Haptoglobin-Related Protein and Renal Salt Wasting in AD
Haptoglobin-related protein without signal peptide induces high prevalence of renal salt wasting and new syndrome of salt wasting in Alzheimer’s disease
John K. Maesaka¹*, Louis J. Imbriano¹, Candace Grant¹, Nobuyuki Miyawaki¹, Rajanandini Muralidharan¹
- Departments of medicine, division of nephrology and hypertension, and the division of neurology, NYU Langone Hospital–Long Island and NYU Grossman Long Island School of Medicine, Mineola, New York, 11501, USA
*Email: [email protected]
OPEN ACCESS
PUBLISHED:31 January 2025
CITATION: Maesaka, J. K., Imbriano, L. J., et al., 2025. Haptoglobin-related protein without signal peptide induces high prevalence of renal salt wasting and new syndrome of salt wasting in Alzheimer’s disease. Medical Research Archives, [online] 13(1).
https://doi.org/10.18103/mra.v13.i1.6287
COPYRIGHT: © 2025 European Society of Medicine. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13.i1.6287
ISSN 2375-1924
ABSTRACT
Our understanding of hyponatremia, defined as a serum sodium < 135 mmol/L, and hyponatremia-related diseases is in a state of flux. A better understanding is now being established by abandoning the ineffective volume approach that has been in existence for over 50 years. We utilized comprehensive pathophysiologic phenomena that brought greater clarity to understanding and identifying the different causes of hyponatremia. An important message was to emphasize the difficulty in differentiating the syndrome of inappropriate secretion of antidiuretic hormone (SIADH) from appropriate antidiuretic hormone secretion in patients with cerebral or renal salt wasting (RSW), a condition that was previously considered to be rare among internists. Both syndromes present with identical clinical parameters that need to be appreciated and emphasized. The new approach demonstrated a high prevalence of RSW in hyponatremic patients from the general medical wards of the hospital to raise the awareness that hyponatremia is not all due to SIADH. We demonstrated natriuretic activity in the plasma of 21 neurosurgical and 17 Alzheimer’s disease patients by performing rat renal clearance studies while infusing plasma from these patients. The natriuretic factor was later identified as haptoglobin related protein without signal peptide in both groups of patients. Interestingly, blood levels of HPRWSP appears to increase progressively as the dementia worsens below a mini mental state examination score of 12 in Alzheimer’s. Additional data suggest that RSW might be present at an early stage of Alzheimer’s and these patients may become more dehydrated as the dementia worsens. Future studies intend to prove the existence and extent of volume depletion in Alzheimer’s and to develop HPRWSP as a biomarker of RSW.
Keywords: Haptoglobin related protein without signal peptide, renal salt wasting in Alzheimer’s disease, cerebral salt wasting, Haptoglobin related protein; Alzheimer Disease; Hyponatremia; Fractional urea excretion; Dehydration; Natriuretic factor; Renal salt wasting; Syndrome of inappropriate antidiuretic hormone secretion.
Introduction
The application of a physiologically derived approach to evaluating patients with hyponatremia and hyponatremia-related diseases has proven to be superior to the ineffective volume approach. In the volume approach, hyponatremic patients are classified as being euvolemic, hypervolemic or hypovolemic despite the general agreement that we are unable to assess accurately the volume status of patients by usual clinical criteria. Differentiating the syndrome of inappropriate secretion of antidiuretic hormone (SIADH) from renal salt wasting (RSW) has proven to be a very difficult task because both syndromes present with identical clinically utilized parameters such as normal renal, thyroid, and adrenal function, hyponatremia, hypouricemia, excreting a concentrated urine where urine osmolality is > plasma osmolality, urine sodium concentration (UNa) usually exceeding 30 mmol/L and having a high fractional excretion of uric acid (FEurate). The only important difference is their volume status where patients with SIADH are volume expanded by an excess of water as compared to being volume depleted in RSW. This diagnostic dilemma has thus created a therapeutic dilemma of water-restricting water-logged patients with SIADH or administering salt water to dehydrated patients with RSW.
The aims of this review are to discuss the inability of the volume approach to differentiate SIADH from RSW and how we utilized sound physiologic phenomena to differentiate both syndromes. This new approach led to an unexpected high prevalence of RSW in hyponatremic patients from the general medical wards of the hospital¹. In our effort to understand how RSW might evolve as a clinical entity, we demonstrated natriuretic activity in the plasma of neurosurgical patients and of Alzheimer disease in rat clearance experiments and later identified haptoglobin related protein without signal peptide (HPRWSP) as the natriuretic factor that causes RSW²⁻⁴. We also introduce RSW as a new syndrome in AD and how these controversial findings led to three debates over the existence of cerebral salt wasting³⁴.
Derivation of physiologic approach to renal salt wasting
We encountered a hyponatremic patient with bronchogenic carcinoma who had all of the clinical parameters noted for SIADH, at a time when the diagnosis of SIADH would have been made. The presence of postural hypotension and reflex tachycardia and brisk increase in serum sodium of > 5 mmol/L after receiving isotonic saline infusion would have been consistent with a hypovolemic state⁵⁻⁶. Based on the proposal that the coexistence of hyponatremia and hypouricemia differentiated SIADH from most other causes of hyponatremia by Beck, we decided to incorporate a more physiologic approach in our work up of this hyponatremic patient⁷.
a) FRACTIONAL URATE EXCRETION
The case of hyponatremia noted above had some complicated features such as coexistent hyponatremia and hypouricemia to favor the diagnosis of SIADH except for the presence of postural hypotension with reflex tachycardia and a greater than 5 mmol/L increase in serum sodium after isotonic saline infusion that were consistent with a hypovolemic state⁶⁻⁷. RSW was also referred to as “cerebral” salt wasting (CSW) where this patient with bronchogenic carcinoma lacked evidence of cerebral disease along with a negative CT scan of brain⁵. Moreover, Beck and others demonstrated that the coexistence of hyponatremia and hypouricemia was accompanied by a unique relationship between serum sodium and FEurate⁷⁻¹⁰. The increased FEurate of > 10% that was associated with hyponatremia normalized to < 10% after correction of hyponatremia in SIADH (figure 1).
