Endometrial Nerve Fibers in Rhesus Monkeys Study
Endometrial Nerve Fibers in Cycling Rhesus Monkeys: Immunohistochemical Study with Frozen Sections
Tatsuo Tomita 1, Kunie Mah 2
- Kunie Mah Oregon National Primate Research Center, Beaverton, OR
- Tatsuo Tomita Department of Integrative Biosciences, Oregon Health and Science University, Portland, OR, USA
OPEN ACCESS
PUBLISHED: 28 Febuary 2025
CITATION: MAH, Kunie; TOMITA, Tatsuo. Endometrial Nerve Fibers in Cycling Rhesus Monkeys: Immunohistochemical Study with Frozen Sections. Medical Research Archives, [S.l.], v. 13, n. 2, mar. 2025. Available at: <https://esmed.org/MRA/mra/article/view/6451>.
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i2.6451
ISSN 2375-1924
Abstract
The presence of nerve fibers in the eutopic primate and human endometrium is unsettled. Fine nerve fibers are present mostly at the endometrial-myometrial interphase, but nerve fibers have not been detected in the functionalis in the normally cycling primate and human endometrium. There are no reports on nerve fibers in the cycling primate and human endometrium. Fine nerve fibers in the functionalis have been reported from women with endometriosis but not women without endometriosis. Several authors claim that immunohistochemical findings of nerve fibers in the functionalis is diagnostic for endometriosis without mentioning menstrual cycle from the women studied for nerve fibers. The current study aimed to detect nerve fibers in the artificially cycling rhesus with uterine frozen sections using neurofilament, CD56 and PGP9.5 as markers for nerve fibers. We detected clumps of fine nonmyelinated nerve fibers together with a few myelinated nerves consistently at the basalis-myometrium interphase and in the basalis with partially spreading into the deep functionalis while there were no nerve fibers in functionalis from all menstrual phases. There were diffusely and thinly distributed fine nerve fibers in the myometrium from all cycling phases. In the rhesus endometrium, there were no nerve fibers in the upper and middle zone of cycling functionalis in both proliferative and secretary phases. The nerve fibers in the basalis grow into to the deep zone functionalis in the eutopic monkey endometrium, thus the normally present nerve fibers in the deep zone functionalis are not specific for endometriosis.
Keywords
endometrium, CD56, immunohistochemistry, neurofilament, nerve fiber, rhesus monkey
Introduction
The presence of nerve fibers in the mammalian endometrium is still unsettled. In rabbits, rats, mongoose and pig, nonmyelinated nerve fibers were detected in the endometrial-myometrium interphase mixed with few myelinated nerve fibers. There were a few literatures on monkey and baboon endometrium revealing most nerve fibers at the endometrial-myometrium interphase and thinly and diffusely in the myometrium. Most immunohistochemical studies had been previously performed in the routinely formalin-fixed and paraffin-embedded human endometrium: innervation of the human endometrium is significantly less than other parts of the uterus such as myometrium, horn and cervix. There are small nonmyelinated nerve fibers at the endometrial-myometrium interphase and diffusely distributed small nerve fibers in the entire myometrium. Many nerve fibers are located adjacent to the blood vessels, mostly arteries but some are freely distributed in the endometrial stroma while fine nerve fibers are located continuously from the basalis to the lower zone functionalis. In women with endometriosis, there were significantly increased nerve fibers in the endometrial functionalis, which has claimed and used as a diagnostic marker for endometriosis. The others reported no correlation of the presence of functionalis nerve fibers in women with endometriosis. In the eutopic women, the highest nerve density was reported in the myometrium with less density in the basalis and few in the functionalis. There have been no reports on nerve fibers in the eutopic cycling women and rhesus monkeys and this study aimed to reveal nerve fibers in the artificially cycling rhesus monkeys. We used mostly neurofilament (NF) and CD56 as markers for nerve fibers. NF has been widely used as a highly specific marker for nonmyelinated Aσ, Aβ, Aδ and B fibers and myelinated adrenergic fibers. CD56 is a marker for fine nerves, fine varicose and sensory nerve endings, cell membranes of ganglion cells and fetal striated muscle. All previously reports on the human endometrium had been performed with routinely formalin-fixed and paraffin-embedded tissues. We had previously demonstrated that immunostaining with frozen sections was superior to the formalin-fixed and paraffin-embedded tissues for immunostaining lymphatic vessels using LYVE-1 and blood vessels using von Willebrand factor and extended immunostaining nerve fibers using frozen tissues from cycling and spayed rhesus monkeys.
