Impact of Climate Change on Health and CCHF Risks
The Effects of Climate Change on Human Health, Vector-borne Diseases, and Crimean-Congo Hemorrhagic Fever
Fatih Dökmedemir1
- Giresun Central Community Health Center, Türkiye
OPEN ACCESS
PUBLISHED: 31 March 2026
CITATION:Dökmedemir, F., 2026. The Effects of Climate Change on Human Health, Vector-borne Diseases, and Crimean-Congo Hemorrhagic Fever. Medical Research Archives, [online] 14(3). https://doi.org/10.18103/mra.v14i3.7321
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v14i3.7321
ISSN 2375-1924
ABSTRACT
Climate change has direct or indirect effects on health. Direct effects; temperature extremes, heat/cold waves, hurricanes, storms, floods and fires; Indirect effects are vectorial diseases, infections, epidemics, water- and food-borne diseases, air pollution and respiratory diseases, stratospheric ozone depletion and UV Radiation, allergic diseases and field dust. Vector-borne diseases are diseases that occur as a result of the bites of arthropods such as mosquitoes, sand flies, ticks and black flies, which transmit the disease agent to humans. More than half of the world’s population is at risk of vector-borne diseases. Vector-borne diseases are problems that threaten public health and require detailed investigation due to their high mortality rate and high level of disability. Global temperature will increase by 1 to 3.5 C° on average by 2100; In relation to this, it is predicted that vector-borne diseases will spread in a wider geography and their prevalence will increase. Ecological change affects the pattern of Crimean-Congo Hemorrhagic Fever and thus can change the risk of transmission. Since it has a low human-to-human transmission potential, it can only cause small epidemics, but it is a disease that should be considered in terms of public health due to its high mortality rates. The benefit of ribavirin has been proven in prevention studies. The efficacy of ribavirin in treatment is controversial. In addition, the risk of transmission of the disease is high in the process of health care delivery practices and it is likely to result in death due to high viral load. It is a public health requirement that organizations dealing with human and animal health around the world develop protection and treatment methods against this disease.
Keywords:
Climate Change, Human Health, Crimean-Congo Hemorrhagic Fever, Zoonotic Diseases, Health Workers.
INTRODUCTION
EFFECTS OF CLIMATE CHANGE ON HEALTH
While it is certain that climate change affects human health, accurately estimating the scale and impact of many climate-sensitive health risks is still difficult. However, scientific advances are increasingly enabling us to assess the increase in disease and mortality rates linked to global warming and to more accurately determine the risks and scale of these health threats.
Climate change has direct or indirect effects on health. Direct effects; temperature extremes, heat/cold waves, hurricanes, storms, floods and fires; Indirect effects are vectorial diseases, infections, epidemics, water- and food-borne diseases, air pollution and respiratory diseases, stratospheric ozone depletion and UV Radiation, allergic diseases and field dust. Its direct effects were effective in Europe in 2003; It emerged with the heat wave that melted 10% of the glacier in the Alps, causing crops to dry out, forest fires and the death of tens of thousands of people, and Hurricane Katrina in 2005, which was powered by the warming of the Gulf of Mexico and destroyed coastal areas. Indirect effects occur with an increase in malaria, dengue fever, viral infections and water- and food-borne diseases due to changes in the geographical and seasonal characteristics of the vectors. The extension of the pollen season due to the increase in temperature also triggers allergic diseases such as asthma. The health effects of climate change will affect many populations in the coming years and will expose billions of people and creatures to this risk.
DIRECT EFFECTS
Temperature Extremes
Heat waves trigger cardiovascular, cerebrovascular and respiratory deaths. Approximately 70,000 deaths occurred as a result of the heat wave in June 2003. It is predicted that the temperature will increase by approximately 3°C in 2071-2100, resulting in an additional 86,000 deaths each year. In a study conducted in the United States using 22 years of data, 4780 people died due to illnesses caused by extreme temperatures and 1203 people died due to hypothermia. The highest death rates due to heat waves are seen in people over the age of 65. In Italy, in the summer of 2003, those over the age of 65 experienced a higher risk of respiratory disease and a 34% higher risk of death on hot days. When the relationship between hot days between 1983 and 2006 in the Catalonia Region of Spain and 503,389 deaths in the same period was examined, it was observed that some external causes such as cardiovascular and respiratory diseases, mental and nervous system disorders, infection and digestive system diseases, diabetes and suicide increased the relationship.
When the seasonal distribution of deaths in hot countries is examined, it is seen that there are more deaths in winter months than in summer months. In the USA, between 1972 and 2002, a total of 16,555 people, approximately 689 people, died every year due to the effects of low temperatures. Cold weather affects blood pressure, resulting in deaths due to blood clots. It was determined that 28 out of 1 million deaths between 1991 and 2015, especially in Eastern Europe, were due to extreme cold.
In 2020, more than 5.2 million people were affected by the flood that occurred as a result of heavy rain in Jiangxi province in eastern China. For the first time since hurricanes began to be observed in the 1960s, Category 5 hurricanes occurred for 4 consecutive years between 2016 and 2019. Five of the 26 Category 5 hurricanes that have occurred in the Atlantic so far have occurred after 2016. In addition to the Atlantic, record hurricanes and typhoons have been observed in the Pacific in the last 10 years. When compared to the 1960s, it is seen that the number of natural disasters has tripled in the last 10 years and the number of people affected by each disaster is higher.
Hurricanes, Storms, Floods and Fires
Hurricanes, floods, tornadoes, snowstorms, windstorms and droughts are the most important extreme weather events. Another important event is forest fires that occur depending on meteorological conditions. Extreme weather events; It also brings with it problems such as deaths, injuries, post-disaster epidemics, migration and malnutrition. During the 2004 tsunami disaster, post-traumatic stress disorder was observed in 14-39% of children in the coastal areas of Sri Lanka.
INDIRECT EFFECTS
Infection and Epidemic Diseases
World Health Organization emphasizes that, infectious diseases that will be affected by climate change are as follows; bird flu, Crimean-Congo Hemorrhagic Fever (CCHF), cholera, sleeping sickness, ebola, parasites, plague, tuberculosis, lyme disease, harmful seaweed, red fever, yellow fever. Infectious diseases caused mass deaths even in developed and prosperous countries such as the USA until the 21st century. Nowadays, a new pathogen is included in the health literature almost every week, and new infectious diseases are described every two to three years.
