Herbal Therapies for CIRS in Pediatric Autism Disorders
Herbal Therapeutics for CIRS Biomarkers in Autism Spectrum Disorders: A Mechanistic and Molecular Approach for Neuroimmune Pathology in Pediatrics
Jodie A. Dashore PhD¹, OTD, RH (AHG), Brian Dashore DO², Scott McMahon MD³, Ritchie Shoemaker MD⁴
- Bionexus Health Clinic, Marlboro, NJ
- NYIT College of Osteopathic Medicine, Glen Head, NY
- Whole World Health Care, Roswell, NM
- ProgeneDX, Pocomoke, MD
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Dashore, JA., Dashore, B., et al., 2026. Herbal Therapeutics for CIRS Biomarkers in Autism Spectrum Disorders: A Mechanistic and Molecular Approach for Neuroimmune Pathology in Pediatrics. Medical Research Archives, [online] 14(1).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v14i1.7223
ISSN 2375-1924
Abstract
As the third article in a four-part series on pediatric neuroimmune disorders, this study evaluates evidence-based herbal therapeutics for biomarker modulation in Chronic Inflammatory Response Syndrome (CIRS). Building on Part 1 (CIRS diagnostic framework), Part 2 (infectious drivers such as Lyme disease and co-infections), and Part 4 (Autoimmune encephalopathy), Part 3 focuses on phytochemicals that target dysregulated markers, including TGF-β1, MMP-9, C4a, VEGF, VIP, and ADH. In a cohort of 1,722 children with treatment-resistant autism spectrum disorder (ASD) and Pediatric Acute-onset Neuropsychiatric Syndrome (PANS), all meeting CIRS criteria via bedside diagnosis, targeted herbal interventions were introduced, with reported improvements including but not limited to cognition, language, behavior, respiratory, sleep, pain, and gastrointestinal function. Mechanistic pathways included modulation of NF-κB, SMAD2/3, and Nrf2, supporting reduction of inflammation and restoration of neurovascular integrity. Bedside diagnostic strategies, necessitated by laboratory constraints, were supported by control data (83 children). Outcomes suggest that herbal therapeutics may serve as viable and valuable adjuncts to the Shoemaker CIRS Protocol, offering improved tolerability and accessibility. This work underscores a systems biology approach to pediatric CIRS management and highlights integrative strategies that bridge conventional and botanical medicine.
Keywords
- Chronic Inflammatory Response Syndrome
- Autism Spectrum Disorder
- Herbal Therapeutics
- Neuroimmune Pathology
- Pediatric Acute-onset Neuropsychiatric Syndrome
Introduction
Chronic Inflammatory Response Syndrome (CIRS) is a complex, environmentally acquired inflammatory illness marked by multisystem, multi-symptom dysregulation of immune, endocrine, vascular, and neurologic function. It arises from sustained exposure to biotoxins, particularly mycotoxins, endotoxins, Actinobacteria, and volatile organic compounds, found in water-damaged indoor environments. In genetically susceptible individuals with HLA-DR haplotypes impairing antigen presentation, these exposures provoke a failure to regulate the innate immune response, leading to a chronic, self-sustaining cascade of inflammation.
Central to the diagnosis and clinical management of CIRS is the quantification of molecular biomarkers that reflect pathophysiologic activity across immune signaling, neurovascular regulation, and endocrine axes. These include transforming growth factor beta-1 (TGF-β1), matrix metalloproteinase-9 (MMP-9), complement component 4a (C4a), vascular endothelial growth factor (VEGF), vasoactive intestinal peptide (VIP), antidiuretic hormone (ADH) with plasma osmolality, and melanocyte-stimulating hormone (MSH). Each of these markers has a defined mechanistic role in disease propagation, whether through fibrogenic signaling, endothelial permeability, redox instability, or neuroendocrine suppression, making them uniquely actionable targets for therapeutic modulation.
Although the Shoemaker Protocol remains the most structured and validated framework for CIRS management, its reliance on pharmaceutical agents, including cholestyramine or colesevelam (for toxin binding), EDTA spray (for MARCoNS eradication), and recombinant VIP (for neuropeptide replacement), presents limitations in tolerability, access, and mechanistic breadth. Furthermore, the protocol’s efficacy is highly contingent on strict environmental avoidance, which is not always feasible. These constraints underscore the need for adjunctive strategies that address underlying inflammatory and redox dynamics while remaining compatible with allopathic standards of care.
The lack of FDA-approved therapies for CIRS has also limited research funding and provider adoption, contributing to delayed diagnosis and inappropriate treatment. Standard pharmacologic interventions targeting symptoms (e.g., SSRIs, stimulants, antihistamines) often fail to resolve the root pathophysiology and may even exacerbate immune dysregulation. Moreover, few interventions outside the Shoemaker paradigm have been systematically evaluated for their capacity to modulate the key biomarkers that define this condition.
Phytochemicals, bioactive constituents of medicinal plants, offer a compelling adjunctive approach for targeting the inflammatory, fibrotic, and neuroendocrine abnormalities seen in CIRS. Many herbal compounds exert effects on canonical molecular pathways relevant to this syndrome, including nuclear factor kappa B (NF-κB), mitogen-activated protein kinase (MAPK), transforming growth factor-beta signaling (SMAD-dependent), and the nuclear factor erythroid 2–related factor 2 (Nrf2) antioxidant response element (ARE) axis. Unlike symptom-based interventions, phytotherapeutics can act upstream at the transcriptional and cytokine level, modulating immune cell polarization, reducing matrix degradation, and restoring vascular integrity.
Herbal agents such as curcumin, baicalin, epigallocatechin gallate (EGCG), and resveratrol have been shown to downregulate MMP-9 expression and inhibit TGF-β1–mediated fibrosis in animal and in vitro models. Others, including licorice, Astragalus membranaceus, and Rhodiola rosea, have demonstrated neuroendocrine modulation, antioxidant activity, and hypothalamic-pituitary-adrenal (HPA) axis stabilization, features highly relevant to VIP, ADH, and MSH regulation. The breadth of these phytochemical mechanisms suggests a capacity to influence not only symptoms but also the upstream biomarker architecture of CIRS itself.
In contrast to empiric botanical prescriptions, this article adopts a biomarker-driven approach, organizing phytotherapeutic interventions by their mechanistic relevance to specific Shoemaker labs. Section III offers detailed analyses of each biomarker, TGF-β1, MMP-9, C4a, VEGF, VIP, ADH/osmolality, and oxidative stress, as discrete pathophysiologic nodes. For each, we identify molecular targets (e.g., NF-κB, SMAD2/3, Nrf2), herbal compounds with preclinical and clinical evidence, and pharmacokinetic considerations such as bioavailability, metabolism, and tissue specificity. All referenced studies are drawn from the PubMed database and evaluated for methodological rigor, molecular specificity, and translational relevance.
This approach aligns with the principles of systems biology, which view chronic illness not as organ-specific failure but as a networked dysfunction of interconnected pathways. By mapping herbal effects onto discrete biochemical targets and immune cascades, the article seeks to bridge the divide between conventional laboratory diagnostics and phytochemical therapeutics. The therapeutic framework presented herein is not intended to supplant the Shoemaker Protocol but to supplement it through mechanistically grounded botanical options that may offer benefit in treatment-resistant or relapsing CIRS cases.
Given the skepticism often surrounding herbal medicine in allopathic contexts, this article emphasizes pharmacological plausibility and avoids generalized claims. Each herb discussed is contextualized within a molecular and clinical framework tied directly to CIRS biomarkers. Rather than promoting herbalism as an alternative model, we propose phytotherapeutics as rational, evidence-informed adjuncts capable of influencing the immune-metabolic architecture of CIRS. As the burden of environmentally acquired chronic illness rises, and the molecular tools for diagnosis evolve, it becomes increasingly critical to explore safe, mechanistically precise therapeutics that integrate with existing protocols and expand the armamentarium of clinicians.
Methods
As the third installment of a four-part retrospective review series this article extends the framework of Part 1 and 2, reviewing herbal therapeutics as an evidence based viable treatment option in a cohort of 1,722 children (aged 7 months–18 years) with treatment-resistant autism spectrum disorder (ASD) and Pediatric Acute-onset Neuropsychiatric Syndrome (PANS) seen at the Bionexus Health Clinic. Of the 1722 patients reviewed, 1326 (78%) were males and 374 were females (22%). All met Shoemaker Chronic Inflammatory Response Syndrome (CIRS) criteria, ≥6/13 multisystem symptoms, including fatigue, cognitive deficits, sensory/autonomic dysfunction, and joint pain.
PANS diagnoses followed published consensus guidelines and Shoemaker’s case definitions. Due to diagnosis-specific associated behavioral and neurological issues, children with ASD often show aversion to blood draws, often making this impossible to accomplish safely. Each child had a positive history of visible mold development and continuous exposure to the interior environment of a water-damaged building (WDB). The children represented fifty-three different nationalities. These comprised twenty-nine European countries, nine Asian countries, one Oceanic nation, three Middle Eastern countries, seven African countries, and four North American countries, including the United States. This includes children from 37 states in the United States. Their ages varied from seven months to 18 years with an average age of 4.5 years. In this group, there were 47 children with an additional diagnosis of Lyme disease as determined by reputable authorities in infectious disease and this analysis has been addressed in Part 2 in the series.
Control cases (n=83; 33 from McMahon et al, 50 from Shoemaker et al) lacked CIRS/PANS features, ensuring diagnostic specificity. Consecutive cases with refractory symptoms received herbal interventions targeting the physiological implications of CIRS biomarkers (TGF-β1, MMP-9, C4a, VEGF, VIP, ADH/osmolality). Interventions included Bionexus proprietary herbal formulations along with herbal extracts adapted from Buhner’s protocol. Dosing was oral, titrated for age and weight, and maintained at targeted doses for 6–18 months. The botanicals were recommended and administered under the close supervision of a registered herbalist, progress was monitored with lab work, and self-dosing is not recommended. Changes in symptoms were qualitatively tracked using simple rating scales completed by parents and clinicians. Clinical monitoring involved structured parental reports and clinician assessed at baseline, 6 months, 12 months and 18 months. Outcomes were documented with improvements reported by parents, teachers and therapists, in various areas of concern. This approach parallels the Lyme disease subset described in Article 2 (n=47), maintaining continuity with prior analyses of herbal modulation in neuroimmune disorders.
The herbals were carefully curated for each patient under the guidance of a Registered Herbalist with 15 years of experience in clinical herbalism and 25 years’ experience in pediatrics. Optimal timing for introduction of the herbal extracts, and step-by-step expert guidance were emphasized throughout. Premium quality ingredients, including biodynamically or organically grown herbals, carefully extracted using cGMP standards, third party tested, and FDA inspected have been used. Additionally, herbal usage safety has been addressed with particular care. Certain herbs can cause interactions when combined with certain medications. Children also process substances differently than adults which needs to be factored in. For this reason, dosing needs to be conservative and closely supervised. Some herbs act broadly and help the body manage stress across many systems. Others are more targeted and are most appropriate when certain markers are elevated. This layered approach reflects a systems view of chronic illness, where problems are connected rather than isolated. It is also stressed that herbal support should not interfere with key steps of the Shoemaker Protocol, such as toxin binding or nasal treatments, and should be spaced carefully. Within this cohort, the children received specific herbal support aimed at biological markers linked to CIRS. These included transforming growth factor beta-1 (TGF-β1), matrix metalloproteinase-9 (MMP-9), complement component 4a (C4a), vascular endothelial growth factor (VEGF), vasoactive intestinal peptide (VIP), and antidiuretic hormone (ADH). Together, these markers reflect inflammation, immune activity, barrier stability, blood flow, and hormone balance.
The herbal approach included compounds such as curcumin, Japanese knotweed, cat’s claw, and baicalin. Specific examples help clarify this. Curcumin and baicalin are described as helping lower excessive TGF-β1 activity and reducing MMP-9 levels. High MMP-9 can weaken the gut lining and the blood–brain barrier. When these barriers are compromised, inflammatory signals can spread more easily through the body and into the brain. Japanese knotweed is noted for its role in supporting VEGF balance, which helps maintain healthy blood vessels in the brain. Other botanicals influence VIP and ADH, hormones involved in sleep, mood, immune tolerance, and fluid balance. This helps explain why changes were seen across several areas of concern rather than in just one area of focus.
A central contribution of this paper lies in how it explains the biological pathways involved. Many plant compounds influence nuclear factor kappa B (NF-κB), which acts like a main switch for inflammation. Others affect SMAD2/3 signaling, which is closely tied to TGF-β1. When TGF-β1 stays too active, it can drive immune imbalance and tissue scarring. Antioxidant systems, such as nuclear factor erythroid 2–related factor 2 (Nrf2), help protect cells from ongoing stress. By working through these pathways, herbal compounds may help calm inflammation at a deeper level rather than only masking symptoms and thus contribute to clinical outcomes. Treatment for CIRS utilizing the established Shoemaker methods, along with alternative treatment utilizing herbal extracts was associated with notable improvements in cognition, motor skills, respiratory, nasal, ophthalmologic, dermatologic, nasal and gastrointestinal health, along with notable gains in speech and language. These findings suggest that CIRS may represent a critical missing component in these treatment-resistant populations, and that addressing all three conditions together may be considered best practice to achieve meaningful and sustained clinical progress.
Results
The authors retrospectively reviewed the charts of 1722 children from 53 countries, diagnosed with ASD, CIRS and PANS. The patients were all previously diagnosed by established specialists in their respective fields. It was noted that a subgroup of 47 children had also been diagnosed with Lyme Disease and co-infections. The control group consisted of data from two separate cohorts of 55 children from a 2009 IACFS study and from a 2018 PANS/PANDAS paper presented at a recent international CIRS conference.
Prior treatment for Autism, Lyme Disease and PANS consisted of recognized conventional therapies used unsuccessfully. These included specialized diets, nutritional supplements, hyperbaric oxygen therapy (HBOT), fecal microbiota transplant (FMT), antibiotics (47 cases), anti-fungal medications, occupational therapy, speech and language therapy, feeding therapy and applied behavior analysis therapy (ABA). Parents reported no significant improvements. As per parental reports, 39 of 47 children given antibiotics experienced gastrointestinal distress while on the prescription medications.
| Control Group | 2009 Cases % | 2018 Cases % | BNHC Group Cases % |
|---|---|---|---|
| No acute or chronic illness identified. | 0 | 0 | 0 |
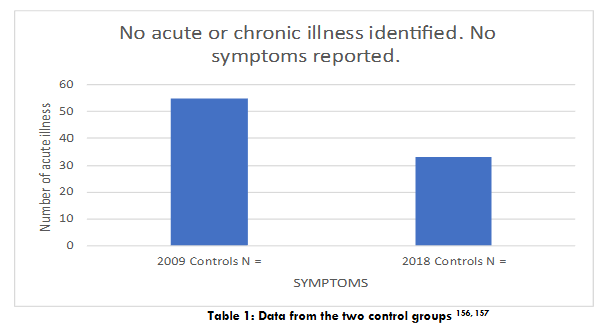
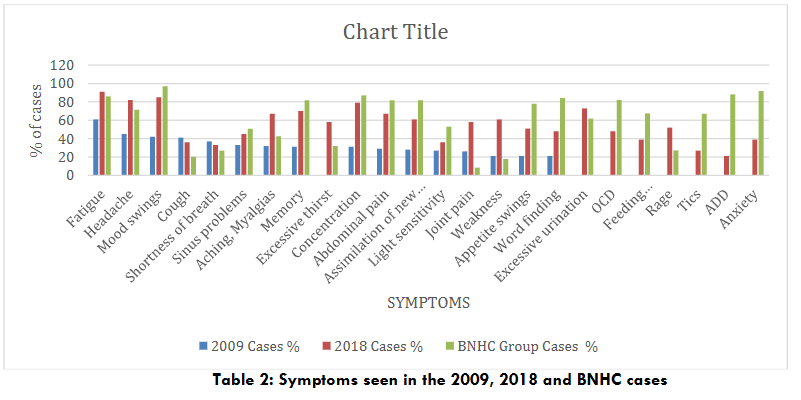
The data from the two studies used for the control group is presented in Table 1. The patients in the control group had no acute or chronic illness and no symptoms. The data for symptoms noted for the cases in the 2009 and 2018 CIRS studies, and BioNexus Health Autism cases is shown in Table 2. The post treatment data highlighting the percentages of improvements observed per symptom is shown in Table 3.
Looking back at the results from Part 1, and 2, the treatment for CIRS utilizing the established Shoemaker methods, including alternative herbal medicine options was associated with notable improvements in cognition, motor skills, respiratory, nasal, and gastrointestinal health, along with gains reported in speech and language. According to these results, treating the illnesses jointly may be the best way to attain the best therapeutic results, and CIRS may be a crucial missing element in these treatment-resistant cases. This information merits international attention given the rapidly increasing prevalence of ASD. It will help alleviate the suffering arising from neuroimmune inflammatory states that can be traumatic for the whole family as a unit, and hinder the functional development of children with ASD.
Discussion
Herbal therapeutics demonstrate significant potential as adjunctive treatments for Chronic Inflammatory Response Syndrome (CIRS) in pediatric autism spectrum disorder (ASD) and Pediatric Acute-onset Neuropsychiatric Syndrome (PANS), as evidenced in a 1,722-child cohort from Bionexus Health Clinic. In this cohort of children treated with phytochemicals (e.g., curcumin, Japanese knotweed, cat’s claw), clinical improvements were observed in the areas of cognition, communication, focus, learning, musculoskeletal recovery, developmental milestones, self-care, speech, respiratory, and gastrointestinal health over a period of 6–18 months. These outcomes align with Article 1’s CIRS framework and Article 2’s findings on Lyme/co-infection-driven neuroimmune dysregulation, suggesting shared inflammatory pathways.
At the molecular level, phytochemicals targeted key CIRS biomarkers, TGF-β1, MMP-9, C4a, VEGF, VIP, and ADH, by modulating nuclear factor kappa B (NF-κB), SMAD2/3, and nuclear factor erythroid 2–related factor 2 (Nrf2) pathways. Curcumin and baicalin reduced TGF-β1–mediated fibrosis and MMP-9–driven matrix degradation, while Japanese knotweed has been shown to balance VEGF which helps promote blood–brain barrier integrity. These mechanisms counter the neuroinflammation and oxidative stress central to CIRS, as detailed in this article’s molecular analyses.
