Improving Chemotherapy Tolerance with Olanzapine and Megestrol
A Retrospective Study on the Use of Short-Term Combination Pharmacotherapy in Improving Weight, Appetite and Tolerance to Cytotoxic Chemotherapy in Patients with Locally Advanced Solid Cancers
Beulah Elizabeth Koshy 1, Linu Abraham Jacob 1, MC Suresh Babu 1, Lokesh KN 1, AH Rudresha 1, LK Rajeev 1, Smitha C Saldanha 1, Kavya Halmandge1
- Department of Medical oncology, Kidwai Memorial Institute of Oncology, Dr. M. H. Marigowda road, Bengaluru Karnataka India
OPEN ACCESS
PUBLISHED: 30 April 2025
CITATION: Koshy, BE., Jacob, LA., et al., 2025. A Retrospective Study on the Use of Short-Term Combination Pharmacotherapy in Improving Weight, Appetite and Tolerance to Cytotoxic Chemotherapy in Patients with Locally Advanced Solid Cancers. Medical Research Archives, [online] 13(4). https://doi.org/10.18103/mra.v13i4.6545
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i4.6545
ISSN 2375-1924
ABSTRACT
Cytotoxic chemotherapy-related toxicities pose significant challenges in the management of solid cancers, particularly in resource-limited settings. This may be because of the increased incidence of cancer precachexia and cachexia that these patients present with. This retrospective observational study assessed the efficacy of administering a combination of T.Olanzapine (5 mg/day) and Syp.Megestrol acetate (800 mg/day) in improving weight gain, appetite, and tolerance to chemotherapy among undernourished cancer patients (BMI <18 kg/m²) presenting with locally advanced solid cancers. A total of 92 patients who received neoadjuvant chemotherapy or chemoradiotherapy from March 2024- June 2024 were divided into 2 cohorts. The patients in cohort A received the combination for up to six weeks, along with the decided neoadjuvant therapy, while the patients in cohort B did not. Cohort A demonstrated significant improvements in weight gain (≥5% increase observed in 90% of patients, OR = 0.0030, 95% CI: 0.0002–0.0585, p=0.0001) and appetite (VAS score +3 in 20 patients, OR = 0.1294, 95% CI: 0.0265–0.6317, p=0.0115; and SGA improvement in 12 patients, OR = 0.0626, p=0.0596) along with a reduction in chemotherapy-induced nausea and vomiting as compared to the patients in Cohort B. Grade ≥3 hematological toxicity occurred in 5 patients belonging to Cohort B vs. none in cohort A (0%, p=0.046, OR = 0.0310, 95% CI: 0.0016–0.5961, p=0.0213). Patients receiving the combination also showed better treatment adherence and completion rates. The study was concluded on November 30th,2024 after the last patient received their last cycle of chemotherapy and was followed up for 3 weeks to check for adverse events. It thus seems to be an effective strategy in improving patient outcomes, especially in socioeconomically disadvantaged populations with higher prevalence of precachexia and cachexia.
Keywords
Chemotherapy, Cachexia, Megestrol Acetate, Olanzapine, Weight Gain, Appetite, Cancer
Introduction
Locally advanced solid cancers present significant challenges in clinical management due to their aggressive nature and the requirement for multimodal treatments, such as chemotherapy and chemoradiotherapy. While these intensive therapies are essential for disease control and improving survival outcomes, they are frequently associated with severe toxicities, including hematological effects like neutropenia and thrombocytopenia, as well as non-hematological side effects such as nausea and vomiting. These adverse events can negatively impact patients’ nutritional status, treatment adherence, and overall quality of life, ultimately compromising the effectiveness of therapy 1,2. Therefore, developing strategies to mitigate these toxicities is critical for optimizing outcomes in this patient population.
Cachexia and malnutrition are particularly prevalent among cancer patients, with up to 80% experiencing weight loss during treatment. This not only impairs physical function but also increases morbidity and mortality. Malnutrition exacerbates treatment-related toxicities and worsens outcomes, but early nutritional intervention and prophylactic use of G-CSFs can enhance therapy tolerance, reduce complications like febrile neutropenia, maintain chemotherapy dose intensity, and improve overall treatment adherence in patients with locally advanced cancers 3,4. Various studies have shown that weight gain during chemotherapy or chemoradiotherapy is associated with improved survival outcomes 5,6,7,8,9,10. Addressing nutritional deficits and appetite stimulation are crucial components of supportive care.
