Histological Analysis of Autologous Dentin Graft Efficacy
Histoligical analysis of ridge augmentation with Autologus Dentin graft and Acellular Dermal matrix: A Descriptive Case Report
Dr. Lanka Mahesh, BDS, MBA, PhD1; Dr. Maurice Salama, DMD2; Dr. Sagrika Shukla3; Dr. Praful Bali, PhD4; Dr. Athreya Rajagopal, PhD5
- Implantologist, Director- The Specialist Clinic, Private practice, New Delhi, India.
- Private practice, Atlanta, Georgia, USA
- MDS (Periodontology), Reader, Dept of Periodontology and Implantology, SGT university, Budhera, Haryana, India
- MDS, Prosthodontist, Director, Bali Dental Clinic, New Delhi, India
- MDS, PhD, Reader, Oral and maxillofacial surgeon, Santosh, to be deemed University, Gaziabad
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Mahesh, L., Salama, M., et al., 2024. Histoligical analysis of ridge augmentation with Autologus Dentin graft and Acellular Dermal matrix: A Descriptive Case Report. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5820
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v12i10.5820
ISSN 2375-1924
ABSTRACT
Aim: to analyze autologus dentin graft and its effectiveness in regeneration histologically.
Material and method: Three implants were placed in mandibular anterior region, for which dentin graft was obtained via grade III mobile teeth 35 and 44, they were extracted, cleaned of all the remnants then grinded into particle size of 300-1200 μm in a sterile grinding chamber. Autologous dentin graft was secured using membrane (Alloderm® GBR membrane, Biohorizons, Inc) with auto tracks. At 4 months during second stage, a bone core was obtained for histological examination using 3.2 mm trephine, the core site was then grafted with beta-TCP and was sutured, following which implant prosthesis was given to the patient.
Results: The surgical procedure and the healing was uneventful and patient did not show any discomfort. Histologically, at 4 months, bone grafted section shows the presence of dentin graft along with formation of new mature bone at the junction and vicinity of graft material.
Conclusion: autologous dentin graft shows promising results as a biocompatible regenerative material with low resorption rate and can be used as a substitute for autogenous bone grafts and synthetic bone grafts.
Keywords: Bone graft, Dentin graft, Regeneration, Membrane
INTRODUCTION
Implant placement has become a desirable option for missing teeth in the current era. However, the remodeling process that occurs in the alveolar bone after teeth extraction possess potential challenge to place dental implants in a restoratively driven position. In order to enhance ridge volume and dimensions, suitable enough for implant placement, lateral bone augmentation is considered as an effective treatment protocol prior to implant placement, which can be engineered through a variety of grafts materials i.e autogenous, allografts, xenografts, alloplastic materials along with membranes. A bone graft substitute should have bimodal behavior supporting both proliferation and differentiation which requires a permeable structure i.e of macropores, micropores, and nanopores. One such bone graft substitute is dentin graft which though being discovered nearly 50 years ago by Urist et al, has gained tremendous popularity recently. Its osteoinductive properties and structural and physicochemical characteristics being similar to those of alveolar bone make it one of the ideal bone graft to be used for bone regeneration.
Dentin graft supports osseous defect regeneration as it constitutes about 90% type I collagen and a similar organic content 35% vs 39% (bone) and inorganic content, 65% vs 61% (bone) along with various other proteins such as glycoproteins, proteoglycans and bone morphogenetic protein (BMP). Also, there are many proteins with are common to both dentin and bone such as, which are common to bone and dentin such as collagen types I, III, and V, bone sialoprotein (BSP), osteopontin (OPN), dentin matrix protein-1 (DMP-1), osteocalcin (OC), and osteonectin (ON). Another point to be considered is the fact that odontoblasts, which synthesized dentin matrix contain abundance of growth factors and contains bioactive molecules required for dentinogenesis. These molecules are released which help in regeneration and repair. released in the presence of bacterial acids or certain dental materials in the case of caries or restorative treatments, causing dentin regeneration and repair. Another important aspect is that in spite of having similar dentin matrix and bone proteins, dentin sialoproteins and dentin phosphoproteins are uniquely found in dentin which include transforming growth factor (TGF), fibroblast growth factor (FGF), insulin-like growth factor (IGFs), BMPs, epidermal growth factor (EGF), PDGF, placenta growth factor (PLGF), vascular endothelial growth factor (VEGF), and angiogenic growth factor (AGF) which when used as a bone graft aid in its regeneration. Aforementioned characteristics are important for the initial stages of bone regeneration, adhesion of osteoblasts and osteoclasts and the deposition of the osteoid. Using a predictable bio-product for regeneration and using a membrane for space maintenance are the two prime requisites and principles of guided bone regeneration (GBR). Using Alloderm® GBR is one such membrane which has been successfully used for regenerative procedures as it can maintain space to prevent ingrowth of soft tissue. Even then, to call regeneration predictable, it becomes paramount to view it histologically. Hence, this study was conducted to analyse regeneration when a dentin graft is being used.
