HPV Vaccine Education for Medical Assistants: Impact Study
Human Papillomavirus Vaccine Education for Medical Assistants as a Strategy to Improve Vaccination Rates
Karlen E. Luthy¹, Alexis G. Peterson¹, Janelle L. B. Macintosh¹, Lacey M. Eden², Michael S. Robinson¹
- Brigham Young University College of Nursing
- Families First Pediatrics
OPEN ACCESS
PUBLISHED: 31 May 2025
CITATION: Luthy, KE., et al., 2025. Human Papillomavirus Vaccine Education for Medical Assistants as a Strategy to Improve Vaccination Rates in 9-year-old Children. Medical Research Archives, [online] 13(5).
https://doi.org/10.18103/mra.v1
315.4644
COPYRIGHT © 2025 European Society of Medicine. This is an open- access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v1 315.4644
ISSN 2375-1924
ABSTRACT
Background: The Human Papillomavirus vaccine protects patients against Human Papillomavirus-related infection and cancers, yet it is underutilized. The American Academy of Pediatrics and the American Cancer Society now recommend starting the vaccine series at age 9 years. Medical Assistants can play a pivotal role in identifying and facilitating Human Papillomavirus vaccination in 9-year-old children and are the focus of this project.
Aim Statement: This project aims to examine whether one educational session can improve the perceptions, knowledge, and comfort level of Medical Assistants regarding Human Papillomavirus vaccination, thus promoting the vaccination in the pediatric clinic setting, especially with 9-year-old patients.
Methods: The project took place at three separate clinics of one pediatric organization in Utah.
Interventions: A 10–15-minute educational session about the Human Papillomavirus vaccine was presented to the Medical Assistants at each clinic, and a pre- and post-survey method was used to analyze the effect of the education on perceptions, knowledge, and comfort level in vaccinating 9-year-old children.
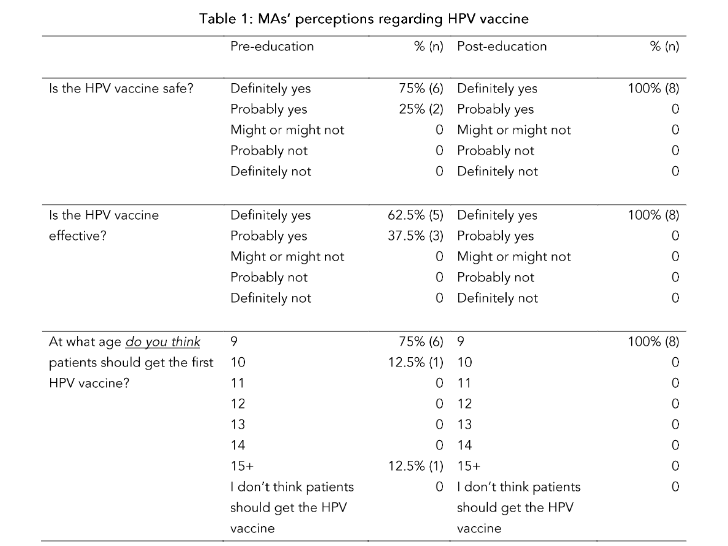
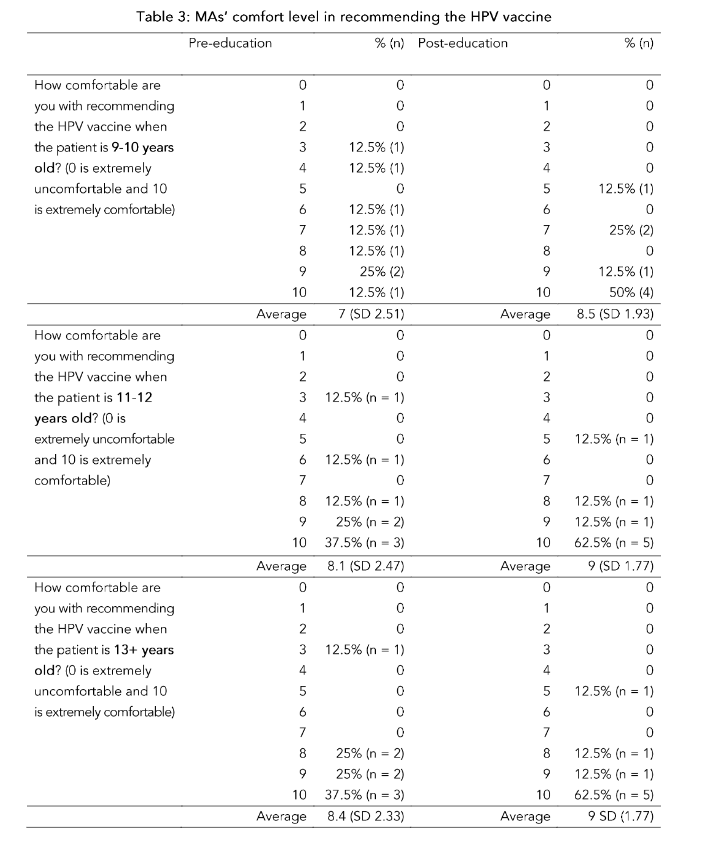
Results: Post-education data showed improvements in Medical Assistants’ perceptions, knowledge, and comfort levels regarding the Human Papillomavirus vaccine. Comfort level of initiating the vaccine series at age 9 increased from 75% pre-survey to 100% post-survey. Pre-survey, 62.5% of Medical Assistants believed the Human Papillomavirus vaccine was effective; post-survey 100% of Medical Assistants believed the Human Papillomavirus vaccine was effective. When comparing pre- and post-survey results, respectively, 75% of Medical Assistants believed the Human Papillomavirus vaccine was safe compared to 100%.