Megestrol acetate, a synthetic progestin, has been widely recognized for its appetite-stimulating and anti-cachectic properties. At a dose of 800 mg/day, it has shown efficacy in promoting weight gain, increasing caloric intake, and enhancing overall well-being in cancer patients 11. However, the use of megestrol should be carefully monitored due to potential side effects such as thromboembolic events, fluid retention, and adrenal suppression, especially in patients with comorbidities or advanced disease 12.
Concurrently, effective management of chemotherapy-induced nausea and vomiting (CINV) is essential for maintaining treatment adherence. Despite advancements in antiemetic protocols, breakthrough nausea and vomiting remain common, significantly impacting patients’ quality of life and nutritional status 13,14. Olanzapine, a second-generation antipsychotic with potent antiemetic properties, has emerged as an effective agent in managing CINV. At a dose of 5 mg/day, olanzapine has been shown to reduce nausea severity, improve food intake, and enhance patient comfort during treatment 15,16.
The combination of megestrol acetate and olanzapine offers a novel pharmacological strategy to address the multifaceted toxicities associated with chemotherapy and chemoradiotherapy. Megestrol acetate primarily focuses on appetite stimulation and weight gain, while olanzapine mitigates nausea and vomiting. Together, they provide a comprehensive approach to supportive care, enabling patients to tolerate and complete their prescribed treatment regimens 17,18,19,20,21,22,23. Although megestrol acetate and olanzapine have demonstrated individual and combined benefits, evidence supporting their combined use in improving tolerance to chemotherapy in patients with locally advanced cancers is limited. There is also a lack of studies assessing the impact of this combination on key outcomes, such as treatment completion rates, incidence of treatment-related toxicities, and patient-reported quality of life.
Two RCTs by Navari et al in 200917 and Sandhya et al in 202318 have looked at the effect of Olanzapine and Megestrol acetate in the management of cancer related anorexia and cachexia. In the RCT by Navari et al, a total of 80 patients who met the definition of Cancer-related anorexia were randomized into two arms – daily megestrol acetate (800 mg /day) or megestrol acetate (800 mg /day) plus olanzapine (5 mg/day) for a period of 8 weeks. The improvement in weight (greater than or equal to 5% weight gain), appetite (+3 in the visual analog scale), nausea (-3 in the visual analog scale), and QOL at 4 and 8 weeks were looked at. The combination of megestrol acetate and olanzapine was found to be statistically superior to the interventional arm of megestrol acetate alone with regards to each of the above-mentioned parameters. In the patients receiving the combination of olanzapine and megestrol acetate, the improvement in weight, appetite, nausea, and QOL was present at 4 weeks and persisted at 8 weeks.
In the RCT by Sandhya et al, 124 patients were randomized into two arms- 63 patients received T. Olanzapine 2.5 mg once a day for 12 weeks and 61 patients received placebo. The proportion of patients with >=5% weight gain after 12 weeks was 60% (35 of 58 patients) versus 9% (5 of 54 patients), favoring olanzapine. The proportion of patients with an improvement in appetite assessed using the VAS from baseline to week 12 was significantly higher in the olanzapine group (43% v 13%; p=.001). The proportion of patients with >=grade 3 toxicities was lower with olanzapine (7 of 58 [12%] v 20 of 54 [37%], P = .002). Dose modification to 100% dose in the second cycle were modified in 16 (28%) patients in the olanzapine group, of whom 12 (75%) had an increase in the dose of chemotherapy to 100%. In the placebo group, 21(39%) had dose modification at cycle 2, of whom only three (14%) patients had an increase in chemotherapy dosage. Therefore, chemotherapy tolerance was significantly improved in the olanzapine group.