MATERIAL AND METHOD
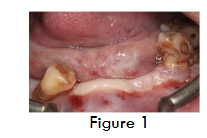
A male patient aged 46yrs visited the dental office with a chief complaint of missing teeth and wanted to get them replaced. On intra oral examination (figure 1), there was grade III mobility in relation to (i.r.t) 35, 44. On radiographic examination, alveolar ridge defect was observed in the mandibular anterior region henceforth implant placement with simultaneous ridge augmentation with autogenous tooth grafts was planned to restore the edentulous arch with implant supported prosthesis, to which the patient readily agreed. On Further examinations such as blood and systematic, no abnormality was detected. A consent was taken from the patient for the same.
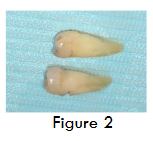
Before commencing with the surgical procedure, Grade III mobile teeth 35 and 44 were extracted. Remnants of Periodontal ligament (PDL) and calculus are removed by tungsten bur (Figure 2). Whole Cleaned teeth are then dried by air syringe, and placed in a sterile grinding chamber of Smart Dentin GrinderTM (figure 3). This device is capable of grinding the roots in 3 secs, furthermore, via vibrating movement of the grinding chamber, particles of less than 1200 μm can be obtained in 20 secs. Particles between 300-1200 μm, suitable for regeneration fall through a sieve to a lower chamber, after which they are immersed in basic alcohol (figure 4) for 10 minutes, in a small sterile glass container. Basic alcohol cleanser consists of 0.5M of NaOH and 30% alcohol (v/v), for defatting, dissolving all organic debris, bacteria and toxins of the dentin particulate. This wet particulate dentin is ready to graft into alveolar bone defects.
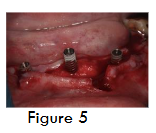
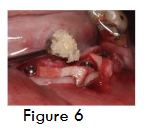
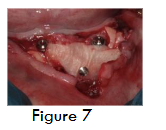
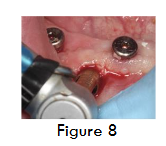
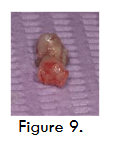
For implant placement, horizontal crestal incision and two vertical incisions were given and full thickness mucoperiosteal flap was elevated. Three Bioner top Dm implants of dimension 4/10 were placed along with cover screws (figure 5). Dentin graft was placed (figure 6), followed Alloderm® GBR membrane (Biohorizons, Inc) which was trimmed, rehydrated in a petri dish with sterile saline solution and secured in position using stabilizing screws (auto tracks) (figure 7) to prevent micromobility. Primary closure is obtained using 3-0 sutures. At 4 months during second stage, a bone core was obtained for histological examination using 3.2 mm trephine (figure 8 and 9), the core site was then grafted with beta-TCP and was sutured, following which implant prosthesis was given to the patient.
RESULTS
Clinically, the results were satisfactory, patient did complain of mild discomfort due to the surgical procedure. On follow-up visit of the patient at 7 days there was no swelling or pain and patient had kept the surgical area free of debris. On radiographic examination at 4 months, implants had integrated well and there was no sign of infection or discomfort to the patient. Clinically there was well keratinized soft tissue all around the implants (figure 8).
