Efficacy of ACTH Test for Aldosterone-Producing Adenoma
Verification of the screening criteria of the ACTH-stimulating test for predicting aldosterone producing adenoma in primary aldosteronism
Toshihiro Kita1
- Department of Projects Research, Frontier Science Research Center, University of Miyazaki, Miyazaki, Japan.
OPEN ACCESS
PUBLISHED: 30 June 2025
CITATION: Kita, T., 2025. Verification of the screening criteria of the ACTH-stimulating test for predicting aldosterone producing adenoma in primary aldosteronism. Medical Research Archives, [online] 13(6). https://doi.org/10.18103/mra.v13i6.6641
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i6.6641
ISSN 2375-1924
Abstract
Adrenal venous sampling (AVS) is essential for the subtype diagnosis of primary aldosteronism (PA); however, it requires a respectable technique and effort. To avoid unnecessary AVS for bilateral PA, we used the adrenocorticotropic hormone-stimulating test (AST) to narrow the unilateral PA before AVS. Here, the efficacy of the criteria was verified in two sets of patients with PA, namely, the nonselected and selected groups. In the nonselected group, patients referred for PA evaluation were mostly admitted to our hospital and underwent AVS if they had positive reactions for at least one of the AST, captopril challenge test, and furosemide upright test. In the selected group, the referred patients underwent AST in the outpatient office, and those who met our criteria were preferentially subjected to AVS. Additionally, some applicants underwent AVS. Of the 43 nonselected patients, only nine (20.9%), 32 (74.4%), and two (4.7%) were diagnosed with unilateral PA, bilateral PA, and considered normal, respectively. Three of the nine unilateral PAs and six of the 32 bilateral PAs did not meet our criteria. In the selected group, 34 patients were screened for AST levels using the new aldosterone measurement method, chemiluminescent enzyme immunoassay, and 14 patients were subjected to AVS. Following AVS, eight of nine patients (88.9%) who met our AST criteria were diagnosed with unilateral PA; conversely, all five patients who did not meet the criteria were diagnosed with bilateral PA (predictive accuracy 92.9%). In conclusion, initial screening with AST appears efficient even in a practical setting for refining unilateral AP before AVS and reducing significant number of patients with bilateral PA from unnecessary AVS.
Keywords
primary aldosteronism, adrenocorticotropic hormone-stimulating test, aldosterone producing adenoma, adrenal venous sampling
Introduction
Primary aldosteronism (PA) is a leading cause of endocrine hypertension. Its prevalence varies from 3.3% to > 20% in patients with hypertension and seems to depend on the criteria of PA. Notably, permissive criteria such as “positive oral sodium suppression test with post-test urine aldosterone ≥ 12 µg/day” resulted in a prevalence of 11.3% even in normotensive participants. Therefore, PA is considered a common disease, and appropriate management is required. Furthermore, guidelines highlight the high prevalence and stronger impact of PA on the cardiovascular and renal systems compared to essential hypertension, emphasizing the necessity of appropriate and early detection of PA. Simultaneous measurements of plasma aldosterone concentration (PAC), plasma renin activity (PRA), and the derived PAC/PRA ratio (ARR) are recommended as the initial screening tests for patients with suspected PA. Awareness of ARR is widely disseminated in practice, and many patients gain awareness at specialized medical institutions. Following confirmatory tests, eligible patients undergo adrenal venous sampling (AVS) to determine the PA subtype, either unilateral PA or bilateral PA. Bilateral PA, mainly caused by idiopathic adrenal hyperplasia, is managed with drug therapy, whereas only unilateral PA, mainly caused by aldosterone producing adenoma (APA), requires surgery. Importantly, AVS demands considerable skill and effort, limiting its availability to only a select number of patients. Additionally, it is more beneficial for unilateral PA than for bilateral PA; thus, advanced refinement of unilateral PA before AVS is very valuable for both patients and clinicians. In a previous study, we proposed diagnostic criteria using the adrenocorticotropic hormone-stimulating test (AST) to identify unilateral PA before AVS. To verify the efficacy of these criteria (PAC/cortisol ratio at 120 min on AST > 1.2), two groups of patients were evaluated. In the first group, patients referred for PA evaluation were mostly admitted to our hospital and underwent AVS if they had positive reactions to one of the three loading tests: AST, captopril challenge test (CCT), and furosemide upright test (FUT). The natural prevalence of unilateral PA and bilateral PA was clarified in this nonselected group. In the second group, the referred patients underwent AST in the outpatient office, and patients who met our criteria were preferentially subjected to AVS. The efficacy of our criteria was verified in the selected group. Since April 2021, radioimmunoassay (RIA) kits for PAC have no longer been available in Japan and have been replaced entirely by chemiluminescent enzyme immunoassay (CLEIA) by the end of June 2021. This alteration had a significant influence on the criteria of screening and confirmatory tests for PA in Japan. At our institute, considerable confusion occurred, and subsequently, a new approach system was established after April 2022. Therefore, some patients from this period of confusion were excluded from this analysis.
