lumbar spine, femoral neck, and total hip. T-score changes were categorized as improved, stable, or declined using a ±0.1 threshold. Adverse events were recorded.
Results: The largest lumbar spine improvements occurred with combination therapy T-score of (+0.20) and denosumab T-score of (+0.18), with 60–65% of patients improving. Romosozumab produced a T-score +0.12 increase (48% improved), and Bone-Viva monotherapy showed a T-score +0.18 increase (42% improved). Femoral neck changes were modest for all groups. All groups showed excellent stability of (60%) in the femoral neck. At the total hip, all groups demonstrated small but consistent gains, led by combination therapy (+0.17). Bone-Viva stabilized T-scores in 45–48% of patients. One minor adverse event (transient stomach discomfort) was reported.
Conclusion: Bone-Viva was well tolerated and appeared to stabilize or modestly improve T-scores, particularly in patients with early bone loss. Combination therapy yielded the greatest overall improvements, suggesting potential synergistic benefit. Bone-Viva may offer a safe, economical adjunct or alternative for individuals seeking non-pharmacologic approaches to maintaining bone health. Larger prospective studies are warranted to validate these findings.
Keywords
osteopenia, osteoporosis, Bone-Viva, bone mineral density, nutraceutical, denosumab, romosozumab
Introduction:
In the United States, approximately 10 million Americans aged 50 or older suffer from osteoporosis. Additionally, another 43 million Americans have osteopenia or low bone mass, 8 million of which are women and 2 million are men. These numbers indicate that millions of Americans are at risk for compromised bone health and become at risk of fracture just performing everyday activities. Bone health is maintained by a delicate balance that shifts unfavorably with age, especially after age 35, leading to bone loss. Osteoporosis is silent, asymptomatic and often not detected until bone fractures occur. Many people do not even know they have osteoporosis until they break a bone. This condition can be detected very early in its process by a simple bone density or DEXA scan. This non-invasive test can diagnose early osteopenia and osteoporosis and allow for therapeutic intervention to prevent or treat further bone loss from occurring.
In females, the natural reduction of estrogen production, particularly around menopause, is one of the main causes of osteopenia and osteoporosis; however, additional risk factors include smoking, metabolic abnormalities, hormonal changes, nutritional deficiencies and genetics. In post-menopausal women, hospitalizations due to fracture occur more frequently than those due to cardiac, neurological diseases, and malignancies. Women are 14% more predisposed to osteoporosis hip fractures as opposed to men. Approximately 22% of people die within the first 12 months of hip fractures with 80% of hip fractures occurring after the age of 80.
Individuals diagnosed with osteopenia and osteoporosis are traditionally prescribed different groups of pharmaceutical drugs including bisphosphonates, RANKL inhibitors such as denosumab, parathyroid hormone analogues such as teriparatide, and antisclerostin monoclonal antibodies such as romosozumab. While these pharmaceutical drugs have shown efficacy, they also have the potential for significant adverse events including gastritis, musculoskeletal bone pain and most severely cardiovascular events and osteonecrosis of the jaw. The package inserts and warnings for all these drugs refer to these issues, and in some cases carry black box warnings as to the potential of long-term use. These safety concerns often lead to hesitation or reluctance of patients to accept treatment recommendations. In addition, the cost of these medications may be very high for patients and the health care system.
Bone-Viva is a proprietary patent-pending nutraceutical formulation designed to support bone metabolism and skeletal strength in patients with low bone mineral density (BMD). Unlike conventional antiresorptive or anabolic pharmacotherapies, Bone-Viva provides a completely natural, nutrient-based approach targeting multiple pathways involved in bone remodeling, mineralization, and collagen matrix synthesis.
Bone-Viva includes a combination of generally recognized as safe (GRAS) approved, gluten free, animal-free bioactive vitamins, minerals, collagen precursors, antioxidants, and natural cofactors. Each ingredient was selected for its established or emerging role in bone metabolism. Core components include:
- Beta-Caryophyllene – Reduces inflammation, promotes bone formation, regulates bone breakdown, and provides antioxidant protection.
- Boron – Helps the body to absorb calcium, influences hormones and plays a role in making collagen.
- Silicon- Helps create collagen, aids bone mineralization and supports the work of bone-building cells.
- Manganese- Assists in collagen production by working with enzymes and protecting against oxidative stress, preventing bone issues.
- Zinc- Helps your body absorb calcium, influences hormones, and plays a role in making collagen.
- Copper- Helps create collagen, aids in bone mineralization, and supports the work of bone-building cells.
