Impact of COVID-19 on Systemic Autoinflammatory Diseases
Effects of the COVID-19 Pandemic in Patients with Systemic Autoinflammatory Diseases. An international survey
S. Nazzar, MD¹; L. Nandini Moorthy, MD (MBBS), MS²; Jennifer Tousseau³; Silvia Lapidus, MD⁴; Leanne Mansfield, MDS; Marinka Twilt, MD, PhD⁶; Grant Schubert, MD, PhD⁷,⁸; Maria J. Gutierrez, MD, MHS, MBA⁹; Smriti Mohan, MD¹⁰; Saskya Angevarell¹¹; Karen Dunant, RN, BSN¹²; Fatma Dedeoglu, MD¹² for the CARRA PFAPA/AutoInflammatory Working Group
-
Division of Rheumatology, University of Central Florida, Nemours Children’s Hospital, Orlando, FL, USA
-
Department of Pediatrics, Rutgers-Robert Wood Johnson Medical Center, New Brunswick, NJ, USA
-
The Autoinflammatory Alliance, San Francisco, CA, USA
-
Division of Pediatric Rheumatology, Hackensack University Medical Center and Hackensack Meridian School of Medicine, Hackensack, NJ, USA
-
Department of Pediatrics, University of Wisconsin School of Medicine and Public Health, Madison, WI, USA
-
Department of Pediatrics, University of Calgary, Alberta Children’s Hospital, Calgary, Alberta, Canada
-
Division of Rheumatology, Cincinnati Children’s Hospital Medical Center, Cincinnati, OH, USA
-
Department of Pediatrics, University of Cincinnati College of Medicine, Cincinnati, OH, USA
-
Division of Pediatric Allergy, Immunology and Rheumatology, Johns Hopkins Hospital University School of Medicine, Baltimore, USA
-
Department of Pediatrics, University of Michigan, Ann Arbor, MI, USA
-
KASZ, Amsterdam, The Netherlands
-
Division of Immunology, Rheumatology Program, Boston Children’s Hospital, Boston, MA, USA
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION Nazzar, S., Moorthy, LN., et al., 2024. Effects of the COVID-19 Pandemic in Patients with Systemic Autoinflammatory Diseases: An international survey. Medical Research Archives, [online] 12(10).
https://doi.org/10.18103/mra.v12i10.5990
COPYRIGHT © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5990
ISSN 2375-1924
Abstract
Background: Early in the COVID-19 pandemic it was recognized that individuals who had worse prognoses had hyperinflammatory reactions associated with SARS CoV-2. This was worrisome for patients with systemic autoinflammatory diseases (SAID) who already have episodic hyperinflammation at baseline.
Aim: To assess the effects of the COVID-19 pandemic and SARS CoV-2 infection on patients with SAID.
Methods: An anonymous survey developed on the Survey Monkey platform was distributed online through social media and support groups of patients with SAID between January 20th and May 17th of 2021.
Results: We analyzed 484 surveys that conveyed data from 593 patients with SAID. Seventy-four percent of the surveys were answered in the United States, US territories, and Canada (n=360/484) and the rest from other countries. Sixty percent (n=330/547) of patients with SAID did not experience changes in the frequency of flares during the first year of the pandemic. Ten percent (n=52/545) of patients had presumed or confirmed COVID-19. Of those, 81% (n=42/52) had mild and moderate symptoms and 4 were hospitalized. Thirty-one percent (n=16/52) experienced flares during or after the infection and 40% (n=21/52) reported post-COVID manifestations. The individuals who had moderate symptoms or were hospitalized for COVID-19 were more likely to report post-COVID manifestations, OR 5.25 (95%CI 1.57-17.6, p=0.0072). Post-COVID integumentary manifestations occurred after asymptomatic and mild COVID-19 whereas neurologic manifestations occurred after acute COVID-19 with moderate symptoms or requiring hospitalization.
Conclusion: During the COVID-19 pandemic, over half of patients with SAID did not experience changes in the frequency of flares despite a decreased exposure to environmental triggers. Few individuals with SAID required hospitalization for COVID-19. The presence and type of post-COVID manifestations were associated with the severity of acute COVID-19 in patients with SAID.
Keywords
COVID-19, systemic autoinflammatory diseases, SAID, hyperinflammation, post-COVID manifestations
Introduction
Systemic autoinflammatory diseases (SAID) are disorders of the innate immune system characterized by systemic inflammation that is not driven by the adaptive immune system. SAID often present with flares or episodes of increased clinical manifestations. In these episodes the affected individual presents with symptoms that may include fever, rashes, oral ulcers, ocular inflammation, chest pain and/or abdominal pain depending on the diagnosis. Several triggers for the flares in autoinflammatory diseases have been identified including stressful events, infections, vaccines, menstruation, and exposure to cold. Other triggers have been associated to specific SAID.
