Immunotherapy in Liver Transplantation: Current Insights
Immunotherapy before and after liver transplantation, where are we now?
Kin Pan Au¹ and Kenneth Siu Ho Chok²
- Department of Surgery, The University of Hong Kong, Hong Kong
- Department of Surgery, Chinese University of Hong Kong, Hong Kong
OPEN ACCESS
PUBLISHED: 30 October 2024
CITATION: Pan Au, K. and Siu Ho Chok, K., 2024. Immunotherapy before and after liver transplantation, where are we now?. Medical Research Archives, [online] 12(10).
https://doi.org/10.18103/mra.v12i10.5692
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5692
ISSN 2375-1924
ABSTRACT
Background: A literature review was performed to identify patients who received immunotherapy before and after liver transplantation. We assessed the safety and efficacy of immunotherapy use before and after liver transplantation.
Methods: In patients who received immunotherapy before liver transplantation, we evaluated the safety of performing the transplant by reviewing the rate of acute rejection and graft loss. We also investigated the potential clinical risk factors leading to rejection and examined the durability of the transplant by assessing the long-term graft survival.
For post-transplant immunotherapy, we evaluated the safety of immunotherapy by examining reported adverse events in patients treated for various malignancies. We also investigated the overall survival rates. To superimpose overall survival after liver transplantation, the authors reached out to the original investigators when necessary information was not provided in the article.
Results: In patients who received immunotherapy before liver transplantation, the rate of acute rejection was 32% (n = 17). Patients who developed acute rejection following immunotherapy had a significantly higher rate of graft loss (11%). The findings are summarized in Table 1.
Conclusions: Immunotherapy is associated with a high risk of rejection in liver transplant patients (36%), accompanied by a high rate of graft loss (11%). Notably, immunotherapy-induced rejection was treatment-resistant, with many episodes not responding to medical treatment and progressing to graft failure. In contrast, spontaneous acute rejection was typically treatment-responsive.
Keywords: immunotherapy, liver transplantation, acute rejection, graft loss, overall survival
Introduction
Immunotherapy has transformed the landscape of systemic cancer treatment over the past decade, offering new hope for patients battling various malignancies. By disrupting immune checkpoint pathways, they enhance and reorient the host’s immune response toward tumours, unleashing the body’s own defences against cancer cells. This paradigm shift is particularly significant in the management of advanced hepatocellular carcinoma (HCC), where systemic therapies have traditionally revolved around targeted therapies. While these targeted approaches have shown modest effectiveness, they often come with significant adverse effects that frequently necessitate dose reductions or even discontinuation of treatment. In contrast, immunotherapy has emerged as a compelling alternative due to its more favourable side-effect profile compared to targeted therapies. As a result, immune checkpoint inhibitors have become integrated into standard treatment protocols for advanced HCC. There is growing interest in leveraging immunotherapy not only for treating advanced disease but also as a potential bridging or downstaging therapy prior to liver transplantation.
However, the application of immunotherapy in the context of liver transplantation raises several concerns and controversies. One significant worry is related to the possibility that immune checkpoint inhibitors might heighten allo-immunity, leading to graft rejection post-transplantation. Immune-related transplant rejection can prove fatal², with a protracted clinical course. This places patients at risk for graft failure and eventual graft loss, complicating an already delicate balance between treating cancer and maintaining transplant viability. On the other hand, recurrent HCC is still frequent after liver transplantation³. The immunosuppressive regimens that are necessary to prevent graft rejection diminish immune surveillance, thereby increasing the likelihood of recurrence. The efficacy of immunotherapy is questionable in this setting because concomitant immunosuppression potentially interfere the immunomodulatory pathways involved. Many cancer immunotherapy trials have excluded liver transplant patients, leading to a scarcity of evidence regarding the role of immune checkpoint inhibitors in this unique population.
In light of these complexities, our research team conducted a mini-review in 2021 to explore the use of immunotherapy in liver transplant recipients². We concluded that the risk of rejection remained a significant barrier to the integration of immunotherapy into post-transplant care. However, evidence and clinical experience in this area have expanded, and there is an increasing interest in employing downstaging immunotherapy for potential liver transplant candidates. In the current study, we aim to review the existing literature on patients who underwent immunotherapy prior to liver transplantation and those who received immunotherapy post-transplant. The objective of this comprehensive review is to summarize the current evidence and provide insights on immunotherapy safety and clinical outcomes in liver transplant patients. By consolidating existing studies, we hope to illuminate the potential for immunotherapy in this challenging setting, paving the way for future clinical applications and research initiatives that could ultimately improve outcomes for patients facing both HCC and the complexities of liver transplantation.
Methods
A comprehensive literature review was conducted on PubMed (United States National Library of Medicine, National Institutes of Health, United States) to identify studies investigating immune checkpoint inhibitors therapy before and after liver transplantation. We also reviewed references from included studies to uncover additional relevant articles. The articles were reviewed to extract patient data including demographics, timing and indication of immunotherapy, concomitant immunosuppression, graft and tumour Programmed death-ligand 1 (PD-L1) status, graft toxicity, treatment response and survivals. The authors were contacted for supplementation when required data is not available.
In the article, survival rates. To supplement the available data, the authors reached out to the original investigators when necessary information was not provided in the article.
In patients who received immunotherapy before liver transplantation, we evaluated the safety of performing the transplant by reviewing the rate of acute rejection and graft loss. We also investigated the potential clinical risk factors leading to rejection and examine the durability of the transplant by assessing the long-term graft survival.