Materials and Methods
Animal care for the rhesus monkeys (Macaca mulatta) was provided by the veterinary staff of the Division of Animal Resources of the Oregon Primate Research Center, Beaverton, OR in accordance with NIH policy for the care and use of laboratory animals. Female rhesus monkeys were oophorectomized and treated sequentially with estradiol (E2) and progesterone (P) to induce menstrual cycles described before. To induce cycles, the oophorectomized animals were first treated with 3cm Silastic capsules (Dow Corning Corp, Midland, MI) packed with crystalline E2 (Sigma) to stimulate development of an induced proliferative phase. After 14 days, a 6cm Silastic capsule packed with crystalline P (Sigma) was implanted sc, and both implants remained in place for 14 days to induce the secretory phase. To induce menstruation, the P implant was removed while the E2 implant was left in place. Menstrual bleeding normally occurs 2-4 days after the P implant was removed. The human endometrium according to the exact menstrual cycle is hard to study from the ethical points of view and this study was undertaken with the artificially cycling rhesus monkeys. The numbers of the animals studied were as follows: the induced menstrual phase (3 days after removing P capsule, E-3p): 5 animals, early proliferative phase (E-7p): 4 animals, late proliferative phase (E-7p): 3 animals, early secretory phase (7 days after placing P capsule, E+ 7P): 3 animals and late secretary phase (E+14P): 4 animals to total 19 animals. A few animals were spayed for 5 months to simulating the postmenopausal state in women. Uteri were procured by necropsy under the appropriate anesthesia at the Oregon National Primate Center. With each uterine histologic sections, the endometrial thickness including the functionalis, basal and total thickness was measured on the H and E-stained tissue sections. For frozen sections, small fresh uterine tissues including myometrium (1 x 1 x 0.5cm) were embedded in OCT matrix (Fisher Scientific, Pittsburgh, PA) and were frozen in liquid propane in the liquid nitrogen as described before and were frozen sectioned at 7 to 10 µm sections. Frozen sections were mounted on Super Plus slides (Fisher Scientific), microwave-irradiated on ice for 3 sec, fixed in 2% paraformaldehyde in phosphate buffer at pH 7.4 for 10 to 15 min at room temperature and immersed twice for 2 min in 85% ethanol. To inhibit endogenous peroxidase activity, sections were incubated with a solution containing glucose oxidase (1U/ml) and sodium azide (10mmll/ml) in PBS for 45 min at 25˚C. Then, sections were incubated with each diluted primary monoclonal NF and monoclonal CD56 and rabbit PGP9.5 antibodies, respectively at 4̊C overnight as reported before. After rinsing and immersion in blocking serum again, sections were incubated with second antibody (1 : 200 dilution) for 30 minutes at room temperature. Final visualization was achieved with the ABC kit (Vector Laboratory, Burlingame, CA) and 0.025 diaminobenzidine tetrahydrochloride in Tris-buffer pH7.6, 0.03 H2O2 to induce brown color. The other sets of frozen sections were processed in the same way in the peroxidase method, then the final visualization was achieved by the alkaline phosphatase Vector Blue kit (SK 5300, Vector Laboratories, Burlingame, CA) for blue coloring. The source of the primary antibodies and each dilution of the antibody were listed as follows: Monoclonal human NF (clone 2F11), Dako System, Carpenteria, CA 1 :100 dilutions Monoclonal CD56, (clone 1E8C9), Dako System, Carpenteria, CA 1: 100 Rabbit PGP9.5 (TGX 17039), Gene Tex, CA 1: 100. The Day 28 specimen (E + 14p) revealed the longest functionalis and total endometrial length (Table 1), and this specimen was studied with 30µm thick sections for peroxidase method and 10µm sections for alkaline phosphatase method, respectively for nerve fibers since this specimen had the most reliable possibility to show nerve fibers in case that nerve fibers were present in the functionalis.
