Efficacy of Short Splint in Zone II Tendon Rehabilitation
Efficacy of the Short Splint versus traditional dorsal splint for rehabilitation of Zone II flexor tendon repairs: study protocol for a randomized controlled trial
Vivien Lees¹˒², Karen Redvers-Chubb¹, Roxana Moscalu¹, Mihaela Moscalu³, Lauren Taylor¹
- Manchester University Hospitals Foundation Trust, Manchester, United Kingdom
- University of Manchester, United Kingdom
- Department of Preventative Medicine and Interdisciplinarity, Grigore T. Popa University of Medicine and Pharmacy, Romania
Abstract
Introduction: Flexor tendon rehabilitation in zone II remains challenging due to the risk of complications such as tendon adhesions and rupture. A previously reported audit of the Manchester Short Splint demonstrated less flexion contracture at the proximal interphalangeal joints at 6 and 12 weeks with greater final range of motion compared with the traditional dorsal forearm splint, without increased tendon rupture. The present study describes the protocol for a randomized controlled trial designed to formally evaluate the short splint.
Methods: This single center randomized controlled trial allocates adults with a single Zone II flexor tendon repair to either a traditional dorsal forearm splint or the Manchester Short Splint. The study is framed to demonstrate superiority of the short splint intervention over the long splint. All tendon repairs use a standard four-strand core repair and epitendinous suture technique. Inclusion criteria for the trial include single finger flexor tendon injury of zone II with up to one but not two simultaneous digital nerve injuries operated on within 4 days of the injury date. Exclusions include children, more complex injuries, those unable to give consent or otherwise co-operate with the trial protocol. Outcomes are assessed at 6 and 12 weeks by a hand therapist blinded to allocation. The primary outcome is total active motion measured with a goniometer. Secondary outcomes include patient-reported measures (Patient Evaluation Measure, Disabilities of the Arm, Shoulder and Hand questionnaire, numerical pain scale) and grip strength at 12 weeks.
Discussion: The study protocol presented aims to evaluate the efficacy of the Manchester Short Splint compared with the traditional dorsal splint in the rehabilitation of Zone II flexor tendon repairs. Earlier observational work suggested potential benefit, but a rigorously designed randomized controlled trial is required to formally assess whether the short splint improves motion without increasing rupture risk. If superiority of the short splint is demonstrated, the findings could support a change in clinical rehabilitation practice and inform the design of a future multicenter trial.
Trial registration: ClinicalTrials.gov Identifier: NCT03850210 21.2.19 Retrospectively registered https://clinicaltrials.gov/ct2/results?cond=&term=NCT03850210&cntry=&state=&city=&dist= See website for WHO Trial Registration Data Set
Keywords:
Short Splint, zone II, flexor tendon repair, rehabilitation, regimen, therapy regime, TAM, PEM
Introduction
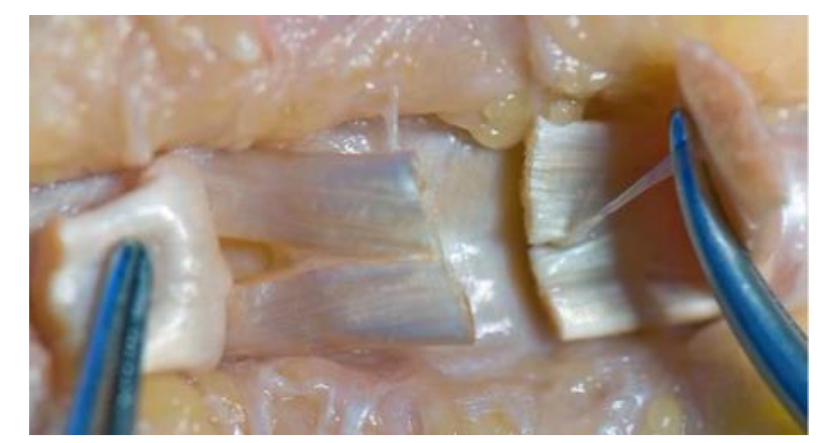
Rehabilitation following flexor tendon repair in zone II remains a recognized challenge. This region often termed no-man’s-land contains both the flexor digitorum profundus (FDP) and superficialis (FDS) tendons enclosed within a narrow fibro-osseous sheath, making it particularly prone to adhesion formation and restricted glide even with minor scar tissue formation (Figure 1). Traditional hand therapy involves immobilization of the wrist in a long dorsal forearm-based splint for six weeks to protect the repair. Wrist immobilisation, however, can reduce the tenodesis effect and limit passive digital extension, contributing to joint contracture and tendon adhesion. Advances in multistrand tendon repair techniques have improved repair strength, creating an opportunity to reconsider safe levels of early motion that may be safely permitted during rehabilitation.
