Headache Prevalence in Neuropathic Pain Patients
Increased Headache Prevalence in Patients with Neuropathic Pain
Jennifer R Cheng1, Megan R Heffernan1, Vi Le1, Hsinlin T Cheng1
- Department of Neurology, Massachusetts General Hospital/ Harvard Medical School, Boston, Massachusetts, United States of Americ
OPEN ACCESS
PUBLISHED: 30 November 2024
CITATION: Cheng, JR., Heffernan, MR., et al., 2024. Increased Headache Prevalence in Patients with Neuropathic Pain. Medical Research Archives, [online] 12(11). https://doi.org/10.18103/mra.v12i11.6075
COPYRIGHT © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i11.6075
ISSN 2375-1924
ABSTRACT
Patients with neuropathic symptoms, including neuropathic pain and autonomic symptoms, frequently have headaches. They were evaluated by neurologists and underwent a diagnostic skin biopsy for small fiber neuropathy at Massachusetts General Hospital from 2018-2020. The correlations of neuropathic pain, autonomic symptoms, and headaches were examined. The results of skin biopsy, nerve conduction velocity/electromyography, and autonomic function testing were analyzed.
Most (73.8%) of the patients who underwent a skin biopsy were females. The mean age was 46.8. Most patients had neuropathic pain (61.3%). In addition, autonomic symptoms were present in 48.0% of patients. The prevalence of headaches was 48.2% in the whole study cohort, with 73.2% of headaches fulfilling the criteria of migraine.
We detected female dominance in patients who had headaches and migraine but not in the neuropathic pain and autonomic symptom groups. The correlations between headaches, migraine, neuropathic pain, and autonomic symptoms were examined. There were statistical correlations between headaches and neuropathic pain (p< 0.0001) and autonomic symptoms (p< 0.01). Similar trends were detected for migraine with neuropathic pain but not with autonomic symptoms.
The symptomatology was analyzed with the results of skin biopsy, nerve conduction velocity/electromyography, and autonomic function testing. Only autonomic symptoms have a significant positive correlation with positive autonomic function testing results. Otherwise, the results of skin biopsy, nerve conduction velocity/electromyography, and autonomic function testing had no significant correlation with neuropathic pain, headaches, and migraine. The lack of positive correlations between peripheral neuropathy studies and headaches suggests centralized mechanisms likely mediate headaches in patients with neuropathic pain and autonomic symptoms.
Keywords
headache, neuropathic pain, autonomic symptoms, migraine, small fiber neuropathy
Introduction
Patients with neuropathic pain (NPP) and autonomic symptoms (AS) are frequently encountered in Neurology practice. Many of these patients with NPP and AS were considered to have small fiber neuropathy (SFN), which affects the small thinly-myelinated (Aδ) and unmyelinated (C) nerve fibers that mediate sensory and autonomic functions. It is a prevalent condition that affects millions of Americans. Most SFN patients experience either positive sensory symptoms of pain or negative symptoms of sensory loss in distal extremities in a length-dependent pattern. Less commonly, some SFN patients have pain that affects other body parts, including the face and trunk. Neuropathic pain is caused by illnesses or injuries that damage peripheral small or large nerve fibers. It ranges from pins and needle sensations to burning, lancinating, shooting, electric-like, and stabbing pain. A subgroup of NPP patients experiences allodynia (non-painful stimulations, such as light touching, which becomes painful) and paresthesia (abnormal quality of sensations). In addition, SFN could affect autonomic nerve fibers, which are unmyelinated nerve fibers that innervate the skin, blood vessels, soft tissues, and internal organs, to generate AS. Small fiber neuropathy could affect the sympathetic and parasympathetic nerves and cause dizziness, digestive difficulties, cardiac arrhythmias, sweating, and genitourinary symptoms. Both NPP and AS could be devastating and cause significant individual physical and functional disability, resulting in significant societal burdens and losses.
