Financial Toxicity in Breast Cancer Management
FINANCIAL TOXICITY IN CANCER MANAGEMENT: Illness costs money. The example of breast cancer
Dr Remy J. Salmon1
- Former Head of Surgery, Institut Curie Paris, FranceProfessor at the Collège de Médecine des Hôpitaux de ParisMember of the French National Academy of SurgeryNational expert, approved by the Cour de cassation (French Supreme Court)
OPEN ACCESS
PUBLISHED: 30 November 2024
CITATION: Salmon, RJ., 2024. FINANCIAL TOXICITY IN CANCER MANAGEMENT: Illness costs money. The example of breast cancer. Medical Research Archives, [online] 12(11). https://doi.org/10.18103/mra.v12i11.5887
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i11.5887
ISSN 2375-1924
ABSTRACT
Progress in cancer treatment is indisputable and concerns all therapeutic areas: surgery, radiotherapy and medical oncology. This progress has been made possible by fundamental and clinical research, at national and international level, which is a source of great enthusiasm for clinicians and researchers alike. Pink October for instance, is a month of great hope for the general public, patients and their carers.
Alas, the reality is not so pink! All this progress comes at a cost, not only in countries with high standards of living, but also in LMICs (Low- and Middle-Income Countries), and amid the general elation of progress, a few Cassandras are trying to alert decision-makers to a health catastrophe looming on the horizon.
The aim of this article is to provide a succinct analysis of the main costs and their recent growth, and to suggest ways of trying to curb this exponential growth in costs so as not to ultimately generate frustration and inadequate or insufficient treatment for our patients.
This article is not intended as a political pamphlet against any particular company, but rather as an alert to our colleagues in the medical and research community, based on a selection of works from the international literature and filtered through personal experience in hospitals and sometimes in humanitarian medicine. This work is necessarily incomplete, according with the numerous aspects of cancer management and the number of people implicated.
Keywords
Financial toxicity, cancer management, breast cancer, healthcare costs, treatment adherence
Introduction
A few days after my arrival at Memorial Sloan Kettering in New York in 1979, I discovered the concept of “financial toxicity”. After presenting a case of Hodgkin’s disease in a young man at the morning “grand round”, my neighbor told me that the patient would not be able to complete the six cycles of chemotherapy because there was no money left on the family credit card. It was a real shock for me, used to living and practicing medicine in a country where all cancer treatments are normally covered at 100%.
Financial toxicity in oncology is getting worse by the day, whatever the nature of the local, regional or systemic treatment. This toxicity can be broken down into 6 main points:
- Screening programs
- The rising cost of treatments themselves
- Indirect costs
- Psychological impact
- Adherence to treatment
- Substitution « treatments »
Using breast cancer as an example, we’ll begin by describing the costs of the various treatments and how they have risen in recent years as a result of medical progress. We’ll then look at the alternatives that are emerging thanks to medical progress, with genetics, data management and, of course, AI.
A few proposals for solutions are then made, for those who govern us to ponder, but the last reference relates to data management: “Federating the players in the ecosystem to liberate the secondary use of health data”, a report by French technocrats.
Financial toxicity can be schematically described in 6 main classes, which may overlap depending on the patient and the stage of her disease. The prices cited in the paper relate to the actual French coverage by our Social Security and are subjects to changes in a near future.
1. Screening Programs
Everyone agrees that when it comes to cancer, the earlier the diagnosis, the better the prognosis. The first national campaigns were developed in Northern Europe several decades ago, and demonstrated their benefits in terms of “early diagnosis”. However, these programs are presently limited by the age range of the candidates and the difficulty of assessing the improvement in survival at national level, and therefore their “cost-effectiveness”. Some countries have already abandoned national screening in favor of individual screening. Experiments targeted in screening populations at high clinical and/or genetic risk are underway.
2. Increased Treatment Costs
The cost of managing a breast cancer patient’s illness has risen sharply in recent decades. Treatments aim to be as “targeted” as possible, guaranteeing the best survival with the fewest side effects, as expressed by therapeutic “de-escalation”. This targeting requires increasingly sophisticated and therefore costly radiological and biological examinations. The same applies to patient follow-up, particularly as survival time increases.
