Endotracheal Tubes: Key Factors in Ventilator-Associated Pneumonia
Ventilator associated pneumonia- Endotracheal tubes the real culprits. A review
Peter R Lichtenthal1, MD Ulf Borg, BS2
- University of Arizona School of Medicine, Tucson, AZ
- Mead CO
OPEN ACCESS
PUBLISHED: 31 March 2025
CITATION: Lichtenthal, P., Borg, U., 2025. Ventilator associated pneumonia- Endotracheal tubes the real culprits. A review. Medical Research Archives, [online] 13(3). https://doi.org/10.18103/mra.v13i3.6454
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i3.6454
ISSN 2375-1924
ABSTRACT
In this review we will outline the reasons for the clinical assumption that the endotracheal tube is the main source of VAP. We have used literature searches in PubMed, EMBASE and Google to obtain the information included in this review. Ventilator-associated pneumonia (VAP) is a hospital acquired infection of the lung that occurs after approximately 48 h of tracheal intubation and mechanical ventilation. We consider VAP a misnomer, the true cause of this infection of lung parenchyma is not primarily caused by mechanical ventilation but by presence of an endotracheal tube. The presence of an endotracheal tube (ETT) represents a major risk factor, as it disrupts the natural protective barriers of the upper airway, allowing direct access to the tracheobronchial tree. As a result, many different endotracheal tube designs have been introduced including different cuff designs, silver coated tubes and tubes with supraglottic suction capabilities. None of these tube designs have significantly decreased the incidence of VAP. Other clinical strategies to reduce the incidence include patient positioning, gastric decontamination and mouth hygiene with antimicrobial soap. Only a combination of the above-mentioned strategies has demonstrated reduction in the incidence of VAP. The prevention and diagnosis of VAP continues to present a clinical conundrum although many factors are involved, we state that the major culprit is the introduction of endotracheal intubation longer than 48 hours. With the introduction of AI the development of prevention and treatment algorithms in concert with clinical and experimental research is required to continue to ameliorate this life threatening complication.
Keywords
Ventilator-associated pneumonia, endotracheal tube, infection, mechanical ventilation, prevention strategies
Introduction
Ventilator-associated pneumonia (VAP) is a nosocomial infection of the lung that occurs after approximately 48 h of tracheal intubation and mechanical ventilation. Up to 36% of critically ill patients are affected and this induces significant antibiotic prescription use which accounts for as much as half of all antibiotic use in the ICU. VAP also increases hospital length of stay and healthcare costs and is associated with increased patient morbidity and mortality. Over the years many clinical protocols have been proposed to reduce the incidence of VAP. We consider VAP a misnomer, the true cause of this infection of lung parenchyma is not primarily caused by mechanical ventilation but by presence of an endotracheal tube. As a result many different endotracheal tube designs have been introduced including different cuff designs, silver coated tubes and tubes with supraglottic suction capabilities. None of these tube designs have significantly decreased the incidence of VAP. The first cuffs were made from latex and require high pressures to seal the airway. The high-pressure seal protected the airways from gastric content aspiration, however, caused major airway injuries. A different cuff design was introduced in the 70s. This cuff had a diameter that was larger than the tracheal diameter and resulted in less pressure on the tracheal wall compared to the latex cuffs. The issue with the new design is that since it is larger than the tracheal diameter the cuff does not completely inflate. The resulting folds in the cuff material allow fluids to leak past the cuff into the airways what has been termed microaspiration. Several studies have demonstrated the fluid leak past the cuff both during animal and human studies. Other clinical strategies to reduce the incidence include patient positioning, gastric decontamination and mouth hygiene with antimicrobial soap. Only a combination of the above-mentioned strategies has demonstrated reduction in the incidence of VAP. In this review we will outline the reasons for the clinical assumption that the endotracheal tube is the main source of VAP. We have used literature searches in PubMed, EMBASE and Google to obtain the information included in this review.