Conclusions: An educational intervention on Human Papillomavirus vaccine improves perceptions, increases knowledge, and boosts comfort levels for Medical Assistants recommending the vaccine in the pediatric clinic setting.
Introduction
Vaccines save millions of lives annually by protecting individuals against more than 20 preventable diseases¹. Along with saving lives, vaccines also decrease the occurrence of long-lasting and potentially devastating effects associated with vaccine-preventable diseases². Vaccines also protect individuals and families from illnesses requiring isolation or quarantine, thus minimizing absenteeism and protecting the employees’ source of income³.
The first human papillomavirus (HPV) vaccine received approval from the Food and Drug Administration for use in 2006⁴. After almost two decades of data, the vaccine is considered highly effective in decreasing the prevalence of HPV infections and, consequently, HPV-associated cancers⁵. Since the introduction of the HPV vaccine, there has been an 88% reduction in HPV infections among adolescent girls and an 81% reduction in HPV infections among young adult women⁶.
As of 2025, the Centers for Disease Control and Prevention routinely recommends initiating the HPV series at age 11–12 years, although the HPV vaccine can be given as early as age 9 years⁷. However, the American Academy of Pediatrics, the National HPV Vaccination Roundtable, and the American Cancer Society (ACS) now recommend routine vaccination with HPV starting at age 9 years⁸. However, only 21% of primary care providers routinely recommend HPV vaccine as early as age 9 years⁹. Most (59%) of primary care providers do not recommend HPV vaccination until the child is at least 11-years-old⁹.
Since the HPV vaccine is safe and effective, it is essential for all healthcare team members, including medical assistants (MAs), to promote HPV vaccination. Medical Assistant responsibilities include providing information about and administering medications, such as vaccinations, to patients under the direction of other healthcare providers (HCPs)¹⁰. Because MAs may be among the first to discuss vaccinations with patients, they must be well-educated and prepared to answer vaccine-related questions. However, vaccine-related patient education training is not included in the core curriculum for MAs¹¹. Hence, MAs may require additional training to build knowledge and confidence in recommending vaccines to patients¹².