For post transplant immunotherapy, we evaluated the safety of immunotherapy by examining rejection rates and early mortality rates among patients treated for various indications. Early mortality was defined as death within 30 days of immunotherapy initiation. We also investigated the efficacy of immunotherapy in patients with recurrent HCC after liver transplantation. We assessed the best treatment response, progression- free survival, and overall survival rates. The treatment response was determined according to the Response Evaluation Criteria in Solid Tumours (version 1.1)⁴. Data was summarized using descriptive statistics, with continuous variables expressed as medians and interquartile ranges. Parametric and non-parametric variables were compared using Student’s t-test and Mann-Whitney U test as appropriate. Categorical variables were expressed as frequencies and percentages and compared using the chi-square test. Survival data was presented using the Kaplan-Meier method and compared using Log-rank test. Data analysis was performed using SPSS 29.0 for Windows (SPSS Inc., Chicago, Illinois, United States). A p-value less than <0.05 was considered statistically significant.
We assessed the safety of immunotherapy by reviewing the rejection rate and mortality in all identified patients treated for various indications. We also looked into patients treated for recurrent HCC after liver transplantation to investigate the efficacy of immunotherapy in this setting. We reviewed the best treatment response, rate of early mortality, progression-free survival and overall survival after immunotherapy. Early mortality was defined as mortality within 30 days from immunotherapy. Treatment response was defined according to the Response Evaluation Criteria in Solid Tumours 1.1⁴. Data was summarized with descriptive statistics. Continuous variables were expressed with medians and interquartile ranges. Parametric and non-parametric variables were compared with Student’s t-test and Mann-Whitney U test where appropriate. Categorical variables were expressed in frequencies and percentages and compared with chi-square test. Survival data was expressed in median, presented with Kaplan-Meier method and compared using Log-rank test. Data was analysed using Statistical Package for the Social Sciences 16.0 (SPSS) for Windows (SPSS Inc., Chicago, IL). Statistical significance was defined by p-value <0.05.
Results
On PubMed, we identified 24 publications and reviewed 52 patients treated on the basis of this study. Among them 44 patients received immunotherapy before undergoing a liver transplantation⁵⁻⁶ ¹⁵⁻⁷ ¹⁴ (supplementary table 1). Fifty-three patients had a prior liver transplantation and subsequently received immunotherapy. Thirty-one were treated for recurrent HCC²⁶ ¹⁵, ³¹ ¹⁷ ²⁴ while 22 were treated for other indications ²⁰ ²⁹ ³⁹ ⁴⁵ ³¹ ³⁸ (supplementary table 2).
ALL PATIENTS
The characteristics of 97 included patients are presented in Table 1. The patient population demonstrated a male predominance, with 79% of patients being male. The median age was 59 years (interquartile range: 50–65). Seventy-four (76%) received immunotherapy for HCC, 10 (10%) for melanoma, and 4 (4%) for squamous cell carcinoma of the skin. Most patients (96%, n = 93) received programmed cell death protein-1 (PD-1) / PD-L1 inhibitors, with nivolumab being used in 60% and pembrolizumab in 22%. Three patients (3%) received cytotoxic T-lymphocyte antigen-4 (CTLA-4) inhibitors, all of whom were being treated for melanoma. One
One patient received both PD-1 and CTLA-4 inhibitors. Nine graft liver and 17 tumour tissue samples were tested for PD-L1 status. Among the tested samples, the rates of positive PD-L1 staining were 44% for graft liver tissue and 53% for tumour tissue, respectively. The rejection rate was 36% (n = 35), and graft loss occurred in 11 patients (11%).
IMMUNOTHERAPY BEFORE LIVER TRANSPLANT
PATIENTS CHARACTERISTICS
Forty-four patients received immunotherapy prior to liver transplantation. The median duration between their last dose of immunotherapy and liver transplantation was 4.2 weeks (interquartile range [IQR], 2.0–10.0 weeks) (Table 1). Most of these patients (43/44) were treated for hepatocellular carcinoma (HCC), which was also the indication for their liver transplant. One patient received immunotherapy for cholangiocarcinoma. All patients received PD-1/PD-L1 inhibitors, with nivolumab being the most commonly used agent (n = 27, 61%), followed by pembrolizumab (n = 9, 20%).
RISK AND IMPLICATIONS OF REJECTION
Eighteen patients experienced rejection, which accounted for a rejection rate of 41% (Table 2). Two patients died due to rejection, and two required retransplantation. This contributed to a 22% graft loss rate in the rejection group, which was significantly higher than the non-rejection group (0%, p = 0.012). Patients who experienced rejection were transplanted earlier following their last dose of immunotherapy, with a median time of 2.9 weeks compared to 8.3 weeks in patients without rejection (p < 0.001). In the rejection group, more patients received pembrolizumab (33% vs. 12%) and fewer patients were treated with nivolumab (39% vs. 77%). The rejection rate associated with pembrolizumab was higher than nivolumab (67% vs. 35%, p = 0.03).
IMMUNOTHERAPY AFTER LIVER TRANSPLANT
PATIENT CHARACTERISTICS
Fifty-three patients who underwent liver transplantation received immunotherapy at a median interval of 3.5 years (IQR 2.0–6.9 years) after transplant (Table 1). Thirty-one patients (58%, n = 31) were treated for recurrent HCC, 19% (n = 10) for melanoma, and 8% (n = 4) for cutaneous squamous cell carcinoma. Most immunotherapies were used as second-line systemic therapy.
