Enhancing Food Quality through Soil and Crop Management
Enhancing Nutritional Quality of Food by Improved Soil and Crop Management
Kentu Lassiter¹, Garrett Mullenix², Sam Rochell³, Guillermo Tellez⁴, Michale T. Kidd¹, Sami Dridi¹, and Walter Bottje¹*
- Center of Excellence for Poultry Science, Division of Agriculture, University of Arkansas, Fayetteville 72701, USA.
- Cargill Corporation, Minneapolis, MN55440, USA.
- Auburn University, Dept. of Poultry Science, Auburn, AL 36849, USA.
- Consultant
OPEN ACCESS
PUBLISHED:30 November 2024
CITATION:Lassiter, K., Mullenix, G., et al., 2024. Effects of a low protein soybean meal diet with and without Spirulina platensis freshwater microalgae on antioxidant systems in broiler liver and muscle tissue. Medical Research Archives, [online] 12(11).https://doi.org/10.18103/mra.v12i11.5697
COPYRIGHT © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i11.5697
ISSN 2375-1924
Abstract
Malnutrition, deficiency of essential micronutrients along with protein and vitamins, affects 2-3 billion people including children below the age of 5, nursing mothers and elderly population. Soil depletion and degradation, affecting 40% of agroecosystems, is one of the factors with adverse effects on nutritional quality of the food. Healthy food is obtained from plants and animals grown on healthy soils. Thus, adoption of proven and innovative science-based options for specific soil/ecoregions and cropping/farming systems are needed to alleviate soil-related constraints and produce nutritious and safe food. Soil pollution and contamination, especially that with some heavy metals (i.e., lead (Pb), mercury (Hg), and arsenic (As)), is another issue because the food may contain toxic levels of some heavy metals and or pesticide residues. Biofortification with micronutrients, such as for selenium (Se) in some Scandinavian countries, can also alleviate the micronutrient deficiencies. Breeding of crops and animals for site-specific situation is another option to promote the “One Health Concept” which states that health of soil, plants, animals, people, ecosystems and planetary processes is one and indivisible. Improving education and implementing policies which reward farmers to adopt modern innovations are among other options to improve diet quality.
Keywords
Malnutrition, Nutritional Quality, Soil Health, Crop Management, Agroecosystems
Introduction
As the world’s population continues to increase and resources become more limited, sustainability and improved efficiency in poultry production has taken on greater interest across the industry. One specific issue facing the poultry industry is providing a cost-effective source of protein in broiler diets. Currently, soybean meal is the preferred choice of protein in broiler diets due to its relatively low cost. However, global human population growth, and the resulting increase in the demand for animal protein has begun to impact the price and availability of soybean meal. Furthermore, using soybean for animal feed competes with its use as a protein source for humans. This issue has led to the search for alternative protein sources (which maintain or improve growth performance and overall bird health) that can be used in poultry diets.
A number of reports have indicated that Spirulina (Arthrospira) platensis is a promising candidate for use as a protein source in animal feed. Spirulina is a blue-green microalgae (cyanobacteria) that naturally grows in marine or freshwater aquatic systems and has good nutritional value due to its high protein content, and a good balance of essential amino acids and fatty acids¹². Earlier studies investigating the use of Spirulina as a feed supplement in livestock animals (e.g.³, including chickens, pigs, cattle, sheep, and rabbits) indicate that the algae potentially offers an improvement in health, production, and meat quality of the animals. A positive attribute of Spirulina in terms of contributing to sustainable poultry feed production is that it can be produced under a variety of environmental conditions with minimal land requirements; whereas soybeans are best suited for growth in (sub-) tropical climates, require more land for cultivation, and have a larger environmental footprint⁴. In addition to its nutritional value and sustainable production, Spirulina contains beneficial bioactive compounds such as phenolic acids, vitamins, minerals, and gamma linoleic acid⁵⁻⁷. Spirulina is also rich in pigments such as carotenoids, specifically xanthophylls and carotenes, and the plant protein phycocyanin, all of which contribute to excellent antioxidant/anti-inflammatory capabilities⁸⁻⁹. The antioxidant/anti-inflammatory compounds contained in the algae are of particular interest as it relates to poultry production. The inclusion of Spirulina in poultry diets has been reported in a number of studies. Birds raised under conventional conditions have shown improved growth performance¹⁰ as well as improved antioxidant activity (Park et al.¹¹) when Spirulina is added to the diet. However, Bonos et al¹² did not show any effect of Spirulina on broiler performance. Recently, Mullenix et al.¹ also showed that the inclusion of Spirulina in low-protein broiler diets reduced the levels of pro-inflammatory cytokines in the circulation¹³. The benefits of algae inclusion on broiler performance and antioxidant status was apparent under heat stress conditions, where the induction of oxidative stress is a major concern¹⁴. Heat stress increased the production of damaging reactive oxygen species in the mitochondria of broiler skeletal muscle¹⁴⁻¹⁵. Other studies reported that adding Spirulina to the diet of heat-stressed broilers improved and/or restored growth performance concomitant with increased antioxidant activity²⁶¹⁶⁻¹⁷.
Numerous antioxidants work in concert in the cell to combat the constant production of oxidants coming from extracellular and intracellular sources (see review by Yu¹⁸). An overview of several redox coupled reactions in the cell is provided in Figure 1. This antioxidant/free radical scavenging milieu are often found in specific cellular compartments (e.g. cytosol and mitochondria) and can interact to rejuvenate each other by donating reducing equivalents. The major nonenzymatic antioxidant in the cell is reduced glutathione (GSH) which is found in mM concentrations in most cells. The rate limiting amino acid and enzyme for synthesis of GSH is cysteine and glutamate cysteine ligase (GCL), respectively. An interorgan circulation of GSH in which GSH is exported from the liver and taken up by other tissues, was first reported by Anderson et al.¹⁹ and later confirmed in broilers Wang et al.²⁰. Thioredoxin is also an important thiol-containing antioxidant in the cell that helps convert numerous oxidized forms of antioxidants back to their reduced state (see review by Arner and Holmgren²¹) via redox reactions. The GSH recycling enzyme system consists of GSH peroxidase that reduces hydrogen and lipid peroxides using reducing equivalents from GSH to form oxidized glutathione (GSSG) that is recycled back to GSH by the action of GSH reductase using reducing equivalents from NADPH. Superoxide dismutase (SOD) catalyzes the reduction of superoxide to hydrogen peroxide. Isoforms of GSH peroxidase and SOD can be found within the cytosol and mitochondrial compartments.
Anderson et al.¹⁹ and later confirmed in broilers Wang et al.²⁰. Thioredoxin is also an important thiol-containing antioxidant in the cell that helps convert numerous oxidized forms of antioxidants back to their reduced state (see review by Arner and Holmgren²¹) via redox reactions. The GSH recycling enzyme system consists of GSH peroxidase that reduces hydrogen and lipid peroxides using reducing equivalents from GSH to form oxidized glutathione (GSSG) that is recycled back to GSH by the action of GSH reductase using reducing equivalents from NADPH. Superoxide dismutase (SOD) catalyzes the reduction of superoxide to hydrogen peroxide. Isoforms of GSH peroxidase and SOD can be found within the cytosol and mitochondrial compartments.