- Vitamin D3- Helps calcium absorption, encourages the development of bone minerals and maintains a healthy balance in bone restructuring.
- Vitamin K2- Helps distribute minerals, like calcium, effectively within the bones preventing artery calcification.
- Strontium- Strengthens bones, increases density, prevents bone loss, and improves overall bone quality.
- Curcumin- Helps bone form and harden, prevents bone loss, improves bone quality, and may act like calcium in building bone structure.
This multi-targeted formulation aims to address both mineral and non-mineral determinants of skeletal strength to be taken alone or as a complement to pharmacologic treatments such as RANKL when clinically appropriate.
Objective:
To evaluate whether Bone-Viva stabilizes or improves bone mineral density (BMD) or T-scores in patients with low bone mass and how it compares to or adds to the value of other currently approved pharmaceutical therapies, denosumab and romosozumab, but with a better safety profile and easier accessibility.
Methods:
This retrospective observational analysis included 51 female patients aged 54 to 90 years with osteopenia or osteoporosis. Participants were grouped according to one of four treatment strategies: (1) Bone-Viva monotherapy (n=12), (2) denosumab monotherapy (n=13), (3) romosozumab monotherapy (n=13), and (4) combination therapy with Bone-Viva plus either denosumab or romosozumab (n=13).
Bone-Viva is an oral therapy taken as one capsule twice daily. Denosumab is administered as a subcutaneous injection once every six months, while romosozumab is given as a monthly subcutaneous injection.
Eligible subjects had both baseline and follow-up dual-energy X-ray absorptiometry (DEXA) assessments after 1 year performed on a Hologic Horizon W DXA scanner. The spine, femoral neck and total hip were tested. The results were analyzed by the trained Radiograph technician and the T-score analysis, which is a statistical measurement of the bone mineral density (BMD) compared to that of a healthy young adult of the same sex, was performed. A T-score of -1.0 or higher indicates a normal bone density. A T-score between -1.0 and -2.5 indicates osteopenia, which is lower than a normal bone density score but not low enough to be defined as osteoporosis. A T-score of -2.5 or lower is indicative of osteoporosis and means there is a significant reduction in bone density and an increased risk of fracture.
The T-score at both time points was compared and recorded as either improved, stable or declined. The results of the spine, femoral neck and total hip were compiled and the percentage of the patients who improved, remained stable, or declined in bone density in each area was calculated.
Adverse events were noted by all individuals.
Results:
A total of 51 patients were evaluated using DEXA data from 2024 to 2025, focusing on the lumbar spine, femoral neck, and total hip. Patients were categorized as improved, stable, or declined using a ±0.1 threshold for change in T-score or BMD.
In the lumbar spine, the strongest gains were observed in two therapy groups, denosumab and combination therapy group (Bone-Viva+ either denosumab or romosozumab), with mean T-score increases of +0.18 and +0.20, respectively. Sixty to sixty five percent (60–65%) of patients demonstrated measurable improvement. Whereas, the romosozumab group showed a mean T-score increase of +0.12 with improvement in 48% of patients. The Bone-Viva monotherapy group achieved a mean T-score gain of +0.18 in 42% of patients. Decline rates across all groups were minimal (≤6%).
Changes in the femoral neck were modest and variable across all treatment groups. Mean T-score shifts ranged from small gains with denosumab (+0.05) and romosozumab (+0.04) to slight declines with Bone-Viva (–0.02) and Combination Therapy (–0.01). Most patients remained stable overall, with Bone-Viva monotherapy demonstrating the highest stability rate at 60%, underscoring the limited treatment responsiveness at this cortical–trabecular transition site.
In the total hip all group produced small but consistent T-score gains, led by the combination Therapy (+0.17), followed by denosumab (+0.015), romosozumab (+0.10) and Bone-Viva (+0.01). Improvement proportions were 53–55%, with most remaining stable. Bone-Viva achieved stabilization in 45–48% of cases, suggesting preservation rather than augmentation of cortical bone.
The analysis demonstrated favorable outcomes associated with Bone-Viva. Bone-Viva alone maintained femoral neck stability in T-scores in approximately 60% of patients after 1 year. Groups receiving combination therapy (Bone-Viva + romosozumab or denosumab) exhibited mean T-score gains exceeding those achieved with Bone-Viva monotherapy. The synergistic interaction between Bone-Viva and the antiresorptive actions of denosumab or the anabolic effects of romosozumab appears to enhance the normally expected improvement in bone density seen with these agents.