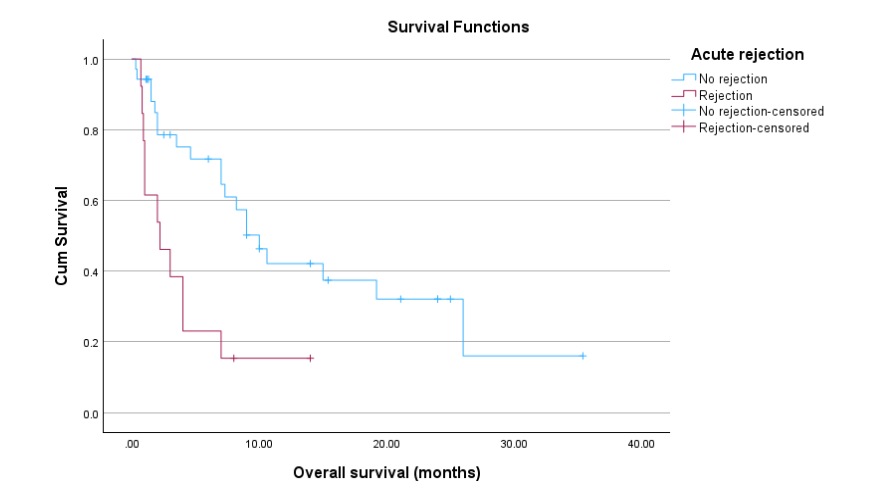
Figure 1. Overall survival of post-transplant patient undergoing immunotherapy stratified by presence and absence of rejection (p=0.003).
IMMUNOTHERAPY AFTER LIVER TRANSPLANT
CHARACTERISTICS OF PATIENTS
Patients who received immunotherapy after liver transplant stratified by indication of immunotherapy are summarized in Table 3.
| Characteristic | HCC | Non-HCC | p-values |
|---|---|---|---|
| Gender (M:F) | 25:5 | 22:4 | 0.432 |
| Age | 61 (46-74) | 62 (55-67) | 0.019 |
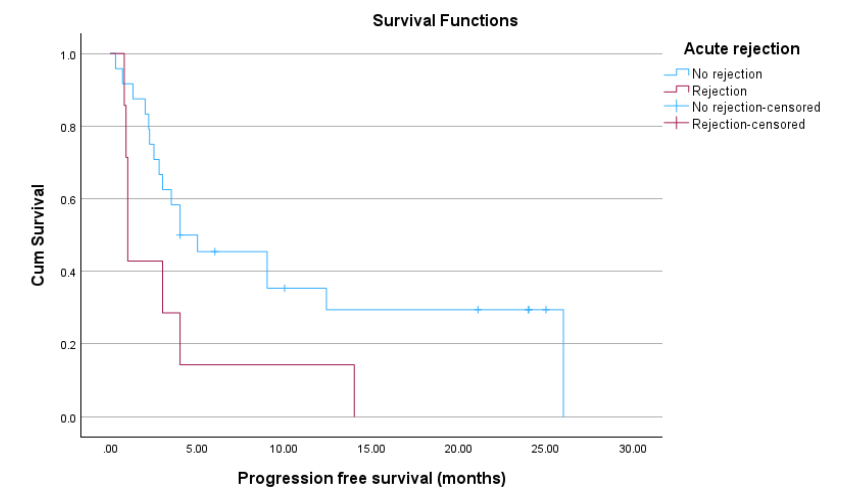
Figure 2. Progression-free survival post-immunotherapy stratified by presence and absence of rejection (p=0.003).
Table 3. Characteristics of patients who received immunotherapy after liver transplant stratified by indication of immunotherapy.
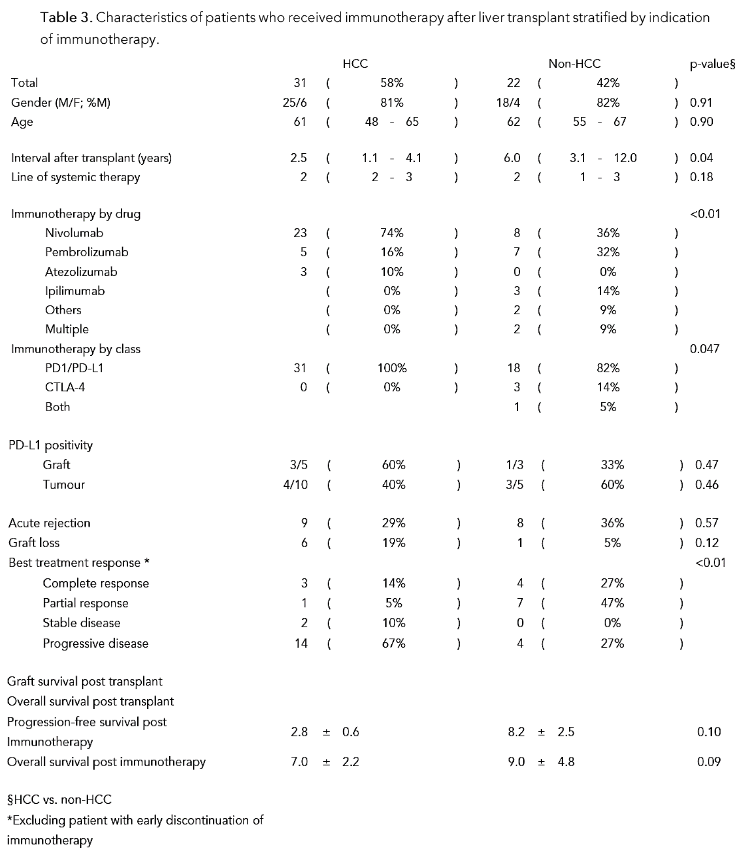
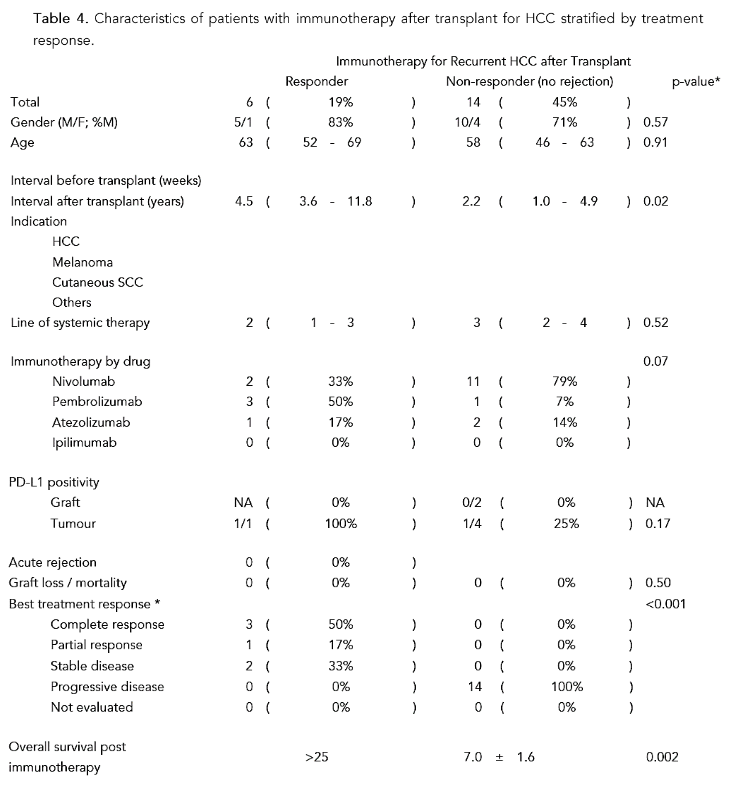
Table 4. Characteristics of patients with immunotherapy after transplant for HCC stratified by treatment response.
