Effects of Tirzepatide on Lipoprotein Lipase in Diabetes
A Case Series of Tirzepatide Treatment in Patients with Type 2 Diabetes, Chylomicronemia and Genetic Variants Affecting Lipoprotein Lipase
Stephan Babirak, MD1 and Mark Henschke Pharm D, DO2
- Metabolic Leader, LLC, PA Scarborough, Maine, USA
OPEN ACCESS
PUBLISHED: 28 February 2025
CITATION: Babirak, S. and Henschke, M., 2025. A Case Series of Tirzepatide Treatment in Patients with Type 2 Diabetes, Chylomicronemia and Genetic Variants Affecting Lipoprotein Lipase. Medical Research Archives, [online] 13(2).
https://doi.org/10.18103/mra.v13i2.6229
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i2.6229
ISSN 2375-1924
ABSTRACT
Background/Aim: Tirzepatide has been shown to improve glycemic control, improve insulin sensitivity, increase adipocyte lipoprotein lipase (LPL) activity and decrease apolipoprotein C3 levels. We previously reported that a patient who had type 2 diabetes (T2DM), persistent chylomicronemia and heterozygosity for LPL deficiency had resolution of his chronic chylomicronemia and near normalization of serum triglycerides in response to tirzepatide therapy. Therefore, the aim of this study was to perform a case series using tirzepatide treatment in a group of patients with T2DM or metabolic syndrome, chylomicronemia and multiple genetic variants affecting LPL to verify similar results in a larger group of patients with Metabolic Chylomicronemia Syndrome.
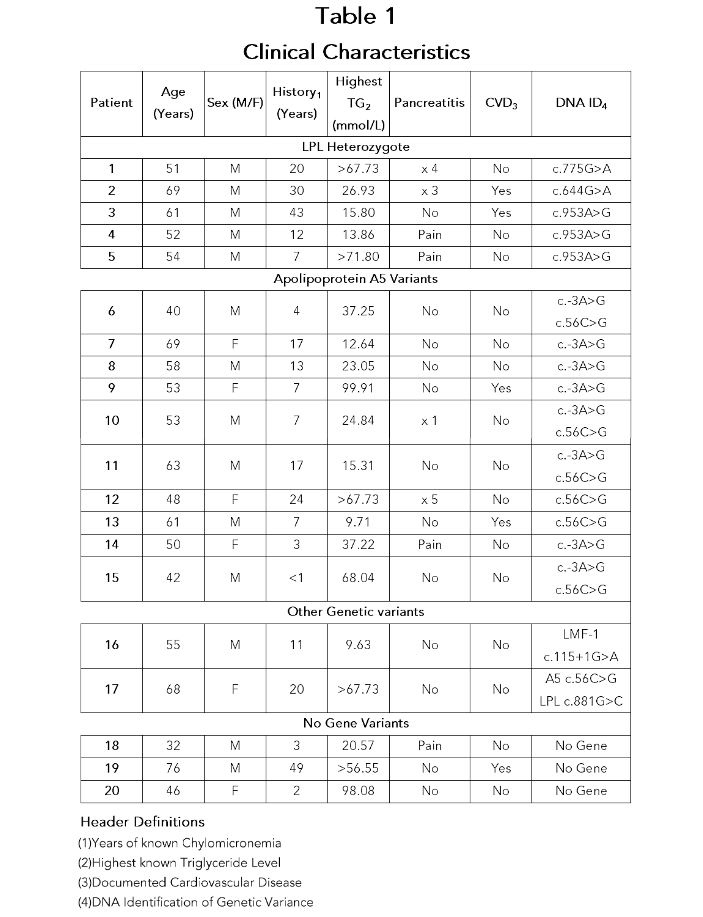
Method: 61 patients with chylomicronemia who were referred to our lipid clinic were screened. Fiftyeight of 61 patients had identifiable genetic variations in one of the 7 genes associated with chylomicronemia and 57/61 had T2DM. Twenty of these chylomicronemic patients (19 with T2DM and 1 with prediabetes) were able to be treated with tirzepatide and 17/20 had a genetic variant associated with a decrease in LPL: 5 having heterozygosity for LPL deficiency, 10 having variants of apolipoprotein A5, 1 with a variant of the LMF-1 gene and one patient with prediabetes who was a compound heterozygote having LPL heterozygosity plus an apolipoprotein A5 variant. Three patients had no identifiable genetic variation associated with chylomicronemia and all were treated with tirzepatide given to maximum tolerated dose.