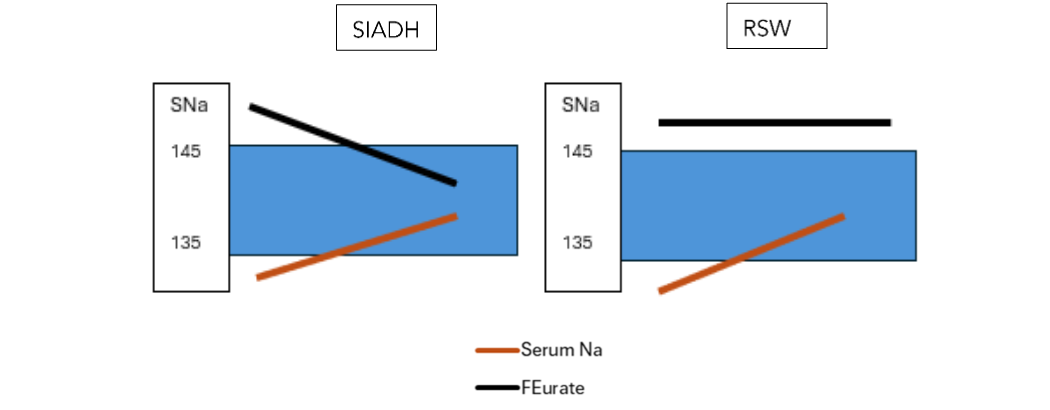
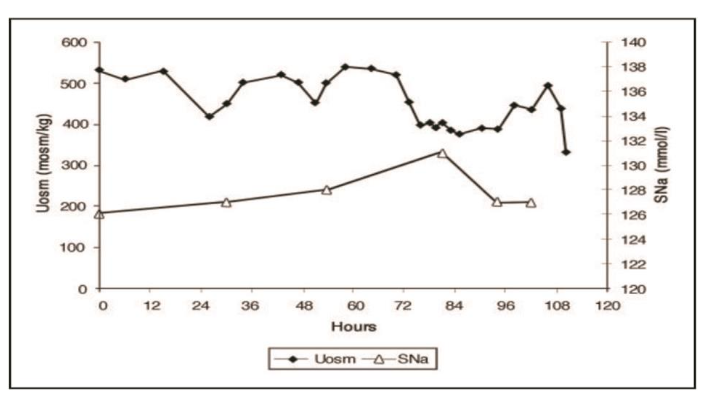 Figure 2A Graph demonstrating urine osmolality (Uosm) and serum sodium (SNa) concentration in response to infusion of isotonic saline in a patient with SIADH – note failure to excrete dilute urines or correct the hyponatremia despite isotonic saline infusion. Reference 13
Figure 2A Graph demonstrating urine osmolality (Uosm) and serum sodium (SNa) concentration in response to infusion of isotonic saline in a patient with SIADH – note failure to excrete dilute urines or correct the hyponatremia despite isotonic saline infusion. Reference 13
In contrast, infusion of isotonic saline in a patient with an unequivocal diagnosis of RSW by having a decreased blood volume and increased renin and aldosterone levels diluted her urine at 13 hours when antidiuretic hormone level in plasma was undetectable and corrected her hyponatremia 48 hours after initiating isotonic saline infusion (figure 2B)¹².
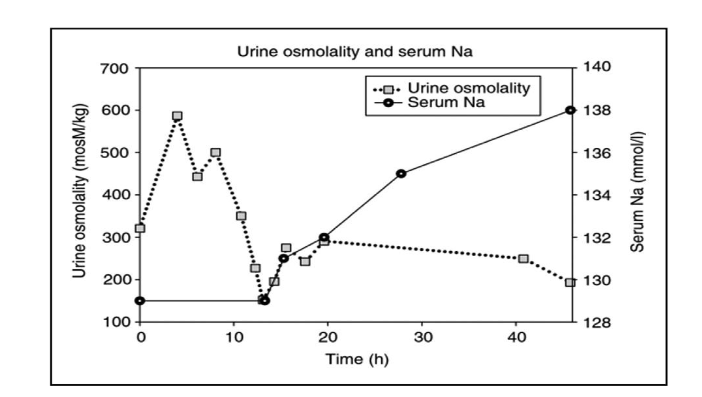
This patient with RSW illustrates the physiologic principle that patients with RSW have an appropriate increase in antidiuretic hormone (ADH) levels as opposed to being inappropriate in SIADH. Under normal conditions, the volume stimulus for ADH secretion is more potent than the osmolar stimulus. A volume depleted patient will, thus, continue to have high ADH levels to maintain the hyponatremia and concentrated urine as long as they remain volume depleted, (figure 3). Isotonic saline infusion eliminates the more potent volume stimulus of ADH secretion to allow the coexistent hypo osmolality to inhibit ADH secretion to dilute the urine by increasing free water excretion and correct the hyponatremia (figures 2B and 3)¹⁷.
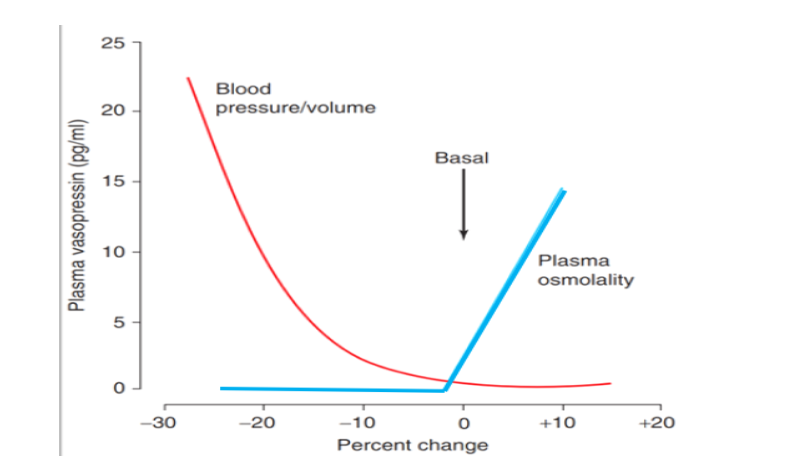 Figure 3. Graph showing antidiuretic hormone response to volume and osmolality where the volume stimulus in red lines is more potent than the osmolar stimulus. Note the marked increase in antidiuretic hormone levels as the patient becomes volume depleted despite the hypoosmolality of plasma¹⁵. Reference 17
Figure 3. Graph showing antidiuretic hormone response to volume and osmolality where the volume stimulus in red lines is more potent than the osmolar stimulus. Note the marked increase in antidiuretic hormone levels as the patient becomes volume depleted despite the hypoosmolality of plasma¹⁵. Reference 17
As noted in figure 4, most of the causes of hyponatremia can be identified with greater accuracy by determining FEurate and being able to differentiate SIADH from RSW by determining FEurate before and after correction of hyponatremia and determining whether isotonic saline infusions will lead to excretion of dilute urines and correction of their hyponatremia.