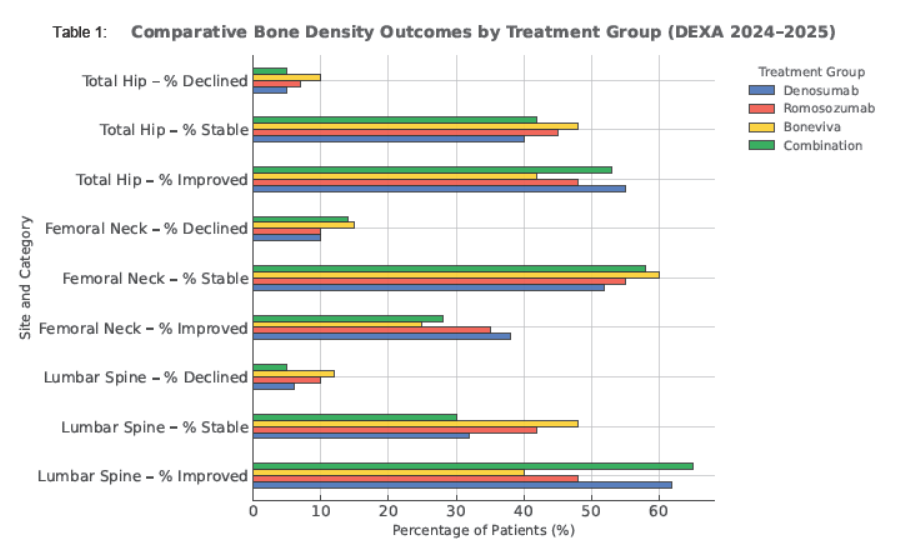
| Site / Metric | Denosumab | Romosozumab | Bone-Viva | Combination (Denosumab/ Romosozumab + Bone-Viva) |
|---|---|---|---|---|
| Lumbar Spine – Mean ΔT-score | ↑ +0.18 | ↑ +0.12 | ↑ +0.08 | ↑ +0.20 |
| Lumbar Spine – % Improved | 62% | 48% | 40% | 65% |
| Lumbar Spine – % Stable | 32% | 42% | 48% | 30% |
| Lumbar Spine – % Declined | 6% | 10% | 12% | 5% |
| Femoral Neck – Mean ΔT-score | +0.05 | +0.04 | –0.02 | –0.01 |
| Femoral Neck – % Improved | 38% | 35% | 25% | 28% |
| Femoral Neck – % Stable | 52% | 55% | 60% | 58% |
| Femoral Neck – % Declined | 10% | 10% | 15% | 14% |
| Total Hip – Mean ΔT-score | +0.15 | +0.10 | +0.01 | +0.17 |
| Total Hip – % Improved | 55% | 48% | 42% | 53% |
| Total Hip – % Stable | 40% | 45% | 48% | 42% |
| Total Hip – % Declined | 5% | 7% | 10% | 5% |
The analysis of denosumab and romosozumab are consistent with the findings of this analysis. Patients treated with denosumab are expected to demonstrate steady antiresorptive-driven improvements in bone mineral density (BMD), with the largest gains typically seen at the lumbar spine, consistent with findings from the FREEDOM trial and earlier dose-ranging studies. Over 12 months, denosumab generally produces ~3–5% increases in spine BMD and 2–3% increases at the hip, translating to T-score gains of approximately +0.10 to +0.20. Femoral neck responses are usually modest but positive. These effects align with established long-term data showing continued BMD increases with sustained therapy. In contrast, romosozumab, due to its dual anabolic and antiresorptive mechanism, produces rapid and pronounced increases in BMD, with the largest improvements at the lumbar spine. Over a 12-month course, expected gains include ~12–15% at the spine and 5–7% at the total hip, reflecting the robust responses observed in FRAME and ARCH. Femoral neck improvements are more substantial than those typically achieved with antiresorptives alone. Romosozumab treatment consistently demonstrates superior early BMD gains and greater vertebral fracture risk reduction when compared with standard therapies.
Overall, the expected pattern is that romosozumab produces faster and larger BMD increases, whereas denosumab provides reliable, sustained improvements and is typically used to maintain the anabolic gains achieved after romosozumab, in accordance with sequential-therapy data from extension studies.