In addition to peripheral neuropathies, NPP and AS could be generated from central brain and spinal cord mechanisms. The evidence of centralized pain is supported by bioimaging and functional imaging studies that demonstrate the activation of somatosensory structures. Good examples of centralized pain are fibromyalgia, temporomandibular disorders, and thalamic pain syndrome. Many patients with symptoms of fibromyalgia have small fiber neuropathy.
It is a common practice that most patients with peripheral NPP and AS to have diagnostic studies, including a diagnostic skin biopsy (SB), nerve conduction studies/electromyography (NCV/EMG), and autonomic function tests (AFT) to establish the diagnosis of small versus large fiber neuropathy. An SB quantifies the degree of neuropathy by measurements of epidermal nerve fiber density; An NCV/EMG measures nerve conduction velocity and resting/active muscle electric activities; and an AFT tests sympathetic/parasympathetic responses to maneuvers such as a tilt table, Valsalva activities, and Quantitative Sudomotor Axon Reflex Testing.
The comorbidity of headaches and neuropathic symptoms is commonly observed in clinical practice. However, the pathological correlations between SFN and headaches are not well-established. Several potential mechanisms connect headache, NPP, and AS. First, many headaches, especially migraine and trigeminal autonomic cephalalgias, are triggered by increased excitability of trigeminal nerve endings that innervate the dura matter and cerebral vasculature. These trigeminal nerve endings, mostly small nerve fibers, expressed calcitonin-gene-related peptides and other nociceptive neurotransmitters. Second, increased peripheral signals generated from small nociceptive nerve endings from cutaneous and neuromuscular components of the head and neck might contribute to tension-type headaches. Third, NPP and AS from SFN in the trigeminal sensory complex could trigger and aggravate the existing headache disorders, resulting in intractable chronic headaches. Fourth, dysautonomia, such as postural orthostatic tachycardia syndrome which could present as a result of SFN, is often considered a trigger and collateral symptom of headache and is frequently overseen by patients and providers. Lastly, sweat, gastrointestinal, and urogenital dysfunctions are common AS that are associated with patients with headaches.
In the current study, we retrospectively studied the correlations between neuropathic (sensory and autonomic) symptoms and headaches. We determined the comorbidities of chronic headaches and migraine by using available medical records and the results of diagnostic studies, including SB, NCV/EMG, and AFT. Our data provide evidence that supports the correlations between neuropathic symptoms and headaches.
Methods
Study design and patient populations
In this retrospective study, we reviewed the medical records of adults (older than 18 years, primarily from the north-eastern United States) with a distal-leg, neurodiagnostic skin biopsy at the Massachusetts General Hospital (MGH), Boston, Massachusetts, USA, from 9/1/2018 to 12/31/2019. The period was chosen before the Coronavirus disease 2019 (COVID-19) pandemic to exclude patients who have COVID-induced neuropathy and headaches. The study was performed following the standards of the Declaration of Helsinki and was approved by the Mass General Brigham Institutional Review Board (Protocol #: 2019P003169).
Inclusion criteria: Patients were evaluated by a neurologist and underwent a diagnostic skin biopsy at MGH.
Exclusion criteria: Patients had neuropathic and autonomic symptoms but were not evaluated by a neurologist.
Primary outcome: Prevalence of headaches and migraine more than 6 months in patients with neuropathic pain and autonomic symptoms. Secondary outcome: Correlation between headache and migraine and the results of SB, NCV/EMG, and AFT.
Diagnostic criteria: Neuropathic pain was assessed by a neurologist according to the guidelines published by EFNS. Briefly, NPP is characterized by spontaneous ongoing or shooting pain and evoked amplified pain responses after noxious or non-noxious stimuli. Patients with NPP have pain described from pins and needle sensations to burning, lancinating, shooting, electric-like, and stabbing pain. A neurologist documented NPP’s characteristics, location, and lateralization in the reviewed medical records. A headache is a general pain affecting one or multiple head areas. The information on headache location and characteristics was documented by a neurologist. Migraine headaches are determined using the criteria from ICHD-3.