A group of 1722 children with ASD and CIRS, previously diagnosed by recognized specialists in CIRS and international experts in autism as per established case definition, were sent to Bionexus specialized clinic for the implementation of CIRS treatment protocols and evaluation of the clinical outcomes. The children were previously classified by their respective autism specialists as resistant to treatment due to no sustained improvements observed after undergoing treatment according to current established protocols for a minimum period of 6 months. As this is a worldwide study, in countries where requisite specialized laboratory tests are often unavailable, the local authorities relied on bedside clinical diagnosis. Children with ASD frequently demonstrate difficulty cooperating with blood draws due to severe behavioral and neurological challenges. All had a positive history of chronic ongoing exposure to the interior environment of a water damaged building (WDB) and visually observed mold growth. This was supported by control data (83 children: 33 from McMahon, 50 from Shoemaker), ensuring diagnostic reliability.
Compared with the Shoemaker Protocol’s reliance on pharmaceuticals (e.g., cholestyramine), herbal interventions may offer advantages in tolerability and accessibility. However, the lack of randomized controlled trials and regional variability in herbal bioavailability may limit causal attribution and generalizability. This study extends Article 1’s systems biology approach and Article 2’s focus on infectious triggers, positioning phytochemicals as potential integrative tools for pediatric CIRS management. By addressing upstream inflammatory cascades and neuroendocrine dysregulation, these interventions may provide a complementary adjunct to conventional therapies. Future research should prioritize multi-omic profiling (e.g., transcriptomics, proteomics) to validate biomarker modulation and longitudinal clinical trials to establish efficacy. As the finale of this four-part series, this work underscores the need for multidisciplinary collaboration to integrate phytotherapeutics into evidence-based care, with the goal of improving developmental outcomes in pediatric neuroimmune disorders.
Functional Overview of CIRS Biomarker Pathways and Molecular Targets of Botanical Extracts
CIRS is best conceptualized as a dynamic network disorder, wherein environmental biotoxins interact with host genetic susceptibility to dysregulate immune signaling, endocrine control, and vascular function. Central to its diagnosis and therapeutic monitoring is the quantification of molecular biomarkers that capture mechanistic disruptions across systems. Rather than isolated markers of inflammation, these biomarkers derived from the Shoemaker Protocol, reflect the convergent biology of tissue injury, redox imbalance, and neuroimmune disintegration. Understanding each within its molecular and clinical context provides a blueprint for therapeutic intervention and biomarker-guided precision care.
Therapeutic Leverage Points Across Biomarker Pathways
Given the interconnectivity of CIRS biomarkers, effective modulation requires targeting nodal regulators rather than isolated markers. The nuclear factor kappa B (NF-κB) pathway serves as a primary pro-inflammatory transcriptional driver and upstream controller of MMP-9, IL-6, TNF-α, and other cytokines. Conversely, nuclear factor erythroid 2–related factor 2 (Nrf2) serves as a master antioxidant switch, upregulating glutathione synthesis, detoxification enzymes, and mitochondrial defense programs.
Other strategic targets include SMAD-dependent TGF-β1 signaling (for fibrotic inhibition), CREB (for neuropeptide regulation), and Th17/Treg balance (for immune recalibration). Maintaining BBB and mucosal barrier integrity through modulation of matrix remodeling and tight junction preservation is also critical. Phytochemicals capable of modulating these axes, through transcriptional, enzymatic, or epigenetic effects, form the basis of rational biomarker-guided herbal therapeutics.
Matrix Metalloproteinase-9 (MMP-9): Gateway to Inflammatory Permeability
MMP-9 is a zinc-dependent proteolytic enzyme primarily secreted by neutrophils, macrophages, and other immune cells during inflammatory responses. Its core function lies in the degradation of extracellular matrix (ECM) proteins, especially type IV collagen in the basement membrane. In the setting of CIRS, sustained MMP-9 elevation drives endothelial permeability, immune cell infiltration, and the release of sequestered inflammatory mediators such as TNF-α and IL-1β, amplifying systemic immune activation. In CIRS patients, elevated serum MMP-9 levels correlate with symptom intensity and barrier tissue vulnerability, particularly in the gut-brain axis and vascular endothelium.
Therapeutically, MMP-9 represents a targetable node via transcriptional inhibition (e.g., suppression of NF-κB and AP-1 pathways), antioxidant support, and stabilization of matrix architecture.
Botanical Modulation of MMP-9 Pathways
Targeting MMP-9 transcription and activity in CIRS requires intervention at the molecular signaling level—primarily through inhibition of NF-κB, attenuation of MAPK cascades, and neutralization of oxidative stress. Several botanicals exhibit multimodal effects across these axes. Epigallocatechin gallate (EGCG), curcumin, and baicalin inhibit NF-κB translocation and suppress MMP-9 gene expression in vitro and in vivo. Resveratrol and rosemary modulate MAPK-dependent upregulation of MMP-9 while restoring redox homeostasis. The following section evaluates these agents’ mechanistic relevance, experimental support, and formulation considerations in the context of biotoxin-induced matrix instability.
Curcumin (Curcuma longa)
Curcumin, the principal curcuminoid of Curcuma longa, has demonstrated potent anti-inflammatory effects across a wide range of preclinical models. A primary mechanism of curcumin’s modulation of MMP-9 involves inhibition of NF-κB and AP-1 activation, leading to suppressed transcription of MMP-9 mRNA in monocytes, endothelial cells, and cancer models. In vascular inflammation models, curcumin has been shown to reduce MMP-9 secretion by attenuating ROS production and stabilizing ECM architecture. In a rat model of cerebral ischemia-reperfusion injury, curcumin administration led to downregulation of MMP-9 expression and preservation of blood–brain barrier integrity. Pharmacokinetically, curcumin’s limited oral bioavailability due to poor solubility and extensive hepatic metabolism has prompted the development of enhanced formulations, including liposomal, nanoparticle, and piperine-adjuvanted forms, all of which demonstrate greater systemic absorption and target tissue delivery.
Baicalin (Scutellaria baicalensis)
Baicalin, a flavone glycoside derived from Scutellaria baicalensis, exerts broad anti-inflammatory and antioxidant effects via downregulation of TLR4/NF-κB signaling and upregulation of Nrf2-mediated antioxidant pathways. In macrophage and microglial activation models, baicalin significantly reduces the production of pro-inflammatory cytokines and MMP-9 expression through suppression of p65 nuclear translocation and modulation of JNK and ERK phosphorylation. In cerebral ischemia and neuroinflammation models, baicalin has been shown to inhibit MMP-9 activity and protect the integrity of the blood–brain barrier, thereby limiting leukocyte infiltration and edema. Notably, baicalin has demonstrated synergistic effects with curcumin in reducing MMP-9 levels and oxidative stress in endotoxin-induced lung injury, suggesting potential for botanical combination strategies. Baicalin is primarily absorbed as baicalein following hydrolysis by intestinal microflora, and its oral bioavailability is moderate but enhanced through micronized and phospholipid-bound delivery forms.
Epigallocatechin-3-gallate (EGCG, from Green Tea)
EGCG, the most bioactive catechin in Camellia sinensis, has been extensively studied for its capacity to inhibit MMP-9 across multiple models of inflammation, cancer, and cardiovascular dysfunction. EGCG modulates gene transcription through inhibition of NF-κB, AP-1, and STAT3, all of which are key inducers of MMP-9 expression under pro-inflammatory conditions. Additionally, EGCG scavenges ROS and downregulates MAPK signaling, thereby interrupting oxidative stress–mediated activation of MMP-9. In endothelial cells exposed to TNF-α, EGCG reduces MMP-9 secretion and restores tight junction protein expression, suggesting potential relevance to blood–brain barrier stabilization in CIRS. In mouse models of lung injury and sepsis, EGCG administration significantly decreased MMP-9 levels in bronchoalveolar fluid and plasma, correlating with improved survival and reduced neutrophilic infiltration. Oral bioavailability of EGCG is variable and affected by pH, metabolism, and binding to plasma proteins, though green tea extract formulations have improved stability and absorption.
Resveratrol (from Polygonum/Fallopia spp.)
Resveratrol, a stilbene polyphenol found in Polygonum cuspidatum and Vitis vinifera, exhibits strong anti-inflammatory and anti-MMP-9 activity through several converging pathways. It activates SIRT1, which inhibits NF-κB transcriptional activity and prevents induction of MMP-9 gene expression in macrophages and endothelial cells. Resveratrol also attenuates MAPK and PI3K/Akt signaling, which modulate MMP-9 secretion in response to oxidative and cytokine stress. In vascular inflammation models, resveratrol reduces MMP-9 release and preserves ECM integrity by downregulating TNF-α and IL-1β–induced matrix degradation. Additionally, resveratrol enhances endothelial nitric oxide synthase (eNOS) activity, improving vascular tone and limiting MMP-9–mediated remodeling. Despite its low oral bioavailability, resveratrol has high cellular uptake and is rapidly metabolized to active glucuronide and sulfate conjugates, which retain partial bioactivity.
Gotu Kola (Centella asiatica)
Centella asiatica is a triterpenoid-rich herb traditionally used for wound healing and neurovascular support. Its primary bioactive compounds, asiaticoside, madecassoside, and asiatic acid, have been shown to modulate MMP-9 levels in tissue remodeling and inflammation models. Gotu kola reduces MMP-9 expression by attenuating NF-κB and MAPK activity and stabilizing ECM architecture, particularly in models of vascular injury and diabetic wound healing. In brain endothelial cells, Centella extracts have demonstrated protective effects against cytokine-induced barrier disruption and reduced MMP-9–mediated permeability, suggesting relevance to blood–brain barrier preservation in CIRS. Although human pharmacokinetic data is limited, bioavailability of triterpenes can be enhanced using ethanolic or lipophilic extracts standardized for asiaticoside content.
Rosemary (Rosmarinus officinalis)
Rosemary contains several phenolic diterpenes, most notably rosmarinic acid and carnosic acid, which suppress MMP-9 expression by inhibiting NF-κB and oxidative stress signaling. In LPS-stimulated macrophages, rosmarinic acid reduces MMP-9 mRNA levels and protein activity while concurrently decreasing ROS production and pro-inflammatory cytokines. In cancer and arthritis models, rosemary extract administration has been associated with lowered MMP-9 levels and reduced tissue invasion. In neuroinflammation models, rosemary constituents have shown potential to modulate glial cell activation and blood–brain barrier permeability, although MMP-9–specific studies in CNS models remain limited. Rosemary extract bioavailability varies with formulation; concentrated ethanolic extracts rich in carnosic acid show enhanced CNS penetration and systemic absorption.
Transforming Growth Factor Beta-1 (TGF-β1): Fibrogenic axis, Immune Skewing, and Barrier Injury
TGF-β1 is a multifunctional cytokine implicated in immune tolerance, tissue remodeling, and fibrogenesis. Importantly, TGF-β1 operates as both a downstream effector of biotoxin-induced signaling and an upstream amplifier of immune and fibrotic pathology. In CIRS, persistently elevated TGF-β1 promotes fibrotic remodeling of airways, gastrointestinal mucosa, and the central nervous system. It skews immune balance away from Tregs toward Th17 polarization, reducing immune regulation while enhancing auto-reactivity and mucosal vulnerability. High TGF-β1 levels have also been associated with increased expression of adhesion molecules on endothelial cells, facilitating leukocyte extravasation and microvascular inflammation. It interacts with oxidative stress, mitochondrial dysfunction, and hypoxia-inducible factor 1-alpha (HIF-1α) signaling, situating it at the intersection of inflammatory, metabolic, and structural dysfunction in CIRS.
As a pleiotropic cytokine that orchestrates a broad range of physiological and pathological processes, including immune homeostasis, extracellular matrix remodeling, epithelial barrier integrity, and fibrosis, TGF-β1 assumes a central mechanistic role by perpetuating immune dysregulation and promoting fibrotic and epithelial injury across multiple organ systems. Unlike other pro-inflammatory cytokines that primarily mediate acute responses, TGF-β1 drives chronicity by sustaining maladaptive remodeling and altering the immunological set point toward tolerance breakdown and tissue stiffness. Its signaling is transduced via the SMAD2/3 pathway, ultimately regulating gene expression involved in epithelial-to-mesenchymal transition (EMT), extracellular matrix production, and T regulatory (Treg) cell function.
In patients with CIRS, persistently elevated TGF-β1 represents a hallmark of unresolved immune activation and failed resolution signaling. Plasma levels of TGF-β1 have been found to be significantly increased in individuals exposed to biotoxins from mold, cyanobacteria, or tickborne illness, with higher concentrations correlating with greater disease severity, fatigue, and cognitive dysfunction. Chronic overexpression of TGF-β1 disrupts immune equilibrium by impairing the function and stability of regulatory T cells (Tregs) while facilitating the expansion of profibrotic and inflammatory pathways. TGF-β1 also cooperates with cytokines, including IL-6, to skew differentiation toward Th17 cells, which have been implicated in autoimmune-like inflammation and blood–brain barrier dysfunction in CIRS and other neuroimmune conditions.
One of the most damaging consequences of TGF-β1 overactivation is the induction of EMT, a process by which epithelial cells lose their polarity and tight junction proteins (e.g., occludin, claudin-1, and ZO-1) and acquire mesenchymal characteristics such as increased motility, vimentin expression, and collagen synthesis. This transdifferentiation not only compromises epithelial and endothelial barriers in organs like the gut, lungs, and brain but also contributes to the accumulation of fibroblasts and myofibroblasts responsible for ECM deposition and tissue stiffness. In the gastrointestinal tract, such changes translate into increased intestinal permeability (“leaky gut”), mucosal immune activation, and microbial translocation, events that further fuel systemic inflammation and perpetuate CIRS pathology.
TGF-β1’s pro-fibrotic effects are equally evident in the lungs, liver, kidneys, and brain microvasculature, where it promotes type I and III collagen deposition, elastin crosslinking, and the accumulation of α-smooth muscle actin (α-SMA)-positive myofibroblasts. These changes are irreversible unless upstream TGF-β1 signaling is modulated and ECM degradation mechanisms (e.g., MMP activity) are restored. TGF-β1 also disrupts the balance between MMPs and their tissue inhibitors (TIMPs), further skewing the tissue remodeling milieu toward pathological fibrosis.
In addition to its fibrotic actions, TGF-β1 exerts direct effects on mitochondrial metabolism and redox balance. It has been shown to impair mitochondrial oxidative phosphorylation, reduce mitochondrial membrane potential, and increase ROS generation, thereby exacerbating oxidative stress and bioenergetic dysfunction, processes commonly observed in the mitochondrial phenotype of CIRS. This intersection between TGF-β1 signaling, redox imbalance, and immune dysregulation underscores the multifaceted role of the cytokine in sustaining the systemic, multi-organ pathology characteristic of CIRS.
Given this vital role, therapeutic strategies targeting TGF-β1 and its downstream effectors are of high clinical relevance. Ideally, such interventions would attenuate SMAD-mediated transcription, reduce ECM synthesis, promote re-epithelialization, and rebalance Treg/Th17 dynamics. Botanical agents that modulate TGF-β1 signaling through diverse mechanisms, including SMAD inhibition, ROS scavenging, EMT blockade, and restoration of epithelial barrier proteins, represent a promising adjunct to conventional Shoemaker protocol interventions. The next section examines five such botanicals, berberine, ashwagandha (Withania somnifera), Scutellaria baicalensis, Salvia miltiorrhiza, and additional Withania withanolides, in the context of mechanistic inhibition of the TGF-β1 axis.
Berberine (from Berberis species)
Berberine is a plant-derived isoquinoline alkaloid found in Berberis vulgaris, Coptis chinensis, and Hydrastis canadensis, with extensive preclinical evidence supporting its antifibrotic and immunomodulatory roles. In hepatic stellate cells, berberine suppresses TGF-β1-induced SMAD2/3 phosphorylation and inhibits transcription of pro-fibrotic genes such as COL1A1, α-SMA, and CTGF. This effect is mediated through upregulation of SMAD7, an inhibitory SMAD protein that blocks receptor-mediated activation of SMAD2/3 complexes.
In pulmonary fibrosis models, berberine has been shown to reduce collagen accumulation and EMT markers while restoring E-cadherin expression and reducing vimentin and fibronectin levels. Berberine also downregulates TGF-β1 production in activated macrophages and attenuates oxidative stress via Nrf2 activation, thereby reducing upstream triggers of the fibrotic cascade. In gut models, berberine preserves epithelial barrier integrity by upregulating tight junction proteins and reducing mucosal inflammation driven by TGF-β1. Its oral bioavailability is poor due to P-glycoprotein efflux and first-pass metabolism, though nanoparticle and phospholipid formulations improve systemic absorption.
Ashwagandha (Withania somnifera)
Ashwagandha, a traditional adaptogenic herb in Ayurvedic medicine, exhibits TGF-β1–modulating effects via its withanolide constituents. Withaferin A, the most studied bioactive, downregulates TGF-β1-induced fibrotic markers in lung, liver, and kidney models through inhibition of SMAD2/3 phosphorylation and suppression of EMT. In bleomycin-induced pulmonary fibrosis, Withaferin A treatment reduced collagen deposition, α-SMA expression, and inflammatory cytokine levels, indicating attenuation of both fibrotic and immune pathways.
Ashwagandha also modulates immune skewing by enhancing FoxP3+ Treg populations while inhibiting pro-inflammatory Th17 differentiation, in part through reduction of TGF-β1/IL-6 synergy that promotes pathogenic Th17 cell development. These dual immunomodulatory and antifibrotic effects may support restoration of immune tolerance in CIRS. Oral bioavailability of withanolides is moderate and can be increased through lipid-based delivery systems or standardized extracts enriched for Withaferin A.
Scutellaria baicalensis (Chinese Skullcap)
Scutellaria baicalensis, a traditional Chinese botanical, contains flavonoids such as baicalin and baicalein with strong anti-inflammatory and antifibrotic properties. In TGF-β1-induced fibroblast activation models, baicalin inhibits SMAD3 nuclear translocation and suppresses transcription of COL1A1 and ACTA2 (α-SMA), thereby preventing ECM remodeling and myofibroblast conversion. In renal and hepatic fibrosis models, baicalin also attenuates EMT and restores epithelial phenotype markers, suggesting interruption of TGF-β1–driven transdifferentiation.
Scutellaria flavonoids additionally enhance Nrf2-mediated antioxidant responses, which indirectly downregulate TGF-β1 signaling by reducing ROS-mediated SMAD activation. In models of inflammatory bowel disease, baicalin restores tight junction expression and reduces mucosal permeability, consistent with modulation of barrier injury driven by cytokine-induced EMT. Oral absorption of baicalin depends on microbial hydrolysis to baicalein, which has greater lipophilicity and bioavailability. Phytosomal formulations improve systemic levels and tissue delivery.