Factors Affecting Global Food and Nutrition Security
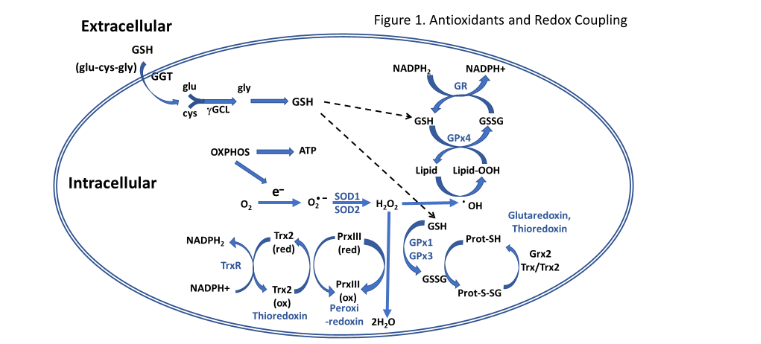
Figure 1. An overview of antioxidant systems and reduction-oxidation reactions in the cell involved in free radical scavenging and antioxidant protection (modified from Bottje, 2017). (See text for details.)
It is well documented that an increase in oxidative stress negatively impacts growth performance of broilers. In several studies comparing birds from the same genetic lineage, broilers with a low feed efficiency phenotype were shown to have higher levels of oxygen radicals and/or protein carbonyls (indicative of oxidative damage to proteins) in several tissues, including breast muscle²², heart muscle²³, liver²⁴⁻²⁵, duodenum²⁶, and lymphocytes²⁷. The inherently elevated metabolic activity in the liver and breast muscle of broilers means that these tissues are potentially more susceptible to oxygen radical production, and thus an increase in oxidative damage. The studies by Bonos et al.¹² and Park et al.¹¹ determined effects of Spirulina on blood antioxidant levels, but did not report effects on tissue antioxidants. It would be beneficial to also determine what antioxidant changes are occurring in the tissues as well as to gain a more complete understanding of how including algae may be beneficial to broiler production. It should be noted that the broilers whose breast muscle were used in the current study were obtained from the same trial conducted and reported by Mullenix et al.¹. The purpose of this study was to determine if Spirulina supplementation in a low-protein broiler diet affected antioxidant status in breast muscle and liver tissues of male broilers, specifically looking at changes in antioxidant gene expression, as well as activities of key antioxidant enzymes, and nonenzymatic concentrations.
Materials and Methods
ANIMAL CARE STATEMENT
The humane care and treatment of animals were approved by the University of Arkansas Institutional Animal Care and Use Committee (IACUC) (IACUC protocol #21002).
ANIMALS AND TISSUES
Ross 708 male broilers were reared in floor pens and provided access to feed and water ad libitum. From day 1 to day 14, all birds were provided a standard commercial starter diet. From d 15 to 37,
the birds were divided into three groups and provided one of three grower diets: 1) A grower diet (Positive control) consisting of a standard corn/soybean meal diet (CON), 2) a negative control, consisting of a low crude protein diet (LCP), and 3) the low crude protein diet in which Spirulina platensis meal was included at 10%, which represented half of the whole protein sources in the LCP plus microalgae (LCP+AL) diet as described by Mullenix et al.¹. Growth performance traits (weight gain, feed intake, feed conversion ratio, mortality) were reported in Mullenix et al. (2021). At the end of the trial, 10 birds were randomly selected from each treatment group, humanely euthanized by cervical dislocation, and breast muscle and liver tissues collected. The collected tissues were immediately frozen in liquid nitrogen and stored at −80°C until further analysis. Body weights were 2.61 ± 0.07, 2.60 ± 0.07, and 2.72 ± 0.08 for birds sampled from the CON, LCP, and LCP+AL, respectively.
RNA ISOLATION AND QUANTITATIVE REAL-TIME PCR
Total RNA was extracted from the breast muscle and liver tissues by TRIzol reagent (Life Technologies, Thermo Fisher Scientific, Carlsbad, CA) according to the manufacturer’s recommendations, DNase-treated, and reverse-transcribed (Quanta Biosciences, Gaithersburg, MD). The concentration and purity of RNA were determined for each sample using a Take 3 microvolume plate and a Synergy HT multimode microplate reader (BioTek, Winooski, VT). The reverse-transcription (RT) products (cDNAs) were amplified by real-time quantitative PCR (7500 real-time PCR system, Applied Biosystems, Thermo Fisher Scientific, Foster City, CA) with Power SYBR Green Master Mix (Applied Biosystems). Oligonucleotide primers used for chicken glutamate cysteine ligase catalytic subunit (GCLC), gamma-glutamyl transferase 2 (GGT2), glutathione peroxidases (GPx1, GPx3, and GPx4), glutathione reductase (GRd), thioredoxin reductase 1 (TrxR), Cu/Zn superoxide (SOD1) and MnSOD (SOD2), and the housekeeping gene ribosomal 18S are summarized in Table 1. The quantitative PCR (qPCR) cycling conditions were 50°C for 2 min, 95°C for 10 min followed by 40 cycles of a two-step amplification program (95°C for 15 s and 58°C for 1 min). A melt curve analysis was applied using the dissociation protocol from the Sequence Detection system to exclude contamination with unspecific PCR products. Relative expressions of target genes were determined by the 2^-ΔΔCt method²⁸.
Table 1
| Gene | Accession Numberᵃ | Primer Sequence (5’ ➔ 3’) | Orientation | Product Size, bp |
|---|---|---|---|---|
| GCLC | XM_040666478.1 | TATGGGGAACCATGTCGGGA (F) / CGGGACACCCTAACCTTGGA (R) | Forward / Reverse | 133 |
| GGT2 | XM_046901324.1 | GCTCTGCGAAAGGAGGACTT (F) / CCATGAGGTTTGTGTGCAG (R) | Forward / Reverse | 76 |
| GPx1 | NM_001277853.2 | TCCCCTGCAACCAATTGC (F) / AGGCCAGGATTCCTCCGTT (R) | Forward / Reverse | 57 |
| GPx3 | NM_001163232.2 | GGGCGGCTGACACTCGAT (F) / CATCTTCCCCGGGACTTTG (R) | Forward / Reverse | 59 |
| GPx4 | NM_001364448.1 | AGAATGCGGAGCAGGAGTG (F) / ATGCAGACAGAGGCCCTGTA (R) | Forward / Reverse | 107 |
| GR | XM_040671422.1 | GAAGCACCAAGTACAGGCAG (F) / CTTACCAAGTTGGCTGTGGC (R) | Forward / Reverse | 97 |
| Gene | Accession Numberᵃ | Primer Sequence (5’ ➔ 3’) | Orientation | Product Size, bp |
|---|---|---|---|---|
| SOD1 | NM_205064.1 | TGGCTTCCCATGTGCATGAAT (F) / AGCACCTCGGCTGGTGACAC (R) | Forward / Reverse | 58 |
| SOD2 | NM_204211.1 | GCTGGAGGCCCAACTACAGT (F) / GGTGGCGTGTGTTGTTTGT (R) | Forward / Reverse | 61 |
| TrxNRD1 | NM_001030762.2 | AGAGCATGACCACCAGCTTTATT (F) / GTGTGAAGAGAAGCCTCAGTATC (R) | Forward / Reverse | 126 |
| 18S | AF173612 | TCCCCTCCCGTTTACCTTGGAT (F) / GCGCTCGTGCAGTAGTA (R) | Forward / Reverse | 60 |
ᵃAccession numbers refer to GenBank (National Center for Biotechnology Information).
