Efficacy of Fibranium Mattress Topper for Chronic Pain
Efficacy of a Novel Fibranium Fiber Mattress Topper for Reducing Chronic Low Back Pain: A Randomized, Double-Blind, Placebo-Controlled Trial
Michael Ofner 1,2, Martin Liebhauser 3, Harald Walach 4,5
- Department of Pathophysiology and Immunology, Medical University Graz, Austria
- Medyco International, Dubai, United Arab Emirates
- ImPuls Institut, Klagenfurt, Austria
- Kazimieras Simonavičius University, Vilnius, Lithuania
- CHS-Institut, Basel, Switzerland
OPEN ACCESS
PUBLISHED: 30 April 2025
CITATION: Ofner, M., Liebhauser, M., et al., 2025. Efficacy of a Novel Fibrin™ Fiber Mattress Topper for Reducing Chronic Low Back Pain: A Randomized, Double-Blind, Placebo-Controlled Trial. Medical Research Archives, [online] 13(4).
https://doi.org/10.18103/mra.v13i4.6494
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i4.6494
ISSN 2375-1924
Abstract
Chronic low back pain is a prevalent and debilitating condition, often with limited treatment options. Previous research has shown that a mattress topper containing ‘Fibrilium’ fibers can effectively reduce chronic low back pain. This study investigated an upgraded fiber, called “Fibranium,” with enhanced composition and manufacturing, hypothesized to improve effects for pain reduction and associated parameters. The aim of this study was to evaluate the efficacy of a Fibranium fiber mattress topper for reducing pain intensity and improving function in individuals with chronic low back pain compared to a placebo topper. This prospective, randomized, double-blind, placebo-controlled trial was conducted with 54 patients (aged 40-70 years) with chronic low back pain (≥ 6 weeks, pain intensity ≥ 5/10 on a Numerical Rating Scale). Participants were randomized to a Fibranium topper (n=27) or a placebo topper (n=27) for 4 weeks. The primary outcome was the McGill Pain Questionnaire total score. Secondary outcomes included the Oswestry Disability Index, Linton-Halldén Score, Pittsburgh Sleep Quality Index, Short Form-36. Data were analyzed using linear models, with baseline scores as covariates. The Fibranium group showed significantly greater improvements in McGill total score (p = 0.000002), Oswestry Disability Index (p = 0.000005), Linton-Halldén Score (p = 0.000010), and SF-36 total score (p = 0.000001) compared to the placebo group. All SF-36 subscales except Emotional Wellbeing showed significant improvements. No significant difference was found in the Pittsburgh Sleep Quality Index total score; however, sleep latency (component 2) improved significantly in the Fibranium group (p = 0.000175). The Fibranium fiber mattress topper demonstrated significant efficacy in reducing pain and improving function in individuals with chronic low back pain. These findings support Fibranium as a non-pharmacological intervention for chronic low back pain.
Keywords: chronic low back pain, Fibranium, mattress topper, randomized controlled trial, pain management, non-pharmacological intervention, infrared radiation
1. Introduction
Chronic low back pain (CLBP) is a leading cause of disability worldwide, significantly impacting individuals and healthcare systems. Current guidelines prioritize non-pharmacological approaches due to concerns over opioid dependency and the limited efficacy of certain pharmacological treatments. Already the NICE-guidelines of 2009 on treating acute low back pain discouraged pharmacological treatments and advised exercise, mobilisation and acupuncture as first-line treatments. While exercise and other active therapies offer modest benefits, the need for additional safe, non-invasive and effective interventions remains.
Our previous research demonstrated that a mattress topper containing “Fibrilium” fibers, derived from Korean clay using nanotechnology, significantly reduced pain and improved function in CLBP patients. This finding provided a strong rationale for exploring enhanced fiber technologies. Other studies have also suggested that modifications to mattress characteristics can positively influence back pain.
The current study investigates “Fibranium,” an upgraded version of the Fibrilium fiber. It incorporates additional crystalline minerals and employs a modified manufacturing process with adjusted temperatures. These modifications are intended to enhance the fiber’s ability to reflect far-infrared radiation (FIR) naturally emitted by the body. In a laboratory experiment conducted in August 2024 at the University of Pisa, the optical properties of two fabric samples — one untreated (without Fibranium fibers) and one treated (containing Fibranium fibers)— were analyzed at 37°C within the spectral range of 7 to 14 µm.