Methods
Study patients
This study included all eligible patients diagnosed with PA who underwent AVS at our hospital between April 2018 and January 2025. In the first period, from April 2018 to June 2021, most referred patients were admitted to our hospital and underwent confirmatory tests, including CCT, FUT, and AST. All patients showing positive reactions in at least one of the AST, CCT, or FUT underwent AVS (nonselected group). In the later period (April 2022 to January 2025), all referred patients underwent AST in our outpatient office, and patients who met our criteria (PAC/cortisol ratio at 120 min >1.2 in AST) and required further evaluation were admitted to our hospital for confirmatory tests and AVS. This study was approved by the ethics committee of the University of Miyazaki, Japan.
Diagnostic procedure for primary aldosteronism
In the nonselected group, patients with an ARR > 20 in PAC (ng/dL) were measured using RIA and considered positive in the screening test. All patients who intended to undergo surgery, if needed, were admitted to our hospital and fed 10 g of sodium chloride per day. Antihypertensive drugs were limited to calcium channel blockers and/or alpha-blockers before the examination. Basal blood sampling for confirmatory tests was performed between 8:00 and 8:30 h after overnight fasting. The AVS was conducted under these conditions.
In the selected group, patients with ARR > 200 or ARR > 100 and PAC > 60 (pg/mL), measured by CLEIA, were considered positive on screening tests. All patients with positive screening test results underwent the AST at our outpatient office. The AST was performed by an expert nurse, and ACTH was administered by the author. Overnight fasting and sodium control were not applied, and the AST was performed in the forenoon. Antihypertensive drugs, except anti-aldosterone antagonists, were permitted during the AST. After hospitalization, the selected group underwent the same conditioning as the nonselected group, followed by confirmatory tests and AVS.
Plasma hormone concentrations were measured using a commercially available laboratory testing service (BML, Tokyo, Japan). In this study, the conversion formula from CLEIA (X) (pg/mL) to RIA (Y) (ng/dL), Y = (1.174X + 42.3)/10 (based on Ref. 9), was used to maintain continuity of the AST criteria. Additionally, new criteria for raw PAC measurements were evaluated. The CCR, FUT, and AST procedures have been described previously. The positive criteria for CCT were PAC/PRA >20 at 60 min in RIA and PAC/PRA >200 at 60 min, or PAC/PRA >100 and PAC >60 pg/mL at 60 min in CLEIA. The positive criterion for UFT was PRA <2.0 at 120 min, while that for AST was PAC/cortisol ratio > 0.85 at the time of maximum PAC value after ACTH injection.
The procedure for AVS has also been described in a previous study. Briefly, AVS was performed by expert radiologists using assistant computed tomography imaging to confirm the correct localization of the microcatheter tip to the right adrenal vein. Following basal sampling from the bilateral adrenal veins, 250 μg of ACTH was administered by an intravenous injection. After 30 min, the second sampling step was performed. According to the JES guidelines, an adrenal venous cortisol concentration greater than 200 μg/ dL after the ACTH stimulation was considered as evidence of an adequate catheterization for the AVS. An adrenal venous aldosterone concentration higher than 1400 ng/dL after ACTH stimulation was considered evidence of aldosterone hypersecretion. In the case of CLEIA measurements for aldosterone, units of aldosterone were converted from pg/mL to ng/dL by dividing by 10. Lateralization of adrenal venous aldosterone hypersecretion is the primary criterion for diagnosing bilateral or unilateral PA subtypes.