Salvia miltiorrhiza (Danshen)
Salvia miltiorrhiza is a polyphenol-rich botanical widely studied in fibrosis and cardiovascular disease. Its main active constituents, tanshinone IIA and salvianolic acid B, exert potent antifibrotic effects by modulating TGF-β1/SMAD signaling. In liver fibrosis models, tanshinone IIA downregulates SMAD2/3 activation, suppresses collagen and α-SMA expression, and induces SMAD7 upregulation, thereby inhibiting profibrotic transcriptional activity. Salvianolic acid B blocks TGF-β1-induced EMT by preserving E-cadherin expression and inhibiting transcription factors such as Snail and Twist.
Salvia compounds also reduce TGF-β1 production in activated macrophages and fibroblasts by inhibiting upstream signaling via ERK and p38 MAPK pathways. In colitis and airway remodeling models, Salvia extracts improve mucosal barrier integrity, reduce epithelial apoptosis, and normalize ECM turnover. Oral bioavailability of tanshinones is low due to rapid metabolism and poor aqueous solubility; however, micelle and liposomal formulations have shown promise in enhancing bioactivity.
Withania somnifera (differentiated phytoconstituent profile)
While ashwagandha and Withaferin A have been addressed above, additional withanolides such as withanoside IV and sitoindosides exhibit independent antifibrotic activities, often through different cellular targets. In cardiac fibroblasts, withanoside IV inhibits TGF-β1-induced myofibroblast differentiation and ECM accumulation via PI3K/Akt-dependent blockade of SMAD nuclear translocation. Other studies suggest that less-characterized withanolides may reduce oxidative stress–driven TGF-β1 overexpression by enhancing mitochondrial function and reducing NADPH oxidase activation.
Beyond direct antifibrotic effects, Withania extracts stabilize epithelial barriers in gut and lung models, reducing permeability and cytokine leakage, in part by modulating junctional protein expression. Bioavailability of whole-root extracts depends on withanolide concentration and formulation strategy, with emulsified and liposomal forms showing improved absorption and CNS penetration.
Complement Component 4a (C4a): Initiator of Innate Immune Cascade and Oxidative Amplification and Redox Damage
The complement system represents a fundamental component of the innate immune response, orchestrating host defense through a finely tuned cascade of serine protease activations. These sequential cleavages produce effector molecules such as C3a, C4a, and C5a, collectively known as anaphylatoxins, that modulate inflammation, chemotaxis, vascular permeability, and cellular oxidative responses. C4a is a cleavage product of the classical complement cascade, formed during the proteolytic activation of C4 by C1 esterase in response to immune complex or pathogen recognition. In CIRS, excessive complement activation, particularly of the C4a and C3a fragments, signifies persistent innate immune stimulation triggered by biotoxin residues or unresolved infection.
The clinical consequence of elevated C4a includes post-exertional fatigue, migratory pain, and neurocognitive dysfunction, symptoms that often mirror mitochondrial dysfunction and autonomic dysregulation. CIRS is marked by persistent innate immune activation, often triggered by biotoxin exposure (e.g., mold, cyanobacteria, Lyme-associated microbes) in genetically susceptible individuals. Elevated plasma levels of C4a serve as a surrogate biomarker for uncontrolled complement activation, correlating with symptom exacerbation, endothelial inflammation, and tissue-level hypoperfusion.
In this context, C4a functions not merely as a byproduct of immune activation but as a driver of sustained oxidative injury and microvascular compromise. C4a is generated during activation of the classical and lectin complement pathways, each of which converges on the proteolytic cleavage of C4 and subsequent formation of the C3 convertase (C4b2a). Elevated C4a drives downstream membrane attack complex (MAC) formation, membrane lysis, and secondary tissue injury. Though historically considered weak compared to C3a and C5a, C4a exerts direct biological effects including endothelial cell activation, increased vascular permeability, and mast cell–independent smooth muscle contraction.
C4a also induces the generation of reactive oxygen species (ROS) via NADPH oxidase, contributing to oxidative stress, mitochondrial dysfunction, and downstream redox-sensitive signaling cascades such as NF-κB and AP-1. Downstream events further amplify inflammation. The cleavage of C3 and C5 yields potent anaphylatoxins C3a and C5a, which bind their respective receptors (C3aR, C5aR1/CD88) on neutrophils, macrophages, endothelial cells, and glia. These interactions trigger calcium influx, ROS production, and release of pro-inflammatory cytokines including tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), and interleukin-6 (IL-6), thereby propagating a feed-forward loop of inflammation and tissue damage. Formation of the terminal complement complex (C5b-9 or MAC) further contributes to mitochondrial membrane permeabilization, epithelial injury, and blood–brain barrier (BBB) compromise, pathologies frequently observed in CIRS patients with neurologic symptoms.
Persistent elevation of C4a in CIRS is thought to result from several converging mechanisms: chronic antigenic stimulation by microbial fragments, failure of complement regulatory proteins (e.g., CD55/DAF, CD59), and inadequate resolution of biofilm-induced immune priming. Mycotoxins and bacterial endotoxins may function as persistent pattern recognition receptor (PRR) ligands, skewing complement homeostasis and maintaining low-grade inflammatory activation. Moreover, breakdown of epithelial and endothelial barriers allows microbial translocation from mucosal sites, especially the gut and sinuses, fueling systemic complement and cytokine cascades.
Clinically, elevated C4a levels are associated with fatigue, pain flares, dizziness, cognitive dysfunction, and sensitivity to re-exposure, a reflection of complement-mediated microcirculatory impairment and redox imbalance. These phenomena are further compounded by mitochondrial damage, hypoxia-inducible factor (HIF) activation, and metabolic inflexibility in affected tissues. Several botanicals demonstrate direct or indirect effects on the complement cascade and associated oxidative stress. These include classical inhibitors of C3/C5 convertases, scavengers of ROS, and modulators of downstream cytokine production. The following agents, licorice (Glycyrrhiza glabra), Artemisia annua, green tea (Camellia sinensis), and Boswellia serrata, offer mechanistic relevance for attenuating C4a-driven pathology.
Licorice (Glycyrrhiza glabra)
Licorice root contains the triterpenoid glycyrrhizin and its aglycone metabolite glycyrrhetinic acid, both of which exhibit immunomodulatory, antiviral, and antioxidant properties. Glycyrrhizin has been shown to inhibit complement activation by directly interfering with C2 cleavage and downstream C3 convertase formation, thereby reducing C4a generation. Additionally, glycyrrhizin inhibits the formation of reactive oxygen species and lipid peroxidation products in neutrophil cultures and ischemia models.
In murine hepatitis and endotoxemia models, glycyrrhizin administration reduces C3a/C5a production and TNF-α levels, suggesting systemic complement attenuation and cytokine control. The compound also preserves endothelial tight junctions and mitochondrial integrity, mitigating complement-driven vascular injury. Safety considerations remain essential: glycyrrhizin mimics mineralocorticoid activity via inhibition of 11β-hydroxysteroid dehydrogenase type 2, potentially causing hypertension, hypokalemia, and sodium retention with chronic use. Deglycyrrhizinated licorice (DGL) formulations minimize these effects but may lack full complement-inhibitory efficacy. Standardized glycyrrhizin-containing extracts, used short-term and under electrolyte monitoring, may offer safer profiles.
Artemisia annua (Sweet Wormwood)
Artemisia annua, the source of artemisinin and its derivatives, is well-known for its antiparasitic and antipyretic properties. Beyond antimalarial applications, artemisinin demonstrates significant immunoregulatory and complement-modulating effects. In animal models of immune complex nephritis, artemisinin reduces C3 and C4 deposition in glomeruli and decreases circulating C3a and C5a levels, suggesting systemic complement suppression. Mechanistically, artemisinin downregulates NF-κB activation and inhibits iNOS-mediated ROS generation, thereby blunting the inflammatory amplification loop initiated by C3a/C5a.
In LPS-stimulated macrophages, artemisinin analogs suppress the expression of pro-inflammatory cytokines (IL-6, TNF-α) and complement proteins, reinforcing their role in controlling innate immune overactivation. Although artemisinin has limited water solubility and short half-life, its lipophilic analogs (e.g., dihydroartemisinin, artesunate) offer improved bioavailability and longer systemic activity. These derivatives have been used in clinical settings for autoimmune disorders and are generally well-tolerated.
Green Tea (Camellia sinensis)
Epigallocatechin-3-gallate (EGCG), the primary catechin in green tea, has been shown to inhibit complement-mediated cytolysis and C3/C5 convertase activity. In erythrocyte lysis assays, EGCG reduced complement-dependent hemolysis in a dose-dependent fashion, implicating terminal pathway inhibition. Additionally, EGCG modulates neutrophil activation and ROS production through suppression of NADPH oxidase and restoration of mitochondrial membrane potential. In neuroinflammation and sepsis models, EGCG administration attenuates oxidative damage, preserves blood–brain barrier function, and downregulates complement-related gene expression. Its dual role as a complement modulator and antioxidant positions EGCG as a potential adjunct in CIRS-related redox and immune dysregulation.
Despite favorable safety profiles, EGCG bioavailability is limited by first-pass metabolism and gastrointestinal degradation. Enteric-coated or liposomal green tea extracts may improve systemic delivery and tissue penetration.
Boswellia serrata (Indian Frankincense)
Boswellia resin contains boswellic acids, particularly acetyl-11-keto-β-boswellic acid (AKBA), which exhibit potent anti-inflammatory and antioxidant properties. AKBA has been reported to downregulate complement component expression, including C3, in inflamed tissues and cultured macrophages. In collagen-induced arthritis models, Boswellia treatment reduced serum C3a levels and joint inflammation, indicating systemic complement suppression. AKBA also scavenges superoxide and inhibits 5-lipoxygenase (5-LOX), thereby reducing ROS generation and lipid peroxidation. In endothelial models, Boswellia extracts preserve barrier integrity and limit cytokine-induced hyperpermeability, which may have relevance for microvascular complications in CIRS.
Boswellia is generally well-tolerated with minimal side effects. Bioavailability remains a limitation, as boswellic acids exhibit poor intestinal absorption. Novel formulations including lecithin-bound or nano-emulsified extracts enhance systemic levels and clinical efficacy.
Vascular Endothelial Growth Factor (VEGF): Oxygenation, Microcirculation, Neurovascular Coupling and Angiogenic Restoration
Vascular endothelial growth factor (VEGF) is a key, heparin binding, angiogenic cytokine with broad physiological influence over vascular integrity, oxygen transport, endothelial cell survival, and neurovascular coupling. Initially identified for its role in endothelial mitogenesis, VEGF is now recognized as a central modulator of the cerebral and systemic microcirculation, acting to balance tissue perfusion with metabolic demands during stress, hypoxia, and inflammation. In the context of CIRS, a paradoxical suppression of VEGF is observed despite ongoing inflammation and tissue injury. Reduced VEGF levels have been correlated with fatigue, cognitive impairment, microvascular ischemia, and mitochondrial dysfunction in affected individuals.
VEGF serves not only as a biomarker of impaired perfusion but also as a therapeutic target for restoring endothelial function and neurovascular resilience. VEGF operates primarily through VEGF receptor-2 (VEGFR-2/KDR/Flk-1), a tyrosine kinase receptor expressed predominantly on vascular endothelial cells. Upon ligand binding, VEGFR-2 undergoes dimerization and autophosphorylation, initiating downstream signaling cascades that include the phosphoinositide 3-kinase (PI3K)/Akt and the mitogen-activated protein kinase (MAPK)/ERK pathways. These pathways converge on endothelial nitric oxide synthase (eNOS), promoting nitric oxide (NO) production, vasodilation, and barrier stabilization. In parallel, VEGF signaling is tightly coupled to hypoxia-inducible factor 1-alpha (HIF-1α), which regulates VEGF transcription in response to oxygen deprivation. This feedback loop ensures that angiogenesis is induced under conditions of hypoxia or oxidative stress.
In CIRS, however, this homeostatic feedback is disrupted. Patients often exhibit low circulating VEGF in the face of significant tissue inflammation and endothelial injury. This anomaly may reflect a state of endothelial exhaustion, oxidative suppression of VEGF transcription, or impaired HIF-1α signaling due to chronic exposure to biotoxins, mitochondrial dysfunction, or environmental hypoxia. Moreover, cytokines such as TNF-α and IL-1β, which are elevated in CIRS, can downregulate VEGF expression in some contexts, further impairing angiogenic repair mechanisms. The consequence is microvascular dropout, reduced oxygen delivery, and increased diffusion distances for nutrients and signaling molecules, particularly in vulnerable tissues such as the brain, muscle, and gastrointestinal tract.
In CIRS patients, paradoxical suppression of VEGF is frequently observed despite the presence of hypoxic stress. This deficiency results in impaired microvascular flow, tissue hypoxia, and neurovascular uncoupling. Such deficits contribute to fatigue, exertional intolerance, and cognitive slowing, hallmarks of the vascular phenotype in biotoxin illness. VEGF suppression may reflect a compensatory downregulation due to chronic inflammation, TGF-β1 antagonism, or dysregulated HIF-1α signaling.
Neurovascular coupling, the process by which neural activity directs local increases in blood flow, is also highly dependent on VEGF and NO signaling. Disruption of this coupling mechanism contributes to the cognitive fatigue, brain fog, and executive dysfunction observed in CIRS. VEGF supports neuronal metabolism through direct neuroprotective actions, including synaptogenesis, blood–brain barrier integrity, and mitochondrial stabilization. Reduced VEGF availability in the CNS has been associated with impaired hippocampal plasticity, memory deficits, and vulnerability to neuroinflammation. Therefore, enhancing VEGF expression or mimicking its downstream effects holds promise for reversing some of the neurological symptoms of CIRS.
Therapeutic modulation of VEGF in CIRS must avoid the promotion of pathological angiogenesis, which is implicated in diseases such as cancer, diabetic retinopathy, and rheumatoid arthritis. The goal is not indiscriminate vessel proliferation but restoration of physiological perfusion and barrier competence. Several botanical agents offer this targeted modulation by activating HIF-1α, eNOS, or PI3K/Akt signaling without excessively promoting angiogenesis in normoxic conditions. These include Ginkgo biloba, Rhodiola rosea, Panax ginseng, Schisandra chinensis, and Cordyceps sinensis, each of which has demonstrated capacity to upregulate VEGF expression under hypoxic or inflammatory stress while also exerting antioxidant, mitochondrial-supportive, and endothelial-protective effects.
These botanicals often support VEGF pathways indirectly by mitigating the redox and inflammatory environment that suppresses VEGF transcription. For example, polyphenols and adaptogens within these herbs can enhance nuclear factor erythroid 2–related factor 2 (Nrf2) activity, reduce reactive oxygen species (ROS), and normalize nitric oxide signaling, thereby removing epigenetic and post-translational blocks on HIF-1α stabilization and VEGF release. Others modulate AMP-activated protein kinase (AMPK) and sirtuin pathways that influence mitochondrial biogenesis and oxygen utilization efficiency, pathways increasingly recognized as critical for VEGF-dependent endothelial health in CIRS.
Given these mechanisms, botanical agents that restore VEGF levels through adaptive and context-sensitive pathways represent an appealing strategy in CIRS management. Their pleiotropic effects allow for rebalancing of angiogenic tone, protection of neurovascular units, and normalization of tissue perfusion without provoking adverse neovascularization. The following subsections explore the VEGF-modulating actions of specific botanicals with clinical and mechanistic relevance to the hypoperfusion, and cognitive decline observed in biotoxin-mediated illness.
Ginkgo biloba
Ginkgo biloba, a gymnosperm with a long history in traditional medicine, contains multiple bioactive constituents including flavonol glycosides (quercetin, kaempferol) and terpene lactones (ginkgolides, bilobalide). These compounds act on cerebral vasculature through antioxidant, anti-inflammatory, and angiogenic pathways. Notably, Ginkgo biloba extract EGb 761 has been shown to upregulate VEGF mRNA and protein expression in ischemic and hypoxic models, largely via HIF-1α stabilization and increased eNOS expression. In a murine hindlimb ischemia model, EGb 761 enhanced capillary density and perfusion, effects that were associated with elevated VEGF and phosphorylated Akt in endothelial cells. In vitro, ginkgolides augment nitric oxide production through upregulation of eNOS and prevent endothelial cell apoptosis under oxidative stress. These actions may counteract the hypoperfusion and endothelial dropout observed in CIRS, where loss of VEGF-mediated support contributes to central and peripheral fatigue.
Neurovascular coupling, the synchronization between neuronal activation and local blood flow, is critically dependent on intact VEGF and NO signaling. Ginkgo biloba has been shown to improve cerebral blood flow and neurovascular responsiveness in aged populations and in models of neurodegeneration. Functional MRI studies in humans reveal increased regional perfusion following Ginkgo supplementation, particularly in areas subserving executive function and memory. These findings align with the cognitive domains frequently impacted in CIRS, suggesting a targeted role for Ginkgo in restoring angiogenic tone and neurovascular integrity.
Pharmacokinetically, Ginkgo biloba extract demonstrates good oral absorption, with peak plasma concentrations occurring within 1–2 hours post-ingestion. Flavonol aglycones are rapidly metabolized via glucuronidation and sulfation but retain partial bioactivity. Terpenoids such as ginkgolide B have longer half-lives and may accumulate in lipid-rich tissues, including the brain. EGb 761 is typically standardized to 24% flavone glycosides and 6% terpene lactones, with clinical doses ranging from 120 to 240 mg/day.
Rhodiola rosea
Rhodiola rosea, an adaptogenic herb native to cold mountainous regions, exerts broad regulatory effects on mitochondrial function, hypoxia tolerance, and angiogenesis. Its principal bioactive constituents, rosavin, salidroside, and tyrosol, are known to modulate HIF-1α stabilization, increase VEGF expression, and enhance cerebral oxygenation in stress models. Experimental evidence indicates that Rhodiola promotes VEGF production through both HIF-1α-dependent and -independent mechanisms. In a hypoxia-reoxygenation model using cultured endothelial cells, salidroside upregulated VEGF mRNA and protein levels via PI3K/Akt and ERK1/2 activation, concurrently increasing eNOS phosphorylation and nitric oxide output. These molecular effects were associated with enhanced tube formation, endothelial migration, and protection against hypoxia-induced apoptosis. In mouse models of ischemic stroke and brain hypoperfusion, Rhodiola treatment increased capillary density, reduced infarct size, and preserved cognitive performance.
In the context of CIRS, where low VEGF is often paralleled by fatigue and cognitive dulling, Rhodiola’s dual impact on perfusion and mitochondrial function may offer a multipronged benefit. Several studies have documented Rhodiola’s capacity to improve mental fatigue, reaction time, and cognitive flexibility under chronic stress and hypoxia, effects potentially mediated through restored neurovascular coupling and synaptic function.
Rhodiola rosea exhibits moderate oral bioavailability. Salidroside is water-soluble and undergoes limited hepatic metabolism, allowing for relatively predictable plasma levels. Rosavin and tyrosol are more lipophilic and may require lipid-based formulations for optimal absorption. Clinical dosing typically ranges from 200 to 600 mg/day of standardized extract (3% rosavins, 1% salidroside).