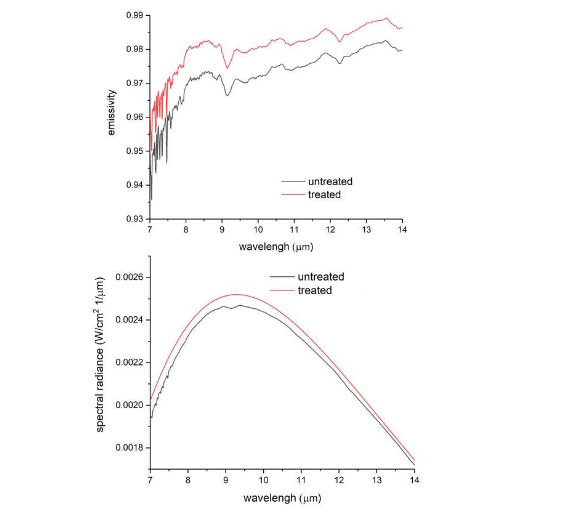
FIR reflection is proposed to improve local circulation, reduce inflammation, and modulate pain signalling pathways. Beyond the well-established effects of FIR, emerging research suggests that interactions within the human biofield may also play a role in physiological regulation and pain perception. The human body emits ultraweak photons, known as biophotons, which are believed to play a role in intercellular communication and metabolic regulation. While the precise function of biophotons remains under investigation, some theories propose that they may influence the structure and function of the interstitial matrix, the connective tissue network that surrounds cells and plays a crucial role in tissue hydration, nutrient transport, and waste removal. It is conceivable that the unique properties of Fibranium, beyond FIR reflection, could interact with these subtle bioenergetic processes. The addition of specific minerals may further alter or amplify these effects, although the precise mechanisms and pathophysiological effects require further investigation.
The primary objective of this study was to evaluate the clinical efficacy of a Fibranium fiber mattress topper in reducing pain intensity compared to a placebo topper.
2. Methods
STUDY DESIGN & ETHICS
This was a prospective, randomized, double-blind, placebo-controlled trial conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. The study protocol received approval from the Carinthian Ethics Committee on January 30, 2024.
PARTICIPANTS:
Inclusion Criteria: Patients were recruited from a general practice and physiotherapy clinic in Klagenfurt, Austria (ImPuls Institute). Inclusion criteria were: (1) chronic low back pain (LBP) of at least 6 weeks duration; (2) current pain level of at least 5 on a 10-point Numerical Rating Scale (NRS); and (3) age between 40 and 70 years.
Exclusion Criteria: Exclusion criteria were: (1) previous osteosynthesis surgeries on the spine; (2) BMI ≥ 35; (3) continuous use of pain medication, especially use of pain medication more than 3 times per week; (4) acute indications for surgery; (5) chronic latent psychiatric illness; and (6) inability to use the product for the 4-week study duration (e.g. holidays, etc).
Recruitment: Patients were recruited from the practice of Dr. Martin Liebhauser (principal investigator) at ImPuls Institute Klagenfurt and through informational outreach to local physiotherapy and massage therapists. The recruitment of patients started on April 8, 2024 and was finished August 26, 2024.
Sample Size Justification: The sample size of 25 participants per group was chosen based on power calculations on the effect sizes of the prior study and a conservatively assumed effect size reduction, aiming at a power of 80% with an effect size of approximately standardized mean difference = 1.
INTERVENTION:
Fibranium Mattress Topper: The Fibranium mattress topper is composed of multiple fabric layers, one of which contains Fibranium fibers—a composite of silicon quartz and mineral nanoparticles that are fused and stabilized within a solid polyester structure. This innovative material is specifically processed for the textile industry to produce yarn and fibers. The exact formulation and manufacturing process remain proprietary to the manufacturer.
Control Group: The placebo topper was designed to be visually and texturally identical to the Fibranium topper, featuring the same fabric layers and construction but without the inclusion of Fibranium fibers. Thus, the two materials were indistinguishable from the outside and the study was easy to blind.
Intervention Protocol: Participants were instructed to place the assigned topper directly on top of their existing mattress and cover it with a thin bed sheet. They were instructed to sleep on the topper every night for a period of 4 weeks. To ensure adherence, participants received regular follow-ups and recorded their progress using an online self-monitoring sheet via Google Forms.
OUTCOME MEASURES:
Primary Outcome Measure: The primary outcome was the McGill Pain Questionnaire (MPQ) total score, a widely used and validated instrument for assessing the intensity and quality of pain. The MPQ provides a comprehensive assessment of both sensory and affective pain dimensions.