Results
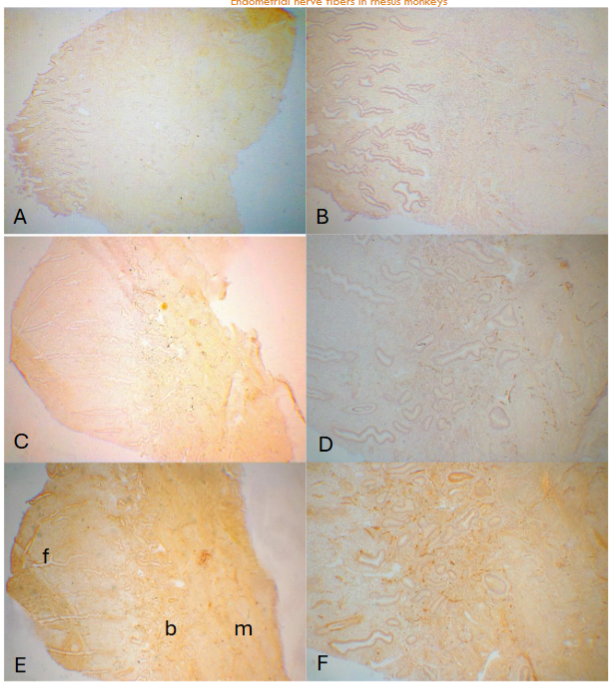
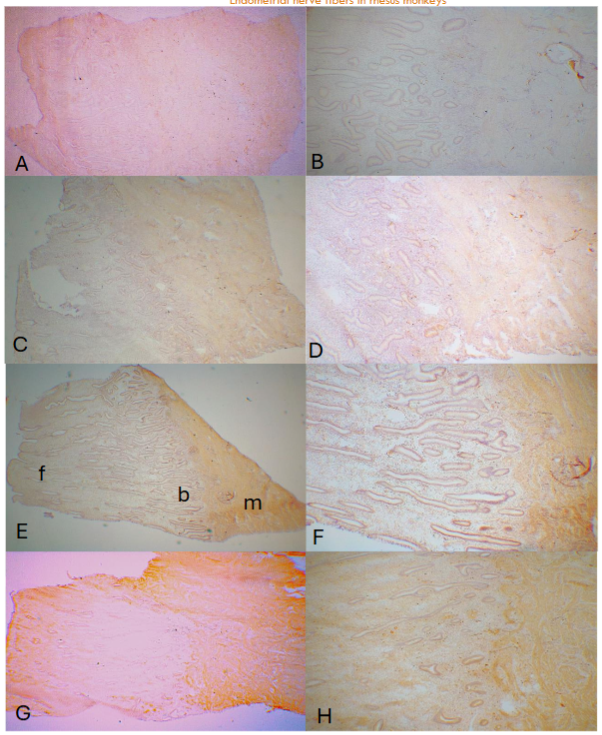
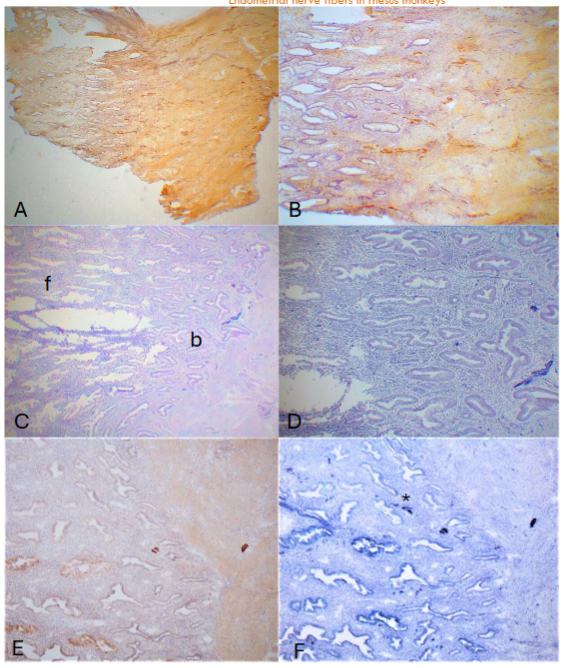
The E capsule has been placed during the entire cycling phases as listed as E. The three day after P capsule was removed stated as E – 3P while the 14th day with P capsule was placed after removing the previously placed P capsule as E + 14P. The day 3 (E – 3P, E continuously placed, and P removed for 3 days) endometrium consisted of thin sloughed off endometrium, corresponding to the remnant of basalis, which measured 438 ± 38 µm in thickness. There were few or no NF immunostained small linear nonmyelinated vertical nerve fibers (20-40µm in length) by CD56 in the sloughed off residual basalis while there were consistently present thin, linear, nonmyelinated nerve fibers scattered in the entire myometrium with relatively more nerve fibers revealed by CD56 than by NF immunostaining (Figure 1-A and -B). Day 7 (E – 7P) endometrium revealed elongating functionalis (1,575 ± 75µm) where there were a few linear nonmyelinated nerve fibers at the basalis-myometrium interphase and in basalis with scattered linear nonmyelinated nerve fibers in the myometrium using NF as a marker (Figure 1-C and -D) and using PGP 9.5 as a marker (Figure 1-E and -F). In the Day 14 (E -14P) specimen, functionalis was more elongated (1,700 ± 58µm) and there were a few myelinated nerve fibers and several nonmyelinated nerve fibers at the basalis-myometrium interphase and nonmyelinated nerve fibers in the myometrium using both NF (Figure 2-A and -B) and CD56 (Figure 2-C and -D). The Day 21 (E + 7P) specimen showed more elongated functionalis (1,800 ± 47µm) with a few nonmyelinated nerve fibers and more linear nonmyelinated nerve fibers in the basalis-myometrium interphase with a few nonmyelinated nerve fibers spreading into the superficial myometrium using NF (Figure 2-E and -F) and CD56 (Figure 2-G and -H). In the Day 28 (E ± 14P) specimen, there were most elongated functionalis (2,225 ±131µm) with numerous thick nerve fibers in the 20 µm-thick section immunostained with CD56 (Figure 3-A and -B) and saw-tooth appearance at the upper luminal functionalis in the 10µm thick sections with a few myelinated nerve fibers and more nonmyelinated nerve fibers at the basalis-myometrium interphase with NF immunostaining using alkaline phosphatase (Figure 3-C and -D) and diffusely, thinly scattered small linear nonmyelinated nerve fibers in the myometrium with both CD56 and NF immunostaining (Figure 3-A to -D). Comparing immunostaining between peroxidase and alkaline phosphatase methods, alkaline phosphatase method detected fine non-myelinated nerve fibers (*), which peroxidase method did not detect although both methods detected thick non-myelinated and myelinated nerve fibers as