Immunotherapy Advances in Esophageal Cancer Treatment
Revolutionizing the Fight: How Immunotherapy is Transforming Esophageal Cancer
Mercedes Tamburelli 1, Javier Castillo 2*
- Gastrointestinal Oncology Unit, Clinical Oncology Service, Hospital Alemán, CABA, Argentina
- Head of the Gastrointestinal Oncology Unit, Oncology Service, Hospital Alemán, CABA, Argentina
*Email: [email protected]
OPEN ACCESS
PUBLISHED: 30 June 2025
CITATION: Tamburelli, M., and Castillo, J., 2025. Revolutionizing the Fight: How Immunotherapy is Transforming Esophageal Cancer. Medical Research Archives, [online] 13(6). https://doi.org/10.18103/mra.v13i6.6582
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i6.6582
ISSN 2375-1924
ABSTRACT
The treatment of esophageal cancer has evolved significantly over the past decade with the emergence of immunotherapy, particularly through the use of immune checkpoint inhibitors (ICIs) such as nivolumab and pembrolizumab. Although gastrointestinal cancers initially showed limited response, recent studies have demonstrated efficacy in selected subgroups, particularly in tumors with high PD-L1 expression or microsatellite instability (MSI-H).
Esophageal cancer consists of two main subtypes: squamous cell carcinoma (SCC) and adenocarcinoma (AC), each with distinct molecular, anatomical and epidemiological characteristic requiring tailored therapeutic approaches. Despite advances in multimodal therapy, overall survival remains low, especially in metastatic disease.
In localized disease, studies such as CROSS and CheckMate-577 have established the role of neoadjuvant chemoradiotherapy followed by surgery, with adjuvant nivolumab now considered standard of care for patients with residual disease. In advanced disease, trials such as KEYNOTE-590 and CHECKMATE-648 have shown survival benefits with the addition of immunotherapy to standard chemotherapy as first-line treatment.
PD-L1 remains a key biomarker for patient selection, although its clinical interpretation is influenced by biological and technical challenges. Furthermore, novel immunotherapeutic strategies involving antibodies targeting TIGIT, LAG-3, and CTLA-4 are currently under investigation.
In conclusion, immunotherapy has redefined the standard of care in esophageal cancer, offering improved outcomes in specific clinical contexts. However, standardization of biomarker evaluation and direct comparison trials are still needed to optimize therapy selection and patient benefit.
Keywords
Immunotherapy, Esophageal Cancer, PD-L1, Immune Checkpoint Inhibitors, Nivolumab, Pembrolizumab
Introduction
Esophageal Cancer (EC) is the eleventh most common malignancy worldwide by incidence with 511,054 new cases in 2022, representing 2.6% of all new cancer cases. Esophageal cancer accounts for 4.6% of cancer deaths in 2022, with 445,391 dying from this disease. However, today it is known that EC should not be considered and treated as a single entity. In fact, there are two subtypes of esophageal carcinoma, esophageal adenocarcinoma (AC) and esophageal squamous cell carcinoma (SCC), which account for most of the cases. Although SCC accounts for 80-90% of cases of oesophageal cancer worldwide, the incidence of AC is rising and has surpassed the incidence rate of SCC in several regions of Europe and North America, as well as certain high-risk areas of Asia, where this change was preceded by economic development and dietary changes.
Squamous cell carcinoma is mainly detected in the upper or middle esophagus, especially in patients with smoke and use alcohol in Eastern Europe or Asia. On the other side, esophageal adenocarcinoma is mainly located in the distal esophagus and is linked with obesity or with the presence of gastroesophageal reflux disease and Barrett’s esophagus. Adenocarcinoma is the most frequent histotype in Europe and North America. Recently, the Cancer Genome Atlas classification confirmed that adenocarcinoma and squamous cell carcinoma are two different entities also from a molecular point of view. Therefore, since adenocarcinoma and squamous cell carcinoma differ from histological, molecular, anatomical and histological points of view, we should consider a different therapeutic approach for each of them, also according to the stage of disease.
Nearly 40% of patients present with metastatic disease at diagnosis. Although survival rates have improved, the prognosis with cytotoxic chemotherapy remains poor, and those with metastatic disease have a five-year survival rate of less than 6%. These figures highlight the urgent need for more effective therapies to address this clinical challenge.
Systemic treatment of metastatic esophageal cancer utilizes a combination of multiple chemotherapeutic agents and standard of care regimen exists. Combination chemotherapy with a platinum and fluoropyrimidine doublet, such as the combination of oxaliplatin + infusional 5-fluorouracil (FOLFOX), capecitabine + oxaliplatin (CAPOX), cisplatin + 5-fluorouracil or cisplatin/capecitabine are common regimens.
The use of immune checkpoint inhibitors in the treatment of malignancies has been expanding rapidly since the first approval for Ipilimumab in 2011 for the treatment of metastatic melanoma. In this context, immunotherapy is a novel and intriguing treatment in this disease. In fact, the results of the landmark clinical trials have led to the approval and use of ICIs in combination with chemotherapy recently.
This review article will focus on the use of ICIs in the treatment of cancers of the esophagus.
Immunotherapies: Mechanism of action
Normally, there is a balance between inhibition and stimulation of the immune system. At immune checkpoints, inhibitory signals are put in place to prevent auto-immune disease. One of the mechanisms tumors can exhibit to escape from an immunologic anti-tumor response, is to express ligands that interact with these immune checkpoints. Hence, one of the many approaches in the field of immunotherapy is to target immune checkpoints. Programmed cell death 1 receptor (PD-1) is a transmembrane receptor expressed on tumor infiltrating lymphocytes. In order to evade immune surveillance, tumors either constitutively or inducibly express the inhibitory transmembrane protein programmed death-ligand 1 (PD-L1). Interaction between PD-L1 and PD-1 inhibits cytotoxic T-cell mediated damage to tumor cells, thereby promoting tumor cell growth. Monoclonal antibodies that block PD-1 (pembrolizumab, nivolumab) and PD-L1 (durvalumab, avelumab, atezolizumab) disrupt this interaction and inhibit cell growth.