Secondary Outcome Measures: Secondary outcomes encompassed a range of measures to assess different aspects of the CLBP experience in addition to the MPQ:
- Oswestry Disability Index (ODI): The ODI is a self-report questionnaire that measures the impact of back pain on daily activities, such as lifting, walking, and sitting.
- Linton-Halldén Score: This questionnaire assesses psychosocial factors associated with back pain, including fear-avoidance beliefs and perceived disability.
- Pittsburgh Sleep Quality Index (PSQI): The PSQI is a self-rated questionnaire that evaluates sleep quality and disturbances over a one-month period.
- Short Form-36 (SF-36): The SF-36 is a widely used health survey that measures eight domains of health-related quality of life: physical functioning, role limitations due to physical health, role limitations due to emotional problems, energy/fatigue, emotional well-being, social functioning, pain, and general health.
- Symptom Score: Twenty pain-associated symptoms were evaluated using a verbal rating scale (0 = never/no symptom; 5 = always/severe symptoms). The individual variables were aggregated into a unidimensional score.
Data Collection Procedures: Data were collected at two time points: baseline (t0, prior to the intervention) and after 4 weeks of using the assigned mattress topper (t1). Assessments at each time point included a clinical examination by the principal investigator, completion of the questionnaires (administered digitally via Google Forms), and physiological measurements. Participants were also provided with a link to an online self-monitoring sheet via Google Forms to report any changes in their symptoms throughout the study period. This procedure allowed real-time tracking of participant experiences.
Randomization and Blinding: Randomization was performed using the web application www.randomizer.org. To ensure balanced group allocation, separate randomization lists were generated for men and women. A delegated person, not directly involved in patient care or data collection, accessed the randomization application and obtained a unique numerical ID for each enrolled participant. This ID corresponded to a pre-packaged mattress topper (either Fibranium or placebo) stored at the clinic. The allocation sequence was concealed from both the patients and the principal investigator, ensuring double-blinding. Blinding was upheld throughout the study, including during data collection and analysis.
Statistical Analysis: Statistical analyses were carried out using Statistica Version 13 (TIBCO Software Inc.). An intention-to-treat (ITT) analysis was performed, including all randomized participants in the analysis according to their assigned group, regardless of adherence to the protocol. Descriptive statistics (means, standard deviations, frequencies, percentages) were used to summarize baseline characteristics and outcome measures. The primary analysis employed linear models with baseline scores of the respective outcome measures as covariates to account for any initial differences between groups. For the primary outcome (MPQ total score), a linear model was fitted with the post-intervention (t1) MPQ score as the dependent variable, the treatment group (Fibranium or placebo) as the independent variable as a random factor, and the baseline (t0) MPQ score as a covariate. Similar linear models were used for the secondary outcome measures (ODI, Linton-Halldén Score, PSQI total score, SF-36 total score, and individual SF-36 subscales). For the PSQI, given the lack of a significant effect on the total score and the skewed distribution of the data, a Poisson regression was initially considered. However, due to limitations in model fit, and given that the primary interest was in comparing groups, a linear model was ultimately used for the PSQI total score, acknowledging the potential limitations. A separate linear model was used to analyze the PSQI component 2 (sleep latency), an essential parameter of sleep quality. Effect sizes were calculated (e.g., Cohen’s d, eta-squared) to quantify the magnitude of the treatment effects.
3. Results
A CONSORT flow chart illustrates the participant flow. Of 62 patients screened, 54 met the inclusion criteria and were randomized to either the Fibranium group (n=27) or the placebo group (n=27) between February 2024 and September 2024. All 54 randomized participants completed the 4-week intervention and follow-up assessments. No participants withdrew or were lost to follow-up, and all were included in the final analysis.