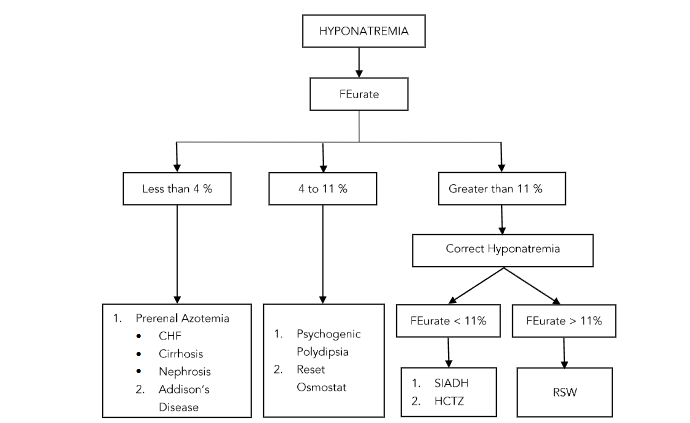
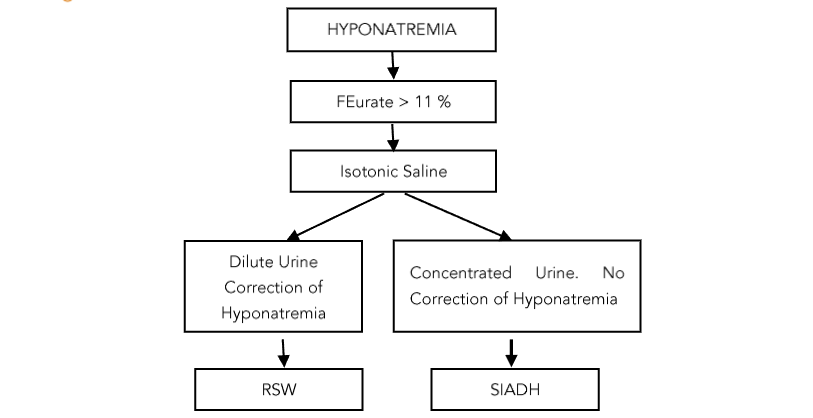
Figure 4. Figure demonstrating the value of determining the fractional excretion of urate (FEurate) in determining most of the causes of hyponatremia and the response of FEurate before and after correction of hyponatremia. Reference 1
High prevalence of renal salt wasting in hyponatremic patients
This new approach to hyponatremia demonstrated the value of determining FEurate and response to isotonic saline infusions to identify with greater accuracy the different causes of hyponatremia in 62 hyponatremic patients from the general medical wards of the hospital. We found 24 (38%) to have RSW, 21 without clinical evidence of cerebral disease to support our proposal to change cerebral to RSW¹⁴. Eleven had persistently increased FEurate after correction of their hyponatremia and 19 diluted their urine within 24 hours after initiating isotonic saline infusions, with 2 having appropriate undetectable levels of plasma ADH when the urine was dilute. Isotonic saline infusions induced excretion of dilute urines to normalize serum sodium within 48 hours in 19 patients with 10 requiring 5% dextrose in water to limit an increase in serum sodium to 6 mmol/L or less in 24 hours to prevent osmotic demyelination¹⁶. Seventeen (27%) had SIADH based on five normalizing a previously increased FEurate after correcting their hyponatremia and 11 failed to excrete dilute urines or correct their hyponatremia when infused with isotonic saline for 72 hours. Nineteen (31%) had a reset osmostat based on a normal FEurate in all 19 patients and 8 spontaneously excreted dilute urine, which is virtually diagnostic of a reset osmostat¹⁹. Of note is that 2 of 17 patients with SIADH, 5 of 19 patients with a reset osmostat and 10 of 24 patients with RSW had baseline UNa less than 20 mmol/L. One patient had a low FEurate because of Addison’s disease and one due to hydrochlorothiazide¹¹. The value of determining FEurate to identify patients with SIADH, reset osmostat and cerebral/renal salt wasting has also been demonstrated in hyponatremic pediatric patients²⁰.
Pathophysiology of renal salt wasting
a) INITIATION PHASE
A comorbid inflammatory condition induces a genetic upregulation of a natriuretic protein, identified as haptoglobin related protein without signal peptide (HPRWSP), that decreases sodium and water reabsorption in the proximal tubule to a point where sodium and water excretion exceeds intake to reduce total body sodium and water and creates a volume depleted state. This can be observed clinically by a rapid increase in urinary volume to induce a volume depleted state with hemodynamic, neural and hormonal adjustments that will respond to isotonic saline infusions⁴. The potent volume stimulus for ADH secretion increases plasma ADH levels to induce hyponatremia as long as the patient is drinking sufficient amounts of water, as virtually all hyponatremic states require sufficient water intake. Water intake must significantly exceed insensible water losses of about 500 ml/day in order to induce hyponatremia.
hyponatremia. This was apparent when daily injections of pitressin to normal human subjects or dogs required an increase in water intake to induce hyponatremia²¹,²². If the subject was receiving isotonic saline infusions with little or no water intake, the development of hyponatremia would be highly unusual unless the patient undergoes the rarely observed desalination where the concentration of sodium and potassium in the urine far exceeds their concentrations in the intake fluid²³. As will be discussed later, patients with RSW can be hyponatremic or normonatremic depending on whether their intake of water is sufficient or whether they received isotonic saline infusions, respectively. Conversely, the lack of sufficient water intake largely defines all cases of hypernatremia.
b) EQUILIBRATED STATE AND CONCEPT OF SALT WASTING ESCAPE
The concept of balance underlies the phenomenon of escape as discussed in greater detail elsewhere²⁴. When discussing sodium balance, our day-to-day intake and output of sodium remain in a state of balance. When this balance is disrupted by RSW, it is understandable that the body cannot sustain the initiating phase of RSW where sodium output exceeds sodium input because this will eventually lead to loss of all exchangeable sodium in the body. To escape this life-threatening outcome, the body escapes this short-lived state by activating hemodynamic, hormonal and neuronal compensations that reduce sodium excretion to match sodium intake to reach a new steady state but at a reduced extracellular volume. The degree of volume depletion will depend on the severity of the defect in sodium transport, and salt intake. Most patients with RSW are seen in this state of renal salt wasting escape and are in sodium balance at a lower extracellular volume which is impossible to determine by usual clinical criteria. The baseline UNa of < 20 mmol/L in 2 of 17 patients with SIADH, 5 of 19 with RO and 10 of 24 patients with RSW had inflammatory conditions that reduced appetite and salt intake when seen during the equilibrated state after they escaped the initiation phase of RSW¹⁴. These data suggest that UNa exceeding 30 to 40 mmol/L has less importance in identifying patients with RSW or even SIADH.
