Imaging Skin Lesions: CT, MRI, Ultrasound, and PET Insights
Review of Diagnostic Radiology Imaging Modalities for Evaluating Skin Lesions Involving the Head and Neck
Daniel Thomas Ginat, MD, MS1; Golnaz Lotfian, MD2
- Department of Neuroradiology, Pritzker School of Medicine, University of Chicago, Chicago, IL
- Department of Diagnostic Radiology and Nuclear Medicine, Rush University Medical Center, Chicago, USA.
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Ginat, DT., and Lotfian, G., 2024. Review of Diagnostic Radiology Imaging Modalities for Evaluating Skin Lesions Involving the Head and Neck. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5763
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v12i10.5763
ISSN 2375-1924
ABSTRACT
While skin lesions are apparent on clinical exam, diagnostic imaging is sometimes warranted to assess the local and distant extent of disease. CT imaging offers high-resolution anatomical detail and is particularly valuable in delineating deep-seated lesions, bone involvement, and assessing metastatic spread. MRI provides excellent soft tissue contrast and is indispensable for evaluating lesions in anatomically complex regions such as the head and neck. Ultrasound, on the other hand, offers real-time imaging, portability, and cost-effectiveness, making it an essential tool in the initial assessment and surveillance of superficial skin lesions. PET imaging, although less commonly employed for primary skin lesion evaluation, plays a crucial role in detecting metastatic disease and assessing treatment response in advanced-stage malignancies. Each modality has its advantages and limitations, and the choice of imaging technique depends on factors such as lesion characteristics, clinical indication, and availability of resources. Integration of multiple imaging modalities with clinical and histopathological correlation enhances diagnostic accuracy and facilitates optimal patient management. This review highlights the roles of CT, MRI, PET, and ultrasound in the comprehensive evaluation of skin lesions, with a focus on their respective indications, advantages, and utility in clinical practice.
Keywords: Skin; imaging; CT; MRI; PET; ultrasound
Introduction
Skin lesions are initially assessed by physical examination. Dermatologic exams can be supplemented by various noninvasive skin imaging techniques, such as skin surface imaging analysis systems, dermoscopy, reflectance confocal microscopy, high-frequency ultrasound, optical coherence tomography, and multiphoton tomography. These modalities can provide high resolution, but limited depth of visualization. For example, optical coherence tomography utilizes infrared broadband light to characterize skin architecture and abnormalities with a resolution between 15 and 3 micrometers, but only to a depth of 1 to 2 mm.
Diagnostic radiology imaging, which includes conventional ultrasound, CT, MRI, and PET, is indicated for soft tissue lesions that cannot be adequately assessed via dermatologic exams and cannot confirmed as benign. Indeed, diagnostic imaging can help identify features suggestive of malignancy, characterize the extent of disease, aid in biopsy planning, and provide important information for staging of cancers. Based on the ACR appropriateness criteria for the evaluation of a superficial soft tissue mass, the following approach can be considered for selecting diagnostic imaging modalities, but can vary based on various factors to individual preferences and specific skin diseases:
- Ultrasound is usually appropriate for the initial imaging.
- MRI without and with contrast is usually appropriate as the next imaging study following nondiagnostic ultrasound.
- When MRI is contraindicated, CT with contrast is usually appropriate as the next imaging study.
- PET is appropriate for staging certain malignancies.
The objective of this article is to review radiologic imaging modalities for evaluating skin lesions.
Computed Tomography
Computed tomography (CT) consists of cross-sectional scans created by combining X-ray images from different angles using computer processing. CT is especially helpful in assessing the degree to which skin lesions invade deeper tissues or organs. CT scans can accurately localize with a typical resolution of less than 1-2 mm, depending on the scan protocol. CT can quantitatively characterize the contents of lesions by measuring Hounsfield Units, which in turn can provide insights regarding the probable diagnosis. CT is especially useful for evaluating lesions that involve osseous structures, such as bone erosions from malignancy and infection, and for detecting calcifications. CT also excels at detecting the presence of gas and foreign bodies. Additionally, CT scans are relatively fast and accessible, which makes CT a convenient modality in emergency situations. Additionally, CT can be used to evaluate suspected deep extension of tumors and infections that may involve bone or soft tissues beyond what is visible in physical examination.