The traditional long dorsal thermoplastic splint immobilizes the wrist in neutral position with a block to 30° of metacarpophalangeal (MCP) joint extension. In contrast, the Manchester Short Splint extends from the proximal wrist crease to the fingertips, permitting maximal wrist flexion and up to 45° of wrist extension, while maintaining a block to 30° of MCP joint extension. A previous observational audit from our center suggested that the short splint resulted in less flexion contracture and greater range of movement at six and twelve weeks, with no increase in rupture rate. These findings require validation in a rigorously designed trial.
This manuscript presents the protocol for that randomized controlled trial. The hypothesis is that the short splint is superior to the traditional long dorsal splint in the rehabilitation of zone II flexor tendon repairs, assessed using total active motion (TAM) at six and twelve weeks. Secondary hypotheses are that the short splint will result in better patient-reported outcomes measured using the Patient Evaluation Measure (Figure 2) and the Disabilities of the Arm, Shoulder and Hand questionnaire and improved grip strength at twelve weeks. A further secondary hypothesis is that the short splint is non-inferior to the traditional splint with respect to tendon rupture. The intention is that the protocol described here will inform a subsequent multicenter study.

Methods/Design
STUDY DESIGN AND SETTING
This is a single-center, single-blind, randomized controlled trial comparing two rehabilitation splints following Zone II flexor tendon repair. Patients and treating therapists are aware of allocation; therapists assessing outcome remain blinded.
PRIMARY AND SECONDARY OUTCOMES
The primary endpoint is TAM measured by goniometer at six weeks and twelve weeks (assessed separately). The secondary endpoints comprise patient reported outcomes at six and twelve weeks, and grip strength measurement at twelve weeks. It is also important to establish that the short splint is not worse than the long splint in terms of rupture rates. As rupture is a relatively rare event (4-6% incidence in recent reported series), it would require a substantial sample size to power the comparison, and it is therefore not a primary endpoint. We envisage a narrative description that would reflect on the rupture rate within the context of published norms.
PARTICIPANTS
Adults (≥ 16 years) with a single Zone II flexor tendon repair performed within 96 hours of injury are eligible. Detailed inclusion and exclusion criteria are shown in Table 1.