Demonstration of the existence of a natriuretic factor in plasma
When it became evident that RSW appeared to be a syndrome that definitely existed, we proposed in 1993 that a circulating natriuretic protein in plasma might be inducing RSW. We performed renal clearance studies in rats by infusing plasma from patients suspected of having RSW. We felt that a rat model would be ideal for purposes of demonstrating a natriuretic activity because it allowed all counteracting forces to come into play as compared to limited options in in-vitro methods. We studied two groups of patients. The first study included 21 neurosurgical patients who commonly suffer from RSW as proven by critical blood volume studies²⁵⁻²⁷. The second study included 18 patients with Alzheimer disease (AD) who were suspected of having RSW because of being hypouricemic from a proposed increase in FEurate²⁸,²⁹. These Alzheimer’s patients indeed had higher FEurates than patients with multi-infarct dementia (MID)/vascular dementia or in age and gender matched controls³¹. We included patients with more advanced AD with mini mental state examination (MMSE) scores <12 to improve our chances of demonstrating natriuretic activity in their plasma³¹. Because urate is transported exclusively in the proximal tubule, we hypothesized that the natriuretic protein had its major effect on the proximal tubule¹¹. We, therefore, included lithium in these studies because lithium was being used as a marker of proximal tubular sodium transport as sodium and lithium were transported on a one- to- one basis only in the proximal tubule²⁹. There were no changes in blood pressures or glomerular filtration rates in both studies. The fractional excretion of lithium increased from a control of 22.3% and 27.2% to 36.6% and 41.7% and FEsodium increased from a control of 0.3% and 0.33% to 0.59% and 0.63% in the neurosurgical and Alzheimer groups, respectively. Utilizing FElithium as a measure of proximal tubular sodium transport allowed us to estimate the enormous capacity of the distal nephron to transport sodium; being 22% (22.3–0.3) and 36.01% (36.6–0.59) and 26.87% (27.2–0.33) and 41.07% (41.7–0.63) reabsorption in the control and neurosurgical and control and AD plasma, respectively. Both studies were published in 1993 at a time when the state of protein analysis prevented us from identifying the natriuretic protein²,³. We eventually identified the natriuretic protein in 2020 in a patient with a subarachnoid hemorrhage (SAH) and another with AD as HPRWSP⁴. The ability to estimate sodium transport in the proximal tubule by determining FElithium identified the proximal tubule as the major site of HPRWSP action. It should also be pointed out that both patients were normonatremic. We identified RSW occurring in the normonatremic SAH patient by documenting the transition from the initiating to the escape phases of RSW⁴. The absence of hyponatremia in the SAH patient can be explained by isotonic saline infusions being a standard of care for these patients to prevent cerebral vasospasm and ischemia of brain, as well as decrease morbidity and mortality when these salt-wasting patients were erroneously water restricted for a “presumed” diagnosis of SIADH³⁰. The normonatremia in the AD patient could be explained by the lack of water intake due to age-related changes in thirst mechanism and their dementia³¹. These data suggest that normonatremic RSW is a common disorder that needs well-defined criteria for identification.
Identification of Haptoglobin Related Protein Without Signal Peptide as inducer of renal salt wasting
The demonstration of a natriuretic protein existing in the plasma of patients with neurosurgical and Alzheimer diseases in 1993 were notable for exposing the possible high prevalence of RSW in both populations of patients. Both studies were characterized by identical increases in FEsodium, FE lithium and stability of blood pressure and glomerular filtrations rates. (2,3) Improvements in protein analyses over the subsequent years simplified identification of the natriuretic factor as HPRWSP in the sera of RSW associated with SAH and a new syndrome of RSW in AD. It is interesting to note that haptoglobin related protein with signal peptide had no natriuretic activity in our rat model
During acute and active inflammation, a number of acute phase reactants are produced in the liver in response to the cytokine IL-6. Among them are the haptoglobin family (HP) which includes three phenotypes, HP 1-1, HP 1-2 and HP 2-2, as well as haptoglobin related proteins with and without signal peptide. All have different activities ranging from binding free hemoglobin, serving as the trypanosome lytic factor, natriuretic activity, and modification of the inflammatory process – some proinflammatory and others anti-inflammatory. Increased HP has been reported in a multitude of conditions, including many infections, diabetes, obesity, cardiovascular disease, peripheral vascular disease, and head injuries³¹–³⁸.
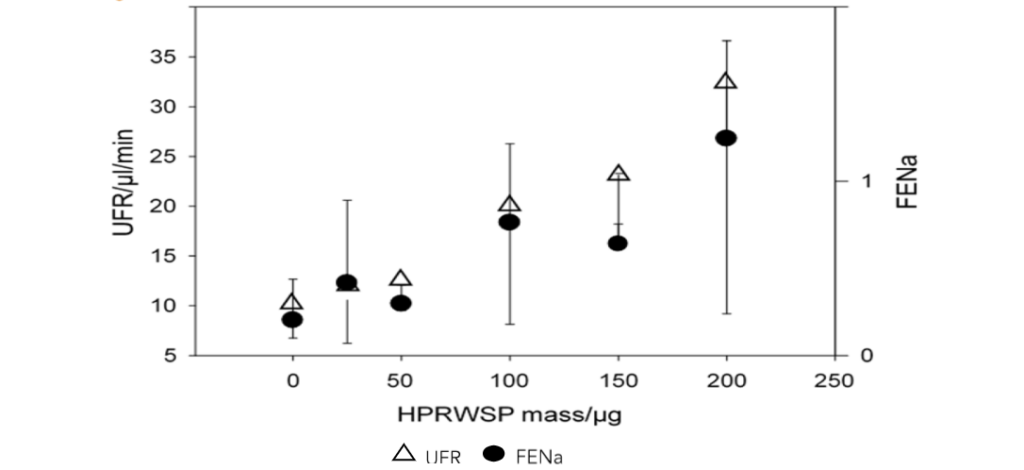
Figure 5. Dose-response increases in fractional sodium excretion and urine flow rates to increasing dose of the natriuretic peptide, haptoglobin-related protein without signal peptide. Reference 4
Haptoglobin related protein (HPR) is a primate specific protein that has a 91% homology with haptoglobin 1-1, a gene locus 2.2 kilobases downstream from the haptoglobin locus and is produced in the liver³⁹. The signal peptide for HPR contains 18–22 amino acids that are located at the N terminus of the mature molecule. The signal peptide with its hydrophobic mid-section is critical for translocating the protein through the endoplasmic reticulum, Golgi apparatus and channels in the fatty cell membrane where signal peptidases cleave the signal peptide before exiting the cell. A protein with a signal peptide will remain in the cytosol—while most if not all proteins are secreted without the signal peptide. Since signal peptides are known to be cleaved prior to protein exiting the cell, there is little mention of proteins being determined with and without the signal peptide, the assumption being that it makes little difference or circulating signal peptides rarely accompany the mature protein molecule. In terms of HPR, however, it is critical to determine how much of the secreted protein contains the signal peptide because HPR with the signal peptide had no natriuretic activity. The retained signal peptide of HPR is unusual but is critical to the formation of the trypanosome lytic factor by combining with high density lipoprotein-apolipoprotein L-1³⁹–⁴².
