Efficacy of Egg-Derived Peptides in Glycemic Control
A review of the efficacy of egg-derived bioactive peptides and hydrolysates on glycemic regulation
Yasaman Mojibi¹, Stepheny C. de Campos Zani², Caroline Richard¹, Catherine B. Chan¹,³
- Department of Agricultural Food and Nutritional Science, University of Alberta, Edmonton, Alberta
- Department of Pharmacy and Pharmaceutical Sciences, University of Alberta, Edmonton, Alberta
- Department of Physiology, University of Alberta, Edmonton, Alberta
OPEN ACCESS
PUBLISHED: 31 October 2025
CITATION: Mojibi, Y., et al., 2025. A review of the efficacy of egg-derived bioactive peptides and hydrolysates on glycemic regulation. Medical Research Archives, [online] 13(10). https://doi.org/10.18103/mra.v1 3i10.6973
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v1 3i10.6973
ISSN: 2375-1924
Abstract
Type 2 diabetes is a chronic metabolic disorder that may lead to serious health complications, including cardiovascular disease, kidney failure, and nerve damage. Furthermore, type 2 diabetes prevalence is projected to exceed 592 million people worldwide by 2035. Bioactive peptides are biological molecules that modulate physiological pathways, exhibiting antidiabetic, antioxidant, anti-inflammatory, antihypertensive, and immunomodulatory activities. Recently egg white has gained attention for developing bioactive peptides. It is hypothesized that consuming these peptides either individually or as part of unpurified or minimally purified hydrolysates may benefit individuals with type 2 diabetes when incorporated into the diet. Although human trials investigating the effects of egg white hydrolysate remain limited, several in vitro and rodent model studies have demonstrated beneficial effects of egg white hydrolysate and other egg-derived bioactive peptides on glucose homeostasis and glycemic control. However, the underlying mechanisms by which egg-derived hydrolysates and peptides affect glucose regulation appear to be diverse. This review summarizes evidence from preclinical and clinical studies investigating the glucoregulatory effects of egg white hydrolysate and egg-derived bioactive peptides in obesity and/or type 2 diabetes. Findings suggest potential benefits through mechanisms such as modulation of body weight, regulation of glucose absorption and uptake, and enhancement of insulin secretion and signaling. Nonetheless, further robust animal studies and clinical trials are needed to enhance our knowledge in this field and advance the use of egg peptides in the management of type 2 diabetes.
Keywords
egg-derived peptides, glycemic regulation, type 2 diabetes, bioactive peptides, insulin sensitivity
Introduction
Type 2 diabetes (T2D) is marked by persistent hyperglycemia resulting from impaired glucose homeostasis. T2D may lead to serious health complications, including cardiovascular disease, kidney failure, and nerve damage. Moreover, T2D prevalence is projected to exceed 592 million people worldwide by 2035. Among the major drivers of this high prevalence is the combination of a sedentary lifestyle with poor diet quality. Conversely, the first line of treatment is to attempt to improve health behaviours, including optimizing dietary intake. Therefore, understanding the potential of healthy dietary patterns or specific food-derived components that can prevent or manage T2D is crucial for the development of nutritional recommendations in the disease context.
Among various dietary patterns to prevent or treat T2D, increased consumption of protein may improve glycemic control. Protein intake promotes satiety by stimulating the release of gastrointestinal hormones and slowing gastric digestion. Consequently, it can reduce energy intake and postprandial glucose levels leading to optimizing glycemic control in individuals with T2D. These physiological activities may be attributed, in part, to bioactive peptides generated during protein digestion. Bioactive peptides are biological molecules that modulate physiological pathways eliciting, for example, antidiabetic, antioxidant, anti-inflammatory, antihypertensive and immunomodulatory activities.
Eggs are a relatively inexpensive, rich source of high-quality protein with all essential amino acids, along with other nutrients such as vitamins and minerals. Considering their various nutritional benefits, recently egg white has gained attention for developing bioactive peptides. It is hypothesized that consuming these peptides individually or as a mixture in unpurified or minimally purified hydrolysates could be beneficial if included as part of the diet of individuals with T2D, although there are limited human trials testing this hypothesis. Currently, only two clinical trials have published information in that regard. One reported that a daily dosage of 5 g of egg-derived lysozyme hydrolysate prepared with alcalase (NWT-03) reduced blood glucose 2 h-post ingestion, an effect that persisted for 2 days in individuals with overweight and obesity with prediabetes or T2D. However, long-term intervention with NWT-03 for 27 days did not affect blood glucose concentration in individuals with metabolic syndrome.
Despite the limited human trials testing the effects of egg white hydrolysate (EWH), there are studies demonstrating the beneficial effects of EWH and/or egg-derived bioactive peptides on glucose homeostasis in vitro or glycemic control in rodents; however, the underlying mechanism by which egg-derived hydrolysates and peptides modulate glucose homeostasis are not clearly elucidated. Therefore, this review aims to discuss studies testing the glucoregulatory properties exerted by EWH and/or egg-derived bioactive peptides in preclinical models of obesity/T2D. Moreover, the effects of NWT-03 hydrolysate in the two clinical studies will be discussed in more detail. Additionally, this review highlights the gaps in this research field that could contribute to shaping future preclinical studies and clinical trials.
Egg-derived hydrolysates and peptides
Bioactive peptides promote physiological functions in the body beyond their nutritional benefits. Once in circulation, bioactive peptides enter cells or bind to cell membrane receptors, activating downstream signalling pathways. Egg protein is a rich source of bioactive peptides. Indeed, proteomics data indicates that there are approximately 70 proteins in the egg white, with most of them exhibiting bioactive properties. Both in vitro and in vivo studies reveal that egg white proteins have many health-related bioactivities including antioxidant, anti-inflammatory, anti-viral, anti-hypertensive, anti-diabetic effects.