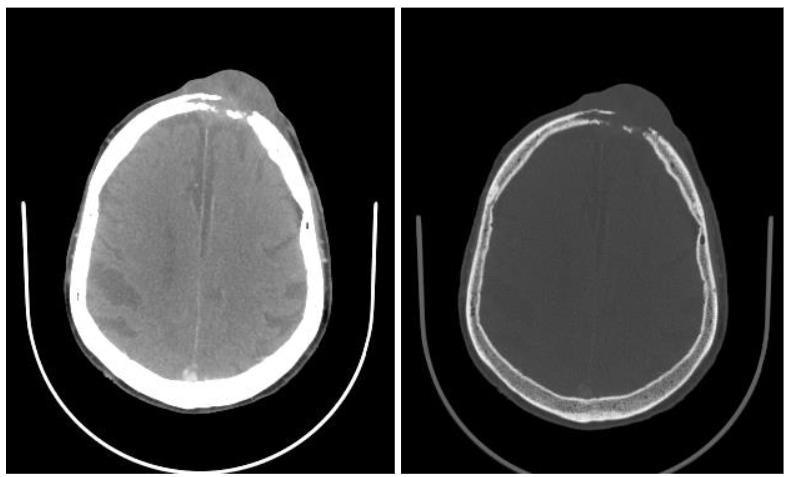
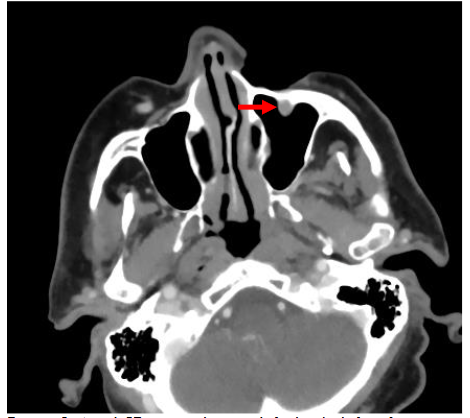
Magnetic Resonance Imaging (MRI)
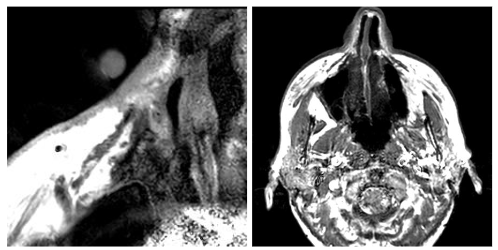
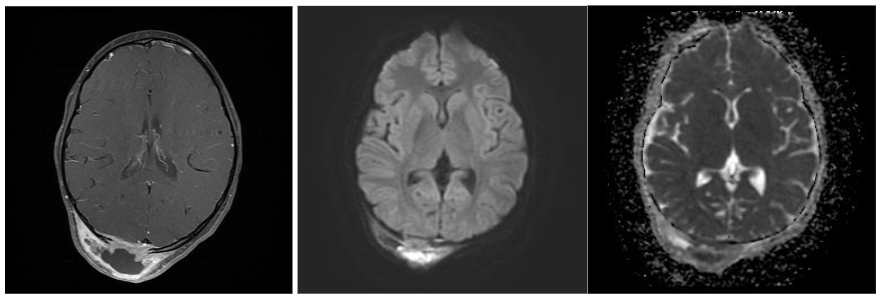
MRI scanners produce cross-sectional images in various planes and different sequences by adjusting the magnetic field and radio waves and provide superior soft tissue delineation. Through the use of several different sequences, MRI is particularly advantageous for characterizing the internal features and margins of lesions, which enhances the accuracy of distinguishing between benign and malignant conditions and informs more precise diagnosis and treatment planning. In particular, non-fatty malignant lesions tend to be ill-defined, display heterogeneous signal characteristics and low diffusivity, and are often larger. With regards to metastatic lymphadenopathy in the neck, CT is generally more sensitive, while MRI is more specific. Likewise, MRI has high diagnostic accuracy in depicting perineural spread from cancers such as squamous cell carcinoma and can be recognized by enlargement and abnormal enhancement of the affected nerve and effacement of any surrounding fat. MRI is also useful for evaluating intracranial metastases from skin cancers and the extent of disease in neurocutaneous syndromes. The use of intravenous contrast and fat suppression can be helpful for highlighting tumors. Furthermore, diffusion weighted imaging typically demonstrates restricted diffusion in pyogenic abscesses. Microscopy coils can provide higher resolution imaging to better delineate the margins of skin tumors and better define involvement of delicate underlying structures for surgical planning. This technique consists of applying small diameter surface coils over the region of interest. Using μMRI methods, it is possible to produce 3D images with an isotropic resolution of 50 μm and smaller.