The signal peptide assumes a critical role of being recognized by the receptor on the trypanosome for endocytosis and eventual lysis of the trypanosome⁴⁰,⁴². Haptoglobin related protein, like haptoglobin, is known to have high-affinity hemoglobin-binding but does not bind to the monocyte/macrophage-specific scavenger receptor CD163⁴³. It is a relatively unknown protein except for being recognized as an important constituent of the trypanosome lytic factor, potential role as a tumor antigen, marker for idiopathic pulmonary fibrosis and now the natriuretic protein that induces RSW⁴⁴,⁴⁵. Because of the important role of the signal peptide in the trypanosome lytic factor, it is important to develop an assay that solely determines blood levels of HPRWSP. Haptoglobin related protein without signal peptide can be developed as a biomarker to identify the potentially large population of RSW patients with and without hyponatremia on first encounter to deliver the appropriate therapy to improve clinical outcomes.
Debates over the existence and prevalence of renal salt wasting
The publication of a high prevalence of RSW in 62 hyponatremic patients in the general medical wards of the hospital and identification of HPRWSP as the natriuretic protein that causes RSW generated much attention from the nephrology community¹. Three debates were conducted in 2022 and 2023: one in April 2022 as part of the annual meeting of the National Kidney Foundation in Boston, another published by the American Society of Nephrology in Kidney360 in June 2022⁴⁶,⁴⁷, and at the annual meeting of the Pediatric Society of Nephrology in Buenos Aires, Argentina in March 2023. None of the debaters arguing against the existence or prevalence of RSW presented data that FEurate determinations and response to isotonic saline infusions do not differentiate SIADH from RSW. It is beyond the scope of this review to discuss the debates except to point out that many criticisms are physiologically incorrect and that two editorials claiming the rarity of cerebral or renal salt wasting cite an article of 49 hyponatremic patients due to a subarachnoid hemorrhage who corrected their hyponatremia in a median of 3 days while infusing isotonic saline infusion alone and erroneously concluded that they all had SIADH⁴⁸–⁵⁰. As discussed earlier, isotonic saline infusion corrects the hyponatremia in RSW but not SIADH. The diagnosis of SIADH was made by the criteria proposed by Schwartz et al without recognizing that these parameters are identical to those found in RSW⁵¹. Any manuscript citing the original criteria for SIADH as the sole means by which the diagnosis of SIADH is made without going through the rigors of differentiating SIADH from RSW should be seriously held in question. Developing HPRWSP as a biomarker of RSW will be able to establish the diagnosis of RSW on first encounter and to identify the appropriate treatment that will improve clinical outcomes.
A new syndrome of renal salt wasting in Alzheimer’s disease
The concept of dehydration or hypovolemia has been frequently used interchangeably but there is a consensus that both represent two distinct clinical entities that is worthy of comment⁵². Dehydration has been classified as “hypertonic dehydration or isotonic dehydration”.
a) HYPERTONIC DEHYDRATION
Hypertonic dehydration characterizes a syndrome that is due to relative and/or absolute abnormalities in total body water where a direct measurement of serum or plasma osmolality of > 300 mOsm/kg excluding the presence of nonelectrolyte solutes such as glucose is the gold standard determination of hypertonic dehydration⁵². Hypertonic dehydration is very applicable to patients with AD because of their failure to drink sufficient amounts of water due to an age-related defect in thirst mechanism and dementia. The major abnormality in hypertonic dehydration is a failure to ingest adequate amounts of water to compensate for insensible water losses or to larger degrees of water loss such as sweating. In this situation the water deficits will induce hypertonicity of the extracellular space, which would osmotically increase water movement out of most cells whose membranes are freely permeable to water. This movement of water minimizes the reduction in extracellular water volume at the expense of a reduction in cell volume⁵².
b) ISOTONIC DEHYDRATION
In contrast to hypertonic dehydration, isotonic dehydration is defined by a state of hypovolemia resulting from water and salt losses such as in prolonged diarrhea, vomiting, hemorrhage and now the newly introduced concept of volume depletion due to a new syndrome of RSW in AD. As discussed earlier, it is extremely difficult to assess the volume status by clinical criteria even by using the National Institute for Health and Care Excellence (NICE) guidelines which can be used to determine whether fluid resuscitation is needed. They suggest the following criteria: 1. systolic blood pressure is < 100 mmHg; 2. Heart rate > 90 beats per minute; 3. Capillary filling pressure > 2 seconds or peripheries are cold to touch; 4. respiratory rate > 20 breaths per minute; 5. National Early Warning Score (NEWS) of 5 or more.
An increase in pulse rate of > 30 beats per minute from a sitting to standing position is consistent with hypovolemia due to significant blood loss⁵³.
A blood urea nitrogen (BUN) to creatinine ratio of > 20 has been shown to be consistent with hypovolemia but we have found it to be absent in patients with RSW due to HPRWSP [8,9]. Because HPRWSP has its major effect on sodium and water transport in the proximal tubule, we have not been able to demonstrate a higher BUN to creatinine ratio in patients with RSW[12,13]. The inhibition of sodium and water transport induced by HPRWSP in the proximal tubule reduces the ability to increase urea concentration in the proximal tubule for passive reabsorption of urea. This is contrasted to hypovolemia in patients with normal kidney function who develop prerenal azotemia, which is characterized by an increase in proximal tubule sodium and water reabsorption to increase the urea concentration gradient for passive urea reabsorption to increase BUN to creatinine ratio, a hallmark of prerenal states. It appears that patients with AD suffer from hypertonic and isotonic dehydration. The failure to recognize volume depletion in AD supports our contention that we cannot accurately assess the volume status of patients by clinical criteria.