2.1 Surgery
Breast cancer patients will see a surgeon in 98% of cases, but it takes 10 years and a lot of money to train a surgeon. Projections show that the number of surgeons is declining and projections at year 2040 announce a deficit of one million surgeons world-wide. The cost of a day’s hospitalization for surgery is €1,700, and can reach €3,500 for a stay in an intensive care unit. The cost of a mastectomy is €290 for a surgeon operating in the strict agreement sector! The operation lasts between 1 and 2 hours, depending on the surgeon’s experience. Currently, over 70% of breast surgery procedures are performed on an outpatient basis.
Until 2023, health insurance and mutual insurance companies considered breast reconstruction to be a cosmetic procedure, and refused to cover the cost, whatever the technique. In 2024, French national recommendations will require all patients undergoing mastectomy to be informed about the various possibilities for immediate or secondary breast reconstruction.
Some surgeons have tried to improve the quality of cosmetic results by using “matrices”. The first financial obstacle is that no official payment system covers the cost of these matrices. They cost around €1,000. Reconstruction can also be carried out using prostheses, autologous tissue (i.e. the patient’s own tissue), myocutaneous flaps and the injection of the patient’s own fat (lipofilling).
Several procedures can be used and prices vary and increase according to the technique: Myocutaneous flaps: latissimus dorsi (€600), DIEP (double inferior epigastric perforator) + micro-vascular anastomosis (€800), and more recently lipofilling, using the patient’s own fat to fill the missing volume (€600 per session, three sessions are generally required), including nipple reconstruction.
There were 62,000 new cases of breast cancer in France in 2023. Some 30% to 40% of patients will be treated by mastectomy, at some stage in their development. Approved hospitals and clinics are currently overwhelmed by these reconstruction procedures, in particular the multiple operating times, which are not strictly speaking cancer procedures. As a result, waiting times are getting longer, or some patients have to be redirected to private practitioners, more or less linked to the cancer center. In such cases, fees can rise considerably, making it more difficult for patients to undergo full reconstruction.
Two comments:
The first is that, thanks to early detection, over 70% of patients can benefit from breast-conserving treatment. These conservative treatments can induce aesthetic deformities, and surgeons must anticipate the quality of their results to prevent these deformities, using so-called “oncoplastic” techniques taught in the majority of surgical training centers. The second point concerns prophylactic surgery for patients carrying a BRCA1-2 mutation and its variants, predisposing them to the development of breast cancer. One of the main indications for “robotic surgery” is prophylactic surgery (to prevent the occurrence of cancer) in these high-risk patients.
A surgical robot costs between €1.2 and €1.4 million, and the annual operating budget for the robotic unit is around €350,000 per year, with technical maintenance at €250,000. There is as yet no specific pricing in France for robotic surgery. To date, randomized trials have demonstrated the statistical “non-inferiority” of robotic surgery to conventional operations, in terms of safety and cancer survival. But at this price, robotic surgery will have some difficulty in demonstrating the real financial benefit of this technique.
In fact, if the price of robot is included in a national vision, including thoracic, urologic GI, head and neck, reduction of hospitalization and various fees are strongly reduced as demonstrated in Denmark or South Korea.
2.2 Radiotherapy
Radiotherapy has made immense progress in recent decades. From Marie Curie’s radiotherapy, to the coupling of treatments with CT scans, to target contouring with AI, photons, protons and hadrons. Making irradiation dependent on breathing, focusing fields according to tumor response, dose fractionation over 25 days in the 80s, to hypo-fractionation over 15 and now five days, IMRT, demonstrate the ongoing evolution of radiotherapy in breast cancer management.
Better targeting of fields to avoid toxicity on surrounding organs has become dogma. Radiotherapy has also proved useful in oligo (small or few) metastatic diseases, and the name “cyber knife” was not chosen at random. Liver and lung metastases can be also controlled by targeted radiotherapy treatments. The same applies to brain metastases, leaving normal brain function in oligo-metastatic disease, instead of the former total irradiation of the brain.
But the price of radiotherapy equipment has been rising steadily, and now varies between 3 and 5 million euros. Pricing for radiation procedures, still in 2024, remains highly variable, depending on the machine used, the number of fractions performed and the time required by doctors and physicists to deliver the right dose to the right target and from one center to another.