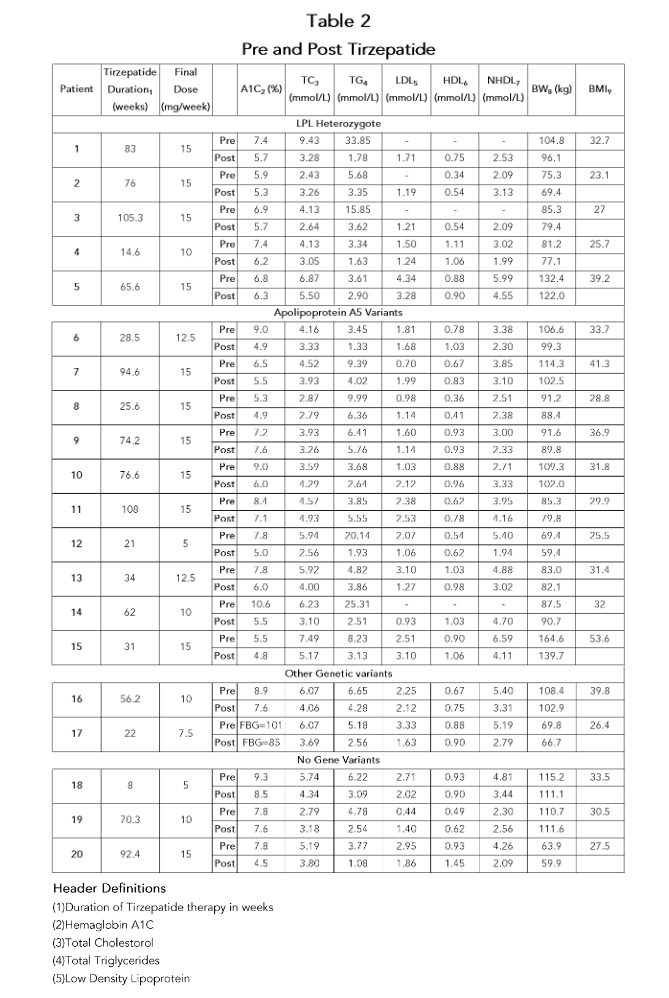
Results: In all patients, there was a resolution of their chylomicronemia. In patients who had a history of chylomicronemia induced pancreatitis or abdominal pain, there were no episodes of pancreatitis or abdominal pain while on tirzepatide. Pre-/post lipid profile averages as follows: total triglyceride decreased from 9.25 to 3.19 mmol/L (-65%, P value < 0.006), total cholesterol decreased from 5.12 to 3.72 mmol/L (-27%, P < 0.0013), LDL cholesterol decreased from 2.14 to 1.72 mmol/L (-20%, P < 0.40), non-HDL cholesterol decreased from 4.09 to 2.97 mmol/L (-27%, P < 0.002), HDL cholesterol increased from 0.74 to 0.84 mmol/L (+13%, P < 0.023). In addition, the average A1c decreased from 7.6% to 6.0% (-15%, P < 0.0001) and body weight decreased from 98.0 to 91.8 kg (-6.5%, P < 0.0001).
Conclusion: In this case series of patients with a history of chylomicronemia, T2DM/metabolic syndrome and multiple gene variants affecting LPL, tirzepatide caused a resolution of chylomicronemia with a significant decrease in total triglyceride, total cholesterol and non-HDL cholesterol levels as well as an increase of HDL cholesterol. Tirzepatide resulted in a significant improvement of A1C as well as a decrease in body weight. The mechanism(s) underlying these effects is(are) not completely understood but warrants further investigation. The results of this case series suggest that tirzepatide may be an agent useful for the treatment of patients with T2DM and chylomicronemia who have genetic variants affecting LPL.
Introduction
Chylomicronemia syndrome was first described in 1981 as a clinical syndrome associated with severely elevated chylomicron and very low density lipoprotein (VLDL) triglyceride with total triglyceride levels > 10 mmol/L or 885 mg/dl, caused by a deficiency in lipoprotein lipase (LPL) activity.¹³ LPL is the rate limiting enzyme responsible for the clearance of triglyceride from the circulation and LPL deficiency can be monogenic or polygenic. The clinical syndrome is characterized by recurrent abdominal pain, pancreatitis, eruptive xanthomas, lipemia retinalis, hepatosplenomegaly and neurologic disorders. Acute pancreatitis caused by chylomicronemia is the most serious complication which can be fatal. There are 2 major syndromes associated w the Chylomicronemia Syndrome.