Autonomic medical history was collected as described by Goldstein and evaluated by a neurologist. Common AS include orthostatic dizziness with tachycardia or hypotension; dyspepsia, constipation, diarrhea from GI motility dysfunctions; increased or reduced sweating; sexual/bladder dysfunction with abnormal urological evaluation; dry mouth/eyes not related to medication use.
Diagnostic studies
Two or three-mm diameter skin punches were collected from the anesthetized standard distal-leg site, 10 cm above the lateral malleolus. The samples were fixed and cut into 50 µm sections. The epidermal nerve fiber densities per mm2 skin surface were measured using PGP9.5 immunohistochemistry analysis as previously described at the MGH skin biopsy laboratory. The results were reported as average pan-neuronal marker PGP9.5-positive epidermal nerve fiber densities per mm2 skin surface area. Measured IENDs less than the 5th centile of the predicted age-matched normal distribution confirmed SFN.
The standardized nerve conduction study and electromyography were performed at the MGH EMG laboratory. Briefly, Sensory conduction studies, including ulnar, median, superficial radial, sural, superficial peroneal, and medial plantar sensory responses were evaluated. Motor nerve conduction studies were performed including ulnar, median, radial, peroneal, and tibial nerves. An electromyogram was performed on the deltoid, biceps, triceps, flex carpi radialis, first interosseous, vastus medialis, anterior tibialis, and gastrocnemius.
The criteria for the diagnosis of polyneuropathy were described by McCorquodale and Smith. The results were reviewed and recorded as normal and abnormal in nerve conduction velocities, action potential amplitudes, and electric muscle activities. A subgroup of patients with AS also had diagnostic composite AFT, consisting of heart rate variability during deep breathing, heart and blood-pressure responses to the Valsalva maneuver, a tilt-table test, and quantitative sudomotor axon reflex testing at the MGH EMG lab.
Statistics
Independent t-tests were used to assess measurements between the 2 study groups, and the chi-square statistic was used to determine the differences in proportions. P values < 0.05 were considered significant. Data were presented as mean ± SD. No corrections for multiple comparisons were applied.
The primary outcome variables were the prevalence of headaches in general and migraine. The statistical analysis for the differences in the prevalence of headache syndromes between test groups was performed using a chi-square test. In a second step, we analyzed the prevalence of headache syndromes stratified by gender (females versus males), age (< 50 versus > 50-year-old), and BMI (< 30 versus >30) analysis was performed by Prism software (GraphPad Software, San Diego, CA, USA). The level of significance was set at 0.05.
Results
STUDY DESIGN AND DATA COLLECTION
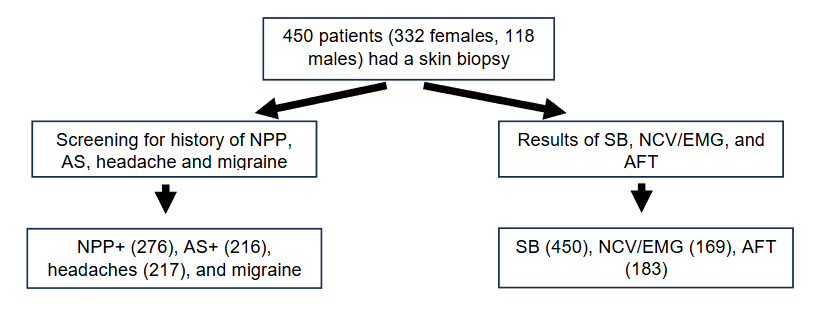
The study was designed as a retrospective chart review. All patients had neurological evaluations by MGH neurologists who requested SB because of documented clinical symptoms of numbness, NPP, and AS from 9/2018 to 12/31/2019. Medical records were reviewed for symptomatology and results of SB, NCV/EMG, and AFT, and the numbers of each group were listed in Figure 1. A total of 450 patients met the inclusion criteria. Their medical records were reviewed by a neurologist for NPP, AS, headaches, and migraine. Collected data detected that 276 patients had NPP, 216 patients had AS, 217 patients had headaches (217), and 159 patients had migraine. In parallel, SB, NCV/EMG, and AFT results were collected. Ninety-four out of 450 patients had a positive SB; Among the 169 patients who had NCV/EMG, 44 had a positive result. One hundred and thirty out of 183 patients had a positive AFT.