Many major pandemics have been recorded in human history, including smallpox, plague, dengue virus, AIDS, influenza, severe acute respiratory syndrome (SARS), West Nile disease, and tuberculosis. The Spanish flu of 1918-1919, which killed more than 50 million people, was recorded as the most devastating epidemic in world history. It was reported that during the SARS CoV epidemic (2003), one of the most dangerous subtypes of the coronavirus family, 8000 infected individuals were recorded worldwide and more than 700 of them died. MERS-CoV 2012, another type of coronavirus that was first seen in 2012 and became a global public health threat with nearly 3000 cases, caused the death of 858 people. The New Coronavirus (COVID-19), which emerged in Wuhan, China in December 2019, was identified in January 2020 as a result of research conducted on patients with respiratory symptoms such as high fever, cough and shortness of breath, and was declared a “pandemic” by the World Health Organization as of March 11, 2020. When this virus has a higher spread rate and infection rate than other viruses in the same category, such as MERS and SARS, both its death rate and economic consequences are too high to compare with these other viruses. Thousands of people have already lost their lives. 60% of new diseases are transmitted from wild animals.
Destruction and alteration of natural ecosystems, decrease in biodiversity, illegal or uncontrolled trade of wild animal species, and bringing together and selling wild and domestic species under unhygienic conditions increase the possibility of transmission of pathogens such as viruses from wild and domestic animals to humans.
With the coronavirus being declared a global epidemic, many opinions have been put forward as to whether the epidemic is linked to climate change. Some scientists state that the origin of coronavirus and climate change are common. According to Dixon, R., as the temperature increases and the glaciers melt, the release of viruses and bacteria frozen in the ice can cause deaths. On the other hand, Harvard University Center for Climate, Health and Global Environment Director Dr. Aaron Bernstein emphasizes that there is no direct evidence that climate change triggered the rapid spread of the coronavirus, but climate change changes our relationship with other species in the world, which is important in terms of health and infection risk. According to Bernstein, as the planet warms, various species living on land and in the sea are heading towards the poles to get away from the heat, which provides the opportunity for pathogens to settle in new hosts by bringing into contact with many species that they normally should not come into contact with. Many of the root causes of climate change also increase the risks of global pandemics. Additionally, deforestation is the largest cause of habitat loss in the world. Habitat loss forces animals to migrate, potentially coming into contact with other animals and humans and sharing microbes.
Inger Andersen, responsible for the United Nations Environment Program (UNEP) declares that, nature is sending us a message through the pandemic and the ongoing climate crisis, as a result of humanity’s damage to the natural world. Andersen emphasizes that the immediate priority is to protect people from the coronavirus and prevent its spread, but in the long term, habitat and biodiversity loss must be combated. Andersen states that 75% of all emerging infectious diseases come from wildlife. Perhaps the only positive effect of the coronavirus has been the mitigation of damage to the environment. However, it is an undeniable fact that this is short-term. Some experts say that the coronavirus will have a negative impact on the climate fight. For example, according to Stanford University Professor Rob Jackson, although the coronavirus crisis has led to a temporary decrease in global carbon emissions, states that will need economic recovery after the epidemic will postpone or even cancel their climate-friendly policies, and this will pose a serious threat to the fight against climate change in the long term. Although it is not yet known for certain whether the coronavirus is a result of climate change, it is thought to be a threat to the climate change action plan in the long term.
Water and Foodborne Diseases
Climate change affects the conditions of “safe drinking water, adequate food and safe shelter”, which are among the social and environmental determinants of health. According to WHO; Between 2030 and 2050, 250,000 increased deaths are expected each year as a result of malnutrition, malaria, diarrhea and heat stress caused by climate change.
Fifty five percent of foodborne diseases occur due to bacteria (Salmonella) and 33% due to viruses. Waterborne diseases; These are mosquito-borne diseases such as typhoid, cholera, diarrhea, skin and eye infections (scabies, trachoma), diseases carried by fleas (epidemic typhus), cercacial dermatitis, malaria and dengue. While the rate of disease spread after rainfall was 51% between 1948 and 1994, this rate increased to 68% in the 90s. It is estimated that foodborne diseases will increase by 5-20% by 2050.
Access to water is one of the most important health issues in the world. There are more than 2 billion people living in arid regions in the world and suffering from diseases caused by malnutrition and lack of access to clean water. In the future, it will be necessary to cope with food and water-borne and food-transmitted diseases, as well as health problems arising from water scarcity.
Most of the important viruses (enteroviruses, rotaviruses, hepatitis A virus and norovirus) are spread through epidemic diseases, water and food. When the analysis of waterborne diseases is made, open water surfaces carry a greater risk in the spread of waterborne diseases after rainfall. Since flood waters pollute drinking water, they indirectly affect the spread of diseases. Acute diarrheal diseases are the leading cause of infant mortality in Eastern Europe. The most important waterborne disease in Western Europe is Cryptosporidium, an intracellular parasite that settles in the respiratory system. It caused an epidemic that affected 400,000 people in the United States in 1993. In England, 5000 cases are reported every year. It is especially seen in seasons with heavy rainfall. Another problem in temperate climates is cercacial dermatitis. Its intermediate hosts are slugs and they are one of the important waterborne parasites in Europe. It is thought that climate change will affect water resources, especially sanitation.
Every year, 210 million people in the USA are admitted to hospital due to gastrointestinal disorders and 6000 of them die. In recent years, 375 million cases of diarrhea have been recorded in hospital records. It is predicted that diarrheal cases in the USA will increase proportionally regardless of age. Most diseases (germs or viruses) that occur through food and water are transmitted through the mouth and feces. An increase in infectious diseases caused by marine microorganisms has also been determined. Increasing sea surface temperature, wind, water currents, storms affect marine ecosystems. Various food poisoning cases have been observed in the USA due to consumption of fish and seafood. With climate change, Pacific-origin, tropical and poisonous lionfish have crossed the Red Sea and started to establish populations in the Mediterranean ecosystem. This invasive species is also seen in Turkish waters and causes serious damage to the Mediterranean ecosystem.