Ipilimumab and tremelimumab target cytotoxic T lymphocyte-associated protein 4 (CTLA-4). CTLA4 is expressed on T cells and binds to antigen presenting cells via CD80 and CD86 receptors. Antibodies that block this binding can also inhibit tumor cell growth. In the last decade, multiple regulatory agencies have approved various immunotherapy agents for the treatment of various solid tumors and hematologic malignancies.
Is PD-L1 the biomarker?
At present, microsatellite instability (MSI)/mismatch repair protein deficiency (dMMR) and PD-L1 expression are the only predictive biomarkers approved for the use of immunotherapy in the context of gastrointestinal malignancies. Programmed death ligand-1 (PD-L1) is a transmembrane protein that plays a crucial role in regulating the immune response, allowing certain tumors to evade detection and destruction by the immune system. The expression of PD-L1 in tumor cells and tumor-infiltrating immune cells is an important predictive biomarker for the response to immune checkpoint inhibitors therapies, such as anti-PD-1 and anti-PD-L1 antibodies.
In the case of esophageal carcinoma, clinical trials have shown that not all patients benefit from immunotherapy. Therefore, it is crucial to identify patients who will respond favorably to this type of treatment. PD-L1 expression occurs in approximately 50-60% of gastroesophageal or gastric adenocarcinoma cases and 20-80% of esophageal squamous cell carcinomas.
Programmed death ligand-1 is detected by immunohistochemistry. In most cases, PD-L1 expression should be evaluated using the combined positive score (CPS) method. An exception is represented by the evaluation of PD-L1 in advanced/metastatic unresectable EC, where the use of the TPS is recommended. PD-L1 positivity is defined as CPS ≥ 1 and TPS ≥ 1. The method of calculating PD-L1 score is shown in

.
There are some factors affecting PD-L1 expression:
- A. BIOLOGICAL VARIABILITY
- Tumor Type: PD-L1 expression varies significantly between different types of cancer.
- Tumor Microenvironment: PD-L1 expression can be induced by the tumor microenvironment, especially by pro-inflammatory cytokines such as interferon-gamma.
- Tumor Heterogeneity: Intratumoral heterogeneity can complicate the interpretation of PD-L1.
- B. TISSUE LOGISTICS
- Fixation Method: Tissue fixation is crucial for antigen preservation. Formaldehyde is the most commonly used fixative, and the duration and conditions of fixation (pH and temperature) can affect the reactivity of the PD-L1 antibody.
- Sample Selection: The choice of sample type (biopsy or surgical specimen) can influence the results. Small or heterogeneous samples may not adequately represent PD-L1 expression throughout the tumor.
- Intertumoral Heterogeneity: PD-L1 expression can be heterogeneous within tumors, as seen in studies comparing expression between primary and metastatic sites.
- C. TECHNICAL FACTORS
- Antibody Specificity and Sensitivity: Variability in the methods used to detect and evaluate PD-L1 expression by IHC is significant. The choice of antibody clone and staining system can influence the results. Different PD-L1 antibody clones, such as Dako 22C3 for Pembrolizumab, Ventana 28-8 for Nivolumab, SP142 for Atezolizumab and SP263, may yield different results. Although a study provided evidence for the potential interchangeability of 22C3 PD-L1 clone (used in the KEYNOTE-061 study) and 28-8 PD-L1 clone (used in the CheckMate 649 study), another recent study showed only moderate concordance rate between the two assays, with higher sensitivity of the 28-8 PD-L1 clone.
- Pathologist Variability: PD-L1 interpretation by IHC is subject to interobserver variability due to the semi-quantitative nature of scoring methods. Concordance between pathologists can vary, especially in the evaluation of immune cells.
In conclusion, PD-L1 expression is a fundamental biomarker in selecting patients for immune checkpoint inhibitor therapies. However, its detection and clinical application are influenced by various biological and technical factors that must be carefully considered to ensure effective and safe treatment. Standardizing staining protocols, ongoing training of pathologists and implementing standardized scoring methods are essential to overcome the challenges and pitfalls in PD-L1 interpretation by IHC.
Immunotherapy in Non-Metastatic Esophageal Cancer
According to international guidelines, multidisciplinary treatment is considered the standard of care in case of non-metastatic Esophageal Cancer. Until recently, the standard of care treatment for locally advanced Esophageal and Gastroesophageal Junction (GEJ) Cancer included neoadjuvant chemoradiation followed by surgery. There was no role for adjuvant therapy following surgery, regardless of whether there was a pathologic complete response or residual disease.
The ChemoRadiotherapy for Oesophageal cancer followed by Surgery Study (CROSS trial) compared chemoradiotherapy (weekly carboplatin and paclitaxel) followed by surgery with surgery alone in 368 patients (75% with adenocarcinoma and 23%, squamous cell carcinoma) and has demonstrated pathological complete response (pCR) rates of 29%. Median overall survival (the primary endpoint) was 49.4 months in the neoadjuvant chemoradiotherapy group versus 24.0 months in the surgery group (Hazard Ratio (HR) 0.65; p= 0.003). Survival benefit persists after 10-year follow-up, with an absolute 10-year overall survival benefit of 13% (38% versus 25%). Treatment-related toxicity was acceptable and there was no increase in surgical morbidity or mortality. In particular, the 5-year overall survival rate of >60% for squamous cell carcinoma in the trimodality arm was substantially higher than those previously reported.
For patients with locally advanced esophageal or GEJ cancer who have residual disease following chemoradiation and surgery, adjuvant nivolumab is the new standard of care. In the pivotal phase III Checkmate 577 trial, 794 patients with locally advanced esophageal or GEJ cancer (squamous histology 29%; adenocarcinoma 71%) who had residual disease following chemoradiation and resection were randomized to 1 year of adjuvant nivolumab versus placebo. Adjuvant nivolumab doubled the median disease-free survival (DFS) compared to placebo (22.4 versus 11.0 months, HR= 0.69 (96.4% CI: 0.56-0.86, p<0.001). The treatment was well tolerated, showing grade 3-4 adverse events in 13% of patients versus 6% in the placebo group. DFS benefit was seen regardless of histological subtype or PD-L1 expression as measured by combined positive score (CPS). Following the results of the Checkmate 577 trial, nivolumab has been approved in 2021 by the Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for clinical use in esophageal cancer patients after neoadjuvant chemoradiation and radical resection without pathological complete response.