PATIENT DEMOGRAPHICS AND PREVALENCE OF PAINFUL SYMPTOMS AND HEADACHES
Most skin biopsies were requested because of documented clinical symptoms of numbness, NPP, and AS. All patients had neurological evaluations by MGH neurologists. In this population, 332 (73.8%) patients were female and 118(26.2%) patients were male. Their mean age was 46.8 (SD: 14.9), with 54.2% below age 50 and 45.8% older than or equal to age 50. The mean BMI was 27.1 (SD: 6.4), with 191 (42.4%) patients with BMI < 25 and 259 (57.6%) patients with BMI > 25.
| Demographics | N(%) |
|---|---|
| Genders | Female 332(73.8) |
| Male 118(26.2) | |
| Age | Mean (SD) 46.8(14.9) |
| <50 244(54.2) | |
| >50 206(45.8) | |
| BMI | Mean (SD) 27.1(6.4) |
| < 25 191(42.4) | |
| > 25 259(57.6) | |
| Total | 450(100) |
THE CORRELATION BETWEEN GENDER, AGE, AND BMI IN THE DEVELOPMENT OF SYMPTOMATOLOGY
The correlation of general painful symptoms (Pain#), NPP, AS, headaches, and migraine with perimeters of gender (female vs male), age (<50 vs >50), and BMI (<25 vs, >25) is demonstrated in Table 2. Three hundred and sixty-four (80.9%) patients had non-specific pain (Pain#, Table 2), including NPP and nociceptive pain from musculoskeletal origin. The mean age of patients with painful symptoms was 46.8 (SD= 14.3), with painful symptoms more likely to develop in patients equal to or older than 50 (p=0.0412, compared patients <50 and >50). Neuropathic pain was reported by 276 (61.3%) patients. Most patients (78.4%) reported NPP affecting more than two body parts, including the face, trunk, and upper and lower extremities. Ninety-eight percent of NPP had bilateral distribution. Eighty-six percent of patients with NPP in head and neck. Two hundred and sixteen (48%) patients also had AS. There was no statistical significance between patients with NPP and AS using the perimeters of gender, age, and BMI.
Two hundred and seventeen (48.2%) patients had headaches, and 159 (35.3%) of these headaches were categorized as migraine. The statistical analysis using gender (female vs male) as a perimeter revealed that significantly more female patients had headaches (p=0.0107) and migraine (p=0.001), compared to male patients. In contrast, no difference in the other perimeters, including age and BMI, was associated with the prevalence of headaches and migraine.
| Patient symptomatology | N(%) | Pain# | NPP | AS | Headaches | Migraine | Total |
|---|---|---|---|---|---|---|---|
| Genders | Female 270(81.3) | 205(61.7) | 165(49.7) | 172(51.8)* | 132(39.8)*** | 332(100) | |
| Male 94(79.7) | 71(60.2) | 51(43.2) | 45(38.1) | 27(22.9) | 118(100) | ||
| Age | Mean (SD) 46.8(14.3) | 46.3(14.5) | 45.1(15.5) | 46.3(14.5) | 46.2(14.8) | ||
| <50 206(84.4) | 153(62.7) | 125(51.2) | 120(49.2) | 89(36.5) | |||
| > 50 158(76.7)* | 123(59.7) | 91(44.2) | 97(47.1) | 70(34.0) | 206(100) | ||
| BMI | Mean ( SD) 26.9(5.7) | 26.8(5.7) | 27(6.0) | 27(6.5) | 26.6(5.9) | ||
| < 25 156(81.7) | 121(63.4) | 90(47.1) | 96(50.3) | 73(38.2) | 191(100) | ||
| > 25 208(80.3) | 155(59.8) | 126(48.6) | 121(46.7) | 86(33.2) | 259(100) | ||
| Total | 364(80.9) | 276(61.3) | 216(48.0) | 217(48.2) | 159(35.3) | 450 (100) |
INCREASE PREVALENCE OF HEADACHES AND MIGRAINE IN PATIENTS WITH NPP AND AS
The correlations between the development of headaches and migraine in patients with NPP are listed in Table 3. The prevalence of headaches was significantly higher in patients with NPP (p<0.0001). In parallel, NPP patients also had a higher prevalence of migraine (p=0.0201). In contrast, patients with AS are also more likely to develop headaches (p=0.0051) but not migraine (p=0.946).