With climate change, diseases whose borders are currently known are expected to cross borders. Chikungunya virus infection is thought to be limited to tropical regions. However, its occurrence in Europe is evidence that the borders have changed. With climate change, not only human mobility, but also the mobility and direction of movement of natural life will change. Changes in bird migrations and changes in the cycle of avian influenza viruses can be given as examples.
Air Pollution and Respiratory Diseases
Air pollution is responsible for an estimated 7 million deaths annually. Air pollutants are mainly; It plays a role in deterioration in respiratory functions, increase in respiratory system diseases, facilitating disease exacerbations in people with chronic respiratory system and heart diseases, and increase in the incidence of cancer and premature death.
Asthma symptoms may worsen due to the increase in fungal spore and pollen density in the air. Again, temperature increases alone can lead to early deaths in the elderly and those with respiratory pathology such as COPD. Some diseases such as upper respiratory tract infections, flu, sinusitis, asthma, bronchitis, chronic bronchitis and even pneumonia are more common in regions where polluted air density is evident. Those most affected by air pollution are children under the age of five, chronic patients (such as asthma, bronchitis, COPD, cardiovascular disease, diabetes) and the elderly. Factors such as low social status (such as homeless people), inability to reach a healthcare facility (e.g. disaster situations), smoking/alcohol habits, and nutritional disorders further increase the impact on these groups.
Effects of Ultraviolet Radiation
Ultraviolet radiation (UVR) is caused by the sun and helps the body produce vitamin D. As with everything, too much is harmful. Depending on the wavelength, it has side effects ranging from skin aging to cancer. Direct health effects of UV rays occur through skin cancers (malignant melanoma), eye diseases, sunburns, effect on the immune system, and indirect effects occur through its harmful effect on DNA. Based on WHO, United Nations Environment Program (UNEP) estimates; It states that a 10% decrease in the amount of stratospheric ozone will result in an additional 300,000 cases of non-melanoma and 4,500 cases of melanoma skin cancer and 1.7 million additional cases of cataracts worldwide each year.
Allergic Diseases
Climate change causes many allergic disorders through pollen at the beginning of spring. The most important of these is hay fever. Increasing CO2 concentration and temperature also causes hay fever to increase. Studies have shown that ragweed pollen counts increase in parallel with increases in temperature and CO2. Additionally, increases in temperature and humidity affect mold concentration. Changing pollen season and duration also affects allergic disease attacks. It can be expected that allergic complaints will increase with the increase in allergen concentration in the air. With the increase in pollen concentration, a worsening of allergic rhinitis symptoms is also observed.
Sahara Dust
One of the largest sources of dust affecting the entire northern hemisphere is the Sahara desert in northwest Africa. Every year, thousands of tons of dust rise from the Sahara and are blown towards the Mediterranean and Aegean Coasts and the Atlantic Ocean, reaching the Caribbean and America. Considering that the size of Saharan dust particles is smaller than 2.5 μg in diameter, it is clear that these particles will easily settle in the lungs. Many epidemiological studies in America and Europe have proven that exposure to dust of this size causes deaths due to respiratory diseases. In addition, these dusts carry microorganisms such as bacteria, viruses, fungi and chemicals found in the soil. Therefore, its negative effects on health are increasing.
Mental Illnesses
Rising temperatures, heat waves, floods, hurricanes, droughts, fires, forest loss and glaciers, the disappearance of rivers, desertification can directly and indirectly cause physical and mental human pathologies. Exposure to natural disasters related to climate and weather conditions can lead to mental health consequences such as anxiety, depression and post-traumatic stress disorder. A significant portion of people affected by these events develop chronic psychological dysfunction. Children, pregnant and postpartum women, people with pre-existing mental illness, the economically disadvantaged, the homeless, and disaster first responders are at high risk for mental health outcomes. Overheating increases both physical and mental health problems and increases the risk of illness and death in people with mental illness. However, there is a serious lack of psychiatric studies on climate change-related mental disorders.
Vector Borne Diseases
Vector-borne diseases are diseases that occur as a result of the bites of arthropods such as mosquitoes, sand flies, ticks and black flies, which transmit the disease agent to humans. Vectors are generally affected by humidity and temperature. A change in vector distribution will also affect human health. Increasing average temperature will affect the distribution and abundance of vectors and cause pathogens to multiply faster.
Temperature and surface waters have significant effects on vector-borne infectious diseases. Mosquito species that spread viral diseases such as dengue and yellow fever and malaria are of particular importance. Mosquitoes need to use stagnant water to breed, and adults need a moist environment to survive. Malaria is an important vector-borne infection affecting more than 100 countries in the world. It is noted that 40% of the world’s population is at risk for malaria and that there is a synergistic effect between climate change and malaria spread. Some changes in land use directly affect disease exposure. Logging in tropical forests has created environments conducive to mosquito-borne diseases such as malaria and dengue fever. However, social economic developments are effective in limiting the spread of the disease.
Approximately 150,000 to 200,000 cases annually in the world are hospitalized and treated with the diagnosis of “hemorrhagic fever with renal syndrome”. Most of these cases are caused by Hanta virus and Seoul virus and are seen in China. In America, approximately 200 cases a year are diagnosed with “Hantavirus pulmonary syndrome”. The number of hantavirus cases has reached levels high enough to pose a public health threat globally. Hantavirus pulmonary syndrome, tick-borne enfesalitis, Lyme disease are associated with rodents. The mouse population in the world has increased greatly in recent years. Mice are carriers of leptospira and cause Weil’s disease.
Pigs, chickens, ducks, etc. that do not pass from person to person. The density and severity of influenza virus (H3N2, H1N1), transmitted through animals, vary depending on weather and climate conditions. Climate change is one of the factors that facilitates the proliferation of the tick population and, accordingly, increases the incidence of tick-borne diseases. CCHF is a fatal viral infection transmitted from ticks to humans and reported from approximately 30 countries. Humans are most often infected through tick bites, contact with patients in the acute phase of the disease, or contact with the blood and tissues of viremic animals. The clinical findings of the disease follow a dramatic course, including bleeding, myalgia and fever.