HISTOLOGICAL EVALUATION
3.2 mm trephine core biopsies (figures 9) were obtained and fixed immediately in neutral buffered formalin solution for 24-48 hours. The specimens were processed after decalcifying in mild decalcifying agent (10% EDTA, pH 7.4). The tissues were processed using standard tissue processing laboratory protocol of dehydration, clearing and infiltration with paraffin wax. Embedding and tissue block preparation was done with paraffin wax. 4 micron thick sections were stained with Hematoxylin and Eosin stains. The slides thus obtained were viewed in research microscope (Olympus BX53) and digital images were captured in low and high magnification (Olympus EPL3).
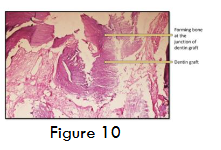
Submitted H and E-stained section shows (in the order from top to bottom) the presence of Gingival tissue overlying Alloderm® which is further underlined by Dentin Graft Material and underlying forming mature bone (figure 10).
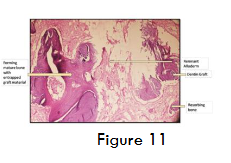
Submitted H and E-stained section shows the presence of dentin graft (figure 11) along with the formation of new mature bone at the junction and vicinity of graft material. Bone shows lamellar arrangement along with entrapped osteocytes and lined by plump osteoblasts. Section shows minimal inflammatory infiltrate indicating uptake of graft material in the absence of inflammatory reaction. Minimal blood vessels indicating healing and process of fibrosis taking place.


DISCUSSION
Autogenous bone graft still remains gold standard for the process of bone augmentation as it is characterized by most effective osteogenic, osteoconductive, osteoinductive and immunogenic properties. However, limited quantity and a second surgical site has led clinicians to use alternatives of autogenous bone which can provide with similar qualities. Autologous dentin graft upto some extent has solved quality (osteoinductive and osteoconductive) concerns as the graft not only shows same effects as autologous bone, it also presents an inert and strong scaffold of dense HA. Dentin graft is a novel graft consisting of four types of calcium phosphate crystals, including hydroxyapatite, tricalcium phosphate, octacalcium phosphate, and amorphous calcium phosphate, which provide a dissolution pattern similar to an autogenous cortical bone. In addition to its osteoinduction, osteoconduction and progressive substitution capabilities, tooth grafts gives a particle size ranging from 300-1200 μm, which is apt for regeneration. There is no chance of cross-infection or problem where some patients refuse allografts and xenografts on the basis of their origin or cases where the osteogenic potential depends upon batches as seen in allografts, also it is cost-effective. The particulate tooth material provides excellent biocompatibility with an absence of antigenicity.
As a result of aforementioned properties and advantages of autogenous tooth/dentin graft many authors and clinicians have started using it for GBR. Jeong et al reported average bone formation of 43.74% after 4 weeks after using autogenous tooth bone (ATB) graft of a miniature pig and concluded that it’s a good substitute for autogenous bone graft. Authors, Kim et al used ATB from humans in guided bone regeneration (GBR) and Lee et al in sinus augmentation, and took the tissue specimen 2 months and 4 months later for histomorphometric analysis. Both the studies found favourable new bone formation and suggested that ATB graft can be used in various bone grafting for regeneration. In a clinical study by Cardaropoli et al, a re-entry at 24 weeks revealed that the graft was incorporated and formed new bone in the extraction socket. The ridge volume was preserved, allowing for successful implant placement with good primary stability and without the need for additional bone augmentation.
Autogenous tooth/dentin graft also results in a significantly higher gain in ridge dimensions (horizontally and vertically) than autogenous bone grafts mainly due to less resorption. Stumbras et al, in a systematic review analyzed the evidence based alveolar ridge preservation techniques and stated that one of the biomaterials which reduces vertical resorption the most is autogenous tooth graft: -0.28 (0.13) mm over an observation period of 4 months. The biological potential of ATB grafts was also confirmed by immunohistochemical (i.e osteocalcin antigen reactivity, biomechanical (i.e removal torque) and microcomputed tomographic (i.e bone volume / tissue volume). As aforementioned, due to presence of growth factors which are slowly released, tooth derived is involved in bone remodeling and equally supports osteointegration of Ti-implants when compared with autogenous cortical bone blocks which has also been histologically confirmed.