According to the President’s Cancer Panel¹³, reducing missed opportunities for HPV vaccination and providing HPV vaccine education are two essential strategies to increase HPV vaccination rates. When properly trained, MAs can improve vaccination rates by identifying eligible patients and providing vaccine education for parents, thus preventing missed opportunities¹⁴. Therefore, this project aims to provide HPV vaccine education to MAs to influence perceptions, increase knowledge, and boost comfort levels when recommending HPV vaccines to parents, especially in the 9-year-old patient population.
PROBLEM DESCRIPTION
In 2020, the Office of Disease Prevention and Health Promotion¹⁵ published a new Healthy People 2030 goal to increase HPV vaccination rates from 48% to 80%. However, in the last 3 years, HPV vaccination rates have only increased by about 10%¹⁶. While there is progress toward reaching the 80% HPV vaccination goal by 2030, achieving the goal may require additional evidence-based programs that are replicable and sustainable¹⁷.
Reasons vaccine-hesitant parents refuse or delay HPV vaccination should be considered. According to the National Immunization Survey-Teen in 2019, these concerns include 1) uncertainty regarding the safety of HPV vaccination; 2) lack of knowledge about the vaccine/belief that the vaccine is unnecessary; and 3) lack of HCP recommendation¹⁸.
One prominent concern of HPV vaccine-hesitant parents is whether or not the HPV vaccination is safe. Sonawane et al.¹⁹ studied almost 40,000 unvaccinated adolescents and found that parents were commonly concerned about the safety of the HPV vaccine. Among the 40,000 unvaccinated
Discussion
In this project, a single educational session of 10–15 minutes improved MAs’ scores, quantifying their HPV vaccine perceptions and knowledge. The MAs’ comfort level also improved across all patient age groups (9–10, 11–12, and 13+). One specific strength of this project is that it specifically studied MAs instead of solely focusing on HCPs, providing unique insights into a vital healthcare team member who is heavily involved in vaccination administration.
Educating MAs on the HPV vaccine may ultimately and positively influence vaccine uptake. Parents who question the HPV vaccine may ask MAs for information since the MA is often the first point of contact in the clinic setting. There is a lack of HPV vaccine confidence among some healthcare professionals⁴⁰; thus, education that improves the perceptions of HPV vaccine safety and efficacy may positively influence the strength of the HPV vaccination recommendation⁴¹. In this project, the MAs’ perceptions regarding HPV vaccine safety and efficacy improved after receiving only one 10–15-minute HPV vaccine education presentation.
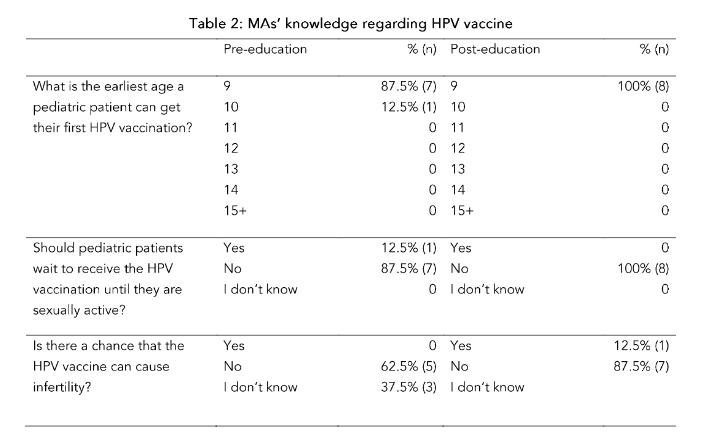
Despite evidence proving the safety of the HPV vaccine⁴⁰, the misconception that the HPV vaccine causes infertility remains²¹,⁴³. Due to insufficient research on this specific misconception, it is unknown if it can be effectively disproved through education alone. In this project, the majority of MAs understood post-education that the HPV vaccine does not cause infertility. There was, however, one post-education participant who still reported infertility as a potential side effect of the HPV vaccine. Due to the small sample size, however, it is unclear if this finding indicates that infertility is a stubborn misconception or if the participant misunderstood the question.