The latest French Cour des Comptes report states: Radiotherapy is constantly and rapidly evolving, mainly in terms of targeting and the number and duration of sessions. These improvements have been so rapid that dedicated agencies and legislators have been overwhelmed in adapting the referentials and nomenclature lists used to calculate and define the new real price of procedures. Between 2015 and 2020, spending on radiotherapy will have risen by far more than the estimated budget for the National Health Insurance Expenditure Target (ONDAM).
To sum up, billing for radiation procedures is still based on a classification system that has remained unchanged since 2004, and is therefore ill-suited to the machines, procedures and, above all, advances in modern oncology. The commission notes that the Regional Health Agencies (ARS), which are supposed to manage this issue, are not competent, relying on insufficient data between public and private centers and the lack of correct information to assess and calculate the real situation, leaving room for non-comparable data from one center to another, making an objective assessment impossible and consequently a big mess in billing.
2.3 Medical Oncology
The Cour des Comptes 2024 report on anti-cancer drugs states in its preamble: Anti-cancer drugs: better regulation while preserving rapid access to innovative treatments. Key figures: In 2020, 433,136 new cases of cancer were diagnosed in France, bringing the number of patients treated each year to 3.4 million. Cancer is the leading cause of death in France (169,910 in 2022). Cancer is the costliest pathology for insurance companies: €22.5 billion in 2021, or 12.1% of total health insurance expenditure. Spending on innovative cancer drugs dispensed in hospitals and included on the “liste en sus” has risen sharply, representing €3.3 billion in 2018 and €5.9 billion in 2022. A recent news from the Lancet Oncology says that Global spending on oncology drugs is expected to reach US$ 409 billion by 2028 up from $223 billion last year according to the IQVIA Institute for human data science.
2.3.1 Hormonal Therapy
Surgical removal of the ovaries, which suppresses estradiol levels, has been the preferred adjuvant treatment when hormone receptors are present on the tumor. This suppression of ovarian secretion is also achieved by LH-RH analogues. There has never been a financial comparison between outpatient surgical oophorectomy (Social Security cost €176.46 for the surgeon + operating room fees) and medical suppression of ovarian function by LH-RH analogues (€347.45 every 3 months) until the decision is made to stop treatment.
After cessation of ovarian function, in patients whose tumors carry hormone receptors, known as hormone-dependent patients, it is possible to give oral anti-estrogens. Tamoxifen was discovered in England over 70 years ago and costs €3.27 per tablet. Aromatase inhibitors took over from Tamoxifen, demonstrating greater efficacy in terms of disease-free survival, but with no statistical benefit in terms of overall survival. Numerous publications have discussed the optimal duration of oral treatments: 5, 7 or 10 years, with no real consensus on the subject. No real medical-economic studies have been carried out on such “cheap” drugs, which have also fallen into the public domain.
In addition, it has been reported that 30-40% of patients discontinue treatment spontaneously after 2-3 years, due to side effects, bone and joint pain and osteoporosis. The price of aromatase inhibitors is around €3 per tablet, but the cost of osteoporosis treatments has never been clearly specified. New drugs have been developed for patients with hormone receptor-positive tumors. Currently reserved for metastatic patients, CDK4/6 inhibitors may be able to replace aromatase inhibitors in high-risk patients, or in patients who develop resistance to aromatase inhibitors, as demonstrated by the appearance of ESR1 gene over-expression during treatment.
2.3.2 Chemotherapy
The cost of chemotherapy has risen steadily since Bonadonna’s publications demonstrating the benefits of adjuvant medical chemotherapy in breast cancer. Since Bonadonna’s CMF protocol (1973), which currently costs €4.40 for methotrexate, €28.75 for 5Fu and €2.87 for folinic acid for a course of treatment, the drugs have fallen into the public domain, and prices have risen logarithmically. If we take the example of triple-negative breast cancer (i.e. without hormone receptors or HER2 over-expression), 10 years ago the price of anthracycline/taxane-based chemotherapy was €6-8,000. The addition of immunotherapy has pushed the price up to €80,000. This price does not include the cost of medicines for immunological diseases that may be induced by immunotherapy, such as skin, thyroid, diabetic or rheumatological diseases.
