Embryo Pooling: Enhancing Ovarian Reserve Outcomes
Role of Embryo pooling in Low ovarian reserve
Keywords:
Low ovarian reserve, embryo pooling, infertility, assisted reproductive technology
Introduction
| No. of Embryos Transferred | No. of Patients |
|---|---|
| Fresh | 24 |
| Frozen | 18 |
| Fresh + Frozen | 38 |
| ET not done | 3 |
The embryos were transferred in either fresh cycle or frozen cycle depending on endometrial conditions. Table 5 shows the distribution of the embryos according to the fresh or frozen stage. In two cases, embryo transfer was done twice as first attempt in both cases resulted in biochemical pregnancy. So, second transfer was done after one month gap. In 3 cases, embryo transfer was not done out of that in two cases embryo didn’t grow and in one case, all embryos were aneuploid.
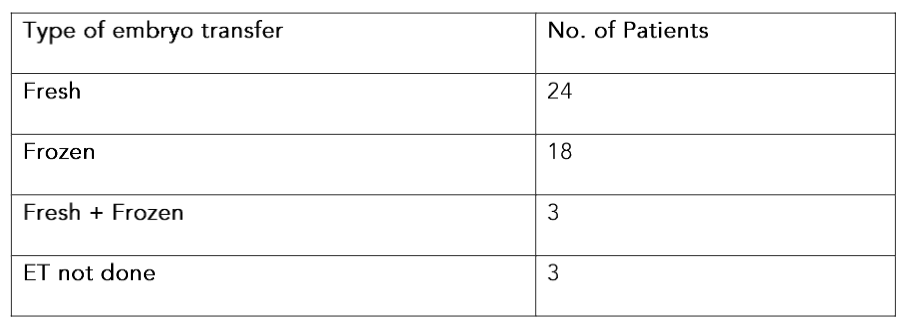
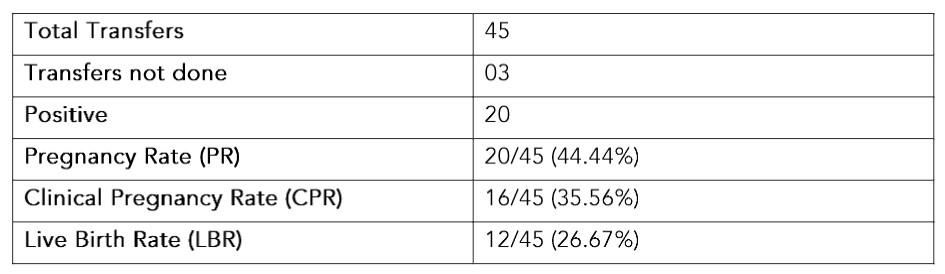
Table 6. Outcome of Embryo Pooling cases
As shown in Table 7, there was no association between the mode of transfer i.e. fresh or frozen, number of embryos transferred and day of embryo transfer with the outcome.
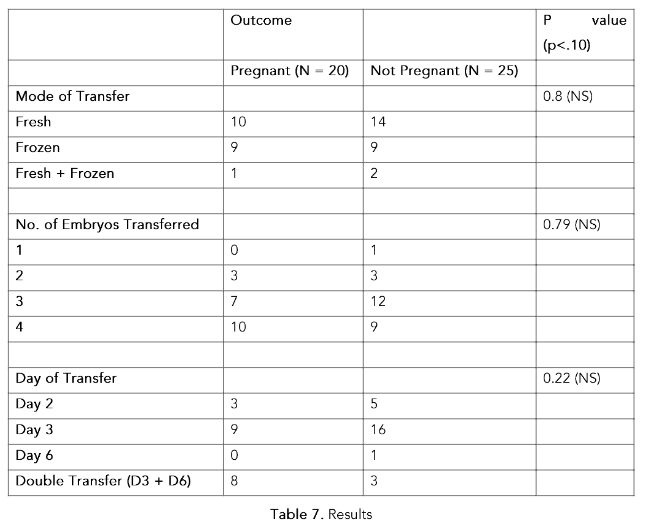
Table 7. Results
Discussion
Low AMH is associated with lower pregnancy outcomes. A large retrospective study performed on 69,336 fresh and 15,458 frozen embryo transfer cycles demonstrated that the areas under the curve (AUC) for AMH as predictor of live birth in fresh cycles and thawed cycles were respectively, 0.631 and 0.540, suggesting that AMH alone is a weak, even if significant, age-independent predictor of live birth after ART.¹⁰
Oocytes may not be retrieved; embryos may not be formed and even when they are formed, they may be less in number and quality. Therefore, the suggestion of multiple cycle stimulation to form minimum of two grade A cleavage stage embryos is suggested.¹¹–¹⁴
It is believed that blastocyst transfer is better than cleavage stage transfer in improving live births. This is true in most cases. However, one cannot forget that there are certain cases, especially in women where the total number of eggs is less that 5, one may not be able to extend embryos to blastocysts and there is risk of no transfer.¹⁵ Also extending culture to blastocyst in those with good egg numbers means freezing fewer embryos so there is also a question of whether the cumulative live birth in case of blastocyst transfer is actually better than cleavage which is contested in many studies.¹⁶,¹⁷ In our clinic we practice transferring embryos at an early stage when the total number of eggs retrieved is less than 5 which according to us gives the patient a better chance of a live birth baby.