Familial chylomicronemia syndrome (FCS) is a rare genetic disease affecting approximately 1/1,000,000 people and caused by inactivating mutations in both alleles of the LPL gene or by other variants in genes including proteins that are required for LPL activity (see below)¹³. Patients who are heterozygotes for LPL deficiency (HeLPL) have an approximate 50% decrease in LPL activity/mass¹⁴ and may also develop chylomicronemia or cardiovascular disease but usually in polygenic fashion or associated with secondary factors¹³,¹⁵,¹⁶. Other variants in genes encoding proteins that are required for LPL activity such as apolipoprotein C2 or A5 (ApoC2 and ApoA5), glycosylphosphatidylinositol anchored high density lipoprotein binding protein (GPIHBP1), lipase maturation factor 1 (LMF-1) as well as other variants have also been described which may or may not be associated with chylomicronemia¹⁶. An inhibitor of LPL activity, apolipoprotein C3 (ApoC3), is another key regulatory protein which can decrease LPL activity directly or indirectly by lipoprotein triglyceride removal unrelated to LPL (17).
Metabolic Chylomicronemia Syndrome (MCS) is a far more common cause of chylomicronemia with an estimated frequency of 1.4% in the United States population which is associated with multiple genetic variants affecting LPL and/or other secondary factors which can reduce LPL activity such as Diabetes Mellitus, hypothyroidism and drugs which can cause hyperlipidemia¹². In approximately 70% of MCS patients, chylomicronemia is associated with hyperglycemia in uncontrolled Type 2 diabetes (T2DM) and genetic variants, drugs or other factors affecting lipoprotein metabolism¹⁸. Uncontrolled hyperglycemia and/or insulin resistance contribute to an increase of VLDL formation in the liver due to the elevated glucose and free fatty acids which are substrate for triglyceride formation. Since insulin is required for LPL synthesis, the LPL activity is reduced in T2DM with insulin deficiency or patients with metabolic syndrome, leading to a decrease in removal of triglyceride rich lipoproteins. In patients with T2DM and/or metabolic syndrome, mild to moderate hypertriglyceridemia is common and may worsen to develop chylomicronemia if coexistent genetic variants of LPL are present in the same patient which may progress to the clinical sequela of chylomicronemia as well as cardiovascular disease. For the T2DM patients who have persistent chylomicronemia and/or chylomicronemia induced pancreatitis, there has not been a very effective treatment to prevent their severe hypertriglyceridemia despite the use of multiple drug regimens³, however there are new agents on the horizon as recently reported in apolipoprotein C3 inhibition¹⁸,²⁰. We previously reported that a patient who had T2DM, persistent chylomicronemia and heterozygosity for LPL deficiency had resolution of his chronic chylomicronemia and near normalization of triglyceride levels in response to tirzepatide therapy²¹. Since tirzepatide has been shown to improve glycemic control as well as improve insulin resistance, decrease free fatty acids, increase adipocyte LPL activity and decrease apolipoprotein C3 levels²², we studied a larger group of patients with T2DM or metabolic syndrome who presented to our lipid clinic with chylomicronemia to verify similar results.
Methods
61 patients with chylomicronemia were referred to Metabolic Leader lipid clinic for evaluation/
References
1) Chait A, Robertson HT, Brunzell JD. Chylomicronemia syndrome in diabetes mellitus. Diabetes Care. 1981; 4:343-8.
2) Brunzell JD, Auwerx JH, Babirak SP, Fujimoto WY, and Hayden MR. Familial Lipoprotein Lipase Deficiency. In: Arteriosclerosis VIII, (Crepaldi G, Gotto AM, and Manzato E eds.), New York, Excerpta Medica; 165-167, 1989.
3) Chait A and Eckel RH. The chylomicronemia syndrome is most often multifactorial. Ann InterMed. 2019;170: 626-634.
4) Brunzell J, Deeb S. Familial lipoprotein lipase deficiency, apo CII deficiency, and hepatic lipase deficiency. In: Scriver C, Beaudet A, Sly W, Vale D, eds. The Metabolic and Molecular Basis of Inherited Disease. 8th ed. New York: McGraw-Hill; 2001:2789-816.
5) Hegele RA, Ginsberg HN, Chapman MJ et al. The polygenetic nature of hypertriglyceridemia: implications for the definition, diagnosis and management. Lancet Diab and Endo. 2014; 2(8): 655-666.
6) Surendran RP, Visser ME, Heemelaar S, Wang J, et al. Mutations in LPL, APOC2, APOA5, GPIHBP1 and LMF1 in patients with severe hypertriglyceridemia. J Intern Med. 2012; 272:185-96.