2.3.3 Targeted Therapies
Additional costs may also arise from a new class of drugs, conjugated antibodies, recently developed for patients with low + or ++ HER2 levels. Historically, tumors expressing the HER2 gene were declared positive if the assay result was +++. Herceptin is a “targeted” treatment that acts specifically on cancers that over-express the HER2 gene. Given by the venous route or subcutaneously for one year, it has moved HER cancers from “poor” prognosis before Herceptin to “good” prognosis in the International Classification of Breast Cancers. The benefit is indisputable in terms of improved survival, but the price is €40,000 a year. The product was considered “cost-effective”. But the concept of HER2 Low tumors has recently emerged, which means that over-expression of one + or two ++ of the HER2 gene could be an indication to benefit from antibody conjugates such as Trastuzumab Deruxtecan, with new molecules currently on the market. The drug is available at a price of €5,165 for one treatment. These treatments are currently reserved for metastatic patients, until disease progression. The reasoning should include the need for cost-effectiveness studies and cost-benefit analyses to assess whether the use of expensive innovations is cost-effective and profitable. Indeed, the cost of adding immunotherapy, Herceptin and conjugated antibodies to the cancer budget over the last 5 years raises questions. However, the cost-efficiency ratio alone is not an exclusive criterion for deciding on new investments. Priority must be given to patients, without innovation jeopardizing the state’s financial equilibrium.
3. Indirect Costs
During the “supportive care” phase of the disease, followed by the “post-cancer” or “cancer survivor” recovery period, most patients are unable to return to work, either full-time or part-time. Add to this the time spent travelling, and the possible need for their husbands to be present during consultations and hospitalizations, and these are “work-free” periods, during which no money comes in.
In France, the vast majority of patients benefit from social security and ALD30 (Long Duration Disease registered in a list of 30 long duration ie chronic disease) in cancerology, which reimburses all cancer-related care. The same cannot be said for supplementary insurance and mutual insurance companies. While some large companies have special programs to alleviate this situation, small companies and self-employed workers can face serious financial problems.
For example, many “non-interventional” therapies are not covered by the French While direct costs can be calculated with varying degrees of precision, so-called “indirect” costs are much more difficult to assess. The journey of cancer survivors does not end with the clinical cure of the disease, as they often face discrimination related to their medical history, years or even decades after their recovery. This discrimination manifests itself in various forms: difficulties in obtaining or keeping a job, difficulties in accessing financial services such as life or travel insurance, as well as loans and mortgages.
The “right to be forgotten” is a major issue that was recently discussed at European level, and has just been reduced from 8 to 5 years. Patients will no longer have “cancer” engraved on their foreheads, which hinders their professional advancement and access to mortgages, for example. Numerous agreements had already been drawn up with insurance companies over the years. The right to be forgotten remains a real issue, but not all players are playing fair. Insurers have the right to refuse to insure someone without justifying their refusal, the amount of monthly insurance payments is not negotiable, and the fact of being ALD30 appears on the carte vitale. In short, Mme FOURNIER’s action at European level is exemplary, but the application of this measure will take “some time” if not a long time.
4. Substitution and Complements
Social security and private insurance can be replaced or supplemented by non-governmental associations, such as patient groups. City assistance, credit cards and bank loans are alternative solutions, linked to the local social network. This search must be led by patients and their families, which represents an additional task when it comes to returning to an ordinary life, in a patient whose energy resources have been severely depleted to keep up with their treatment.
To summarize, when cancer is diagnosed, several factors can aggravate the risk of financial toxicity:
- If the patient is the family breadwinner,
- How much do other family members earn?
- Income level, given that the lower the income level, the greater the financial toxicity
- What was the level of debt before the cancer diagnosis?
- How does cancer and its treatment affect the ability to work?
- How do health and disability insurance cover costs in addition to social security reimbursements?
However, if we stay as we are and move towards an ideal world in which every patient should be tested, using a blood test analyzing circulating cells and/or DNA, to find out if they have cancer, to identify targets and, after using NGS, to decide on the best available treatment, who will pay and, finally, where and when should we eventually stop treatments and on the basis of what criteria?
5. Psychological Impact
Cancer itself causes stress and anxiety. Financial pressure can increase these psychological traumas, for example, in the face of loan repayments for younger patients, or pension contributions for older ones. This financial stress can have a negative effect on the patient’s ability to concentrate on treatment and maintain a social environment. These costs are difficult to measure, especially as psychological trauma may not become apparent until it is too late.