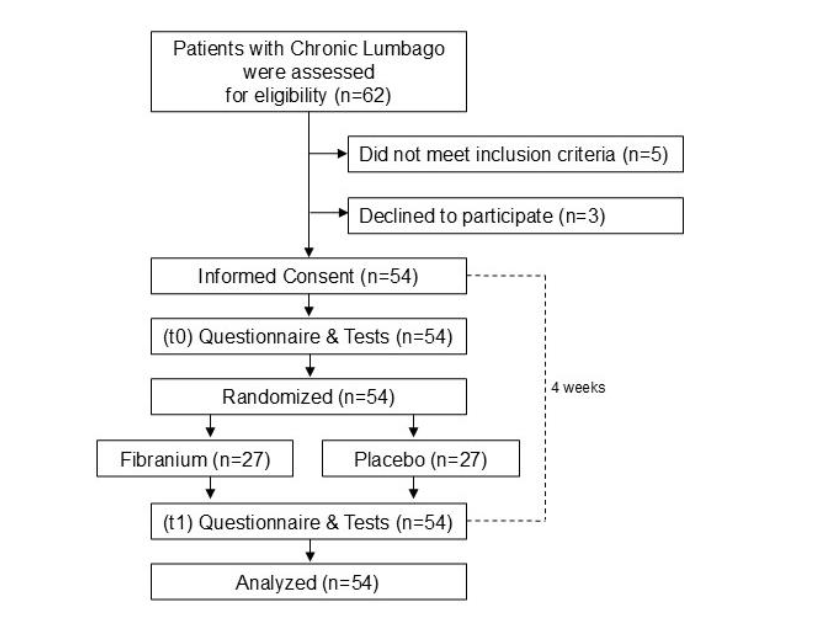
Table 1 presents the baseline demographic and clinical characteristics of the two groups, as well as the baseline scores of the outcome variables. The groups were well-matched on all baseline variables, including age, weight, height, duration of back pain, relationship with the doctor, and expectation of improvement. Table 2 presents the scores of the two groups after treatment, including the primary and secondary variables.
| Fibranium (n = 27) | Placebo (n = 27) | Total (n = 54) | |
|---|---|---|---|
| Gender (male) | 15 (55.6%) | 12 (44.4%) | 27 |
| Pain Duration (yrs) | 10.78 (9.3) | 10.44 (8.6) | 10.61 (8.8) [1-40] |
| Height (cm) | 175.44 (7.8) | 175.04 (8.0) | 175.24 (7.8) [159-193] |
| Weight (kg) | 79.37 (13.6) | 77.96 (14.4) | 78.67 (13.9) [52-120] |
| Age (yrs) | 52.92 (7.8) | 51.30 (6.7) | 52.11 (7.3) [41-79] |
| Relationship with Doctor | 3.07 (0.3) | 3.07 (0.3) | 3.07 (0.3) [3-4] |
| Expectation | 6.93 (2.4) | 7.0 (1.8) | 6.96 (2.1) [0-10] |
| Education | Basic 17 (63%) | 15 (55.6%) | 32 |
| A-Level 5 (18.5%) | 5 (18.5%) | 10 | |
| College 0 | 2 (7.4%) | 2 | |
| University 5 (18.5%) | 5 (18.5%) | 10 | |
| Smoker: | No 23 (85.2%) | 24 (88.9%) | 47 |
| 1-10 2 (3.7%) | 2 (3.7%) | 4 | |
| 11-20 2 (7.4%) | 1 (3.7%) | 3 | |
| >20 1 (3.7%) | 1 (3.7%) | 2 | |
| Alcohol | Never 5 (18.5%) | 5 (18.5%) | 10 |
| Rarely 9 (33.3%) | 11 (40.7%) | 20 | |
| Sometimes 13 (48.1%) | 10 (37%) | 23 | |
| Often 0 | 1 (3.7%) | 1 | |
| Coffee | Never 1 (3.7%) | 6 (22.2%) | 7 |
| Rarely 2 (7.4%) | 0 | 2 | |
| Sometimes 0 | 1 (3.7%) | 1 | |
| Often 11 (40.7%) | 1 (3.7%) | 12 | |
| Daily 13 (48.1%) | 19 (70.4%) | 32 | |
| Sport | Rarely 2 (7.4%) | 5 (18.5%) | 7 |
| Sometimes 11 (40.7%) | 7 (26%) | 18 | |
| Often 11 (40.7%) | 11 (40.7%) | 22 | |
| Daily 3 (11.1%) | 4 (14.8%) | 7 | |
| McGill Pain Total | 23.67 (12.8) | 24.15 (9.6) | 23.91 (11.2) [8-50] |
| SF 36 | Physical Functioning 75.92 (23.2) | 80.37 (15.1) | 78.15 (19.5) [15-100] |
| Role Physical | 73.15 (39.2) | 55.56 (39.4) | 64.35 (39.9) [0-100] |
| Role Emotional | 90.12 (29.0) | 79.01 (38.3) | 84.57 (34.1) [0-100] |
| Energy | 52.78 (10.0) | 49.07 (9.6) | 50.93 (9.9) [30-75] |
| Emotional Wellbeing | 44.59 (7.5) | 41.04 (8.8) | 42.81 (8.3) [24-68] |
| Social Functioning | 80.09 (20.0) | 75.0 (20.2) | 77.55 (20.1) [25-100] |
| Pain | 51.85 (23.7) | 55.28 (21.6) | 53.56 (22.5) [12.5-100] |
| General Health | 63.70 (17.1) | 65.18 (18.6) | 64.44 (17.7) [15-95] |
| Sum Average | 66.53 (13.7) | 62.56 (14.4) | 64.54 (14.0) [32.6-84.4] |
| Pittsburgh Sleep Total | 6.92 (3.7) | 7.11 (3.8) | 7.02 (3.7) [2-19] |
| Oswestry Total | 9.30 (6.9) | 9.78 (4.8) | 9.54 (5.9) [1-24] |
| Linden-Hallden Total | 79.22 (26.7) | 78.11 (27.7) | 78.67 (27.0) [28-141] |
| Symptoms Sum Score | 29.70 (18.9) | 29.07 (12.2) | 29.39 (15.7) [7-87] |