It is widely believed and practiced that transferring fewer number of embryos leads to lower multiple pregnancy rate. While that is true it also leads to lower pregnancy rate per embryo transfer in women with advanced maternal age and women with multiple
1. Palgamkar JB, Jindal DK, Sawkar SV et al.Anti-Mullerian Hormone Levels in Indian Women Seeking Infertility Treatment: Are Indian Women Facing Early Ovarian Senescence? J Hum Reprod Sci. 2021 Oct-Dec;14(4):380-385. doi: 10.4103/jhr s.jhrs_71_21. Epub 2021 Dec 31. PMID: 35197683; PMCID: PMC8812391.
2. Ferraretti AP, La Marca A, Fauser BC, Tarlatzis B, Nargund G, Gianaroli L; ESHRE working group on Poor Ovarian Response Definition. ESHRE consensus on the definition of ‘poor response’ to ovarian stimulation for in vitro fertilization: the Bologna criteria. Hum Reprod. 2011 Jul;26(7):1616-24. doi: 10.1093/humrep/der092. Epub 2011 Apr 19. PMID: 21505041.
3. Abu-Musa A, Haahr T, Humaidan P. Novel Physiology and Definition of Poor Ovarian Response; Clinical Recommendations. Int J Mol Sci. 2020 Mar 19;21(6):2110. doi: 10.3390/ijms210 62110. PMID: 32204404; PMCID: PMC7139860.
4. Zhao D, Fan J, Wang P, Jiang X, Yao J, Li X. Age-specific definition of low anti-Mullerian hormone and associated pregnancy outcome in women undergoing IVF treatment. BMC Pregnancy Childbirth. 2021 Mar 5;21(1):186. doi: 10.1186/s12 884-021-03649-0. PMID: 33673814; PMCID: PMC7 936413.
5. Revelli A, Biasoni V, Gennarelli G, Canosa S, Dalmasso P, Benedetto C. IVF results in patients with very low serum AMH are significantly affected by chronological age. J Assist Reprod Genet. 2016 May;33(5):603-609. doi: 10.1007/s10815-016-067 5-7. Epub 2016 Feb 18. PMID: 26888025; PMCID: PMC4870438.
6. Rasool S, Shah D. Fertility with early reduction of ovarian reserve: the last straw that breaks the Camel’s back. Fertil Res Pract. 2017 Oct 11;3:15. doi: 10.1186/s40738-017-0041-1. PMID: 29046817; PMCID: PMC5637249.
7. Sunkara SK, Rittenberg V, Raine-Fenning N, Bhattacharya S, Zamora J, Coomarasamy A. Association between the number of eggs and live birth in IVF treatment: an analysis of 400 135 treatment cycles. Hum Reprod. 2011 Jul;26(7): 1768-74. doi: 10.1093/humrep/der106. Epub 2011 May 10. PMID: 21558332.
8. Cobo A, Garrido N, Crespo J, José R, Pellicer A. Accumulation of oocytes: a new strategy for managing low-responder patients. Reprod Biomed Online. 2012 Apr;24(4):424-32. doi: 10.1016/j.rbm o.2011.12.012. Epub 2012 Jan 8. PMID: 22386762.
9.https://dhr.gov.in/sites/default/files/ART%20Rules%202022%20notification%20dated%2007062022%20published%20on%2008%20june.pdf
10. Tal R, Seifer DB, Wantman E, Baker V, Tal O. Antimüllerian hormone as a predictor of live birth following assisted reproduction: an analysis of 85,062 fresh and thawed cycles from the Society for Assisted Reproductive Technology Clinic Outcome Reporting System database for 2012-2013. Fertil Steril. 2018 Feb;109(2):258-265. doi: 10.1016/j.fert nstert.2017.10.021. Epub 2018 Jan 11. PMID: 2933 1235.
11. Vermey BG, Chua SJ, Zafarmand MH et al. Is there an association between oocyte number and embryo quality? A systematic review and meta-analysis. Reprod Biomed Online. 2019 Nov;39(5): 751-763. doi: 10.1016/j.rbmo.2019.06.013. Epub 2019 Jul 3. PMID: 31540848.