| INCLUSION CRITERIA | EXCLUSION CRITERIA |
|---|---|
| Adult patient (16 yrs or older) | Children under 16 yrs of age |
| Single Zone II flexor tendon division with or without FDS division. Includes single nerve injury on same digit. | Patients with special needs and vulnerable groups |
| Time interval of injury to operation <96 hours (4 days) | Patients lacking capacity or motivation to participate in planned physiotherapy |
| Patients unable to attend physiotherapy for requisite number of sessions for social and economic reasons | Patients unable to understand English sufficient to complete rehabilitation |
| Flexor tendon injury greater than 4 days previous | Two digital nerve injuries in the same finger as the flexor tendon injury |
| More than one finger with FDP injury | Multilevel FDP injury in one finger |
| Complex injury with soft tissue defect | Complex injury with underlying fracture |
PROCESSES, INTERVENTIONS AND COMPARISON
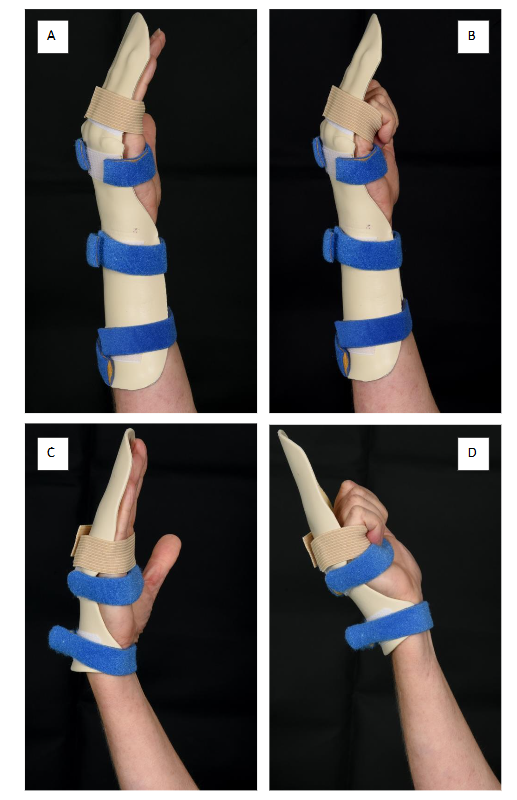
The specific intervention is to test the use of a short splint against the traditional long dorsal forearm splint in zone II flexor tendon repair rehabilitation. Figure 3 illustrates the two splint designs: the traditional long dorsal splint (Fig. 3A and 3B) and the short splint (Fig. 3C and 3D).
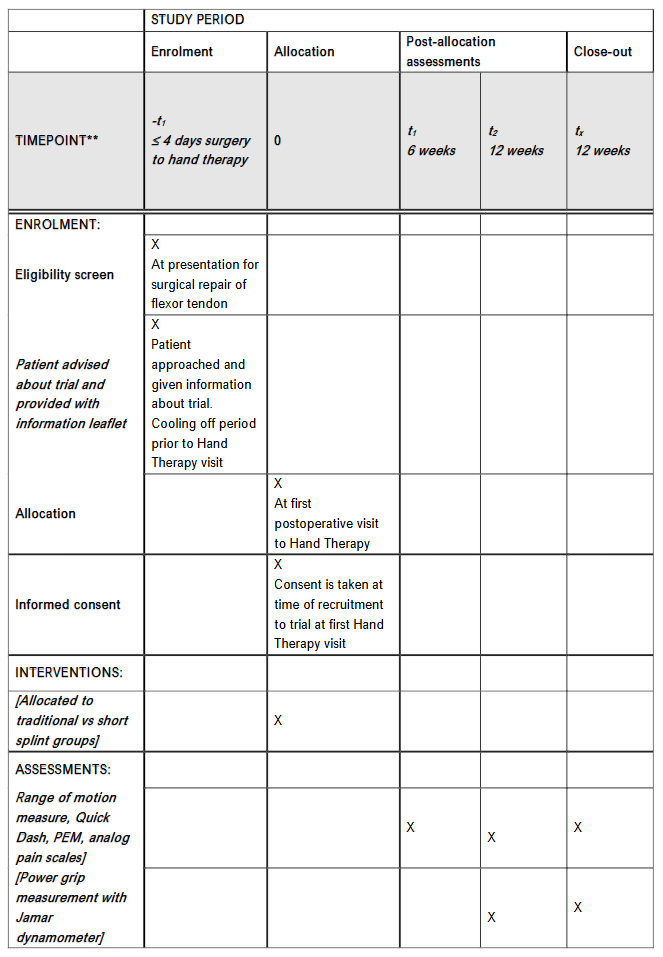
The study timelines and assessment schedule are detailed in the SPIRIT diagram (Figure 4). The rehabilitation protocol modifies the traditional exercise program to engage controlled wrist movement and harness the tenodesis effect, which promotes passive interphalangeal joint extension. Detailed measurements are taken at six and twelve weeks by an assessing therapist who remains blinded to the splint type used. All splints are discontinued at six weeks, and the number and frequency of therapy visits are identical in both groups.