Enzymatic hydrolysis is the main process for generating egg-derived bioactive peptides. Proteins are hydrolyzed either by gastrointestinal proteases such as pepsin, or non-gastrointestinal proteases such as alcalase or thermoase, which have wider cleavage sites compared to gastrointestinal proteases and can optimize the production of bioactive peptides. Various methods such as liquid chromatography can be used to identify individual peptides in the hydrolysates in an attempt to identify those with biological function. However, this increases the cost of production and may be unnecessary to derive therapeutic benefits, since hydrolysate mixtures have been shown metabolic improvements in preclinical models.
Effects of egg-derived hydrolysates and peptides on glucose homeostasis and insulin sensitivity in preclinical models
Glucoregulation is the main goal in diabetes management, and can be achieved by either directly modulating glucose absorption/reabsorption (i.e. ingestion of carbohydrate foods with low glycemic index or via pharmacotherapy to inhibit renal glucose reabsorption) or by modulating insulin secretion and/or insulin sensitivity to increase glucose uptake by the cells. There are several ways to influence insulin sensitivity, including body weight (BW) management, dietary interventions, physical activity and pharmacotherapy. In this review, we focus on studies evaluating the role of EWH (Table 1) and egg-derived bioactive peptides (Table 2) on indices of glucoregulation in vivo. For the included studies, we first provide an overview of the general outcomes observed, followed by the proposed mechanisms of action, the translation to human studies and the gaps present in this field of research.
METHODOLOGY
Initially 232 articles were identified in PUBMED based on the following key words: [Egg-derived peptide OR Egg peptide OR Egg white hydrolysate OR Egg protein hydrolysate OR Ovotransferrin OR Ovalbumin] AND [Glucose OR Insulin OR Glycemic control OR Glucose tolerance] AND [Mouse OR mice OR Rat OR Rats]. Only articles in English, published between January 2009 and June 2025, and animal studies involving mouse or rat models assessing glycemic markers were included in this review. Non-English articles, review papers, and studies published prior to 2009 were excluded. In addition, studies evaluating the effects of EWH or egg-derived peptides exclusively in vitro or in vivo studies in which no glycemic marker was measured were excluded. In vitro studies were used later to support proposed mechanisms in the discussion but were not included in Tables 1 or 2 as they do not follow the inclusion criteria. Following the application of the mentioned criteria, 13 animal studies were identified and included in this review. Another article that met the mentioned criteria was added in August 2025, as identified through the Web of Science database. Considering our focus on glycemic regulation, from the selected papers the information regarding the effects of in vivo studies of egg-derived hydrolysates and bioactive peptides on body weight, glucose homeostasis and insulin sensitivity were extracted and can be found in Tables 1 and 2. The data was extracted by two independent reviewers (YM, SCCZ) to check for the accuracy of the information retrieved before proceeding to drawing conclusions. For human studies, we searched ClinicalTrials.gov using the keywords Egg white hydrolysate and Egg-derived peptide as intervention. Among those studies, only two were found that included measurements of blood glucose.
| First Author, year | Animal model*; study duration | Intervention (Enzymes used) | Results/outcomes | Proposed mechanism of action |
|---|---|---|---|---|
| Cao, 2024 | C57BL/6J mice; HFD + low STZ; 14 wk | EWH 500 or 1000 mg/kg BW | BW: Food intake: N/A EWH 1000 ↓ FBG; GT, HOMA-IR | ↓ FBG – inhibition of gluconeogenesis and ↑ FAHFAs |
| Elhadad, 2024 | 4 wk old C57BL/6 mice; HFD; 14 wk | OVAHs 4% w/w (T or T+P) | BW: Both ↓; Food intake: Both OVAHs ↑ GT; ITT ↑ glucose uptake by ↑ insulin signaling in muscle via ↑ p-AKT and p-Irβ, p-AMPKα | N/A |
| Ochiai, 2014a | 3 wk old Wistar rats; HFSD; 8 wk | EW 286.2 g/kg + EWH 394.4 g/kg | BW: Food intake: ↓ FBG ↓ SCD indices and G6PD activity of the liver and gastrocnemius muscle | N/A |
| Ochiai, 2014b | 5 wk old GK rats vs Wistar rats; 6 wk | EWH 267.6 g/kg | BW: Food intake: ↓ FBG, HOMA-IR ↓ SCD indices in soleus muscle | N/A |
| Ochiai, 2017 | 4 wk old NSY mice; HFSD; 8 wk | EW 268.0g/kg + EWH 275.5 g/kg | BW: EWH ↑ GT; FBG, HOMA-IR, ITT ↓ intestinal glucose absorption and/or ↑ peripheral glucose uptake | N/A |
| Garcés-Rimón, 2018 | 8 wk old Zucker fatty rats; 12 wk | EWH1 (P), EWH2 (AP 433P) 750 mg/kg | BW: N/A EWH1 ↓ HOMA-IR; FBG | N/A |
| Jahandideh, 2019 | 8 wk old Sprague Dawley rats; HFD; 12 wk | EWH 1-4 g/100g diet (T+P) | BW, food intake: 4% EWH ↑ GT and insulin sensitivity; FBG, HOMA-IR 4 % EWH ↑ adipocyte differentiation | N/A |
| Moreno-Fernández, 2018 | 8 wk old Wistar rats; HFD+high dextrose; 20 wk | EWH 1 g/kg (P) | BW ↓ Food intake: FBG, HOMA-IR N/A | N/A |
| Wang, 2012 | 10 wk old obese ZDF; 15 wk | NWT-03 1 g/kg (Alcalase) | BW: Food intake: N/A FBG but ↓ long term renal damage | ↓ oxidative stress and ↓ COX expression, ↓ ACE and DPP4 activities |