Panax ginseng
Panax ginseng exerts neurovascular benefits through its ginsenosides, particularly Rg1, Rb1, and Rg3, which promote angiogenesis and endothelial stability. These compounds activate HIF-1α and PI3K/Akt signaling, upregulating VEGF expression and enhancing nitric oxide (NO) production via eNOS phosphorylation. In cerebral ischemia models, ginsenosides restore perfusion and promote capillary growth while improving cognitive outcomes through BDNF–VEGF co-induction. Additionally, ginseng mitigates oxidative stress via Nrf2 activation, stabilizing mitochondrial dynamics in hypoxic endothelial cells. These effects are especially relevant to CIRS, where chronic vascular hypoperfusion and low VEGF contribute to fatigue and neurocognitive dysfunction. Ginsenosides undergo microbial deglycosylation into active forms such as compound K, improving bioactivity. Fermented and liposomal ginseng extracts enhance systemic absorption, overcoming limitations of poor solubility and first-pass metabolism.
Schisandra chinensis
Schisandra chinensis lignans, including schisandrin A and B, promote VEGF-mediated angiogenesis through activation of HIF-1α and MAPK pathways. In cerebral ischemia models, schisandrin B enhances VEGF expression and supports neovascular remodeling. Schisandra also preserves endothelial function by increasing eNOS activity and reducing endothelin-1, promoting capillary perfusion and oxygen delivery. In endothelial cell cultures, it reduces ROS and TNF-α–induced apoptosis, suggesting dual roles in vascular protection and redox stabilization. These mechanisms align with CIRS pathology, where VEGF depletion and microvascular injury underlie central fatigue and neuroinflammation. Schisandra’s bioavailability is enhanced by ethanol extraction and micronization techniques. Its combination of mitochondrial preservation, anti-apoptotic signaling, and VEGF upregulation makes it a promising candidate for restoring neurovascular coupling in biotoxin-associated syndromes.
Cordyceps sinensis
Cordyceps sinensis, a medicinal fungus rich in cordycepin and polysaccharides, has demonstrated pro-angiogenic effects via upregulation of VEGF and eNOS expression. In hypoxic endothelial and myocardial models, Cordyceps enhances HIF-1α signaling and promotes NO-mediated vasodilation, improving tissue perfusion and oxygenation. These actions support its traditional use for fatigue and respiratory insufficiency, both core features of CIRS. Cordyceps also reduces mitochondrial oxidative damage and inflammatory cytokines, contributing to endothelial repair. In models of ischemia-reperfusion injury, it preserves microvascular architecture and improves ATP generation. Cordycepin further modulates AMPK and Nrf2 signaling, supporting cellular adaptation to metabolic stress. Oral bioavailability is moderate, but hot-water extracts, and mycelial fermentation products demonstrate improved efficacy and systemic delivery. Cordyceps’ combined angiogenic and antioxidant effects directly counteract the hypoperfusion and redox dysfunction characteristic of low-VEGF states in CIRS.
Vasoactive Intestinal Peptide (VIP): Neuroimmune Synchronizing, Neuropeptide Signaling and Circadian Immune Balance
Vasoactive intestinal peptide (VIP) is a multifunctional, pleiotropic neuropeptide critical for maintaining homeostasis across the neuroendocrine-immune interface. Synthesized predominantly in the hypothalamus, suprachiasmatic nucleus (SCN), and peripheral parasympathetic neurons, VIP governs circadian rhythm regulation, anti-inflammatory signaling, and T cell homeostasis through tightly orchestrated cyclic AMP (cAMP)-mediated pathways. It signals through G protein–coupled VPAC receptors to elevate intracellular cAMP and activate CREB-dependent transcription. In the immune system, VIP suppresses pro-inflammatory cytokine release (e.g., IL-6, TNF-α), enhances Treg differentiation, and preserves epithelial barrier integrity.
In the context of CIRS, VIP is often pathologically suppressed, particularly in the later phases of biotoxin illness, reflecting neuroimmune exhaustion and circadian dysregulation. VIP deficiency contributes to immune disinhibition, disrupted circadian rhythms, and autonomic instability. VIP exerts its biological effects primarily through G protein–coupled receptors VPAC1 and VPAC2, which activate adenylate cyclase and increase intracellular cAMP. This cascade stimulates downstream transcription factors, most notably cAMP response element-binding protein (CREB), which modulates the transcription of anti-inflammatory cytokines (e.g., IL-10), neurotrophic factors (e.g., BDNF), and genes involved in circadian entrainment.
VIP’s role in promoting regulatory T cell (Treg) stability and suppressing Th1/Th17 polarization has been well-documented, positioning it as a key endogenous modulator of immune tolerance. VIP also attenuates NF-κB and MAPK activity in microglia and peripheral macrophages, reducing TNF-α, IL-6, and IL-1β secretion and thereby stabilizing neuroinflammatory tone. In CIRS, low VIP levels are associated with disrupted circadian biology, persistent inflammation, and impaired blood–brain barrier (BBB) function. Hypothalamic–pituitary–adrenal (HPA) axis desynchrony, characterized by dysregulated ACTH and cortisol rhythms, often parallels VIP suppression, suggesting a collapse of both top-down and bottom-up regulatory axes. Moreover, VIP deficiency correlates with reduced cerebral perfusion, cognitive slowing, and fatigue, hallmark symptoms of advanced biotoxin exposure. Importantly, exogenous VIP administration during the final stages of the CIRS Protocol has been shown to restore neurovascular coupling, improve oxygen delivery, and normalize cytokine profiles.
CREB is central to VIP-mediated effects, functioning as a transcriptional integrator of circadian light signaling and inflammatory suppression. In the SCN, CREB activation by VIP promotes Per1 and Per2 transcription, reinforcing circadian gene oscillations and synchronizing peripheral clocks. In the immune system, CREB enhances IL-10 and FOXP3 expression, supporting tolerogenic pathways and restraining chronic inflammation. Conversely, impaired CREB signaling, as seen in chronic stress, oxidative injury, or sleep disruption, may exacerbate immune dysregulation and reduce neurotrophic support.
cAMP, the secondary messenger linking VIP receptor activation to nuclear transcriptional control, also plays a vital role in BBB integrity, mitochondrial respiration, and neuroplasticity. Chronic cAMP suppression, whether due to receptor downregulation or oxidative degradation, has been implicated in microglial priming and mitochondrial uncoupling in neuroinflammatory disorders. Thus, restoring VIP–cAMP–CREB axis function is an essential therapeutic priority in CIRS.
A variety of botanicals modulate cAMP signaling, enhance CREB phosphorylation, and support neuroimmune synchronization. In particular, certain adaptogenic and nootropic herbs promote neurotrophic balance, circadian rhythm restoration, and resilience against immune provocation. The following section explores five botanicals—Lion’s Mane, Bacopa monnieri, Magnolia officinalis, Mucuna pruriens, and Albizia julibrissin, with mechanistic relevance to VIP signaling and its downstream neuroimmune pathways.
Hericium erinaceus (Lion’s Mane)
Lion’s Mane, a nootropic and neurotrophic mushroom, contains erinacines and hericenones that cross the blood–brain barrier and stimulate nerve growth factor (NGF) synthesis in astrocytes. While not directly influencing VIP gene expression, NGF–TrkA signaling synergizes with VIP-induced CREB activation, particularly in limbic and hypothalamic regions implicated in neuroimmune regulation. In a hippocampal neuronal culture model, Lion’s Mane extracts increased phosphorylation of CREB and promoted neurite outgrowth via ERK and cAMP pathways. These mechanisms mirror the trophic and circadian regulatory roles of VIP in suprachiasmatic nucleus (SCN) and hypothalamic circuitry.
In vivo, oral administration of Hericium erinaceus improved cognitive function and reduced depressive behaviors in animal models, accompanied by increased hippocampal neurogenesis and BDNF mRNA expression, an axis partially downstream of CREB. These neurogenic effects may support repair of hypothalamic injury and cortical dysregulation common in CIRS patients with VIP deficiency. Moreover, polysaccharides in Lion’s Mane exhibit immunomodulatory activity, enhancing dendritic cell maturation and restoring Th1/Th2 balance, further contributing to VIP’s homeostatic immune role.
Bacopa monnieri
Bacopa monnieri is a well-established adaptogen and cognitive enhancer that modulates neurotrophin expression, cholinergic transmission, and stress axis activity. Its active compounds, bacosides A and B, activate CREB via cAMP-dependent pathways and upregulate BDNF and NGF in hippocampal and cortical neurons. In this context, Bacopa indirectly supports VIP-mediated neuroimmune communication by stabilizing circadian outputs and enhancing neuroplasticity in hypothalamic-pituitary-limbic networks.
Animal studies have demonstrated that Bacopa reverses corticosterone-induced impairments in dendritic complexity and CREB phosphorylation, indicating restoration of cAMP/PKA signaling. These actions are particularly relevant to the neuroendocrine disarray in CIRS, where low VIP is associated with flattened cortisol rhythms, limbic atrophy, and disrupted feedback within the HPA axis.
In microglial cultures, Bacopa extract suppresses LPS-induced TNF-α and IL-6 production, reduces ROS generation and preserves mitochondrial function, mechanisms that indirectly support VIP production by minimizing hypothalamic neuroinflammation. Additionally, Bacopa enhances expression of circadian clock genes such as BMAL1 and PER2 in hypothalamic explants, implicating its utility in resynchronizing chrono disrupted patients with VIP loss.
Magnolia officinalis
Magnolia officinalis, a traditional botanical used for mood regulation and neuroinflammation, contains bioactive neolignans honokiol and magnolol that modulate both neurotransmitter systems and intracellular signaling cascades. These compounds enhance cAMP levels via PDE inhibition and potentiate GABA-A receptor activity, aligning with VIP’s role in anxiolysis and circadian gating. In hippocampal neurons, honokiol increases CREB phosphorylation and BDNF expression, conferring both antidepressant and neurotrophic effects.
Magnolia extracts also suppress microglial activation and inhibit expression of pro-inflammatory mediators (TNF-α, COX-2, iNOS) via downregulation of NF-κB and MAPK pathways, thus attenuating the neuroinflammatory milieu that impairs VIP production. In a study looking at chronic stress, Magnolia administration restored circadian cortisol patterns and improved REM sleep architecture, indicating a capacity to rebalance HPA and SCN outputs.
Pharmacokinetically, honokiol is rapidly absorbed and crosses the blood–brain barrier, with lipophilic formulations enhancing CNS bioavailability. Its dual role as a cAMP modulator and immunoneuroendocrine stabilizer positions Magnolia as a promising adjunct for restoring VIP tone in CIRS.
Mucuna pruriens
Mucuna pruriens, a leguminous plant rich in L-DOPA, directly replenishes central dopaminergic tone, an upstream regulator of VIP secretion from hypothalamic and limbic neurons. VIP-expressing neurons in the SCN and anterior hypothalamus exhibit dopaminergic innervation, and stimulation of D1 receptors increases VIP release and CREB activation. Thus, Mucuna’s enhancement of dopaminergic transmission may augment neuropeptide signaling relevant to Shoemaker’s VIP-deficient cohort.
In animal models of Parkinsonism and neuroinflammation, Mucuna extract restored striatal dopamine levels, reduced pro-inflammatory cytokines (e.g., IL-1β, TNF-α), and increased antioxidant enzyme expression, consistent with protection of VIP-producing circuits. Its polyphenolic constituents also upregulate mitochondrial biogenesis and reduce microglial activation—processes implicated in hypothalamic injury and VIP loss in CIRS. Standardized Mucuna extracts offer controlled L-DOPA delivery, with improved tolerability compared to synthetic formulations. Adjunct use with herbal adaptogens may synergistically support dopaminergic–VIP axis reconstitution.
Albizia julibrissin
Albizia julibrissin, or mimosa tree bark, is a traditional anxiolytic and sedative with emerging evidence for neuroimmune modulation. Its saponins and flavonoids modulate cAMP signaling and enhance CREB-mediated transcription of BDNF and other neuropeptides in hippocampal neurons. These effects align with VIP’s regulatory roles in synaptic plasticity, circadian entrainment, and emotional processing.
One study demonstrated Albizia extract restored diurnal rhythm of corticosterone and normalized SCN electrical activity, indicating a stabilizing effect on the hypothalamic circadian pacemaker. Additionally, Albizia attenuates stress-induced hippocampal inflammation and downregulates microglial TNF-α and IL-1β expression, protecting regions integral to VIP expression and output. Albizia’s central actions are complemented by peripheral immunomodulation, including suppression of mast cell degranulation and modulation of Th2 cytokines. These multifaceted properties support their use in VIP restoration, especially in CIRS patients exhibiting autonomic and limbic dysregulation.
Antidiuretic Hormone (ADH) and Plasma Osmolality: Fluid Balance and Neurohypophyseal Derailment
Antidiuretic hormone (ADH), also termed arginine vasopressin (AVP), is a pivotal neuropeptide for the regulation of extracellular fluid volume, serum osmolality, and vascular tone. Synthesized by magnocellular neurons within the supraoptic (SON) and paraventricular (PVN) nuclei of the hypothalamus, ADH is transported axonally to the posterior pituitary, where it is stored and secreted in response to osmotic or hemodynamic stimuli. Upon release, ADH binds to vasopressin receptors, primarily V1a, V1b, and V2, which are distributed across vascular smooth muscle, pituitary corticotrophs, and renal collecting duct epithelial cells, respectively. The most relevant for fluid homeostasis is the V2 receptor, located on the basolateral surface of principal cells in the collecting ducts, where ADH binding triggers insertion of aquaporin-2 (AQP2) channels into the apical membrane, facilitating water reabsorption and concentrating urine
109.
In CIRS, inappropriate suppression of ADH relative to elevated osmolality suggests hypothalamic injury or receptor resistance, contributing to polydipsia, frequent urination, orthostatic intolerance, and salt wasting. These findings reflect neurohypophyseal stress and hypothalamic-pituitary axis disruption, a consistent feature across environmentally acquired illnesses with central effects.
In the pathophysiology of CIRS regulation of this AVP-V2-AQP2 axis has emerged as a hallmark abnormality. Characteristic laboratory findings include inappropriately low serum ADH concentrations in the
context of elevated serum osmolality and sodium, reflecting a breakdown in hypothalamic osmo sensing or posterior pituitary secretion¹⁰,¹¹. This paradoxical hypodipsic hyperosmolality results in aquaresis, extracellular volume depletion, and secondary activation of compensatory systems such as the sympathetic nervous system and RAAS. Patients often present with profound thirst, polyuria, orthostatic intolerance, and reduced urine specific gravity, all reflective of a functional diabetes insipidus–like state.
On a molecular level, ADH–V2 receptor engagement leads to Gs protein–coupled activation of adenylate cyclase, increased intracellular cAMP, and subsequent activation of protein kinase A (PKA). PKA phosphorylates AQP2 at serine residues, promoting vesicular trafficking to the apical membrane of collecting duct cells¹¹⁰. Chronic disruption of this signaling, whether via biotoxin-mediated neuroinflammation, autoantibody formation, or mitochondrial injury in hypothalamic-pituitary structures, can lead to failure of AQP2 mobilization and water reabsorption. This effect is compounded by cytokine interference; TNF-α and IL-6 have been shown to downregulate AQP2 transcription and impair vasopressin receptor sensitivity through JAK/STAT and SOCS signaling, further diminishing renal responsiveness.
The renin–angiotensin–aldosterone system (RAAS) represents a critical co-regulator of volume homeostasis, acting synergistically with ADH to ensure sodium and water retention. Juxtaglomerular cells in the afferent arterioles of the kidney release renin in response to reduced renal perfusion pressure or sympathetic stimulation. Renin cleaves angiotensinogen to generate angiotensin I, which is converted to angiotensin II by angiotensin-converting enzyme (ACE). Angiotensin II binds AT1 receptors to initiate systemic vasoconstriction, stimulate thirst centers, promote ADH secretion, and upregulate sodium reabsorption in the proximal nephron. It also stimulates adrenal release of aldosterone, which acts on mineralocorticoid receptors in the distal tubules to increase epithelial sodium channel (ENaC) expression and Na⁺/K⁺-ATPase activity, promoting sodium retention and potassium excretion¹¹¹.
In the context of CIRS, RAAS integrity is frequently compromised. Chronic inflammation and oxidative stress impair mitochondrial function in juxtaglomerular cells, leading to insufficient renin release and blunted angiotensin II generation. Moreover, the adrenal cortex may be hypo-responsive due to cytokine-induced damage or disrupted HPA axis feedback. This contributes to inadequate aldosterone secretion and defective sodium conservation, particularly in the face of biotoxin-induced vascular permeability and salt loss. Importantly, low aldosterone levels in combination with low ADH exacerbate extracellular volume contraction, impair baroreceptor-mediated compensation, and perpetuate hypovolemia and orthostatic intolerance.
Recent evidence also implicates central mechanisms in the osmoregulatory dysfunction of CIRS. Hypothalamic inflammation, often driven by chronic TLR activation or oxidative injury, alters the firing threshold of osmoreceptor neurons and reduces the set point for AVP release. Furthermore, oxidative and excitotoxic damage to glial cells within the median eminence and organum vasculosum of the lamina terminalis (OVLT), key circumventricular organs lacking a complete blood–brain barrier, may impair the transduction of peripheral osmotic signals to neuroendocrine outputs. These disruptions mirror findings in other neuroinflammatory syndromes characterized by dysnatremia and volume instability.
Taken together, the coordinated dysfunction of the ADH–AQP2 system and RAAS in CIRS reflects a multi-tiered breakdown in central and peripheral osmoregulatory circuits. The result is a phenotype marked by inappropriate diuresis, hypovolemia, and an impaired ability to maintain electrolyte and volume homeostasis in the face of environmental, infectious, or biotoxin-related stressors. Therapeutic strategies targeting this axis may involve both neuroprotective agents that restore hypothalamic-pituitary integrity and agents that enhance renal responsiveness to AVP and RAAS hormones. The following botanicals have been identified for their ability to modulate vasopressin signaling, enhance aquaporin expression, or support RAAS activity in the context of volume dysregulation observed in CIRS.