Patients are counselled about the study during their surgical admission by a site investigator. Those willing to participate are provided with a patient information leaflet. Formal recruitment and consent take place at the first postoperative attendance in the hand therapy department, where a site-investigator therapist confirms eligibility, answers questions, and completes written informed consent.
Surgery is performed according to the department’s standard protocol using a four-strand core suture (3/0 Prolene) and a 5/0 epitendinous repair, with venting of the pulleys as required. The trial evaluates only the postoperative rehabilitation phase.
Patients are typically reviewed by hand therapy 4-5 days after surgery, allowing time to consider participation. Randomization occurs at this first therapy appointment. Block randomization is used to ensure equal numbers in each arm (block size = 4). Allocation sequences are computer-generated using randomization software (www.randomizr.org) and coded to maintain allocation concealment. Site investigators remain unaware of upcoming allocations to maintain equipoise and minimize recruitment bias.
At both six- and twelve-week assessments, splints are removed by the treating therapist prior to review by the blinded assessing therapist. Patients are instructed not to disclose their splint type. The treating therapist and patient are aware of allocation; therefore, this is a single-blind study. Patients who decline participation receive standard long dorsal splinting. All therapy is delivered by experienced hand therapists. Participants who withdraw or lose capacity are removed from the study. In practice, this means that patients in the short-splint group revert to traditional splinting if they withdraw during the first six weeks; long-splint participants continue standard care but are excluded from study-specific assessments.
All patients attend weekly therapy sessions for the first six weeks. Following splint removal at six weeks, all patients follow the same exercise, stretching and night-splint regimen for residual contracture, with return to unrestricted activity at twelve weeks. Both groups follow an early combined passive flexion and active motion program, with the exercise sequence emphasizing full passive flexion stretches to maximize passive digital motion prior to commencing active motion. Active flexion exercises are initiated from the distal interphalangeal (DIP) joint to maximize differential glide, avoiding excessive end-range effort. Active digital extension exercises are encouraged to reduce flexion contractures. In Group A (long dorsal splint) exercises are performed with the wrist in neutral and MCP joints blocked at 30° flexion. In Group B (short splint) exercises are performed with the wrist extended up to 45° during flexion and maximally flexed during digital extension, maintaining 30° MCP flexion. Palmar thermoplastic finger splints are provided at night for any patient with incomplete interphalangeal joint extension. All participants are instructed on light functional hand use (excluding the injured digit) and to wear the study splint continuously for six weeks.