A new syndrome of renal salt wasting in Alzheimer’s disease induced by haptoglobin related protein without signal peptide
We previously demonstrated the presence of natriuretic activity in the plasma of 21 and 18 patients with neurosurgical diseases and Alzheimer disease, respectively²,³. We have already shown a high prevalence of RSW in hyponatremic patients from the general medical wards of the hospital and for the first time demonstrate the possibility of RSW occurring in normonatremic patients with AD. We decided to study patients with advanced AD to improve our chances of demonstrating natriuretic activity in their plasma and surprisingly found FElithium to increase rapidly as mini mental state examination (MMSE) scores decreased from 12 to 0 (figure 6)³. Note the progressive increase in FElithium as mini mental state examination scores decrease from 12 to 0. Because there is a dose-responsive increase in FElithium to increasing doses of the natriuretic peptide, blood levels of the natriuretic peptide must be increasing to augment patients’ volume depletion. It is important to note that there is possibility of volume depletion being present at very early stages of dementia.
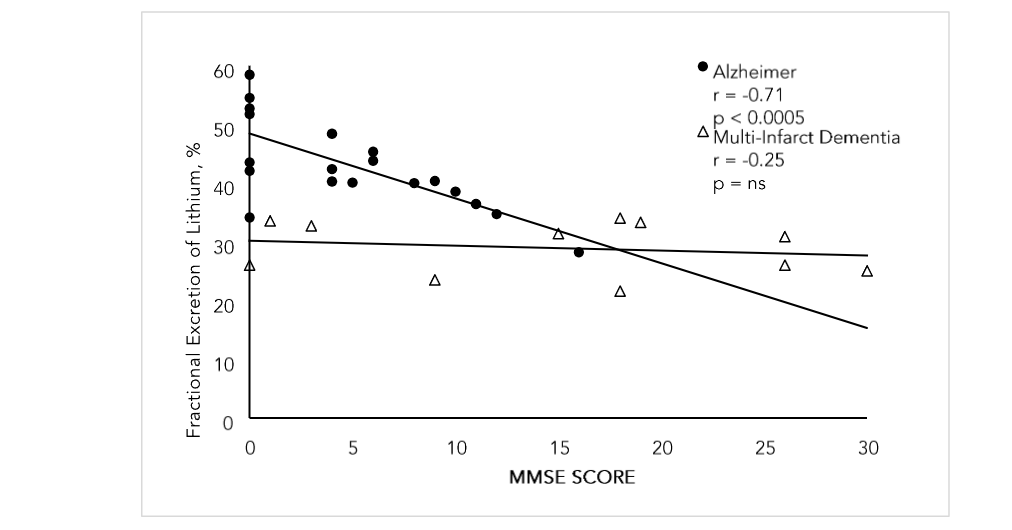
FIGURE 6: Graph showing fractional excretion of lithium in rats infused with plasma of patients with advanced Alzheimer’s disease as compared to patients with multi-infarct dementia. MMSE = mini mental state examination score. Reference 3
Since there was a dose response increase in FEsodium and FElithium with HPRWSP, the blood levels and severity of RSW must have been increasing as the patient became more demented, (figures 5 and 6)¹,³. It is interesting to note that levels of haptoglobin (HP) and HPR appear to have a parallel relationship in human plasma⁵⁴. Haptoglobin levels have been shown to be higher at a time when the patient with AD is at an early stage when they have mild cognitive impairment (MCI) and increase further as the degree of dementia increases⁵⁵,⁵⁶. Haptoglobin 2-2 genotype has been reported to be associated with renal salt wasting in patients with aneurysmal SAH suggesting that the blood levels of HPRWSP were increased to induce RSW as haptoglobin did not have natriuretic activity despite being increased in our patient with SAH and RSW⁴⁰,⁵⁷. These data suggest that the increasing HP levels at early stages of AD may be associated with increased levels of HPRWSP and increasing volume depletion as the dementia worsens, (figure 5)⁵⁵,⁵⁶. Future studies should address the possibility that RSW due to HPRWSP can serve as a biomarker of RSW at different stages of dementia in AD, including MCI. The failure to demonstrate natriuretic activity in the plasma of patients at earlier stages of AD in our rat studies can be explained by rat clearance protocol where the rats were exposed to 2.5 ml of plasma given over a 4.5-hour period²,³. As will be discussed below, this is contrasted to the persistent exposure of AD kidneys to the natriuretic protein that may be present at earlier stages of AD.
Differences in upregulation of the gene that increases production of haptoglobin related protein without signal peptide
It appears that the gene that upregulates the production of HPRWSP has variable degrees of potency and duration. Our study of RSW in the general medical wards of the hospital appears to demonstrate a relatively short period of upregulation of the gene because RSW subsided after resolution of their comorbid, inflammatory condition in 1 to 3 weeks¹. In AD, however, there appears to be a persistent upregulation of the gene and HPRWSP levels. This means that every organ of the body will be exposed to HPRWSP as long as AD exists. Because of the persistent exposure of all organs of the body to this novel and relatively uninvestigated protein, there is an inclination to wonder about the effect of HPRWSP on all organs of the body, especially brain.
Future studies intend to develop HPRWSP as a biomarker of RSW at all stages of AD, including those with MCI. Because HPR with the signal peptide did not have any natriuretic activity, it is important to determine HPRWSP levels in all of these studies. The present ELISA for determining HPR levels in plasma does not differentiate whether HPR has or lacks the signal peptide. Although most or all signal peptides are cleaved by signal peptidases before exiting the cell, HPR with signal peptide is present in the circulation as a key element in the trypanosome lytic factor⁴⁰–⁴². We are in the process of developing strategies to determine HPRWSP levels in our studies. It will also be interesting to develop an inhibitor to HPRWSP to treat patients with RSW and to assess clinical outcomes, especially brain function in AD. An inhibitor to HPRWSP will improve treatment of all patients with RSW because isotonic saline infusions significantly increase urine output and nocturia to create poor quality of life issues. Our sense is that RSW is probably a common syndrome in AD that highlights our perceived inability to assess the volume status of patients by usual clinical criteria. Lastly, we must emphasize the probability of RSW occurring commonly in normonatremic patients, especially in neurosurgical patients and in intensive care units.
In conclusion, we describe the development of a more physiologic approach to evaluating patients with hyponatremia and potentially large population of normonatremic patients with RSW. This physiologic approach was found to be superior to the ineffective volume approach. This new approach showed a high prevalence of RSW in hyponatremic patients from the general medical wards of the hospital that were contrary to the view among internists that RSW was a rare clinical entity. Moreover, rat clearance studies demonstrated natriuretic activity in the plasma of patients with neurosurgical diseases and with AD. The natriuretic factor was later identified as HPRWSP. These controversial data led to three debates that contested the existence and prevalence of cerebral salt wasting which is more appropriately being termed RSW. None of the doubters of RSW have provided data to prove our physiologic approach to be wrong. Future studies intend to develop HPRWSP as a biomarker of RSW in hyponatremic and normonatremic patient with RSW, including the probability of a high prevalence of a new syndrome of RSW in AD. The biomarker will identify patients with RSW on first encounter at which time the appropriate therapy will improve clinical outcomes.