The Fibranium group experienced significantly greater improvements across multiple outcome measures compared to the placebo group (Table 2). This included a highly significant reduction in the primary outcome, the McGill Pain Questionnaire (MPQ) total score, as well as significant improvements in the Oswestry Disability Index (ODI) and the Linton-Halldén Score, indicating reduced disability and improved psychosocial factors, respectively. Although the overall Pittsburgh Sleep Quality Index (PSQI) score did not differ significantly, sleep latency showed a significant improvement in the Fibranium group. Furthermore, the Fibranium group demonstrated significantly better scores on the overall Short Form-36 (SF-36), and all of its subscales except for Emotional Wellbeing, signifying a broad improvement in health-related quality of life.
| Outcome Measure | Fibranium (n=27) | Placebo (n=27) | p-value for group factor | Effect Size partial η2 | |
|---|---|---|---|---|---|
| McGill Total Score | 10.43 (7.95, 12.91) | 19.94 (17.46, 22.42) | 0.000002 | 0.37 | |
| Oswestry Disability Index | 4.47 (2.93, 6.01) | 10.01 (8.48, 11.55) | 0.000005 | 0.34 | |
| Linton-Halldén Score | 53.83 (47.71, 59.94) | 74.91 (68.80, 81.03) | 0.000010 | 0.32 | |
| PSQI Total Score | 4.55 (3.22, 5.89) | 5.33 (4.20, 6.47) | 0.187 | 0.03 | |
| PSQI Sleep Latency | 1.02 (0.78, 1.26) | 1.85 (1.61, 2.09) | 0.000175 | 0.24 | |
| SF-36 Total Score | 77.44 (73.94, 80.94) | 63.19 (59.69, 66.69) | 0.000001 | 0.39 | |
| SF-36 Subscales | Physical Functioning | 94.4 | 86.7 | 0.006 | 0.16 |
| Role Physical | 93.1 | 72.6 | 0.02 | 0.12 | |
| Role Emotional | 94.1 | 75.1 | 0.05 | 0.09 | |
| Energy | 50.3 | 43.6 | 0.02 | 0.12 | |
| Emotional Wellbeing | 38.2 | 39.8 | 0.4 | 0.02 | |
| Social Functioning | 92.7 | 70.3 | 0.00002 | 0.34 | |
| Pain | 81.5 | 55.0 | 0.00005 | 0.31 | |
| General Health | 76.1 | 61.5 | 0.00007 | 0.31 | |
| Symptoms Sum Score | 17.73 (13.98, 21.47) | 28.34 (21.87, 24.64) | 0.000001 | 0.24 |
No adverse events or side effects related to the use of either the Fibranium or placebo mattress toppers were reported during the study.
4. Discussion
SUMMARY OF FINDINGS:
This randomized, double-blind, placebo-controlled trial provides compelling evidence that a mattress topper containing Fibranium fibers is effective in reducing pain and improving function in individuals with chronic low back pain. The Fibranium group demonstrated statistically significant improvements in the primary outcome measure (MPQ total score) and in a majority of the secondary outcome measures, including the Oswestry Disability Index, Linton-Halldén Score, and the overall SF-36 score, as well as most of the SF-36 sub-domains.
INTERPRETATION OF RESULTS:
Improvements across multiple independent measures suggest that Fibranium mattress toppers could serve as an effective treatment option for chronic low back pain. The magnitude of the observed improvements in the Fibranium group is clinically meaningful, particularly given the chronic nature of the participants’ pain and the often-limited success of existing treatments. Statistical effect sizes are large for all outcome variables except for the PSQI total score. These findings align with and expand upon our previous research on the Fibrilium fiber. Notably, the effect sizes observed in the current study with Fibranium appear to be even larger than those seen with Fibrilium for several key outcomes, suggesting that the modifications incorporated into the Fibranium fiber (additional crystalline minerals and altered manufacturing process) may have enhanced its therapeutic properties.