GLUTATHIONE ANALYSIS
Analysis of glutathione in breast muscle and liver tissues was conducted using a glutathione assay kit (#703002, Cayman Chemical, Ann Arbor, MI) following the manufacturer’s recommended procedure. The assay was used to measure total glutathione (TGSH), which is the sum of reduced glutathione (GSH) and oxidized glutathione (GSSG)²⁹. The assay is based on the enzymatic recycling method originally described by Tietze³⁰. The expression of TGSH and GSSG is reported as µM/mg protein.
GLUTATHIONE PEROXIDASE ANALYSIS
Analysis of glutathione peroxidase (GPx) activity in breast muscle and liver tissues was conducted using a GPx assay kit (#703102, Cayman Chemical, Ann Arbor, MI) following the manufacturer’s recommended procedure. The assay is based on the spectrophotometric assay described by Paglia and Valentine³¹. The activity of GPx is reported as nmol/min/mg protein.
GLUTATHIONE REDUCTASE ANALYSIS
The measurement of glutathione reductase (GRd) activity in breast muscle and liver tissues was performed using a GRd assay kit (#703202, Cayman Chemical, Ann Arbor, MI) following the manufacturer’s recommended procedure. GRd activity is reported as nmol/min/mg protein.
SUPEROXIDE DISMUTASE ANALYSIS
Analysis of superoxide dismutase (SOD) activity in breast muscle and liver tissues was conducted using a SOD assay kit (#706002, Cayman Chemical, Ann Arbor, MI) following the manufacturer’s recommended procedure. The expression of SOD activity is reported as U/mg protein.
THIOREDOXIN REDUCTASE ANALYSIS
The measurement of thioredoxin reductase (TrxR) activity in breast muscle and liver tissues was performed using a thioredoxin reductase assay kit (#10007892, Cayman Chemical, Ann Arbor, MI) following the manufacturer’s recommended procedure. TrxR activity is reported as nmol/min/mg protein.
THIOBARBITURIC ACID REACTIVE SUBSTANCES (TBARS) ANALYSIS
Measurement of the lipid peroxidation product malondialdehyde (MDA) in breast muscle and liver tissues was conducted using a TBARS assay kit (#10009055, Cayman Chemical, Ann Arbor, MI) following the manufacturer’s recommended procedure. The expression of MDA is reported as nM/mg tissue.
STATISTICAL ANALYSES
In the qPCR and oxidative assay studies, comparison of mean expression values between the three experimental diets were made using one way analysis of variance (ANOVA) and student t-test with Graph Pad Prism (version 7.0). Differences were considered significant at P ≤ 0.05.
Results
LIVER
In the liver, mRNA expression of γGT, the enzyme needed for hydrolysis of GSH and transport into the cell, was highest in the LCP+AL group and lowest in the CON group (Figure 2A). expression of GCL, the rate limiting enzyme in GSH synthesis, was elevated (P<0.05) in the LCP-AL group compared to CON-fed and LCP fed birds (Figure 2B). Levels of hepatic TGSH were highest in the CON-fed birds and lowest in birds receiving the LCP diet (Figure 2C). Both GSSG and TBARS, (indices of oxidative stress in the cytosolic and lipid compartments, respectively), were higher in the LCP-AL group compared to the LCP group (Figure 2D and 2E). The percentage of GSSG in TGSH (insert, Figure 2D) was 0.06% for CON, 0.04% for LCP groups, and 0.2% for the LCP AL group. There was no difference in mRNA expression in SOD1 (CuZn SOD) between treatment groups (Figure 2F). Whereas SOD2 expression was higher in the LCP+AL group compared to CON values (Figure 2G), overall SOD activity was highest in CON and lowest in the LCP AL group (Figure 2H). The mRNA expression of TrxR was higher in the LCP+AL group compared to CON (Figure 2I), but there were no differences in TrxR enzyme activity (Figure 2J). Expression and activity of GSH recycling enzymes are shown in Figure 2K to 2P. In general, the highest expression was observed in the LCP AL group, and lowest values observed in the CON group. Liver activity of GPx was lower in the LCP AL group compared to both the CON-fed and LCP-fed (GPx) (Figure 2N). There was a marginal reduction of Grd activity (P = 0.09) in the LCP+AL group compared to the LCP group (Figure 2P).
Concentration of Micronutrients in Pulses
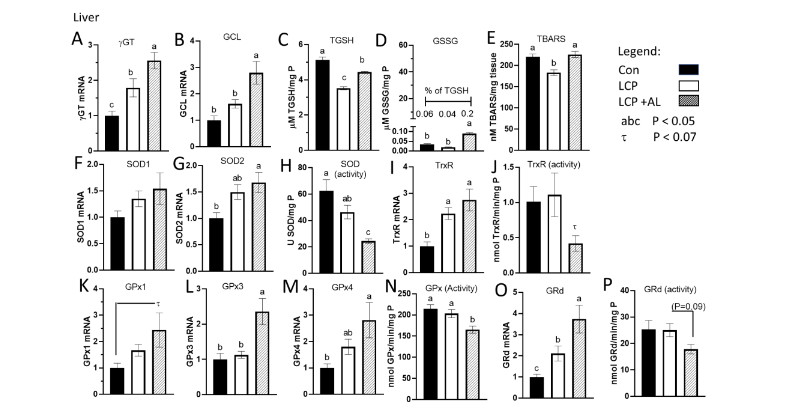
Figure 2.
Effect of feeding Spirulina platensis microalgae on antioxidant gene expression and enzyme activities in liver. Broilers were either a standard corn-soy based Control (CON, 20% CP), a low crude protein diet (LCP, 17% CP), or the LCP diet with half of the soybean meal (10% of the diet) was replaced with microalgae (LCP + AL, 17% CP). Relative gene expression is presented in arbitrary units compared to values obtained in the CON diet, concentrations of total GSH (TGSH) and oxidized GSH (GSSG) are presented in µmole per mg P, and enzyme activities are presented in units per mg tissue P. Data are presented for: A) gamma-glutamyl transpeptidase (γGT), B) glutamate cysteine ligase (GCLC), C) Total GSH (TGSH), D) oxidized glutathione (GSSG), E) thiobarbituric acid reducing substance (TBARS), F) superoxide dismutase 1 (SOD1), G) SOD2, H) SOD enzyme activity, I) thioredoxin (Trx), J) TrxR enzyme activity, K) GSH peroxidase 1 (GPx1); L) GPx3, M) GPx4, N) GPx enzyme activity, O) glutathione reductase (GRd), and P) GRd enzyme activity. Data are presented as mean ± SEM (n = 7–10/group). abc Means with different letters are significantly different (P < 0.05).