Conflict of Interest:
The authors have no conflicts of interest to declare.
Funding Statement:
This review received no external funding.
Acknowledgements:
Conception /writing – John Maesaka
Louis J. Imbriano, M.D. [this author contributed equally to this work]
Candace Grant, M.D. [this author contributed equally to this work]
Nobuyuki Miyawaki, M.D. [The author reviewed multiple revision of the manuscript, helped develop tables and graphs and reviewed the literature]
Rajanandini Muralidharan, M.D. [Author reviewed the manuscript with major contributions made on recruiting and discussing neurologic diseases and Alzheimer disease patients]
The authors wish to thank Maria Kollarus for her outstanding contribution to executing the very different protocol that demonstrated the high prevalence of RSW in the general medical wards of the hospital.
References
1. Maesaka JK, Imbriano LJ, Miyawaki N. High prevalence of renal salt wasting without cerebral disease as cause of hyponatremia in general medical wards. Am J Med Sci. 2018;356:15–22.
2. Maesaka JK, Venkatesan J, Piccione JM, et al. Plasma natriuretic factor[s] in patients with intracranial disease, renal salt wasting and hyperuricosuria. Life Sci 1993; 52:1875–1882.
3. Maesaka JK, Wolf-Klein G, Piccione JM, et al. Hypouricemia, abnormal renal tubular urate transport, and plasma natriuretic factor[s] in patients with Alzheimer’s disease. J Am Geriatr Soc 1993; 41:501–506.
4. Maesaka JK, Imbriano LJ, Pinkhasov A, et al. Identification of a novel natriuretic protein in patients with cerebral-renal salt wasting- Implications for enhanced diagnosis. Am Jour Med Sci 2021; 361:261-268.
5. Maesaka JK, Batuman V, Yudd M, et al. Hyponatremia and hypouricemia: differentiation from SIADH. Clin Nephrol 1990; 33:174–178.
6. Chung H.-M, Kluge R, Schrier, R.W. Clinical assessment of extracellular fluid volume in hyponatremia. Am. J. Med. 1987, 83, 905–908.
7. Beck LH. Hypouricemia in the syndrome of inappropriate secretion of antidiuretic hormone. N Engl J Med 1979; 301:528–530.
8. Assadi F.K. John E.G. Hypouricemia in Neonates with Syndrome of Inappropriate Secretion of Antidiuretic Hormone. Pediatr. Res. 1985; 19, 424–427.
9. Mee, E.J.D, Van Assendelft P.B, Nieuwenhuis M.G. Elevation of uric acid clearance caused by inappropriate antidiuretic hormone secretion. Acta Med. Scand. 1971; 189, 69–72.
10. Sonnenblick M, Rosin A. Increased uric acid clearance in the syndrome of inappropriate secretion of antidiuretic hormone. Isr. J. Med Sci. 1988; 24, 20–23.
11. Weinman EJ, Steplock D, Suki WN, et al. Urate reabsorption in proximal convoluted tubule of the rat kidney. Am J Physiol 1976; 231:509–515.
12. Maesaka JK, Miyawaki N, Palaia T, et al.. Renal salt wasting without cerebral disease: value of determining urate in hyponatremia. Kidney Int. 2007;71:822–826.
13. Bitew S, Imbriano L, Miyawaki N, et al. More on renal salt wasting without cerebral disease, response to saline infusion. Clin J Amer Soc Nephol. 2009;4:309–315.
14. Maesaka JK, Imbriano LJ, Ali NM, et al. Is it cerebral or renal salt wasting? Kidney Int 2009; 76:934–938.
15. Bartter FC, Schwartz WB. The syndrome of inappropriate secretion of antidiuretic hormone. Am J Med 1967; 42:790–805.
16. Jancic N, Verbalis JG. Evaluation and management of hypo-osmolality in hospitalized patients. Endocrinol Metab Clin North Am 2003; 32:459-481.
17. Stricker E.M, Verbalis J. Water intake and body fluids. In Fundamental Neuroscience, 2nd ed.; Squire, L.E., Roberts, J.L., Spitzer, N.C., Zigmond, M.J., McConnell, S.K., Bloom, F.E., Eds.; Elsevier: La Jolla, CA, USA, 2003; pp. 1011–1029.
18. Sterns RH. Disorder of plasma sodium-causes, consequences and correction. N Engl J Med 2015;372:55–65
19. Imbriano LJ, Ilamathi E, Ali NM, et al. Normal fractional urate excretion identifies hyponatremic patients with reset osmostat. J Nephrol 2012; 25:833–838.
20. Assadi F, Mazaheri M. Differentiating syndrome of inappropriate ADH, reset Osmostat, cerebral/ renal salt wasting using fractional urate excretion. J Pediatr Endocrinol Metab 2020 Nov12;34(1):137-140. Doi:10.1515/jpem-2020-0379. PMID:33180045
21. Jaenike JR, Waterhouse C. The renal response to sustained administration of vasopressin and water in man. J Clin Endocrinol Metab 1961; 21:231–242.
22. Levinsky NG, Davidson DG, Berliner RW. Changes in urine concentration during prolonged administration of vasopressin and water. Am J Physiol 1959; 196:451–456.
23. Steele A, Gowrishankar M, Abrahamson S, et al. Postoperative hyponatremia despite near-isotonic saline infusion: a phenomenon of desalination. Ann Intern Med 1997; 126:20–25.
24. Maesaka JK, Imbriano L, Miyawaki N. Determining fractional urate excretion rates in hyponatremic conditions and improved methods to distinguish cerebral/renal salt wasting from the syndrome of inappropriate secretion of antidiuretic hormone. Frontiers in Medicine. November 2018; Volume 5: article 319 000
25. Nelson PB, Seif SM, Maroon JC, et al. Hyponatremia in intracranial disease: perhaps not the syndrome of inappropriate secretion of antidiuretic hormone [SIADH]. J Neurosurg 1981; 55:938–941.
26. Wijdicks EF, Vermeulen M, ten Haaf JA, et al. Volume depletion and natriuresis in patients with a ruptured intracranial aneurysm. Ann Neurol 1985; 18:211–216.
27. Sivakumar V, Rajshekhar V, Chandy MJ. Management of neurosurgical patients with hyponatremia and natriuresis. Neurosurgery 1994; 34:269–274.