Statistical analysis
All statistical analyses were performed using SPSS Statistics version 22 (IBM Japan, Tokyo, Japan). The significance of the differences between the two groups was evaluated using an unpaired t-test for parametric variables or the Mann-Whitney U test for nonparametric variables. Data are expressed as the means ± SD, and a value of P < 0.05 indicates statistical significance.
Results
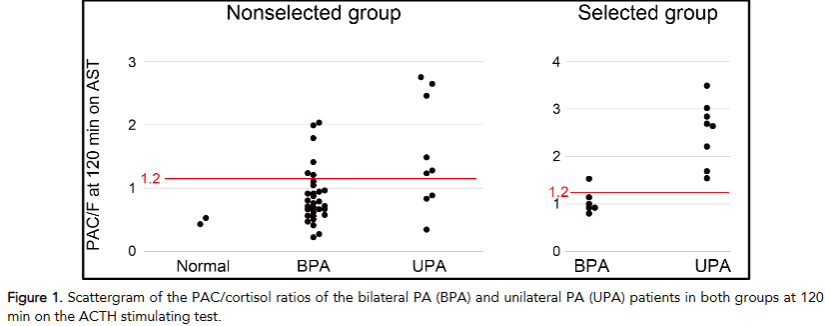
In the nonselected group, 49 patients were hospitalized, and five did not undergo AVS because of low reactivity on AST and negative reactions for CCT and FUT. One patient who did not achieve satisfactory AVS results was excluded. After AVS, two patients were judged as normal, and in the remaining 41 individuals, nine and 32 were diagnosed with unilateral PA and bilateral PA, respectively. In the selected group, 34 patients received AST; two did not meet diagnostic criteria for PA, while 10 and 22 individuals were over the 1.2 criteria on AST and under the 1.2 criteria on AST, respectively. One patient highly suspected of having unilateral PA rejected AVS for personal reasons and was excluded from this analysis. Nine and six patients over and under the 1.2 criteria who strongly wished to obtain a final diagnosis underwent AVS, respectively. After AVS, one patient meeting the 1.2 criteria was misclassified as bilateral PA, and the remaining 13 patients matched the predictive diagnosis by AST. The baseline characteristics of the patients are summarized in Table 1.
| Non-selected group | Selected group | ||||||
|---|---|---|---|---|---|---|---|
| UPA | BPA | UPA | BPA | ||||
| N | 9 | 32 | 8 | 6 | |||
| Male/Female | 4/5 | 11/21 | 0.65 | 3/5 | 1/5 | 0.57 | |
| Age (year old) | 55.8 ± 8.7 | 48.6 ± 13.7 | 0.15 | 46.3 ± 7.6 | 43.7 ± 12.6 | 0.64 | |
| Serum potassium (mmol/L) | 3.5 ± 0.4 | 4.0 ± 0.4 | 0.001 | 3.2 ± 0.6 | 3.8 ± 0.5 | 0.099 | |
| PAC (ng/dL) | 21.2 ± 8.9 | 14.3 ± 7.2 | 0.021 | ー | ー | ||
| PAC (pg/mL) | ー | ー | 247.5 ± 100.8 | 115.9 ± 64.5 | 0.017 | ||
| PRA (ng/mL/h) | 0.5 ± 0.4 | 0.6 ± 0.5 | 0.50 | 0.6 ± 0.6 | 0.7 ± 0.4 | 0.88 | |
| Adrenal tumor on CT | 5/9 (55.6%) | 2/32 (6.3%) | 0.008 | 8/8 (100%) | 0/6 (0%) | 0.008 | |
| PAC/PRA at 60min on CCT | 57.1 ± 51.3 | 25.5 ± 30.3 | 0.024 | 610 ± 626 | 176 ± 281 | 0.14 | |
| Positive rate on CCT | 7/9 (77.8%) | 14/32 (43.8%) | 0.13 | 4/8 (50.0%) | 1/6 (16.7%) | 0.35 | |
| Positive rate on CCT (>100) | ー | ー | 6/8 (75.0%) | 2/6 (33.3%) | 0.23 | ||
| PRA at 120min on FUT | 1.3 ± 1.2 | 2.6 ± 2.7 | 0.16 | 1.4 ± 1.0** | 1.0 ± 0.7 | 0.42 | |
| Positive rate on FUT | 8/9 (88.9%) | 18/32 (56.3%) | 0.15 | 4/7 (57.1%)** | 6/6 (100%) | 0.23 | |
| ACTH stimulating test | PAC (ng/dL)max/F | 2.2 ± 1.0 | 1.6 ± 0.7 | 0.044 | 3.3 ± 0.6 | 2.6 ± 0.7 | 0.087 |
| PAC (ng/dL)/F at 120min | 1.5 ± 0.9 | 0.9 ± 0.4 | 0.002 | 2.5 ± 0.7 | 1.1 ± 0.3 | <0.001 | |
| PAC (pg/mL)/F at 120min | ー | ー | 19.8 ± 5.8 | 7.5 ± 2.3 | <0.001 |
PAC: plasma aldosterone concentration, PRA: plasma renin activity, CCT: captopril challenge test, FUT: furosemide upright test, ACTH: adrenocorticotropic hormone, F: cortisol