We did not detect any headache patients who met the criteria of cluster headache and trigeminal autonomic cephalagias.
| The prevalence of headaches and migraine in patients with neuropathic pain and autonomic symptoms | N (%) | +NPP | -NPP | Total |
|---|---|---|---|---|
| Headaches | 151(69.6)**** | 66(30.4) | 217 (100) | |
| Migraine | 109(68.6)* | 50(31.4) | 159 (100) | |
| Total | 276(61.3) | 174(38.7) |
| The prevalence of headaches and migraine in patients with autonomic symptoms | N (%) | +AS | -AS | Total |
|---|---|---|---|---|
| Headaches | 119(54.8)** | 98(45.2) | 217 (100) | |
| Migraine | 97(41.6) | 136(58.4) | 159 (100) | |
| Total | 216(48.0) | 234(52.0) |
THE CORRELATION BETWEEN TEST RESULTS AND THE DEVELOPMENT OF PAINFUL AND AUTONOMIC SYMPTOMS
A diagnostic SB, EMG, and AFT are frequently requested for patients with peripheral neurological symptoms. All 450 patients received an SB. Among them, 94 (20.9%) patients had positive SB results. Patients who were female (p=0.0011), > 50-year-old (p=0.0053), and had a BMI > 25 (p<0.0001) were more likely to have a positive skin biopsy. However, positive SB results were not significantly correlated with the development of painful symptoms, including NPP, headaches, and migraine. In contrast, patients with a positive SB were more likely to have AS (p=0.0343).
An NCV/EMG is a diagnostic tool for large fiber neuropathy. Forty-four out of 313 (26%) patients with headaches had positive NCV/EMG results with the diagnosis of either peripheral neuropathy or radiculopathy. Female gender and older (> 50) were more likely to have a positive NCV/EMG result. The results of NCV/EMG are not associated with the development of NPP, headaches, and migraine.
An AFT was performed on 183 patients. AS is the only symptom that correlates significantly with positive AFT results. None of the painful symptoms, including NPP, headaches, and migraine were significantly associated with positive AFT results.
| The correlation between test results and symptoms | N (%) | SB+ | EMG+ | AFT+ | Total |
|---|---|---|---|---|---|
| Genders | Female 57(17.2)** | 25(21.2)* | 104(71.2) | 332(100) | |
| Male 37(31.4) | 19(37.3) | 26(70.3) | 118(100) | ||
| Age | <50 39(16.0) | 14(17.1) | 79(73.1) | 244(100) | |
| > 50 55(26.7)** | 29(34.1)* | 48(66.7) | 206(100) | ||
| BMI | < 25 26(13.6) | 18(31.0) | 61(76.3) | 191(100) | |
| > 25 68(26.3)**** | 26(23.4) | 69(67.0) | 259(100) | ||
| Symptoms | NPP 58(21.0) | 24(8.7) | 246(89.1) | 276 100) | |
| Headaches 43(19.8) | 21(27.3) | 69(69.7) | 217(100) | ||
| Migraine 51(21.9) | 15(25.0) | 61(72.6) | 233(100) | ||
| AS 36(16.7)* | 15(22.7) | 128(73.1)** | 216(100) | ||
| Total | 94(20.9) | 44(26.0) | 130(71.0) |
Discussion
Neuropathic symptoms, especially NPP and AS, are prevalent symptoms that affect 7-10% of the population worldwide. Together, they cause a significant loss of quality of life and a heavy burden on our societies. In parallel, headache is a common symptom frequently encountered in Neurology practice. Here, we report a positive correlation between NPP and AS and headaches.