| Wu, 2025 | 6 wk old Wistar rats on HFD; 4 wk | EWH 1 g/kg (P+pancreatin) | BW ↓ Food intake ↓ ↑ GT; IR | Suppressed ghrelin secretion through mTOR pathway activation |
| First Author, year | Animal model*, study duration | Intervention (Hydrolysis enzyme) | Results/outcomes | Proposed mechanism of action |
|---|---|---|---|---|
| de Campos Zani, 2022 | 4 wk old C57BL/6 mice on HFD; 14 wk | IRW 15 and 45 mg/kg BW | IRW 45 ↓ BW Food intake: IRW 45 ↓ FBG, HOMA-IR; ↑ GT | Skeletal muscle: ↑ glucose uptake; ↑ AKT/GLUT4 and AMPKα/GLUT4 |
| de Campos Zani, 2023 | 5 wk old C57BL/6 mice on HFD; 14 wk | PEP2 45 mg/kg BW | BW and food intake: PEP2 ↓ IR; FBG, HOMA-IR, GT | ↑ systemic IR. WAT: ↓ lipolysis and ↓ spillover of lipids, ↑ insulin signaling, PPARγ |
| Liu, 2022 | 9 wk old C57BL/6J mice on HFD; 8 wk | IRW or IQW 0.03 g/L | BW: IQW ↓, IRW Food intake: N/A Both ↓ FBG, ↑ GT and ↓ IR | ↓ lipid deposition, maintained energy balance, and reprogrammed gut microbiota |
| de Campos Zani, 2024 | 5 wk old C57BL/6 mice on HFD; 14 wk | IRW 45 mg/kg BW | ↓ Food intake: ↑ GT; ↓ FBG | Regulated hepatic lipid metabolism, ↑ mitochondria OxPhos capacity |
Results
OVERALL CHARACTERISTICS OF THE INCLUDED STUDIES
Characteristics of the selected studies are shown in Tables 1 and 2. All studies aimed to investigate the effect of EWH or egg-derived peptides on glycemic control in T2D/obesity rodent models. Among 14 studies, seven used either high fat diet (HFD) fed mice or spontaneous T2D Nagoya-Shibata-Yasuda (NSY) mice. The remaining studies used rats, which were either HFD induced obesity/T2D or spontaneous genetic T2D models, including Goto-Kakizaki (GK) rats as non-obese and Zucker diabetic fatty rats (ZDF) as obese rats. The effects of feeding EWH and individual peptides on glycemic control were assessed in comparison to control groups, involving HFD fed rodents without EWH or peptides.
Regarding the interventions, the studies used whole EWH, ovalbumin hydrolysates (OVAH), Newtricious-03 (NWT-03) or individual egg white peptides, including IRW (isoleucine, arginine, and tryptophan), IQW (isoleucine, glutamine, tryptophan), or QAMPFRVTEQE (PEP2).
BODY WEIGHT AND FOOD INTAKE
Increased BW and food intake are associated with the development of insulin resistance (IR) and T2D. While some studies revealed that neither EWH nor bioactive peptides altered BW gain, others reported a reduction in BW gain of rodents. Most of the studies reporting a decrease in BW were related to the ingestion of EWH with fewer studies evaluating single white egg-derived peptides. Because there are several bioactive peptides in an EWH attributing the observed reduction in BW gain to a single peptide is impossible.
For example, compared with a high-fat/high dextrose diet (HFSD) control, Moreno-Fernández et al. observed a decrease of approximately 20% BW gain after the addition of an EWH generated with pepsin hydrolysis to the diet. However, others observed smaller decreases in BW gain after EWH or single peptide ingestion. The production of EWH/peptides can affect the palatability of the diet (i.e., hydrolysis can increase bitterness); therefore, it is noteworthy that the reductions in BW reported by most of these studies were independent of changes in food intake. One study did report a decrease in food intake in rats receiving EWH, which was attributed to its effect in suppressing the hormone ghrelin. Based on these findings, it is plausible to speculate that the greater reduction in BW in whole EWH studies might be due to the additive effect of the peptides found in hydrolysates, compared to the effect of single peptide supplementation.
FASTING BLOOD GLUCOSE AND HOMA-IR
In T2D, persistent elevation in fasting blood glucose (FBG) caused by disrupted insulin secretion and/or IR is a key clinical marker of impaired glucose homeostasis. IR is a condition in which cells of the body exhibit a diminished response to insulin action, particularly in the skeletal muscle, adipose tissue and liver. This impaired insulin signaling decreases glucose uptake and prevents the inhibition of lipolysis and gluconeogenesis during fed states thereby contributing to hyperglycemia and hyperlipidemia. These are hallmark metabolic characteristics of T2D.
Homeostasis model assessment of IR (HOMA-IR) is a surrogate measure of IR that considers the interaction between FBG and fasting insulin concentrations in humans and can also be a suitable tool to evaluate IR in mice. Addition of EWH to the diet of T2D mouse models decreased FBG concentration compared with their respective controls in two independent studies. Only one of them reported a parallel reduction in HOMA-IR. However, Ochiai’s group found no effect of EWH on FBG and HOMA-IR when using a different animal model, suggesting that the effect of the EWH might be dependent on certain genetic background. Moreover, two studies from another group using an EWH produced with pepsin found no significant changes in FBG, while one of them saw improvement in HOMA-IR.
Supplementing the diet of mice on HFD with the single peptides IRW or IQW decreased FBG concentration compared with their respective controls. Similarly, HOMA-IR was decreased by IRW as reported by the same research team. On the other hand, the same group also reported that supplementation with a different peptide (PEP2) did not alter FBG levels.