After the educational intervention, the greatest increase in comfort levels was seen in the 9–10-year-old age group, with an increase of 1.5 points (out of 10). Although the increase in vaccine comfort level is noteworthy, a few MAs were still less comfortable than one may expect after receiving HPV vaccine education. Interestingly, similar results were seen in a pre- and post-education study among medical students. After receiving HPV vaccine education, the medical students were asked if the HPV vaccine caused more benefits than harm. Again, a few medical students were not convinced that the HPV vaccine’s benefits outweighed the risk of harm⁴⁴. Given that the MAs and the medical students had one educational session, all participants’ confidence in HPV vaccine benefits could be improved with a series of HPV vaccine education sessions.
The results of this project show that MAs employed at the pediatric clinics in Salt Lake County, Utah, already had a foundational understanding of the HPV vaccine before receiving the HPV vaccine education. These findings, however, are unusual when compared to the literature. For example, vaccine acceptance and support seem to be related to the healthcare workers’ years of education. Thus, physicians are typically the strongest proponents of vaccines⁴⁵. Still, this project showed that MAs’ perceptions, knowledge, and comfort levels regarding the HPV vaccine can be improved with only one educational session.
LIMITATIONS
Like all projects, there are limitations. In this project, only one pediatric group with three locations was included. Therefore, the success of this project may not be generalizable to other MAs working with different pediatric populations. Furthermore, while all eight of the MAs participated in the education, the sample size was very small. However, the project could easily be replicated in other pediatric groups with a larger sample. The training and pre- and post-education questionnaires were created by a group of vaccination experts and HCPs to evaluate perceptions, knowledge, and comfort level of HPV vaccine for a specific group of MAs, but the training and the pre- and post-questionnaires could be easily adapted to fit the needs and goals of any pediatric practice. Additionally, only one training was held, so it is unknown whether the MAs’ perceptions, knowledge, and comfort levels regarding HPV vaccine would continue to improve with a series of trainings.
Conclusion
While the safety of the HPV vaccine is well established, it is still underutilized in the pediatric population, especially among 9–10-year-olds. Medical Assistants can play an essential role in improving HPV vaccine uptake and were, therefore, included in this project to determine whether one educational session could positively influence their perceptions, knowledge, and comfort levels. While the one educational session did not completely improve the comfort level of all the MAs, there was still considerable improvement. These findings are important to the medical community as they show that a relatively inexpensive and straightforward intervention can result in positive changes for MAs. In turn, MAs may be more likely to help increase HPV vaccination rates for pediatric patients.
Recommendations for future projects include incorporating a multi-pronged approach to increase HPV vaccination rates among 9-year-olds such as, including an HPV vaccine eligibility alert in the electronic health record, education for all members of the healthcare team in the clinic setting, and an in-clinic champion who consistently promotes the intervention over a specific amount of time, such as a year. Another recommendation is to deliver a series of educational seminars throughout the year to reinforce HPV education. A final recommendation is to increase the sample size or expand the intervention to include other clinics to verify reproducibility.
Acknowledgements:
The authors would like to thank the Myrtie Fulton Endowment Fund for funding this project. The authors would also like to thank Lacey M. Eden, DNP, FNP-c, for assistance with coordinating the project with the clinic locations.