Infections have long been recognized as triggers for flares in patients with SAID. The innate arm of the immune system is able to recognize preserved structures of infectious pathogens through membrane-bound and cytosolic receptors that include Toll-like receptors (TLRs) and nucleotide-binding oligomerization domain (NOD)-like receptors (NLRs). Activating these receptors subsequently triggers the inflammatory cascade. The stimulation of TLR-2, TLR-4 and NOD-2 of peripheral monocytes of patients with active HIDS leads to higher IL-1α, IL-1β, IL-6, TNF and activated caspase 1 when compared to monocytes from healthy controls.
Multiple viral agents have been implicated in the onset and relapses of Adult-onset Still’s disease. There is evidence of infectious agents triggering Behçet’s disease recurrences, including SARS CoV-2.
During the first year of the COVID-19 pandemic, many individuals experienced mandated lockdowns that led to limited environmental exposures, particularly to infectious agents which are known triggers for flares for specific SAID. Interestingly, there was an increase in new patient evaluations for recurring fevers in North America. Moreover, it was noted that some individuals infected with SARS-Cov-2 developed a hyperinflammatory state. This was concerning for patients with autoinflammatory conditions who have a lower threshold for inflammation.
The aims of this cross-sectional survey are to describe changes in the frequency of SAID flares during the first year of the pandemic, and to assess the outcome of SAID patients who had COVID-19.
Methods
Two patient organizations, the Autoinflammatory Alliance and KAISZ Netherlands, and the Childhood Arthritis and Rheumatology Research Alliance (CARRA) PFAPA/Autoinflammatory Diseases working group collaborated to develop an online questionnaire in the Survey Monkey platform to capture the experience of the patients with autoinflammatory diseases during the pandemic. Providers promoted the survey by informing the patients in their clinics and patient organizations did so through social media. The social media platforms used were Facebook, Twitter, RareConnect and email servers. The survey was granted exempt status from the Boston Children’s Institutional Board Review with approval number IRB-P00034465.
SURVEY
The survey was created and distributed online by patient organizations using the Survey Monkey platform between January 20th and May 17th of 2021. The questions inquired about the patients’ experiences in the first year of the COVID-19 pandemic (2020). Patients with SAID or their caregivers responded to the survey and submitted it anonymously. The survey included questions about the patient with SAID and about each of their affected children if applicable.
The questions were part of a larger survey intended for patients with SAID, but for the purposes of this study we analyzed 16 questions of which 4 were open-ended. The open-ended questions inquired about the country in which the respondent resides, reasons for changes in medications during the pandemic, access to care during the pandemic, and experience with COVID-19 illness, including post-COVID symptoms for each patient with SAID.
The closed-ended, multiple-choice questions included questions about geographic location, and for each family member with SAID we asked about their specific SAID diagnosis, changes in the frequency of their flares, changes in their fevers, changes in medications, if they had ever been diagnosed with COVID-19, and if so, the patient and/or parent perceived severity of acute SARS CoV-2 infection, and COVID-19 related flares. We inquired about sheltering practices of SAID patients and their caregivers from March through June and from July through December of 2020 due to changes in the mandated lockdowns. The specific times were determined because schools started reopening in August 2020.
The first question divided the cohort into two groups depending on whether they resided in the United States, US territories, and Canada, or in other countries. If the respondent was in the survey for residents from the United States, US territories, and Canada and answered that they were not residing in these countries, they were redirected to the survey for other countries and vice versa. The survey was only available in English for all participants.
The second question was a filter to make sure that the respondents had SAID, or a child diagnosed with SAID with the option, “No, I do not have an autoinflammatory disease, and no, I do not have a child with an autoinflammatory disease.” By committing to this response, they were not allowed to continue answering the survey.
Surveys were excluded from the analysis when truncated based on their response to the first question, if they answered that neither they nor their children had an autoinflammatory disease in the second question, or if they did not answer any of the COVID-19 questions.
The data were analyzed in Excel with means and percentages. Odds ratios were calculated using the online MedCalc tool. Missing data for specific questions were removed for the analysis.
Results
CHARACTERISTICS OF THE SAMPLE
There was a total of 751 survey entries. Of those, 42 were truncated based on country of residence, 10 answered that neither they or their children had a SAID diagnosis, 210 were removed because they did not answer the COVID-19 questions and 3 were removed because the specified diagnosis was not a SAID. During the analysis, 2 surveys were found to be duplicates and were removed, for a total of 484 surveys comprising 593 patients in the final analysis.
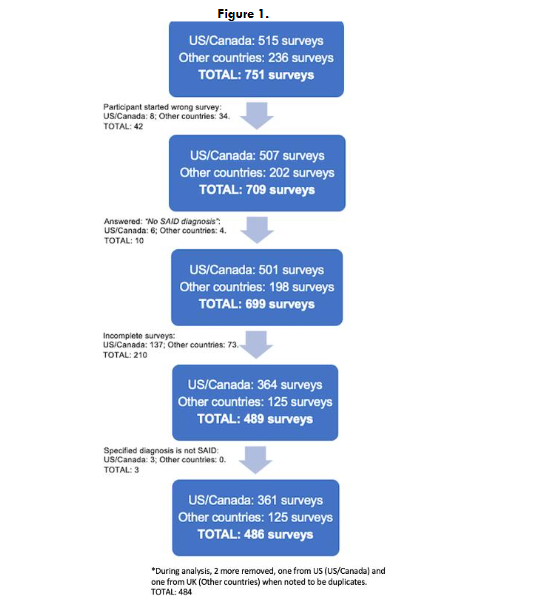
Seventy-four percent of the surveys included in the final analysis were completed by residents of the United States, US territories, and Canada (n=360/484). The remainder were from other countries with a predominance of Western European countries (n=83/484, 17%).