As wildlife is invaded, the number of these diseases will continue to increase. More than 40% of tick-borne pathogens have been discovered in the last two decades. Lime disease is transmitted to humans and other animals from ticks. According to research, the risk of contracting this disease is much higher in areas with poor vertebrate animal diversity, such as forests smaller than two hectares and habitats with significantly damaged integrity.
With climate change, some infectious diseases those prevalences dropped in Türkiye have begun to be more diagnosed nowadays. West Nile Virus (WNV) Infection, which was first detected in the West Nile region of Uganda in 1937 and is most common in Africa, West Asia and the Middle East, has started to be seen in our country as of August 2010. This mosquito-borne disease is usually seen during summer and early autumn.
The distribution of schistosomiasis, a water-related parasitic disease whose intermediate host is the water snail, may be affected by climatic factors. The length of dry seasons and the density of the human population in a region of Brazil are the most important factors in the distribution and abundance of Schistosomiasis. There was an inverse relationship between prevalence rate and length of dry seasons over larger areas. Recent studies conducted in China show that schistosomiasis has increased in response to warming in the past decade.
More than half of the world’s population is at risk of vector-borne diseases. Vector-borne diseases threaten public health and require detailed examination because they cause high mortality rates and high levels of disability. Vector-borne diseases are an important problem for developing countries in tropical regions today. Global temperature will increase by an average of 1 to 3.5 C° by 2100; It is estimated that vector-borne diseases will spread in a wider geography and their prevalence will increase accordingly. Therefore, analyzing vector distributions; It is necessary to predict their distribution against current and future climate scenarios and to develop intervention programs.
Ecological niche models; It aims to characterize the environmental conditions required by a species for the long-term maintenance of the existing population without having to migrate. Defining these conditions; It enables the determination of the potential distribution of species. Models calibrated using today’s data can also be projected into future climate scenarios.
Since the health effects of global climate change began to be investigated, vector-borne diseases have become a focal point due to the sensitivities of arthropod vector biology and the increased prevalence of vector-borne pathogens with climate change.
Increasing human population, landscape, agricultural practices, and habitat fragmentation cause climate change, which leads to an increase in infectious diseases and the emergence of new infectious diseases.
By definition, global climate change; It is a global phenomenon that emphasizes long-term changes in meteorological events such as temperature, precipitation and wind. The expected change in the world’s climate is the temperatures that occur especially in higher latitudes. Additionally, changes in precipitation make some regions prone to drought. Increasing climate change and meteorological events (severe storms, extreme heat, heavy rainfall, etc.) drive the emergence of vector-borne diseases in several ways:
- Polar spread of vector and vector-borne diseases as weather warms in temperate regions. Additionally, with warming weather, the equatorial boundaries of species are shrinking towards the poles.
- The possibility of tropical and subtropical vector-borne diseases spreading to temperate regions and creating endemicity.
- Increased vector and vector-borne pathogen survival with increasing temperatures in recipient areas
- Increase in vector and vector-borne pathogens in tropical and subtropical regions
- Increased spread of tropical and subtropical vector-borne pathogens with increased human migration due to climate change
- With increasing climate change, the re-emergence of endemic vector-borne diseases and the change in the genotypes of vector-borne pathogens.
Effect of Climate-Related Environmental Variables on Physiological and Ecological Characteristics of Hosts, Pathogens and Vectors
Environmental change can have direct effects on physiological and ecological traits of species, such as pathogen resistance, life cycle parameters, or geographic range. Responses of species to environmental change include physiological acclimation and evolutionary adaptation.
Climate change affects geographic distribution of hosts, vectors and pathogens
Climate variables; It acts as an environmental filter that has a direct impact on the geographic distribution of hosts, pathogens and vectors. The expansion of the suitable temperature range in which a species can survive causes the geographical distribution of this species to increase. However, climate change factors cause not only geographical distribution expansions but also geographical distribution shifts. For example, malaria in tropical countries; It is less common in high altitude areas. Because relatively low temperatures in mountainous regions are not suitable for vector life. With increasing temperatures due to global warming, the presence of malaria is expected to increase in tropical mountainous regions.
Climate change affects life cycle characteristics of hosts, vectors and pathogens
Climate variables; can influence disease dynamics by modulating the survival, development, and reproduction of hosts, pathogens, and vectors. The direct consequence of changing climatic conditions is reduced survival of the pathogen in the environment. For example, the survival time of Avian Influenza A virus (AIV), which is transmitted through water contaminated with feces, varies depending on water temperature. An inverse relationship was found between increasing water temperature and AIV survival. Therefore, AIV cases are expected to decrease with increases in water temperatures.
Effects of climate change on the physiological capacities of hosts, vectors and pathogens
Changes in host susceptibility to a pathogen depend on changes in the host immune system. Although ecological factors such as spatial distribution affect the host and pathogen together, the host immune system is decisive in the occurrence of infection. Climate change is known to modulate the immune system. This affects the pathogen’s ability to infect the host. Additionally, climate effects on host immunity may also have implications for pathogen evolution. Since the immune system is an important selection mechanism for pathogens, climate-induced deficiencies in host immunity may alter pathogen evolution and lead to the emergence of new virulent strains.
One of the ecological mechanisms that increase host susceptibility to pathogen infection and interaction with pathogens is changes in animal distribution and interaction. Seasonal migration of animals is thought to reduce parasite prevalence in host species. However, challenging conditions such as drought cause animals to gather in water bodies and pathogen transmission increases.
Ecology and Crimean-Congo Hemorrhagic Fever Disease
In recent years, new outbreaks of CCHF have emerged in many Balkan countries, southwestern Russia and Türkiye. Türkiye is experiencing the largest epidemic recorded to date, with the number of cases increasing since 2002. Climate change, which has a significant impact on the reproduction rate of hyalomma ticks, which are vectors, plays a major role among the reasons for the re-emergence of CCHF. The fact that many animals live as hosts in the geography where CCHF occurs provides a favorable environment for the disease to spread in these regions.