Immunotherapy for Recurrent HCC after Transplant
Discussions
Despite ongoing concerns about rejection, the use of immunotherapy in liver transplant patients continues to grow. In 2021, we identified 25 patients receiving immunotherapy in the literature, which increased to 97 patients in 2024². In the post-transplant setting, immunotherapy is being used in select patients with recurrent HCC and de-novo malignancies who have failed multiple lines of systemic therapy. Notably, there has been a significant expansion in the use of immunotherapy in pre-transplant patients, with 44 patients receiving immunotherapy for bridging and downstaging before transplant. A consistent finding in the expanded cohort is that immunotherapy is associated with a high risk of rejection in liver transplant patients (36%), accompanied by a high rate of graft loss (11%). Notably, immunotherapy-induced rejection was treatment-resistant, with many episodes not responding to medical treatment and progressing to graft failure. In contrast, spontaneous acute rejection was typically treatment-responsive⁴⁶ ⁴⁸. To better understand the clinical risk factors for rejection, we investigated the clinical predictors of rejection in both scenarios: immunotherapy before liver transplant and immunotherapy after liver transplant.
IMMUNOTHERAPY BEFORE TRANSPLANT
Patients who received liver transplantation after immunotherapy were found to have a higher risk of acute rejection if they received ICI shortly before transplantation. The half-life of nivolumab is approximately 20 days⁴⁹. Pembrolizumab has a half-life of 26 days⁵⁰. However, the biological effects of PD-L1 inhibitors on T-cell function may persist longer than their half-lives. Studies have shown that PD-L1 receptor occupancy can be observed for up to months after immune checkpoint inhibitor use (Safety and Activity of Anti–PD-L1 Antibody in Patients with Advanced Cancer). After a single administration of nivolumab, PD-1 occupancy on lymphocytes was observed for 14 weeks⁵¹. In clinical practice, immune-related hepatitis can occur months after starting treatment or even after stopping immune checkpoint inhibitor treatment⁵².
Pooled data from available literature showed that patients with acute rejection after liver transplantation had a shorter median washout period (2.9 weeks) compared to those without rejection (8.3 weeks). Most rejection cases occurred in patients with a washout period of less than 6 weeks (17/26, rejection risk 65%), while only one out of 18 patients who underwent transplant more than 6 weeks after ICI suffered from rejection (rejection risk 5.6%). Based on these laboratory and clinical findings, a washout period of at least 6 weeks may be reasonable to reduce the risk of fatal rejection. Nevertheless, some individuals may still experience rejection, even with prolonged washout periods. Phenotypic variations might determine individual susceptibility to effect of PD-L1 blockade. Further preclinical and translational studies are needed to elucidate the underlying mechanisms and identify high-risk individuals at risk of prolonged effects of PD-L1 blockade.
We observed a higher rejection rate associated with pembrolizumab over nivolumab (67% vs. 35%, p = 0.03). However, most rejection episodes with pembrolizumab (n = 5) were contributed by one case series¹⁵. In this series, 9 rejections were reported but only 4 of them were biopsy proven. The others were clinical rejections which improved with titrating up the immunosuppression. Our observation was in contrast to the results of a systematic review published by Fisher et al. in 2020 which reported higher rejection rate associated with nivolumab than pembrolizumab (25% vs. 10%)⁵⁴. However, Fisher et al.’s report also included patients with kidney and heart transplant, and the findings were not statistically significant. Due to the limitation in methodology and incoherent results, the observation needs to be carefully interpreted.
IMMUNOTHERAPY AFTER TRANSPLANT
In our previous review, we reported that patients who underwent long-term liver transplantation were less likely to experience rejection when treated with immunotherapy². Our latest findings from an expanded cohort of 53 patients confirm this observation, revealing a significant association between earlier immunotherapy and rejection after liver transplantation. Specifically, patients who developed rejection received immunotherapy at a median time of 2.9 years after transplant, compared to 4.0 years in those who did not experience rejection (p = 0.04). This underscores the importance of timing of immunotherapy in liver transplant recipients.