BREAST MUSCLE
The expression of γGT was higher in the LCP+AL-fed birds compared to the CON and LCP groups (Figure 3A) whereas GCLC gene expression was lower (P = 0.067) in the LCP and LCP+AL groups compared to CON (Figure 3B). Total GSH and GSSG levels were higher in breast muscle of the LCP AL group compared to both the CON and LCP groups (Figure 3C and 3D). There were no differences in TBARS between groups (Figure 3E). Whereas expression of SOD1 (CuZn) was marginally lower (P< 0.09) in the LCP and LCP AL birds (Figure 3F), SOD2 (Mn) was lower in the LCP+AL compared to the other 2 groups (Figure 3G). Overall SOD activity was higher in muscle of the LCP+AL-fed birds compared to CON (Figure 3H). The LCP+AL-fed birds exhibited higher TrxR expression and enzyme activity compared to the other groups (Figure 3I and 3J). With respect to GPx, only GPx3 was elevated in the LCP+AL group, but there were no treatment differences in GPx1 and GPx4 expression or GPx activity (Figure 3K–N). Both GRd expression (Figure 3O) and activity (Figure 3P) were higher in the LCP+AL group compared to the CON but were not different to the LCP values.
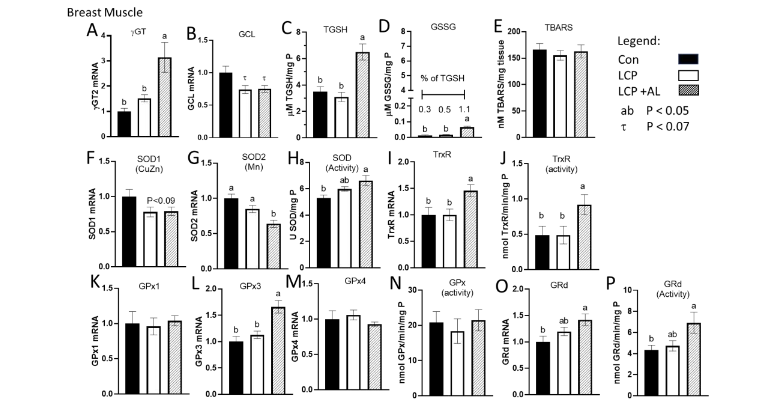
Effect of feeding Spirulina platensis microalgae on antioxidant gene expression and enzyme activities in breast muscle. Broilers were either a standard corn-soy based Control (CON, 20% CP), a low crude protein diet (LCP, 17% CP), or the LCP diet in which half of the soybean meal (10% of the diet) was replaced with microalgae (LCP + AL, 17% CP). Relative gene expression is presented in arbitrary units compared to values obtained in the CON diet, concentrations of total GSH (TGSH) and oxidized GSH (GSSG) are presented in µmole per mg P, and enzyme activities are presented in units per mg tissue P. Data are presented for: A) gamma-glutamyl transpeptidase (γGT), B) glutamate cysteine ligase (GCLC), C) Total GSH (TGSH), D) oxidized glutathione (GSSG), E) thiobarbituric acid reducing substance (TBARS), F) superoxide dismutase 1 (SOD1), G) SOD2, H) SOD enzyme activity, I) thioredoxin (TrxR), J) TrxR enzyme activity, K) GSH peroxidase 1 (GPx1), L) GPx3, M) GPx4, N) GPx enzyme activity, O) glutathione reductase (GRd), and P) GRd enzyme activity.
Data are presented as mean ± SEM (n = 7–10/group).
abc: Means with different letters are significantly different (P < 0.05).
Discussion
We previously reported that feeding a low crude protein (LCP) diet in which half of the soybean meal was replaced by microalgae (Spirulina platensis) meal (LCP+AL), had no adverse effect on broiler growth performance (comparing the LCP and LCP+AL diets)¹. Crafton³² (2022) also reported no difference in growth performance in broilers fed diets containing 2% microalgae with or without 8% distiller dried grains. Birds fed the LCP+AL diet exhibited reductions in bacterial translocation from the gut, foot pad lesions, as well as circulating cytokines, and an increase in pigmentation of skin and muscle¹. The increase in pigmentation was most likely due to higher levels of carotenoids in the LCP+AL diet and could be hypothesized to reduce oxidative stress. Spirulina platensis microalgae is a very good source of antioxidants and thus can be expected to be beneficial in helping combat cellular oxidative stress (e.g. Kumar et al.³³). Oxidative stress occurs when the levels of free radical production overcome the host’s physiological ability to effectively scavenge them, ultimately resulting in DNA, protein, and lipid damage. Thus, a major goal of the current study was to investigate the impact of a low crude protein (LCP) diet (17% vs 21% crude protein in a control diet, [CON]), and one in which half of the LCP soybean meal was replaced with Spirulina platensis (LCP+AL) on antioxidant systems (gene expression, enzyme activity) and in tissues (liver and breast muscle) obtained in male broilers.
Previous studies conducted in human cell lines, hamsters, rats, fish, chickens, and rabbits have shown that administration or dietary supplementation with Spirulina platensis protects animals against inflammation, scavenges free radicals, and
attenuates lipid peroxidation and DNA damage¹¹,³⁴,³⁷. Analysis of Spirulina indicates that the pigment protein phycocyanin and the vitamin A precursor β-carotene are the two primary active compounds in the microalgae, with both taking part in antioxidant and anti-inflammatory activities (reviewed by Wu et al.⁴⁰). Reports have shown that phycocyanin effectively decreases lipid peroxidation, the formation of peroxyl, alkoxyl, and hydroxyl radicals, nitrite production, and iNOS expression⁴¹,⁴². β-carotene has also been characterized as having antioxidant effects by inhibiting the intracellular accumulation of reactive oxygen species and protecting membranes against singlet oxygen–mediated lipid peroxidation¹⁸,⁴³. Anti-inflammatory activity of β-carotene in mice macrophages stimulated by lipopolysaccharide (LPS) or interferon gamma (IFNγ) is due to down-regulation of several genes associated with inflammation (COX-2, TNF-α, IL-1β, IL-6, and IL-12) that is likely due to inhibition of NF-KB and iNOS promoters⁴⁴,⁴⁵.
There was a decrease in TGSH and GSH (not shown) in the liver in the LCP-fed birds compared to CON-fed birds, that was partially ameliorated in birds fed the LCP+AL diet (Figure 2C). However, there was also evidence of increased oxidative stress as indicated by the increase in GSSG and TBARS (Figure 2D and 2E). There were also elevations in TGSH and GSSG in breast muscle of LCP+AL birds, but no difference in TBARS (representing generalized lipid oxidation) compared to LCP-fed (Figure 3C, D, and E). It should be noted that despite the elevation in GSSG in liver and muscle, the ratio of TGSH and GSSG was 99.8 in liver and 98.9 in breast muscle and on the low end of an acceptable range of 1 to 5%¹⁸.