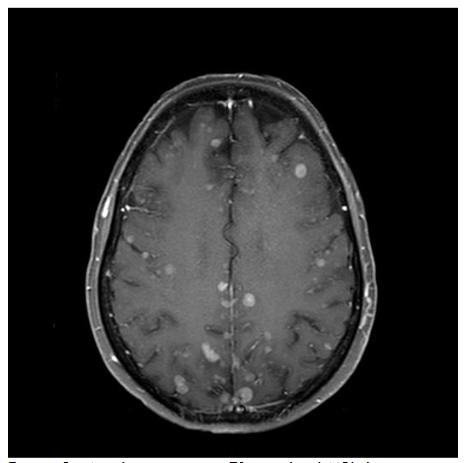
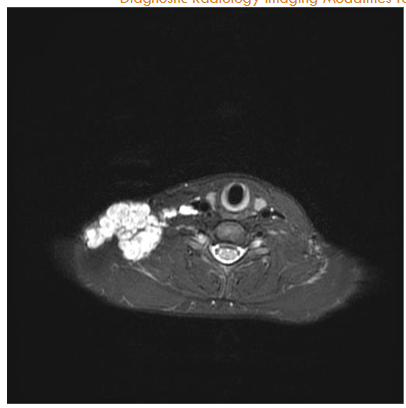
Ultrasound
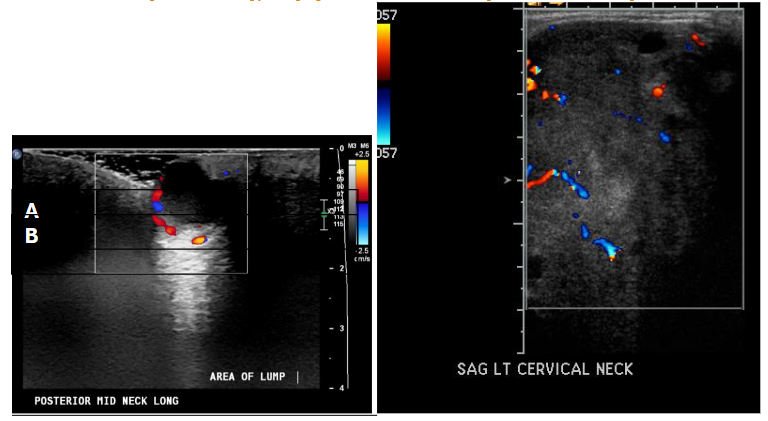
Ultrasound is a non-invasive imaging modality that creates real-time images of the body’s internal structures using high-frequency sound waves. Depending on frequency and characteristics of the transducer, ultrasound machines can produce lateral resolutions ranging from 0.1 to 1.0 mm, and axial resolution ranging from 0.1 to 0.5 mm. Furthermore, ultrasound can penetrate up to 5-10 cm deep. Lesions can be characterized as hyperechoic versus hypoechoic based on their contents, in which fat, nerves, bone, calcifications are hyperechoic, muscle is isoechoic, and fluid is hypoechoic. Furthermore, Doppler ultrasound can be useful for detecting blood flow and thus help differentiate fluid containing versus solid lesions.
Positron Emission Tomography (PET)
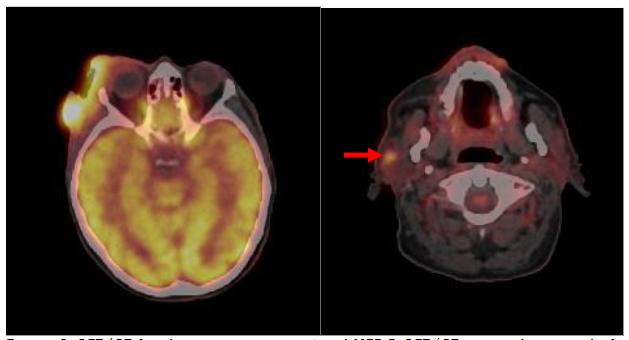
PET provides insights regarding the physiological and metabolic processes and often uses 18FDG, which is a positron-emitting radiotracer. These photons are detected by detectors encircling the patient’s body, allowing three-dimensional pictures to be reconstructed that emphasize high-tracer-uptake regions. PET imaging is used in many medical specialties, such as neurology, cardiology, and oncology, to monitor illness development, diagnose conditions, and evaluate treatment response. PET is an important tool in personalized medicine and clinical research because of its capacity to view molecular processes at the cellular level. PET imaging can be used for staging skin cancers and uses radiotracers, such as 18FDG. PET imaging can help detect additional sites of disease involvement beyond what is obvious on clinical examination or conventional imaging and therefore is useful for staging skin cancer patients. While Standard Update Values (SUV, quantitative measurement of metabolic activity) can vary based on the type of cancer, SUV values of > 2.5 are typically associated with malignancy. By detecting areas of increased metabolic activity, PET can aid in staging, treatment planning, and monitoring response to therapy in patients with cutaneous malignancies. While it can provide useful physiologic insights, PET has a relatively limited spatial resolution of about 4 to 6 mm, and it is not as specific as it is sensitive. Therefore, PET scans can be prone to false-positive and false-negative results for the evaluation of malignancy. In particular, infection and inflammation and sometimes normal physiologic activity can lead to increased FDG uptake, certain types of malignancy and small lesions might display low FDG uptake. To address these limitations, using complementary imaging modalities such as CT, MRI, and ultrasound can provide additional diagnostic information. Furthermore, a correlation with clinical and histological findings is essential for accurately interpreting PET results and ensuring a reliable diagnosis.