This retrospective observational study included precachectic and cachectic patients with a Body Mass Index (BMI) of less than 18 who presented to the Department of Medical Oncology, with a diagnosis of locally advanced solid cancer. These patients were offered olanzapine and megestrol acetate as part of their supportive care regimen. The primary objective of the study was to observe the proportion of patients who achieved a weight gain of ≥5% of their original weight. Additionally, improvement in appetite measured by Visual Analog Scale (VAS) and Subjective Global Assessment (SGA) scales and tolerance to chemotherapy which was defined as reduction in both haematological and non-haematological adverse events were looked for.
This approach is particularly relevant in rural Indian cancer patients, who exhibit higher levels of intolerance to standard chemotherapy regimens; likely due to higher prevalence of anorexia and cancer cachexia that worsens with intensive treatments. These patients experience an increased incidence of both hematological and non-hematological toxicities, often requiring upfront or mid-treatment dose modifications, prophylactic growth factor administration, and, in severe cases, hospital admissions or prolonged Intensive Care Unit (ICU) stays. Despite all the prophylactic interventions, 10–20% of patients still experience significant toxicities.
By addressing the dual challenges of malnutrition and CINV with a fixed-dose pharmacotherapy regimen comprising megestrol acetate and olanzapine, this study seeks to enhance chemotherapy tolerance, increase total body weight and appetite, and reduce treatment-related toxicities within a fixed period of six weeks. This simple, scalable strategy could improve patient outcomes, set new standards for supportive care, and enable patients to complete their prescribed treatments with fewer complications.
Methodology
PATIENT SELECTION
Patients with age greater than or equal to 18 years with BMI less than 18 kg/m2 and histopathologically confirmed locally advanced or metastatic head and neck, gastroesophageal, hepatopancreatic biliary, colorectal, breast and lung cancers were included. Patients found to be unfit for standard of care chemotherapy or requiring dose adjustment/delay (>1 week) or discontinuation due to CTCAE Grade ≥3 adverse events were also included. The exclusion criteria consisted of the following: patients aged >70 years, with hematological malignancies, chronic diseases causing end-organ damage (like Chronic Kidney Disease (CKD), Chronic Liver Disease (CLD), Ischemic Heart Disease (IHD), patients reliant on parenteral nutrition, patients with lipid storage disorders, hypercoagulability, or thromboembolism history and whose medical records are incomplete.
The combination of T.Olanzapine (5 mg/day) and Syp.Megestrol acetate (800 mg/day) was offered to all the patients who belonged to the under-weight category (defined pre-emptively as BMI<18 mg/m2). However, only roughly half of the patients were able to afford this combination as it was not available under the government subsidy scheme.
STUDY DESIGN AND TREATMENT REGIMEN
All patients seen in the Outpatient department during the study period (1st March 2024- 30th June 2024) who fit the inclusion criteria were considered for the study. A total of 92 patients who were planned for neoadjuvant chemotherapy or chemoradiotherapy were divided into 2 cohorts. The patients in cohort A received the combination of T. Olanzapine 5mg/day and Megestrol acetate 800 mg/day for up to six weeks, along with the decided neoadjuvant therapy, while the patients in cohort B did not. The patients in cohort A received the drug combination till their weight increased to 5% or more than that at presentation or till a maximum period of 6 weeks.
STUDY VISITS AND ASSESSMENT PROCEDURES
This retrospective study reviewed the medical records of patients treated at our centre during the study period. Patients in both groups underwent routine assessments and follow-up evaluations as part of their clinical care.
DATA COLLECTION AND ASSESSMENT PROCEDURES
The study included a review of demographic data (age, gender, height, and weight) and medical information, including detailed history (diet and appetite), physical examinations (BMI, BSA, mid-arm circumference), nutritional status assessments (Subjective Global Assessment), and laboratory data by trained medical personnel. Appetite and nausea were assessed using Visual Analog Scales (VAS), where a score of zero indicated no symptom and a score of ten indicated the maximum severity. A change in VAS scores by three or more points over time was considered clinically significant.
Patients’ weight, appetite level, nausea severity, quality of life (QOL), and symptom intensity were assessed weekly during their treatment period as part of standard follow-up. Adverse events were documented based on general physical examinations and patient-reported symptoms during weekly follow-ups.