Etiology
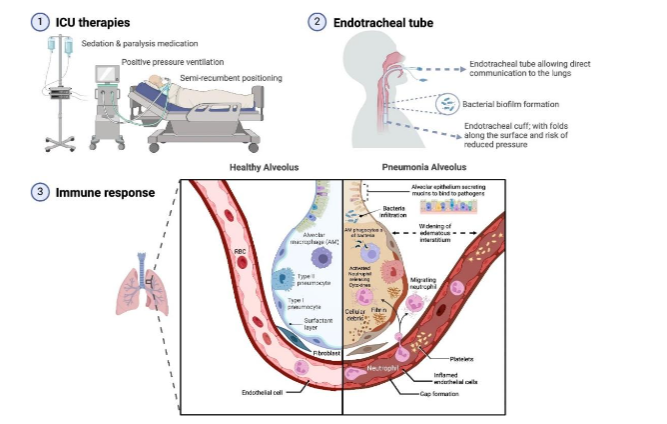
Ventilator-associated pneumonia should not be confused with tracheitis. Tracheitis is the presence of clinical signs of purulent tracheal discharge, fevers, respiratory distress, and the presence of bacteria and white blood cells in the tracheal aspirate without radiological signs of pneumonia. Ventilator-associated pneumonia includes clinical signs of purulent tracheal discharge, fevers, respiratory distress, and microbiological signs of the presence of microorganisms along with white blood cells in the tracheal aspirate along with radiological evidence of pneumonia. Ventilator-associated pneumonia is most typically bacterial. Common organisms are Staphylococcus aureus (28.4 %), Pseudomonas aeruginosa (25.2 %), and other gram negatives such as Klebsiella and Enterobacter species (26.6%). In a large retrospective review completed in the ICU settings of three hospitals, microbiology was the same across adult and pediatric hospitals however, polymicrobial infections are increasing. Anaerobic bacteria are a rare source of Ventilator-associated pneumonia and can play a role in polymicrobial infections. Nosocomial viruses and fungi are rare causes of pneumonia in immunocompetent hosts. Antibiotic-sensitive community-acquired organisms is most likely the cause of early pneumonia (less than 4 days after admission), and late pneumonia (more than 4 days) is more likely due to antibiotic-resistant organisms. The presence of an endotracheal tube (ETT) represents a major risk factor, as it disrupts the natural protective barriers of the upper airway, allowing direct access to the tracheobronchial tree. A volume, low-pressure cuff may minimize gross aspiration but does not prevent micro aspirations. Other risk factors include entrainment of pooled secretions, loss of cuff pressure and ETT movement. Silent microaspiration may be a result of pooled secretions from the oropharynx, nasal sinuses and stomach, which are susceptible to leaking below the cuff. Epithelial damage and impeded cough, swallowing and mucociliary clearance are all results of the ETT presence. ICU therapies, such as sedation, paralysis, positive pressure ventilation and the influence of gravity associated with semi-recumbent positioning enhance respiratory compromise. Biofilm initiates within hours of intubation and is found on the ETT surface in 95% of mechanically ventilated patients is an additional source of VAP. The biofilm can become dislodged and acts as a reservoir for infective microorganisms, which may enter the distal airways and subsequently lead to VAP. The longer the duration of mechanical ventilation, the greater the incidence of VAP.
Treatment and Management
Once ventilator-associated pneumonia is suspected and appropriate culture samples are sent, empirical therapy will be based on the duration of intubation and hospitalization, prior or current antibiotic therapy, the severity of clinical susceptibility patterns. Initial broad-spectrum therapy with coverage of gram-negative bacilli, including P. aeruginosa, and possibly methicillin-resistant S. aureus is generally appropriate. Treatment is then narrowed based on subsequent culture data and clinical and radiographic findings. Below are the risk factors which when mediated will reduce the incidence of ventilator-associated pneumonia: (1) treatment strategies that reduce the incidence of colonization of oropharynx or stomach, (2) strategies that reduce gastric reflux and aspiration like depressed mental status, supine position, nasogastric tubes, (3) reduction of duration of intubation and mechanical ventilation, and (4) remove factors that interfere with adequate pulmonary toilet, thoracic or abdominal surgery or immobilization. Additional strategies for decreasing the incidence of ventilator-associated pneumonia are: (1) judicious use of reflux medications, (2) removal of unnecessary nasogastric tubes, and (3) daily evaluation of readiness to extubate, pain-control, and maintenance of mechanical ventilation system are all part of protocols ICUs use to decrease the rate of ventilator-associated pneumonia. The use of endotracheal tube with subglottic suction for removal of potential harmful matter may be controversial. We include a review of this clinical intervention which was our response to Society for Healthcare Epidemiology (SHEA). The recommendations attempted to assist hospitals in prioritizing and implementing strategies to prevent ventilator-associated pneumonia (VAP) in adults, children, and neonates. These new recommendations update their published 2014 prevention strategies, and several recommendations have been added, removed, or changed. According to the new guidelines, although subglottic secretion drainage (SSD) has been shown to lower VAP rates, there is insufficient evidence about its impact on the duration of mechanical ventilation, length of stay (LOS), mortality, and costs. However, 2 recent meta-analyses have demonstrated that use of endotracheal tubes (ETTs) with SSD reduced VAP rates by 44%-50%. Further, routine use of SSD may reduce the risk of postoperative VAP in patients undergoing cardiac surgery, with those undergoing intraoperative continuous and intermittent SSD reporting a 70% reduction in the rate of postoperative VAP. Although