Durvalumab is another anti-PD-L1 monoclonal antibody under investigation in upper gastrointestinal malignancies and other solid malignancies. The role of durvalumab in locally advanced esophageal and GEJ adenocarcinoma in the adjuvant setting was evaluated in a phase II study Big Ten Consortium with early results presented at the 2019 American Society of Clinical Oncology Annual Meeting. The use of durvalumab was evaluated after neoadjuvant, concurrent chemoradiotherapy and R0 resection in locally advanced disease. With the addition of durvalumab to standard tri-modality treatment, patients had 1-year relapse-free survival (RFS) of 79.2% and 1-year overall survival of 95.5% with low rates of grade 3 adverse events (12.5%). Historically, trimodality therapy resulted in 1-year RFS of 50%. Given the tolerability of durvalumab in the adjuvant setting, and nearly 30% absolute improvement in 1-year RFS, further large studies are needed to assess its safety and efficacy.
Moreover, many interesting phase II trials are underway for patients in a curative setting. The ongoing phase II TAPESTRY trial (NCT04595149) investigates the potential of bintrafusp alfa in combination with definitive chemoradiotherapy in esophageal squamous cell carcinoma. Bintrafusp alfa is an agent that binds to transforming growth factor-β (TGF-β) receptor, involved in cell growth, as well as to PD-L1. Thus, it exerts a simultaneous inhibitory function to both Tumor Growth Factor-β and PD-L1.
For locally advanced esophageal squamous cell carcinoma, the PALACE-2 trial (NCT04435197) is ongoing and investigates pembrolizumab in combination with neoadjuvant chemoradiation according to CROSS trial. Earlier results of a small phase Ib trial have shown that this regimen was well-tolerated and achieved a pathological complete response in 56% of patients. The trial is currently proceeding as a phase II study.
Some phase III studies are evaluating the potential of pembrolizumab or durvalumab in conjunction with definitive chemoradiation for individuals with locally advanced esophageal and GEJ cancer. Another study, a phase II-III ECOG-ACRIN 2174, is evaluating the role of dual immune checkpoint inhibition with nivolumab (anti-PD-1) plus ipilimumab (anti-CTLA-4) for resectable locoregional esophageal and GEJ adenocarcinoma.
The phase III KEYNOTE-975 trial is evaluating the efficacy of adding pembrolizumab to definitive chemoradiotherapy in locally advanced esophageal or GEJ cancers (both histologies). The results of this study will help define the role of immunotherapy as first-line treatment option in patients with esophageal cancer who are not eligible for curative surgery.
Similarly, lymphocyte activation gene 3 (LAG-3) is another immune checkpoint inhibitory receptor regulating T cell activation although its exact mechanisms is as yet not fully understood. Combined anti-LAG-3 therapy with Relatlimab and anti-PD-1 or anti-PD-L1 therapy has shown promising clinical activity and has been approved (with nivolumab) in melanoma patients. A phase 1b study evaluated neoadjuvant Nivolumab or Nivolumab-Relatlimab in combination with chemoradiotherapy in 32 patients with resectable stage II/stage III gastroesophageal cancer and showed encouraging outcomes in terms of major pathological response (mPR), relapse free and overall survival but higher rate of grade 3-4 adverse events in the nivolumab plus relatlimab group.
SKYSCRAPER-07 is a phase III trial that assess consolidation atezolizumab, as well as dual inhibition with atezolizumab and tiragolumab, a TIGIT antibody in unresectable esophageal squamous cell carcinoma patients, whose cancers have not progressed following definitive chemoradiotherapy.
Immunotherapy in Metastatic Disease
Patients with unresectable or metastatic esophageal cancer have a poor prognosis and survival has historically been <1 year; however, the use of immune checkpoint inhibitors in combination with chemotherapy has recently improved survival for this patient group. Standard first-line chemotherapy for esophageal cancer is a platinum/fluoropyrimidine doublet. Most randomized trials have been conducted in esophageal adenocarcinoma and data are extrapolated to squamous cell carcinoma; however, multiple phase II studies support platinum/fluoropyrimidine treatment in a squamous cell population.
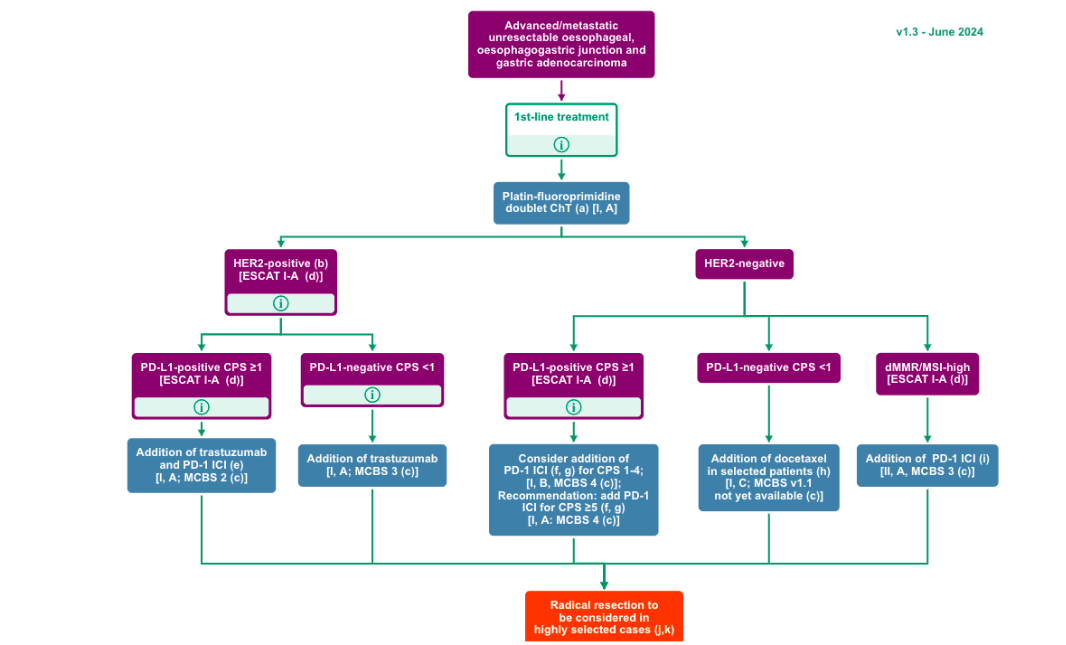
Treatment of advanced adenocarcinoma of the esophagus and GEJ should be in line with the European Society of Medical Oncology Clinical Practice Guidelines for gastric cancer as shown in
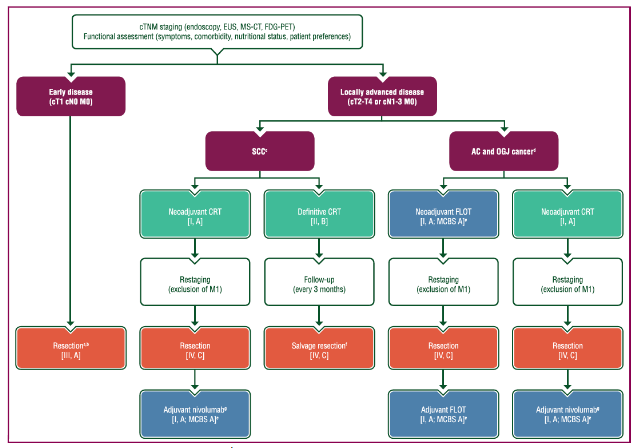
and a proposed algorithm for the treatment of advanced esophageal squamous cell carcinoma is shown in
.