This should not be unexpected since the liver is continuously exposed to oxidative stress conditions arising from: 1) the detoxification of xenobiotics and other substances that have been absorbed in the intestines and delivered to the liver via the portal vein, and 2) the substantial and continuous production of reactive oxygen species requirements of the organ. Analysis of multiple components of the glutathione system was conducted to determine how gene expression and enzyme activity was altered in response to microalgae. In addition to its vital role as a participant in antioxidant defense and redox homeostasis, the glutathione system is also a key part of cell signaling, cellular metabolism, and immunomodulation. The peptide glutathione (GSH) is an essential antioxidant in that it detoxifies reactive oxygen species (ROS), reactive nitrogen species (RNS), and hydroxyl radicals. The liver is one of the organs with the highest GSH content but interestingly, gene expression for glutamate cysteine ligase (GCL), the rate-limiting enzyme in GSH synthesis, was significantly increased in the liver of birds fed the LCP+AL diet; however, GCL gene expression was significantly lower in the breast muscle of LCP+AL birds. This difference in GCL gene expression between the two tissue types may be partially explained by the interorgan transfer of GSH¹⁹,²⁰. It is possible that supplementation of the broiler diet with Spirulina is further stimulating gene expression of enzymes involved in glutathione synthesis in the liver (i.e. increasing GCL, GPx, GRd gene expression), which is followed by the increased production and exportation of GSH into the general circulation and uptake by tissues (e.g. muscle) for resynthesis. Previous work by Wang et al.²⁰ has demonstrated the ability of the avian liver to export GSH into the general circulation. Since GCL activity is regulated by GSH via a negative feedback loop⁴⁶, an increase in GSH activity (as observed in this study) in the breast muscle through interorgan transfer from the liver may be at least partially responsible for the reduced GCL expression observed in the breast muscle of broilers fed the LCP+AL diet. GSH also serves as a cofactor for enzymes in the GSH recycling system⁴⁷,⁴⁸. There are multiple isozymes of GPx that have been identified, whose function is to reduce the oxidation of hydroperoxides to water and alcohols. Hydroperoxide is using GSH
relation to the present study, GPx1 is the most abundant isozyme and found in the cytosol, mitochondria, and nucleus of most tissues; whereas GPx3 is a secreted form found in plasma in various organs, and GPx4 (i.e. phospholipid GPx) is a membrane-bound isozyme found in the cytosol, mitochondria, and nucleus of different tissues⁵⁰,⁵¹. Gene expression levels of GPx1, GPx3, and GPx4 were observed in breast muscle and liver tissues collected during the present study. In liver tissue, expression of all three GPx isozymes was up-regulated in birds fed the LCP+AL diet when compared to the Standard diet, however only GPx3 was up-regulated in breast muscle. Expression of the gene encoding glutathione reductase (GRd), which is the enzyme responsible for catalyzing the reduction of oxidized glutathione (GSSG) to GSH was significantly increased in both tissue types of birds fed the LCP+AL diet. GRd is very important for preserving a healthy redox balance since its activity is responsible for replenishing and maintaining a high ratio of GSH to GSSG⁵². Similar to GRd, gene expression of the enzyme thioredoxin reductase (TrxR), whose actions regenerate reduced thioredoxin in the antioxidant system, was up-regulated in both breast muscle and liver tissue. Both GRd and TrxR are important components in the regulation of redox status and maintenance of redox homeostasis. The suggestion that the glutathione and thioredoxin systems work in concert during the scavenging of H₂O₂⁵³ would help explain the similar patterns seen in gene expression of the two reductases following dietary treatment with microalgae in the current study.
In addition to investigating the GSH system, superoxide dismutase (SOD) gene expression following the inclusion of microalgae in the diet was also studied. Superoxide is the most-prevalent free radical under normal cellular physiological conditions, therefore SOD is an important antioxidant and is the primary contributor to the first line of antioxidant defense against ROS formation and subsequent damage to DNA, lipids, proteins, and carbohydrates⁵⁴,⁵⁵. In the present study there was no difference in relative mRNA expression of SOD1 in liver and breast muscle tissue between the three diets; however, expression of SOD2 in the LCP+AL-fed birds was significantly up-regulated in liver, but significantly down-regulated in the breast muscle, further supporting the observations that there are tissue-specific differences in the avian antioxidant defense system. In fact, inherent tissue-specific differences in the chicken antioxidant profile have been previously reported⁵⁵. Further investigation of the specific microalgae Spirulina platensis reveals some possible explanations for the obtained results. As stated earlier, Spirulina platensis contains carotenoids, a class of natural pigments that serve as a major dietary source of vitamin A, participates in the scavenging of free radicals, and are further divided into two groups consisting of xanthophylls and carotenes based on their chemical structure⁵⁶. When these compounds are incorporated into the diet, they are absorbed into the body through passive diffusion via intestinal epithelial cells and metabolized⁵⁷. In addition to its scavenging properties, reports have indicated that carotenoids activate the network of antioxidant defenses by interacting with transcription factors that regulate it. The redox-sensitive transcription factor Nrf2 (nuclear factor-erythroid factor 2-related factor 2) is of particular interest since it is considered the master regulator in adaptive oxidative stress responses⁵⁸. The activation of Nrf2 results in it translocating to the nucleus where it binds to the antioxidant response element (ARE) and induces the transcription of several antioxidant enzymes, including GCL, GPx, GRd, SOD, TrxR⁵⁹. Interestingly, dietary regulation of the antioxidant pathway involving Nrf2 and ARE has been discussed previously. The review by Stefanson and Bakovic⁶⁰ cites a number of dietary phytochemicals that have been shown to upregulate the transcription of antioxidant enzymes in the absence of increasing induced oxidative stress; this would include carotenoids⁶¹,⁶². Therefore, an increase in the presence of carotenoids in the diet of animals
subsequent activation of the Nrf2 pathway would give a possible explanation as to why a number of antioxidant genes were upregulated (in the absence of oxidative stress induction) in the tissues of birds fed the LCP+AL diet.
In this study the inclusion of Spirulina platensis in a low crude protein broiler diet also affected antioxidant and antioxidant enzyme activity levels. Once again, the effects of the microalgae give rise to tissue-specific differences that will be discussed below. The results obtained in the liver tissue are peculiar, given that in the LCP+AL-fed birds the expression of several genes involved in glutathione synthesis and recycling were significantly up-regulated, but there was no change in glutathione levels and the activity of nearly every enzyme measured was significantly reduced. The reduction in liver SOD, GPx, GR, and TrxR activity may very well be attributed to the increased radical scavenging ability provided by Spirulina’s endogenous antioxidants (i.e. carotenoids, flavonoids, phycocyanins). It must also be considered that Spirulina platensis contains its own antioxidant defense system that is used to control ROS levels and protect cells from oxidative damage that occurs from environmental stress or the photosynthetic process. Analysis of the algae has shown that it produces several antioxidant enzymes including SOD¹⁷,³³,⁴³ as well as peroxidase, catalase, and ascorbate peroxidase⁶⁴. It appears that there is the potential for several different antioxidant enzymes to be absorbed and metabolized from the algae in a supplemented diet, ultimately leading to a reduction in the activity of host antioxidant enzymes as seen in the liver tissue. In contrast, in the breast muscle tissue a significant increase in antioxidant (total GSH and reduced GSH) and antioxidant enzyme activity (GRd, SOD, TrxR) was observed in birds fed LCP+AL. The differences in gene and enzyme activity between the two tissues cannot be fully explained at this time and further investigation is indeed warranted.