Seventy-six percent (n=450/593) of the patients included in the survey provided their SAID diagnosis. The top 5 SAID diagnosis were cryopyrin-associated periodic syndromes (CAPS) (n=108/450, 24%), undifferentiated systemic autoinflammatory disease (USAID) (n=70/450, 16%), periodic fever, aphthous stomatitis, pharyngitis and cervical adenitis (PFAPA) syndrome (n=64/450, 14%), systemic juvenile idiopathic arthritis (SJIA) or adult onset Still’s disease (AOSD) (n=37/450, 8%), and tumor necrosis factor receptor-associated periodic syndrome (TRAPS) (n=32/450, 7%).
| Specific Diagnosis of Patients with SAID | USA | Other countries | Total |
|---|---|---|---|
| Cryopyrin-associated periodic syndromes (CAPS) | 108 (24%) | n/a | 108 (24%) |
| Undifferentiated systemic autoinflammatory disease (USAID) | 70 (16%) | n/a | 70 (16%) |
| Periodic fever, aphthous stomatitis, pharyngitis, cervical adenitis (PFAPA) | 64 (14%) | n/a | 64 (14%) |
| Systemic juvenile idiopathic arthritis (SJIA) or adult onset Still’s disease (AOSD) | 37 (8%) | n/a | 37 (8%) |
| Tumor necrosis factor receptor-associated periodic syndrome (TRAPS) | 32 (7%) | n/a | 32 (7%) |
CHANGE IN THE FREQUENCY OF FLARES
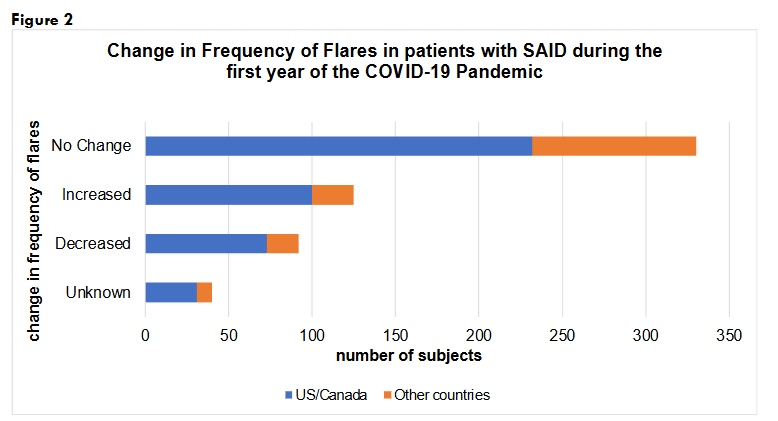
Sixty percent (n=330/547) of the participants experienced no change in the frequency of their flares during the pandemic, whereas 23% (n=125/547) reported increased frequency, 17% (n=92/547) had decreased frequency of flares of their baseline SAID and 7%, n=40/547 marked “unknown”. There is data missing from 46 individuals.
CHANGE IN FEVER PATTERN DURING FLARES
Only 55% (n=327/593) of the patients answered this question. Twenty-one percent of individuals with SAID (n=68/327) reported not typically having fevers with their flares and seventeen percent (n=57/327) reported “Feverless flares now.” Fourteen percent, (n=46/327), reported “Fevers are not as high with flares,” 9% (n=30/327) had “Lower temperatures”, and 10% (n=31/327) reported higher fevers with flares. Twenty-nine percent (n=95/327) responded “unknown”.
CHANGE IN MEDICATIONS DURING THE PANDEMIC
Forty-seven percent (n=273/584) of patients had no change in their medications and 11% (n=63/584) were not on any treatment during the pandemic. Among those who reported that there was a change in their medications (n=248/584, 43%), the most frequent reason was to improve control or reduce flares (n=67/248, 27%), the second most common was new diagnosis (n=18/248, 7%), and the third was adverse effects (n=12/248, 5%). Difficulty obtaining medication, switching to a different medication, weight adjustment, infection other than COVID-19, increased cases of COVID-19 in the area, allergic reaction, absence of symptoms, and switching providers were less frequently reported reasons for change in medications. Only 2 individuals reported that the change in medication was due to COVID-19 illness. There was no response from 9 individuals.
ACCESS TO CARE
Forty-four percent (n=152/349) reported no barriers. Telehealth (n=50/349, 14%) and phone appointments (n=10/349, 3%) were reported as available options for health care delivery.
The most frequent barrier reported was lack of available appointments or cancellations (n=72/349, 21%), followed by feeling unsafe due to COVID-19 (n=19/349, 5%), inability to communicate with providers (n=12/349, 3%), obtaining medications (n=7/349, 2%), multiple COVID-19 tests (n=6/349, 1.7%), being turned away due to fever during SAID flare following COVID protocols (n=4/349, 1.2%), one-parent rule (n=3/349, 0.9%), and canceled flights (n=2/349, 0.6%). There is data missing from 28% of respondents (n=135/484).