MECHANISMS OF ACTION:
Although the precise mechanisms of Fibranium’s effects remain unclear, it is hypothesized to primarily enhance the reflection of far-infrared radiation (FIR) naturally emitted by the body. FIR is known to penetrate tissues and has been associated with various physiological benefits, including enhanced local blood circulation, reduced inflammation, and modulation of pain signaling pathways. Additionally, biophotons may play a role in cellular communication, metabolic regulation, and pain modulation. Biophotons are also hypothesized to contribute to axonal growth and thereby to be the mechanistic principle for neuroplasticity. This would be a potential rationale for modifying pain memory, which is crucial in chronic pain processes. Furthermore, the interstitial matrix, a dynamic, interconnected network of connective tissue, is increasingly recognized as a key regulator of physiological processes, influencing tissue mechanics, cellular signalling, and overall homeostasis. Through vagal innervation of the interstitial matrix peripheral effects such as those from FIR can reach central pathways and modulate pain afferents, similar to principles that have been shown for antinociceptive effects of acupuncture. The integration of specific crystalline minerals into Fibranium fibers is believed to enhance FIR reflection and may contribute to additional biophysical effects. First basic research results have shown this to be true. However, further research is needed to fully elucidate these potential mechanisms.
Mattress composition has been shown to play a role in back pain treatment, and medium hard mattresses are considered best. We did not advise patients on this aspect, and the mattress top offered in this study did not have any influence on the hardness of the mattress, as it was only comparatively thin, like a bed sheet. Infrared therapy for pain treatment has been researched in various studies and shown good effects, supporting the rationale of Fibranium. These other interventions, however, are either contingent on devices, which need to be implemented, worn, applied or, like sauna, require some other activity. The benefit of the Fibranium fibre is its non-invasive character which does not require any additional time, energy source, or effort on part of the patients.
STRENGTHS AND LIMITATIONS:
This study has several strengths, including a randomized, double-blind, placebo-controlled design, which minimizes the risk of bias, a well-defined study population, the use of validated outcome measures, and a high completion rate. However, a limitation is that the population has been changed compared to the initial study, including only people aged >40 years, excluding those with a BMI >35 and with a high intake of pain killers. The results may therefore not be generalizable to all patients with chronic low back pain. The sample size was sufficient, and statistical power high, due to the large clinical effects. However, for broader generalizability larger studies with more diverse patients, or different pain syndromes might be advisable. A critical reader might point out that we have calculated multiple linear models, in addition to analyzing the main outcome, MPQ, altogether six for the secondary outcomes, in addition to the main model. As we had a clearly defined primary outcome, we do not see this as a problem, especially as the statistical significance was so high that it would survive correction for another 100 multiple tests. All secondary outcomes, excluding the PSQI total score, showed clear and significant results, supporting our hypothesis. The effect sizes were all large and clinically meaningful. The lack of significant effect for the Pittsburgh Sleep Inventory Total score was likely due to a floor effect: Participants in this study did not have clinically meaningful problems with their sleep. We analyzed the component 2, sleep latency, especially, because informal feedback of the patients regarding better sleep had alerted us to this possibility. No other subgroup analyses not reported here have been conducted.
The significant improvement in sleep latency (PSQI component 2) observed in the Fibranium group, despite the lack of a significant effect on the overall PSQI score, is intriguing. This finding suggests that Fibranium may exert some influence on sleep regulation. Potential mechanisms could involve improved thermal comfort during sleep due to FIR reflection, or modulation of autonomic nervous system activity.
CLINICAL IMPLICATIONS:
The Fibranium mattress topper offers a promising non-pharmacological and non-invasive approach for managing CLBP. Its ease of use, lack of reported adverse effects, and demonstrated efficacy across multiple outcome measures make it a potentially valuable addition to the treatment armamentarium for this common and often-challenging condition.
FUTURE RESEARCH:
Further research is warranted to build upon these findings. Future studies should:
- Investigate the long-term effects of Fibranium use in CLBP, with follow-up assessments beyond 4 weeks.
- Explore the efficacy of Fibranium in other pain conditions, such as fibromyalgia, osteoarthritis or menstruation related pain.
- Conduct laboratory studies to more precisely elucidate the physiological effects of Fibranium, including investigations of its impact on inflammatory markers, tissue perfusion, and neural activity.