these findings are very promising, they were not considered when reclassifying SSD recommendations in the new guidelines. Studies that have evaluated reductions in the duration of mechanical ventilation with SSD are limited to patients expected to require >48-72 hours of mechanical ventilation. However, it is difficult to determine, at the time of intubation, which patients will remain on mechanical ventilation >48 hours. Even so, a recent meta-analysis reported that SSD delayed time to VAP by 2.66-4.04 days. In regard to cuff material, the new guidelines contend that ultrathin polyurethane cuffs are inconsistently associated with lower VAP rates and have no, or negative, impact on duration of mechanical ventilation, LOS, or mortality. The main reference used to make this determination was a study that reported that polyurethane cuffs did not reduce bacterial colonization or VAP compared with cylindrical PVC cuffs. However, tracheal colonization was already present at the time of intubation in several patients, and determining whether the cuff influenced tracheal colonization in these patients was difficult. Regarding ETT cuff shape, the guidelines suggest that tapered ETTs are inconsistently associated with lower VAP rates and have no, or negative, impact on duration of mechanical ventilation, LOS, or mortality. The only referenced study for tapered cuffs is a meta-analysis that reported no difference in VAP rates or outcomes when tapered cuffs were used. However, additional factors, including cuff underinflation, may influence risk of microaspiration over time. None of the included studies accounted for cuff-pressure management in the prevention of VAP. Underinflation, even when episodic, can lead to microaspiration of secretions regardless of cuff shape. In previous research examining intraoperative aspiration and its association with postoperative pneumonia using dye above the cuff at time of intubation, the use of tapered-shaped cuffs had a protective role against aspiration (ie, no dye leaked into the trachea). This publication was not cited in the new guidelines. We understand that demonstrating that any of the ETT characteristics mentioned reduces mortality, LOS or days on ventilation, a sample size of thousands would be necessary for a single randomized controlled trial. The prevention of VAP is achieved through a bundled approach, with a variety of measures and interventions instituted at the same time to reduce the incidence of VAP. An evaluation of different strategies to reduce VAP reported that very high compliance rates, >90%, were significantly associated with reduction in VAP rates, with long-term compliance contributing to VAP rates close to zero. No single characteristic of the ETT will result in significant reduction of VAP; however, neglecting the published information does not justify recommendations to not use the devices.
Discussion
The cuff on the endotracheal tube plays an important role in protecting the airways from gastric content while allowing for positive pressure ventilation. The presence of an endotracheal tube (ETT) represents a major risk factor for VAP, as it disrupts the natural protective barriers of the upper airway, allowing direct access to the tracheobronchial tree. A volume, low-pressure cuff may minimize gross aspiration but does not prevent micro aspirations. Additional risk factors include entrainment of pooled supraglottic secretions, poor cuff pressure management and ETT movement during change in position of the patient. Silent micro-aspiration may be a result of pooled secretions from the oropharynx, nasal sinuses and stomach, which may be accessing the airways through folds in the cuff. Epithelial damage and impeded cough, swallowing and mucociliary clearance are all results of the ETT presence. Common ICU therapies, such as sedation, paralysis, positive pressure ventilation and the influence of gravity associated with semi-recumbent positioning adds to the risk of developing VAP. Therefore, we disagree with the published SHEA recommendation of the utilization of ETTs with lumen for subglottic secretion drainage (SSD) and agree with Lorente et.al that it can help prevent VAP in patients requiring mechanical ventilation for more than 48 hours.
Conclusion
The diagnosis of VAP continues to present a clinical conundrum although many factors are involved, we state that the major culprit is the introduction of endotracheal intubation longer than 48 hours. With the introduction of AI the development of prevention and treatment algorithms in concert with clinical and experimental research is required to continue to ameliorate this life threatening complication.
Conflict of Interest:
Peter Lichtenthal: I have no conflict of interest with any parties related to the material discussed in this manuscript.
Ulf Borg: have no conflict of interest with any parties related to the material discussed in this manuscript.
Funding Statement:
None.
Acknowledgements:
None.
References:
- Kollef, M. H. What is ventilator-associated pneumonia and why is it important? Respir. Care 2005, 50, 714-721.
- Barbier, F., Andremont, A., Wolff, M. & Bouadma, L. Hospital-acquired pneumonia and ventilator-associated pneumonia: recent advances in epidemiology and management. Curr. Opin. Pulm. Med. 2013, 19, 216-228.