One interesting result from this study that should be addressed is the increase in oxidized glutathione (GSSG) observed in both tissues of birds from the LCP+AL group, indicating that there may be increased oxygen radical production being addressed by the glutathione system. Although carotenoids such as β-carotene are known for their antioxidant activity, a number of studies have indicated that these compounds may also function as a prooxidant as well under certain conditions. The prooxidant potential of carotenoids⁶⁷ becomes more favorable in the face of increasing oxygen partial pressure (pO₂) and/or increasing carotenoid concentrations in tissues, resulting in more favorable conditions for autoxidation that leads to the formation of carotenoid alkoxyl/peroxyl radicals⁶⁸. Also, studies conducted in rats and mice indicate that the accumulation of carotenoids in tissues can disturb electron-transport between mitochondria during ATP production, resulting in the generation of ROS⁶⁹,⁷⁰. However, it is interesting to note that birds may be more resistant to mammals as suggested by Johnson and Hill⁷¹. Mayne and Parker⁷² reported that the accumulation of dietary beta-carotene in chicken liver is also accompanied by increased levels of the antioxidants α-tocopherol and retinol, potentially providing a pathway to arrest carotenoid pro-oxidation. Also, birds have a lower basal rate of electron leak and subsequent ROS formation compared to mammals⁷³. Therefore, it should be noted that although there was an increase in breast muscle and liver GSSG in birds fed the algae-supplemented diet, the ratio of GSH to GSSG (an indicator of oxidative stress) did not deviate outside of what would be considered a “normal” physiological range for a healthy redox balance (unpublished observations). In addition, there was no change in measured MDA levels (an indicator of lipid peroxidation) in breast muscle and liver tissue from the LCP+AL diet when compared to the Control diet. This antioxidant/prooxidant capability of carotenoids is interesting and may help to provide further insight into the tissue-specific results seen in this study. Experiments in both rats and birds where animals were fed carotenoid-supplemented diets reported that carotenoids accumulate and are stored in the liver⁶⁹,⁷⁴,⁷⁵. Different levels of carotenoid accumulation between the liver tissue and breast muscle tissue may provide further explanation for the observed tissue-specific differences in antioxidant responses; however, tissue carotenoid levels were not examined in this study and future work looking at the antioxidant properties of microalgae may warrant further investigation into how distribution of carotenoids among tissues affects antioxidant responses in those tissues.
Conclusion
The results from this study indicate that the supplementation of a low crude protein diet with Spirulina platensis protein meal enhances the antioxidant status of breast muscle and liver tissue in male broilers. Breast muscle showed an increase in GPx3, GRd, Trx gene expression and an increase in SOD, GPx, GRd, SOD, and TrxR activity, while liver expression of GPx1, GPx3, GPx4, GRd, SOD2, and TrR was upregulated, while there was a significant reduction in the activity of several antioxidant enzymes (GPx, GRd, SOD, TrxR). It is proposed here that the inclusion of microalgae in the diet directly protects against oxidative stress by increased radical scavenging due to compounds originating from the microalgae (i.e. phycocyanins, carotenoids, flavonoids, and other vitamins). The direct scavenging of radicals via the microalgae may explain why there was a reduction in antioxidant enzyme activity in the liver tissue. Spirulina platensis may also enhance antioxidant protection in an indirect fashion through the activation of the antioxidant transcription factor Nrf2 and the subsequent antioxidant response element (ARE) to induce gene expression of antioxidant enzymes. However, the increase in GSSG seen in this tissue suggests that higher inclusion of microalgae may become a stressor due to the prooxidant potential of β-carotene. The antioxidant defense pathway of organisms is complex and interacts with a number of other pathways (i.e. immunomodulatory, anti-inflammatory) that may affect its actions. Also, other factors such as interorgan circulation of glutathione originating from the liver, the differences in metabolism of various tissues, and genetic selection for muscle growth must be taken into consideration as well. As a result, the differences in gene expression and enzyme activity observed between the two tissue types should not be unexpected and indeed requires further investigation. The inclusion of Spirulina platensis into poultry diets as an alternative protein source has benefits and should be pursued further.
Conflict of Interest:
The authors declare there are no competing interests.
Funding Statement:
None.
List of Abbreviations
ENM: Environmental Nutrient Management, SOM: Soil Organic Matter, Zn: Zinc, Cu: Copper, Fe: Iron, Mn: Manganese, Se: Selenium, Hg: Mercury, S: Sulfur, N: Nitrogen.
References
1. von der Goltz J, Dar A, Fishman R, Mueller N, Barnwal P, McCord G. Health Impacts of the Green Revolution: Evidence from 600,000 births across the Developing World. JOURNAL OF HEALTH ECONOMICS. 2020;74.
doi:10.1016/j.jhealeco.2020.102373
2. Eliazer Nelson ARL, Ravichandran K, Antony U. The impact of the Green Revolution on indigenous crops of India. Journal of Ethnic Foods. 2019;6(1):8-8. doi:10.1186/s42779-019-0011-9
3. Singh M. Micronutrient Deficiencies in Crops and Soils in India. In: Micronutrient Deficiencies in Global Crop Production. ; 2008:93-125. doi:10.1007/978-1-4020-6860-7_4
4. Popkin B. The nutrition transition and obesity in the developing world. JOURNAL OF NUTRITION. 2001;131(3):871S-873S. doi:10.1093/jn/131.3.871S
5. World Health Organization. Global Report on Diabetes.; 2016.
6. Debnath S, Dey A, Khanam R, et al. Historical shifting in grain mineral density of landmark rice and wheat cultivars released over the past 50 years in India. SCIENTIFIC REPORTS. 2023; 13(1). doi:10.1038/s41598-023-48488-5
7. Cakmak I. Plant nutrition research: Priorities to meet human needs for food in sustainable ways. PLANT AND SOIL. 2002;247(1):3-24. doi:10.1023/A:1021194511492
8. Ramkumar D, Marty A, Ramkumar J, et al. Food for thought: Making the case for food produced via regenerative agriculture in the battle against non-communicable chronic diseases (NCDs). ONE HEALTH. 2024;18. doi:10.1016/j.onehlt.2024.100734
9. Lal R. Soil organic matter content and crop yield. Journal of Soil and Water Conservation. 2020;75(2):27A-32A. doi:10.2489/jswc.75.2.27A
10. Howard A. An Agricultural Testament. Oxford University Press; 1943:228.
http://journeytoforever.org/farm_library/howardAT/ATtoc.html
11. LAL R. Managing soil quality for humanity and the planet. Frontiers of Agricultural Science
and Engineering. 2020;7(3):251-251. doi:10.15302/J-FASE-2020329
12. Lady Eve Balfour. Towards a Sustainable Agriculture: The Living Soil. Faber and Faber Ltd; 1943.
13. Lal R. Integrating Animal Husbandry With Crops and Trees. Frontiers in Sustainable Food Systems. 2020;4:113-113. doi:10.3389/fsufs.2020.00113
14. Janssens I, Roobroeck D, Sardans J, et al. Negative erosion and negative emissions: Combining multiple land-based carbon dioxide removal techniques to rebuild fertile topsoils and enhance food production. FRONTIERS IN CLIMATE. 2022 ;4. doi:10.3389/fclim.2022.928403
15. Ross M. Welch, Robin D. Graham, Ismail Cakmak. Linking Agricultural Production Practices to Improving Human Nutrition and Health. Published online November 19, 2014.