Temperature and humidity are the main determinants of the geographic distribution and habitat of ticks. Increased temperature has an impact on annual and seasonal survival patterns of tick populations; It especially affects life cycles by changing them. For the presence of the CCHF factor, the land surface temperature must be at least 18.91 degrees. However, a land temperature of 18.16 degrees does not allow the CCHF agent to survive. These temperature limits explain the consistent relationship of CCHF with high temperature and season. Compared to other ticks, hyalomma ticks are better adapted to hot, dry climates. This feature causes hyalomma ticks to become active in late spring and remain continuously active throughout the summer and into early autumn. Microecological factors (e.g., vegetation, microclimate) can affect tick host dynamics and provide an explanation for the heterogeneity that may occur in infection levels even in endemic regions.
Humans influence the ecology of CCHF and thus may alter the risk of transmission. Decrease in agricultural activities, changes in land use, decrease in rabbit hunting, reshaping of animal husbandry, conflicts and wars are associated with CCHF epidemics in Russia, Bulgaria, Kosovo and Türkiye.
Crimean-Congo Hemorrhagic Fever
Zoonotic diseases are infectious diseases that can be transmitted between animals and humans. Approximately 60% of all human diseases and about 75% of emerging infectious diseases are of zoonotic origin. These types of diseases have high impacts on human health, livelihoods, animals and ecosystems.
Epidemiological Pattern
Agent Crimean-Congo hemorrhagic fever virus (CCHFV) is an enveloped RNA virus belonging to the Nairovirus genus of the Bunyaviridae family. Nairoviruses are enveloped and therefore susceptible to external conditions. They cannot survive without a host. The virus is rapidly inactivated at 56°C in 30 minutes and by ultraviolet light, and is sensitive to 1% hypochlorite and 2% glutaraldehyde.
Contagion The virus exists through a tick-vertebrate life cycle. The primary vectors are ticks of the genus Hyalomma. Ticks of the genus Hyalomma serve as both reservoirs and vectors of the CCHF virus. Wild and domestic mammals, including farm animals such as sheep, goats, and cattle, are secondary hosts. Transmission of the virus to humans occurs through tick bites and contact of mucous membranes and open wounds with the blood and body fluids of viremic animals.
History CCHF was first observed among Soviet soldiers in the summer of 1944. The disease was called Crimean-Congo hemorrhagic fever. Later, in 1956, the Congo virus was detected in a febrile patient in Zaire. It was understood in 1969 that both viruses were the same virus. After that, the disease was called Crimean-Congo hemorrhagic fever.
Geographic and Epidemiological Patterns
The main risk factors for the disease are occupations related to animal husbandry (especially livestock activities involving sheep and goats), and agricultural and agropastoral work. These occupations indirectly involve tick exposure. Tick exposure refers to direct physical contact, tick removal from humans and animals, and exposure to ticks in the home environment. It is seen less frequently in healthcare workers (doctors and nurses), veterinarians, and butchers. Many studies show that increasing age increases the risk of transmission. A study conducted in Türkiye found the highest seropositivity rate among those aged 31-40 years. Since the majority of participants were male, gender roles could not be assessed. However, a history of tick bites is a significant risk factor. According to the study, the most important risk factors for transmission are livestock farming and animal-related occupations.
Since its initial identification, the disease has been reported in numerous countries, including Zaire, Uganda, Saudi Arabia, the United Arab Emirates, Pakistan, Europe, Russia, Iran, and South Africa. Sporadic cases and large outbreaks have also been detected in various regions such as Kosovo and Kenya.
In the last decade, CCHF has started to reappear in the Balkan countries, Türkiye, the southern and western regions of the Russian Federation, and Ukraine. The reasons for the re-emergence of CCHF include agricultural practices and hunting activities, host-tick-virus dynamics, and anthropogenic factors such as animal movements.
Global Situation
CCHF is still endemic in Bulgaria, where a major outbreak occurred in 1954 and 1955. Between 1953 and 2008, 1,568 CCHF cases were reported in Bulgaria, with a general fatality rate of 17%. Endemic areas include Shumen, Razgrad, Veliko Tarnovo, Plovdiv, Pazardzhik, Haskovo, Kardzhali, and Burgas. Blagoevgrad, located near the Greek border in Bulgaria, was known until recently for its low CCHF endemic rate. Outbreaks of CCHF were also recorded in Albania in 2001 and 2003, and in Kosovo in 2001. After 27 years without any human cases, CCHF reappeared in the southwestern regions of the Russian Federation in 1999. In the Russian Federation, more than 1300 clinical cases were detected between 2000-2009 with a mortality rate of 3.2%.
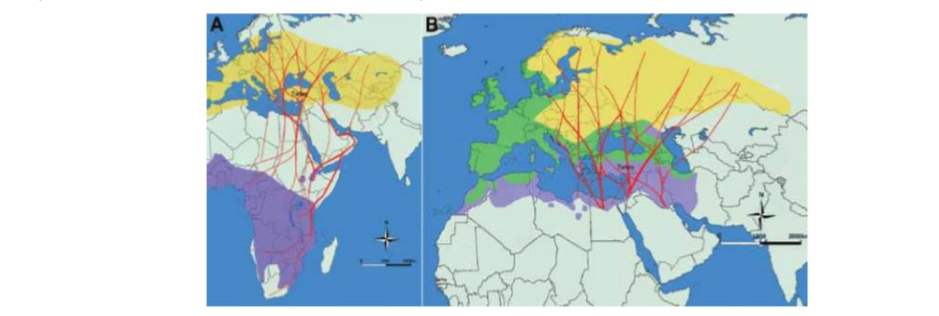
New cases were also identified in Georgia, Kazakhstan, Tajikistan, Iran, and Pakistan in 2009. The geographical distribution of CCHF corresponds to the geographical distribution of ticks of the genus Hyalomma. H. Marginatum, the main CCHF virus vector in Europe, is found in Albania, Bulgaria, Cyprus, France, Greece, Italy, Kosovo, Moldova, Portugal, Romania, Russia, Serbia, Spain, Türkiye, and Ukraine. The tick was also detected in the Netherlands and Southern Germany in 2006. Given the wide distribution of the CCHF vector, the large number of animals that can serve as hosts, and the favorable climatic and ecological conditions in many European countries bordering the Mediterranean, it is possible that the spread of CCHF disease will increase.