6. Treatment Adherence
Adherence to oral treatments, particularly hormone therapy, has become a real concern in recent years. Between 30% and 40% of patients abandon their treatment, mainly because of its cost, side effects and insufficient explanations provided by doctors and nurses. It is particularly difficult to assess the financial impact of this discontinuation of treatment, because although it is possible to track the purchase of medication and the rate at which it is renewed, this does not constitute proof of adherence to treatment. Self-medication remains an uncontrollable phenomenon.
Finally, there are socio-cultural barriers to delivering clear, accurate information, which are difficult to assess. What is certain, however, is that our governments will not be able to continue feeding the healthcare budget in the short term as they are currently doing.
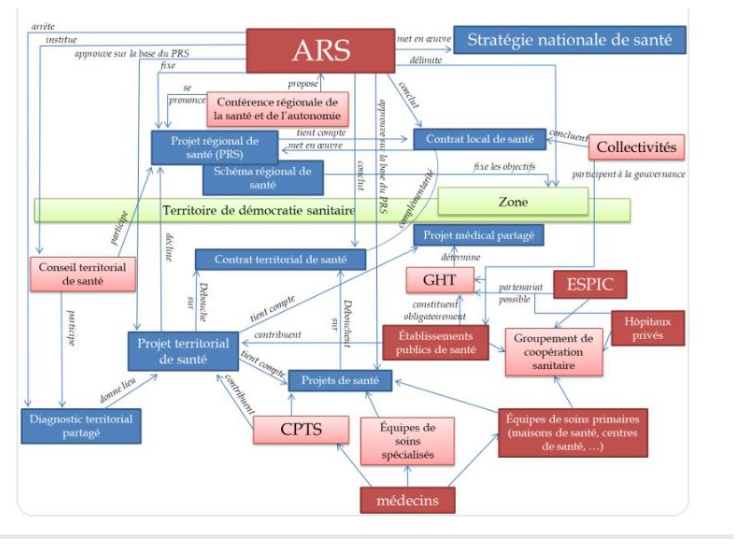
Draft solutions
To solve a problem, the first thing to do is to recognize it and have the will to resolve it, but as this diagram, which only concerns the CPTS (Communautés Professionnelles de Santé), an example of an administrative mille-feuille, shows, there’s still a long way to go. The solution can only come from a national consensus, and the first step is for our managers, both political and health, to change their paradigm and see healthcare as an investment, not a cost. It is important, however, that it should be a productive investment, provided that the expenditure is technically efficient.
A healthy population costs less than a population with costly diseases. Good health saves on healthcare costs and is good for the economy, while reducing social inequalities. Patients want to regain their health and be treated quickly, eventually cured, without pain or anxiety, surrounded by their families. Doctors want to keep their patients healthy, keep abreast of new developments and earn enough money to live well, pay their bills and raise their children.
Such an article would be incomplete without references to the pharmaceutical industry and politics or politicians! Big Pharma wants to remain competitive, and claims that research and development is as essential as it is costly to find new molecules, and that it has no choice but to make profits with blockbusters. Politicians want to be re-elected, and one of the best ways to do this is to demonstrate to potential voters that they have passed good laws to keep them healthy, possibly by preventing the onset of disease. In France, we’ve had 8 ministers of health over the past 5 years, and the most recent one will soon be faced with these issues.
The administrations in charge of the healthcare system are numerous, spread across different ministries – health, of course, but also finance, research, education and government agencies such as HAS, ARS, etc. – making for slow decision-making processes. Of course we need to save money, but who’s going to start?
For the same expenditure (as a % of GDP), we could significantly improve the system’s efficiency by improving the productive and allocative efficiency of spending. The majority of doctors will not accept a reduction in their fees, and do not want to lose their “power” and freedom to prescribe. No one has ever seen medical administrations or agencies cut their own budgets or staff. Between pandemics, migratory flows and various wars, politicians have their own emergencies to deal with, before cutting back on healthcare. What’s more, they want to be re-elected every 3 to 5 years, so they need quick, visible results to show their potential voters.
In red: Administrative people In green: University teachers In blue: Medical students. This diagram, recently published on the net, can be applied to healthcare in France.