The risk of spontaneous acute cellular rejection decreases over time after transplantation, as the recipient’s immune system develops tolerance to the graft. This phenomenon, known as systemic chimerism, occurs when donor leukocytes disseminate to the recipient⁵⁴ ⁵⁵. As a result, immunosuppression can often be tapered or even discontinued in long-term transplant recipients⁵⁶. It is logical to deduce that the risk of immunotherapy-induced rejection would also decrease with time. However, our findings suggest that this effect may be less pronounced in the context of immunotherapy. Spontaneous acute rejection unrelated to immunotherapy tends to occur within the first few years after transplant⁵⁷. While the risk of spontaneous rejection largely decreases beyond the first year⁵⁸, the risk of post-immunotherapy rejection appears to persist further. In our cohort, the median time from transplant to rejection was 2.9 years, with one case of fatal rejection reported after receiving immunotherapy for recurrent post-liver transplant HCC¹⁸. Our data suggest that a prolonged period of waiting time may be necessary before the risk of rejection decreases significantly. Therefore, it is crucial not to neglect this risk in the first few years after transplantation. To confirm these findings and establish a more definitive safe window for immunotherapy initiation, further studies are needed.
Allograft PD-L1 expression was evaluated in 8 patients who underwent immunotherapy following liver transplantation. Despite the limited sample size, our findings suggest that positive PD-L1 staining on liver grafts may increase the risk of acute rejection following treatment with immune checkpoint inhibitors. PD-L1 plays a critical role in immunoregulation and preventing allograft rejection. It is expressed on graft hepatocytes, and it binds to PD-1 on infiltrating lymphocytes thereby downregulating the immune response and preventing acute cellular rejection⁵⁹. PD-L1 expression in liver allografts may indicate ongoing immune regulation to mitigate immune activation. However, when the PD-1/PD-L1 axis is blocked, infiltrating lymphocytes lose regulatory control, leading to unopposed immune activation and unchecked allograft injury⁶⁰.
Timing of assessment may be critical for accurately predicting the risk of rejection based on graft PD-L1 expression. Nordness et al. reported a fatal case of graft rejection following post-transplant use of immune checkpoint inhibitor⁵. While the pre-implant graft biopsy was negative for PD-L1 staining, the repeated biopsy during rejection revealed significant PD-L1 expression on infiltrating donor Kupffer cells and antigen-presenting cells. The dynamic expression of PD-L1 on grafts may be influenced by the interplay between hepatocytes and infiltrating lymphocytes. Therefore, the implications of PD-L1 expression in different timing must be carefully evaluated. Graft biopsies before implantation and exposure to donor alloimmunity might not forecast the risk of rejection following engraftment. Further research is needed to determine the optimal time interval for assessing allograft PD-L1 status after transplantation.
EFFICACY IN TREATING RECURRENT HEPATOCELLULAR CARCINOMA
When evaluating treatment response, a significant proportion of patients (35.2%, n = 11) were excluded due to premature termination of treatment, primarily attributed to rejection. These results suggested that safety of immunotherapy has to [be] addressed before its potential efficacy could be fully assessed. Our findings suggest that immunotherapy administered after a longer period following transplant is not only associated with a lower risk of rejection but also with better treatment responses for recurrent HCC. This observation is consistent with the analysis by Kayali et al. [25]. One notable difference is that we excluded patients whom treatment was prematurely discontinued due to rejection or other reasons. The improved survival benefits observed in patients with late recurrence may be attributed to better tumour biology. Our experience shows that patients with early HCC recurrence, regardless of treatment modality, have a poorer prognosis⁶¹. Unfortunately, most HCC recurrences occur early after liver transplantation³. In our cohort, patients who received immunotherapy for HCC recurrence were
treated earlier after transplant compared to those treated for de novo malignancies (median time from transplant 2.5 vs. 6.0 years, p = 0.04). Patients with late HCC recurrence may be better candidates for immunotherapy due to reduced rejection risk and potentially improved oncological outcomes.
From our results, Pembrolizumab appeared to be associated with better oncological outcomes. More patients in the responding group received Pembrolizumab, and this group also had the longest overall survival. This finding is consistent with the observation of survival benefits reported for Pembrolizumab over Nivolumab in unresectable primary HCC⁶². Unfortunately, the disease status of these patients was not available for comparison. Their potential confounding effects should be considered when interpreting the outcomes. Last but not least, we suggest reviewing the post-transplant tumour PD-L1 status as it is logical to assume that it affects treatment response. PD-L1 expression in tumours has been shown to predict treatment response in unresectable primary HCC⁶³. In our cohort, the percentage of PD-L1 positivity was higher in the responding group (100% vs. 25%, p = 0.17), although the sample size was too limited to draw statistical conclusions. In future studies, explant tumour PD-L1 status can be reviewed when patients are contemplated for immunotherapy.
Conclusion
The current study has several limitations. The subjects were sampled from individual case reports and series, making it vulnerable to publication bias, where patients with extreme outcomes are more likely to be reported. The methodology of the individual studies is heterogeneous, and not all rejection episodes were biopsy proven. The diversified immunosuppressive regimens may have affected rejection and response rates. The small sample size also limits the generalisability of the study. Based on the current available evidence, rejection remains a major obstacle to the use of immunotherapy in liver transplant patients. Rejection is associated with a significant risk of graft loss and mortality.
To ensure patient safety, it is essential to identify patients at risk of developing rejection. We observed several clinical associations with acute rejection, including a short washout period in patients who received immunotherapy prior to transplant, as well as short duration from transplant and graft PD-L1 positivity in post-liver transplant patients. We also revealed better survival benefits in patients with late recurrence of HCC after liver transplantation. These observations will allow clinicians to refine patient selection. To provide more evidence on the use of immune checkpoint inhibitor in liver transplant recipients, an international registry would be ideal to collect information on immunotherapy in liver transplant patients. This will allow for a systematic collection of data until more robust clinical trials become available. Finally, laboratory and translational studies are also needed to enhance our understanding of the complex interplay between the immune system, tumour neoantigens, and graft alloantigens. These will allow direction of host immunity to maximise anti-tumour effect while maintaining allograft tolerance. With better understanding and insight, we can achieve more desirable outcomes and allow more transplant patients benefit from immunotherapy.