KEYNOTE-028 was one of the first studies to investigate the safety and activity of pembrolizumab (anti-PD-1) in the treatment of squamous cell carcinoma (78%) and adenocarcinoma of the esophagus or GEJ. This phase Ib study included 23 patients, 43% had PD-L1-positive tumors and 87% of patients having received at least 2 lines of prior therapy. Overall response rate was 30% with 17% of patients experiencing grade 3-4 treatment-related adverse events. Given the potential for a significant population of patients with esophageal cancer who could benefit from immunotherapy, clinical trials were undertaken to assess for safety and efficacy.
Expanding on this, the phase II KEYNOTE-180 evaluated pembrolizumab in 121 patients with metastatic esophageal squamous cell and adenocarcinoma and Siewert type 1 gastroesophageal junction adenocarcinomas who had failed at least two prior lines of therapy, regardless of PD-L1 expression status. The majority of patients enrolled were squamous cell carcinomas (52%) and 47.9% of the patients had PD-L1 CPS ≥10. ORR was just 9.9% and 12.4% of patients experiencing grade 3-4 TRAEs. Objective response rates (ORR) for patients with squamous histology was 14.3%, but just 5.2% with adenocarcinoma histology. Furthermore, for those with PD-L1 combined positive score (CPS) ≥10 who represented 48% of the total cohort, ORR was 13.8% but just 6.3% for those who were PD-L1 CPS <10.
Next, came KEYNOTE-181, a phase III trial which randomized 628 patients with advanced esophageal adenocarcinoma or squamous histology (64% of patients) as second line (after one previous line of treatment) to either pembrolizumab or chemotherapy (paclitaxel, docetaxel or irinotecan). Final analysis was undertaken 16 months after the last randomization, at which median OS for those with PD-L1 CPS ≥10 (37%) was 9.3 months with pembrolizumab and 6.7 months for chemotherapy (HR= 0.69; p= 0.0074). The 12-month survival rate was 43.0% in the pembrolizumab group and 20.4% in the chemotherapy group. The co-primary end point of overall survival in patients with squamous cell carcinoma was not met (median OS was 8.2 months and 7.1 months, respectively); but this difference did not reach the prespecified boundaries for significance (HR= 0.78; p= 0.0095). The 12-month survival rate was 39.4% in the pembrolizumab group and 24.9% in the chemotherapy group.
Additionally, the trial showed no difference in median overall survival in the entire population, regardless of PD-L1 and histology (7.1 versus 7.1 months, HR= 0.89; p= 0.056). Grade 3-4 adverse events occurred in 40.9% of those receiving chemotherapy and 18% of those in the pembrolizumab arm, suggesting this is a more tolerable option for those with previously treated disease. Based on KEYNOTE-181, the Food and Drug Administration approved pembrolizumab as second line therapy in patients with locally advanced or metastatic esophageal with squamous histology with PD-L1 CPS ≥10 but the European Medicine Agency has not afforded approval in this setting.
Also, the phase III trial ATTRACTION-3 randomized 419 patients from USA, Europe and Asia with esophageal squamous cell carcinoma to single agent nivolumab versus chemotherapy (investigator’s choice of paclitaxel or docetaxel) as a second-line treatment after failure of a fluoropyrimidine-based treatment. Overall survival favored nivolumab compared with chemotherapy (10.9 vs. 8.4 months; HR= 0.77, p= 0.019) and this benefit was seen in the nivolumab group regardless of PD-L1 CPS score. Patients in the nivolumab arm experienced less toxicity than patients in the chemotherapy group (18% versus 63% grade 3-4 AEs, respectively). Food and Drug Administration and the European Medicine Agency have approved the use of nivolumab for this indication.
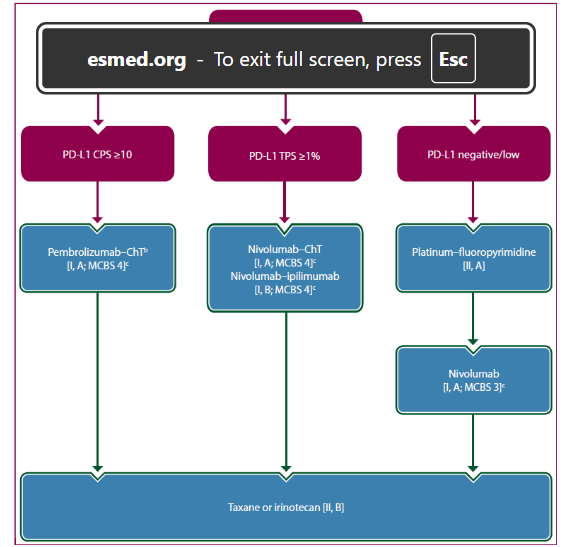
Figure 4. Treatment algorithm for advanced esophageal SCC.¹⁷
First Line Treatment: Doublet chemotherapy based on platin and fluoropyrimidine/taxanes is the standard of care in the first-line treatment for metastatic esophageal carcinomas. However, the survival outcomes with these agents are still disappointing with median overall survival of 8-10 months.