Türkiye
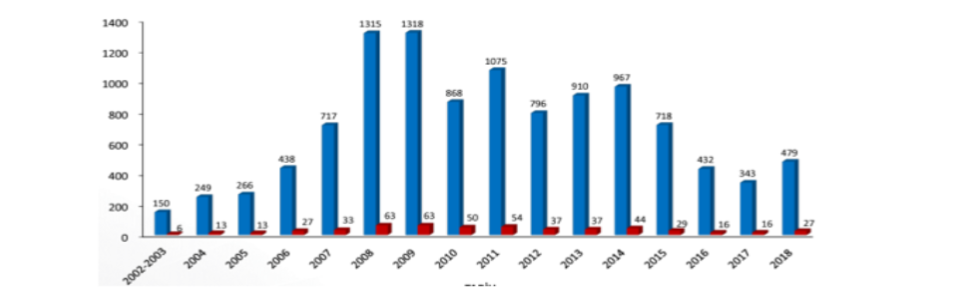
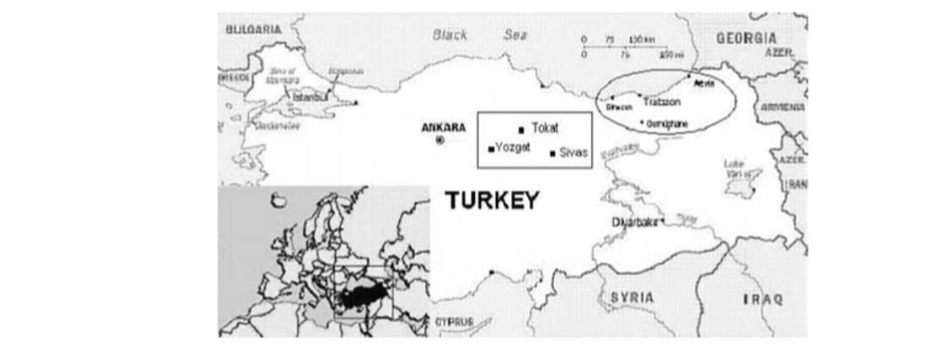
In Türkiye, Tokat, Sivas, and Yozgat have become the epicenters of the disease. Cases in these regions have been the subject of ongoing epidemiological studies. In 2002, the year CCHF was first detected in Türkiye, no deaths were observed among suspected cases. However, in 2003, 2 out of 10 detected cases died due to severe visceral bleeding. In various studies conducted in Türkiye, the seroprevalence of CCHF has been found to be between 1.6% and 19.6%. However, the disease burden in the world and in Türkiye is much higher than estimated due to undetected cases. In addition, undetected cases have prevented adequate and reliable epidemiological studies from being carried out and have led to the disease being underestimated. Yet, the disease is a global public health threat.
| Years | Population | Number of Cases | Morbidity Rate (100,000) | Death Toll | Mortality Rate (1,000,000) |
|---|---|---|---|---|---|
| 2008 | 71.517.100 | 1315 | 1.84 | 63 | 0.88 |
| 2009 | 72.561.312 | 1318 | 1.82 | 63 | 0.87 |
| 2010 | 73.772.988 | 868 | 1.18 | 50 | 0.68 |
| 2011 | 74.724.269 | 1075 | 1.44 | 54 | 0.72 |
| 2012 | 75.627.384 | 796 | 1.05 | 37 | 0.49 |
| 2013 | 76.667.864 | 910 | 1.19 | 37 | 0.48 |
| 2014 | 77.695.904 | 967 | 1.24 | 44 | 0.57 |
| 2015 | 78.741.053 | 718 | 0.91 | 29 | 0.37 |
| 2016 | 79.814.871 | 432 | 0.54 | 16 | 0.20 |
| 2017 | 80.810.525 | 343 | 0.42 | 16 | 0.20 |
Conclusion and Recommendations
Unlike other viruses, human-to-human transmission of CCHF is typically limited. Therefore, when considering countermeasures, priority should be given to blocking the routes of spread. Various factors in the transmission dynamics of the CCHF virus, including a complex transmission cycle involving both ticks and vertebrate hosts, are often overlooked by current strategies but owe their existence to the One Health approach necessary for disease prevention and control. This means elucidating the transmission routes, knowing and implementing preventive measures.
Possible causes of the emergence and/or re-emergence of CCHF include anthropogenic factors such as changes in agricultural activities, habitat fragmentation and the import of infected animals and ticks. The potential impact of climate change on the spread of the disease should also not be overlooked.
Recent increases in case numbers in Türkiye and southwestern Asia have demonstrated the public health impacts of this re-emerging disease. Its potential for zoonotic transmission to humans is high due to its wide geographical distribution and persistent circulation in nature. Unlike other emerging viruses, human-to-human transmission of CCHF after transmission via animal hosts or ticks is limited and results in small outbreaks. The importance of limiting spread for human health supports prioritizing measures to mitigate its potential for transmission.
The “One Health” concept recognizes the interconnectedness of human, animal, and environmental health; it is a disease prevention approach that strives to address human health within a broader context, making changes and intervening in an interdisciplinary manner. The World Organisation for Animal Health (OIE) and the World Health Organization (WHO) have published a health-based framework entitled “Triple Guidelines for Addressing Zoonotic Diseases.” A significant portion of the guidelines focuses on a multi-sectoral disease control and health approach.
The Guide’s Contents:
I) Strategic planning and emergency preparedness,
II) Monitoring and information sharing on zoonotic diseases,
III) Coordinated research and response,
IV) Joint risk assessment for zoonotic disease prevention measures,
V) Risk reduction, risk communication and community involvement,
VI) Workforce development.