COVID-19 IN PATIENTS WITH SAID
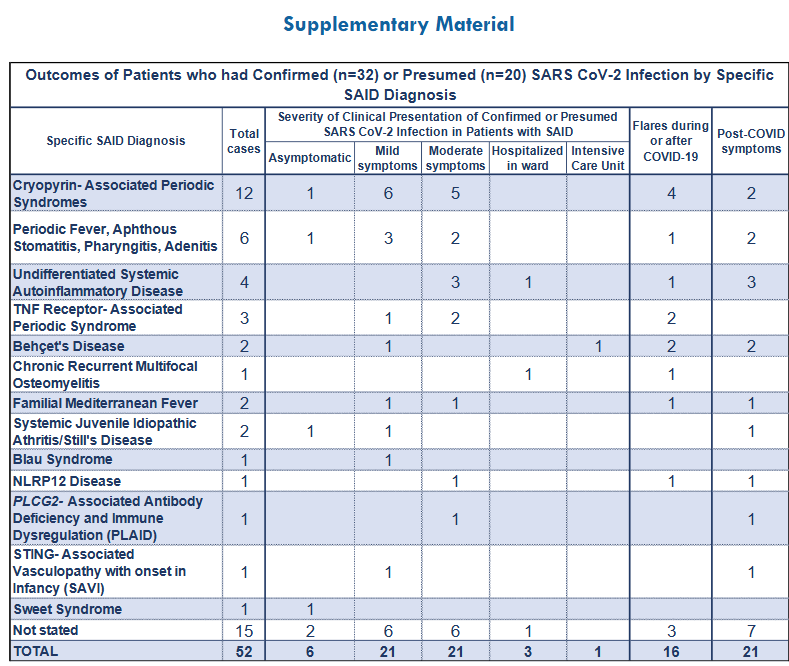
Ten percent (n=52/545) of the patients with SAID who were accounted for in the survey reported physician-diagnosed (n=20/52, 38%) or confirmed SARS-CoV-2 infection (n=32/52, 62%). Of those, 11.5% (n=6/52) were asymptomatic, 40% (n=21/52) had mild symptoms, 40% (n=21/52) had moderate symptoms, 6% (n=3/52) were hospitalized in the wards, and 2% (n=1/52) required admission to the intensive care unit (ICU). Thirty-one percent (n=16/52) experienced flares during (n=9/52, 17%) or after (n=7/52, 14%) COVID-19 infection.
CLINICAL PRESENTATION OF SARS-COV-2 INFECTION AND CHANGE IN FREQUENCY OF SAID FLARES
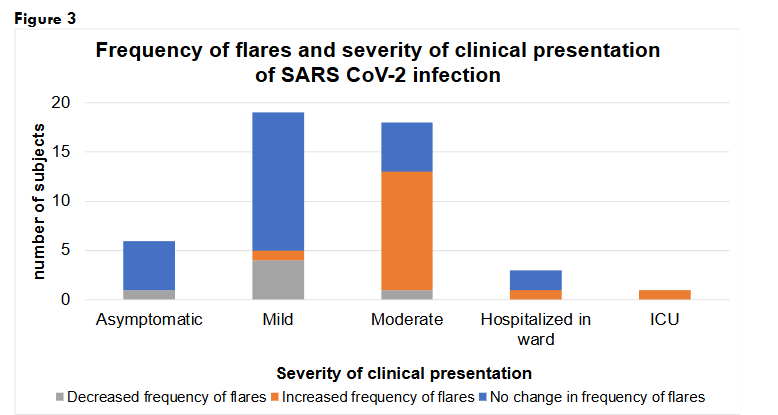
Among the individuals who had asymptomatic SARS CoV-2 infection, 83% (n=5/6) reported no change in the frequency of flares and 17% (n=1/6) had decreased flares. For the mild COVID-19 presentations, 67% (n=14/21) had no change, 19% (n=4/21) had decreased, and 5% (n=1/21) had increased frequency of flares. For the individuals who had moderate symptoms with COVID-19, 57% (n=12/21) had increased frequency of flares, 24% (n=5/21) had no change, and 5% (n=1/21) had decreased flares. In the patients who were hospitalized, 2 out of 3 had no change in flares and 1 had increased flares. Finally, the patient who was hospitalized in the ICU reported increased frequency of flares. Individuals with increased frequency of flares were more likely to have moderate COVID-19 or to require hospitalization, OR 42 (95%CI 4.74-372, p=0.0008).
POST-COVID SYMPTOMS
A total of 21 patients had prolonged or post-COVID symptoms that were predominantly from the immunologic (n=7/21, 33%), neurologic (n=6/21, 29%) and respiratory systems (n=6/21, 29%). Constitutional post-COVID manifestations were reported by 14% (n=3/21). Two patients (n=2/52, 3.8%) had multisystem inflammatory syndrome in children (MIS-C).