Unilateral PA showed advanced hypokalemia and stronger reactions to CCT than it did to bilateral PA, but not to FUT. However, these differences were not observed in the selected group, probably due to the small number of patients. In the selected group, positive rates of CCT were low, potentially due to changes in the PAC measurement method. If the cutoff CCT was >100, positive ratios in the selected group were similar to those in the nonselected group.
Discussion
This study compared the acquisition rate of the unilateral PA in AVS between two groups of patients before and after the practical application of the AST criteria prior to AVS. Although this remains an exploratory attempt, advanced refinement of the unilateral PA before AVS using the AST appears to be useful in a practical setting. A significant reduction in non-unilateral PA patients (bilateral PA + normal) was achieved to obtain similar numbers of unilateral PA (nine and eight) in AVS; the 34 non-unilateral PA patients decreased to six patients (p = 0.015). This approach may improve work efficiency in PA diagnosis and reduce the burden on patients and medical staff.
Attempts to predict bilateral PA without AVS have been made using several approaches. In a meta-analysis evaluating the superiority of predictive tools, an algorithm combining several factors showed the highest specificity (95%) but low sensitivity (28%). This algorithm included combined parameters such as no adrenal nodule on CT, serum potassium > 3.8 mmol/L, ARR < 550 (PAC: pg/mL), and female sex. The characteristics of bilateral PA patients in the nonselected group appeared to match this algorithm, thus suggesting an unbiased patient population. The predictive ability of our AST criteria for bilateral PA was as follows: specificity, 66.7% and 100%; sensitivity, 81.3% and 83.3% in the nonselected and selected groups, respectively. These abilities were comparable to those reported by other groups using AST for subtype diagnosis of PA.
Notably, high specificity is desirable to avoid the misdiagnosis of unilateral PA as bilateral PA, which may lead to a lost opportunity for the operative treatment of APA. Moderate specificity in the nonselected group was caused by the relatively high responsiveness of cortisol after ACTH stimulation; as a result, the PAC/cortisol ratio at 120 min decreased with high cortisol levels. Although the possibility of complicated Cushing’s syndrome was ruled out by an overnight 1 mg dexamethasone suppression test, further measures may be needed. Conversely, the hit ratio of patients with a high PAC/cortisol ratio diagnosed with unilateral PA was satisfactorily high, as shown in the selected group. The well-organized AST in our outpatient office may have contributed to this consistent result. Our criteria maintained a relatively high sensitivity, but a considerable number of bilateral PA cases showed a high PAC/cortisol ratio in AST. On a trial basis, prolongation of AST was applied to 11 patients in the nonselected group, and blood sampling was continued every 30 min until 180 min after ACTH administration. Five patients with a PAC/cortisol ratio >1.2 at 120 min were included in this trial, and only patients with unilateral PA maintained this level until 150 min. This may provide a clue for improving the sensitivity of AST, but the burden of AST will increase.