- Examine the potential synergistic effects of combining Fibranium with other non-pharmacological interventions, such as exercise or cognitive-behavioral therapy.
- Further investigate the effects of Fibranium on sleep using more objective measures, such as polysomnography, to better understand its impact on sleep architecture and regulation.
- Consider larger and more diverse populations to improve generalizability.
Conclusion:
This study provides robust evidence that a Fibranium fiber mattress topper is a safe and effective intervention for reducing pain and enhancing function in individuals with chronic low back pain. The Fibranium topper offers a promising non-pharmacological option for managing this prevalent and debilitating condition. The observed improvements in pain, disability, and quality of life highlight its potential clinical utility.
References
- Global Burden of Disease Collaborative Network. Global Burden of Disease Study (GBD 2019) Results. Seattle, United States: Institute for Health Metrics and Evaluation (IHME);2020.
- Qaseem A, Wilt TJ, McLean RM, Forciea M, Physicians; ftCGCotACo. Noninvasive treatments for acute, subacute, and chronic low back pain: A clinical practice guideline from the american college of physicians. Annals of Internal Medicine. 2017;166(7):514-530.
- Savigny P, Kuntze S, Watson P, et al. Low Back Pain: early management of persistent non-specific low back pain. London: National Collaborating Centre for Primary Care and Royal College of General Practitioners; 2009.
- Chou R, Deyo R, Friedly J, et al. Nonpharmacologic Therapies for Low Back Pain: A Systematic Review for an American College of Physicians Clinical Practice Guideline. Ann Intern Med. 2017;166(7):493-505.
- Ofner M, Treven M, Walach H. A new nano-fibre mat has a strong benefit for chronic low-back pain patients – Results of a double-blind randomised trial. Clinical Research and Trials. 2019;5:1-6.
- Tonelli MR. Optische Kennzeichnung der Proben von Cafissi in dem Spektralbereich von 4-14 um [Optical characterisation of probes of “cafissi” in the spectral range of 4 – 14 micrometer]. Pisa: CNRNano: Istituto Nanoscienze – Consiglio Nazionale di Ricerche; 2013.
- Tsai S-R, Hamblin MR. Biological effects and medical applications of infrared radiation. Journal of Photochemistry and Photobiology B: Biology. 2017;170:197-207.
- Vatansever F, Hamblin MR. Far infrared radiation (FIR): Its biological effects and medical applications. Photonics & Lasers in Medicine. 2012;1(4):255-266.
- Hammerschlag R, Levin M, McCraty R, et al. Biofield physiology: A framework for an emerging discipline. Global Advances in Health and Medicine. 2015;4(suppl):35-41.
- Ives JA, van Wijk EPA, Bat N, et al. Ultraweak Photon Emission as a Non-Invasive Health Assessment: A Systematic Review. PLoS ONE. 2014;9(2):e87401.
- van Wijk R, van Wijk EPA, Wiegant FAC, Ives J. Free radicals and low-level photon emission in human pathogenesis: State of the art. Indian Journal of Experimental Biology. 2008;46:273-309.
- Popp FA. Biophoton emission. Experientia. 1988;44:443-444.
- Zarkeshian P, Kergan T, Ghobadi R, Wilten N, Simon C. Photons guided by axons may enable backpropagation-based learning in the brain. . 2022;12(1):20720. PMID: 36456619. Scientific Reports. 2022;12(1):20720.
- Pollack GH. The Fourth Phase of Water: Beyond Solid, Liquid, and Vapour. Seattle: Ebner & Sons; 2013.
- Bordoni B, Marelli F. Emotions in motion: myofascial interoception. Complementary Medicine Research. 2017;24:110-113.
- Langevin HM, Keely P, Mao J, et al. Connecting (t)issues: How research in fascia biology can impact integrative oncology. Cancer Research. 2016;76(21):6159-6162.
- Tozzi P. A unifying neuro-fasciagenic model of somatic dysfunction – underlying mechanisms and treatment – part I. Journal of Bodywork and Movement Therapies. 2015;19:310-326.
- Tozzi P. A unifying neuro-fasciagenic model of somatic dysfunction – underlying mechanisms and treatment – Part 2. Journal of Bodywork and Movement Therapies. 2015;19:526-543.
- Ofner M, Walach H. The Vegetative Receptor-Vascular Reflex (VRVR) – A New Key to Regeneration. Frontiers in Physiology – Autonomic Neuroscience. 2020;11:547526.