The majority of patients were female (73.8%). Eighty percent of patients who had a skin biopsy for neuropathic symptoms had painful symptoms, with 60.3% of patients having NPP. This prevalence is higher than the reported range (25-50%) of NPP in diabetic neuropathy in the extremities. The discrepancy is likely because our NPP group included pain from large fiber neuropathy, such as radicular pain, centralized pain, and pain from non-neuronal sources, but presents with neuropathic features.
Our study did not detect a demographical perimeter correlating with the prevalence of NPP and AS. Similar to the current result, there is no definite association between age, gender, and BMI in developing NPP in diabetic neuropathy. These findings are distinct from the data from the general population that chronic neuropathic pain is more frequent in females and older (>50 years of age) and most commonly affects the lower back and lower limbs, neck, and upper limbs. This difference is likely because of the variable patient population and symptom characteristics.
Our results demonstrate a close connection between headaches, migraine, and NPP. The connection between headaches, especially daily persistent headaches, and neuropathic pain has been reported only in constant post-traumatic headaches. Although NPP types are not specified in our study because of the limitation of chart review, most of the patients (86%) had cervical radicular or myelopathic pain that affects the upper cervical segments (C1-C3) and/or cranial neuralgias. This trend was discussed in the literature by Ashina and colleagues, who characterized the prevalence of cervical neuropathic pain in patients with primary headaches. In addition, most of the secondary headaches from occipital neuralgia and cervicogenic headaches have neuropathic features. Our results suggest there are interactions between cervical neuropathic pain and headache, similar to well-established cervicogenic headaches from cervical musculoskeletal pathologies.
Headaches have been reported to be associated with dysautonomia. Unfortunately, autonomic symptoms are frequently underreported in current clinical practice. In a Polish study, hypotension was reported in 26.9% of migraine patients. However, it is unclear if the low blood pressure in migraine patients is related to autonomic dysfunction. In the pediatric population, the prevalence of dysautonomia peaks in parallel with migraine. In addition, cranial autonomic symptoms have strong connections with migraine and other headaches, especially trigeminal autonomic cephalalgias. Dysautonomia could be related to central brain mechanisms that increase parasympathetic activities. Recently, a strong correlation between dysautonomia and headache was reported after COVID-19 infection. The pathomechanisms of dysautonomia are complex and have been well-reviewed by Gazerani and Clairns. Our results are in agreement with the reports in the literature that AS is positively correlated with the prevalence of headaches in our study population. However, the same trend was not detected in migraine and AS. This discrepancy could be related to the limitation of the chart review process when the headaches were not well-characterized enough to be identified as migraine in the medical records.
Over the last three decades, skin punch biopsy has become the gold standard for diagnosing small fiber neuropathies with sensory and autonomic symptoms. In the current study, a positive skin biopsy was more likely to be detected in groups with female gender, > 50 years of age, and large (>25) BMI. These trends are consistent with the published data on skin biopsies in the general population. Our results suggest that a positive skin biopsy result only correlates with AS but not NPP, HA, or migraine. These findings indicate that headaches and migraine are more likely to occur in patients with probable and possible but not definite SFN as defined by the Diabetic Neuropathy Study Group of the European Association for the Study of Diabetes (NEURODIAB).
In parallel, our data did not support the association of positive AFT results, including the diagnosis of postural orthostatic tachycardia syndrome with headaches and migraine. However, our study is limited because only a fraction of patients (130 out of the 216 patients with AS) with AS had AFT. Dedicated research for AFT and headaches is necessary to determine if a diagnosis of autonomic disorders is related to the development of headaches in patients with dysautonomia.