Assessments at six and twelve weeks are performed by the blinded therapist, who is one of the site investigators. The primary outcome is the TAM of the PIP and DIP joint of the affected finger. Secondary outcome measures comprise Visual Analogue Scale for pain score (from no pain to very severe pain), PEM, Quick DASH patient questionnaire at six and twelve weeks, and grip strength at 12 weeks. Tendon rupture is an additional secondary outcome. All data and adverse events are recorded in Case Report Forms (CRFs). Table 2 outlines the assessments, investigations, and interventions undertaken by the hand therapy team, highlighting which activities exceed standard care. The assessment by the site investigator hand therapist includes:
- Quick DASH, PEM
- pain scales (Numerical Analogue Scale 0-10)
- range of motion of injured digit proximal and distal interphalangeal joint range of motion using digital goniometer
- grip strength at 12 weeks using Jamar dynamometer on 2nd setting (power grip is not tested earlier to avoid potential tendon rupture)
| Details of assessments and interventions undertaken by the hand therapists |
|---|
| First postoperative visit (within 5 days of flexor tendon repair operation) |
| *Recruitment into trial with counselling regarding same by the investigating hand therapist. Trial consent form is completed, and patient is randomized to short splint (intervention) or long dorsal splint (control) |
| Fashioning of thermoplastic splint. Advice to patient on exercises to be undertaken including safe use of the hand. |
| Patient reviewed weekly thereafter at 2,3,4, and 5 weeks as part of standard care by treatment hand therapist. During visit the patient is further advised and undertakes supervised exercises by the treating hand therapist. Integrity and glide of tendon repair are assessed. Same for trial and non-trial patients. |
| *Trial patients – any adverse events are reported in CRF having been notified to a site investigator |
| 6-week review. Splint is discontinued and removed by the treating hand therapist. Advice is given on driving and return to normal activities of daily living and sporting and recreational activities. The treating hand therapist then hands over to the assessing hand therapist (site investigator) who is ‘blinded’ as to which splint has been used. The assessing hand therapist is a different one from the therapist who recruited the patient initially to the trial. Assessing hand therapist measures range of motion of the affected finger is made and recorded using goniometer. Also measures contralateral finger range of motion to allow calculation of the TAM as a percentage of what is normal for that patient. Integrity and glide of tendon repair are assessed. Data is recorded. |
| *Trial patients have their measurements performed by an assessing hand therapist after removal of the splint (single blind protocol). Any adverse events are reported in CRF. In addition, the PEM, Quick DASH and VAS PROMs scales are administered and recorded in the CRF. |
| 12-week review. Patients who attend will have their range of motion and grip strength recorded using Jamar dynamometer on the second setting (mean of 3 separate recordings of grip). Integrity and glide of tendon repair are assessed. Data is recorded. |
| *Trial patients have their measurements performed by an assessing hand therapist (single blind protocol). Any adverse events are reported in CRF. In addition the PEM, Quick DASH and VAS PROMs scales are administered and recorded in the CRF. |
Adverse events (AEs) are recorded in participants CRFs and managed according to Good Clinical Practice (GCP) standards. The treating therapist notifies a site-investigator therapist, who assigns causality. Serious AEs are reported within 24 hours to the Principal Investigator and trial sponsor. Any trial participant suffering a serious adverse event will be followed up and onward referral to an appropriate clinician made, as clinically indicated. Potential Serious Adverse Device Effects (SADEs), both anticipated (SADE) or unanticipated (USADE) may occur. Because both splint designs use identical materials, a biological adverse reaction (SUSAR) is not expected. Surgical wound infection would be classified as unrelated to device configuration. Tendon ruptures would be categorized as SADEs but affected patients would remain in the study for data capture. Tenolysis procedures would fall outside the twelve-week study period and will therefore not be reported within the timeline of the trial. Any additional operations within the trial window are recorded in the CRF.
TRIAL MANAGEMENT
The trial team meets regularly to review study conduct, recruitment, and compliance with GCP. The Principal Investigator (PI) is responsible for all ethics submissions, amendments, and progress reports. Approved amendments are circulated to site investigators and the Clinical Trials Management Team (CTMT). An independent reviewer from the CTMT performs annual on-site audits of compliance and documentation. The investigating team are scientifically independent of the sponsor (the Trust). Interim analyses and stopping guidelines are overseen by the trials team and PI with statistical advice. Recruitment is supported through ongoing education of new surgical staff as site investigators. Potential participants are identified at the daily trauma meeting, assisted by the research nurse and trauma coordinator. Reasons for non-recruitment are reviewed regularly. Continuity of care within the hand therapy department supports participant retention. Non-attenders are contacted once to offer a rescheduled appointment.
Trial ethical approval has been obtained from the Research Ethics Committee 14/NW/1486 IRAS project ID 159054. The Trust as sponsor is acting for ancillary and post-trial care as the need arises.