well. In the above four cycling stages, there were no myelinated and nonmyelinated nerve fibers in the upper and middle zone functionalis using both NF and CD56 with peroxidase and alkaline phosphatase methods. In the specimens from rhesus spayed for 5 months, the endometrium was simulated for the postmenopausal endometrium where there were extremely thickened basalis (500-750µm) with no attached functionalis, and there were thin, diffusely increased fine linear vertical nonmyelinated nerve fibers in the thickened basalis by NF and CD56 immunostaining but there were no immunostained nerve fibers in the basalis using PGP9.5 staining. In the myometrium, there were markedly and diffusely increased fine linear nerve fibers with large nerve bundles, myelinated and numerous nonmyelinated nerve fibers around the cross sections of arteries with NF and CD56 but partially immunostained by PGP9.5 around the arteries.
| Menstrual phase (4) | E-3p | Basalis (µm) | Functionalis (µm) | Total length (µm) |
|---|---|---|---|---|
| 1 | 400 µm | 0 µm | 400 µm | |
| 2 | 500 | 0 | 500 | |
| 3 | 500 | 0 | 500 | |
| 4 | 350 | 0 | 350 | |
| Mean ± SE | 438 ± 38 µm | 0 ± 0 | 438 ± 38 µm |
Early proliferative phase (4) E-7p
| Animals | Basalis (µm) | Functionalis (µm) | Total length (µm) |
|---|---|---|---|
| 1 | 400 µm | 1,000 µm | 1,400 µm |
| 2 | 440 | 1,260 | 1,700 |
| 3 | 300 | 1,200 | 1,500 |
| 4 | 200 | 1,500 | 1,700 |
| Mean ± SE | 335 ± 47 µm | 1,240 ± 103 µm | 1,575 ± 75 µm |
Late proliferative phase (3) E-14p
| Animals | Basalis (µm) | Functionalis (µm) | Total length (µm) |
|---|---|---|---|
| 1 | 400 µm | 1,000 µm | 2,200 µm |
| 2 | 400 | 1,600 | 2,000 |
| 3 | 300 | 1,700 | 2,000 |
| Mean ± SE | 367 ± 27 µm | 1,700 ± 58 µm | 2,067 ± 115 µm |
Early secretary phase (3) E +7p
| Animals | Basalis (µm) | Functionalis (µm) | Total length (µm) |
|---|---|---|---|
| 1 | 440 µm | 1,700 µm | 2,200 µm |
| 2 | 400 | 1,800 | 2,200 |
| 3 | 200 | 1,900 | 2,100 |
| Mean ± SE | 347 ± 103 µm | 1,800 ± 47 µm | 2,167 ± 27 µm |
Late secretary phase (4) E+14p
| Animals | Basalis (µm) | Functionalis (µm) | Total length (µm) |
|---|---|---|---|
| 1 | 300 µm | 2,000 µm | 2,300 µm |
| 2 | 300 | 2,100 | 2,500 |
| 3 | 400 | 2,200 | 2,600 |
| 4 | 400 | 2,600 | 3,000 |
| Mean ± SE | 350 ± 25 µm | 2,225 ± 131 µm | 2,600 ± 147 µm |
Discussion
The presence of nerve fibers in the primate and human endometrium has not been settled. Nerve fibers in the endometrium are less than those in cervix and vagina. In the eutopic rhesus monkey and baboons, nerve fiber density was the highest in the myometrium and there was no reported study of nerve density between basalis and myometrium. In this study, rhesus monkeys were induced with menstrual cycle placing E capsule throughout the cycle, producing the proliferative