References
1. World Health Organization. Vaccines and immunization. 2023 [cited 27 September 2024]. In: World Health Organization Website [Internet]. Available from:
https://www.who.int/health-topics/vaccines-and-immunization#tab=tab_1
2. American Academy of Pediatrics. 14 diseases you almost forgot about (thanks to vaccinApril 2021 [cited 3 December 2024]. In: Healthy Children Website [Internet]. Available from:
https://www.healthychildren.org/English/safety-prevention/immunizations/Pages/Vaccination-Protects-Against-These-Diseases.aspx
3. Centers for Disease Control and Prevention. Why CDC is involved in global immunization. 4 September 2024 [cited 3 December 2024]. In: Centers for Disease Control and Prevention Website. Available from:
https://www.cdc.gov/globalhealth/immunization/why/index.html
4. Kaiser Family Foundation. The HPV vaccine: Access and use in the U.S. 8 October 2024 [cited 3 December 2024]. In: Kaiser Family Foundation Website. Available from:
https://www.kff.org/womens-health-policy/fact-sheet/the-hpv-vaccine-access-and-use-in-the-u-s/
5. Centers for Disease Control and Prevention. HPV vaccination. 20 August 2024 [cited 3 December 2024]. In: Centers for Disease Control and Prevention Website. Available from:
https://www.cdc.gov/hpv/vaccines/index.html
6. Centers for Disease Control and Prevention. Reasons to get HPV vaccine. 3 July 2024 [cited 3 December 2024]. In: Centers for Disease Control and Prevention Website. Available from:
https://www.cdc.gov/hpv/vaccines/reasons-to-get.html?CDC_AAref_Val=https://www.cdc.gov/hpv/parents/vaccine/six-reasons.html
7. Centers for Disease Control and Prevention. Human papillomavirus (HPV) vaccine safety. 31 July 2024 [cited 3 December 2024]. In: Centers for Disease Control and Prevention Website. Available from:
https://www.cdc.gov/vaccine-safety/vaccines/hpv.html?CDC_AAref_Val=https://www.cdc.gov/vaccinesafety/vaccines/hpv-vaccine.html
8. American Cancer Society and the National HPV Vaccination Roundtable. N.D. [cited 1 May 2025]. In: HPV Roundtable Website. Available from: https://hpvroundtable.org/wp-content/uploads/2023/05/HPV_Roundtable-HPV_Why_Age_9_Sales_Sheet_WEB.pdf
9. Kong WY, Huang Q, Thompson P, Grabert BK, Brewer NT, Gilkey MB. Recommending HPV vaccination at age 9: A national survey of primary care professionals. Acad Pediatri. 2022; 22(4): 573-580. doi:10.1016/j.acap.2022.01.008
10. American Association of Medical Personnel. Registered medical assistant. 2024 [cited 3 December 2024]. In: American Association of Medical Personnel Website. Available from:
https://www.aamp.net/services/registered-medical-assistant.html
11. Commission on Accreditation of Allied Health Education Programs. Standards and guidelines for the accreditation of education programs in medical assisting. 4 April 2022 [cited 3 December 2024]. In: CAAHEP Website. Available from:
https://assets.website-files.com/5f466098572bfe97f28d59df/62f4194eb9b831023c2dd848_2022-03_MA_BoD%20Approved_FINAL%20STANDARDS%20w%20App%20A.pdf
12. Mills J, Van Winkle P, Shen M, Hong C, Hudson S. Physicians’, nurses’, and medical assistants’ perceptions of the human papillomavirus vaccine in a large integrated health care system. Perm J. 2016;20(4):15-205. doi:10.7812/TPP/15-205
13. President’s Cancer Panel. Accelerating HPV vaccine uptake: Urgency for action to prevent cancer. A report to the President of the United States from the President’s Cancer Panel. February 2014 [cited 3 December 2024]. In: National Cancer Institute. Available from:
https://deainfo.nci.nih.gov/Advisory/pcp/annualReports/HPV/PDF/PCP_Annual_Report_2012-2013.pdf
14. Centers for Disease Control and Prevention. Fostering a culture of immunization in your practice. 12 September 2024 [cited 3 December 2024]. In: Centers for Disease Control and Prevention Website. Available from:
https://www.cdc.gov/immunization-training/hcp/courses/index.html#cdc_training_materials_section_4-fostering-a-culture-of-immunization-in-your-practice
15. Office of Disease Prevention and Health Promotion. Increase the proportion of adolescents who get recommended doses of the HPV vaccine – IID-08. Data. August 2020 [cited 3 December 2024]. In: Healthy People 2030. Available from:
https://odphp.health.gov/healthypeople/objectives-and-data/browse-objectives/vaccination/increase-proportion-adolescents-who-get-recommended-doses-hpv-vaccine-iid-08/data
16. Office of Disease Prevention and Health Promotion. Increase the proportion of adolescents who get recommended doses of the HPV vaccine – IID-08. August 2020 [cited 3 December 2024]. In: Healthy People 2030. Available from:
https://health.gov/healthypeople/objectives-and-data/browse-objectives/vaccination/increase-proportion-adolescents-who-get-recommended-doses-hpv-vaccine-iid-08
17. Office of Disease Prevention and Health Promotion. Healthy People 2030 framework. August 2020 [cited 11 December 2024]. In: Healthy People 2030. Available from:
https://health.gov/healthypeople/about/healthy-people-2030-framework
18. Rositch AF, Liu T, Chao C, Moran M, Beavis AL. Levels of parental human papillomavirus vaccine hesitancy and their reasons for not intending to vaccinate: Insights from the 2019 National Immunization Survey-Teen. J Adolesc Health. 2022; 71(1):39-46. doi:10.1016/j.jadohealth.2022.01.223
19. Sonawane K, Lin YY, Damgacioglu H, et al. Trends in human papillomavirus vaccine safety concerns and adverse event reporting in the United States. JAMA Netw Open. 2021;4(9).