| Subtype | 120 min | 150 min |
|---|---|---|
| APA | 2.75 | 2.55 |
| APA | 2.46 | 2.01 |
| APA | 1.28 | 1.34 |
| IHA | 1.21 | 1.15 |
| IHA | 1.41 | 0.95 |
APA: aldosterone producing adenoma, IHA: idiopathic hyperaldosteronism
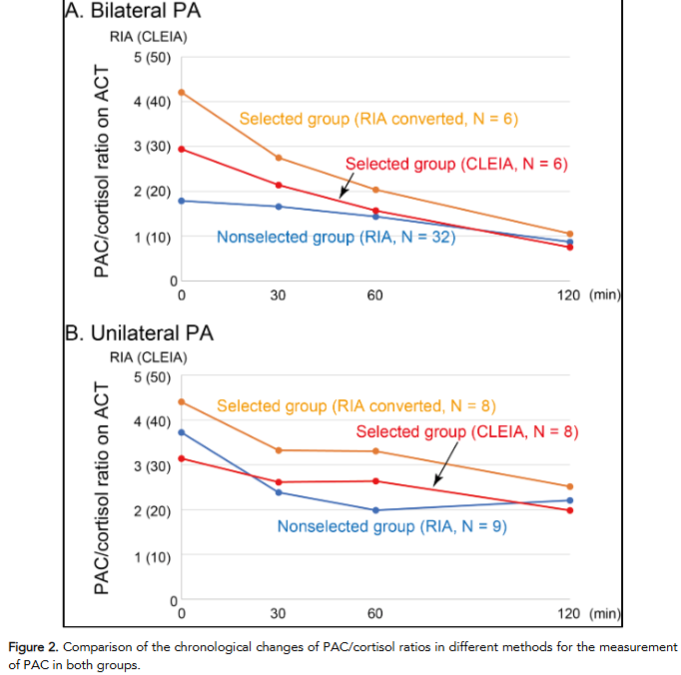
The mathematically derived raw PAC value in CLEIA corresponding to the PAC/cortisol ratio was 8.5. Indeed, among screened patients in the selected group (n = 34), those who had a PAC/cortisol ratio over 1.2 at 120 min using converted RIA values also exceeded the criterion of 10 in raw CLEIA values. The superiority of CLEIA for PAC measurement has been reported 9-11; thus, these alternative criteria for CLEIA will be useful in PA diagnosis.
Conclusion
The application of our criteria using the AST for advanced refinement of the unilateral PA before AVS worked well in a practical setting, and a significant number of patients with bilateral PA avoided unnecessary AVS. Aldosterone measurement using CLEIA seems to be superb for AST, and 10 seems to be the reliable threshold of the PAC/cortisol ratio at 120 min for the selection of unilateral PA. Additional evaluations with a larger number of patients may advance the utility of AST in PA diagnosis.
Conflict of Interest Statement:
None.
Funding Statement:
None.
Acknowledgements:
None.
References:
- Charoensri S, Turcu AF. Primary Aldosteronism Prevalence – An Unfolding Story. Exp Clin Endocrinol Diabetes. 2023; 131: 394-401. doi: 10.1055/a2066-2696.
- Brown JM, Siddiqui M, Calhoun DA, Carey RM, Hopkins PN, Williams GH, Vaidya A. The unrecognized prevalence of primary aldosteronism: A cross-sectional study. Ann Intern Med. 2020; 173: 10-20. doi: 10.7326/M20-0065.
- Milliez P, Girerd X, Plouin PF, Blacher J, Safar ME, Mourad JJ. Evidence for an increased rate of cardiovascular events in patients with primary aldosteronism. J Am Coll Cardiol. 2005; 45: 1243-1248. doi: 10.1016/j.jacc.2005.01.015.
- Born-Frontsberg E, Reincke M, Rump LC, Hahner S, Diederich S, Lorenz R, Allolio B, Seufert J, Schirpenbach C, Beuschlein F et al. Cardiovascular and cerebrovascular comorbidities of hypokalemic and normokalemic primary aldosteronism: Results of the German Conn’s Registry. J Clin Endocrinol Metab. 2009;94:1125-1130. doi: 10.1210/jc.2008-2116.