phase, and placing P capsule to induce the secretory phase, then removing P capsule to induce menstruation. We studied the entire 28-day cycle studying nerve fibers in the endometrium and myometrium. In the cycling rhesus endometrium, there was consistent presence of fine nonmyelinated and occasional myelinated nerve fibers at the basalis-myometrial interphase, and myometrium was diffusely innervated with fine nonmyelinated fine nerve fibers. We did not detect nerve fibers in the upper and middle functional zones in the entire menstrual cycle in the rhesus endometrium. In the human uterus, there was consistent presence of nonmyelinated fine nerve fibers at the basalis-myometrial interphase and diffusely present fine nerve fibers in the myometrium. Many authors reported that nerve fibers were detected in the functionalis using NF and PGP9.5 in the endometrium from women with endometriosis but not in women without endometriosis. A few authors reported no difference of nerve fibers in the functionalis between women with endometriosis and women without endometriosis. Those who reported the presence of nerve fibers in the functionalis used it as a diagnostic tool for diagnosing endometriosis. By laparoscopic biopsy, Boker et al and other further claimed that the presence of nerve fibers in the functionalis from women with mild and moderate endometriosis was at 95% sensitivity and 100% specificity, to which we are skeptical since it is practically impossible to histopathologically define zonation of functionalis on the fragmented laparoscopy specimens. This claim must be verified by more study in a detailed study of nerve fibers regarding zones of functionalis and basalis since their biopsy-obtained specimens were not reliable for precise zonation of functionalis for nerve fibers in the endometrium. The previously reported studies all used routinely formalin-fixed and paraffin-embedded tissues, which have proven to be not the best way to detect nerve fibers for lymphatic and blood vessels. We used frozen section to detect nerve fibers in this study since immunohistochemical staining for nerve fibers has shown much more prevailing using frozen section compared to using formalin-fixed and paraffin-embedded tissues. We used NF as a specific marker for nerve fibers, which immunohistochemically stains nonmyelinated A, B fibers and small fibers and we added CD56, which immunohistochemically stains thin sensory nerve fibers, fine varicose and sensory nerve endings and cell membrane of ganglion cells with better immunostaining for small nonmyelinated nerve fibers than NF in our experience. PGP9.5 is a pan neural marker originally isolated from whole human brain as is found neurons, peripheral nerves and neuroendocrine cells but is not specific for neural and neural sheath tumors.