STATISTICAL ANALYSIS AND POWER CALCULATION
The statistical analysis of the variables of interest will be carried out in SPSS v.29. For continuous variables, we will assess the averages and standard deviation or the medians, depending on the normal distribution of the values.
Regarding the primary analysis of TAM, this will be conducted by comparing the two patient groups and the given timepoints (6 and 12 weeks). Thus, different versions of the t-test will be applied, including independent samples t-tests and paired t-test.
Sample size calculation and power analysis will be determined based on the effect size, statistical power (1-β), and type I error rate (α). The effect size is defined as the minimum detectable difference in TAM values (between groups), expressed as a mean difference with standard deviation (mean: 20; SD: 30). The significance level (α) was set at <0.05, while power (1-β) was set at >0.9 (90%). The proportion of subjects assigned to Group 1 will be 0.5 (50%), and to Group 2 will also be 0.5 (50%). Therefore, based on the formula N = (n1 n2 +1)∙(Zα/2 +Z1−β)2∙σ2/n1 n2∙d2, we obtained a minimum number of 48 patients in group 1 and 48 in group 2, respectively. For qualitative variables, we will analyze frequencies (absolute and relative %) and will perform comparisons between groups based on the results of non-parametric tests (Fisher’s exact test as appropriate).
The value of the significance level p < 0.05 will confirm the alternative hypothesis (H1).
INTERIM ANALYSIS
An interim analysis will not be carried out, as this is a small-scale exploratory trial to investigate the effectiveness of the new short splint. Early stopping for futility is unlikely as the new splint did not appear to perform worse than the control splint in our previous audit (Peck et al, 2014). There will be no issue of multiple testing as there are not multiple primary outcomes. On the point of potential missing data, the sample size has been increased to account for this.
DATA MANAGEMENT
The study complies with Caldicott Principles and GDPR. Participants are recorded on the Local Portfolio Management System and R-PEAK using anonymized study IDs. Raw data are entered manually and transcribed into an encrypted electronic database stored on Trust computers. Hard-copy CRFs are maintained securely on site. The raw data transcription into electronic format will be undertaken by a member of the hand therapy team. Primary data will be processed on Trust computers by the authors and secondary data transported without patient identifiers on encrypted pen drives. Processed anonymised data will be stored on secure personal devices of the investigators for analysis, consistent with institutional policy.
Trial data management is subject to regular audit led by the Clinical Trials Management team of the Trust. The auditor is independent of the trial investigating team. Trial documentation is audited annually by an independent CTMT reviewer. Processed data are retained for ten years and available on reasonable request from the PI. The full participant data will be stored for less than 12 months from the conclusion of the study (electronic and hard copy).
On study completion, results will be submitted for peer-reviewed publication and presented at national meetings. A summary of findings will be provided to participants by letter and reviewed with the Patient and Public Involvement and Engagement (PPIE) group.
Discussion
The present study represents the first randomized controlled trial to evaluate the Manchester Short Splint in rehabilitation following zone II flexor tendon repair. Earlier observational work suggested potential benefit, but a prospective trial is required to provide high-quality evidence regarding efficacy and safety before wider adoption.
Adhesions remain a frequent and function-limiting complication after Zone II repair (4-10% of cases), and the rehabilitation approach remains the primary modifiable factor influencing outcome in routine practice. Outcomes in Zone II flexor tendon repairs have improved with stronger multistrand core repairs and early active (or combined) motion protocols; these developments provide a sound biomechanical and clinical rationale for splinting strategies that permit controlled wrist motion. Multicenter experience also emphasizes that shorter or out-of-splint exercises can be safe and efficient in compliant patients, provided repairs use robust multistrand techniques and therapy is supervised, aligning with the principles behind our short-splint regimen. Should the short splint be found to be superior to the traditional long splint, then this will have important implications for how flexor tendon injuries are rehabilitated in future in the context of a 4-strand repair.