12. Liu R, Zhang Q, Geng L et al. What is the optimal number of embryos to transfer for POSEIDON group 1 and group 2? A retrospective study. J Ovarian Res. 2024 May 31;17(1):117. doi: 10.1186/s13048-024-01443-y. PMID: 38822354; PMCID: PMC1114 0960.
13. Hu X, Ding C, Zhang D et al. Embryo pooling: a promising strategy for managing insufficient number of embryos in preimplantation genetic diagnosis. Gynecol Endocrinol. 2017 Nov; 33(11):867-871. doi: 10.1080/09513590.2017.134 7778. Epub 2017 Jul 6. PMID: 28682646.
14. Kamath MS, Mascarenhas M, Kirubakaran R, Bhattacharya S. Number of embryos for transfer following in vitro fertilisation or intra-cytoplasmic sperm injection. Cochrane Database Syst Rev. 2020 Aug 21;8(8):CD003416. doi: 10.1002/14651858.C D003416.pub5. PMID: 32827168; PMCID: PMC80 94586.
15. Neblett MF 2nd, Kim T, Jones TL et al. Is there still a role for a cleavage-stage embryo transfer? F S Rep. 2021 Jun 29;2(3):269-274. doi: 10.1016/j.xfr e.2021.06.004. PMID: 34553150; PMCID: PMC844 1559.
16. Fernández-Shaw S, Cercas R, Braña C, Villas C, Pons I. Ongoing and cumulative pregnancy rate after cleavage-stage versus blastocyst-stage embryo transfer using vitrification for cryopreservation: impact of age on the results. J Assist Reprod Genet. 2015 Feb;32(2):177-84. doi: 10.1007/s10815-014-0387-9. Epub 2014 Nov 18. PMID: 25403438; PMCID: PMC4354180.
17. Glujovsky D, Farquhar C, Quinteiro Retamar AM, Alvarez Sedo CR, Blake D. Cleavage stage versus blastocyst stage embryo transfer in assisted reproductive technology. Cochrane Database Syst Rev. 2016 Jun 30;(6):CD002118. doi: 10.1002/146 51858.CD002118.pub5. Update in: Cochrane Database Syst Rev. 2022 May 19;5:CD002118. PMID: 27357126.
18. Gleicher N, Kushnir VA, Barad DH. Risks of spontaneously and IVF-conceived singleton and twin pregnancies differ, requiring reassessment of statistical premises favoring elective single embryo transfer (eSET). Reprod Biol Endocrinol. 2016 May 3;14(1):25. doi: 10.1186/s12958-016-0160-2. PMID: 27142226; PMCID: PMC4855800.
19. Practice Committee of the American Society for Reproductive Medicine. Electronic address: [email protected]; Practice Committee of the Society for Assisted Reproductive Technology. Guidance on the limits to the number of embryos to transfer: a committee opinion. Fertil Steril. 2017 Apr;107(4):901-903. doi: 10.1016/j.fertnstert.201 7.02.107. Epub 2017 Mar 11. PMID: 28292618.
20. Masschaele T, Gerris J, Vandekerckhove F, De Sutter P. Does transferring three or more embryos make sense for a well-defined population of infertility patients undergoing IVF/ICSI? Facts Views Vis Obgyn. 2012;4(1):51-8. PMID: 24753889; PMCID: PMC3991445.
21. Greco E, Litwicka K, Arrivi C et al. Accumulation of oocytes from a few modified natural cycles to improve IVF results: a pilot study. J Assist Reprod Genet. 2013 Nov;30(11):1465-70. doi: 10.1007/s10815-013-0103-1. PMID: 24077861; PMCID: PMC3879935.
22. Vora AV, Nadkarni P, Singh P N, Nadkarni V, Nadkarni A.Case reports for embryo banking – for women who want their own. Int JReprod Contracept Obstet Gynecol 2019;8:2926-31.
23. Lee KS, Lin MH, Hwu YM, Yang JH, Lee RK. The live birth rate of vitrified oocyte accumulation for managing diminished ovarian reserve: a retrospective cohort study. J Ovarian Res. 2023 Mar 3;16(1):49. doi: 10.1186/s13048-023-01128-y. PMID: 36869354; PMCID: PMC9983267.
24. Trokoudes KM, Pavlides C, Zhang X. Comparison outcome of fresh and vitrified donor oocytes in an egg-sharing donation program. Fertil Steril. 2011 May;95(6):1996-2000. doi: 10.1016/j.fe rtnstert.2011.02.035. Epub 2011 Mar 15. PMID: 21406304.