- Dworkin RH, Turk DC, Farrar JT, et al. Core outcome measures for chronic pain clinical trials: IMMPACT recommendations. Pain. 2005;113:9-19.
- Kiss I, Müller H, Abel M. The McGill Pain Questionnaire – German version. A study on cancer pain. Pain. 1987;29(2):195-207.
- Melzack R, ed Pain. Measurement and Assessment. New York: Raven Press; 1983.
- Mannion AF, Junge A, Fairbank JC, Dvorak J, Grob D. Development of a German version of the Oswestry Disability Index. Part 1: cross-cultural adaptation, reliability, and validity. European Spine Journal. 2006;15:55-65.
- Fairbank J, Davies J, Couper J, al. e. The Oswestry low back pain disability questionnaire. Physiotherapy. 1980;66:271-273.
- Linton SJ, Halldén K. Can we screen for problematic back pain? A screening questionnaire for acute and subacute back pain. Clinical Journal of Pain. 1998;14(3):209-215.
- Backhaus J, Junghanns K, Broocks A, Riemann D, Hohagen F. Test-retest reliability and validity of the Pittsburgh Sleep Quality Index in primary insomnia. Journal of Psychosomatic Research. 2002;53(3):737-740.
- Buysse D, Reynolds C, Monk T, Berman S, Kupfer D. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Research. 1989;28:193-213.
- Bullinger M, Kirchberger I. Der SF-36 Fragebogen zum Gesundheitszustand (SF-36). Handbuch für die deutschsprachige Fragebogenversion. Göttingen: Hogrefe; 1998.
- Popp F-A. Biophotons – Background, experimental results, theoretical approach and applications. Frontier Perspectives. 2002;11(1):16-28.
- Sandkühler J. Learning and memory in pain pathways. Pain. 2000;88:113-118.
- Bordoni B, Marelli F, Morabito B, Castagna R, Sacconi B, Mazzucco P. New proposal to define the fascial system. Complementary Medicine Research. 2018;25:257-262.
- Schleip R, Findley TW, Chaitow L, Huijing PA, eds. Fascia: The Tensional Network of the Human Body. London: Churchill Livingstone/Elsevier; 2012.
- Schleip R. Fascia as an organ of communication. In: Schleip R, Findley TW, Chaitow L, Huijing PA, eds. Fascia: The Tensional Network of the Human Body. London: Churchill Livingstone; 2012:77-79.
- Pomeranz B. Acupuncture analgesia – basic research. In: Stux G, Hammerschlag R, eds. Clinical Acupuncture. Scientific Basis. Heidelberg: Springer; 2001:1-28.
- Bergholdt K, Fabricius RN, Bendix T. Better backs by better beds? Spine (Phila Pa 1976). 2008;33(7):703-708.
- Kovacs FM, Abraira V, Peña A, et al. Effect of firmness of mattress on chronic non-specific low-back pain: randomised, double-blind, controlled, multicentre trial. The Lancet. 2003;362(9396):1599-1604.
- Alfano AP, Taylor AG, Foresman PA, et al. Static magnetic fields for treatment of fibromyalgia: A randomized controlled trial. Journal of Alternative and Complementary Medicine. 2001;7:53-64.
- Caggiari G, Talesa GR, Toro G, Jannelli E, Monteleone G, Puddu L. What type of mattress should be chosen to avoid back pain and improve sleep quality? Review of the literature. Journal of orthopaedics and traumatology: official journal of the Italian Society of Orthopaedics and Traumatology. 2021;22(1):51.
- Ervolino F, Gazze R. Far infrared wavelength treatment for low back pain: Evaluation of a non-invasive device. Work (Reading, Mass). 2015;53(1):157-162.
- Oosterveld FG, Rasker JJ, Floors M, et al. Infrared sauna in patients with rheumatoid arthritis and ankylosing spondylitis. A pilot study showing good tolerance, short-term improvement of pain and stiffness, and a trend towards long-term beneficial effects. Clin Rheumatol. 2009;28(1):29-34.
- York RM, Gordon IL. Effect of optically modified polyethylene terephthalate fiber socks on chronic foot pain. BMC Complement Altern Med. 2009;9:10.
- Gale GD, Rothbart PJ, Li Y. Infrared therapy for chronic low back pain: a randomized, controlled trial. Pain research & management. 2006;11(3):193-196.
- Masuda A, Koga Y, Hattanmaru M, Minagoe S, Tei C. The effects of repeated thermal therapy for patients with chronic pain. Psychother Psychosom. 2005;74(5):288-294.
“`