doi:10.1001/jamanetworkopen.2021.24502
20. Beavis A, Krakow M, Levinson K, Rositch AF. Reasons for lack of HPV vaccine initiation in NIS-Teen over time: Shifting the focus from gender and sexuality to necessity and safety. J Adolesc Health. 2018;63(5):652-656. doi:10.1016/j.jadohea lth.2018.06.024
21. Marshall S, Fleming A, Moore AC, Sahm LJ. Views of parents regarding human papillomavirus vaccination: A systematic review and meta-ethnographic synthesis of qualitative literature. Res Social Adm Pharm. 2019;15(4):331-337. doi:10.101 6/j.sapharm.2018.05.013
22. Kepka D, Bodson J, Lai D, et al. Diverse caregivers’ HPV vaccine-related awareness and knowledge. Ethn Health. 2021;26(6):811-826. doi:10.1 080/13557858.2018.1562052
23. Adjei Boakye E, Nair M, Abouelella DK, et al. Trends in reasons for human papillomavirus vaccine hesitancy: 2010-2020. Pediatrics. 2023;151(6). doi:10.1542/peds.2022-060410
24. Lu PJ, Yankey D, Fredua B, et al. Association of provider recommendation and human papillomavirus vaccination initiation among male adolescents aged 13-17 years-United States. J Pediatr. 2019;206:33-41.e1. doi:10.1016/j.jpeds.2 018.10.034
25. Kong WY, Bustamante G, Pallotto IK, et al. Disparities in healthcare providers’ recommendation of HPV vaccination for U.S. adolescents: A systematic review. Cancer Epidemiol Biomarkers Prev. 2021;30 (11):1981-1992. doi:10.1158/1055-9965.EPI-21-0733
26. Lake P, Fuzzell L, Brownstein NC, et al. HPV vaccine recommendations by age: A survey of providers in federally qualified health centers. Hum Vaccin Immunother. 2023;19(1):2181610. doi:10.10 80/21645515.2023.2181610
27. Rani U, Darabaner E, Seserman M, Bednarczyk RA, Shaw J. Public education interventions and uptake of human papillomavirus vaccine: A systematic review. J Public Health Manag Pract. 2022;28(1). doi:10.1097/PHH.0000000000001253
28. Dike SN, Cesario SK, Malecha A, Nurse R. An education intervention to increase human papillomavirus vaccination confidence and acceptability: A randomized controlled trial. Oncol Nurs Forum. 2023;50(4):423-436. doi:10.1188/2 3.ONF.423-436
29. Dixon BE, Zimet GD, Shan X, et al. An educational intervention to improve HPV vaccination: A cluster randomized trial. Pediatrics. 2019;143(1). doi:10.1542/peds.2018-1457
30. Beck A, Bianchi A, Showalter D. Evidence-based practice model to increase human papillomavirus vaccine uptake: A stepwise approach. Nurs Womens Health. 2021;25(6):430-436. doi:10.101 6/j.nwh.2021.09.006
31. Bernstein TA, Broome M, Millman J, Epstein J, Derouin A. Promoting strategies to increase HPV vaccination in the pediatric primary care setting. J Pediatr Health Care. 2022;36(2). doi:10.1016/j.pe dhc.2021.10.009
32. Centers for Disease Control and Prevention. HPV vaccination recommendations. 16 November 2021 [cited 11 December 2024]. In: Centers for Disease Control and Prevention Website. Available from: https://www.cdc.gov/vaccines/vpd/hpv/hcp/recommendations.html