Taken together, the conflicting effects of EWH or single peptides in improving FBG or HOMA-IR might be due to the different animal model used to assess FBG or due to the proteases used in producing the peptides, which could generate a different pool of bioactive peptides present in the hydrolysate. Unfortunately, the protease used in some of the studies was not specified, therefore, we cannot confirm this aspect.
GLUCOSE TOLERANCE
Glucose tolerance, which refers to the ability of the body to maintain euglycemia, is impaired in T2D. Glucose tolerance in fasted rodents is measured by administering a glucose challenge either orally (OGTT) or intraperitoneally (ipGTT) and measuring glucose in the blood for 1 to 3 hours. The interpretation of results includes both insulin-mediated and non-insulin-mediated mechanisms of glucose disposal and is aided if insulin is measured simultaneously.
A research group found in two different studies that supplementation of a HFD with either OVAH in mice or EWH in rats, both generated with pepsin and thermoase, improved glucose tolerance following an OGTT. Additionally, another research group reported a similar improvement in the OGTT after EWH supplementation. In this study, pepsin and pancreatin were used for the hydrolysis process. Another study reported an improvement in the OGTT (but not in the ipGTT) at 60, 90 and 180 minutes, after EWH supplementation in a spontaneous T2D rat model. Yet, when the AUC was calculated, despite a reduction of more than 50%, the result did not reach statistical significance. A fourth independent group did not observe changes in glucose tolerance following addition of EWH in a HFD and low-dose streptozotocin-induced T2D mice.
In terms of single peptides affecting glucose tolerance, IRW supplementation improved OGTT and ipGTT. IQW also improved ipGTT in this study. These data suggests that the effects of IRW on glucose tolerance might be independent of incretin hormones, which can augment glucose clearance via their effects on insulin secretion, considering that ipGTT bypasses the intestine. On the other hand, providing a different peptide (PEP2) in the diet did not affect OGTT in a HFD-fed mice that developed prediabetes.
The inconsistent effects of EWH in improving glucose tolerance might be explained by the pool of the peptides present in the hydrolysate, considering that the studies reporting improvements in glucose tolerance all had in common pepsin as one of the hydrolytic enzymes, while those finding no improvements did not specify the protease used.
DIRECT MEASURES OF INSULIN SENSITIVITY
Insulin sensitivity is often assessed in preclinical models by the insulin tolerance test (ITT). The ITT indicates how effectively glucose is cleared from circulation via insulin sensitive tissues following an intraperitoneal administration of insulin. The measurements are done over a 1-2 hour period with intervals ranging from 15 to 30 minutes.
While the majority of studies did not report any improvements in ITT after EWH in either mice on a HFD or rats on a HFD or peptide supplementation in mice on a HFD, EWH made with thermoase and pepsin were effective in improving IR after ITT in rats on a HFD. This intervention also elicited an improvement in the OGTT, suggesting that the modulation of glucose tolerance by that EWH might be mediated by an insulin-dependent pathway. Furthermore, two independent research groups found that PEP2 and IRW or IQW improved IR. The improvement in ITT observed with PEP2 supplementation was confirmed by an hyperinsulinemic-euglycemic clamp, which is considered the gold standard for measuring insulin sensitivity.
Considering the similar improvements in ITT and glucose tolerance by EWH and IRW (an ovotransferrin-derived peptide), and the lack of improvements in OGTT by PEP2 (ovalbumin-derived peptide) or in the ITT by OVAH, the effects observed might be due to the combined action of the individual peptides present in the EWH after thermoase and pepsin hydrolysis rather than the effect of individual peptides. The additive effects of ovalbumin and ovotransferrin peptides in the EWH possibly contributed to its greater influence in obese/T2D models compared to only OVAH. Nevertheless, more studies from different groups using the same peptides or EWH would be necessary to confirm the reproducibility of the effects mentioned above.
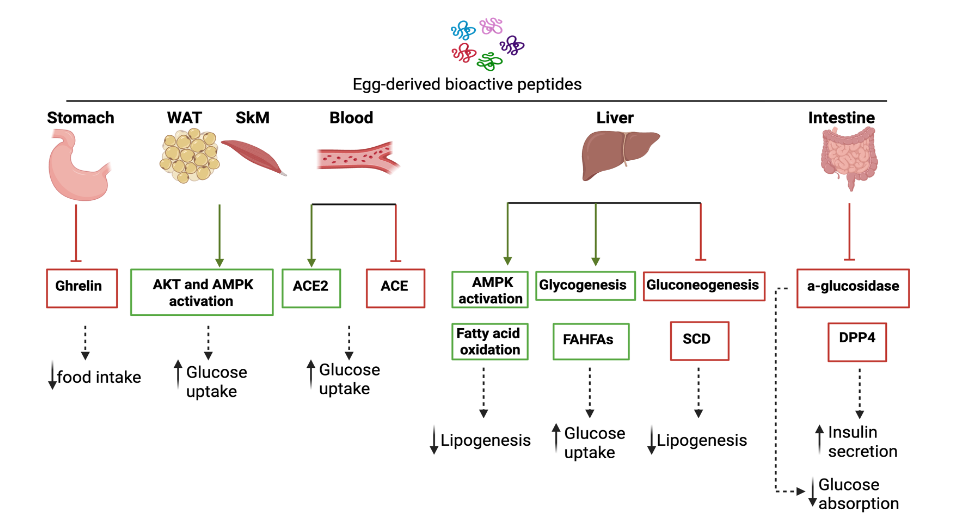
Proposed mechanisms of action for glucoregulatory effects of EWH and peptides
As mentioned before, several physiological processes are modulated in the regulation of glucose homeostasis. The proposed mechanisms by which egg hydrolysates or single peptides might be exerting their effects based on the data presented in Tables 1 and 2. In addition, in vitro studies suggest some mechanisms behind the glucoregulatory effects of EWH and egg-derived peptides, such as α-glucosidase inhibition by RVPSLM peptide isolated from EWH generated with alcalase and dipeptidyl peptidase 4 (DPP-4) inhibition by EWH generated with peptidase 433P and BC Pepsin. α-glucosidase is an enzyme that increases glucose absorption by hydrolyzing carbohydrates to glucose; therefore, α-glucosidase inhibitory peptides would be predicted to decrease glucose absorption and reduce blood glucose as suggested by others, but this effect has not been confirmed in vivo. DPP-4 is an enzyme that reduces insulin secretion by inactivating the incretin hormones glucose-dependent insulinotropic polypeptide (GIP) and GLP-1. Therefore, inhibiting its activity could improve glycemic control; a hypothesis that remains to be investigated.