Conflict of Interest:
None
Acknowledgements:
None
References
1. Finn RS, Qin S, Ikeda M, et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N Engl J Med. 2020;382(20):1894-1905. doi:10.1056/NEJMOA1915745/SUPPL_FILE/NEJMOA1915745_DATA-SHARING.PDF
2. Au KP, Chok KSH. Immunotherapy after liver transplantation: Where are we now? World J Gastrointest Surg. 2021;13(10):1267. doi:10.4240/WJGS.V13.I10.1267
3. Au KP, Chok KSH. Multidisciplinary approach for post-liver transplant recurrence of hepatocellular carcinoma: A proposed management algorithm. World J Gastroenterol. 2018;24(45):5081-5094. doi:10.3748/wjg.v24.i45.5081
4. Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228-247. doi:10.1016/j.ejca.2008.10.026
5. Nordness MF, Hamel S, Godfrey CM, et al. Fatal hepatic necrosis after nivolumab as a bridge to liver transplant for HCC: Are checkpoint inhibitors safe for the pretransplant patient? Am J Transplant. 2020;20(3):879-883. doi:10.1111/AJT.15617
6. Schwacha-Eipper B, Minciuna I, Banz V, Dufour JF. Immunotherapy as a Downstaging Therapy for Liver Transplantation. Hepatology. 2020;72(4):1488-1490. doi:10.1002/HEP.31234
7. Chen GH, Wang G Bin, Huang F, et al. Pretransplant use of toripalimab for hepatocellular carcinoma resulting in fatal acute hepatic necrosis in the immediate postoperative period. Transpl Immunol. 2021;66. doi:10.1016/J.TRIM.2021.101386
8. Chen Z, Hong X, Wang T, et al. Prognosis after liver transplantation in patients treated with anti- PD-1 immunotherapy for advanced hepatocellular carcinoma: case series. Ann Palliat Med. 2021;10 (9):9354361-9359361. doi:10.21037/APM-21-999
9. Dehghan Y, Schnickel GT, Hosseini M, et al. Rescue liver re-transplantation after graft loss due to severe rejection in the setting of pre-transplant nivolumab therapy. Clin J Gastroenterol. 2021;14 (6):1718-1724. doi:10.1007/S12328-021-01521-4
10. Sogbe M, López-Guerra D, Blanco-Fernández G, Sangro B, Narváez-Rodriguez I. Durvalumab as a Successful Downstaging Therapy for Liver Transplantation in Hepatocellular Carcinoma: The Importance of a Washout Period. Transplantation. 2021;105(12):E398-E400. doi:10.1097/TP.0000000000003855
11. Tabrizian P, Florman SS, Schwartz ME. PD-1 inhibitor as bridge therapy to liver transplantation? Am J Transplant. 2021;21(5):1979-1980. doi:10.1111/AJT.16448
12. Aby ES, Lake JR. Immune Checkpoint Inhibitor Therapy Before Liver Transplantation—Case and Literature Review. Transplant Direct. 2022;8(4):E1304. doi:10.1097/TXD.0000000000001304
13. Kang E, Martinez M, Moisander-Joyce H, et al. Stable liver graft post anti-PD1 therapy as a bridge to transplantation in an adolescent with hepatocellular carcinoma. Pediatr Transplant. 2022; 26(3). doi:10.1111/PETR.14209
14. Schnickel GT, Fabbri K, Hosseini M, et al. Liver transplantation for hepatocellular carcinoma following checkpoint inhibitor therapy with nivolumab. Am J Transplant. 2022;22(6):1699-1704. doi:10.1111/AJT.16965
15. Wang T, Chen Z, Liu Y, et al. Neoadjuvant programmed cell death 1 inhibitor before liver transplantation for HCC is not associated with increased graft loss. Liver Transpl. 2023;29(6):598-606. doi:10.1097/LVT.0000000000000083
16. Wang K, Guo WX, Chen MS, et al. Multimodality Treatment for Hepatocellular Carcinoma With Portal Vein Tumor Thrombus: A Large-Scale, Multicenter, Propensity Mathching Score Analysis. Medicine (Baltimore). 2016;95(11) :e3015. doi:10.1097/MD.0000000000003015
17. De Toni EN, Gerbes AL. Tapering of Immunosuppression and Sustained Treatment With Nivolumab in a Liver Transplant Recipient. Gastroenterology. 2017;152(6):1631-1633. doi:10.1053/j.gastro.2017.01.063
18. Friend BD, Venick RS, McDiarmid S V., et al. Fatal orthotopic liver transplant organ rejection induced by a checkpoint inhibitor in two patients with refractory, metastatic hepatocellular carcinoma. Pediatr Blood Cancer. 2017;64(12):e26682. doi:10.1002/pbc.26682
19. Varkaris A, Lewis DW, Nugent FW. Preserved Liver Transplant After PD-1 Pathway Inhibitor for Hepatocellular Carcinoma. Am J Gastroenterol. 2017;112(12):1895-1896. doi:10.1038/ajg.2017.387
20. Deleon TT, Salomao MA, Aqel BA, et al. Pilot evaluation of PD-1 inhibition in metastatic cancer patients with a history of liver transplantation : the Mayo Clinic experience. J Gastrointest Oncol. 2018;(suppl 4S). doi:10.21037/jgo.2018.07.05
21. Gomez P, Naim A, Zucker K, Wong M. A Case of Hepatocellular Carcinoma (HCC) Immunotherapy Inducing Liver Transplant Rejection. Am J Gastroenterol. 2018;113.
22. Gassmann D, Weiler S, Mertens JC, et al. Liver allograft failure after nivolumab treatment—A Case report with systematic literature research. Transplant Direct. 2018;4(8). doi:10.1097/TXD.0000000000000814
23. Nasr F, Al Ghoche A, Diab S, et al. Pembrolizumab Monother-Apy in Relapsed Hepatocellular Carcinoma Post Living Donor Liver Transplantation and Sorafenib. Vol 1.; 2018. https://www.elsevier.com/books/robbins-and-cotran-patho-. Accessed February 7, 2021.