- Rossi GP, Bernini G, Desideri G, Fabris B, Ferri C, Giacchetti G, Letizia C, Maccario M, Mannelli M, Matterello MJ et al. Renal damage in primary aldosteronism: Results of the PAPY Study. Hypertension. 2006; 48: 232-238. doi: 10.1161/01.HYP.0000230444.01215.6a.
- Funder JW, Carey RM, Mantero F, Murad MH, Reincke M, Shibata H, Stowasser M, Young WF Jr. The management of primary aldosteronism: Case detection, diagnosis, and treatment: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2016;101:1889-1916. doi:10.1210/jc.2015-4061.
- Naruse M, Katabami T, Shibata H, Sone M, Takahashi K, Tanabe A, Izawa S, Ichijo T, Otsuki M, Omura M, et al. Japan Endocrine Society clinical practice guideline for the diagnosis and management of primary aldosteronism 2021. Endocr J. 2022; 69: 327-359. doi: 10.1507/endocrj.EJ21-0508.
- Kita T, Furukoji E, Sakae T, Kitamura K. Efficient screening of patients with aldosterone-producing adenoma using the ACTH stimulation test. Hypertens Res. 2019; 42: 801-806. doi: 10.1038/s41440-018-0191-5.
- Nishikawa T, Omura M, Kawaguchi M, Takatsu A, Satoh F, Ito S, Kurihara I, Itoh H, Yanase T, Shibata H et al. Calibration and evaluation of routine methods by serum certified reference material for aldosterone measurement in blood. Endocr J. 2016; 63: 1065-1080. doi: 10.1507/endocrj.EJ16-0304.
- Teruyama K, Naruse M, Tsuiki M, Kobayashi H. Novel chemiluminescent immunoassay to measure plasma aldosterone and plasma active renin concentrations for the diagnosis of primary aldosteronism. J Hum Hypertens. 2022; 36: 77-85. doi: 10.1038/s41371-020-00465-5.
- Ozeki Y, Tanimura Y, Nagai S, Nomura T, Kinoshita M, Shibuta K, Matsuda N, Miyamoto S, Yoshida Y, Okamoto M et al. Development of a New Chemiluminescent Enzyme Immunoassay Using a Two-Step Sandwich Method for Measuring Aldosterone Concentrations. Diagnostics (Basel). 2021; 11: 433. doi: 10.3390/diagnostics11030433.
- Onozawa S, Murata S, Tajima H, Yamaguchi H, Mine T, Ishizaki A, Sugihara H, Oikawa S, Kumita S. Evaluation of right adrenal vein cannulation by computed tomography angiography in 140 consecutive patients undergoing adrenal venous sampling. Eur J Endocrinol. 2014; 170: 601-608. doi: 10.1530/EJE-13-0741.
- Nishikawa T, Omura M, Satoh F, Shibata H, Takahashi K, Tamura N, Tanabe A, Task Force Committee on Primary Aldosteronism, The Japan Endocrine Society, The Japan Endocrine Society. Guidelines for the diagnosis and treatment of primary aldosteronism–The Japan Endocrine Society 2009. Endocr J. 2011; 58: 711-721. doi: 10.1507/endocrj.ej11-0133.
- Ng E, Gwini SM, Zheng W, Fuller PJ, Yang J. Predicting bilateral subtypes of primary aldosteronism without adrenal vein sampling: A systematic review and meta-analysis. J Clin Endocrinol Metab. 2024; 109: e837-e855. doi: 10.1210/clinem/dgad451.
- Jiang Y, Zhang C, Wang W, Su T, Zhou W, Jiang L, Zhu W, Xie J, Ning G. Diagnostic value of ACTH stimulation test in determining the subtypes of primary aldosteronism. J Clin Endocrinol Metab. 2015; 100: 1837-1844. doi: 10.1210/jc.2014-3551.
- Moriya A, Yamamoto M, Kobayashi S, Nagamine T, Takeichi-Hattori N, Nagao M, Harada T, Tanimura-Inagaki K, Onozawa S, Murata S et al. ACTH stimulation test and computed tomography are useful for differentiating the subtype of primary aldosteronism. Endocr J. 2017; 64: 65-73. doi: 10.1507/endocrj.EJ16-0297.