Astragalus membranaceus
Astragalus membranaceus, a core adaptogenic herb in Traditional Chinese Medicine, demonstrates renoprotective and osmoregulatory properties through modulation of aquaporin expression and the renin–angiotensin–aldosterone system (RAAS). Polysaccharides and saponins in Astragalus enhance renal sodium retention while reducing proteinuria and glomerular hyperfiltration, especially under inflammatory stress conditions¹¹²,¹¹³. Astragalus has been shown to upregulate aquaporin-2 in vasopressin-deficient models and stabilize arginine vasopressin receptor-2 (AVPR2) mRNA expression in the renal medulla¹¹⁴. These effects support its use in cases of CIRS-related polyuria and sodium loss. Astragaloside IV, its principal active constituent, additionally suppresses angiotensin II–induced ROS and fibrosis, protecting against RAAS-mediated tubular injury. Its safety profile is favorable, though it may potentiate sodium retention in salt-sensitive individuals, requiring monitoring in volume-overloaded states.
Pueraria lobata (Kudzu)
Pueraria lobata, or kudzu root, contains isoflavones such as puerarin and daidzin that modulate hypothalamic-pituitary signaling, vascular tone, and renal perfusion. In models of water deprivation, puerarin promotes AVP release from the hypothalamic supraoptic nucleus and enhances vasopressin receptor responsiveness in collecting duct epithelia¹¹³. These actions align with its traditional use in managing dehydration, hangover, and thermoregulatory stress. Kudzu extracts also attenuate angiotensin II–induced hypertrophy in renal cells and reduce expression of aldosterone synthase, mitigating downstream salt retention and fibrosis¹¹⁴. The vasodilatory effects of puerarin via nitric oxide and cAMP pathways contribute to improved renal perfusion and osmotic regulation. While generally well tolerated,
Pueraria’s phytoestrogenic effects may require caution in hormonally sensitive individuals or long-term use.
Eucommia ulmoides
Eucommia ulmoides, a medicinal tree bark known for its antihypertensive and nephroprotective properties, influences fluid balance via RAAS modulation and nitric oxide–dependent vasodilation. Lignans and iridoids from Eucommia downregulate renin expression and suppress angiotensin II–mediated vasoconstriction and aldosterone secretion¹¹⁵. In hypertensive models, Eucommia extract reduces urinary sodium excretion and increases aquaporin-2 expression in the renal collecting duct, indicating potentiation of vasopressin signaling under volume-depleted conditions¹¹⁵. These effects may be particularly useful in CIRS patients with hypovolemia or excessive diuresis. Eucommia’s antioxidant and anti-inflammatory actions further protect hypothalamic osmoregulatory neurons from ROS-mediated injury. The herb is generally well tolerated, though hypotensive synergy with ACE inhibitors should be considered.
Ziziphus jujuba (Jujube)
Ziziphus jujuba, or jujube fruit, offers neuroendocrine and renal effects relevant to osmolality regulation in chronic inflammatory states. Rich in saponins, flavonoids, and cyclic AMP analogs, jujube modulates hypothalamic neuropeptides including CRH and AVP, supporting HPA-axis normalization¹¹⁶. In dehydration models, jujube increases pituitary vasopressin release and promotes AQP2 expression in renal tubules, supporting water reabsorption and plasma osmolality restoration (Park et al., 2015). Additionally, jujube exhibits aldosterone-suppressing effects via 11β-HSD inhibition and antioxidant regulation of RAAS feedback loops. These mechanisms may attenuate sodium wasting and polyuria observed in low-ADH phenotypes of CIRS. Ziziphus is traditionally used as a mild sedative; somnolence may occur at high doses, although no electrolyte disturbances have been reported.
Lepidium meyenii (Maca)
Lepidium meyenii, or maca root, is an Andean adaptogen with emerging evidence for neuroendocrine and osmoregulatory effects. Macamides and macaenes, its bioactive lipids, modulate hypothalamic-pituitary signaling and elevate AVP and ACTH in stress models, enhancing fluid retention and sodium conservation¹¹⁷. In rodent studies, maca supplementation preserves plasma osmolality and reduced polyuria following water deprivation, suggesting improved hypothalamic vasopressin output¹¹⁸. Its antioxidant properties also support neurohypophyseal neurons against oxidative injury, a mechanism relevant to AVP and ADH dysregulation in CIRS. Although generally well tolerated, maca may transiently raise blood pressure in sensitive individuals and should be used cautiously in volume-overloaded states. It shows promise as a tonic for adrenal and hypothalamic restoration under chronic biotoxin-induced stress.
Melanocyte-Stimulating Hormone (MSH): Terminal Integrator of Neuroimmune and Barrier Function
MSH is a cleavage product of pro-opiomelanocortin (POMC), secreted from the hypothalamus, with potent anti-inflammatory, antimicrobial, and gut-regulatory properties. It regulates mucosal immunity, pain sensitivity, sleep patterns, and microbial containment in mucous membranes²⁰.
MSH suppression is a hallmark of chronic biotoxin exposure, particularly when upstream regulators such as VIP and leptin are dysregulated¹⁰. MSH is a downstream barometer of unresolved inflammation and neuroendocrine collapse, often indicating the failure of multiple upstream systems.
Oxidative Stress: A Convergent Amplifier of Biomarker Dysregulation
Oxidative stress in CIRS is not merely a downstream effect of inflammation but functions as a cross-cutting disruptor of cellular, mitochondrial, and genomic stability. ROS production is amplified by complement activation (C4a), ECM degradation (MMP-9), and hypoxic signaling (via VEGF suppression), creating a network of feed-forward loops that impair cellular redox homeostasis²¹.
Excess ROS destabilize mitochondrial electron transport chains, induce mtDNA mutations, and oxidize membrane lipids, all of which further impair immune resolution. Moreover, oxidative stress inhibits Treg function and enhances Th17-driven inflammation, thereby compounding immune polarization driven by TGF-β1.
Because ROS can directly impair transcriptional programs (e.g., Nrf2 inhibition) and disrupt barrier tissue function, their control represents a high-yield therapeutic target. Antioxidant strategies must focus not just on scavenging ROS, but on restoring upstream redox sensing, mitochondrial biogenesis, and Nrf2 signaling.
Oxidative Stress and Mitochondrial Dysfunction
Oxidative stress and mitochondrial dysfunction are central features of CIRS, contributing to its multisystemic symptomatology and treatment resistance. Mitochondria are critical regulators of cellular energy metabolism, redox homeostasis, and innate immune signaling. However, under chronic biotoxin exposure or unresolved inflammation, mitochondrial membranes become destabilized, respiratory chain function is impaired, and reactive oxygen species (ROS) are excessively produced, fueling a pathological loop of oxidative damage and energy failure¹¹⁹.
One of the key endogenous defense mechanisms against oxidative stress is the nuclear factor erythroid 2–related factor 2 (Nrf2) pathway. Under basal conditions, Nrf2 is sequestered in the cytoplasm by Kelch-like ECH-associated protein 1 (Keap1), which targets it for ubiquitin-mediated degradation. Oxidative stress or electrophilic modification of Keap1 cysteine residues leads to Nrf2 stabilization, nuclear translocation, and transcriptional activation of antioxidant response element (ARE)–containing genes²². These genes encode for critical enzymes such as heme oxygenase-1 (HO-1), NAD(P)H:quinone oxidoreductase 1 (NQO1), glutathione S-transferases (GSTs), and superoxide dismutase (SOD), which collectively reduce ROS and preserve redox equilibrium.
In the context of CIRS, Nrf2 signaling appears to be functionally suppressed despite elevated oxidative stress markers, leading to unchecked lipid peroxidation, protein carbonylation, and mitochondrial DNA (mtDNA) damage¹⁹. Mitochondrial membrane potential (Δψm), a key indicator of mitochondrial integrity, becomes depolarized under oxidative assault, impairing ATP synthesis via oxidative phosphorylation (OXPHOS). This not only disrupts cellular energy supply but also promotes mitochondrial permeability transition pore (mPTP) opening, cytochrome c release, and caspase activation, mechanisms that underlie programmed cell death and tissue damage in CIRS-affected organs¹²⁰.
Damage to electron transport chain (ETC) complexes I–IV further exacerbates ROS production due to electron leakage, particularly at coenzyme Q and complex III sites. As a result, redox imbalance perpetuates further mitochondrial dysfunction, creating a vicious cycle of oxidative injury and impaired recovery. Inflammatory cytokines such as TNF-α and IL-1β also compromise mitochondrial biogenesis and fusion–fission dynamics, interfering with cellular resilience and adaptation¹²¹. Mitochondrial dysfunction disrupts calcium buffering and amplifies endoplasmic reticulum stress, processes implicated in neuroinflammation, fatigue, and cognitive decline commonly observed in CIRS.
Restoration of mitochondrial health requires both suppression of oxidative damage and stimulation of mitochondrial repair processes. Upregulation of Nrf2 activity has been shown to promote mitochondrial biogenesis via peroxisome proliferator–activated receptor gamma coactivator-1 alpha (PGC-1α) and to improve the expression of genes involved in mitochondrial quality control, such as mitofusin-2 (MFN2) and optic atrophy 1 (OPA1). Pharmacological and botanical activation of Nrf2 may therefore hold therapeutic relevance for CIRS patients, particularly those with persistent fatigue, myalgia, and cognitive impairment resistant to standard interventions.
Silybum marianum (Milk Thistle)
Silybum marianum contains silymarin, a complex of flavonolignans with potent antioxidant and mitochondrial-protective effects. Silymarin activates Nrf2 and upregulates phase II detoxification enzymes, including HO-1 and NQO1, while suppressing lipid peroxidation and mitochondrial ROS. In hepatocyte models, silybin stabilizes mitochondrial membrane potential and inhibits permeability transition pore opening, mitigating apoptosis under oxidative challenge. These effects are relevant in CIRS patients presenting with multiorgan involvement and inflammatory vulnerability. Silymarin also attenuates inflammatory cytokine expression and may indirectly modulate MMP-9 activity through NF-κB inhibition. Its capacity to stabilize redox tone and support mitochondrial fidelity across hepatic, renal, and CNS tissues makes it a systems-level adjunct in redox-sensitive CIRS phenotypes.
Polygonum cuspidatum (Japanese Knotweed)
Polygonum cuspidatum is rich in resveratrol, a polyphenol with pleiotropic mitochondrial and anti-inflammatory effects. Resveratrol activates SIRT1 and PGC-1α, enhancing mitochondrial biogenesis, electron transport chain (ETC) efficiency, and ATP production. Additionally, it stimulates Nrf2 and reduces mitochondrial ROS in models of neuroinflammation and metabolic stress. In murine studies, resveratrol downregulates MMP-9 and TGF-β1 expression, both of which are key players in CIRS pathophysiology²². Its BBB-penetrant profile and neurovascular protective effects also position it as a modulator of VIP-linked neuroimmune networks. Resveratrol’s oral bioavailability is low, but micronized and liposomal formulations improve pharmacokinetics, ensuring systemic redox impact¹²⁴.
Centella asiatica (Indian Pennywort)
Centella asiatica contains triterpenoid saponins such as asiaticoside and madecassoside, known to modulate mitochondrial function, Nrf2 activation, and angiogenesis³. These compounds enhance mitochondrial respiration and reduce ROS accumulation in neuroglial and hepatic models. Centella upregulates antioxidant enzymes (SOD, CAT, GPx) and supports neuronal mitochondrial integrity, particularly under hypoxic or inflammatory stress. It also promotes VEGF expression and neurovascular remodeling, supporting cross-talk with VIP-related pathways. In clinical studies, Centella has improved fatigue, mood, and cognitive parameters, symptoms frequently observed in mitochondrial dysfunction–driven CIRS exacerbations. Its adaptogenic and mitochondria-stabilizing profile supports broad application across organ systems.
Paeonia lactiflora (White Peony)
Paeonia lactiflora, through its active compound paeoniflorin, exhibits anti-inflammatory, antioxidative, and mitochondrial-preserving properties. Paeoniflorin reduces mitochondrial calcium overload, stabilizes membrane potential, and enhances ATP production in stress models¹²³. In immune cells, it suppresses NF-κB and downregulates pro-inflammatory cytokines including IL-1β and TNF-α. These effects may indirectly reduce MMP-9 activity and restore redox balance. Paeonia also improves cerebral blood flow and supports synaptic plasticity, overlapping mechanistically with VIP-neuroimmune modulation. In experimental autoimmune models, it has shown benefit in multi-organ inflammation, making it particularly suited for systemic oxidative burden seen in CIRS.
Tinospora cordifolia (Guduchi)
Tinospora cordifolia is a well-characterized adaptogen and immunomodulator that enhances mitochondrial antioxidant defense via Nrf2 activation and upregulation of glutathione synthesis. Its polyphenolic constituents reduce oxidative stress–induced mitochondrial damage and restore mitochondrial complex I and IV activity in hepatocellular and neuronal models. Tinospora also modulates pro-inflammatory signaling through inhibition of MAPK and NF-κB, suppressing downstream MMP-9 expression¹⁴⁷. In models of sepsis and liver injury, it improves mitochondrial membrane stability and reduces apoptotic signaling, which may be relevant for patients with persistent inflammation and organ impairment in CIRS. Its systemic antioxidant actions are complemented by
immune-normalizing properties, supporting multi-organ resilience.
Natural Binders for Jarisch–Herxheimer Reactions and Toxins: Mechanisms and Evidence
Antimicrobial therapy to eliminate infections often causes a Jarisch–Herxheimer reaction, also known as a die off reaction. This can be observed in individuals receiving botanical antimicrobials, similar to conventional antibiotic therapy. The process of pathogen elimination can trigger a systemic inflammatory response by unleashing proinflammatory cytokines, lipoproteins, and endotoxin-like compounds. The emergence of symptoms such as fever, gastrointestinal issues, malaise, and myalgia is promoted by cytokines such tumor necrosis factor α (TNF-α) and interleukins 6 and 8. After consuming botanical antimicrobials, symptoms usually start to appear one to two hours later and usually fade away in a day or two. In general, severity and infection burden seem interrelated.
In children with ASD and PANS associated with infectious triggers like Lyme Disease, effective detoxification strategies must address enterohepatic recirculation of lipophilic toxins, many of which are excreted via bile. Binding agents act within the gastrointestinal tract to adsorb these compounds and facilitate fecal elimination, reducing systemic reabsorption and inflammatory signaling¹²⁵,¹²⁶,¹²⁷,¹²⁸.
Toxin binders are a vital part of any successful antimicrobial and detox program since they help minimize die off symptoms, aid in detoxification, and ease the strain on elimination pathways. Children with ASD are often exposed to xenobiotics and biotoxins¹⁵². A systematic review from 2014 identified that the most common toxicants associated with ASD included pesticides, polychlorinated biphenyls (PCBs), phthalates, solvents, toxic waste sites, air pollutants, and heavy metals, with the strongest evidence found for air pollutants and pesticides¹⁵¹,¹⁵². While we highlight the usage of toxin binders effective in the binding of various microbial and environmental toxins, including mycotoxins, it is important to note their efficacy in treatment of CIRS has not been shown. Pertaining to all of the binders discussed below, objective benefit in treatment of CIRS has not been demonstrated.
Activated charcoal, a porous carbon-based adsorbent, has demonstrated broad-spectrum binding to various biotoxins. Its high surface area enables electrostatic and van der Waals interactions with hydrophobic toxins especially aflatoxin B1 and zearalenone¹²⁵,¹²⁶. While effective, charcoal may also bind micronutrients, necessitating judicious use.
Bentonite and montmorillonite clays, primarily composed of aluminum phyllosilicates, offer strong binding potential for microbial toxins, other biotoxins and environmental pollutants via ion-exchange and adsorption mechanisms. These clays have been extensively studied in animal feed and in vitro gastrointestinal models, showing significant reductions in toxin bioavailability¹²⁷. Clinical trials using processed montmorillonite show safety and efficacy in humans.
Chlorella, a freshwater green microalga, binds biotoxins and heavy metals through cell wall–associated polysaccharides and glycoproteins. In murine models, chlorella reduced aflatoxin B1–induced hepatotoxicity and genotoxicity¹²⁸. Due to its high nutritional content, it might be considered a safer option in sensitive populations. However, contamination with other cyanobacterial toxins remains a significant adverse effect of chlorella and a strong concern for CIRS patients.
Glucomannan, a soluble dietary fiber derived from Amorphophallus konjac, exhibits dose-dependent adsorption of aflatoxins, zearalenone, and T-2 toxins. It remains unabsorbed through the gut, forming a viscous gel that physically entraps mycotoxins¹²⁹. Its tolerability profile supports its use in patients with altered bowel motility, though we have no objective data confirming benefit in CIRS.
Modified citrus pectin (MCP), a low molecular weight pectic polysaccharide, binds demonstrated heavy metal chelation, and emerging evidence suggests potential toxin binding via galactose-rich domains¹³⁰. MCP is fermentable by gut microbiota, which may further enhance its detoxification synergy. Yet, no publications of objective data have shown benefit in CIRS.
Humic and fulvic acids, natural humic substances derived from decayed organic matter, exhibit amphipathic properties that enable interaction with diverse biotoxins. In vitro models demonstrate their affinity for various toxins and pollutants like via hydrogen bonding and cation exchange¹³¹. Their anti-inflammatory effects add additional value to individualized protocols. There is no evidence of any benefit in CIRS.
Probiotic strains, particularly Lactobacillus plantarum, L. rhamnosus, and Saccharomyces boulardii, bind biotoxins including neurotoxins, mycotoxins through peptidoglycan and polysaccharide-rich cell walls. These strains can immobilize aflatoxin B1, zearalenone, and ochratoxin A, and in some cases, enzymatically degrade toxins¹³²,¹³⁵. Soil-based bacilli like Bacillus subtilis also demonstrate mycotoxin degradation via manganese peroxidase activity¹³³. There is no evidence of any benefit in CIRS.
Okra: A closer look at bile acid binding natural binders
Cholestyramine, a bile acid–binding, cholesterol-lowering drug is the preferred binder used in the CIRS protocol with proven results. The bile acid–binding potential of foods like okra and other vegetables has been studied to assess their ability to decrease cholesterol¹⁵³,¹⁵⁴. Because less fat is absorbed and less cholesterol is used to produce bile acid, lowering bile acid recirculation also lowers cholesterol. Studies show the mucilaginous properties of okra along with botanical binding properties of few other vegetables can be a valuable resource for those seeking natural bile acid binders. In vitro bile acid binding properties for beets (18%), okra (16%), eggplant (14%), asparagus (10%), carrots (8%), green beans (7%), cauliflower (6%), and turnips (1%), as compared to cholestyramine, with a statistical significance of P ≤ .05. It was observed that in
vitro bile acid binding was greatly enhanced by steam cooking¹⁵³.
A multimodal approach using complementary binders offers enhanced theoretical efficacy against diverse microbial and biotoxins while minimizing nutrient loss, gastrointestinal side effects and assistance with detoxification, biotoxins binding and die off reactions.