- Timsit, J. F., Esaied, W., Neuville, M., Bouadma, L. & Mourvllier, B. Update on ventilator-associated pneumonia. F1000Res 2017 6, 2061.
- Safdar, N., Dezfulian, C., Collard, H. R. & Saint, S. Clinical and economic consequences of ventilator-associated pneumonia: a systematic review. Crit. Care Med. 2005, 33, 2184-2193.
- Zolfaghari, P. S. & Wyncoll, D. L. The tracheal tube: gateway to ventilator-associated pneumonia. Crit. Care 2011, 15, 310.
- GoleNaz A. Kohbodi; Venkat Rajasurya; Asif Noor Ventilator-Associated Pneumonia. NCBI Bookshelf. StatPearls Publishing; 2025 Jan-. 1-7 NBK [507711].
- W M Shelly, R B Dawson, I A May. Cuffed tubes as a cause of tracheal stenosis J Thorac Cardiovasc Surg. 1969 May;57(5):623-7.
- Colice GL Technical standards for tracheal tubes. Clin Chest Med. 1991 Sep;12(3):433-48.
- Lichtenthal PR, Maul D, Borg U. Do tracheal tubes prevent microaspiration? Br J Anaesth. 2011 Nov;107(5):821-2. doi: 10.1093/bja/aer312.
- Jain V, Vashisht R, Yilmaz G, Bhardwaj A. StatPearls [Internet]. StatPearls Publishing; Treasure Island (FL): Jul 31, 2023. Pneumonia Pathology. [PubMed: 30252372].
- Iosifidis E, Pitsava G, Roilides E. Ventilator-associated pneumonia in neonates and children: a systematic analysis of diagnostic methods and prevention. Future Microbiol. 2018 Sep;13:1431-1446.
- Lynch, J. P.III. Hospital-acquired pneumonia: risk factors, microbiology, and treatment. CHEST 119, 2001, 373S-384SS.
- Mietto, C., Pinciroli, R., Patel, N. & Berra, L. Ventilator associated pneumonia: evolving definitions and preventive strategies. Respir. Care 2013, 58, 990-1007.
- Young, P. J., Pakeerathan, S., Blunt, M. C. & Subramanya, S. A low-volume, low-pressure tracheal tube cuff reduces pulmonary aspiration. Crit. Care Med. 2006, 34, 632-639.
- Carter, E. L. et al. Strategies to prevent ventilation-associated pneumonia: the effect of cuff pressure monitoring techniques and tracheal tube type on aspiration of subglottic secretions: an invitro study. Eur. J. Anaesthesiol. 2014, 31, 166-171.
- Craven, D. E. & Hjalmarson, K. I. Ventilator-associated tracheobronchitis and pneumonia: thinking outside the box. Clin. Infect. Dis. 2010, 51, S59-S66.
- Niederman, M. S. The clinical diagnosis of ventilator-associated pneumonia. Respir. Care 2005, 50, 788-796 discussion 807-12.
- Jackson, L. & Owens, M. Does oral care with chlorhexidine reduce ventilator-associated pneumonia in mechanically ventilated adults? Br. J. Nurs. 2019, 28, 682-689.
- Goetz, R. L., Vijaykumar, K. & Solomon, G. M. Mucus clearance strategies in mechanically ventilated patients. Front Physiol. 2022, 13, 834716.
- Konrad, F., Schreiber, T., Brecht-Kraus, D. & Georgieff, M. Mucociliary transport in ICU patients. Chest 1994, 105, 237-241.
- Chastre, J. & Fagon, J. Y. Ventilator-associated pneumonia. Am. J. Respir. Crit. Care Med. 2002, 165, 867-903.
- Lorente, L., Lecuona, M., Jiménez, A., Mora, M. L. & Sierra, A. Ventilator-associated pneumonia using a heated humidifier or a heat and moisture exchanger: a randomized controlled trial [ISRCTN88724583]. Crit. Care 2006, 10, R116.
- Deem, S. & Treggiari, M. M. New endotracheal tubes designed to prevent ventilator-associated pneumonia: do they make a difference? Respir. Care 2010, 55, 1046-1055.
- Adair, C. G. et al. Implications of endotracheal tube biofilm for ventilator-associated pneumonia. Intensive Care Med. 1999, 25, 1072-1076.
- Morris A. C. Management of pneumonia in intensive care. J. Emgy Crit. Care Med. 2018, 2 https://doi.org/10.21037/jeccm.2018.11.06.
- Biel, M. A. et al. Reduction of endotracheal tube biofilms using antimicrobial photodynamic therapy. Lasers Surg. Med. 2011, 43, 586-590.