We looked at the severity of SARS-CoV-2 infection related to post-COVID symptoms. Seventeen percent (n=1/6) of asymptomatic patients, 24% (n=5/21) of patients who had mild COVID-19 symptoms, 57% (n= 12/21) of patients who had moderate symptoms, 2 out of the 3 patients who were hospitalized in the ward, and the patient who was hospitalized in the ICU reported post-COVID manifestations. Those who had moderate symptoms or were hospitalized had higher odds of having post-COVID symptoms compared to those who had mild and asymptomatic presentations, OR 5.25 (95%CI 1.57-17.6, p=0.0072).
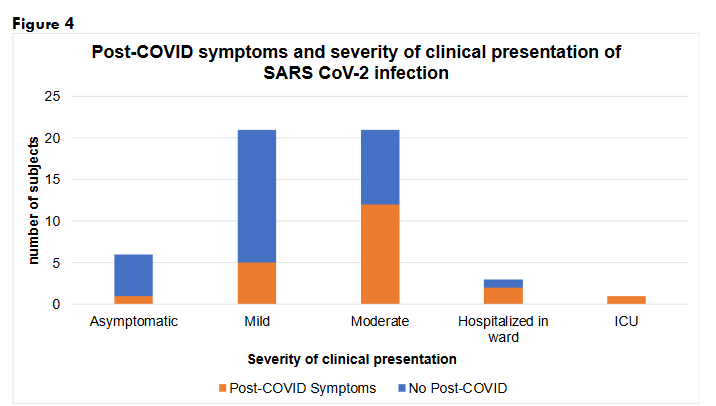
The patient who had asymptomatic SARS CoV-2 infection had COVID toes as a post-COVID manifestation. The patients who had mild COVID-19 had respiratory (n=2/5), immunologic (n=2/5), integumentary (n=1/5), and musculoskeletal (n=1/5) post-COVID symptoms. Those who had moderate COVID-19 reported post-COVID symptoms from multiple organ-systems, mostly immunologic (n=4/12), neurologic (n=3/12), respiratory (n=3/12) and constitutional (n=3/12) symptoms. The patients who were hospitalized in the ward had neurologic (n=2/2) and immunologic (n=1/2) symptoms and the patient in the ICU had neurologic, respiratory, and cardiovascular symptoms.
Discussion
The primary aim of this project was to describe the experiences of patients with SAID with COVID-19. Interestingly, despite the reduced exposure to possible environmental triggers with the lockdowns, the frequency of SAID flares was unchanged in over half of the patients. The mandated sheltering and isolation tested the influence of external factors as triggers of flares in patients with SAIDs. This strengthens the notion that there may be intrinsic factors from the disease process and from the individual such as emotional distress triggering flares.
Patients with SAID reported having feverless flares. The reported decrease in the temperature of the fevers could represent a phenomenon that occurs over time in the natural history of some SAID or because of medication adjustments. It could also imply the intensity of the fever during the episodes is a factor that depends on external elements to which the patients had less exposure during the pandemic.
From our survey, 44% (n=152/349) of respondents answered that the pandemic did not affect their care. Similarly, Wu et al. conducted a survey in China between April and May 2020 and 37.9% patients answered that the pandemic did not pose a challenge to their disease treatment and follow-up visits. They also identified obtaining medications as a challenge during the pandemic.
In the initial stages of the pandemic the prevalence of COVID-19 illness was low in our group and the majority had mild or moderate symptoms. This finding is in concordance with what is reported in the literature from that same period. Most patients with autoinflammatory conditions had asymptomatic SARS-CoV-2 infection or were able to manage at home. The patients that required hospitalization had other known risk factors for severe disease, e.g, older age, hypertension, and obesity. The patients who required hospitalization and had no other known risk factors for severe disease had diagnosis of A20 haploinsufficiency (Behçet’s-like syndrome) and autoimmune lymphoproliferative syndrome (ALPS). Only a large French cohort reported increased frequency of severe disease in patients with autoinflammatory conditions, but there was no mention of risk factors related to poor prognosis.
There were no fatalities related to COVID-19 in our cohort but a recent systematic review by Alhumaid et al. about patients with inborn errors of immunity reported 2 fatalities in children with autoinflammatory conditions (2/67) who had COVID-19. MIS-C was higher in that review occurring in 26.9% (18/67) of cases. In our cohort only 2 (n=2/52, 3.8%) MIS-C cases were reported.
Of note, the literature suggests that severe COVID-19 was associated with interferon dysfunction, in some cases due to neutralizing interferon antibodies and in others due to genetic defects affecting the interferon pathway. It is plausible that an increased inflammatory response in some patients with SAID resulted in enhanced viral clearance and ended up being protective factor against severe COVID-19 illness.
In our data, we found that the severity of the clinical presentation of COVID-19 illness appeared to be associated with changes in the frequency of flares of their baseline SAID. Unfortunately, the limitations of the survey data would not permit us to determine if the change in frequency of flares occurred before or after SARS-CoV-2 infection.