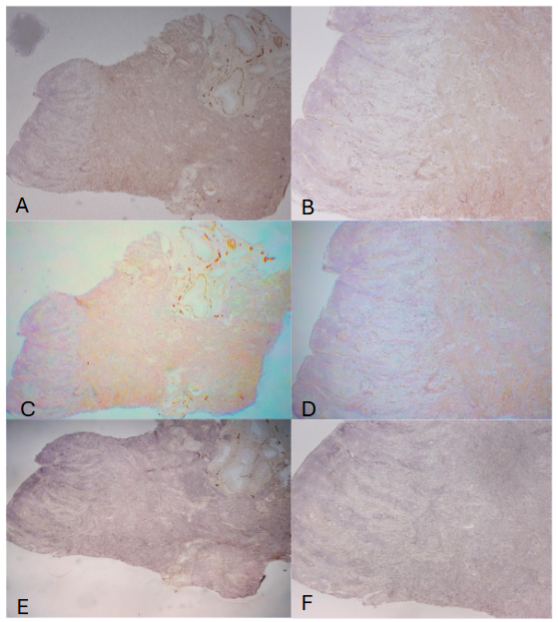
Compared to the normal peritoneum, Tokushige et al claimed increased nerve fiber in the peritoneal endometriotic lesions, which is a dislocated basalis with no attached functionalis. These nerve fibers were unmyelinated sensory C, a mixture of sensory Aδ and myelinated adrenergic fibers. However, there were much more nonmyelinated fine nerve fibers in the basalis compared to occasional presence of myelinated nerve in the rhesus monkeys in our study. They used CGRP (calcitonin gene-related peptide), neuropeptide Y (NPY) and substance P (SP) as markers for sensory C and sensory Aδ. They further used Vacht (vesicular acetylcholine transporter) for cholinergic, VIP (vasoactive polypeptide) for parasympathetic nerve and tyrosine hydroxylase for myelinated adrenergic nerve marker. Tokushige et al further published nerve density of nerve fibers in the women untreated for endometriosis as follows: functionalis;11 ± 5mm2, basalis;18 ± 8mm2 and myometrium;3 ± 1mm2. Endometriosis is a dislocation of basalis, which is normally innervated with fine nerve fibers, thus endometriotic lesions represented by basalis are also innervated with fine nerve fibers and there are no functionalis elements in endometriotic lesions. The presence of nerve fibers in the endometriotic lesions supports endometrial/progenitor cell theory, that resides in the basalis bases and endometrial progenitor cells give rise to endometrial, endothelial, nerve and stromal cells. The five-month spayed animals simulated the postmenopausal women. The endometrium revealed thickened basalis with no functionalis, showing extensively increased nonmyelinated fine nerve fibers in the basalis by NE and CD56 immunostaining while there was no immunostained fine nerves using PGP9.5 immunostaining. There were numerous nerve fibers detected around arteries in the myometrium, among which immunostaining using NF and CD56 stained circular nerve fibers around the arteries but PGP9.5 immunostained nerve fibers partially, not completely surrounding the arteries. There were diffusely increased nonmyelinated fine nerve fibers in the entire myometrium with myelinated thick nerves adjacent to arteries by all NF, CD56 and PGP9.5 immunostaining at relatively less immunostained by PGP9.5 compared to NF and CD56 immunostaining. As shown in the spayed rhesus, PGP9.5 is a pan neural marker and is not the best marker for nerve fibers compared to NF and CD56 since small nerve fibers in the endometrial basalis may not be adequately immunohistochemically stained as shown in the spayed rhesus. Compared to the normal peritoneum, Tokushige et al claimed increased nerve fiber in the peritoneal endometriotic lesions, which is a dislocated basalis, and the normal basalis is also innervated with fine nerve fibers same as the dislocated endometrium in the peritoneal endometriotic lesions. Tokushige et al’s original reports were the presence of nerve fibers in the functionalis from women with endometriosis, and absent nerve fibers in the eutopic women without endometriosis, the latter finding was the same for the cycling rhesus monkeys in our study. They published nerve fibers without the zonation of the functionalis and their high-power microphotographs did not provide the precise zone location of nerve fibers in the endometrium. There has not been reports regarding endometrial nerve fibers from normally cycling women and rhesus monkeys. In the myometrium, there were diffusely distributed sensory C and sensory Aδ and a few myelinated adrenergic nerve fibers, which transmit menstrual and chronic pelvic pain in tandem with uterine contraction. Comparing nerve fibers between endometrium and myometrium, myometrium is consistently distributed with fine nerve fibers throughout the menstrual cycle, while basalis also is distributed with fine nerve fibers continuous from the endometrial-basalis interphase. But there are no nerve fibers in the upper and middle zone functionalis in both proliferative and secretary phases. In the secretarial phase, a few fine nerve fibers infiltrate to the lower zone functionalis adjacent to the basalis. Thus, this normal secretary functionalis is distributed with a few fine nerve fibers and the presence of nerve fibers in this lower zone functionalis is not diagnostic for endometriosis from the current study. Reduced total nerve density was reported during estrus and nerve density recovered during menstruation in the rat. Since no nerve fibers were detected in the upper and middle zone functionalis, menstrual and chronic pelvic pain would be less likely transmitted by endometrial nerve but more likely transmitted through the diffusely distributed, sensory non-myelinated nerve fibers in the myometrium. Uterine innervation in the endometrium may play significant roles in various conditions such as menstrual pain, chronic pelvic pain with or without endometriosis, infertility, spontaneous abortion and others.