33. Lazcano-Ponce E, Torres-Ibarra L, Cruz-Valdez A, et al. Persistence of immunity when using different human papillomavirus vaccination schedules and booster-dose effects 5 years after primary vaccination. J Infect Dis. 2018;219(1):41-49. doi:10.1 093/infdis/jiy465
34. Minihan AK, Bandi P, Star J, et al. The association of initiating HPV vaccination at ages 9-10 years and up-to-date status among adolescents ages 13-17 years, 2016-2020. Hum Vaccin Immunother. 2023;19(1):2175555. doi:10.1080/21 645515.2023.2175555
35. Troiano G, Nardi A. Vaccine hesitancy in the era of COVID-19. Public Health. 2021;194:245-251. doi:10.1016/j.puhe.2021.02.025
36. Peretti-Watel P, Larson HJ, Ward JK, Schulz WS, Verger P. Vaccine hesitancy: Clarifying a theoretical framework for an ambiguous notion. PLoS Curr. 2015;7. doi:10.1371/currents.outbreaks.6 844c80ff9f5b273f34c91f71b7fc289
37. Crawford R. Healthism and the medicalization of everyday life. Int J Health Serv. 1980;10(3):365-388. doi:10.2190/3H2H-3XJN-3KAY-G9NY
38. Johnson BB, Swedlow B. Cultural Theory’s contributions to risk analysis: A thematic review with directions and resources for further research. Risk Anal. 2021;41(3):429-455. doi:10.1111/risa.13 299
39. Taylor AC, Hopkins LW, Moore G. Increasing human papillomavirus immunization in the primary care setting. Nurse Pract. 2021;46(10):37-42. doi:10.1 097/01.NPR.0000790528.06533.66
40. Vu M, King AR, Jang HM, Bednarczyk RA. Practice-, provider- and patient-level facilitators of and barriers to HPV vaccine promotion and uptake in Georgia: A qualitative study of healthcare providers’ perspectives. Health Educ Res. 2020;35(6):512-523. doi:10.1093/her/cyaa026
41. Sackey ME, Markey K, Grealish A. Healthcare professional’s promotional strategies in improving human papillomavirus (HPV) vaccination uptake in adolescents: A systematic review. Vaccine. 2022;40 (19):2656-2666. doi:10.1016/j.vaccine.2022.03.054
42. Schmuhl NB, Mooney KE, Zhang X, et al. No association between HPV vaccination and infertility in U.S. females 18-33 years old. Vaccine. 2020;38(24): 4038-4043. doi:10.1016/j.vaccine.2020.03.035
43. Wubu A, Balta B, Cherie A, Bizuwork K. Perception about human papillomavirus vaccination among middle adolescent school girls in Addis Ababa, Ethiopia 2023: Qualitative study. BMC Womens Health. 2023;23(1):520. doi:10.1186/s12905-023-02660-1
44. Berenson AB, Hirth JM, Fuchs EL, Chang M, Rupp RE. An educational intervention to improve attitudes regarding HPV vaccination and comfort with counseling among US medical students. Hum Vaccin Immunother. 2020;16(5):1139-1144. doi:10.1 080/21645515.2019.1692558
45. Shekhar R, Sheikh AB, Upadhyay S, et al. COVID-19 vaccine acceptance among health care workers in the United States. Vaccines. 2021;9(2): 119. doi:10.3390/vaccines9020119