Regarding post-COVID manifestations, the most common symptoms reported by the surveyed were immunologic, neurologic, and respiratory. In another large survey-based international study about long-COVID, almost all patients experienced systemic (99.7%, 95%CI 99.49%-99.84%) and ear, nose, and throat (100%) symptoms. Notably, gastrointestinal symptoms are commonly reported in the general population, but was only reported by one patient with SAID in our survey.
In our cohort, the development of post-COVID symptoms appeared to be associated with the severity of acute COVID-19 in patients with SAID. Skin manifestations were seen after mild or asymptomatic COVID-19 while the patients who had moderate symptoms or were hospitalized had neurological post-COVID symptoms. In the study by Taquet et al., patients in the general population with more severe COVID-19 illness had significantly more features post-COVID, the most frequent being cognitive difficulties and the least common being myalgias and headaches.
Limitations include selection bias due to the need for internet access, level of comfort with the use of technology, variable availability of healthcare resources for the appropriate diagnosis of SAIDs, language barrier, and missing data. Most of the surveys submitted were from North America and Europe. Age and gender of the patients with SAID were not asked in the questionnaire to decrease survey fatigue as well as the possibility of the affected families or individuals to be identified. The question about sheltering practices was removed from the manuscript by consensus among the authors. The question included caregivers without SAID making it susceptible to misinterpretation. Another limitation was that we intended to capture patients who experience low temperatures with their flares given that it has been informally reported by patients. However, the option “lower temperature” did not specify a cutoff temperature value. At the time of the survey development and distribution there was no standard definition of post-COVID symptoms. The period that was addressed in the survey was 2020 in the early stages of the pandemic when there was limited testing availability.
Future directions include to determine the effects of specific SARS-COV-2 variants, repeat infection and COVID-19 immunization on the severity of acute COVID-19 and post-COVID manifestations in patients with SAID.
Conclusions
- The COVID-19 pandemic provided a natural experiment on the role of isolation on patients with systemic autoinflammatory diseases. The period of isolation during the pandemic did not affect the frequency of flares in 60% of patients with SAID.
- Asymptomatic and mild acute COVID-19 presentations were more likely to be associated with integumentary post-COVID manifestations and were not associated with change in the frequency of flares. On the other hand, moderate acute COVID-19 and those who required hospitalization were more likely to report increased frequency of flares and neurologic post-COVID manifestations.
- From our data, 8% of patients with SAID who were infected with the SARS CoV-2 virus were hospitalized and there were no fatalities reported.
Conflicts of Interest Statement
Lakshmi N Moorthy with NJ Pediatrics (NJAAP) and Bristol-Myers Squibb. Sivia Lapidus was awarded grants from the Shotmyer Family and the Hearst foundations. Grant Schulert with Ipinovyx, Novartis, SOBI and Boehringer Ingelheim. Karen Durrant, Jennifer Tousseau, Saskya Angevare and Fatma Dedeoglu are board members of the Autoinflammatory Alliance. Saskya Angevare is board member of KAISZ and ENCA. Karen Durrant is the patient organization representative for ISSAID. Fatma Dedeoglu serves as Education Committee Chair of ISSAID, was an invited guest speaker for ISSAID 2023, an invited speaker at ACR Convergence 2023 and receives royalties up-to-date.
Funding Statement
There was no financial support, grants or benefits from commercial sources utilized for the elaboration of this manuscript.
Acknowledgments
We acknowledge Mariana Correia-Marques for her contributions in the preparation of the manuscript, and Giomilsy Ramos for assistance with literature search. The authors wish to acknowledge CARRA and the ongoing Arthritis Foundation financial support of CARRA.
References
- Ben-Chetrit E, Gattorno M, Gul A, et al. Consensus proposal for taxonomy and definition of the autoinflammatory diseases (AIDs): a Delphi study. Ann Rheum Dis. 2018;77(11):1558-1565. doi:10.1136/annrheumdis-2017-212515
- van der Hilst, Jeroen C H et al. “Long-term follow-up, clinical features, and quality of life in a series of 103 patients with hyperimmunoglobulinemia D syndrome.” Medicine vol. 87,6 (2008): 301-310. doi:10.1097/MD.0b013e318190cfb7
- Kishida, Dai et al. “Triggering factors for febrile attacks in Japanese patients with familial Mediterranean fever.” Clinical and experimental rheumatology vol. 38 Suppl 127,5 (2020): 76-79.