Acknowledgement
We would like to express our sincere thanks to Drs Robert M Brenner and Ov D Slayden for kindly allowing me to use normal tissues of rhesus monkey at their research laboratory of the Oregon National Primate Research Center, Beaverton, OR, USA. Their critical advice and comments are highly appreciated, which prompted me to pursue this project.
References
- Brenner, RM, Slayden, OD: Cyclic changes in the primate oviduct and endometrium. The physiology of reproduction. 2nd ed. E. Knobil, JD Neal, pp541-567, Raven Press, 1994, NT, NY.
- Bartelmez, GW: The phases of the menstrual cycle and their interpretation in terms of pregnancy cycle. Am J Obstet Gynecol 1957, 74(5):931-955.
- Dallenbach, FD, Vonderlin, D: The innervation of the human endometrium. Arch fur Gynekologie 1973, 15(4):365-376.
- Latini, C, Fratini, A, Morroni, M, Marzioni, D, Castelluci, M: Remodeling of uterine innervation. Cell Tissue Res 2008, 334(1):1-6.
- Padylula, HA, Coles, LG, McCracken, JA, King, NWJr, Lungcope, C et al: A zonal pattern of cell proliferation in rhesus endometrium during the estrogen surge. Biol Repro 1984, 31(5):1103-1118.
- Okulicz, WC, Ace, CI, Scarrell, R: Zonal changes in proliferation in the rhesus endometrium during late secretary phase and menses. Proc Exp Bil Med 1997, 214(2):132-138.
- Donnez, O, Soares, M, Defrere, S, Dehoux, JP, Langrendonickt, M: Nerve density in deep nodular endometric lesion induced in baboon. Fertil Steril 2023, 100(4):1144-1150.
- Quinn, MJ, Kirk, N: Differences in uterine innervation at hysterectomy. Am J Obstet Gynecol 2002, 187(6):1515-1520.
- Tokushige, N, Markham, R, Russell, P, Fraser, IS: High density of small nerve fibers in the functional layer of the endometrium in women with endometriosis. Hum Reprod 2006, 21(3):782-787.
- Tokushige, N, Markham, R, Russell, P, Fraser, IS: Different types of small nerve fibers in eutopic endometrium and myometrium in women with endometriosis. Fertil Steril 2007, 88(4):795-803.
- Tokushige, N, Markham, R, Russell, P, Fraser, IS: Effects of hormonal treatment on nerve fibers in endometrium and myometrium in women with endometriosis. Fertil Steril 2007, 90(5):1589-803.
- Tokushige, N, al-Jefout, M, Salih, H, Fraser, IS: Endometrial nerve fibers in endometriosis. Iran J Reprod Med 2007, 5(3):81-88.
- Zhang, X, Lu, B, Huang, X, Su, H, Zhou, C et al: Inner- vation of endometrium and myometrium in women with painful adenomyosis and uterine fibroids. Fertil Steril 2010, 94(2):1300-1307.
- Boker, A, Kyma, CM, Vercruysse, L, Fassbender, A, Gevaert, O et al: Density of small diameter sensory nerve fibres in endometrium: semi-invasive diagnostic test for mining to mild endometriosis. Hum Reprod 2009, 24(12):3025-3032.
- Fraser, IS, Bersinger, NA: Detection of pan neuronal marker PGP9.5 in eutopic endometrium from women with and without endometriosis. Arch Gynecol Obstet 2015, 191(1):85-91.
- Yadav, G, Rao, M, Gothwal, M, Singh, P, Kathuria, P et al: Detection of nerve fibers in the eutopic endometrium of women with endometriosis, uterine fibroids and adenomyosis. Obstet Gynecol 2021, 64(5):454-461.
- Zavallos, HBV, McKinnon, B, Tokushige, N, Miller, MD, Fraser, IS et al: Detection of pan neuronal marker PGP9.5 in eutopic endometrium from women with and without endometriosis. Arch Gynecol Obstet 2015, 291(1):85-91.
- Atwal, G, du Plessis, D, Arnstrove, G, Slade, R, Quinn, M: Uterine innervation after hysterectomy for chronic pelvic pain with and without endometriosis. Am J Obstet Gynecol 2005, 193(5):1650-1655.
- Schlaeffer, WW: Neurofilaments: Structure and implication in disease. J Neuropath Exp Biol 1987, 46(2):46(2):117-119.