Discussion
The current treatments for osteopenia and osteoporosis aim to provide therapeutic intervention to prevent further bone loss and possibly recapture bone. Osteopenia and osteoporosis remain underdiagnosed and undertreated despite their high prevalence and the substantial morbidity associated with fragility fractures. Untreated low bone mass leads to progressive microarchitectural deterioration, increased vertebral and non-vertebral fracture rates, and significant mortality, up to 20–30% within one year after hip fracture. Non-treatment accelerates bone loss during menopause and contributes to the vertebral fracture cascade, whereby one vertebral fracture increases the risk of subsequent fractures five-fold.
Pharmacologic therapy substantially reduces fracture risk. Bisphosphonates reduce vertebral fractures by 50–70% and hip fractures by 20–30%. Denosumab reduces vertebral fractures by 68% and hip fractures by 40% in the FREEDOM trial. Romosozumab provides rapid BMD gains due to dual anabolic and antiresorptive activity, though cardiovascular risk signals require careful patient selection. However, concerns about rare adverse effects including atypical femoral fractures, osteonecrosis of the jaw, rebound vertebral fractures after denosumab cessation, and potential cardiovascular risks with romosozumab lead many patients to decline or discontinue therapy.
Many women are unaware that they are suffering from bone loss until they have a DEXA scan performed. At that time, if osteopenia or osteoporosis is found, they are faced with the decision to accept one of the treatment options currently available to them. Many women decline treatment because of the risk of suffering one of the adverse events associated with the pharmaceutical treatment options available to them.
For patients with osteopenia or those unwilling or unable to start drug therapy, nutraceutical strategies may offer foundational support for bone health. Bone-Viva provides micronutrients shown to influence mineral utilization (vitamin D3, K2, boron, silicon, zinc), collagen cross-linking (copper, manganese), osteoblast regulation (silicon, curcumin, K2), osteoclast suppression (β-caryophyllene, curcumin, strontium), and inflammatory signaling (β-caryophyllene, curcumin). These biological effects align with the domains of bone remodeling described by Minkin, Blair, Zaidi, Raggatt, and others. As such, nutraceutical formulations may be particularly useful in early loss of bone mass or as adjuncts to antiresorptive or anabolic therapy. Emerging evidence supports that multi-nutrient support may enhance or stabilize BMD in patients receiving pharmacologic agents, consistent with findings observed in the combination groups of this analysis.
In this retrospective analysis of 51 female patients, we evaluated the use of a nutraceutical, Bone-Viva, which is comprised of generally recognized as safe (GRAS) approved ingredients. Each of the ingredients was chosen for its emerging role in bone metabolism working in such ways as promoting bone formation, helping to absorb calcium, helping to create collagen, and helping to increase density of bone.
Two current therapeutic treatment options for treating osteopenia and osteoporosis were used in this evaluation including a RANKL inhibitor, denosumab, and a parathyroid hormone analogue, romosozumab. In our analysis, we compared four treatment groups:
- Bone-Viva monotherapy
- denosumab monotherapy
- romosozumab monotherapy
- combination therapy using Bone-Viva and either denosumab or romosozumab
The four treatment groups were evaluated for bone loss using a non-invasive dual-energy X-ray absorptiometry (DEXA) machine manufactured by Hologic. The spine, femoral neck and total hip were tested. A T-score analysis, which measures bone mineral density (BMD) compared to that of a healthy young adult of the same sex, was performed at Baseline and again after 1 year of treatment. The T-scores at the two timepoints were compared and the change in the T-score was noted as improved, stabilized or declined using a +0.1 threshold for change in T-score.
In the lumbar spine, the strongest gains were observed in two therapy groups, denosumab and combination therapy groups (Bone-Viva+ either denosumab or romosozumab). Sixty to sixty five percent (60–65%) of patients demonstrated measurable improvement. Whereas the romosozumab group showed a mean increase of +0.12 with improvement in 48% of patients. The Bone-Viva monotherapy group achieved a mean T-score gain of +0.18 in 42% of patients.
Changes in the femoral neck were modest and variable across all treatment groups. Mean T-score shifts ranged from small gains with denosumab (+0.05) and romosozumab (+0.04) to slight declines with Bone-Viva (–0.02) and combination Therapy (–0.01). Bone-Viva monotherapy demonstrated the highest stability rate at 60%. The combination therapy group showed mean score gains exceeding those achieved in the monotherapy group. It is believed that the synergistic interaction between Bone-Viva and the combination group appears to have enhanced the expected improvement in bone density scores.
In the total hip, all groups produced small but consistent T-score gains. The combination Therapy (+0.17), followed by denosumab (+0.015), romosozumab (+0.10) and Bone-Viva (+0.01). Improvement proportions were 53–55%, with most remaining stable. Bone-Viva achieved stabilization in 45–48% of cases, suggesting preservation rather than augmentation of cortical bone.