We anticipate that the principal challenges on the study will be those around recruitment. The subjects of the study will have sustained injury which requires treatment through the urgent care pathway of the trauma service and the wider department will need to be involved in the identification of potential patients to the study. This will necessitate ongoing efforts to train new staff members as site investigators expert in the conduct of the study.
Recruitment issues may be encountered due to the number of patients being excluded because of strict injury-related criteria for study entry. Large retrospective data identify prolonged time from injury to surgery, multiple tendon injuries and concomitant vascular injury as independent risk factors for adhesions; our 96-hour window and restricted inclusion criteria were chosen to reduce these confounders and protect internal validity. It is important these injuries receive due priority to be treated within the 4-day window.
The burdens for the patient are minimal in this study with the same number of visits as would normally be offered for full rehabilitation of the flexor tendon injury. Individual sessions may take slightly longer to accommodate specific assessments and the recording of those assessments.
The study design is that of single blind involving separation of the treating hand therapist from the assessing hand therapist (site investigator). It is possible therefore that the treating team may come to a view on which regimen is working better before we come to the end of the study. It will be important to avoid conveying our own subjective impressions to patients who are on the study and the site investigators will be briefed on this point.
In terms of the selection of primary and secondary endpoints we have used TAM as our objective measure of range of motion of the joints. Our original audit suggested that there was significant difference between groups at 6 weeks and, while patients improved their ranges in both short and long splint groups between 6 and 12 weeks, the difference at 12 weeks bore similar relationship to that observed at 6 weeks albeit that the absolute ranges had increased in both groups. We have chosen on this basis to assess both time points independent of one another. In a recent randomized study comparing early active motion with a modified Kleinert protocol, grip strength and multiple PROMs were superior at three months, but no difference was found with TAM, and all measures converged by twelve months. This supports our focus on early timepoints and the inclusion of PROMs alongside TAM. With respect to the secondary end point of grip strength this can only be measured at 12 weeks as measurement at 6 weeks could risk rupture of the repair. In addition, the differences in grip strength between male and female participants will be greater than the potential difference generated by the two forms of splints. As such even larger case numbers would be required to do the appropriate analysis.
We anticipate that retention of patients once on the study will be a challenge, particularly at the 12-week time point. The reason for including this time point is to assess whether any early differences in range of movement at 6 weeks persisted at 12 weeks. The inclusion of the 12-week time point also improves our understanding how quickly patients are getting back to work and is therefore important for both clinical and socioeconomic reasons. The potential provision of inconvenience payments to patients would be something to consider in any future study.
Conclusion
This study protocol outlines a pragmatic randomized controlled trial evaluating a novel rehabilitation splint following Zone II flexor tendon repair. The protocol incorporates standardized surgical techniques, blinded outcome assessment, and validated functional and patient-reported measures to ensure methodological rigor. Its findings will provide prospective data on the feasibility, safety, and potential functional advantage of the short splint. Should the short splint be demonstrated to offer superior outcomes without increasing rupture risk, this would have significant implications for clinical practice. The design is simple, low-cost, and readily reproducible across hand therapy units using standard materials. Confirmation of benefit could lead to widespread adoption and standardization of this approach.
Declarations:
ETHICS APPROVAL AND CONSENT TO PARTICIPATE:
North West Haydock Research Ethics Committee IRAS project ID: 159054
TRIAL REGISTRATION:
ClinicalTrials.gov Identifier: NCT03850210
All patient participants undergo full informed consent process as part of the recruitment to the trial.
CONSENT FOR PUBLICATION
Image Consent Publication Statement: The images Figures 1A-D inclusive are of my own hand and I give consent for publication in the Trials Journal.
Vivien Lees 1.5.20
DATA AVAILABILITY STATEMENT
The datasets generated and/or analyzed during the current study are not publicly available due but are available from the corresponding author on reasonable request.
COMPETING INTERESTS:
None to declare
FUNDING:
No grant was sought or awarded for this study. The study has been registered. The Trust will cover the costs of Investigators time as they undertake their trial roles alongside their regular duties. There will be no payments to participants, investigators or Lead Investigator.