- Yuan, A, Rao, MV, Veervanna, Nixon, RA: Neurofilaments at a glance. J Cell Sci, 2012, 125(14):3257-3263.
- Mechtersheimer, G, Stauder, M, Moller, P: Expression of the natural killer cell-associated CD56 and CD57 in human neural and striated muscle cells and their tumors. Cancer Res 1991, 51(4):1300-1307.
- Tomita, T, Mah, K: Lymphatic and blood vessels in normal rhesus monkey organs by immunohistochemical staining with frozen sections: Structure and function relationship. Med Res Arch 2024, 12(9).
- Tomita, T: Immunohistochemical staining for nerve fibers in normal tissues with frozen sections. Structure and function relationship. Med Res Arch 2023, 11(6).
- Hogden, GD: Surrogate embryo transfer combined with estrogen-progesterone therapy in monkeys. JAMA 1983, 350(16):2167-2171.
- Slayden, OD, Koji, T, Brenner, RM: Microwave stabilization enhances immunocytochemical detection of estrogen receptor in frozen sections of macaque oviduct. Endocrinol 1995, 136 (9):4012-4021.
- Nayak, NR, Critchley, HOD, Slayden, OD, Menrad, A, Schwalisz, K et al: Progesterone withdrawal up-regulates vascular endothelial growth factor type 2 in the superficial zone stroma of the human and macaque endometrium. J Clin Endocrinol Metab 2000, 85(9):3443-3452.
- Slayden, OD, Nayak, NR, Burton, KA, Chwalisz, K, Cameron, ST et al: Progesterone antagonists increase androgen receptor expression in the rhesus macaque and human endometrium. J Clin Endocrinol Metab 2001, 86(6):2668-2679.
- Cao, W, Mah, K, Carroll, RS, Slayden, OD, Brenner, RM: Progesterone withdrawal up-regulates fibronectin and integrin during menstruation and repair in the rhesus macaque endometrium. Hum Reprod 2007, 22(12):3223-3231.
- Jackson, P, Thompson, RJ: The dementia of new human brain-specific protein by high-resolution two-dimensional polyacrylamide gel electrophoresis. J Neurol Sci 1981, 49(3):429-438.
- Campbell, LK, Thomas, JR, Lamps, LW, Schmdler, BR, Folge, AL: Protein gene product 9.5 (PGP9.5) is not a specific marker of neural and nerve sheath tumors. An immunohistochemical study of 95 mesenchymal neoplasms. Mod Pathol 2003, 16(10):963-969.
- Tokushige, N, Markham, R, Russell, P, Fraser, IS: Effects of hormonal treatment in endometrium and myometrium in women with endometriosis. Fertil Steril 2008, 90(5):1589-1598.
- Leendecker, G, Horbertz, M, Kunz, M: Endometriosis results from the dislocation of basal endometrium. Human Reprod 2002, 17(10):2725-2736.
- Gargett, C, Masuda, H: Adult stem cells in the endometrium. Molec Human Reprod 2010, 16(11):818-834.
- Zoubina, EV, Fan, Q, Smith, PG: Variation in uterine innervation during estrous cycle in rat. J Comp Neurol 1998, 397(4):561-571.
- Berkley, K, Dmitrieva, N, Cutis, KC, Papka, RE: Innervation of ectopic endometrium in a rat model of endometriosis. Proc Aca Sci USA 2004, 101(30):11094-11098.
- Rosenwak, Z, Seegar-Jones, G: Menstrual pain: its origin and pathogenesis. J Repro Med, 1980, 25(4 suppl):207-212.
- Kobayashi, H, Yamada, Y, Morioka, S, Niiro, E, Shigematsu, A et al: Mechanism of pain generation for endometriosis-associated pelvic pain. Arch Gynecol Obstet 2014, 289(1):13-21.
- Pinsard, M, Mouchet, N, Dions, L, Bessede, T, Bertrand, M, et al: Anatomic and functional mapping of human uterine innervation. Fertil Steril 2022, 117(6):1279-1288.
- Brauer, A, Smith, PG: Estrogen and female reproductive tract innervation: cellular and molecular mechanisms of autonomic neuroplasticity. Auton Neurosci 2014, 187(1):1-17.
- Astruc, A, Roux, L, Robin, F, Sall, NS, Dion, L et al: Advanced insights into human uterine innervation: Implication for endometriosis and pelvic pain. J Clin Med 2024, Mar.1, 13(5):1433.