- Wu, D., Wu, C., Zhang, S. & Zhong, Y. Risk factors of ventilator-associated pneumonia in critically III patients. Front Pharm. 2019, 10, 482.
- Chomton M, Brossier D, Sauthier M, Vallières E, Dubois J, Emeriaud G, Jouvet P. Ventilator-Associated Pneumonia and Events in Pediatric Intensive Care: A Single Center Study. Pediatr Crit Care Med. 2018 Dec;19(12):1106-1113. [PubMed: 30234676].
- Martin-Loeches I, Rodriguez AH, Torres A. New guidelines for hospital-acquired pneumonia/ventilator-associated pneumonia: USA vs. Europe. Curr Opin Crit Care. 2018 Oct;24(5):347-352. [PubMed: 30063491].
- Niederman MS. Antibiotic treatment of hospital-acquired pneumonia: is it different from ventilator-associated pneumonia? Curr Opin Crit Care. 2018 Oct;24(5):353-360. [PubMed: 30028739].
- Klompas M, Branson R, Cawcutt K, Crist M, Eichenwald EC, Greene LR, Lee G, Maragakis LL, Powell K, Priebe GP, Speck K, Yokoe DS, Berenholtz SM. Strategies to prevent ventilator-associated pneumonia, ventilator-associated events, and nonventilator hospital-acquired pneumonia in acute-care hospitals: 2022 Update Infect Control Hosp Epidemiol. 2022 Jun;43(6):687-713. doi: 10.1017/ice.2022.88. Epub 2022 May 20.
- Klompas, M, Branson, R, Eichenwald, EC, et al. Strategies to prevent ventilator-associated pneumonia in acute-care hospitals: 2014 update. Infect Control Hosp Epidemiol 2014;35 suppl 2:S133-S154. doi: 10.1017/S0899823X00193894.
- Pozuelo-Carrascosa, DP, Herráiz-Adillo, Á, Alvarez-Bueno, C, Añón, JM, Martínez-Vizcaíno, V, CaveroRedondo, I. Subglottic secretion drainage for preventing ventilator-associated pneumonia: an overview of systematic reviews and an updated meta-analysis. Eur Respir Rev 2020;29:190107. doi: 10.1183/16000617.0107-2019.
- Sanaie, S, Rahnemayan, S, Azizi, S, et al. Comparison of subglottic vs nonsubglottic secretion drainage in prevention of ventilator-associated pneumonia: a systematic review and meta-analysis. Trends Anaesthesia Crit Care 2022;43:23-29. doi: 10.1016/j.tacc.2022.02.002.
- K, Park, J-B, Park, WB, et al. Effect of perioperative subglottic secretion drainage on ventilator-associated pneumonia after cardiac surgery: a retrospective, before-and-after study. J Cardiothor Vasc Anesth 2021;35:2377-2384. doi: 10.1053/j.jvca.2020.09.126.
- Lacherade, J-C, Azais, M-A, Pouplet, C, Colin, G. Subglottic secretion drainage for ventilator-associated pneumonia prevention: an underused efficient measure. Ann Translat Med 2018;6:422. doi: 10.21037/atm.2018.10.40.
- Philippart, F, Gaudry, S, Quinquis, L, et al. Randomized intubation with polyurethane or conical cuffs to prevent pneumonia in ventilated patients. Am J Resp Crit Care Med 2015;191:637-645. doi: 10.1164/rccm.201408-1398OCC.
- Maertens, B, Blot, K, Blot, S. Prevention of ventilator-associated and early postoperative pneumonia through tapered endotracheal tube cuffs: a systematic review and meta-analysis of randomized controlled trials. Crit Care Med 2018;46:316-323. doi: 10.1097/CCM.0000000000002889.
- Rompaey, K, Umbrain, V, Poelaert, J. Assessment of intraoperative microaspiration: does a modified cuff shape improve sealing? Acta Anaesthesiol Scand 2013;57:873-880. doi: 10.1111/aas.12119.
- Alecrim, RX, Taminato, M, Belasco, A, Longo, MCB, Kusahara, DM, Fram, D. Strategies for preventing ventilator-associated pneumonia: an integrative review. Revista brasileira de enfermagem 2019;72:521-530. doi: 10.1590/0034-7167-2018-0473.
- Leonardo Lorente, María Lecuona, Alejandro Jiménez, Lisset Lorenzo, Isabel Roca, Judith Cabrera, Celina Llanos, María L Mora. Continuous endotracheal tube cuff pressure control system protects against ventilator-associated pneumonia. Crit Care. 2014 Apr 21;18(2):R77. doi: 10.1186/cc13837.