When it comes to GBR, the concept talks about exclusion of undesired cells for proliferation of bone cells to guide them towards regeneration and establishing the missing structure for function and aesthetics, having said so, placing a membrane is paramount which acts as a space maintainer. For the same purpose, Alloderm® GBR was used, which is a collagen-based membrane prepared from dermis processed to remove its antigenicity and cellularity via cryoprecipitate. Structurally speaking, it is similar to the alloderm used for recession coverage, hence alloderm® GBR also has a connective tissue and basement membrane side, however the orientation is unimportant. Since it is used for GBR, it varies in thickness and functions as an effective barrier membrane. In this study, keeping anatomy into consideration, connective tissue side was placed facing the flap surface. Also, the results of this study are in accordance with aforementioned researchers, by 4 months, formation of new mature bone at the junction and vicinity of graft material could be seen, along with entrapped osteocytes and lined by plump osteoblasts. Also, minimal inflammatory infiltrate has been observed indicating good uptake of the graft material.
CONCLUSION
Autogenous graft shows promising results, with structures similar to bone. advantage of tooth used as a graft is its protein content which allows osteoinduction and autogenous collagenated hydroxyapatite with type I collagen which allows osteoconduction and excellent biocompatibility, absence of antigenicity and progressive substitution capabilities. however, in a systematic review, authors stated that dentin graft showed an increased risk of dehiscence, particularly in block grafted sites ranging from 12.96% to 34.38%. Hence, long-term study with large number of surgical sites treated with ATB is needed to scientifically prove predictability of dentin graft. However, the current data available does not state any quality or safety risks, nor it questions its regenerative potential. Hence it can be used in cases which require regeneration, provided the tooth used is not infected, and if the quantity is limited it can be mixed with other graft materials such as a xenograft to resolve resorption issues.
Source(s) of support: None
Acknowledgement: None
Conflict of interest: None
Contributions of all authors:
- Dr. Lanka Mahesh – Conceptualization
- Dr. Maurice Salama – Data curation
- Dr. Sagrika Shukla – Formal analysis
- Dr. Praful Bali – Funding acquisition
- Dr. Athreya Rajagopal – Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing
REFERENCES
- Mahesh L, Calvo Guirado JL, Shukla S. et al. Clinical and radiographic findings without the use of bone substitute materials in extraction sockets and delayed implant placement- A case series. J Oral Biol Craniofac Res. 2020;10(2):141-145. doi: 10.1016/j.jobcr.2020.03.011.
- Schwarz F, Hazar D, Becker K. et al. Short-term outcomes of staged lateral alveolar ridge augmentation using autogenous tooth roots. A prospective controlled clinical study. J Clin Periodontol. 2019;46(9):969-976. doi: 10.1111/jcpe.13161.
- Calvo- Guirado JL, Montilla AB, De Aza P. et al. Particulated, Extracted Human Teeth Characterization by SEM- EDX Evaluation as a Biomaterial for socket preservation: An in vitro study. Materials 2019;12:380.
- Yeomans JD, Urist MR. Bone Induction by Decalcified Dentine Implanted into Oral, Osseous and Muscle Tissues. Arch Oral Biol. 1967;12:999–1008. doi: 10.1016/0003-9969(67)90095-7.
- Bang G, Urist MR. Bone Induction in Excavation Chambers in Matrix of Decalcified Dentin. Arch Surg 1967;94:781–789. doi: 10.1001/archsurg.1967.01330120035008
- Liu Y, Yu L, Zhang D. et al. Manufacture and preliminary evaluation of acellular tooth roots as allografts for alveolar ridge augmentation. J Biomed Master Res A 2022;110(1):122-130.doi.org/10.1002/jbm.a.37270.
- Schwarz F, Mihatovic I, Popal-Jensen I. et al. Influence of autoclavation on the efficacy of extracted tooth roots used for vertical alveolar ridge augmentation. J Clin Periodontol. 2019;46(4):502-509. doi: 10.1111/jcpe.13090.
- kim YK. Bone graft material using teeth. J korean Assoc. Oral maxillofacial surgery. 2012;38:134-138.
- Butler WT, Ritchie H. The Nature and Functional Significance of Dentin Extracellular Matrix Proteins. Int J Dev Biol 1995;39:169–179.
- Schmidt-Schultz TH, Schultz M. Intact Growth Factors Are Conserved in the Extracellular Matrix of Ancient Human Bone and Teeth: A Storehouse for the Study of Human Evolution in Health and Disease. Biol Chem. 2005;386:767–776. doi: 10.1515/BC.2005.090.