24. Rammohan A, Reddy MS, Farouk M, Vargese J, Rela M. Pembrolizumab for metastatic hepatocellular carcinoma following live donor liver transplantation: The silver bullet? Hepatology. 2018;67(3):1166-1168. doi:10.1002/hep.29575
25. Kumar S. Nivolumab-Induced Severe Allograft Rejection in Recurrent Post-Transplant Hepatocellular Carcinoma. Am J Gastroenterol. 2019;114. doi:10.14309/01.ajg.0000598472.41771.5f
26. Amjad W, Kotiah S, Gupta A, Morris M, Liu L, Thuluvath PJ. Successful Treatment of Disseminated Hepatocellular Carcinoma After Liver Transplantation With Nivolumab. J Clin Exp Hepatol. 2020;10(2): 185-187. doi:10.1016/J.JCEH.2019.11.009
27. Anugwom C, Leventhal T. Nivolumab-Induced Autoimmune-Like Cholestatic Hepatitis in a Liver Transplant Recipient. ACG case reports J. 2020;7(7):e00416. doi:10.14309/CRJ.0000000000000416
28. Al Jarroudi O, Ulusakarya A, Almohamad W, Afqir S, Morere J-F. Anti-Programmed Cell Death Protein 1 (PD-1) Immunotherapy for Metastatic Hepatocellular Carcinoma After Liver Transplantation: A Report of Three Cases. Cureus. 2020;12(10). doi:10.7759/cureus.11150
29. Owoyemi I, Vaughan LE, Costello CM, et al. Clinical outcomes of solid organ transplant recipients with metastatic cancers who are treated with immune checkpoint inhibitors: A single-center analysis. Cancer. 2020;126(21):4780-4787. doi:10.1002/CNCR.33134
30. Ben Khaled N, Roessler D, Reiter FP, Seidensticker M, Guba M, De Toni EN. Extending the Use of Atezolizumab and Bevacizumab to a Liver Transplant Recipient: Need for a Posttransplant Registry. Liver Transpl. 2021;27(6):928-929. doi:10.1002/LT.26011
31. Rudolph M, Shah SA, Quillin R, et al. Immune checkpoint inhibitors in liver transplant: a case series. J Gastrointest Oncol. 2023;14(2):1141-1148. doi:10.21037/JGO-22-922/COIF)
32. Ranganath HA, Panella TJ. Administration of ipilimumab to a liver transplant recipient with unresectable metastatic melanoma. J Immunother. 2015;38(5):211. doi:10.1097/CJI.0000000000000077
33. Morales RE, Shoushtari AN, Walsh MM, Grewal P, Lipson EJ, Carvajal RD. Safety and efficacy of ipilimumab to treat advanced melanoma in the setting of liver transplantation. J Immunother Cancer. 2015;3(1):22. doi:10.1186/s40425-015-0066-0
34. Biondani P, De Martin E, Samuel D. Safety of an anti-PD-1 immune checkpoint inhibitor in a liver transplant recipient. Ann Oncol. 2018;29(1): 286-287. doi:10.1093/annonc/mdx548
35. Dueland S, Guren TK, Boberg KM, et al. Acute liver graft rejection after ipilimumab therapy. Ann Oncol. 2017;28(10):2619-2620. doi:10.1093/annonc/mdx281
36. Schvartsman G, Perez K, Sood G, Katkhuda R, Tawbi H. Immune checkpoint inhibitor therapy in a liver transplant recipient with melanoma. Ann Intern Med. 2017;167(5):361-362. doi:10.7326/L17-0187
37. Kuo JC, Lilly LB, Hogg D. Immune checkpoint inhibitor therapy in a liver transplant recipient with a rare subtype of melanoma. Melanoma Res. 2018;28(1):61-64. doi:10.1097/CMR.0000000000000410
38. Tio M, Rai R, Ezeoke OM, et al. Anti-PD-1/PD-L1 immunotherapy in patients with solid organ transplant, HIV or hepatitis B/C infection. Eur J Cancer. 2018;104:137-144. doi:10.1016/j.ejca.2018.09.017
39. Chen JA, Esteghamat N, Kim EJ, et al. PD-1 Blockade in a Liver Transplant Recipient With Microsatellite Unstable Metastatic Colorectal Cancer and Hepatic Impairment. J Natl Compr Canc Netw. 2019;17(9):1026-1030. doi:10.6004/JNCCN.2019.7328
40. Lee BT, Horwich BH, Chopra S, Ahearn A, Han HH. Checkpoint Inhibitor-Induced Rejection of a Liver Allograft: A Combination of Acute T Cell-Mediated and Antibody-Mediated Rejection. Liver Transpl. 2019;25(12):1845-1848. doi:10.1002/LT.25622
41. Braun M, Fuchs V, Kian W, et al. Nivolumab Induced Hepatocanalicular Cholestasis and Liver Rejection in a Patient With Lung Cancer and Liver Transplant. J Thorac Oncol. 2020;15(9):e149-e150. doi:10.1016/j.jtho.2020.01.026
42. Tsung I, Worden FP, Fontana RJ. A Pilot Study of Checkpoint Inhibitors in Solid Organ Transplant Recipients with Metastatic Cutaneous Squamous Cell Carcinoma. Oncologist. 2021;26(2): 133-138. doi:10.1002/ONCO.13539
43. Bittner A, Radke J, Eurich D, et al. Cerebral EBV-positive PTLD controlled by PD-1 checkpoint blockade in a liver transplant patient. Leuk Lymphoma. 2021;62(8):2026-2029. doi:10.1080/10428194.2021.1889537
44. Brumfiel CM, Patel MH, Aqel B, Lehrer M, Patel SH, Seetharam M. Immune checkpoint inhibitor therapy in a liver transplant recipient with autoimmune disease and metastatic cutaneous squamous cell carcinoma. JAAD Case Reports. 2021;14:78. doi:10.1016/J.JDCR.2021.05.012
45. Kondo T, Kawachi S, Nakatsugawa M, et al. Nivolumab for recurrent/metastatic hypopharyngeal squamous cell carcinoma in a liver transplant recipient. Auris Nasus Larynx. 2022;49(4):721-726. doi:10.1016/J.ANL.2021.02.002
46. Rodríguez-Perálvarez M, Rico-Juri JM, Tsochatzis E, Burra P, De la Mata M, Lerut J. Biopsy-proven acute cellular rejection as an efficacy endpoint of randomized trials in liver transplantation: a systematic review and critical appraisal. Transpl Int. 2016;29(9):961-973. doi:10.1111/tri.12737
47. Thurairajah PH, Carbone M, Bridgestock H, et al. Late acute liver allograft rejection; A study of its natural history and graft survival in the current era. Transplantation. 2013;95(7):955-959.