28. Kasa M, Bierma TJ, Waterstraat F Jr, et al. Routine blood chemistry screen: a diagnostic aid for Alzheimer’s disease. Neuroepidemiology, 1989 ; 8[5]pp.254-256
29. Hayslett JP, Kashgarian M. A micro puncture study of the renal handling of lithium. Pflug Arch 1979; 380:159–163.
30. Wijdicks EF, Vermeulen M, Hijdra A, et al. Hyponatremia and cerebral infarction in patients with ruptured intracranial aneurysm: is fluid restriction harmful. Ann Neurol. 1985;17:137–140.
31. Begg DP. Disturbances of thirst and fluid balance associated with aging. Physiol Behav. 2017 Sep 1;178:28-34. doi: 10.1016/j.physbeh.2017.03. 003. Epub 2017 Mar 4. PMID: 28267585.
32. Arredouani, M.; Matthijs, P.; Van Hoeyveld, E.; Kasran, A.; Baumann, H.; Ceuppens, J.L.; Stevens, E. Haptoglobin directly affects T cells and suppresses T helper cell type 2 cytokine release. Immunology 2003, 108, 144–151.
33. Levy, A.P. Haptoglobin: A major susceptibility gene for diabetic cardiovascular disease. Isr. Med. Assoc. J. IMAJ 2004, 6, 308–310.
34. Quaye, I.K.; Ababio, G.; Amoah, A.G. Haptoglobin 2-2 Phenotype is a Risk Factor for Type 2 Diabetes in Ghana. J. Atheroscler. Thromb. 2006, 13, 90–94.
35. Quaye, I.K. Haptoglobin, inflammation and disease. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 735–742.
36. Delanghe, J.; Langlois, M.; Duprez, D.; De Buyzere, M.; Clement, D. Haptoglobin polymorphism and peripheral arterial occlusive disease. Atherosclerosis 1999, 145, 287–292.
37. Yildirim, C.; Kekec, Z.; Sozuer, E.M.; Ikizceli, I.; Avsarogullari, L. Blood levels of acute phase reactants with traffic accidents. Soudni Lekarstvi 2004, 49, 25–29
38. Arredouani, M.S.; Kasran, A.; Vanoirbeek, J.A.; Berger, F.G.; Baumann, H.; Ceuppens, J.L. Haptoglobin dampens endotoxininduced inflammatory effects both in vitro and in vivo. Immunology 2005, 114, 263–271.
39. Imrie HJ, Fowkes FJ, Migot-Nabias F, et al. Individual variation in levels of haptoglobin-related protein in children from Gabon. PLoS ONE. 2012;7:e49816.
40. Harrington JM, Nishanova T, Rose Pena S, et al. A retained secretor signal peptide mediates high density lipoprotein [HDL] assembly and function of haptoglobin-related protein. J Biol Chem 2014;289 number 36:24811-24820
41. Owji H, Nezafat N, Negahdaripour M, et al. A comprehensive review of signal peptides: structure, roles, and applications. Euro J Cell. 2018; 97[6]:422–441.
42. Hardwick RJ, Menard A, Sironi M, et al. Haptoglobin [HP] and haptoglobin-related protein [HPR] copy number variation, natural selection, and trypanosomiasis, Hum Genet 2014; 133: 69-83 DOI 10.1007/s00439-013-1352-x
43. Nielsen MJ, Petersen SV, Jacobsen C, et al. Haptoglobin-related protein is a high-affinity hemoglobin-binding plasma protein. Blood. 2006;108:2846–2849.
44. Saraswat M, Joenvaara S, Tohmola T, et al. Label-free plasma proteomics identifies haptoglobin-related protein as candidate marker of idiopathic pulmonary fibrosis and dysregulation of complement and oxidative pathways. Scientific Reports 2020;10:7787 https://doi.org/10.1038/s41598-020-64759-
45. Kuhajda FP, Katumuluwa AI, Pasternak GR. Expression of haptoglobin-related protein and its potential role as a tumor antigen. Proc Natl Acad Sci 1989;86:1188-1192
46. Maesaka JK, Imbriano LJ. Debates in Nephrology. Cerebral salt wasting is a real cause of hyponatremia: PRO. Kidney360. Published ahead of print 2022 DOI: 10.34067/KID.0001422022.
47. Sterns R, Rondon-Berrios H. Debates in Nephrology. Cerebral salt wasting is a real cause of hyponatremia: CON. Kidney360 published ahead of print, 2022; DOI 10.3406/KID.0001412022a
48. Verbalis J.G Hyponatremia with intracranial disease: not often cerebral salt wasting. J Clin Endocrinol Metab 2014;99:59-62 https://doi.org/10.1210/jc.2013-4289
49. Verbalis J.G. The curious story of cerebral salt wasting. Fact or fiction. Clin J Amer Soc Nephrol. 2020;15:1666-1668. DOI:https://doi.org/10.2215/CJN.00070120
50. Hannon MJ, Behan LA, O’Brien MM, et al. Hyponatremia following mild/moderate subarachnoid hemorrhage is due to SIAD and glucocorticoid deficiency and not cerebral salt wasting. J Clin Endocrinol Metab 2014;99:291-298.
51. Schwartz WB, Bennett W, Curelop S. A syndrome of renal sodium loss and hyponatremia probably resulting from inappropriate secretion of antidiuretic hormone. Am J Med 1957;23:529–42.
52. Lacey J, Corbett J, Forni L, et al. A multidisciplinary consensus on dehydration: definitions, diagnostic methods and clinical implications. Annala Med. 2019;51,NOS,3-4,232-251. https://doi.org/10.1080/07853890.2019.1628352l
53. NICE clinical guidelines 174. Intravenous fluid therapy in adults in hospital. London: NICE 2013
54. Skytthe MK, Sørensen AL, Hennig D, et al., Haptoglobin-related protein in human plasma correlates to haptoglobin concentrations and phenotypes. Scand J Clin Lab Invest. 2022 Oct;82 (6):461-466. doi: 10.1080/00365513.2022.212207 6. Epub 2022 Sep 21. PMID: 36129375.
55. Song IU, Kim YD, Chung SW, et al. Association between serum haptoglobin and the pathogenesis of Alzheimer’s disease. Int Med. 2015;54:453–456.
56. Cen-Jing Zhu, Guo-Xin Jiang, Jin-Mei Chen, et al. Serum haptoglobin in Chinese patients with Alzheimer’s disease and mild cognitive impairment: A case-control study. Brain Res Bull. 2018 Mar:13 7:301-305. Doi:10.1016/j.brainresbull.2018.01.005
57. Murthy SB, Caplan J, Levy AP, et al. Haptoglobin 2-2 genotype is associated with cerebral salt wasting syndrome in aneurysmal subarachnoid hemorrhage. Neurosurgery. 2016;78:71–76.