A first solution is to stop separating public hospitals from private practice. This arbitrary distinction between “good” medicine in public hospitals and “bad” one in “for-profit” establishments (what an ugly word!!) is totally counterproductive and serves only to drive certain political agendas, without ever leading to a constructive situation. While the healthcare offer is competitive in some major cities, this is far from being the case across the whole country, which is currently undergoing desertification rather than competition.
If there is any danger, it comes from large financial groups that own more and more healthcare establishments, pharmaceutical laboratories, biological laboratories, radiology practices and even emergency services.
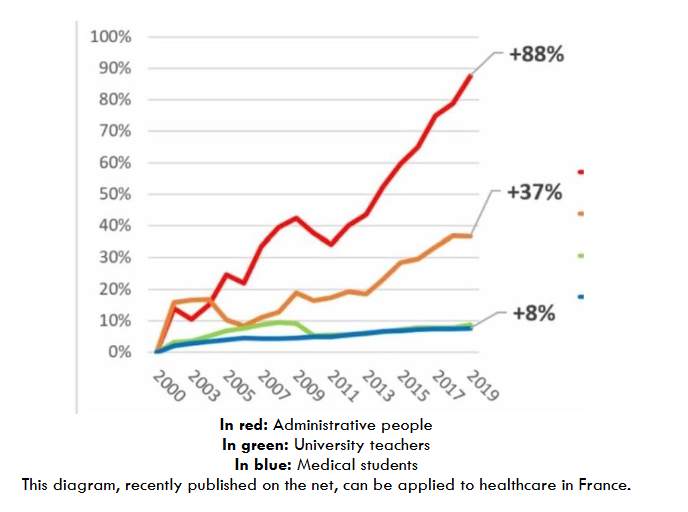
In the short term, the financial weight of these groups and the number of patients treated in their establishments will be such that the “supervisory authorities” risk becoming dependent on their proposals. Medical practices vary according to speciality and region, and harmony does not always reign between the different medical unions. However, we saw during the COVID pandemic that doctors were able to put aside their differences and, when necessary, unite in the face of disease. It’s just a question of asking their opinion to the professionals and asking the right questions.
Artificial intelligence is ubiquitous, especially in radiology, pathology and genetics, but although the contributions of this AI are said to be increasing dramatically, it will take time and money before it can replace and then supplant current medicine. In addition, conventional radiology, pathology and blood testing, among others, are likely to disappear, and resistance from those involved in these fields is likely to be very strong.
Targeted diagnosis and treatment is another option. Molecular biology, next-generation sequencing, circulating cells, DNA or RNA, could make it possible to find the right diagnosis, appropriate treatment and follow-up for each patient, with the help of RWD (Real World Data) and RWE (Real World Evaluation) and AI. Being able to determine who will be ill, when and what the right treatment will be is no longer a dream.
National and international groups emerged during the last decade G7 Cancer Group Grand Challenge, for instance who are able to gather international funding and select specific international laboratories focused on a specific project decided by an ad hoc committee. This solution is very efficient avoiding the spread of the available money on a myriad of laboratories, with limited funding.
These human genome projects are being developed all over the world, and the most recent article published on this subject in one of the latest issues of Lancet Oncology shows its value in selecting the most appropriate treatment in childhood cancers, modifying treatment in 7% of patients, but WGS (Whole Genome Human) did not lead to a change in disease management for around 93% of patients. In the case of sarcoma, 33% of sarcoma patients who underwent WGS had their treatment plan changed to a more personalized approach using a known targeted intervention, conversely, as in the childhood cancer study, 67% of patients who underwent WGS had their treatment plan unchanged. These approaches are developing in all solid tumors, but at what cost?
For example, the Orbis Project investigated the clinical benefit, time to acceptance and price of new molecules accepted by the FDA in the USA, Great Britain and Scotland. 244 anti-cancer molecules were analyzed. Of the 24 accepted, the gain in progression-free survival was 4.1 months, and there was no benefit in overall survival. The average monthly price of these drugs ranged from US$13 to US$37,000.
The creation of digital twins has also been proposed, which would avoid the difficulties associated with lengthy and costly randomized trials. This technology could also be used to train young surgeons and radiotherapists, thus avoiding the need for training on patients.