Another mechanism involves enzymes in the angiotensin-converting enzyme (ACE) pathway, a major regulator of blood pressure. IRW is a peptide known for its anti-hypertensive activity. Although initially thought to be an ACE inhibitor, IRW acts via ACE2 upregulation, such as in mesenteric arteries at the mRNA level in vitro, and at the protein level in the kidneys and aorta of spontaneously hypertensive rats. ACE2, a homologue of ACE, cleaves angiotensin II (Ang II) to produce Ang (1-7). Ang (1-7) attenuates Ang II-induced vasoconstriction and, importantly, promotes glucose transporter 4 (GLUT4) translocation through binding to Mas receptor, which together results in reduced blood pressure and blood glucose concentrations. In a HFD obese model, de Campos Zani et al. reported that enhanced glucose tolerance observed after IRW supplementation was independent of ACE inhibition but ACE2 activity in plasma increased following IRW supplementation, supporting this hypothesis.
Obesity plays a key role in the pathophysiology of IR/T2D by promoting inflammation through the release of pro-inflammatory cytokines from immune cells located in adipose tissue, including M1 macrophages, B cells, and T cells. This chronic low-grade inflammation can impair insulin signaling, thereby contributing to the development of IR. Thus, reduction of BW is associated with improved glycemic markers in clinical studies of individuals with obesity and T2D. This is consistent with the findings in rodents supplemented with EWH or peptides, in which all studies reporting a reduction in BW gain also observed an improvement in glucose tolerance. Additionally, among studies reporting decreased BW gain, one reported a decrease in FBG and two reported a reduction in FBG and enhancement of insulin sensitivity.
Decreased fat accumulation and BW also lead to improved insulin sensitivity, in which decreased stearoyl-CoA desaturase (SCD) in skeletal muscle and liver may play an important role. SCD regulates fat metabolism by catalyzing the conversion of saturated fatty acids to unsaturated fatty acids. Reductions in SCD could be responsible for decreased lipogenesis, which is beneficial for insulin sensitivity. Still on the topic of modulating hepatic lipid metabolism to improve glycemic markers, it is proposed the IRW might act via AMPKα activation to increase fatty acid oxidation capacity in the liver preventing hepatic fat accumulation and contributing to a reduction in BW and improved glucose tolerance. Another mechanism for the effect of EWH or peptides on BW gain could be via inhibition of ghrelin secretion through activation of mechanistic target of rapamycin (mTOR) signaling pathway in the gastric fundus. Ghrelin is a hunger hormone secreted by the endocrine cells in the stomach that controls energy metabolism via its action in the hypothalamus, including increasing food intake and consequently BW gain. mTOR is a serine/threonine protein kinase that regulates energy metabolism in response to nutrients and the activation of mTOR signaling pathway is negatively linked to ghrelin secretion. Even though these studies indicate a link between BW changes and glycemic markers, in some studies reduction in BW and improved glycemic markers are not correlated. This suggests that EWH and peptides might be acting through another mechanism, such as increasing GLP-1 or energy expenditure to improve glucose homeostasis.
Human studies and egg ingestion: Are the results translational?
The ingestion of eggs has been studied in clinical trials, including in healthy individuals and those with obesity or T2D. A meta-analysis of ten randomized controlled trials assessed the adverse effects of whole egg consumption in individuals at risk or diagnosed with T2D and found that ingestion of six to 12 eggs weekly within a healthy diet recommended for cardiovascular health did not negatively impact T2D. However, whole eggs also did not improve BW or glycemic markers, including FBG and HOMA-IR.
Therefore, the use of EWH or peptides could provide several advantages. For example, egg white is an allergen for some individuals and the process of hydrolysis can decrease the allergenicity of the egg white. Moreover, the process of hydrolysis of egg white could potentially increase the solubility in water and improve the gastric digestion and absorption of nutrients. Therefore, there is a strong rationale for investigating the glycemic effects of EWH or egg-derived bioactive peptides in individuals with obesity and T2D. Nonetheless, there are currently limited clinical trials investigating the glucoregulation effects of EWH in humans, which showed either a beneficial effect or neutral effect of NWT-03 on glucose tolerance markers. To our knowledge, there are no clinical trials testing the bioavailability of a single egg-derived peptide from EWH.