25. Domingues TS, Aquino AP, Barros B et al. Egg donation of vitrified oocytes bank produces similar pregnancy rates by blastocyst transfer when compared to fresh cycle. J Assist Reprod Genet. 2017 Nov;34(11):1553-1557. doi: 10.1007/s10815-017-1017-0. Epub 2017 Aug 16. PMID: 28815355; PMCID: PMC5699994.
26. Setti AS, Braga DPAF, Iaconelli A, Borges E. Fresh oocyte cycles yield improved embryo quality compared with frozen oocyte cycles in an egg-sharing donation programme. Zygote. 2021 Jun;29(3):234-238. doi: 10.1017/S0967199420000842. Epub 2021 Jan 18. PMID: 33455591.
27. Sadeghi MR. Oocytes/Embryos Banking: A Vague Hope for Poor Responder Women. J Reprod Infertil. 2018 Jul-Sep;19(3):123-124. PMID: 30167391; PMCID: PMC6104429.
28. Practice Committee of the American Society for Reproductive Medicine and the Practice Committee for the Society for Assisted Reproductive Technologies. Electronic address: [email protected]. Guidance on the limits to the number of embryos to transfer: a committee opinion. Fertil Steril. 2021 Sep;116(3):651-654. doi: 10.1016/j.fertnstert.202 1.06.050. Epub 2021 Jul 28. PMID: 34330423.
29. Çelik S, Turgut NE, Cengiz Çelik D et al. The effect of the pooling method on the live birth rate in poor ovarian responders according to the Bologna criteria. Turk J Obstet Gynecol. 2018 Mar; 15(1):39-45. doi: 10.4274/tjod.62447. Epub 2018 Mar 29. PMID: 29662715; PMCID: PMC5894535
30. McCormack CD, Leemaqz SY, Furness DL, Dekker GA, Roberts CT. Anti-Müllerian hormone levels in recurrent embryonic miscarriage patients are frequently abnormal, and may affect pregnancy outcomes. J Obstet Gynaecol. 2019 Jul;39(5):623-627. doi: 10.1080/01443615.2018.1552669. Epub 2019 Mar 27. PMID: 30917731.
31. Busnelli A, Somigliana E, Cirillo F, Levi-Setti PE. Is diminished ovarian reserve a risk factor for miscarriage? Results of a systematic review and meta-analysis. Hum Reprod Update. 2021 Oct 18;27(6):973-988. doi:10.1093/humupd/dmab018. PMID: 34254138.
32. Blumenfeld Z. What Is the Best Regimen for Ovarian Stimulation of Poor Responders in ART/IVF? Front Endocrinol (Lausanne). 2020 Apr 17;11:192. doi: 10.3389/fendo.2020.00192. PMID: 32362870; PMCID: PMC7180183.
33. Davis OK. IVF stimulation: protocols for poor responders. Methods Mol Biol. 2014;1154: 329-41. doi: 10.1007/978-1-4939-0659-8_15. PMID: 24782017.
34. Jeve YB, Bhandari HM. Effective treatment protocol for poor ovarian response: A systematic review and meta-analysis. J Hum Reprod Sci. 2016 Apr-Jun;9(2):70-81. doi: 10.4103/0974-1208.183515. PMID: 27382230; PMCID: PMC4915289.
35. Katsika ET, Bosdou JK, Goulis DG, Grimbizis GF, Kolibianakis EM. Higher live birth rate following transdermal testosterone pretreatment in poor responders: a systematic review and meta-analysis. Reprod Biomed Online. 2023 Jan;46(1):81-91. doi: 10.1016/j.rbmo.2022.09.022. Epub 2022 Oct 7. PMID: 36369150.
36. Caligara C, Navarro J, Vargas G, Simón C, Pellicer A, Remohí J. The effect of repeated controlled ovarian stimulation in donors. Hum Reprod. 2001 Nov;16(11):2320-3. doi: 10.1093/humrep/16.11.2320. PMID: 11679512.
37. Pailis M, Sapir O, Lande Y et al. Consecutive ovarian stimulation is beneficial in patients with a poor response to high-dose follicle-stimulating hormone. Gynecol Endocrinol. 2021 Nov;37(11): 995-999. doi: 10.1080/09513590.2021.1908991. Epub 2021 Apr 9. PMID: 33834936.
38. Silverberg KM, Klein NA, Burns WN, Schenken RS, Olive DL. Consecutive versus alternating cycles of ovarian stimulation using human menopausal gonadotrophin. Hum Reprod. 1992 Aug;7(7):940-4. doi: 10.1093/oxfordjournal s.humrep.a137775. PMID: 1430133.
39. Ron-El R, Raziel A, Herman A et al. Ovarian response in repetitive cycles induced by menotrophin alone or combined with gonadotrophin releasing hormone analogue. Hum Reprod. 1990 May;5(4): 427-30. doi: 10.1093/oxfordjournals.humrep.a137116. PMID: 2141846.