https://www.fao.org/about/meetings/icn2/preparations/document-detail/en/c/224907/
16. Maitin-Shepard M, Werner E, Feig L, et al. Food, nutrition, and fertility: from soil to fork. AMERICAN JOURNAL OF CLINICAL NUTRITION. 2024;119(2):578-589. doi:10.1016/j.ajcnut.2023.12.005
17. Brezeanu C, Brezeanu P, Calara M, Ambarus S, Cristea T. THE ROLE OF FOOD LEGUME SPECIES IN THE CONTEXT OF SUSTAINABLE AGRICULTURE, FOOD SECURITY, AGROBIODIVERSITY, CONSERVATION AND HUMAN HEALTH. SCIENTIFIC PAPERS-SERIES B-HORTICULTURE. 2021;65(1):390-399.
18. Ojiewo C, Keatinge D, Hughes J, et al. The Role of Vegetables and Legumes in Assuring Food, Nutrition, and Income Security for Vulnerable Groups in Sub-Saharan Africa. WORLD MEDICAL & HEALTH POLICY. 2015;7(3):187-210. doi:10.1002/wmh3.148
19. FAO, IFAD, UNICEF, WFP, WHO. The State of Food Security and Nutrition in the World 2024: Financing to end hunger, food insecurity and malnutrition in all its forms. Published online 2024. https://www.fao.org/publications/home/fao-flagship-publications/the-state-of-food-security-and-nutrition-in-the-world/en
20. Masso C, Baijukya F, Ebanyat P, et al. Dilemma of nitrogen management for future food security in sub-Saharan Africa – a review. SOIL RESEARCH. 2017;55(5-6):425-434. doi:10.1071/SR16332
21. Drinkwater L, Snapp S. Advancing the science and practice of ecological nutrient management for smallholder farmers. FRONTIERS IN SUSTAINABLE FOOD SYSTEMS. 2022;6. doi:10.3389/fsufs.2022.921216
22. Commoner B. The Closing Circle: Nature, Man, and Technology. Knopf; 1971:326.
https://books.google.com/books?id=lpYwAAAAMAAJ
23. Wezel A, Herren B, Kerr R, Barrios E, Gonçalves A, Sinclair F. Agroecological principles and elements and their implications for transitioning to sustainable food systems. A review. AGRONOMY FOR SUSTAINABLE DEVELOPMENT. 2020;40(6). doi:10.1007/s13593-020-00646-z
24. Deckelbaum R, Palm C, Mutuo P, DeClerck F. Econutrition: Implementation models from the Millennium Villages Project in Africa. FOOD AND NUTRITION BULLETIN. 2006;27(4):335-342. doi:10.1177/156482650602700408
25. Rempelos L, Baranski M, Wang J, et al. Integrated Soil and Crop Management in Organic Agriculture: A Logical Framework to Ensure Food Quality and Human Health? AGRONOMY-BASEL. 2021;11(12). doi:10.3390/agronomy11122494
26. Leakey R, Leakey R. Twelve Principles for Better Food and More Food From Mature Perennial Agroecosystems.; 2017:387. doi:10.1016/B978-0-12-805356-0.00037-4
27. Rattan Lal. Neglected and Under-Utilized Crop Species for Food and Climate Security. Presented at: ICARDA, AgMIP, and UM6P Conference on NUS in the MENA Region; February 5, 2024; ICARDA, Rabat, Morocco.
28. Shelenga T, Kerv Y, Perchuk I, et al. The Potential of Small Grains Crops in Enhancing Biofortification Breeding Strategies for Human Health Benefit. AGRONOMY-BASEL. 2021;11(7). doi:10.3390/agronomy11071420
29. Chivandi E, Mukonowenzou N, Nyakudya T, Erlwanger K. Potential of indigenous fruit-bearing trees to curb malnutrition, improve household food security, income and community health in Sub-Saharan Africa: A review. FOOD RESEARCH INTERNATIONAL. 2015;76:980-985. doi:10.1016/j.foodres.2015.06.015
30. Lal R. Soil degradation as a reason for inadequate human nutrition. Food Security. 2009;1 (1):45-57. doi:10.1007/s12571-009-0009-z
31. Deckersi J, Steinnes E. State of the art on soil-related geo-medical issues in the world. In: Sparks D, ed. ADVANCES IN AGRONOMY, VOL 84. Vol 84.; 2004:1-35. doi:10.1016/S0065-2113(04)84001-X
32. Bridget Elworthy, Henrietta Courtauld. The Land Gardeners: Soil to Table: Recipes for Healthy Soil and Food. Thames and Hudson; 2023.
33. Berkhout ED, Malan M, Kram T. Better soils for healthier lives? An econometric assessment of the link between soil nutrients and malnutrition in Sub-Saharan Africa. PloS one. 2019;14(1):1-1. doi:10.1371/journal.pone.0210642
34. Gregorio G. Progress in breeding for trace minerals in staple crops. JOURNAL OF NUTRITION. 2002;132(3):500S-502S. doi:10.1093/jn/132.3.500S
35. Kaur L, Sharma R, Singh G, Dhaliwal S. Agronomic Biofortification of Mungbean [Vigna radiata (L.) Wilczek] Grain with Zinc to Combat Zinc Malnutrition. JOURNAL OF SOIL SCIENCE AND PLANT NUTRITION. 2023;23(4):6206-6215. doi:10.1007/s42729-023-01478-y
36. Pal V, Singh G, Dhaliwal S. Agronomic biofortification of chickpea with zinc and iron through application of zinc and urea. COMMUNICATIONS IN SOIL SCIENCE AND PLANT ANALYSIS. 2019; 50(15):1864-1877. doi:10.1080/00103624.2019.1648490
37. Singh M, Singh K. Agronomic zinc biofortification of wheat. AGROCHIMICA. 2019;63 (4):307-317. doi:10.12871/00021857201941
38. Maqbool M, Beshir A. Zinc biofortification of maize (Zea mays L.): Status and challenges. PLANT
BREEDING. 2019;138(1):1-28. doi:10.1111/pbr.12658
39. Prom-u-thai C, Rashid A, Ram H, et al. Simultaneous Biofortification of Rice With Zinc, Iodine, Iron and Selenium Through Foliar Treatment of a Micronutrient Cocktail in Five Countries. FRONTIERS IN PLANT SCIENCE. 2020;11. doi:10.3389/fpls.2020.589835
40. Ebbisa A. Mechanisms underlying cereal/ legume intercropping as nature-based biofortification: A review. Food Production, Processing and Nutrition. 2022;4(1). doi:10.1186/s43014-022-00096-y