Translation of studies in rodents presented in this review to humans still remains to be investigated. And yet, translating doses of EWH or individual egg-derived peptides from preclinical rodent studies to clinical human studies is challenging due to several reasons such as different metabolism between species. Indeed, rodents need more protein than human in general and have higher metabolic rate. Some of these studies used a low dose, which could be more translatable to human physiology, while the other studies used a very high dose making it impossible to translate to clinical trials. In the above mentioned pre-clinical studies, rats typically consumed 750 mg/kg of BW of EWH daily, which accounts for approximately 3.3% of their total daily energy intake. Considering an average daily energy intake in humans (2000 to 2500 kcal/day), this would translate to approximately 66 to 82 kcal per day derived from the protein supplement. Therefore, the equivalent human dose of EWH would be approximately 16 to 20 g per day. Given that the recommended daily intake of available protein supplements on the market (ex whey protein) is between 20-25 g, the selected dose in these preclinical studies would be relevant when translated to human clinical trial. With this in mind and considering the above mentioned effects observed in diet-induced models of obesity/prediabetes after IRW supplementation, our group is conducting a double-blind randomized cross-over study, (registered in clinicaltrials.gov as NCT06555393) to investigate the acute effects of IRW peptide from EWH by thermoase (20 g daily) on blood glucose levels and its bioavailability in individuals at risk of or diagnosed with T2D. An amount of 20 g of EWH produced using thermoase and pepsin has approximately 22 mg of the peptide IRW. Based on pre-clinical trials, we anticipate that this dosage of IRW will be sufficient to promote beneficial effects in humans in terms of glucose regulation and IRW bioavailability.
Conclusion
Existing preclinical studies reveal that EWH and bioactive peptides could potentially play an important role in managing glycemia. However, inconsistent effect on FBG, glucose tolerance, insulin sensitivity and BW outcomes across preclinical studies could stem from use of animal models with different physiology and age, peptide sources (hydrolysates and peptides with different amino acid sequences), dosage, and administration of the peptides (gavage and mixed in the diet).
Moreover, none of the selected studies used female rodents. Although female rodents have higher insulin sensitivity and are not as prone to develop the IR phenotype compared to male rodents, future studies should investigate potential sex differences in the response to EWH and peptides to make results more translatable to the human population. Additionally, none of the studies quantified peptide concentration in plasma of the animals. The extent to which a peptide is absorbed into circulation and reaches its intended tissue defines its bioavailability, which is affected by several factors such as amino acid sequence and peptide molecular weight. Therefore, studies are needed in this regard. Considering the number of peptides present in the EWH, there are still limited studies investigating the effects of individual peptides on glycemic control; further studies are needed to explore these effects in preclinical models and facilitate the design of standardized clinical trials.
In summary, the results presented in this review indicate a potential beneficial effect of EWH and single egg-derived bioactive peptides in the regulation of glucose homeostasis. This might occur through modulation of BW, glucose absorption/uptake, enhancing insulin secretion or insulin signaling. Nevertheless, more robust animal studies and clinical trials investigating the translatability of these findings to humans will enhance our knowledge in this field and advance the use of egg-peptides in the management of T2D.
References
- Goyal R, Singhal M, Jialal I. Type 2 Diabetes. [Updated 2023 Jun 23]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls.; 2023. https://www.ncbi.nlm.nih.gov/books/NBK513253/
- Kharroubi AT. Diabetes mellitus: The epidemic of the century. World J Diabetes. 2015;6(6):850. doi:10.4239/wjd.v6.i6.850
- Huang X, Wu Y, Ni Y, Xu H, He Y. Global, regional, and national burden of type 2 diabetes mellitus caused by high BMI from 1990 to 2021, and forecasts to 2045: analysis from the global burden of disease study 2021. Front Public Health. 2025;13: 1515797. doi:10.3389/fpubh.2025.1515797
- Weisman A, Fazli GS, Johns A, Booth GL. Evolving Trends in the Epidemiology, Risk Factors, and Prevention of Type 2 Diabetes: A Review. Can J Cardiol. 2018;34(5):552-564. doi:10.1016/j.cjca.2018.03.002
- Anjom-Shoae J, Feinle-Bisset C, Horowitz M. Impacts of dietary animal and plant protein on weight and glycemic control in health, obesity and type 2 diabetes: friend or foe? Front Endocrinol (Lausanne). 2024;15:1412182. doi:10.3389/fendo.2024.1412182