While the conclusion of this article is relatively full of hope, it also highlights the limits of progress. The scale of the investment in time, money, laboratory infrastructure and personnel that will be required to implement routine WGS in healthcare systems cannot be underestimated. Staff, resources and funds are rapidly running out in an overburdened healthcare system, where dangerous delays in cancer care are becoming normalized and waiting lists for diagnostic tests are at record levels. Any effort to make complex genomic services more widely available must take into account the cost to healthcare systems, and should only be routine practice for patients for whom there is a proven link between genomic testing and survival. This will require an initial screening process, with questions raised about the identification and training of those who will have to make the decisions, or not, to perform a WGS or rather a circulating cell assay.
The costs of these new technologies will have to be compared with conventional approaches, and it will be some time before they are accessible to the vast majority of the population.
In other words, medicine is made for patients, and listening to patients is becoming increasingly important in decision-making at some cancer centers and large pharmaceutical companies.
The next step will be not only to seek patients’ opinions and increasingly ask them to sign an “informed consent”, thus protecting doctors from lawsuits, but also to include them in the treatment decision-making process. Our American colleagues have also recently developed the concept of “choosing wisely”, mainly reserved for elderly and heavily-treated patients in situations where survival benefit must be weighed against quality of life.
Conclusion
No one is arguing that things need to change urgently and radically, because financial toxicity is likely to kill far more people than disease in the short term. Attempts to bring about change will come up against many difficulties, and our politicians will be very wary and cautious. The power of doctors and large medical and economic agencies, largely run by technocrats, will be called into question, and this is likely to lead to strong negative reactions. But patients need to be at the heart of this “revolution”, and the use of patient associations, patient experts, PRO’S and so-called “wise persons”, in decision-making processes where they are directly concerned.
Bibliography
- Financial Toxicity Among Patients With Breast Cancer Worldwide: A Systematic Review and Meta-analysis. Ehsan AN, Wu CA, Minasian A, et al. JAMA Netw Open. 2023 Feb 1;6(2):e2255388
- Federating ecosystem players to unleash the secondary use of healthcare data Report 2023 Jérôme MARCHAND-ARVIER, Pr Stéphanie ALLASSONNIERE, Aymeril HOANG, Dr Anne-Sophie JANNOT Rapporteurs 150 Pages INCa publications Organized breast cancer screening program Institut National du Cancer https://www.e-cancer.fr
- Organized breast cancer screening Santé publique France https://www.santepubliquefrance.fr Jul 31, 2023
- Global demand for cancer surgery and an estimate of the optimal surgical workforce between 2018 and 2040: a population-based modelling study Sathira Kasun Perera, Susannah Jacob, Brooke E Wilson, et al. Lancet Oncology 2021,22,182-89
- Will surgery be disappeared from the treatment of breast cancer. Salmon R.J. Arch of breast cancer 2023 10, 99-102
- Do we need acellular dermal matrix in pre pectoral breast reconstruction, Systematic review and meta analysis. Nolan IT, Farajzadeh MM, Boyd CJ, et al. Plast Reconstr Aesthet Surg. 2023 Nov;86:251-260
- Analysis of online materials regarding DIEP and TRAM flap autologous breast reconstruction. Bruce JC, Batchinsky M, Van Spronsen NR, et al Plast Reconstr Aesthet Surg. 2023 Jul;82:81-91 Breast Reconstruction Combining Lipofilling and Prepectoral Prosthesis after Radiotherapy.
- Razzouk K, Fitoussi A, Al Khori N, Pasquier J, Chouchane L, Tabrizi AR. Plast Reconstr Surg Glob Open. 2020 May 26;8(5):e2659. doi: 10.1097/GOX.0000000000002659.
- Improving breast cancer surgery: a classification and quadrant per quadrant atlas for oncoplastic surgery. Clough KB, Kaufman GJ, Nos C, Bucci Mazza I, Sarfati IM. Ann Surg Oncol. 2010 May;17(5):1375-91
- Robotic Versus Conventional or Endoscopic-assisted Nipple-sparing Mastectomy and Immediate Prosthesis Breast Reconstruction in the Management of Breast Cancer: A Prospectively Designed Multicentre Trial Comparing Clinical Outcomes, Medical Cost, and Patient-reported Outcomes (RCENSM-P). Lai HW, Chen DR, Liu LC, Chen ST, et al. Ann Surg. 2024 Jan 1;279(1):138-146.