7) Fojo SS, Brewer HB. Hypertriglyceridemia due to genetic defects in lipoprotein lipase and apolipoprotein C-II. J Intern Med. 1992; 231:669-677.
8) Calandra S, Priore Oliva C, Tarugi P, Bertolini S. APOA5 and triglyceride metabolism, lesson from human APOA5 deficiency. Curr Opin Lipidol. 2006; 17:122-7.
9) Rios JJ, Shastry S, Jasso J, Hauser N, Garg A, Bensadoun A, et al. Deletion of GPIHBP1 causing severe chylomicronemia. J Inherit Metab Dis. 2012; 35:531-40.
10) Hegele RA, Berberich AJ, Ban MR, et al. Clinical and biochemical features of different molecular etiologies of familial chylomicronemia. J Clin Lipidol 2018; 12: 920–927.
11) Haller JF, Mintah IJ, Shihanian LM, Stevis P, Buckler D, AlexaBraun CA, et al. ANGPTL8 requires ANGPTL3 to inhibit lipoprotein lipase and plasma triglyceride clearance. J Lipid Res. 2017; 58:1166- 1173.
12) Chi X, Britt EC, Shows HW, Hjelmaas AJ, Shetty SK, Cushing EM, et al. ANGPTL8 promotes the ability of ANGPTL3 to bind and inhibit lipoprotein lipase. Mol Metab. 2017; 6:1137-1149.
13) Hegele RA, Ginsberg HN, Chapman MJ, Nordestgaard BG, Kuivenhoven JA, Averna M, et al; European Atherosclerosis Society Consensus Panel. The polygenic nature of hypertriglyceridemia: implications for definition, diagnosis, and management. Lancet Diabetes Endocrinol. 2014; 2:655-66.
14) Babirak SP. The detection and characterization of the heterozygote state for LPL deficiency. Arteriosclerosis 1989: 9(3): 316-334.
15) Babirak SP, Brown BG, and Brunzell JD. Premature coronary disease, elevated apolipoprotein B, and low lipoprotein lipase. Arteriosclerosis 8: 575a, 1988.
16) Chait A, Brunzell JD. Severe hypertriglyceridemia: role of familial and acquired disorders. Metabolism. 1983; 32:209-214.
17) Gaudet D, Brisson D, Tremblay K, et al. Targeting APOC3 in the familial chylomicronemia syndrome. N Engl J Med 2014; 371: 2200–06.
18) Witztum JL, Gaudet D, Freedman SD, Alexander VJ, Digenio A, Williams KR, Yang Q, Hughes SG, Geary RS, Arca M, Stroes ESG, Bereron J, Soran H, Civeira H, Hemphill L, Tsimikas S, Blom DJ,O’Dea and Bruckert E. Volanesorsen and triglyceride levels in Familial chylomicronemia syndrome. N Engl J Med 2019; 381:531-542.
19) Stroes ESG, Alexander VJ and Karwatowska-Prokopczuk E. Olezarsen, acute pancreatitis, and familial pancreatitis syndrome. N Engl J Med 2024; 390:1781-92.
20) Watts GF, Rosenson RS, Hegele RA, et al. Plozasiran for Managing persistent chylomicronemia and pancreatitis risk. N Engl J Med Sept 2 2024, doiEpub ahead of print, PMID; 39225259.
21) Babirak SP. Tirzepatide therapy in a patient with type 2 diabetes mellitus, chylomicronemia and heterozygosity for lipoprotein lipase deficiency. AACE Clinical Case Report 2023; 9;128-130.
22) Wilson JM, Nikooienejad A, Robins DA, Roell WC, et al. The dual glucose-dependent insulinotropic peptide and glucagon-likepeptide-1 receptor agonist, tirzepatide, improves lipoprotein biomarkers associated with insulin resistance and cardiovascular risk in patients with type 2 diabetes. Diabetes Obes Metabolism.2020:22(12): 2451-2459.
23) Pirro V, Roth KD, Lin Y et al. effects of tirzepatide, a dual GIP and GLP–1 receptor agonist, on lipid and metabolite profiles in subjects with type 2 diabetes. J Clin Endocrinol Metab. 2022; 7 (2): 363–378.
24) Kim SJ. Activation of lipoprotein lipase by glucose-dependant insulinotropic polypeptide in adipocytes. J Biol Chem. 2007; 282:8557-8567.
25) Kim SJ, Nian C, McIntosh CH.GIP increases human adipocyte LPL expression through CREB and TORC2-mediated trans-activation of the LPL gene. J Lipid Res. 2010; 51(11):3145-57.