- Finkelman RD, Mohan S, Jennings JC. Et al. Quantitation of Growth Factors IGF-I, SGF/IGF-II, and TGF-Beta in Human Dentin. J Bone Miner Res. 1990;5:717–723. doi: 10.1002/jbmr.5650050708.
- Kanazirski N, Kanazirska P. Auto-Tooth Bone Graft Material for Reconstruction of Bone Defects in the Oral Region: Case Reports. Folia Med. 2022;64:162–168. doi:0.3897/folmed.64.e59099.
- Minetti E, Palermo A, Malcangi G. et al. Dentin, Dentin Graft, and Bone Graft: Microscopic and Spectroscopic Analysis. J Funct Biomater 2023;14:272. https://doi.org/10.3390/jfb14050272.
- kim YK, Kim SG, Oh JS. et al. Analysis of the inorganic component of autogenous tooth bone graft material. J Nanosci Nanotechnol 2011;11:7442-7445. doi: 10.1166/jnn.2011.4857.
- Bindeman I, Hallel G, Nardy C. et al. A Novel Procedure to Process Extracted Teeth for Immediate Grafting of Autogenous Dentin. J Interdiscipl Med Dent Sci 2014;2(6):56-63 DOI: 10.4172/2376-032X.1000154.
- Kim YK, Lee J, Kim KWB. et al. Analysis of organic components and osteoinductivity in autogenous tooth bone graft material. J Korean Assoc Maxillofac Plast Reconstr Surg 2013;35:353-59. DOI.org/10.14402/jkamprs.2013.35.6.353.
- Lanka M, Calvo- Guirado JL, Shukla S. et al. Histological study to compare the appropriate particle size of beta tcp for socket preservation. INT J SCI RES 2023;12(2):1-4. DOI: 10.36106/ijsr/3503764.
- Jeong HR, Hwang JH, Lee JK. Effectiveness of autogenous tooth bone used as a graft material for regeneration of bone in miniature pig. J Korean Assoc Oral Maxillofac Surg. 2011;37:375–379. DOI:org/10.5125/jkaoms.2011.37.5.375.
- Kim SG, Kim YK, Lim SC. et al. Histomorphometric analysis of bone graft using autogenous tooth bone graft. Implantology. 2011;15:134–141. Doi.org/10.1016/j.0000.2012.04.018
- Lee JY, Kim YK, Kim SG.et al. Histomorphometric study of sinus bone graft using various graft material. J Dent Rehabil Appl Sci. 2011;27:141–147.
- Cardaropoli D, Nevins M, Schupbach P. New Bone Formation Using an Extracted Tooth as a Biomaterial: A Case Report with Histologic Evidence. Int J Perio Rest Dent. 2019 Mar/Apr;39(2):157-163. doi: 10.11607/prd.4045.
- Stumbras A, Kuliesius P, Januzis G. et al. Alveolar Ridge Preservation after Tooth Extraction Using Different Bone Graft Materials and Autologous Platelet Concentrates: a Systematic Review . J Oral Maxillofac Res 2019;10(1):1-14. doi: 10.5037/jomr.2019.10102.
- Schwarz F, Golubovic V, Mihatovic I. et al. Periodontally diseased tooth roots used for lateral alveolar ridge augmentation. A proof-of-concept study. J Clin Periodontol 2016;43(9):797-803. doi: 10.1111/jcpe.12579.
- Schwarz F, Hazar D, Becker K. et al Efficacy of autogenous tooth roots for lateral alveolar ridge augmentation and staged implant placement. A prospective controlled clinical study. J Clin Periodontol 2018;45(8):996-1004.
- Sabitha S, Arun KV, Priya MS. et al. Clinical and histological evaluation of alloderm GBR and BioOss in the treatment of Siebert’s class I ridge deficiency. J Indian Soc Periodontol 2008;12(3):73-78. DOI: 10.4103/0972-124X.44099
- Gharpure AS, Bhatavadekar NB. Clinical Efficacy of Tooth-Bone Graft: A Systematic Review and Risk of Bias Analysis of Randomized Control Trials and Observational Studies. Implant Dent. 2018;27(1):119-134. doi: 10.1097/ID.0000000000000687.