41. McCally M. Environment and health: an overview. CANADIAN MEDICAL ASSOCIATION JOURNAL. 2000;163(5):533-535.
42. Malik A, Akhtar R, Grohmann E. Environmental Deterioration and Human Health: Natural and Anthropogenic Determinants.; 2014:421. doi:10.1007/978-94-007-7890-0
43. Wei J, Rahim S, Wang S. Role of Environmental Degradation, Institutional Quality, and Government Health Expenditures for Human Health: Evidence From Emerging Seven Countries. FRONTIERS IN PUBLIC HEALTH. 2022;10. doi:10.3389/fpubh.2022.870767
44. Russo R. Environmental Rights are a Human Right to a Healthy Environment: A Brief Review. International Journal of Life Science and Agriculture Research. 2023;02(12):476-479. doi:10.55677/ijlsar/V02I12Y2023-01
45. Lal R. Laws of sustainable soil management. Agronomy for Sustainable Development. 2008;29(1):7-9. doi:10.1051/agro:2008060
46. Crutzen PJ. Geology of mankind. Nature. 2002;415(6867):23-23. doi:10.1038/415023a
47. Berti PR, Desrochers RE, Van HP, et al. The process of developing a nutrition-sensitive agriculture intervention: a multi-site experience. FOOD SECURITY. 2016;8(6):1053-1068. doi:10.1007/s12571-016-0625-3
48. Lal R. Home gardening and urban agriculture for advancing food and nutritional security in response to the COVID-19 pandemic. Food Security. 2020; 12(4):871-876. doi:10.1007/s12571-020-01058-3
49. Wang M, Li B, Li S, Song Z, Kong F, Zhang X. Selenium in Wheat from Farming to Food. JOURNAL OF AGRICULTURAL AND FOOD CHEMISTRY. 2021; 69(51):15458-15467. doi:10.1021/acs.jafc.1c04992
50. Govasmark E, Singh BR, MacLeod JA, Grimmett MG. Selenium concentration in spring wheat and leaching water as influenced by application times of selenium and nitrogen. Journal of Plant Nutrition. 2008;31(2):193-203. doi:10.1080/01904160701853605
51. JOHNSSON L. TRENDS AND ANNUAL FLUCTUATIONS IN SELENIUM CONCENTRATIONS IN WHEAT-GRAIN. PLANT AND SOIL. 1991;138 (1):67-73. doi:10.1007/BF00011809
52. Eriksson J, Dahlin A, Sohlenius G, Söderström M, Öborn I. Spatial patterns of essential trace element concentrations in Swedish soils and crops. GEODERMA REGIONAL. 2017;10:163-174. doi:10.1016/j.geodrs.2017.07.001
53. Olof Sundström. Selenium in soil and winter wheat: analysis of soil-crop-inventory data. Published online 2018. https://stud.epsilon.slu.se/13364/
54. Alexander J, Olsen A. Selenium – a scoping review for Nordic Nutrition Recommendations 2023. FOOD & NUTRITION RESEARCH. 2023;67. doi:10.29219/fnr.v67.10320
55. López-Bellido F, Sanchez V, Rivas I, López-Bellido R, López-Bellido L. Wheat grain selenium content as affected by year and tillage system in a rainfed Mediterranean Vertisol. FIELD CROPS RESEARCH. 2019;233:41-48.
doi:10.1016/j.fcr.2019.01.006
56. BRUCE A. SWEDISH VIEWS ON SELENIUM. ANNALS OF CLINICAL RESEARCH. 1986;18(1):8-12.
57. Lal R. The Soil-Human Health-Nexus. (Lal R, ed.). Taylor & Francis; 2020.
58. Basset C. Soil security: The cornerstone of national security in an era of global disruptions. Soil Security. 2024;16:100154. doi:10.1016/j.soisec.2024.100154
59. Fact.MR. Agricultural Equipment Market Is Forecasted to Expand at a CAGR of 4.3%, Reach US$ 194.4 Billion by 2034.; 2024.
https://www.globenewswire.com/news-release/2024/09/04/2940322/0/en/Agricultural-Equipment-Market-is-Forecasted-to-Expand-at-a-CAGR-of-4-3-Reach-US-194-4-Billion-by-2034-Fact-MR-Report.html#:~:text=Rockville%2C%20MD%2C%20Sept.,4.3%25%20from%202024%20to%202034.
60. Doocy S, Walton S, West KP. Famine: Causes, consequences and responses. In: Caballero B, ed. Encyclopedia of Human Nutrition (Fourth Edition). Academic Press; 2023:411-419. doi:10.1016/B978-0-12-821848-8.00088-3
61. Dugassa B. Public Health Impacts of Famine in the Horn of Africa. American Journal of Public Health Research. 2019;7(5):171-181. doi:10.12691/ajphr-7-5-2
62. Roseboom T, de Rooij S, Painter R. The Dutch famine and its long-term consequences for adult health. Early Human Development. 2006;82 (8):485-491. doi:10.1016/j.earlhumdev.2006.07.001
63. Lumey LH, Li C, Khalangot M, Levchuk N, Wolowyna O. Fetal exposure to the Ukraine famine of 1932-1933 and adult type 2 diabetes mellitus. Science (New York, NY). 2024;385:667-671. doi:10.1126/science.adn4614
64. Klimek P, Thurner S. The lasting effects of famine. Science. 2024;385(6709):606-607.
doi:10.1126/science.adr1425
65. Martínez-Ballesta M, Lopez-Perez L, Mercedes M, López-Berenguer C, Fernández-García N, Carvajal M. Agricultural practices for enhanced human health. Phytochemistry Reviews. 2007;7: 251-260. doi:10.1007/s11101-007-9071-3
66. McCullum-Gomez C. Using sustainable agriculture to improve human nutrition and health. J Community NutritionJ Community Nutrition. 2004;618:18-25.
67. Montgomery D, Biklé A, Archuleta R, Brown P, Jordan J. Soil health and nutrient density: preliminary comparison of regenerative and conventional farming. PeerJ. 2022;10:e12848. doi:10.7717/peerj.12848
68. McLennon E, Dari B, Jha G, Sihi D, Kankarla V. Regenerative agriculture and integrative permaculture for sustainable and technology driven global food production and security. AGRONOMY JOURNAL. 2021;113(6):4541-4559. doi:10.1002/agj2.20814
69. Giller KE, Hijbeek R, Andersson JA, Sumberg J. Regenerative Agriculture: An agronomic perspective. Outlook on Agriculture. Published online March 2021:0030727021998063-0030727 021998063. doi:10.1177/0030727021998063