- Targeted RT study: results on early toxicity of targeted therapies and radiotherapy. Konnerth D, Gaasch A, Westphalen CB, Heinrich K, et al. Radiat Oncol. 2024 Aug 29;19(1):113
- Cour des Comptes report on radiotherapy 2023 https://www.ccomptes.fr
- Cour des Comptes report chapter VI 2024 https://www.ccomptes.fr
- Rising costs of cancer medicines Devi S Lancet oncol 25,1262, 2024
- Trial. Iwase T, Saji S, Iijima K, Higaki et al. J Clin Oncol. 2023 Jun 20;41(18):3329-3338
- The CDK4/6 inhibitor revolution – a game-changing era for breast cancer treatment. Morrison L, Loibl S, Turner NC. Nat Rev Clin Oncol. 2024 Feb;21(2):89-105.
- Switch to fulvestrant and palbociclib versus no switch in advanced breast cancer with rising ESR1 mutation during aromatase inhibitor and palbociclib therapy (PADA-1): a randomised, open-label, multicentre, phase 3 trial. Bidard FC, Hardy-Bessard AC, Dalenc F; et PADA-1 investigators. Lancet Oncol. 2022 Nov;23(11):1367-1377
- Cost-effectiveness of pembrolizumab plus chemotherapy as first-line treatment in PD-L1-positive metastatic triple-negative breast cancer. Huang M, Fasching P, Haiderali A, Pan W, et al. Immunotherapy. 2022 Sep;14(13):1027-1041.
- Antibody-Drug Conjugates in Solid Tumor Oncology: An Effectiveness Payday with a Targeted Payload. Kondrashov A, Sapkota S, Sharma A, et al JJ. Pharmaceutics. 2023 Aug 19;15(8):2160. doi: 10.3390/pharmaceutics15082160
- Rising costs of cancer medicines Devi S. Lancet oncol 2024, 25, 1262 Adherence to oral hormonal anticancer agents in breast cancer. Gori S, Modena A, Foglietta J, et al. Tumori. 2023 Jun;109(3):262-268.
- Protecting cancer survivors from financial discrimination throughout the EU: A cross-European perspective. Meunier F. Eur J Cancer. 2024 Sep;209:114238. doi: 10.1016/j.ejca.2024.114238. Epub 2024 Jul 20.
- Ending financial discrimination for cancer survivors: embedding the right to be forgotten in legislation across Europe. Lawler M., Scocca G., Meunier F. Lancet Oncology 2024, 25 (9) 1123-1126
- Markus H, Chandrananda D, Moore E, Mouliere F, et al. Refined characterization of circulating tumor DNA through biological feature integration. Sci Rep. (2022) 12:1928. doi: 10.1038/s41598-022-05606-z
- Updates on clinical use of liquid biopsy in colorectal cancer screening, diagnosis, follow-up, and treatment guidance. Front Cell Dev Biol (2021) 9:660924. doi: 10.3389/fcell.2021.660924
- Les itinérants de la santé, quel futur pour notre système de santé Ed Michalon 2024 Bizard F.
- Artificial intelligence in breast cancer: application and future perspectives. Yan S, Li J, Wu W. J Cancer Res Clin Oncol. 2023 Nov;149(17):16179-16190.
- Advancing precision oncology with large, real-world genomics and treatment outcomes data. [No authors listed] Nat Med. 2022 Aug;28(8):1544-1545.
- Incorporating whole-genome sequencing into cancer care. The Lancet Oncology. 2024 Aug;25(8):945.
- Clinical benefit, reimbursement outcomes and prices of FDA approved cancer drugs reviewed through Project Orbis. Jenei K., Gentilini A., Haslam A., Prasad V. Lancet oncol. 2024 Vol.25 979-988
- Digital twins in cancer research and treatment: A future for personalized medicine. Heudel P.E. 1Renard F. 2, A. 2. Bull cancer 2023 Oct;110(10):1085-1087. doi:10.1016/j.bulcan.2023.07.007. Epub 2023 Sep 1.
- Choosing wisely in oncology: necessity and obstacles. Saletti P, Sanna P, Gabutti L, Ghielmini M. ESMO Open. 2018 Jul 11;3(5):e000382. doi:10.1136/esmoopen-2018-000382