At the end of the 6-week study period or upon achieving ≥5% weight gain, patients were re-evaluated with repeat physical examinations, VAS assessments, and laboratory tests. The records were reviewed to determine whether patients resumed or initiated standard-of-care chemotherapy after intervention. The incidence of hematological and non-hematological toxicities (especially CTCAE Grade 3 or higher) post-intervention was recorded to assess the impact of the combination therapy on treatment tolerance.
STATISTICAL METHODS
The baseline characteristics were utilized by descriptive analysis. Continuous variables were reported as mean ± standard deviation (SD) or median with interquartile range (IQR) depending on their distribution, assessed using the Mann-Whitney test. Categorical variables were summarized as frequencies and percentages, and the incidence of severe toxicities and adverse events were calculated. Differences between groups were evaluated using the independent t-test or Mann-Whitney U test for continuous variables and the chi-square test or Fisher’s exact test for categorical variables. The study was powered for a 90% chance of detecting a 10% difference in weight and appetite in each arm. This was based on the predicted response from the data available in the literature. The percentage of patients with improvement in weight, appetite, nausea, and QOL for the 6 weeks periods was calculated separately for each treatment regimen. The mean, median, and standard deviation of the maximum symptom scores for 6 weeks for each treatment regimen were calculated.
Results:
Among the 49 patients in Cohort A, 42 patients completed the chemotherapy/chemo-radiotherapy, with 7 dropouts (14.2%). In contrast, the cohort B group that had 43 patients initially had 12 dropouts (27.9%).
| Physical and Demographic Characteristics | Cohort A (n=49) | Cohort B (n=43) |
|---|---|---|
| Median Age | 51 (Range:43) | 54(Range:46) |
| M:F ratio | 1.45:1 | 1.23:1 |
| ECOG PS | 1 38 | 40 |
| 2 11 | 3 | |
| 3 0 | 0 | |
| Median BMI | 17.7 (Range:7) | 17.9(Range:6) |
| Median BSA | 1.41 (Range: 0.45) | 1.43 (Range: 0.54) |
| Median VAS | 10 (Range: 6) | 10 (Range: 4) |
| SGA | A 37 | 40 |
| B 11 | 3 | |
| C 1 | 0 | |
| Median MAC | 28 (Range:7) | 29(Range:8) |
| Clinical Characteristics | Cohort A (n=49) | Cohort B (n=43) |
|---|---|---|
| H&N Cancers | 26 | 20 |
| GIT Cancers | 16 | 17 |
| Breast Cancers | 3 | 1 |
| Cervical Cancers | 3 | 5 |
| Lung Cancer | 1 | 0 |
| Median ANC | 5090 (Range: 11260) | 4875 (Range: 10670) |
| Median Albumin | 4.0 (2.10) | 4.0 (2.50) |
The physical, demographic, and clinical characteristics of the patients assessed are presented in Table 1A and 1B. Cohort A (n=49) represents the group that received the combination of Olanzapine and Megestrol acetate and cohort B (n=43), the group that did not. The median age was 51 and 54 years, respectively, with a slightly higher male-to-female ratio in the cohort A (1.45:1 vs. 1.23:1). Most patients had an ECOG Performance Status of 1, with no cases of PS 3. Median BMI and BSA were similar across groups (BMI: 17.7 vs. 17.9; BSA: 1.41 vs. 1.43). Both groups reported a median VAS score of 10, with slight variations in range. Nutritional status (SGA) indicated most patients were well-nourished (SGA-A), and median MAC was slightly higher in Cohort B (29 cm vs. 28 cm). Overall, the groups were well-balanced at baseline. The clinical characteristics of patients in both arms showed similar distributions across cancer types, in which Head and Neck (H&N) cancers were the most prevalent.