- Shenavandeh S, Asis M, Eftekhari MH, et al. The Patients’ Beliefs Regarding the Role of Food, Mucosal Trauma, Menstruation, and Psychological Stress in the Recurrence of Behçet’s Disease Symptoms. J Med Life. 2020;13(2):164-169. doi:10.25122/jml-2019-0153
- Karadag O, Tufan A, Yazisiz V, et al. The factors considered as trigger for the attacks in patients with familial Mediterranean fever. Rheumatol Int. 2013;33(4):893-897. doi:10.1007/s00296-012-2453-x
- Schwabe, Peters. Familial Mediterranean Fever in Armenians. Analysis of 100 cases. Medicine 1974;53(6):453
- G. Yenokyan, H.K. Armenian. Triggers for attacks in Familial Mediterranean Fever: application of the case-crossover design. Am J Epidemiol, 175 (2012), pp. 1054-1061
- Avagyan, Amaryan et al. Influence of Some Environmental Factors on Manifestation of Familial Mediterranean Fever in Children: Clinical and Genetic Aspects. Caucasus Journal of Health Sciences and Public Health, June 2018;2(2)
- Korkmaz, Cengiz et al. “Familial Mediterranean fever: the molecular pathways from stress exposure to attacks.” Rheumatology (Oxford, England) vol. 59,12 (2020): 3611-3621. doi:10.1093/rheumatology/keaa450
- Volle G, Fraison JB, Gobert D, et al. Dietary and Nondietary Triggers of Oral Ulcer Recurrences in Behçet’s Disease. Arthritis Care Res (Hoboken). 2017;69(9):1429-1436. doi:10.1002/acr.23155
- Lachmann HJ, Papa R, Gerhold K, et al. The phenotype of TNF receptor-associated autoinflammatory syndrome (TRAPS) at presentation: a series of 158 cases from the Eurofever/EUROTRAPS international registry. Ann Rheum Dis. 2014;73(12):2160-2167. doi:10.1136/annrheumdis-2013-204184
- Maggio, Maria Cristina, and Giovanni Corsello. “FMF is not always “fever”: from clinical presentation to “treat to target”.” Italian journal of pediatrics vol. 46,1 7. 15 Jan. 2020, doi:10.1186/s13052-019-0766-z
- Christiansen, Mette et al. “XIAP deficiency and MEFV variants resulting in an autoinflammatory lymphoproliferative syndrome.” BMJ case reports vol. 2016 bcr2016216922. 28 Sep. 2016, doi:10.1136/bcr-2016-216922.
- Stoffels, Monique et al. “TLR2/TLR4-dependent exaggerated cytokine production in hyperimmunoglobulinaemia D and periodic fever syndrome.” Rheumatology (Oxford, England) vol. 54,2 (2015): 363-8. doi:10.1093/rheumatology/keu341
- Jia J, Shi H, Liu M, et al. Cytomegalovirus Infection May Trigger Adult-Onset Still’s Disease Onset or Relapses. Front Immunol. 2019;10:898. Published 2019 Apr 24. doi:10.3389/fimmu.2019.00898
- Yu X, Zheng H. Refractory Genital HPV Infection and Adult-Onset Still Disease: A Case Report and Literature Review. Medicine (Baltimore). 2016;95(15):e3169. doi:10.1097/MD.0000000000003169
- Torres-Aguilar H, Sosa-Luis SA, Aguilar-Ruiz SR. Infections as triggers of flares in systemic autoimmune diseases: novel innate immunity mechanisms. Curr Opin Rheumatol. 2019;31(5):525-531. doi:10.1097/BOR.0000000000000630
- Ozcifci G, Aydin T, Atli Z, et al. The incidence, clinical characteristics, and outcome of COVID-19 in a prospectively followed cohort of patients with Behçet’s syndrome. Rheumatol Int. 2022;42(1):101-113. doi:10.1007/s00296-021-05056-2
- McPeake JR, Hirst WJ, Brind AM, Williams R. Hepatitis A causing a second episode of virus-associated haemophagocytic lymphohistiocytosis in a patient with Still’s disease. J Med Virol. 1993;39(2):173-175. doi:10.1002/jmv.1890390216
- Walker UA, Luetke Volksbeck S, Kuemmerle-Deschner J. Flare of a Cryopyrin-associated Periodic Syndrome Following Vaccination with Neisseria Meningitidis Polysaccharides. J Rheumatol. 2018;45(6):878-879. doi:10.3899/jrheum.171037
- Jeyaratnam J, Ter Haar NM, Lachmann HJ, et al. The safety of live-attenuated vaccines in patients using IL-1 or IL-6 blockade: an international survey. Pediatr Rheumatol Online J. 2018;16(1):19. Published 2018 Mar 21. doi:10.1186/s12969-018-0235-z
- Park SY, Lee KH. Adult-onset Still’s Disease after BNT162b2 mRNA COVID-19 Vaccine. J Korean Med Sci. 2021;36(50):e344. Published 2021 Dec 27. doi:10.3346/jkms.2021.36.e344
- Flare-up of adult-onset Still’s disease after receiving a second dose of BNT162b2 COVID-19 mRNA vaccine
- Peet CJ, Papadopoulou C, Sombrito BRM, Wood MR, Lachmann HJ. COVID-19 and autoinflammatory diseases: prevalence and outcomes of infection and early experience of vaccination in patients on biologics. Rheumatol Adv Pract. 2021;5(2):rkab043. Published 2021 Aug 23.
- Grasland A, Le Maître F, Pouchot J, Hazera P, Bazin C, Vinceneux P. Maladie de Still de l’adulte après vaccination contre l’hépatite A et B? [Adult-onset Still’s disease after hepatitis A and B vaccination?]. Rev Med Interne. 1998;19(2):134-136. doi:10.1016/s0248-8663(97)83425-x
- Padgett, Christina. “Recurrence of Symptoms Associated with Menstruation in a Patient with a History of Periodic Fevers.” Journal of pediatric and adolescent gynecology vol. 33,4 (2020): 429-431. doi:10.1016/j.jpag.2020.03.008
- Guzelant, Gul et al. “Exacerbation of Behçet’s syndrome and familial Mediterranean fever with menstruation.” Clinical and experimental rheumatology vol. 35 Suppl 108,6 (2017): 95-99.