Systems-Based Integration of Botanicals with the Shoemaker Protocol
The Shoemaker Protocol for CIRS provides a phase-based therapeutic architecture addressing biotoxin-induced immune and neuroendocrine dysregulation. While pharmacologic interventions remain the backbone of this protocol, systems-based integration of botanical therapeutics can enhance treatment response, target overlapping molecular pathways, and bridge gaps in patient resilience. Botanical compounds, selected for mechanistic specificity and temporal alignment, may augment detoxification, downregulate inflammatory biomarkers, restore neuroendocrine tone, and recalibrate immune regulation without disrupting the pharmacokinetics or pharmacodynamics of standard agents. This section systematically maps phytochemical interventions onto Shoemaker’s five-phase model using molecular targets such as MMP-9, TGF-β1, VEGF, VIP, ADH, and MSH.
Botanical adaptogens such as Rhodiola, Schisandra, Panax ginseng, and Bacopa exert pleiotropic effects across multiple axes—mitochondrial, immune, and neuroendocrine—offering resilience-enhancing properties rather than discrete molecular inhibition or activation. These agents stabilize physiological function under stress via modulation of HPA tone, cAMP signaling, and cytokine balance. They are ideally suited for buffering systemic volatility in CIRS patients undergoing biotoxin clearance.
Integration Without Disruption of Pharmaceutical Timelines
A critical consideration in botanical integration is compatibility with Shoemaker’s pharmaceutical regimen. Binding agents such as cholestyramine may non-selectively bind lipophilic plant constituents, reducing bioavailability. Therefore, antioxidant herbs should be administered at least two hours apart from binders. Similarly, intranasal antibiotics for MARCoNS should not be co-formulated with antimicrobial essential oils unless clinically evaluated for mucosal tolerance and botanical synergy¹³⁴.
TGF-β1 modulators such as Salvia miltiorrhiza and Baicalin should be introduced after toxin clearance to prevent interference with necessary immune activation during early detox phases. Neuropeptide-enhancing herbs should coincide with phase 4–5 interventions when pharmacologic VIP or DDAVP is introduced. Attention to formulation—liposomal, fermented, or ethanol-extracted forms—can optimize CNS and systemic delivery while avoiding pharmacokinetic conflicts.
Through synchronized botanical integration across Shoemaker’s phases, clinicians can leverage the systems biology of phytotherapy to enhance precision, resilience, and repair in biotoxin-affected patients.
Considerations for Safety, Dosing, and Pharmacokinetics
In the clinical application of botanical therapeutics for CIRS, careful attention must be paid to pharmacokinetic variability, safety profiles, and herb–drug interactions. As phytotherapeutic interventions span fractions within integrative frameworks, their compatibility with the Shoemaker Protocol depends not only on mechanistic alignment with biomarker pathology, but also on rigorous understanding of dose–response relationships, metabolic liabilities, and contraindications. This section provides a scientific analysis of key safety and pharmacokinetic factors influencing the deployment of botanicals already introduced in this article, with emphasis on cytochrome P450 modulation, bioavailability enhancement strategies, pediatric considerations, and botanical quality control.
Cytochrome P450 Interactions and Herb–Drug Synergy or Interference
Multiple botanicals employed in CIRS have demonstrated clinically significant interactions with the cytochrome P450 (CYP) enzyme superfamily, which mediates phase I hepatic biotransformation of xenobiotics. Curcumin, a diarylheptanoid polyphenol from turmeric, inhibits CYP3A4, CYP1A2, and CYP2C9 isoenzymes at physiologically relevant concentrations. This activity may potentiate plasma concentrations of concurrently administered pharmaceuticals metabolized by these pathways, such as corticosteroids, or statins, which are often used in the CIRS population.
Ginkgo biloba, extensively used for its angiogenic and cognitive effects, also modulates CYP enzymes. Standardized extracts inhibit CYP2C9 and CYP3A4 while inducing CYP2B6 and CYP2C19, with potential to influence clearance rates. Clinical caution is warranted with medications that require tight plasma control, such as warfarin, given Ginkgo’s additional antiplatelet effects. Similarly, Berberine has been shown to inhibit CYP2D6 and CYP3A4, which can increase the risk of drug accumulation and toxicity when co-administered with tricyclic antidepressants, macrolide antibiotics, or calcium channel blockers.
While CYP modulation can pose risks, it may also offer therapeutic synergy. Baicalin from Scutellaria baicalensis enhances hepatic detoxification by upregulating phase II glucuronidation via UGT1A1, potentially supporting biotoxin elimination without interfering with phase I oxidation. However, concurrent inhibition of efflux transporters such as P-glycoprotein by curcumin and berberine necessitates further vigilance for drug–herb interactions in polypharmacy contexts¹³⁵.
Bioavailability Optimization and Delivery Platforms
Several botanical compounds discussed in this review suffer from poor oral bioavailability due to rapid metabolism, low aqueous solubility, or poor gastrointestinal absorption. Curcumin, despite its pleiotropic molecular effects, demonstrates low systemic
exposure in humans. Co-administration with piperine from black pepper (Piper nigrum) has been shown to increase curcumin bioavailability by up to 2000% via inhibition of glucuronidation. Piperine-containing formulations, however, should be used cautiously in individuals with hepatic impairment or those on narrow therapeutic index drugs.
Lipophilic compounds such as resveratrol and boswellic acids also exhibit limited bioavailability, prompting the development of liposomal, nanoparticle, and phospholipid-complex formulations to enhance plasma concentrations and tissue penetration. Standardized Ginkgo extracts (e.g., EGb 761) utilize acetone–water extraction to ensure reproducible terpene lactone content and optimize absorption across the blood–brain barrier.
For highly polar constituents like baicalin or berberine, advanced delivery systems such as self-microemulsifying drug delivery systems (SMEDDS) and solid lipid nanoparticles (SLNs) have demonstrated improved pharmacokinetic profiles in animal models. Integration of such technologies can increase the therapeutic index of botanicals used in CIRS, particularly in tissues protected by barrier systems (e.g., CNS, sinuses).
Pediatric Use in CIRS-like Presentations
Although the Shoemaker Protocol is typically validated in adult populations, a subset of pediatric patients with autism spectrum disorder (ASD), PANS/PANDAS, or idiopathic neuroinflammatory syndromes may fulfill criteria for CIRS. In this context, safety and efficacy of botanicals in children must be carefully evaluated. Lion’s Mane (Hericium erinaceus), Bacopa monnieri, and Tinospora cordifolia have shown favorable neurodevelopmental safety profiles in animal studies and small clinical trials.
However, pediatric pharmacokinetics are distinct from adults due to developmental variability in hepatic enzyme expression, renal clearance, and blood–brain barrier permeability. For example, berberine may accumulate to higher levels in children due to immature CYP3A and P-glycoprotein activity, posing risk of bradycardia or hypotension. Licorice, even at modest doses, may increase the risk of pseudo-hyperaldosteronism and potassium depletion, particularly in low-bodyweight children with renal salt-wasting syndromes.
Appropriate pediatric formulations, alcohol-free glycerites, powdered extracts, or liposomal suspensions, should be prioritized, with dosing based on body surface area or weight (mg/kg) and monitored by a trained integrative clinician. Furthermore, select botanicals (e.g., Ginkgo biloba) lack sufficient pediatric safety data and should be avoided unless clinically indicated.
Herb Quality, Adulteration, and Standardization
Therapeutic reliability and safety of botanical preparations depend heavily on raw material quality, standardized extract content, and absence of adulterants. Good Agricultural and Collection Practices (GACP) and GMP-certified manufacturing protocols are essential to minimize batch variability and contamination. Adulteration remains a major concern, especially in imported herbal products; berberine-containing species are often substituted with lower-cost Coptis chinensis analogues, while Ginkgo preparations may be spiked with flavonol-rich extracts from unrelated plants.
Standardization ensures reproducible bioactivity by quantifying marker compounds such as curcuminoids (curcumin), ginkgolides (Ginkgo), bacosides (Bacopa), or withanolides (Withania somnifera). However, full-spectrum extracts may confer synergistic benefits not captured by single-constituent quantification. Herbal fingerprinting using HPLC, LC–MS/MS, or DNA barcoding should be employed by suppliers to verify identity and purity.
Heavy metal contamination (e.g., lead, arsenic), microbial load, and pesticide residues should be evaluated according to pharmacopeial standards, especially for botanicals sourced from regions with lax environmental oversight. Clinicians should request certificates of analysis (COAs) from manufacturers and preferentially use botanicals evaluated by third-party laboratories.
Specific Safety Concerns and Contraindications
Certain botanicals present unique toxicological risks that must be addressed in clinical use. Licorice (Glycyrrhiza glabra) inhibits 11β-hydroxysteroid dehydrogenase type 2, leading to cortisol-mediated mineralocorticoid receptor activation. This can induce sodium retention, hypertension, hypokalemia, and metabolic alkalosis. In CIRS patients with salt sensitivity or RAAS dysregulation, deglycyrrhizinated licorice (DGL) formulations may be preferable, though DGL lacks systemic anti-inflammatory activity.
Berberine, while a potent AMPK activator and antimicrobial, alters gut microbiota composition and may reduce microbial diversity with prolonged use¹³⁷. This is relevant in CIRS patients with MARCoNS or gut dysbiosis, where microbiome balance is critical. Additionally, berberine’s inhibition of cardiac hERG channels suggests caution in patients with QT prolongation risk.
Ginkgo biloba’s antiplatelet effects, mediated by ginkgolide B’s PAF antagonism, raise bleeding concerns in patients on anticoagulants or in perioperative settings. Curcumin may also potentiate bleeding risk via inhibition of thromboxane synthesis and platelet aggregation. Combined use of such botanicals with NSAIDs, warfarin, or SSRIs requires careful monitoring.
While botanical therapies offer promising molecular synergies with CIRS pathophysiology, their clinical utility must be framed within a rigorous pharmacological context. Individualized dosing, pediatric sensitivity, pharmacogenetic interactions, and quality assurance are non-negotiable parameters for safe integration. The path to phytotherapeutic adoption within the Shoemaker Protocol requires both mechanistic precision and cautious implementation informed by pharmacokinetic science and safety pharmacology.
Conclusion
The pathophysiology of CIRS involves convergent disruption of immune, vascular, neuroendocrine, and mitochondrial axes, driven by chronic biotoxin exposure and sustained by impaired host compensatory signaling. This multidimensional dysfunction challenges reductionist treatments, necessitating a systems-based approach. Guided by molecular biomarkers, botanical agents emerge not only as symptom mitigators but as precision tools capable of restoring disrupted feedback loops across CIRS networks.
Curcumin, baicalin, and resveratrol downregulate MMP-9 via MAPK and NF-κB inhibition, reducing endothelial permeability and leukocyte trafficking. Fibrotic remodeling, driven by TGF-β1, is attenuated by berberine, Scutellaria flavones, and Salvia diterpenoids, which inhibit SMAD2/3 signaling and restore Treg/Th17 balance. Complement dysregulation, exemplified by elevated C4a, is countered by Boswellia and Artemisia annua, which inhibit complement activation and reduce ROS, recalibrating Nrf2/NF-κB signaling. Botanicals also modulate VEGF, with EGCG and curcumin restoring angiogenic balance by suppressing pathological overexpression while correcting hypoxic deficits.
Within the hypothalamic-pituitary axis, herbs such as Hericium, Bacopa, and Mucuna engage neurotrophic and circadian pathways, aligning with VIP’s role in immune tolerance, sleep, and mood regulation. Astragalus and Ziziphus improve ADH function through aquaporin and receptor modulation, addressing osmoregulatory imbalance. Mitochondrial resilience is supported by Silybum, Tinospora, and Centella, which activate Nrf2, stabilize membrane potential, and enhance electron transport efficiency. These mechanisms complement Shoemaker interventions, which depend on adequate mitochondrial capacity for detoxification and neuroendocrine recovery.
A unifying theme is the ability of botanicals to selectively engage disrupted signaling while preserving physiological complexity. Adaptogens such as Rhodiola, ginseng, and Schisandra buffer multi-organ stress and modulate HPA tone, while pathway-specific agents like curcumin or berberine act on dominant biomarkers during specific protocol phases. Evidence-based integration with pharmaceuticals requires attention to herb–drug interactions, quality assurance, and standardized dosing, especially in pediatrics.
As the third of this four-part series, these findings integrate Part 1’s diagnostic framework and Part 2’s infectious insights, positioning herbal therapeutics as viable adjuncts for pediatric CIRS management. Future research should prioritize clinical trials and multi-omic profiling to validate efficacy and safety, advancing multidisciplinary care for complex neuroimmune disorders.
Acknowledgement
During the editing process of all 4 papers in this series, Microsoft Excel was used for assistance with the figures and tables. Microsoft Word editor was used for spell check and formatting purposes and Grammarly (which includes Grammarly AI) has been utilized for editing assistance for improved grammar and structural organization of original content for non-analytical language refinements. The generated responses have been fully reviewed, edited, proofread, and checked for accuracy and the lead author takes full responsibility for accuracy, originality, and ethical integrity.
References
1. Shoemaker RC, . Surviving Mold: Life in the Era of Dangerous Buildings. Otter Bay Books; 2010.
2. McMahon SW. Pediatric chronic inflammatory response syndrome (CIRS). In: Nutrition and Integrative Medicine for Clinicians, Volume Two. CRC Press/Taylor & Francis; 2023: chapter 4, 22 pages.
3. Shoemaker RC, House D, Ryan JC. Structural brain abnormalities in patients with inflammatory illness acquired following exposure to water-damaged buildings: a volumetric MRI study using NeuroQuant®. Neurotoxicol Teratol. 2014;45:18-26. PMID: 24946038.
4. Shoemaker, R., House, D. and Ryan, J. (2013) Vasoactive intestinal polypeptide (VIP) corrects chronic inflammatory response syndrome (CIRS) acquired following exposure to water-damaged buildings. Health 05(03):396-401. doi: 10.4236/health.2013.53053.
5. Panahi Y, Hosseini MS, Khalili N, Naimi E, Simental-Mendía LE, Majeed M, Sahebkar A. Effects of curcumin on serum cytokine concentrations in subjects with metabolic syndrome: a post-hoc analysis of a randomized controlled trial. Biomed Pharmacother. 2016 Aug;82:578-82. PMID: 27470399.
6. Zhao Y, Wu J, Liu X, Chen X, Wang J. Decoding nature: multi-target anti-inflammatory mechanisms of natural products in the TLR4/NF-κB pathway. Front Pharmacol. 2025 Jan 14;15:1467193. PMID: 39877388
7. Lu J, Zhong Y, Lin Z, et al. Baicalin alleviates radiation-induced epithelial–mesenchymal transition of primary type II alveolar epithelial cells via TGF-β and ERK/GSK3β signaling pathways. Biomed Pharmacother. 2017 Nov;95:1219-1224. PMID: 28931214
8. Curr Oncol. 2006 Feb;13(1):14-26. PMID: 17576437.
9. Vandooren J, Van den Steen PE, Opdenakker G. Biochemistry and molecular biology of gelatinase B or matrix metalloproteinase-9 (MMP-9): the next decade. Crit Rev Biochem Mol Biol. 2013;48(3):222-72. PMID: 23547785
10. Kirk SL, Karlik SJ. VEGF and vascular changes in chronic neuroinflammation. J Autoimmun. 2003 Dec;21(4):353-63. PMID: 14624758.
11. Massagué J. TGFβ signalling in context. Nat Rev Mol Cell Biol. 2012 Oct;13(10):616-30. PMID: 22992590.
12. Letterio JJ, Roberts AB. Regulation of immune responses by TGF-β. Annu Rev Immunol. 1998;16:137-161. PMID: 9597127.
13. Frangogiannis NG. Transforming growth factor–β in tissue fibrosis. J Exp Med. 2020;Feb 13;217(3):e20190103. PMID: 32997468.
14. Ricklin D, Hajishengallis G, Yang K, Lambris JD. Complement: a key system for immune surveillance and homeostasis. Nat Immunol. 2010 Sep;11(9):785-97. PMID: 20720586.
15. Sarma JV, Ward PA. The complement system. Cell Tissue Res. 2011 Jan;343(1):227-35. PMID: 20838815.
16. Olsson AK, Dimberg A, Kreuger J, Claesson-Welsh L. VEGF receptor signalling—in control of vascular function. Nat Rev Mol Cell Biol. 2006 May;7(5):359-371. PMID: 16633338.
17. Robinson ES, Khankin EV, Karumanchi SA, Humphreys BD. Hypertension induced by vascular endothelial growth factor signaling pathway inhibition: mechanisms and potential use as a biomarker. Semin Nephrol. 2010 Nov;30(6):591-601. doi: 10.1016/j.semnephrol.2010.09.007. PMID: 21146124
18. Gonzalez-Rey E, Delgado M. Vasoactive intestinal peptide and regulatory T-cell induction: a new mechanism and therapeutic potential for immune homeostasis. Trends Mol Med. 2007 Jun;13(6):241-51. PMID: 17467339.
19. Gray MR, Thrasher JD, Crago R, et al. Mixed mold mycotoxicosis: immunological changes in humans following exposure to water-damaged buildings. Arch Environ Health. 2003;58(7):410-420. doi:10.1080/00039896.2003.11879142. PMID: 15143854
20. Brzoska T, Luger TA, Maaser C, Abels C, Böhm M. Alpha-melanocyte-stimulating hormone and related tripeptides: biochemistry, anti-inflammatory and protective effects in vitro and in vivo, and future perspectives for the treatment of immune-mediated inflammatory diseases. Endocr Rev. 2008 Aug;29(5):581-602. PMID: 18612139.
21. Dröge W. Free radicals in the physiological control of cell function. Physiol Rev. 2002 Jan;82(1):47-95. PMID: 11773609
22. Ma Q. Role of Nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol. 2013;53:401-426. PMID: 23294312.
23. Feng D, Zou J, Su D, Mai H, Zhang S, Li P, Zheng X. Curcumin prevents high-fat diet-induced hepatic steatosis in ApoE−/− mice by improving intestinal barrier function and reducing endotoxin and liver TLR4/NF-κB inflammation. Nutr Metab (Lond). 2019 Nov 15;16:79. PMID: 31788011
24. Zhu T, Chen Z, Chen G, Wang D, Tang S, Deng H, Wang J, Li S, Lan J, Tong J, Li H, Deng X, Zhang W, Sun J, Tu Y, Luo W, Li C. Curcumin Attenuates Asthmatic Airway Inflammation and Mucus Hypersecretion Involving a PPARγ-Dependent NF-κB Signaling Pathway In Vivo and In Vitro. Mediators Inflamm. 2019 Apr 3;2019:4927430.PMID: 31073274
25. Gu XS, Wang F, Zhang CY, Mao CJ, Yang J, Yang YP, Liu S, Hu LF, Liu CF. Neuroprotective Effects of Paeoniflorin on 6-OHDA-Lesioned Rat Model of Parkinson’s Disease. Neurochem Res. 2016 Nov;41(11):2923-2936. Epub 2016 Jul 22. PMID: 27447883.
26. Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB. Bioavailability of curcumin: problems and promises. Mol Pharm. 2007 Nov-Dec;4(6):807-18. PMID: 17999464.
27. Bao M, Liang M, Sun X, Mohyuddin SG, Chen S, Wen J, Yong Y, Ma X, Yu Z, Ju X, Liu X. Baicalin Alleviates LPS-Induced Oxidative Stress via NF-κB and Nrf2-HO1 Signaling Pathways in IPEC-J2 Cells. Front Vet Sci. 2022 Jan 25;8:808233. PMID: 35146015.
28. Shen B, Zhang H, Zhu Z, Ling Z, Zeng F, Wang Y, Wang J. Baicalin Relieves LPS-Induced Lung Inflammation via the NF-κB and MAPK Pathways. Molecules. 2023; 28(4):1873. PMID: 36838858
29. Fang J, Zhu Y, Wang H, Cao B, Fei M, Niu W, Zhou Y, Wang X, Li X, Zhou M. Baicalin Protects Mice Brain From Apoptosis in Traumatic Brain Injury Model Through Activation of Autophagy. Front Neurosci. 2019 Jan 9;12:1006. PMID: 30686973
30. Fu YJ, Xu B, Huang SW, Luo X, Deng XL, Luo S, Liu C, Wang Q, Chen JY, Zhou L. Baicalin prevents LPS-induced activation of TLR4/NF-κB p65 pathway and inflammation in mice via inhibiting the expression of CD14. Acta Pharmacol Sin. 2021 Jan;42(1):88-96. Epub 2020 May 26. PMID: 32457419
31. Huang T, Liu Y, Zhang C. Pharmacokinetics and Bioavailability Enhancement of Baicalin: A Review. Eur J Drug Metab Pharmacokinet. 2019 Apr;44(2): 159-168. doi: 10.1007/s13318-018-0509-3. PMID: 30209794.
32. Annabi B, Lachambre MP, Bousquet-Gagnon N, Page M, Béliveau R. Green tea polyphenol (−) epigallocatechin-3-gallate inhibits MMP-2 secretion and MT1-MMP-driven migration in glioblastoma cells. Biochim Biophys Acta. 2002 Jan 30;1542(1-3):209-20. PMID: 11853893.
33. Sang S, Lambert JD, Ho CT, Yang CS. The chemistry and biotransformation of tea constituents. Pharmacol Res. 2011 Aug;64(2):87-99. PMID: 21371557.
34. Liu W, Dong M, Bo L, Li C, Liu Q, Li Y, Ma L, Xie Y, Fu E, Mu D, Pan L, Jin F, Li Z. Epigallocatechin-3-gallate ameliorates seawater aspiration-induced acute lung injury via regulating inflammatory cytokines and inhibiting JAK/STAT1 pathway in rats. Mediators Inflamm. 2014;2014:612593. doi: 10.1155/ 2014/612593. Epub 2014 Feb 20. PMID: 24692852
35. Walle T. Bioavailability of resveratrol. Ann N Y Acad Sci. 2011 Jan;1215(1):9-15. PMID: 21261636.
36. Yuyun X, Xi C, Qing Y, Lin X, Ke R, Bingwei S. Asiatic acid attenuates lipopolysaccharide-induced injury by suppressing activation of the Notch signaling pathway. Oncotarget. 2018 Jan 23;9(19):15036-15046. doi: 10.18632/oncotarget.24542. PMID: 29599924
37. Wattanathorn J, Mator L, Muchimapura S, Tongun T, Pasuriwong O, Piyawatkul N, Yimtae K, Sripanidkulchai B, Singkhoraard J. Positive modulation of cognition and mood in the healthy elderly volunteer following the administration of Centella asiatica. J Ethnopharmacol. 2008 Mar 5;116(2):325-32. Epub 2007 Dec 4. PMID: 18191355
38. Yesil-Celiktas O, Sevimli C, Bedir E, Vardar-Sukan F. Inhibitory effects of rosemary extracts, carnosic acid and rosmarinic acid on the growth of various human cancer cell lines. Plant Foods Hum Nutr. 2010 Jun;65(2):158-63. PMID: 20449663.
39. Osakabe N, Yasuda A, Natsume M, et al. Rosmarinic acid, a major polyphenolic component of perilla (Perilla frutescens), reduces lipopolysaccharide-induced liver injury in D-galactosamine-sensitized mice. Free Radic Biol Med. 2002 Sep 15;33(6):798-806. PMID: 12208367.
40. Al-Sereiti MR, Abu-Amer KM, Sen P. Pharmacology of rosemary (Rosmarinus officinalis Linn.) and its therapeutic potentials. Indian J Exp Biol. 1999 Feb;37(2):124-30. PMID: 10641130.
41. Robertson IB, Rifkin DB. Regulation of the bioavailability of TGF-β and TGF-β-related proteins. Cold Spring Harb Perspect Biol. 2016 Jun 1;8(6):a021907. PMID: 27252363.
42. Border WA, Noble NA. Transforming growth factor beta in tissue fibrosis. N Engl J Med. 1994 Nov 10;331(19):1286-92. PMID: 7935686.
43. Das R, Xu S, Quan X, Nguyen TT, Kong ID, Chung CH, Lee EY, Cha SK, Park KS. Upregulation of mitochondrial Nox4 mediates TGF-β-induced apoptosis in cultured mouse podocytes. Am J Physiol Renal Physiol. 2014 Jan;306(2):F155-67. Epub 2013 Nov 20. PMID: 24259511
44. Chitra P, Saiprasad G, Manikandan R, Sudhandiran G. Berberine inhibits Smad and non-Smad signaling cascades and enhances autophagy against pulmonary fibrosis. J Mol Med (Berl). 2015 Sep;93(9):1015-31. Epub 2015 Apr 17. PMID: 25877860.
45. Liu X, Liang Q, Wang Y, Xiong S, Yue R. Advances in the pharmacological mechanisms of berberine in the treatment of fibrosis. Front Pharmacol. 2024 Sep 20;15:1455058. PMID: 39372209
46. Yang G, Zhao Z, Zhang X, Wu A, Huang Y, Miao Y, Yang M. Effect of berberine on the renal tubular epithelial-to-mesenchymal transition by inhibition of the Notch/snail pathway in diabetic nephropathy model KKAy mice. Drug Des Devel Ther. 2017 Mar 31;11:1065-1079. PMID: 28408805.
47. Li C, Yin X, Xie C, Zeng J, Song C, Yang G, Zhang J, Chen S, Wei P, Wang Z, Gu M, Li W, An J, Pan Y. Berberine attenuates TNBS-induced colitis in mice by improving the intestinal microbiota. Front Microbiol. 2024 Aug 29;15:1463005.PMID: 39268532
48. Domitrović R, Jakovac H, Marchesi VV, Blažeković B. Resolution of liver fibrosis by isoquinoline alkaloid berberine in CCl₄-intoxicated mice is mediated by suppression of oxidative stress and upregulation of MMP-2 expression. J Med Food. 2013 Jun;16(6):518-28. Epub 2013 Jun 4. PMID: 23734997.
49. Gu J, Chen C, Wang J, Chen T, Yao W, Yan T, Liu Z. Withaferin A Exerts Preventive Effect on Liver Fibrosis through Oxidative Stress Inhibition in a Sirtuin 3-Dependent Manner. Oxid Med Cell Longev. 2020 Sep 24;2020:2452848. PMID: 33029279; PMCID: PMC7532400.
50. Kaileh M, Vanden Berghe W, Boone E, Essawi T, Haegeman G. Withaferin A strongly elicits IκB kinase β hyperphosphorylation concomitant with potent inhibition of its kinase activity. J Biol Chem. 2007 Feb 16;282(7):4253-4264. PMID: 17150968.
51. Bani S, Gautam M, Sheikh FA, et al. Selective Th1 up-regulating activity of Withania somnifera aqueous extract in an experimental system using flow cytometry. J Ethnopharmacol. 2006 Aug 11;107(1):107-15. PMID: 16603328.
52. Biswas, Pronama & Mathur, Dishali & Dinesh, Jinal & Dinesh, Hitesh & Desai, Belaguppa Manjunath Ashwin. (2025). Computational Analysis using Multi-ligand Simultaneous Docking of Withaferin A and Garcinol Reveals Enhanced BCL-2 and AKT-1 Inhibition. 10.48550/arXiv.2505.08632.
53. Tang B, Qiao H, Meng F, Sun X. Glycyrrhizin attenuates endotoxin-induced acute liver injury after partial hepatectomy in rats. Braz J Med Biol Res. 2007 Dec;40(12):1637-46. PMID: 17994167.
54. Zheng L, Zhang C, Li L, Hu C, Hu M, Sidikejiang N, Wang X, Lin M, Rong R. Baicalin ameliorates renal fibrosis via inhibition of transforming growth factor β1 production and downstream signal transduction. Mol Med Rep. 2017 Apr;15(4):1702-1712. PMID: 28260014.
55. Zhang Y, Zhang S, Chen X. Tanshinone IIA protects against cardiac fibrosis through inhibition of β-tubulin expression. J Biol Regul Homeost Agents. 2018 Nov-Dec;32(6):1451-1455. PMID:30574749.
56. Pan RH, Xie FY, Chen HM, Xu LZ, Wu XC, Xu LL, Yao G. Salvianolic acid B reverses the epithelial-to-mesenchymal transition of HK-2 cells that is induced by transforming growth factor-β. Arch Pharm Res. 2011 Mar;34(3):477-83. PMID:21547681.
57. Wu C, Chen W, Ding H, Li D, Wen G, Zhang C, Lu W, Chen M, Yang Y. Salvianolic acid B exerts anti-liver fibrosis effects via inhibition of MAPK-mediated phospho-Smad2/3 at linker regions in vivo and in vitro. Life Sci. 2019 Dec 15;239:116881. PMID:31678285.
58. Feng PP, Fang XS, Zhao SH, Fu JY, Zhang HT, Yi YL, Li CY, Jiang CL, Chen DP. Salvianolic acid B decreases interleukin-1β-induced colitis recurrence in mice. Chin Med J (Engl). 2020 Jun 20;133(12):1436-1444. PMID:32472783.
59. Wang X, Lee WYW, Or PMY, Yeung JHK. Pharmacokinetic interaction studies of tanshinones with tolbutamide, a model CYP2C11 probe substrate, using liver microsomes, primary hepatocytes and in vivo in the rat. Phytomedicine. 2010 Mar;17(3-4):203-11. PMID:19679455.
60. Hamza A, Amin A, Daoud S. The protective effect of a purified extract of Withania somnifera against doxorubicin-induced cardiac toxicity in rats. Cell Biol Toxicol. 2008 Jan;24(1):63-73. PMID: 17520333.
61. Paul S, Chakraborty S, Anand U, et al. Withania somnifera (L.) Dunal (Ashwagandha): a comprehensive review on ethnopharmacology, pharmacotherapeutics, biomedicinal and toxicological aspects. Biomed Pharmacother. 2021 Nov;143:112175. PMID: 34649336
62. Ward PA. The dark side of C5a in sepsis. Nat Rev Immunol. 2004 Feb;4(2):133-142. PMID: 15040586.
63. Carroll MC. The complement system in regulation of adaptive immunity. Nat Immunol. 2004 Oct;5(10):981-6. PMID: 15454921
64. Shoemaker RC, House D, Ryan JC. Structural brain abnormalities in patients with inflammatory illness acquired following exposure to water-damaged buildings: a volumetric MRI study using NeuroQuant®. Neurotoxicol Teratol. 2014 Sept;45:18-26. PMID: 24946038.
65. Fujisawa Y, Sakamoto M, Matsushita M, Fujita T, Nishioka K. Glycyrrhizin inhibits the lytic pathway of complement—possible mechanism of its anti-inflammatory effect on liver cells in viral hepatitis. Microbiol Immunol. 2000;44(9):799-804. PMID: 11092245.
66. Visavadiya NP, Narasimhacharya AVRL. Hypocholesterolaemic and antioxidant effects of Glycyrrhiza glabra (Linn) in rats. Mol Nutr Food Res. 2006 Nov;50(11):1080-6. PMID: 17054099.
67. Liu DK, Dong HF, Liu RF, Xiao XL. Baicalin inhibits the TGF-β1/p-Smad3 pathway to suppress epithelial-mesenchymal transition-induced metastasis in breast cancer. Oncotarget. 2020 Jul 21;11(29):2863-2872. PMID: 32754303.
68. Sadek MT, El-Abd SS, Ibrahim MAA. Effect of chronic unpredictable mild stress on adrenal cortex of adult rat and the possible protective role of licorice extract: a histological and immunohistochemical study. Egypt J Histol. 2021;44(4):887-901. doi:10.21608/ejh.2020.45532.1369.
69. Jin Q, Liu T, Chen D, Yang L, Mao H, Ma F, Wang Y, Li P, Zhan Y. Therapeutic potential of artemisinin and its derivatives in managing kidney diseases. Front Pharmacol. 2023 Feb 15;14:1097206. PMID: 36874000; PMCID: PMC9974673.
70. Zheng D, Liu T, Yu S, Liu Z, Wang J, Wang Y. Antimalarial Mechanisms and Resistance Status of Artemisinin and Its Derivatives. Trop Med Infect Dis. 2024 Sep 20;9(9):223. PMID: 39330912
71. Meshnick SR. Artemisinin: mechanisms of action, resistance and toxicity. Int J Parasitol. 2002; Dec 4;32(13):1655-60. PMID: 12435450.
72. Sahu A, Lambris JD. Structure and biology of complement protein C3, a connecting link between innate and acquired immunity. Immunol Rev. 2001 Apr;180:35-48. PMID: 11414361.
73. Ammon HPT. Modulation of the immune system by Boswellia serrata extracts and boswellic acids. Phytomedicine. 2010 Sep;17(11):862-7. PMID: 20696559.
74. Kapil A, Moza N. Anticomplementary activity of boswellic acids—a C3-convertase inhibitor of the classical complement pathway. Int J Immunopharmacology. 1992 Oct;14(7):1139–43. PMID: 1452399.
75. Safayhi H, Mack T, Sabieraj J, Anazodo MI, Subramanian LR, Ammon HP. Boswellic acids: novel, specific, nonredox inhibitors of 5-lipoxygenase. J Pharmacol Exp Ther. 1992 Jun;261(3):1143-6. PMID: 1602379.
76. Li Y, Yao J, Han C, et al. Quercetin, inflammation and immunity. Nutrients. 2016;8(3):167. PMID: 26999194.
77. Rogerio AP, Kanashiro A, Fontanari C, et al. Anti-inflammatory activity of quercetin and isoquercitrin in experimental murine allergic asthma. Inflamm Res. 2007;56(10):402-408. PMID: 17909804.
78. Boots AW, Haenen GR, Bast A. Health effects of quercetin: from antioxidant to nutraceutical. Eur J Pharmacol. 2008;585(2-3):325-337. PMID: 18417119.
79. Guo Y, Bruno RS. Endogenous mechanisms and exogenous factors that modulate bioavailability of quercetin and quercetin metabolites. J Nutr Biochem. 2015;26(3):201–210. PMID: 25468612
80. Marti HJ, Bernaudin M, Bellail A, Schoch H, Euler M, Petit E, Risau W. Hypoxia-induced vascular endothelial growth factor expression precedes neovascularization after cerebral ischemia. Am J Pathol. 2000 Mar;156(3):965-76. doi: 10.1016/ S0002-9440(10)64964-4. PMID: 10702412
81. Greenberg DA, Jin K. From angiogenesis to neuropathology. Nature. 2005 Dec 15;438(7070): 954-959. PMID: 16355213.
82. Tang Y, Zhou G, Yao L, Xue P, Yu D, Xu R, Shi W, Yao X, Yan Z, Duan JA. Protective effect of Ginkgo biloba leaves extract, EGb761, on myocardium injury in ischemia reperfusion rats via regulation of TLR-4/NF-κB signaling pathway. Oncotarget. 2017 Sep 28;8(49):86671-86680. PMID: 29156826
83. Sun BL, Hu DM, Yuan H, Ye WJ, Wang XC, Xia ZL, Zhang SM, Wang LX. Extract of Ginkgo biloba promotes the expression of VEGF following subarachnoid hemorrhage in rats. Int J Neurosci. 2009;119(7):995-1005. PMID: 19466634.
84. Koltermann A, Hartkorn A, Koch E, Fürst R, Vollmar AM. Ginkgo biloba extract EGb 761 increases endothelial nitric oxide production in vitro and in vivo. Cell Mol Life Sci. 2007;64(13):1715-1722. PMID: 17497242.
85. Tchantchou F, Xu Y, Wu Y, Christen Y, Luo Y. EGb 761 enhances adult hippocampal neurogenesis and phosphorylation of CREB in transgenic mouse model of Alzheimer’s disease. FASEB J. 2007;Aug 21(10):2400-2408. PMID: 17356006.
86. Wesnes KA, Ward T, McGinty A, Petrini O. The cognitive, subjective, and physical effects of a Ginkgo biloba/Panax ginseng combination in healthy volunteers with neurasthenic complaints. Psychopharmacology (Berl). 1997;33(4):677-683. PMID: 9493479.
87. DeFeudis FV, Drieu K. Ginkgo biloba extract (EGb 761) and CNS functions: basic studies and clinical applications. Curr Drug Targets. 2000;Jul;1(1):25-58. PMID: 11475535.
88. Panossian A, Wikman G. Evidence-based efficacy of adaptogens in fatigue, and molecular mechanisms related to their stress-protective activity. Curr Clin Pharmacol. 2009;Sept 4(3):198-219. PMID: 19500070.
89. Zhong ZF, Han J, Zhang JZ, Xiao Q, Chen JY, Zhang K, Hu J, Chen LD. Neuroprotective effects of salidroside on cerebral ischemia/reperfusion-induced behavioral impairment involves the dopaminergic system. Front Pharmacol. 2019;Dec 13:1433. PMID: 31920641
90. Spasov AA, Wikman GK, Mandrikov VB, Mironova IA, Neumoin VV. A double-blind, placebo-controlled pilot study of the stimulating and adaptogenic effect of Rhodiola rosea SHR-5 extract on the fatigue of students caused by stress during an examination period with a repeated low-dose regimen. Phytomedicine. 2000;Apr;7(2):85-89. PMID: 10839209.
91. Ghafouri-Fard S, Balaei N, Shoorei H, Hasan SMF, Hussen BM, Talebi SF, Taheri M, Ayatollahi SA. The effects of Ginsenosides on PI3K/AKT signaling pathway. Mol Biol Rep. 2022 Jul;49(7):6701-6716. Epub 2022 Feb 27. PMID: 35220526.