- Moreno-Fernández S, Garcés-Rimón M, Miguel M. Egg-derived peptides and hydrolysates: A new bioactive treasure for cardiometabolic diseases. Trends Food Sci Technol. 2020;104:208-218. doi:10.1016/j.tifs.2020.08.002
- Wang X, Son M, Meram C, Wu J. Mechanism and potential of egg consumption and egg bioactive components on type-2 diabetes. Nutrients. 2019;11(2):357. doi:10.3390/nu11020357
- Plat J, Severins N, Mensink RP. Improvement of pulse wave velocity and metabolic cardiovascular risk parameters through egg protein hydrolysate intake: A randomized trial in overweight or obese subjects with impaired glucose tolerance or type 2 diabetes. J Funct Foods. 2019;52:418-423. doi:10.1016/j.jff.2018.11.020
- Nijssen KMR, Joris PJ, Mensink RP, Plat J. Longer-term effects of the egg-protein hydrolysate NWT-03 on arterial stiffness and cardiometabolic risk markers in adults with metabolic syndrome: a randomized, double-blind, placebo-controlled, crossover trial. Eur J Clin Nutr. 2023;77(10):982-988. doi:10.1038/s41430-023-01305-8
- Son M, Wu J. Egg white hydrolysate and peptide reverse insulin resistance associated with tumor necrosis factor-α (TNF-α) stimulated mitogen-activated protein kinase (MAPK) pathway in skeletal muscle cells. Eur J Nutr. 2019;58(5):1961-1969. doi:10.1007/s00394-018-1753-7
- Son M, Chan CB, Wu J. Egg White Ovotransferrin-Derived ACE Inhibitory Peptide Ameliorates Angiotensin II-Stimulated Insulin Resistance in Skeletal Muscle Cells. Mol Nutr Food Res. 2018;62(4):1700602. doi:10.1002/mnfr.201700602
- Wang Y, Landheer S, van Gilst WH, et al. Attenuation of Renovascular Damage in Zucker Diabetic Fatty Rat by NWT-03, an Egg Protein Hydrolysate with ACE- and DPP4-Inhibitory Activity. PLoS One. 2012;7(10):e46781. doi:10.1371/journal.pone.0046781
- Jahandideh F, De Campos Zani SC, Son M, et al. Egg white hydrolysate enhances insulin sensitivity in high-fat diet-induced insulin-resistant rats via Akt activation. Br J Nutr. 2019;122(1):14-24. doi:10.1017/S0007114519000837
- Garcés-Rimón M, González C, Vera G, et al. Pepsin egg white hydrolysate improves glucose metabolism complications related to metabolic syndrome in zucker fatty rats. Nutrients. 2018;10(4):441. doi:10.3390/nu10040441
- Ochiai M, Azuma Y. Egg White Hydrolysate Improves Glucose Tolerance in Type-2 Diabetic NSY Mice. J Nutr Sci Vitaminol. 2017;63:422-429. doi:10.3177/jnsv.63.422
- Moreno-Fernández S, Garcés-Rimón M, González C, et al. Pepsin egg white hydrolysate ameliorates metabolic syndrome in high-fat/high-dextrose fed rats. Food Funct. 2018;9:78-86. doi:10.1039/c7fo01280b
- Elhadad N, de Campos Zani SC, Chan CB, Wu J. Ovalbumin Hydrolysates Enhance Skeletal Muscle Insulin-Dependent Signaling Pathway in High-Fat Diet-Fed Mice. J Agric Food Chem. 2024;72(27):15248-15255. doi:10.1021/acs.jafc.4c01008
- de Campos Zani SC, Wang R, Veida-Silva H, et al. An Egg White-Derived Peptide Enhances Systemic Insulin Sensitivity and Modulates Markers of Non-Alcoholic Fatty Liver Disease in Obese, Insulin Resistant Mice. Metabolites. 2023;13(2):174. doi:10.3390/metabo13020174
- de Campos Zani SC, Son M, Bhullar KS, Chan CB, Wu J. IRW (Isoleucine Arginine Tryptophan) Improves Glucose Tolerance in High Fat Diet Fed C57BL/6 Mice via Activation of Insulin Signaling and AMPK Pathways in Skeletal Muscle. Biomedicines. 2022;10(6):1235. doi:10.3390/biomedicines10061235
- Liu Z, Ding S, Jiang H, Fang J. Egg Protein Transferrin-Derived Peptides Irw (Lle-Arg-Trp) and Iqw (Lle-Gln-Trp) Prevent Obesity Mouse Model Induced by a High-Fat Diet via Reducing Lipid Deposition and Reprogramming Gut Microbiota. Int J Mol Sci. 2022;23(19). doi:10.3390/ijms231911227
- Cao X, Chen L, Lu K, et al. Egg white-derived peptides reduced blood glucose in high-fat-diet and low-dose streptozotocin-induced type 2 diabetic mice via regulating the hepatic gluconeogenic signaling and metabolic profile. Food Funct. 2024;15(13):7003-7016. doi:10.1039/d4fo00725e
- Ochiai M, Kuroda T, Matsuo T. Increased muscular triglyceride content and hyperglycemia in Goto-Kakizaki rat are decreased by egg white hydrolysate. Int J Food Sci Nutr. 2014;65(4):495-501. doi:10.3109/09637486.2013.879288
- Ochiai M, Matsuo T. Effect of egg white and its hydrolysate on stearoyl-CoA desaturase index and fat accumulation in rat tissues. Int J Food Sci Nutr. 2014;65(8):948-952. doi:10.3109/09637486.2014.937800
- Liao W, Jahandideh F, Fan H, Son M, Wu J. Egg Protein-Derived Bioactive Peptides: Preparation, Efficacy, and Absorption. Adv Food Nutr Res. 2018;85:1-58. doi:10.1016/bs.afnr.2018.02.001
- Bao X, Wang Z, Wu J. Bioactives of egg white proteins and peptides. In: Handbook of Egg Science and Technology. CRC Press; 2023:411-442. doi:10.1201/9781003254430-29
- De Campos Zani SC, Wu J, Chan CB. Egg and soy-derived peptides and hydrolysates: A review of their physiological actions against diabetes and obesity. Nutrients. 2018;10(5):549. doi:10.3390/nu10050549
- Wu T, Wang W, Hai N, et al. Egg white peptides suppress ghrelin secretion from the stomach by activating the mTOR signaling pathway in rats. Food Res Intl. 2025;216:116710. doi:10.1016/j.foodres.2025.116710
- DeFronzo RA, Ferrannini E, Groop L, et al. Type 2 diabetes mellitus. Nat Rev Dis Primers. 2015;1:15019. doi:10.1038/nrdp.2015.19