The use of immunotherapy in combination with chemotherapy has recently changed the treatment landscape in this setting. KEYNOTE-590 established a new standard of care for combined chemotherapy with immunotherapy in the first line setting for patients with advanced disease. KEYNOTE-590 is a phase III trial that included 749 patients (Squamous 73%, Adenocarcinoma 27%) with metastatic esophageal and gastroesophageal junction tumors (Siewert I) who did not receive any previous treatment for metastatic disease, regardless of PD-L1 status. This patients were randomized to receive pembrolizumab or placebo in combination with chemotherapy (cisplatin and 5-fluorouracil) until progression or unacceptable toxicity. Pembrolizumab plus chemotherapy was superior to placebo plus chemotherapy for overall survival in all randomized patients (12.4 versus 9.8 months; HR 0.73; p < 0.0001). Greater benefits were seen with the addition of immunotherapy for those with both PD-L1 CPS ≥10 and squamous histology with an improvement of 5 months (13.9 versus 8.8 months; HR 0,57; p<0·0001), 3 months in patients with squamous cell carcinoma (12.6 versus 9.8 months; HR= 0.72; p=0·0006) and 4 months in patients with PD-L1 ≥10 (13.5 versus 9.4 months; HR= 0.62; p<0.0001). Same positive results were reported for progression free survival (PFS). However, in the exploratory analysis there was no benefit to adding pembrolizumab to chemotherapy in patients with PD-L1 <10. The combined treatment had a manageable safety profile; however, 72% of patients in the experimental arm had a grade 3-4 adverse events versus 68% in the control arm. The most frequent grade 3–4 adverse events in the combination arm were decreased neutrophil count (23%), neutropenia (14%), anemia (12%) and nausea (7%). Additionally, 26% of patients in the pembrolizumab arm versus 12% in the placebo arm had immune-related adverse events (grade 3: 7% versus 2%); among them, hypothyroidism (11%), pneumonitis (6%) and hyperthyroidism (6%) were the most common. Those data were confirmed after a longer follow-up. Interestingly, in the updated analysis, 20% of patients in the experimental arm versus 6% in the control arm have maintained the response for a long period (≥24 months). Based on these data, the Food and Drug Agency approved this regimen for esophageal adenocarcinoma and squamous cell carcinoma and Siewert 1 gastroesophageal junction carcinomas. The European Medicine Agency, however, afforded approval only for those with PD-L1 CPS ≥10. As the role of adjuvant nivolumab has been established in esophageal tumors as per CHECKMATE-577, there are questions regarding when to re-challenge with immunotherapy for those with relapsed disease. The general consensus actually is to consider immunotherapy if at least 6 months has elapsed since prior adjuvant immunotherapy.
Nivolumab plus chemotherapy and nivolumab plus ipilimumab have also shown activity in patients with previously untreated, metastatic esophageal squamous cell carcinoma. CHECKMATE-648 was a phase 3 study that randomized 970 patients (only squamous histology) to nivolumab plus chemotherapy, nivolumab plus ipilimumab or chemotherapy alone. Nivolumab or nivolumab plus ipilimumab were administered for a maximum of two years. The trial was not designed to compare the nivolumab plus chemotherapy and nivolumab plus ipilimumab arms directly, rather comparing each separately with chemotherapy alone. Median overall survival favored nivolumab plus chemotherapy over chemotherapy alone (13.2 versus 10.7 months; HR=0.74; p=0.002) and nivolumab plus ipilimumab over chemotherapy (12.7 versus 10.7 months; HR=0.78; p<0.01). In patients with PD-L1 CPS ≥1 disease, there was a survival benefit for both experimental arms and a progression free survival benefit for nivolumab plus chemotherapy but not for nivolumab plus ipilimumab.
Regarding the safety profile, grade 3–4 adverse events were reported in 47% of the nivolumab plus chemotherapy arm, 32% of the nivolumab plus ipilimumab arm and 36% of the chemotherapy arm. Based on these results, both nivolumab plus chemotherapy and nivolumab plus ipilimumab are considered effective first-line treatment for squamous histology but the benefit for immunotherapy in patients with PD-L1 negative tumors is questionable. Given the superior response rate and progression free survival for nivolumab plus chemotherapy, the optimal population for nivolumab plus ipilimumab is not well defined.
The ESCORT-1 trial randomized 595 chinese patients with naïve metastatic esophageal squamous cell carcinoma to receive chemotherapy (cisplatin and paclitaxel) as first-line treatment, alone or in combination with camrelizumab (anti-PD-1). The trial met its primary endpoints; the combination of camrelizumab plus chemotherapy improved overall survival (median: 15.3 versus 12 months; HR= 0.70, p= 0.001) and progression free survival (median: 6.9 versus 5.6 months, HR= 0.56, p= 0.001) with a manageable safety profile (grade 3–4 adverse events: 63.4% versus 67.7% in the combination and control arm, respectively). However, we should consider that the trial included only asian patients and there was no selection according to PD-L1 status.
Tislelizumab is a novel anti-PD-1 approved in China that has reported phase III level efficacy in combination with chemotherapy in esophageal cancer. RATIONALE 306, a phase 3 trial within a global population, randomized 649 patients with previously untreated advanced esophageal squamous cell carcinoma to chemotherapy with tislelizumab or placebo regardless of PD-L1 status. Median overall survival in the immunotherapy arm was 17.2 months in comparison with 10.6 months for chemotherapy alone (HR= 0.66; p< 0.0001). Neither the European Medicine Agency nor Food and Drug Administration approved this combination in this setting to date.