This guide provides a comprehensive analysis for the prevention of zoonotic diseases. An effective surveillance system provides data on disease incidence and prevalence. It provides information about high-risk behaviors or practices at the vector/host/human stages and guides the development and implementation of preventive measures. Due to the wide geographical range of Hyalomma ticks, their multi-stage life cycle and corresponding variability, surveillance values should be coordinated across many sectors to provide information about CCHFV in humans, animals and ticks. Therefore, all organizations working in the human and animal health sectors, including WHO, Food and Agriculture Organization (FAO) and OIE, should be in communication with each other.
Recently, five levels have been proposed to influence country-specific CCHF surveillance systems. The levels are based on case incidence, potential for disease transmission to humans, and the availability of systems.
- Level 1 country: The virus is endemic in these countries.
- Level 2 country: Contains cases of irregular autochthonous humans.
- Level 3-4 country: There are no documented human cases in the country, but ecological data, including the presence of Hyalomma ticks, suggest that cases may occur.
- Level 5 countries: Countries for which no information is available.
CCHF surveillance strategies naturally differ between different country-specific levels. For example, level 1-2 countries require a robust strategy as human cases may be disorganized and there may be a lack of public awareness. In contrast, level 5 countries should focus their surveillance particularly on the presence of the tick vector and not initially on identifying seropositive individuals among humans.
Animal movements can cause the sample collection site to differ from the encounter site, making the interpretation of serological data difficult. Furthermore, these ticks feed on small mammals and then move to a different large mammal host for adulthood, exhibiting a two-host life cycle. This also complicates serological monitoring.
Although the role of other tick species in the transmission and management of the CCHF virus is not well-defined, the virus has been detected in other tick species (Amblyomma, Rhipicephalus, Dermacentor, Ixodes). Assessing vector adequacy is crucial, particularly for understanding the potential for CCHF occurrence in new geographic regions where non-Hyalomma ticks may support the transmission cycle or play a role in cryptic transmission.
There is no globally accepted vaccine against CCHF. However, an inactivated vaccine of Soviet Socialist Republics origin has been used in Bulgaria since 1974.
CCHF virus is a high-risk pathogen for public health. Due to the high morbidity and mortality rates associated with CCHF infection, the development of medical countermeasures for the prevention and treatment of the disease is an urgent necessity. Healthcare workers in endemic areas are at risk of infection after unprotected contact with infected blood and body fluids. With climate change, the threat of CCHF to public health is increasing, and the disease needs to be given greater attention.
Conflict of Interest Statement: None.
Funding Statement: None.
Acknowledgements: None.
ORCID ID: 0000-0001-8998-8585
References:
- Nur, N., Sümer, H., “Kentleşme, Küresel Isınma ve İklim Değişikliğinin Sağlık Üzerindeki Etkileri”,
www.erciyes…dergisi.org (18.05.2020) - ACA, https://www.eea.europa.eu/tr/isaretler/isaretler-2015/gorusme/iklim-degisikligi-ve-insansagligi/download.pdf (16.04.2020)
- Eker EO., “İklim Değişikliğinin İnsan Sağlığına Etkileri, İklim Değişikliği Alanında Ortak Çabaların Desteklenmesi Projesi (İklimİN)”, Çevre ve Şehircilik Bakanlığı, 59 syf.
- Çelik, S., Bacanlı, H., Görgeç, H., “Küresel İklim Değişikliği ve İnsan Sağlığına Etkileri”, Ankara
- Cianconi, P., Betro S., Janiri L., “Impact of Climate Change on Mental Health: A Systematic Descriptive Review”, Front. Psychiatry, 06 March 2020,
https://doi.org/10.3389/fpsyt.2020.00074, Rome, Italy - Evei Kiraz, E. D., “İklim Değişikliğinin İnsan Sağlığına Etkileri, İklim Değişikliği Alanında Ortak Çabaların Desteklenmesi Projesi (İklimİN)”, Çevre ve Şehircilik Bakanlığı, 59 syf.
- ACA, “Avrupa Çevre Durumu ve 2020 Görünümü: SOER 2020 Raporu”, SOER, 2020,
https://www.eea.europa.eu/soer/2020 (23.04.2020) - Özkocak, V., Koç, F., Gültekin, T., “Pandemilere Antropolojik Bakış: Koronavirüs (Covid-19) Örneği”, Araştırma Makalesi, eISSN: 1308-2140, 14 syf.
- WWF., “Doğanın Yok Oluşu ve Pandemilerin Yükselişi, İnsanların ve Gezegenin Sağlığını Korumak”, 22 syf., İtalya
- IRAM., https://iramcenter.org/koronavirus-ve-iklim-degisikligi/ (25.06.2020)
- Sabancı Univ.-CDB., “İklim Değişikliği ve Coronavirus (COVID-19) İlişkisi”,
https://cdpturkey.sabanciuniv.edu/tr/content/iklim-coronavirus (15.06.2020) - TKHHÖKP., “Türkiye Kronik Hava Yolu Hastalıklarını Önleme ve Kontrol Programı, Türkiye’nin Hava Kirliliği ve İklim Değişikliği Sorunlarına Sağlık Açısından Yaklaşım”, ISBN: 978-975-590-342-2, Sağlık Bakanlığı, No: 811, Ankara
- Çimen, M., Öztürk, S., “Küresel Isınma, İklim Değişikliğinin Solunum Sistemi Üzerine Etkisi ve Büyükşehir Bronşiti”, www.tip.fusabil.org (15.05.2020)
- Weir K., “Climate change is threatening mental health”, July/August 2016, Vol 47, No. 7, Print version: page 28