- Veit M, van Asten R, Olie A, Prinz P. The role of dietary sugars, overweight, and obesity in type 2 diabetes mellitus: a narrative review. Eur J Clin Nutr. 2022;76(11):1497-1501. doi:10.1038/s41430-022-01114-5
- de Campos Zani SC, Berg E, Jiang X, et al. IRW improves diet-induced non-alcoholic fatty liver disease by reducing steatosis associated with increased capacity for oxidative phosphorylation. J Funct Foods. 2024;112:105976. doi:10.1016/j.jff.2023.105976
- Samuel VT, Shulman GI. The pathogenesis of insulin resistance: Integrating signaling pathways and substrate flux. J Clin Invest. 2016;126(1):12-22. doi:10.1172/JCI77812
- Bowe JE, Franklin ZJ, Hauge-Evans AC, King AJ, Persaud SJ, Jones PM. Assessing glucose homeostasis in rodent models. J Endocrinol. 2014;222(3):13-25. doi:10.1530/JOE-14-0182
- Jagannathan R, Neves JS, Dorcely B, et al. The oral glucose tolerance test: 100 years later. Diabetes Metab Syndr Obes. 2020;13:3787-3805. doi:10.2147/DMSO.S246062
- Moro C, Magnan C. Revisited guidelines for metabolic tolerance tests in mice. Lab Anim (NY). 2025;54:16-23. doi:10.1038/s41684-024-01473-5
- Yu Z, Yin Y, Zhao W, et al. Novel peptides derived from egg white protein inhibiting alpha-glucosidase. Food Chem. 2011;129(4):1376-1382. doi:10.1016/j.foodchem.2011.05.067
- Garcés-Rimón M, López-Expósito I, López-Fandiño R, Miguel M. Egg white hydrolysates with in vitro biological multiactivities to control complications associated with the metabolic syndrome. Eur Food Res Technol. 2016;242(1):61-69. doi:10.1007/s00217-015-2518-7
- Pan G, Lu Y, Wei Z, Li Y, Li L, Pan X. A review on the in vitro and in vivo screening of α-glucosidase inhibitors. Heliyon. 2024;10(18):e37467. doi:10.1016/j.heliyon.2024.e37467
- Majumder K, Liang G, Chen Y, Guan L, Davidge ST, Wu J. Egg ovotransferrin-derived ACE inhibitory peptide IRW increases ACE2 but decreases proinflammatory genes expression in mesenteric artery of spontaneously hypertensive rats. Mol Nutr Food Res. 2015;59(9):1735-1744. doi:10.1002/mnfr.201500050
- Liao W, Bhullar KS, Chakrabarti S, Davidge ST, Wu J. Egg White-Derived Tripeptide IRW (Ile-Arg-Trp) is an Activator of Angiotensin Converting Enzyme 2. J Agric Food Chem. 2018;66(43):11330-11336. doi:10.1021/acs.jafc.8b03501
- Bindom SM, Lazartigues E. The sweeter side of ACE2: Physiological evidence for a role in diabetes. Mol Cell Endocrinol. 2008;302(2):193. doi:10.1016/J.MCE.2008.09.020
- Zatterale F, Longo M, Naderi J, et al. Chronic Adipose Tissue Inflammation Linking Obesity to Insulin Resistance and Type 2 Diabetes. Front Physiol. 2020;10:1607. doi:10.3389/fphys.2019.01607
- Look AHEAD Research Group WR. Long-term Effects of a Lifestyle Intervention on Weight and Cardiovascular Risk Factors in Individuals With Type 2 Diabetes Mellitus Four-Year Results of the Look AHEAD Trial. Arch Intern Med. 2010;170:1566-1575. doi:10.1001/archinternmed.2010.334
- Akalu Y, Molla MD, Dessie G, Ayelign B. Physiological Effect of Ghrelin on Body Systems. Int J Endocrinol. 2020;2020:1385138. doi:10.1155/2020/1385138
- Oh KJ, Han HS, Kim MJ, Koo SH. CREB and FoxO1: Two transcription factors for the regulation of hepatic gluconeogenesis. BMB Rep. 2013;46(12):567-574. doi:10.5483/BMBRep.2013.46.12.248
- Jaiswal N, Gavin MG, Quinn WJ, et al. The role of skeletal muscle Akt in the regulation of muscle mass and glucose homeostasis. Mol Metab. 2019;28:1-13. doi:10.1016/j.molmet.2019.08.001
- Islam MS, Loots DT. Experimental rodent models of type 2 diabetes: A review. Methods Find Exp Clin Pharmacol. 2009;31(4):249-261. doi:10.1358/mf.2009.31.4.1362513
- Hahn MK, Giacca A, Pereira S. In vivo techniques for assessment of insulin sensitivity and glucose metabolism. J Endocrinol. 2024;260(3):e230308. doi:10.1530/JOE-23-0308
- Jackson SJ, Andrews N, Ball D, et al. Does age matter? The impact of rodent age on study outcomes. Lab Anim. 2017;51(2):160-169. doi:10.1177/0023677216653984
- Richard C, Cristall L, Fleming E, et al. Impact of Egg Consumption on Cardiovascular Risk Factors in Individuals with Type 2 Diabetes and at Risk for Developing Diabetes: A Systematic Review of Randomized Nutritional Intervention Studies. Can J Diabetes. 2017;41(4):453-463. doi:10.1016/j.jcjd.2016.12.002
- Ballmer-Weber BK, Brockow K, Fiocchi A, et al. Hydrolysed egg displays strong decrease in allergenicity and is well tolerated by egg-allergic patients. Allergy Eur J Allergy Clin Immunol. 2016;71(5):728-732. doi:10.1111/all.12852
- Lai M, Chandrasekera PC, Barnard ND. You are what you eat, or are you? the challenges of translating high-fat-fed rodents to human obesity and diabetes. Nutr Diabetes. 2014;4(9):e135. doi:10.1038/nutd.2014.30
- Li Q, Liao W, Fan H, Wu J. Optimization and Scale-Up Preparation of Egg White Hydrolysate With Angiotensin I Converting Enzyme Inhibitory Activity. J Food Sci. 2018;83(6):1762-1768. doi:10.1111/1750-3841.14158
- Sun X, Acquah C, Aluko RE, Udenigwe CC. Considering food matrix and gastrointestinal effects in enhancing bioactive peptide absorption and bioavailability. J Funct Foods. 2020;64. doi:10.1016/j.jff.2019.103680