Finally, the results of the ORIENT-15 and JUPITER-06 trials were recently presented. The ORIENT-15 trial is a phase III study that has investigated the role of Sintilimab in combination with chemotherapy as a first-line treatment for metastatic esophageal squamous cell carcinoma. This trial randomized 659 patients to receive sintilimab plus chemotherapy or placebo plus chemotherapy. The patients were included regardless of PD-L1 status; however, the trial assessed PD-L1 positivity by both tumor proportion score (TPS) and combined positive score (CPS). The results showed that the addition of sintilimab to chemotherapy was superior in terms of overall survival (16.7 versus 12.5 months, HR= 0.62; p< 0.0001) and progression free survival (7.2 versus 5.7 months, HR= 0.55, p< 0.0001) in the study population. However, patients with PD-L1 CPS ≥10 had the greatest benefit (median Overall Survival: 17.2 versus 13.6 months, HR= 0.63; p= 0.0018; median Progression Free Survival: 8.3 versus 6.4 months, HR= 0.58; p< 0.0001) with a comparable safety profile (grade 3–5 adverse events: 59.9% versus 54.5%). Of note, 97.1% of the study population was from China. The authors concluded that sintilimab plus chemotherapy could be considered a new therapeutic option in the first-line treatment for metastatic esophageal squamous cell carcinoma.
The JUPITER-06 trial is a phase III trial that is evaluating the efficacy and safety of toripalimab as a first-line treatment for metastatic esophageal squamous histology in an Asian population. The trial randomized 514 patients to receive toripalimab plus chemotherapy or chemotherapy alone, followed by toripalimab as a maintenance treatment or placebo, regardless of PD-L1 status. The first results showed that the addition of toripalimab to chemotherapy has improved overall and progression free survival in all the subgroups (median OS: 17 versus 11 months, HR= 0.58; p= 0.00037) with comparable safety profile to chemotherapy alone (grade 3–5 adverse events: 73.2% versus 70.0%). However, the rate of fatal events was higher if indirectly compared to other trials in this field (8.2%). Final results are awaited.
There are a host of novel immune-based therapies under investigation both as stand-alone agents and in combination with existing anti PD-L1 agents. PD-L1 and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) represent the two main immune checkpoint receptors currently targeted with current immunotherapy, by limiting tumoral methods of immune escape. T cell immunoreceptor with immunoglobulin and ITIM domain (TIGIT) represents a new immune receptor target, as the TIGIT pathway is a regulator of T-cell and natural killer cell (NK) recognition of tumor cells, playing a role in both the adaptive and immune response against them. Gastroesophageal cancers have been shown to utilize this pathway to limit anti-tumor immune response and high TIGIT expression has been suggested to correlate with poorer prognosis. Multiple different anti-TIGIT monoclonal antibodies are under investigation across a host of different malignancies. This includes STAR-221, a phase III trial looking at domvanalimab (anti-TIGIT) with zimberelimab (anti-PD1) and chemotherapy versus nivolumab and chemotherapy in the first-line treatment of gastroesophageal adenocarcinoma. In the case of patients with locally advanced esophageal squamous cell carcinoma who cannot undergo surgery after neoadjuvant chemoradiation, the phase III SKYSCRAPER-07 (NCT04543617) is comparing tiragolumab (anti-TIGIT) plus atezolizumab (anti-PD-L1 agent) versus atezolizumab versus placebo as maintenance treatment in patients who have not progressed after chemoradiotherapy.
| Trial | N | PD-L1 positivity | Histology | Treatment arms | OS (months) | PFS (months) | ORR (%) |
|---|---|---|---|---|---|---|---|
| KEYNOTE-590 | 749 | CPS>10: 50% | SCC 73%, AC 27% | Chemo/Placebo, Chemo/Pembrolizumab | 9.8 | 12.4 | 5.8 |
| CHECKMATE-648 | 970 | TPS >1: 49% | SCC 100% | Chemo alone, Chemo/Nivolumab, Nivolumab/Ipilimumab | 10.7 | 13.2 | 12.7 |
| ESCORT-1 | 595 | TPS >1: 55% | SCC 100% | Chemo/Placebo, Chemo/Camrelizumab | 12.0 | 15.3 | 5.6 |
| RATIONALE 306 | 649 | TPS >10: 37% | SCC 100% | Chemo/Placebo, Chemo/Tislelizumab | 10.6 | 17.2 | 5.6 |
| ORIENT-15 | 659 | CPS >10: 57% | SCC 100% | Chemo/Placebo, Chemo/Sintilimab | 12.5 | 16.7 | 5.7 |
| JUPITER-06 | 514 | CPS >1: 78% | SCC 100% | Chemo/Placebo, Chemo/Toripalimab | 11.0 | 17.0 | 5.5 |
Conclusions
The treatment of esophageal cancers has begun to evolve in the era of immunotherapy and immunotherapy has changed the standard of care in the adjuvant treatment after neoadjuvant chemoradiation and residual disease after surgery and in first-line treatment for metastatic esophageal cancer. Additional novel agents are currently being tested in this field. Nevertheless, the lack of direct comparison in this setting as well as the heterogeneity of the study population and methods (e.g., PD-L1 score and assessment) do not allow to select a single best choice of anti-PD-1 inhibitor as the first-line treatment at this time in metastatic disease. In the non-Asian population, pembrolizumab plus chemotherapy and nivolumab plus chemotherapy are the two options with the most consistent data up to date; however, the first is recommended in Europe only in patients with PD-L1 ≥10. In an Asian population, the combination of camrelizumab, sintilimab or toripalimab with chemotherapy could be options in this setting. The use of biomarkers in determining the potential for efficacy of immune checkpoint inhibition are being investigated to aid in improving patient selection to maximize benefit seen with these agents.
References
1. Ferlay J, Ervik M, Lam F, Laversanne M, Colombet M, Mery L, Piñeros M, Znaor A, Soerjomataram I, Bray F (2024). Global Cancer Observatory: Cancer Today. Lyon, France: International Agency for Research on Cancer. Available from: https://gco.iarc.who.int/today, accessed 09 MAY 2025.
2. Arnold M, Abnet CC, Neale RE, et al. Global burden of 5 major types of gastrointestinal cancer. Gastroenterology. 2020;159(1). doi:10.1053/j.gastro.2020.02.068
3. Cancer Genome Atlas Research Network; Analysis Working Group: Asan University; BC Cancer Agency; Integrated genomic characterization of oesophageal carcinoma. Nature. 2017;541(7636):169-175. doi:10.1038/nature20805
4. Cancer of the esophagus – cancer stat facts. SEER. Accessed May 09, 2025. https://seer.cancer.gov/statfacts/html/esoph.html.
5. Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12(4):252-264. Published 2012 Mar 22. doi:10.1038/nrc3239
6. Madan A, Uronis HE, Strickler JH. A narrative review of the evolving role of immunotherapy in the management of esophageal and gastric cancer. J Gastrointest Oncol. 2022;13(4):2007-2019. doi:10.21037/jgo-22-55
7. Marcus L, Lemery SJ, Keegan P, Pazdur R. FDA Approval Summary: Pembrolizumab for the Treatment of Microsatellite Instability-High Solid Tumors. Clin Cancer Res. 2019;25(13):3753-3758. doi:10.1158/1078-0432.CCR-18-4070
8. Mastracci L, Grillo F, Parente P, et al. PD-L1 evaluation in the gastrointestinal tract: from biological rationale to its clinical application. Pathologica. 2022;114(5):352-364. doi:10.32074/1591-951X-803
9. Heskamp S, Hobo W, Molkenboer-Kuenen JD, et al. Noninvasive Imaging of Tumor PD-L1 Expression Using Radiolabeled Anti-PD-L1 Antibodies. Cancer Res. 2015;75(14):2928-2936. doi:10.1158/0008-5472.CAN-14-3477
10. Rebelatto MC, Midha A, Mistry A, et al. Development of a programmed cell death ligand-1 immunohistochemical assay validated for analysis of non-small cell lung cancer and head and neck squamous cell carcinoma. Diagn Pathol. 2016;11(1):95. Published 2016 Oct 8. doi:10.1186/s13000-016-0545-8
11. Munari E, Zamboni G, Lunardi G, et al. PD-L1 Expression Heterogeneity in Non-Small Cell Lung Cancer: Defining Criteria for Harmonization between Biopsy Specimens and Whole Sections [published correction appears in J Thorac Oncol. 2019 Apr;14(4):744. doi: 10.1016/j.jtho.2019.01.018.]. J Thorac Oncol. 2018;13(8):1113-1120. doi:10.1016/j.jtho.2018.04.017
12. Bodor JN, Boumber Y, Borghaei H. Biomarkers for immune checkpoint inhibition in non-small cell lung cancer (NSCLC). Cancer. 2020;126(2):260-270. doi:10.1002/cncr.32468
13. Hendry S, Byrne DJ, Wright GM, et al. Comparison of Four PD-L1 Immunohistochemical Assays in Lung Cancer. J Thorac Oncol. 2018;13(3):367-376. doi:10.1016/j.jtho.2017.11.112
14. Ahn S, Kim KM. PD-L1 expression in gastric cancer: interchangeability of 22C3 and 28-8 pharmDx assays for responses to immunotherapy. Mod Pathol. 2021;34(9):1719-1727. doi:10.1038/s41379-021-00823-9
15. Yeong J, Lum HYJ, Teo CB, et al. Choice of PD-L1 immunohistochemistry assay influences clinical eligibility for gastric cancer immunotherapy. Gastric Cancer. 2022;25(4):741-750. doi:10.1007/s10120-022-01301-0
16. Cooper WA, Russell PA, Cherian M, et al. Intra- and Interobserver Reproducibility Assessment of PD-L1 Biomarker in Non-Small Cell Lung Cancer. Clin Cancer Res. 2017;23(16):4569-4577. doi:10.1158/1078-0432.CCR-17-0151
17. Obermannová R, Alsina M, Cervantes A, et al. Oesophageal cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022;33(10):992-1004. doi:10.1016/j.annonc.2022.07.003
18. NCCN Clinical Practice Guidelines in Oncology. Esophageal and Esophagogastric Junction Cancer. Version 3.2024 – April 26, 2024. Available online: https://www.nccn.org/professionals/physician_gls/pdf/esophageal.pdf,accessed 28 DIC 2024
19. van Hagen P, Hulshof MC, van Lanschot JJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366(22):2074-2084. doi:10.1056/NEJMoa1112088
20. Eyck BM, van Lanschot JJB, Hulshof MCCM, et al. Ten-Year Outcome of Neoadjuvant Chemoradiotherapy Plus Surgery for Esophageal Cancer: The Randomized Controlled CROSS Trial. J Clin Oncol. 2021;39(18):1995-2004. doi:10.1200/JCO.20.03614
21. Kelly RJ, Ajani JA, Kuzdzal J, et al. Adjuvant Nivolumab in Resected Esophageal or Gastroesophageal Junction Cancer [published correction appears in N Engl J Med. 2023 Feb 16;388(7):672. doi: 10.1056/NEJMx220014.]. N Engl J Med. 2021;384(13):1191-1203. doi:10.1056/NEJMoa2032125
22. Mamdani H, Schneider BJ, Abushahin LI, et al. Safety and efficacy of durvalumab following multimodality therapy for locally advanced esophageal and GEJ adenocarcinoma: Results from Big Ten Cancer Research Consortium Study. J Clin Oncol. 2019;37(15_suppl):4058-4058. doi:10.1200/jco.2019.37.15_suppl.4058
23. Valkema MJ, Mostert B, Lagarde SM, Wijnhoven BPL, van Lanschot JJB. The effectivity of targeted therapy and immunotherapy in patients with advanced metastatic and non-metastatic cancer of the esophagus and esophago-gastric junction. Updates Surg. 2023;75(2):313-323. doi:10.1007/s13304-022-01327-0
24. Shah MA, Bennouna J, Doi T, et al. KEYNOTE-975 study design: a Phase III study of definitive chemoradiotherapy plus pembrolizumab in patients with esophageal carcinoma. Future Oncol. 2021;17(10):1143-1153. doi:10.2217/fon-2020-0969
25. Wang L, Chen M, Kato K, et al. A phase 3 randomized, double-blind, placebo-controlled, Multicenter, Global Study of durvalumab with and after chemoradiotherapy in patients with locally advanced, unresectable esophageal squamous cell carcinoma: Kunlun. J Clin Oncol. 2022;40(4_suppl). doi:10.1200/jco.2022.40.4_suppl.tps373