- Türkiye Cumhuriyeti Sağlık Bakanlığı, “Ülkemizde İklim Değişikliğinin Sağlık Üzerine Olumsuz Etkilerinin Azaltılması Ulusal Programı ve Eylem Planı”
- Tekbaş, F., Vaizoğlu, S., Oğur, R., Güler, Ç., “Küresel Isınma, İklim Değişikliği ve Sağlık Etkileri”, ISBN: 1307-9649, Ankara
- Ergönül, Ö., “Türkiye’de Yeni Bir Enfeksiyon: Kırım Kongo Kanamalı Ateşi”, Sted, cilt 15, sayı 6, ss. 98-106
- Luis E. Escobar, Daniel Romero-Alvarez, Renato Leon, Manuela A., Lepe-Lopez., “Declining Prevalence of Disease Vectors Under Climate Change”, DOI: 10.1038/srep39150
- Ogden NH, Lindsay LR., “Effects of Climate and Climate Change on Vectors and Vector-Borne Diseases: Ticks Are Different”,
http://dx.doi.org/10.1016/j.pt.2016.04.015 - Taştan R., Özer C., Ocuç A., “Vektörlerle Bulaşan Hastalıklar: İklim Değişikliği ve Küreselleşmenin Tetiklediği Yeni Tehdit”,
https://www.researchgate.net/publication/332718308 - Milena G., Marie-P., Ryser-D., Thomas W., Helmut S., “Climate change and infectious diseases of wildlife: Altered interactions between pathogens, vectors and hosts”, Current Zoology 59 (3): 427–437, 2013
- Maltezou HC, Papa A., “Crimean-Congo hemorrhagic fever: Risk for emergence of new endemic foci in Europe?”, Travel Medicine and Infectious Disease (2010) 8, 139–143
- Ari HO, İşlek E., Özatkan Y., MK Bilir., Karakaş F., “Türkiye’de Zoonotik Hastalıkların Hastalık Yükü ve Maliyeti”, 2020,
https://www.tuseb.gov.tr/tuseb/uploads/yayinlar/makaleler/pdf/08-06-2020_5ede03c557655_tusebrapor01_turkiyede_zoonotik_hastaliklarin_hastalik_yuku_ve_maliyeti.pdf - Karti SS., Odabaşı Z., Korten V., Yılmaz M., Sonmez M., Caylan R., et al., “Crimean-Congo Hemorrhagic Fever in Turkey”,
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC330426/ - Zohaib A., Saqib M., Athar MA., Hussain MH., Sial A., Tayyab MH., “Crimean-Congo Hemorrhagic Fever Virus in Humans and Livestock, Pakistan, 2015–2017”, doi: 10.3201/eid2604.191154
- Ozcan E., Ozkul A., “Investigation of Crimean-Congo hemorrhagic fever virus in ruminant species”
- slaughtered in several endemic provinces in Turkey / Archives of Virology (2020) 165:1759–1767
- https://doi.org/10.1007/s00705-020-04665-9
- Albayrak Z. An investigation of the seroprevalence of Crimean-Congo Hemorrhagic Fever and Lumpy Skin Disease in domesticated water buffaloes in northern Turkey
https://www.researchgate.net/publication/339618149 - Kar S, Rodriguez SE, Akyildiz G, Crimean-Congo hemorrhagic fever virus in tortoises and Hyalomma aegyptium ticks in East Thrace, Turkey: potential of a cryptic transmission cycle
https://parasitesandvectors.biomedcentral.com/articles/10.1186/s13071-020-04074-6 - Arteaga LA, Sardon MA, Bellido JLM, Santigo MBV., Lista MVC., Aban JL., et al., Seroprevalence of Crimean-Congo hemorrhagic fever in humans in the World Health Organization European region: A systematic review
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7067482/ - Ergönül Ö, Keske S., Çeldir MG, Kara İA., Peshnichnaya N., Abuova G., Systematic Review and Meta-analysis of Postexposure Prophylaxis for Crimean-Congo Hemorrhagic Fever Virus among Healthcare Workers
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6106438/ - Ahmed A., Tahir MJ., Siddiqi AR., Dujaili J., Potential of Crimean-Congo Hemorrhagic Fever outbreak during Eid-Ul-Adha Islamic festival and COVID-19 pandemic in Pakistan
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7362032/ - Türkiye Cumhuriyeti Sağlık Bakanlığı, Halk Sağlığı Genel Müdürlüğü, Zoonotik ve Vektörel Hastalıklar Daire Başkanlığı
https://hsgm.saglik.gov.tr/tr/zoonotikvektorel-kkka (07.12.2020) - Ergönül Ö, Çelikbaş A, Dokuzoğuz B, Eren Ş., Baykam N., Esener H., Characteristics of patients with Crimean-Congo hemorrhagic fever in a recent outbreak in Turkey and impact of oral ribavirin therapy. doi: 10.1086/422000. Epub 2004 Jul 2.
- Leblebicioğlu H., Ercioglu, Yavuz KE, Hokelek M., Acici M., Yilmaz H., Role of Migratory Birds in Spreading Crimean-Congo Hemorrhagic Fever, Turkey
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4111188/ - Güneş T, Engin A, Poyraz O, Elaldi N., Kaya S., Dokmetas I., et al., Crimean-Congo hemorrhagic fever virus in high-risk population, Turkey / doi: 10.3201/eid1503.080687
- Sorvillo TE, Rodriguez SE, Hudson P., Carey M., Moruguez LL., Siprooulou CF., et al., Towards a Sustainable One Health Approach to Crimean-Congo Hemorrhagic Fever Prevention: Focus Areas and Gaps in Knowledge / DOI: 10.3390/tropicalmed5030113
- Tipih T., Burt FJ., Crimean–Congo Hemorrhagic Fever Virus: Advances in Vaccine Development BioResearch Open Access. Jun 2020. 137–150
http://doi.org/10.1089/biores.2019.0057 - Maltezou HC, Andonova L, Bouloy M, Ergönül O., Jogenain F., Kalvatchev N., et al., Crimean-Congo hemorrhagic fever in Europe: current situation calls for preparedness / Euro Surveill. 2010 Mar 11;15(10):19504
- Celikbas AK, Dokuzoguz B, Baykam N., Gok SE., Eroglu MN., Midilli K., et al., Crimean-Congo Hemorrhagic Fever among Health Care Workers, Turkey
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3844981/ - Center for Disease Control and Prevention. CDC 24/7.
https://www.cdc.gov/vhf/crimean-congo/exposure/index.html - Aydın H, Uyarık MH, Karamesa M., Sozdutmaz I., Timurkan MO., Gulen A., Serological Investigation of Occupational Exposure to Zoonotic Crimean-Congo Hemorrhagic Fever Infection
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7311122/