AUTHORS CONTRIBUTIONS:
The authors declare that they have no competing interests.
There is no contribution by professional writers to this manuscript.
ACKNOWLEDGEMENTS:
Authors listed on this publication have made contribution in line with the expected contributions specification.
In addition, the authors acknowledge Fiona Peck for work in development of the short splint design, Emma Birkin and Alison Roe for their contributions in the earlier parts of the study. All Site Investigators and colleagues for assisting in recruitment of patients to the study. Bradley Tallon for trial support. Emma Barrett Department of Medical Statistics for power calculation and advice on statistical methodology.
References:
- Wimbiscus M, Sullivan L, Lee J, Vinson A, Torres-Guzman RA, Stehr J, Bhandari L. Advances and Challenges in Zone 2 Flexor Tendon Repairs. Annals of Plastic Surgery. 2024 Dec 1;93(6S): S138-43.
- Peck FH, Bucher CA, Watson JS, Roe A. A comparative study of two methods of controlled mobilization of flexor tendon repairs in zone 2. J Hand Surg Br. 1998, 23: 41-5.
- Thien TB, Becker JH, Theis JC. Rehabilitation after surgery for flexor tendon injuries in the hand. Cochrane Database Syst Rev. 2004: CD003979.
- Small JO, Brennen MD, Colville J. Early active mobilization following flexor tendon repair in zone 2. J Hand Surg Br. 1989, 14: 383-91.
- Neiduski RL, Powell RK. Flexor tendon rehabilitation in the 21st century: A systematic review. J Hand Therapy 2019; 32: 165-174.
- Peck F, Roe AE, Ng CY, Duff C, McGrouther DA, Lees VC. The Manchester short splint: A change to splinting practice in the rehabilitation of zone II flexor tendon repairs. Hand Therapy 2014; 19(2): 47-53.
- Savage R. The influence of wrist position on the minimum force required for active movement of the interphalangeal joints. J Hand Surg Br 1988; 13(3): 262-8.
- Woythal L, Homer P, Brorson S. Splints, with or without wrist immobilization, following surgical repair of flexor tendon lesions of the hand: A systematic review. Hand Surg and Rehab 2019; 38: 217-222.
- Kaskutas V, Powell R. The impact of flexor tendon rehabilitation restrictions on individuals’ independence with daily activities: implications for hand therapists. J Hand Ther. 2013, 26: 22-8; quiz 9.
- Hardwick JT, Tan JJ, Foster MA and Titley OG. A systematic review of 2-strand versus multistrand core suture techniques and functional outcome after digital flexor tendon repair. J Hand Surg Am 2014; 39(4): 686-695.
- Vinitpairot C, Yik JH, Haudenschild DR, Szabo RM, Bayne CO. Current trends in the prevention of adhesions after zone 2 flexor tendon repair. Journal of Orthopaedic Research. 2024 Oct;42(10):2149-58.
- Tang JB. Rehabilitation after flexor tendon repair and others: a safe and efficient protocol. Journal of Hand Surgery (European Volume). 2021 Oct;46(8): 813-7.
- Tang JB, Lalonde D, Harhaus L, Sadek AF, Moriya K, Pan ZJ. Flexor tendon repair: recent changes and current methods. Journal of Hand Surgery (European Volume). 2022 Jan;47(1):31-9.
- Jia Q, Chen D, Guo J, Luo X, Alimujiang A, Zhang J, Hu N, Liu Y, Xie Z, Ma C. Risk factors associated with tendon adhesions after hand tendon repair. Frontiers in Surgery. 2023 Apr 18;10:1121892.
- Renberg M, Turesson C, Borén L, Nyman E, Farnebo S. Rehabilitation following flexor tendon injury in Zone 2: a randomized controlled study. Journal of Hand Surgery (European Volume). 2023 Sep;48(8):783-91.