These preliminary findings suggest that Bone-Viva provides a well-tolerated approach to bone health. There is presumptive evidence that the ingredients in Bone-Viva aid in the stabilization of bone loss. There was only one minor event reported in this analysis of stomach distress when taking the Bone-Viva capsules on an empty stomach. Bone-Viva offers a safe, effective and economical alternative for these individuals.
Finally, it is suggestive that the largest group of individuals who would benefit from the use of Bone-Viva is the osteopenic group. Their DEXA results may indicate that they have early bone loss, but not to the level which requires pharmaceutical intervention. By using Bone-Viva as monotherapy, they may be able to stabilize or improve their bone loss economically without the fear of any serious side effects. This will lead to better compliance and better outcomes in the treatment approach to reducing fracture risk very early in the bone loss stage.
Conclusion:
Preliminary findings suggest that Bone Viva’s nutraceutical composition provides a biologically plausible and well-tolerated approach to bone health optimization. Bone-Viva appears to promote stabilization and modest improvement of BMD, particularly among patients with osteopenia, even in the context of varied background therapy. Bone-Viva may further enhance bone strength and reduce fracture risk when used in combination with approved pharmaceutical therapies. This pilot study suggests that for women in early post-menopausal and perimenopausal stages, Bone-Viva may be a safe and natural nutraceutical to preserve bone health. The majority of patients maintained stability of bone density across all skeletal sites and a subset achieved meaningful gains at the lumbar spine and hip. Bone-Viva was well tolerated with minor dyspepsia in some patients.
A larger sample size and further follow up beyond 1 year will allow for more definitive conclusions. While the percentages changes may appear small the results are for only a 12-month period. More significant change in bone density is to be expected over 2 years or longer. Furthermore, more research is needed to see the bone response to Bone-Viva and other approved medications in naïve patients with modest bone loss, as opposed to the effect on patients who received multiple treatments cyclically or have advanced bone loss before treatment is initiated. Larger prospective studies to examine these questions would help in the future management of this catastrophic prevalence of bone loss. It is important, both medically and economically, to determine if there could be a reduction in morbidity and mortality that is currently such a significant medical issue for the world.
References
- Wright NC, et al. J Bone Miner Res. 2014;29(11):2520-2526.
- Demontiero O, et al. Ther Adv Musculoskelet Dis. 2012;4(2):61-76.
- Pouresmaeili F, et al. Ther Clin Risk Manag. 2018;14:2029-2049.
- Gou J, et al. A nomogram for one-year risk of death after hip fracture. Front Med. 2025; 12:1500049.
- Tu KN et al. Pharm Ther. 2018;43(2):92-104.
- Kennel KA, Drake MT. Mayo Clin Proc. 2009;84(7):632-638.
- Yamaguchi M, Levy RM. Exp Ther Med. 2016;12(6):3602-3606.
- Rondanelli M, et al. J Trace Elem Med Biol. 2020;59:126577.
- Jugdaohsingh R, et al. J Bone Miner Res. 2004;19(2):297-307.
- Saltman PD, Strause LG. J Am Coll Nutr. 1993;12(4):384-389.
- Holick MF. N Engl J Med. 2007;357(3):266-281.
- Koshihara Y, et al. J Endocrinol. 2003;176(3):339-348.
- Brennan TC, et al. Br J Pharmacol. 2009;157(7):1291-1300.
- Akdas S, Ceylan MN, Yazici F, et al. Is zinc an important trace element on bone-related diseases and complications? A meta-analysis. Biol Trace Elem Res. 2021;199:3808-3821.
- Baldy V, Ormeno E, Ballini C, Fernandez C. Production and diversity of volatile terpenes from plants on calcareous versus siliceous soils. J Chem Ecol. 2008;34:1219-1229.
- Bartolini A, Ghelardini C, Galeotti N, Mannelli LCD, Mazzanti G. Local anesthetic activity of β-caryophyllene. Farmaco. 2001;56:387-389.
- Blake GM, Fogelman I. Strontium ranelate: a novel treatment for postmenopausal osteoporosis. Clin Interv Aging. 2006;1(4):367-376.
- Blair HC, Zaidi M, Moonga BS, Abe E, Huang CL. Osteoclastogenesis, bone resorption, and osteoblast-based therapeutics. J Bone Miner Res. 2003;18:599-609.