Several possible mechanisms could link the somatosensory and autonomic systems with the trigeminovascular system that mediates headaches and migraine. First, because of the negative correlation between test results of peripheral neuropathy and symptoms of NPP, AS, headaches, and migraine, there could be interactions between NPP, headaches, and migraine in the central nervous system. Central sensitization is an essential concept for chronic NPP. In this phenomenon, the interactions between cervical and trigeminal neuropathic inputs with the cervical trigeminal vascular pathways, especially the connections between the descending trigeminal nucleus/tract with the C2 and C3 nociceptive pathways, could contribute to headaches and migraine in patients with cervical NPP. Second, microglial-mediated inflammation from NPP and AS could serve as a local link between trigeminovascular pathways with the nearby somatosensory and autonomic systems in the brain stem and thalamus. Third, pro-inflammatory cytokines and chemokines from NPP could initiate a remote activating effect to trigger the trigeminovascular pathways contributing to headaches and migraine.
Conclusion
In conclusion, our study demonstrated a positive correlation between NPP, AS, and headaches and migraine. However, these correlations are not established based on the positive results of SB, NCV/EMG, and AFT. These findings suggest that there could be central mechanisms in the brain and spinal cord that connect headaches and migraine with NPP and AS.
Conflicts of Interests
The authors have no conflicts of interest.
Funding Statement
The study is funded by The Ruth Freeman Fund for Pain – HMS NO 60-111-1102-2.
Acknowledgments
The authors thank Dr. Merit Cudkowicz for her leadership in the support of our study.
References
- Hovaguimian A, Gibbons CH. Diagnosis and treatment of pain in small-fiber neuropathy. Curr Pain Headache Rep. Jun 2011;15(3):193-200. doi:10.1007/s11916-011-0181-7
- Devigili G, Cazzato D, Lauria G. Clinical diagnosis and management of small fiber neuropathy: an update on best practice. Expert Rev Neurother. Sep 2020;20(9):967-980. doi:10.1080/14737175.2020.1794825
- Colloca L, Ludman T, Bouhassira D, et al. Neuropathic pain. Nat Rev Dis Primers. Feb 16 2017;3:17002. doi:10.1038/nrdp.2017.2
- Baron R, Binder A, Wasner G. Neuropathic pain: diagnosis, pathophysiological mechanisms, and treatment. Lancet Neurol. Aug 2010;9(8):807-19. doi:10.1016/S1474-4422(10)70143-5
- Goldberger JJ, Arora R, Buckley U, Shivkumar K. Autonomic Nervous System Dysfunction: JACC Focus Seminar. J Am Coll Cardiol. Mar 19 2019;73(10):1189-1206. doi:10.1016/j.jacc.2018.12.064
- Bakkers M, Faber CG, Hoeijmakers JG, Lauria G, Merkies IS. Small fibers, large impact: quality of life in small-fiber neuropathy. Muscle Nerve. Mar 2014;49(3):329-36. doi:10.1002/mus.23910
- Dydyk AM, Givler A. Central Pain Syndrome. StatPearls. 2023.
- Ducreux D, Attal N, Parker F, Bouhassira D. Mechanisms of central neuropathic pain: a combined psychophysical and fMRI study in syringomyelia. Brain. Apr 2006;129(Pt 4):963-76. doi:10.1093/brain/awl016
- Clauw DJ. Fibromyalgia: a clinical review. JAMA. Apr 16 2014;311(15):1547-55. doi:10.1001/jama.2014.3266
- Harper DE, Schrepf A, Clauw DJ. Pain Mechanisms and Centralized Pain in Temporomandibular Disorders. J Dent Res. Sep 2016;95(10):1102-8. doi:10.1177/0022034516657070
- Dydyk AM, Munakomi S. Thalamic Pain Syndrome. StatPearls. 2023.