92. Li Y, Guan Y, Wang Y, Yu CL, Zhai FG, Guan LX. Neuroprotective Effect of the Ginsenoside Rg1 on Cerebral Ischemic Injury In Vivo and In Vitro Is Mediated by PPARγ-Regulated Antioxidative and Anti-Inflammatory Pathways. Evid Based Complement Alternat Med. 2017;2017:7842082. Epub 2017 Jun 1. PMID: 28656054
93. Bian XB, Yu PC, Yang XH, Han L, Wang QY, Zhang L, Zhang LX, Sun X. The effect of ginsenosides on liver injury in preclinical studies: a systematic review and meta-analysis. Front Pharmacol. 2023 May 11;14:1184774. PMID: 37251340
94. Fang Y, Pan J, Wang P, Wang R, Liang S. A comprehensive review of Schisandrin B’s preclinical antitumor activity and mechanistic insights from network pharmacology. Front Pharmacol. 2025 Feb 10;16:1528533. PMID: 39995410
95. Ehambarampillai, D., Wan, M.L.Y. A comprehensive review of Schisandra chinensis lignans: pharmacokinetics, pharmacological mechanisms, and prospects in disease prevention and treatment. Chin Med 20, 47 (2025). https://doi.org/10.1186/s13020-025-01096-z
96. Krishna KV, Ulhas RS, Malaviya A. Bioactive compounds from Cordyceps and their therapeutic potential. Crit Rev Biotechnol. 2024 Aug;44(5):753-773. Epub 2023 Jul 30. PMID: 37518188.
97. Shoemaker RC, House DE. Sick building syndrome (SBS) and exposure to water-damaged buildings: time series study, clinical trial and mechanisms. Neurotoxicol Teratol. 2006 Sep-Oct;28(5):573-88. Epub 2006 Aug 7. PMID: 17010568.
98. Delgado M, Ganea D. Vasoactive intestinal peptide: a neuropeptide with pleiotropic immune functions. Amino Acids. 2013 Jul;45(1):25-39. PMID:22139413.
99. Ma, Bing-ji, Jin-Wen Shen, Hai-You Yu, Yuan Ruan, Ting-Ting Wu and Xu Zhao. Hericenones and erinacines: stimulators of nerve growth factor (NGF) biosynthesis in Hericium erinaceus. Mycology 2010 1(2): 92 – 98.
100. Mori K, Inatomi S, Ouchi K, Azumi Y, Tuchida T. Improving effects of the mushroom Yamabushitake (Hericium erinaceus) on mild cognitive impairment: a double-blind placebo-controlled clinical trial. Phytother Res. 2009 Mar;23(3):367–372. PMID: 18844328
101. Chiu C-H, Chyau C-C, Chen C-C, Lee L-Y, Chen W-P, Liu J-L, Lin W-H, Mong M-C. Erinacine A-Enriched Hericium erinaceus Mycelium Produces Antidepressant-Like Effects through Modulating BDNF/PI3K/Akt/GSK-3β Signaling in Mice. International Journal of Molecular Sciences. 2018 Jan 24; 19(2):341. PMID: 29364170
102. Bhattacharya SK, Bhattacharya A, Kumar A, Ghosal S. Antioxidant activity of Bacopa monniera in rat frontal cortex, striatum and hippocampus. Phytother Res. 2000 May;14(3):174-179. PMID:10815010
103. Shinomol GK, Muralidhara. Bacopa monnieri modulates endogenous cytoplasmic and mitochondrial oxidative markers in prepubertal mice brain. Phytomedicine. 2011 Feb 15;18(4):317-326. PMID:20850955.
104. Russo A, Izzo AA, Borrelli F, Renis M, Vanella A. Free radical scavenging capacity and protective effect of Bacopa monniera L. on DNA damage. Phytother Res. 2003 sep;17(8):870–875. PMID: 13680815.
105. Chuang DY, Chan MH, Zong Y, Sheng W, He Y, Jiang JH, Simonyi A, Gu Z, Fritsche KL, Cui J, Lee JC, Folk WR, Lubahn DB, Sun AY, Sun GY. Magnolia polyphenols attenuate oxidative and inflammatory responses in neurons and microglial cells. J Neuroinflammation. 2013 Jan 29;10:15. PMID: 23356518.
106. Zhang J, Chen Z, Huang X, Shi W, Zhang R, Chen M, Huang H, Wu L. Insights on the Multifunctional Activities of Magnolol. Biomed Res Int. 2019 May 23;2019:1847130. PMID: 31240205.
107. Hanin I. Central neurotransmitter function and its behavioral correlates in man. Environ Health Perspect. 1978 Oct;26:135-41. PMID: 31279.
108. Manyam BV, Dhanasekaran M, Hare TA. Neuroprotective effects of the antiparkinson drug Mucuna pruriens. Phytother Res. 2004 Sep;18(9):706–712. PMID: 15478206.
109. Birnbaumer M. Vasopressin receptors. Trends Endocrinol Metab. 2000 Dec;11(10):406-410. PMID: 11091117.
110. Fushimi K, Sasaki S, Marumo F. Phosphorylation of serine 256 is required for cAMP-dependent regulatory exocytosis of the aquaporin-2 water channel. J Biol Chem. 1997 Jun;272(23):14800-14804. PMID: 9169447.
111. Hall JE. Guyton and Hall Textbook of Medical Physiology. 12th ed. Elsevier Saunders; 2011.
112. Meng X, Wei M, Wang D, et al. Astragalus polysaccharides protect renal function and affect the TGF-β/Smad signaling pathway in streptozotocin-induced diabetic rats. J Int Med Res. 2020 May;48(5):300060520903612. PMID: 32475187
113. Gao HL, Yang Y, Tian H, Xu SL, Li BW, Fu LY, Liu KL, Shi XL, Kang YM, Yu XJ. Puerarin Alleviates Blood Pressure via Inhibition of ROS/TLR4/NLRP3 Inflammasome Signaling Pathway in the Hypothalamic Paraventricular Nucleus of Salt-Induced Prehypertensive Rats. Nutrients. 2024 Aug 6;16(16):2580. PMID: 39203718
114. Zhou X, Bai C, Sun X, et al. Puerarin attenuates renal fibrosis by reducing oxidative stress induced-epithelial cell apoptosis via MAPK signal pathways in vivo and in vitro. Ren Fail. 2017 Nov;39(1):423-431. PMID: 28335679.
115. Gong M, Zhang H, Liu X, et al. Effect of Eucommia ulmoides leaves on hyperuricemia and kidney injury induced by a high-fat/high-fructose diet in rats. Iran J Basic Med Sci. 2022 Apr ;25(4):527-535. PMID: 35656068.
116. Chen J, Liu X, Li Z, Qi A, Yao P, Zhou Z, Dong TTX, Tsim KWK. A Review of Dietary Ziziphus jujuba Fruit (Jujube): Developing Health Food Supplements for Brain Protection. Evid Based Complement Alternat Med. 2017;2017:3019568. Epub 2017 Jun 7. PMID: 28680447.
117. Gonzales GF, Cordova A, Vega K, et al. Effect of Lepidium meyenii (maca) on sexual desire and its absent relationship with serum testosterone levels in healthy adult men. Andrologia. 2006 Dec;34(6): 367-372. PMID: 12472620.
118. Ai Z, Cheng AF, Yu YT, Yu LJ, Jin W. Antidepressant-like behavioral, anatomical, and biochemical effects of petroleum ether extract from maca (Lepidium meyenii) in mice exposed to chronic unpredictable mild stress. J Med Food. 2014 May;17(5):535-542. PMID: 24730393
119. Naviaux RK, Naviaux JC, Li K, et al. Metabolic features of chronic fatigue syndrome. Proc Natl Acad Sci U S A. 2016 Sep 13;113(37):E5472-E5480. PMID: 27573827.
120. Kroemer G, Galluzzi L, Brenner C. Mitochondrial membrane permeabilization in cell death. Physiol Rev. 2007 Jan;87(1):99-163. PMID: 17237344.
121. Picard M, McManus MJ, Gray JD, et al. Mitochondrial functions modulate neuroendocrine, metabolic, inflammatory, and transcriptional responses to acute psychological stress. Proc Natl Acad Sci U S A. 2015 Dec 1;112(48):E6614-E6623. PMID: 26627253.
122. Kong AN, Yu R, Hebbar V, Chen C, Mandlekar S, . Signal transduction events elicited by cancer prevention compounds. Mutat Res. 2000 Sep 1;480-481:231–241. PMID: 11506817.
123. Wu F, Ye B, Wu X, et al. Paeoniflorin on Rat Myocardial Ischemia Reperfusion Injury of Protection and Mechanism Research. Pharmacology. 2020;105(5-6):281-288. PMID: 31618740.
124. Hope J. A review of the mechanism of injury and treatment approaches for illness resulting from exposure to water-damaged buildings, mold, and mycotoxins. Scientific World Journal. 2013 Apr 18;2013:767482. PMID: 23710148
125. Lauterburg BH, Dickson ER, Pineda AA, Carlson GL, Taswell HF. Removal of bile acids and bilirubin by plasmaperfusion of U.S.P. charcoal-coated glass beads. Gastroenterology. 1979 Oct;94(4):585-592. PMID: 479668.
126. Rotter RG, Frohlich AA, Marquardt RR. Influence of dietary charcoal on ochratoxin A toxicity in Leghorn chicks. Can J Vet Res. 1989 Oct;53(3):449-453. PMID: 2590872.
127. Wang JS, Luo H, Billam M, et al. Short-term safety evaluation of processed calcium carbonate montmorillonite clay (Clay) in humans. Food Addit Contam. 2005 Mar;22(3):270-279. PMID: 16019795.
128. Jubert C, Mata JE, Bench G, et al. Effects of chlorophyll and chlorophyllin on low-dose aflatoxin B1 pharmacokinetics in human volunteers. Cancer Prev Res (Phila). 2009 Dec;2(12):1015-1022. PMID: 19952359.
129. Devreese M, Girgis GN, Tran ST, et al. The effects of feed-borne Fusarium mycotoxins and glucomannan in turkey poults based on specific and non-specific parameters. Food Chem Toxicol. 2014 Jan;63:69-75. PMID: 24200858.
130. Zhao ZY, Liang L, Fan X, et al. The role of modified citrus pectin as an effective chelator for heavy metals lead in children hospitalized with toxic lead levels. Altern Ther Health Med. 2008 Jul-Aug;14(4):34-38. PMID: 18616067.
131. Yang HL, Chen CS, Chang WH, et al. Humic acid induces apoptosis in human premyelocytic leukemia HL-60 cells. Life Sci. 2004 Aug 27;75(1):1817-31. PMID: 15302226.
132. Hamidi A, Ghanbarpour R, Alizadeh A, Khoshbakht R. The aflatoxin B1 isolating potential of two lactic acid bacteria. Asian Pac J Trop Biomed. 2013 Sep;3(9):732-736. PMID: 23998015.
133. Qin X, Su X, Tu T, et al. Enzymatic degradation of multiple major mycotoxins by dye-decolorizing peroxidase from Bacillus subtilis. Toxins (Basel). 2021 Jun 19;13(6):429. PMID: 34205294.
134. Bozin B, Mimica-Dukic N, Simin N, Anackov G. Characterization of the volatile composition of essential oils of some Lamiaceae spices and the antimicrobial and antioxidant activities of the entire oils. J Agric Food Chem. 2006 Mar 8;54(5):1822-1828. PMID: 16506839.
135. Lee LS, Andrade AS, Flexner C. Interactions between natural health products and antiretroviral drugs: pharmacokinetic and pharmacodynamic effects. Clin Infect Dis. 2006 Oct 15;43(8):1052-9. Epub 2006 Sep 8. PMID: 16983620.
136. Joshi K, Chavan P, Warude D, Patwardhan B. Molecular markers in herbal drug technology. Curr Sci Asso. 2004;87(2):159–165.
137. Habtemariam S. Berberine pharmacology and the gut microbiota: a hidden therapeutic link. Pharmacol Res. 2020 May;155:104722. PMID: 32105754.
138. Prato A, Gulisano M, Scerbo M, Barone R, Vicario CM, Rizzo R. Diagnostic approach to pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections (PANDAS): a narrative review of literature data. Front Pediatr. 2021 Oct 27;9:746639. PMID: 34778136
139. Shoemaker RC, House DE. A time-series study of sick building syndrome: chronic, biotoxin-associated illness from exposure to water-damaged buildings. Neurotoxicol Teratol. 2005 Jan-Feb;27(1):29-46. PMID: 15681119.
140. Swedo SE, Leckman JF, Rose NR. From research subgroup to clinical syndrome: Modifying the PANDAS criteria to describe PANS (Pediatric Acute-onset Neuropsychiatric Syndrome). Pediatrics & Therapeutics. 2012 Jan;(2):1-8. doi:10.4172/2161-0665.1000113
141. McMahon SW, Smith J. Pediatric Quirks of CIRS and PANS. Paper presented at: Surviving Mold Annual Conference; May 4th, 2018; Salisbury, MD.
142. Shoemaker RC, Maizel MS. Exposure to interior environments of water-damaged buildings causes a CFS-like illness in pediatric patients: a case/control study. Bull IACFS ME. 2009;17(2):69-81.
143. Buhner S. Herbal Antibiotics, 2nd Edition: Natural Alternatives for Treating Drug-Resistant Bacteria. Storey Publishing; 2015.
144. Talbott SM, Talbott JA, Pugh M. Effect of Magnolia officinalis and Phellodendron amurense (Relora®) on cortisol and psychological mood state in moderately stressed subjects. J Int Soc Sports Nutr. 2013 Aug 7;10(1):37. PMID: 23924268.
145. Bishan Huang, Yingyao Wu, Chan Li, Qingfa Tang, Yuanwei Zhang. Molecular basis and mechanism of action of Albizia julibrissin in depression treatment and clinical application of its formulae. Chinese Herbal Medicines,2023 15(2):201-213. https://doi.org/10.1016/j.chmed.2022.10.004.
146. Ma J, Fan S, Chen J, Gu Y, Lin S. Messenger RNA expressions of vasopressin system and aquaporin-2 in adriamycin-induced nephrotic rats and effects of astragalus membranaceus. Chin Med J (Engl). 1999 Dec;112(12):1068-72. PMID: 11721440.
147. Wu W, Huang J, Han P, Zhang J, Wang Y, Jin F, Zhou Y. Research Progress on Natural Plant Molecules in Regulating the Blood-Brain Barrier in Alzheimer’s Disease. Molecules. 2023 Nov 16;28(22):7631. PMID: 38005352
148. Kong J, Wang Y, Xia K, Zang N, Zhang H, Liang X. New insights into the antibacterial and quorum sensing inhibition mechanism of Artemisia argyi leaf extracts towards Pseudomonas aeruginosa PAO1. 3 Biotech. 2021 Feb;11(2):97Epub 2021 Jan 27. PMID: 33520583.
149. Philip, S., Tom, G., Balakrishnan Nair, P. et al. Tinospora cordifolia chloroform extract inhibits LPS-induced inflammation via NF-κB inactivation in THP-1cells and improves survival in sepsis. BMC Complement Med Ther 21, 97 (2021). https://doi.org/10.1186/s12906-021-03244-y.
150. Wood JM, Sbar E. Jarisch-Herxheimer Reaction. [Updated 2025 Jul 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK557820/
151. Rossignol DA, Genuis SJ, Frye RE. Environmental toxicants and autism spectrum disorders: a systematic review. Transl Psychiatry. 2014 Feb 11;4(2):e360. PMID: 24518398.
152. Faber S, Fahrenholz T, Wolle MM, Kern JC 2nd, Pamuku M, Miller L, Jamrom J, Skip Kingston HM. Chronic exposure to xenobiotic pollution leads to significantly higher total glutathione and lower reduced to oxidized glutathione ratio in red blood cells of children with autism. Free Radic Biol Med. 2019 Apr; 134:666-677. PMID: 30763613.
153. Talwinder S. Kahlon, Mei-Chen M. Chiu, Mary H. Chapman, Steam cooking significantly improves in vitro bile acid binding of beets, eggplant, asparagus, carrots, green beans, and cauliflower, Nutrition Research. 2007 27(12), Issue 12, 750-755. ISSN 0271-5317, doi.org/10.1016/j.nutres.2007.09.011.
154. T.S. Kahlon, M.H. Chapman, G.E. Smith. In vitro binding of bile acids by okra, beets, asparagus, eggplant, turnips, green beans, carrots, and cauliflower. Food Chemistry,2007 103(2), 676-680. ISSN 0308-8146, doi.org/10.1016/j.foodchem.2006.07.056.
155. Lam TI, Tam CC, Stanker LH, Cheng LW. Probiotic Microorganisms Inhibit Epithelial Cell Internalization of Botulinum Neurotoxin Serotype A. Toxins (Basel). 2016 Dec 16;8(12):377. PMID: 27999281.
156. Dashore, JA., Dashore, B., et al., 2025. Chronic Inflammatory Response Syndrome: Exploring Neuroimmune Pathology and Multisystem Framework for Differential Diagnosis in Pediatrics Part 1. Medical Research Archives, [online] 13(11). https://doi.org/10.18103/mra.v13i11.6952
157. Dashore, J., A., Dashore B., McMahon, S., Shoemaker R., Autism Spectrum Disorders and Lyme Disease: Exploring Shared Neuro-lnflammatory and Immune Pathways. Medical Research Archives, [online] 13(11). https://doi.org/10.18103/mra.v13i11. 7019
Most read articles by the same author(s)
- Jodie A. Dashore, PhD, OTD, RH (AHG), Brian Dashore, DO, Scott McMahon, MD, Ritchie Shoemaker, MD, Chronic Inflammatory Response Syndrome: Exploring Neuroimmune Pathology and Multisystem Framework for Differential Diagnosis in Pediatrics- Part 1 , Medical Research Archives: Vol 13 No 10 (2025): Vol.13, Issue 10, October 2025
- Jodie A. Dashore, PhD, Brian Dashore, DO, Scott McMahon, MD, Ryan Shoemaker, MD, Autism Spectrum Disorders and Lyme Disease: Exploring Shared Neuro-Inflammatory and Immune Pathways , Medical Research Archives: Vol 13 No 11 (2025): Vol.13, Issue 11, November 2025
- Jodie A. Dashore, PhD, Brian Dashore, DO, Scott McMahon, MD, Ritchie Shoemaker, MD, Pediatric Neuroimmune and Inflammatory Overlap: Immunopathology of PANS/PANDAS in Autism Spectrum Disorder , Medical Research Archives: Vol 14 No 1 (2026): Vol.14, Issue 1, January 2026