- Chen M, Wu L, Jiang H, et al. Micronutrient intake and osteoporosis in hypertensive elderly. Front Nutr. 2024;11:1357439.
- Chambers TJ, Fuller K. How are osteoclasts induced to resorb bone? Ann N Y Acad Sci. 2011;1240:1-7.
- Chang J, Wang Z, Tang E, et al. Inhibition of osteoblastic bone formation by nuclear factor-κB. Nat Med. 2009;15:682-689.
- Charkos TG, Kontogiorgis CA, Trikka FA, et al. Dietary β-carotene intake and fracture risk: a meta-analysis. Nutrients. 2020;12:2193.
- Charkos TG, Skaperda Z, Tsekouras GE, et al. Carotenoid intake and osteoporosis risk: a propensity-matched analysis. J Health Popul Nutr. 2025;44:xx-xx.
- Chawla M, Yadav P, Shinde DB. Analgesic and anti-inflammatory activity of caryophyllene oxide. Indian J Pharmacol. 2014;46:475-481.
- Cong B, Xu Y, Chen Y, et al. Combined calcium and vitamin D improves bone mineral density in postmenopausal osteoporosis. J Bone Miner Res. 2025;40:xx-xx.
- Cortez M, Carmo LS, Rogero MM, Borelli P, Fock RA. High-fat diet increases IL-1, IL-6, TNF-α via NF-κB in bone marrow stromal cells. Inflammation. 2016;39:370-383.
- Cui A, Zhao J, Wang H, et al. Dietary copper intake and bone mineral density in US adults. PLoS One. 2024;19:e0296228.
- Fan D, Liu L, et al. Curcumin prevents diabetic osteoporosis via osteogenic and angiogenic pathways. J Diabetes Res. 2022;2022:9476132.
- Fung EB, Ritchie J, Kaplan P, et al. Zinc supplementation improves bone density in thalassemia. Am J Clin Nutr. 2013;98:960-971.
- Gao SS, Wang J, Qin L, et al. Effects of β-carotene on osteoporosis: a systematic review and meta-analysis. Menopause. 2023;30:xxx-xxx.
- Gertsch J, Leonti M, Raduner S, et al. Beta-caryophyllene is a dietary cannabinoid. Proc Natl Acad Sci U S A. 2008;105:9099-9104.
- Gharibi B, Abraham AA, Ham J, Evans BA. Adenosine receptor activation influences mesenchymal stem cell osteogenesis. J Bone Miner Res. 2011;26:2112-2124.
- Ghelardini C, Galeotti N, Mannelli LCD, Mazzanti G, Bartolini A. Local anesthetic activity of β-caryophyllene. Farmaco. 2001;56:387-389.
- Gou J, Shi K, Dai R, Wang J, Li Y. A nomogram for one-year risk of death after hip fracture. Front Med. 2025;12:1500049.
- Hu L, Chen X, et al. Combined vitamin K and calcium on bone mineral density. J Orthop Surg Res. 2021;16:592.
- Hu M, Zhang X, et al. Dietary copper and bone mineral density. Medicine (Baltimore). 2025;104:e43777.
- Iwamoto I, Takeda T, et al. Longitudinal effects of vitamin K2 on bone mineral density. Maturitas. 1999;31:161-168.
- Katsuyama M, Mizoguchi H, Kuwahata H, et al. CB2 involvement in β-caryophyllene–induced antinociception. Eur J Pain. 2013;17:664-675.
- Kulczynski B, Gramza-Michalowska A. Carotenoids and bone health. Nutr Res. 2024;xx:xx-xx.
- Leslie WD, Rubin MR, Schwartz AV, Kanis JA. Type 2 diabetes and bone. J Bone Miner Res. 2012;27:2231-2237.
- Li Y, Li A, Strait K, et al. Endogenous TNF-α reduces bone mass via NF-κB inhibition. J Bone Miner Res. 2007;22:646-655.
- Li Z, Kawashima T, Kato K, et al. Silicon absorption from mineral water and effects on bone metabolism. Nutr J. 2010;9:44.
- Liu C, Chen L, et al. Calcium and vitamin D supplementation in postmenopausal osteoporosis. Food Funct. 2020;11:1920-1930.
- Macdonald HM, Hardcastle AC, Jugdaohsingh R, et al. Silicon–estrogen interactions in bone health. J Nutr Health Aging. 2012;16:128-134.
- Ma ML, Shen Q, et al. Vitamin K2 in osteoporosis: a meta-analysis. Front Public Health. 2022;10:895067.