- Akar S, Soyturk M, Onen F, Tunca M. The relations between attacks and menstrual periods and pregnancies of familial Mediterranean fever patients. Rheumatol Int. 2006;26(7):676-679. doi:10.1007/s00296-005-0041-z
- Kumei, Shima et al. “Cold Exposure Related Fever with an Mediterranean Fever (MEFV) Gene Mutation.” Internal medicine (Tokyo, Japan) vol. 56,16 (2017): 2233-2236. doi:10.2169/internalmedicine.8274-16
- Sharma M, Ferguson PJ. Autoinflammatory bone disorders: update on immunologic abnormalities and clues about possible triggers. Curr Opin Rheumatol. 2013;25(5):658-664. doi:10.1097/BOR.0b013e328363eb08
- Nakashimai, Hideyuki et al. “Febrile attacks triggered by milk allergy in an infant with mevalonate kinase deficiency.” Rheumatology international vol. 36,10 (2016): 1477-8. doi:10.1007/s00296-016-3522-3
- Mansfield L, Lapidus S, Nazzar S, Moorthy LN, Adler-Shohet F, Hollander M et al. Increase in pediatric recurrent fever evaluations during the first year of the COVID-19 pandemic in North America. Front. Pediatr. 2023; 11
- Manson JJ, Crooks C, Naja M, Ledlie A, Goulden B, Liddle T et al. COVID-19-associated hyperinflammation and escalation of patient care: a retrospective longitudinal cohort study. Lancet Rheumatol. 2020; 2: 594-602.
- MedCalc Software Ltd. Odds ratio calculator. https://www.medcalc.org/calc/odds_ratio.php (Version 22.009; accessed August 8, 2023)
- Levinsky Y, Butbul Aviel Y, Ahmad SA, Broide M, Gendler Y, Dagan N, et al. PFAPA flares observed during COVID outbreak: can emotional stress trigger PFAPA attacks? A multicenter cohort study. Pediatr Rheumatol Online J. 2022;20(1):46.
- Wu N, Zhao M, Wu D, Yu K, Shen M. COVID-19 pandemic and systemic autoinflammatory diseases management: a cross-sectional survey. Rheumatol Int. 2021; 41: 1541-1543
- Haslak F, Yildiz M, Adrovic A, Sahin S, Koker O, Aliyeva A, et al. Management of childhood-onset autoinflammatory diseases during the COVID-19 pandemic. Rheumatol Int. 2020; 40: 1423-1431.
- Pablos JL, Galindo M, Carmona L, Lledó A, Retuerto M, Blanco R, et al. Clinical outcomes of hospitalised patients with COVID-19 and chronic inflammatory and autoimmune rheumatic diseases: a multicentric matched cohort study. Ann Rheum Dis. 2020; 79: 1544-1549.
- Bourguiba R, Delplanque M, Vinit C, Ackermann F, Savey L, Grateau G, et al. Clinical course of COVID-19 in a cohort of 342 familial mediterranean fever patients with a long-term treatment by colchicine in a french endemic area. Ann Rheum Dis. 2021; 80: 539-540.
- Welzel T, Samba SD, Klein R, van den Anker JN, Kuemmerle-Deschner JB. COVID-19 in autoinflammatory diseases with immunosuppressive treatment. J Clin Med. 2021; 10: 605
- FAI2R /SFR/SNFMI/SOFREMIP/CRI/IMIDIATE consortium and contributors. Severity of COVID-19 and survival in patients with rheumatic and inflammatory diseases: data from the french RMD COVID-19 cohort of 694 patients. Ann Rheum Dis. 2020; 80: 527-538.
- Meyts I, Bucciol G, Quinti I, et al. Coronavirus disease 2019 in patients with inborn errors of immunity: An international study. J Allergy Clin Immunol. 2021;147(2):520-531. doi:10.1016/j.jaci.2020.09.010
- Shields AM, Burns SO, Savic S, Richter AG; UK PIN COVID-19 Consortium. COVID-19 in patients with primary and secondary immunodeficiency: The United Kingdom experience. J Allergy Clin Immunol. 2021;147(3):870-875.e1. doi:10.1016/j.jaci.2020.12.620
- Alhumaid S, Al Mutared KM, Al Alawi Z, et al. Severity of SARS-CoV-2 infection in children with inborn errors of immunity (primary immunodeficiencies): a systematic review. Allergy Asthma Clin Immunol. 2023; 19: 69
- Davis HE, Assaf GS, McCorkell L, et al. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. EClinicalMedicine. 2021; 38: 101019.
- Taquet M, Dercon Q, Luciano S, Geddes JR, Husain M, Harrison PJ. Incidence, co-occurrence, and evolution of long-COVID features: a 6-month retrospective cohort study of 273,618 survivors of COVID-19. PLoS Med. 2021; 18: 1003773.
Supplementary Material