Conclusion
The assessment of skin lesions often requires a multimodal approach that incorporates CT, MRI, ultrasound, and PET imaging. In distinct elements of lesion characterization, localization, and staging, each modality has advantages of its own and complements the others. Ultrasound allows for the real-time assessment of superficial lesions, PET detects metabolic activity indicative of malignancy, MRI delivers greater soft tissue contrast, and CT imaging provides specific anatomical information. Through the utilization of various imaging modalities in conjunction with clinical and histological correlation, medical practitioners can accurately diagnose patients with cutaneous lesions, precisely stage their cases, and effectively plan their therapy. In order to significantly enhance diagnostic accuracy and patient outcomes in dermatologic oncology, future research efforts should concentrate on optimizing imaging protocols, developing image interpretation algorithms, and investigating novel imaging technologies. To maximize the use of imaging modalities in the thorough evaluation of skin lesions and eventually enhance patient care, radiologists, dermatologists, and oncologists must continue their collaboration.
Conflicts of Interest:
None
Funding:
None
References
- Ning X, Jiang L, Yu R, Sheng Y, Li M, Ouyang H, Xu J, Cui Y. Advances in the Application of Noninvasive Skin Imaging Techniques in Acne Scars. Am J Clin Dermatol. 2024 Sep;25(5):823-835.
- Sattler E, Kästle R, Welzel J. Optical coherence tomography in dermatology. J Biomed Opt. 2013 Jun;18(6):061224.
- Expert Panel on Musculoskeletal Imaging:; Kransdorf MJ, Murphey MD, Wessell DE, Cassidy RC, Czuczman GJ, Demertzis JL, Lenchik L, Motamedi K, Pierce JL, Sharma A, Walker EA, Ying-Kou Yung E, Beaman FD. ACR Appropriateness Criteria® Soft-Tissue Masses. J Am Coll Radiol. 2018 May;15(5S):S189-S197.
- Teixeira PA, Gay F, Chen B, Zins M, Sirveaux F, Felblinger J, Blum A. Diffusion-weighted magnetic resonance imaging for the initial characterization of non-fatty soft tissue tumors: correlation between T2 signal intensity and ADC values. Skeletal Radiol. 2016 Feb;45(2):263-71.
- Sun J, Li B, Li CJ, Li Y, Su F, Gao QH, Wu FL, Yu T, Wu L, Li LJ. Computed tomography versus magnetic resonance imaging for diagnosing cervical lymph node metastasis of head and neck cancer: a systematic review and meta-analysis. Onco Targets Ther. 2015 Jun 8;8:1291-313.
- Abdullaeva U, Pape B, Hirvonen J. Diagnostic Accuracy of MRI in Detecting the Perineural Spread of Head and Neck Tumors: A Systematic Review and Meta-Analysis. Diagnostics (Basel). 2024 Jan 4;14(1):113.
- Mukherji SK, Chenevert TL, Castillo M. Diffusion-weighted magnetic resonance imaging. J Neuroophthalmol. 2002 Jun;22(2):118-22.
- Nakayama M, Tabuchi K, Nakamura Y, Hara A. Basal Cell Carcinoma of the Head and Neck. Journal of Skin Cancer. 2011;2011:1-9.
- Fabiszewska E, Pasicz K, Grabska I, Skrzyński W, Ślusarczyk-Kacprzyk W, Bulski W. Evaluation of Imaging Parameters of Ultrasound Scanners: Baseline for Future Testing. Pol J Radiol. 2017 Dec 15;82:773-782.
- Mahajan S, Barker CA, Singh B, Pandit-Taskar N. Clinical Value of 18F-FDG-PET/CT in Staging Cutaneous Squamous Cell Carcinoma. Nuclear Medicine Communications. 2019;40(7):744-751.
- Moses WW. Fundamental Limits of Spatial Resolution in PET. Nucl Instrum Methods Phys Res A. 2011 Aug 21;648 Supplement 1:S236-S240.
- Yeh R, Amer A, Johnson JM, Ginat DT. Pearls and Pitfalls of 18FDG-PET Head and Neck Imaging. Neuroimaging Clin N Am. 2022 May;32(2):287-298.