26. Eads JR, Weitz M, Catalano PJ, et al. A phase II/III study of perioperative nivolumab and Ipilimumab in patients (PTS) with locoregional esophageal (e) and gastroesophageal junction (GEJ) adenocarcinoma: Results of a safety run-in—a trial of the ECOG-acrin cancer research group (EA2174). J Clin Oncol. 2021;39(15_suppl):4064-4064. doi:10.1200/jco.2021.39.15_suppl.4064
27. Kelly RJ, Landon BV, Zaidi AH, et al. Neoadjuvant nivolumab or nivolumab plus LAG-3 inhibitor relatlimab in resectable esophageal/ gastroesophageal junction cancer: a phase Ib trial and ctDNA analyses. Nat Med. 2024;30(4):1023-1034. doi:10.1038/s41591-024-02877-z
28. Goodman KA, Xu R, Chau I, et al. Skyscraper-07: A phase III, randomized, double-blind, placebo-controlled study of atezolizumab with or without tiragolumab in patients with unresectable ESCC who have not progressed following definitive concurrent chemoradiotherapy. J Clin Oncol. 2022;40(4_suppl). doi:10.1200/jco.2022.40.4_suppl.tps374
29. Doi T, Piha-Paul SA, Jalal SI, et al. Safety and antitumor activity of the anti–programmed death-1 antibody pembrolizumab in patients with advanced esophageal carcinoma. J Clin Oncol. 2018;36(1):61-67. doi:10.1200/jco.2017.74.9846
30. Shah MA, Kojima T, Hochhauser D, et al. Efficacy and Safety of Pembrolizumab for Heavily Pretreated Patients With Advanced, Metastatic Adenocarcinoma or Squamous Cell Carcinoma of the Esophagus: The Phase 2 KEYNOTE-180 Study. JAMA Oncol. 2019;5(4):546-550. doi:10.1001/jamaoncol.2018.5441
31. Kojima T, Shah MA, Muro K, et al. Randomized Phase III KEYNOTE-181 Study of Pembrolizumab Versus Chemotherapy in Advanced Esophageal Cancer. J Clin Oncol. 2020;38(35):4138-4148. doi:10.1200/JCO.20.01888
32. Kato K, Cho BC, Takahashi M, et al. Nivolumab versus chemotherapy in patients with advanced oesophageal squamous cell carcinoma refractory or intolerant to previous chemotherapy (ATTRACTION-3): a multicentre, randomised, open-label, phase 3 trial [published correction appears in Lancet Oncol. 2019 Nov;20(11):e613. doi: 10.1016/S1470-2045(19)30646-1.]. Lancet Oncol. 2019;20(11):1506-1517. doi:10.1016/S1470-2045(19)30626-6
33. Sun JM, Shen L, Shah MA, et al. Pembrolizumab plus chemotherapy versus chemotherapy alone for first-line treatment of advanced oesophageal cancer (KEYNOTE-590): a randomised, placebo-controlled, phase 3 study [published correction appears in Lancet. 2021 Nov 20;398(10314):1874. doi: 10.1016/S0140-6736(21)02487-9.]. Lancet. 2021;398(10302):759-771. doi:10.1016/S0140-6736(21)01234-4
34. Metges JP, Kato K, Sun JM, et al. First-line pembrolizumab plus chemotherapy versus chemotherapy in advanced esophageal cancer: longer-term efficacy, safety, and quality-of-life results from the phase 3 KEYNOTE-590 study. J Clin Oncol. 2022;40(4_suppl):241. doi:10.1200/jco.2022.40.4_suppl.241
35. Doki Y, Ajani JA, Kato K, et al. Nivolumab Combination Therapy in Advanced Esophageal Squamous-Cell Carcinoma. N Engl J Med. 2022;386(5):449-462. doi:10.1056/NEJMoa2111380
36. Luo H, Lu J, Bai Y, et al. Effect of Camrelizumab vs Placebo Added to Chemotherapy on Survival and Progression-Free Survival in Patients With Advanced or Metastatic Esophageal Squamous Cell Carcinoma: The ESCORT-1st Randomized Clinical Trial. JAMA. 2021;326(10):916-925. doi:10.1001/jama.2021.12836
37. Xu J, Kato K, Raymond E, et al. Tislelizumab plus chemotherapy versus placebo plus chemotherapy as first-line treatment for advanced or metastatic oesophageal squamous cell carcinoma (RATIONALE-306): a global, randomised, placebo-controlled, phase 3 study [published correction appears in Lancet Oncol. 2024 Mar;25(3):e102. doi: 10.1016/S1470-2045(24)00018-4.]. Lancet Oncol. 2023;24(5):483-495. doi:10.1016/S1470-2045(23)00108-0
38. Shen L, Lu Z-H, Wang J-Y, et al. LBA52 Sintilimab plus chemotherapy versus chemotherapy as first-line therapy in patients with advanced or metastatic esophageal squamous cell cancer: First results of the phase III orient-15 study. Ann Oncol. 2021;32. doi:10.1016/j.annonc.2021.08.2132
39. Wang ZX, Cui C, Yao J, et al. Toripalimab plus chemotherapy in treatment-naïve, advanced esophageal squamous cell carcinoma (JUPITER-06): A multi-center phase 3 trial. Cancer Cell. 2022;40(3):277-288.e3. doi:10.1016/j.ccell.2022.02.
40. Lynch E, Duffy AG, Kelly RJ. Role of Immunotherapy in Gastroesophageal Cancers-Advances, Challenges and Future Strategies. Cancers (Basel). 2023;15(22):5401. Published 2023 Nov 14. doi:10.3390/cancers15225401
41. Klempner SJ, Shitara K, Sison A, et al. Star-221: A randomized, open-label, Multicenter, Phase 3 trial of domvanalimab, zimberelimab, and chemotherapy versus nivolumab and chemotherapy in previously untreated, locally advanced, unresectable or metastatic gastric, gastroesophageal junction, and esophageal adenocarcinoma. J Clin Oncol. 2023;41(16_suppl). doi:10.1200/jco.2023.41.16_suppl.tps4206