- Farhad K, Oaklander AL. Fibromyalgia and small-fiber polyneuropathy: What’s in a name? Muscle Nerve. Nov 2018;58(5):611-613. doi:10.1002/mus.26179
- Tesfaye S, Boulton AJ, Dyck PJ, et al. Diabetic neuropathies: update on definitions, diagnostic criteria, estimation of severity, and treatments. Diabetes Care. Oct 2010;33(10):2285-93. doi:10.2337/dc10-1303
- Raasing LRM, Vogels OJM, Veltkamp M, van Swol CFP, Grutters JC. Current View of Diagnosing Small Fiber Neuropathy. J Neuromuscul Dis. 2021;8(2):185-207. doi:10.3233/JND-200490
- Iyengar S, Johnson KW, Ossipov MH, Aurora SK. CGRP and the Trigeminal System in Migraine. Headache. May 2019;59(5):659-681. doi:10.1111/head.13529
- Edvinsson JCA, Vigano A, Alekseeva A, et al. The fifth cranial nerve in headaches. J Headache Pain. Jun 5 2020;21(1):65. doi:10.1186/s10194-020-01134-1
- Messlinger K, Fischer MJ, Lennerz JK. Neuropeptide effects in the trigeminal system: pathophysiology and clinical relevance in migraine. Keio J Med. 2011;60(3):82-9. doi:10.2302/kjm.60.82
- Terrier LM, Fontaine D. Intracranial nociception. Rev Neurol (Paris). Sep 2021;177(7):765-772. doi:10.1016/j.neurol.2021.07.012
- Cohen SP, Mao J. Neuropathic pain: mechanisms and their clinical implications. BMJ. Feb 5 2014;348:f7656. doi:10.1136/bmj.f7656
- Wig R, Oakley CB. Dysautonomia and Headache in the Pediatric Population. Headache. Oct 2019;59(9):1582-1588. doi:10.1111/head.13659
- Haensch CA, Tosch M, Katona I, Weis J, Isenmann S. Small-fiber neuropathy with cardiac denervation in postural tachycardia syndrome. Muscle Nerve. Dec 2014;50(6):956-61. doi:10.1002/mus.24245
- Lacomis D. Small-fiber neuropathy. Muscle Nerve. Aug 2002;26(2):173-88. doi:10.1002/mus.10181
- Cruccu G, Sommer C, Anand P, et al. EFNS guidelines on neuropathic pain assessment: revised 2009. Eur J Neurol. Aug 2010;17(8):1010-8. doi:10.1111/j.1468-1331.2010.02969.x
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. Jan 2018;38(1):1-211. doi:10.1177/0333102417738202
- Goldstein DS, Cheshire WP, Jr. The autonomic medical history. Clin Auton Res. Aug 2017;27(4):223-233. doi:10.1007/s10286-017-0425-7
- Oaklander AL, Herzog ZD, Downs HM, Klein MM. Objective evidence that small-fiber polyneuropathy underlies some illnesses currently labeled as fibromyalgia. Pain. Nov 2013;154(11):2310-6. doi:10.1016/j.pain.2013.06.001
- Lauria G, Hsieh ST, Johansson O, et al. European Federation of Neurological Societies/Peripheral Nerve Society Guideline on the use of skin biopsy in the diagnosis of small fiber neuropathy. Report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society. Eur J Neurol. Jul 2010;17(7):903-12, e44-9. doi:10.1111/j.1468-1331.2010.03023.x
- McCorquodale D, Smith AG. Clinical electrophysiology of axonal polyneuropathies. Handb Clin Neurol. 2019;161:217-240. doi:10.1016/b978-0-444-64142-7.00051-5
- Freeman R, Chapleau MW. Testing the autonomic nervous system. Handb Clin Neurol. 2013;115:115-36. doi:10.1016/B978-0-444-52902-2.00007-2
- van Hecke O, Austin SK, Khan RA, Smith BH, Torrance N. Neuropathic pain in the general population: a systematic review of epidemiological studies. Pain. Apr 2014;155(4):654-662. doi:10.1016/j.pain.2013.11.013
- Blyth FM. Global burden of neuropathic pain. Pain. Mar 2018;159(3):614-617. doi:10.1097/j.pain.0000000000001127
- Themistocleous AC, Ramirez JD, Shillo PR, et al. The Pain in Neuropathy Study (PiNS): a cross-sectional observational study determining the somatosensory phenotype of painful and painless diabetic neuropathy. Pain. May 2016;157(5):1132-1145. doi:10.1097/j.pain.0000000000000491