doi:10.1097/TP.0b013e3182845f6c
48. Jain A, Reyes J, Kashyap R, et al. Long-term survival after liver transplantation in 4,000 consecutive patients at a single center. Ann Surg. 2000;232(4): 490-500. doi:10.1097/00000658-200010000-00004
49. Brahmer JR, Hammers H, Lipson EJ. Nivolumab: targeting PD-1 to bolster antitumor immunity. Future Oncol. 2015;11(9):1307-1326. doi:10.2217/FON.15.52
50. Dang TO, Ogunniyi A, Barbee MS, Drilon A. Pembrolizumab for the treatment of PD-L1 positive advanced or metastatic non-small cell lung cancer. Expert Rev Anticancer Ther. 2016;16(1):13. doi:10.1586/14737140.2016.1123626
51. Brahmer JR, Drake CG, Wollner I, et al. Phase I Study of Single-Agent Anti–Programmed Death-1 (MDX-1106) in Refractory Solid Tumors: Safety, Clinical Activity, Pharmacodynamics, and Immunologic Correlates. J Clin Oncol. 2010;28 (19):3167-3175. doi:10.1200/JCO.2009.26.7609
52. Vitale G, Lamberti G, Comito F, et al. Anti-programmed cell death-1 and anti-programmed cell death ligand-1 immune-related liver diseases: from clinical pivotal studies to real-life experience. Expert Opin Biol Ther. 2020;20(9):1047-1059. doi:10.1080/14712598.2020.1762562
53. Fisher J, Zeitouni N, Fan W, Samie FH. Immune checkpoint inhibitor therapy in solid organ transplant recipients: A patient-centered systematic review. J Am Acad Dermatol. 2020;82(6):1490-1500. doi:10.1016/j.jaad.2019.07.005
54. Bohne F, Martínez-Llordella M, Lozano JJ, et al. Intra-graft expression of genes involved in iron homeostasis predicts the development of operational tolerance in human liver transplantation. J Clin Invest. 2012;122(1):368-382. doi:10.1172/JCI59411
55. Martínez-Llordella M, Lozano JJ, Puig-Pey I, et al. Using transcriptional profiling to develop a diagnostic test of operational tolerance in liver transplant recipients. J Clin Invest. 2008;118(8): 2845-2857. doi:10.1172/JCI35342
56. de la Garza RG, Sarobe P, Merino J, et al. Trial of complete weaning from immunosuppression for liver transplant recipients: Factors predictive of tolerance. Liver Transplant. 2013;19(9):937-944. doi:10.1002/lt.23686
57. Anand AC, Hübscher SG, Gunson BK, McMaster P, Neuberger JM. Timing, significance, and prognosis of late acute liver allograft rejection. Transplantation. 1995;60(10):1098-1103. doi:10.1097/00007890-199511270-00007
58. Fischer L, Saliba F, Kaiser GM, et al. Three-year Outcomes in de Novo Liver Transplant Patients Receiving Everolimus with Reduced Tacrolimus: Follow-Up Results from a Randomized, Multicenter Study. Transplantation. 2015;99(7):1455-1462. doi:10.1097/TP.0000000000000555
59. Shi XL, Mancham S, Hansen BE, et al. Counter-regulation of rejection activity against human liver grafts by donor PD-L1 and recipient PD-1 interaction. J Hepatol. 2016;64(6):1274-1282. doi:10.1016/J.JHEP.2016.02.034
60. Morita M, Fujino M, Jiang G, et al. PD-1/B7-H1 interaction contribute to the spontaneous acceptance of mouse liver allograft. Am J Transplant. 2010;10(1):40-46. doi:10.1111/J.1600-6143.2009.02859.X
61. Au KP, Fung JYY, Dai WC, Chan ACY, Lo CM, Chok KSH. Verifying the Benefits of Radical Treatment in Posttransplant Hepatocellular Carcinoma Oligo-recurrence: A Propensity Score Analysis. Liver Transpl. 2022;28(1):51-64. doi:10.1002/LT.26251
62. Kuo HY, Han MZ, Liao CH, et al. Real-World Comparative Effectiveness of Nivolumab versus Pembrolizumab in Patients with Unresectable Hepatocellular Carcinoma. Pharmaceutics. 2022; 14(11). doi:10.3390/PHARMACEUTICS14112263
63. Yang Y, Chen D, Zhao B, et al. The predictive value of PD-L1 expression in patients with advanced hepatocellular carcinoma treated with PD-1/PD-L1 inhibitors: A systematic review and meta-analysis. Cancer Med. 2023;12(8):9282-9292. doi:10.1002/CAM4.5676