| Treatment Characteristics | Cohort A (n=49) | Cohort B (n=43) |
|---|---|---|
| 2 weekly TPF | 13 | 12 |
| 3 weekly TPF | 12 | 6 |
| FOLFIRINOX | 2 | 3 |
| Gem-Cis | 2 | 2 |
| CAPEOX | 3 | 6 |
| FOLFOX | 1 | 1 |
| FLOT | 1 | 0 |
| 3 weekly P+C | 7 | 5 |
| 3 FEC- 3 DOCE | 3 | 1 |
| NACT/RT | 5 | 7 |
The distribution of treatment characteristics across two arms (cohort A n=49 and cohort B n=42) is presented in table 2. Group 1 had a higher number of patients receiving 2/3-weekly TPF regimens, and specific chemotherapy combinations like 3 weekly P+C and FEC-Docetaxel. Cohort B had slightly more patients treated with the CAPEOX regimen and NACT/RT, suggesting differences in treatment strategies. Out of the 49 patients in Cohort A, 34 (69.3%) of them were administered prophylactic GCSF therapy while only 23 (53.4%) patients in Cohort B received the same.
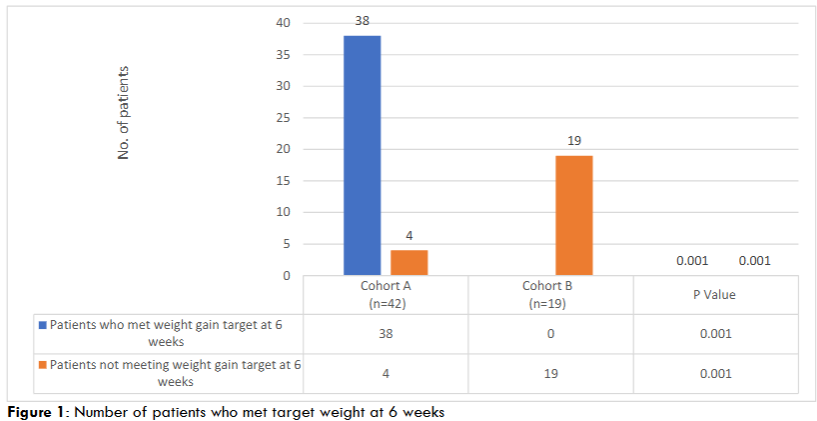
Out of the total patients who completed the study (42 patients in Cohort A and 19 patients in Cohort B), Figure 1 compares the number of patients who met or did not meet the weight gain target at 6 weeks between the cohort A group (n=42) and the cohort B group (n=19). In the cohort A group, a significant improvement in weight gain (≥5% increase) was observed in 90% of patients (n=38). In contrast, none of the patients in the cohort B group met the weight gain target, with all 19 failing to achieve it. The differences between the two groups are statistically significant, as indicated by a p-value of 0.001 for both outcomes.
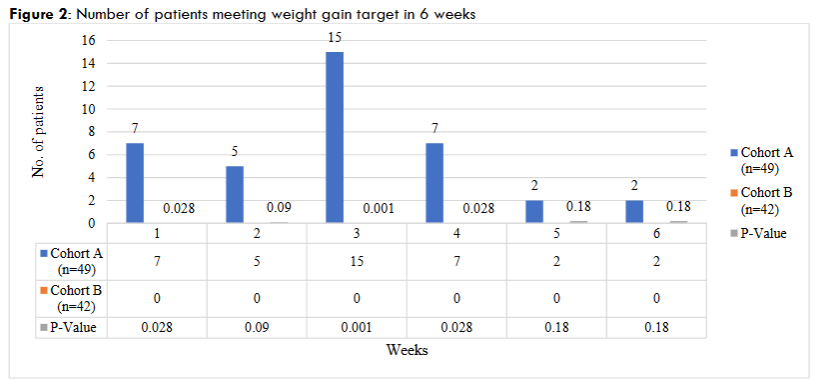
Figure 2 illustrates the number of patients who met the target weight gain in the cohort A group (n=49) and cohort B group (n=42) across six weeks, along with corresponding p-values for statistical significance. In the cohort A group, peaks in patient outcomes are observed at Week 3 (15 patients) and Week 4 (7 patients), with smaller numbers in other weeks. The cohort B group shows no outcomes across all weeks. Statistically significant differences between the groups are noted at Weeks 1 (p=0.028), 3 (p=0.001), and 4 (p=0.028), while Weeks 2, 5, and 6 show no significant differences.
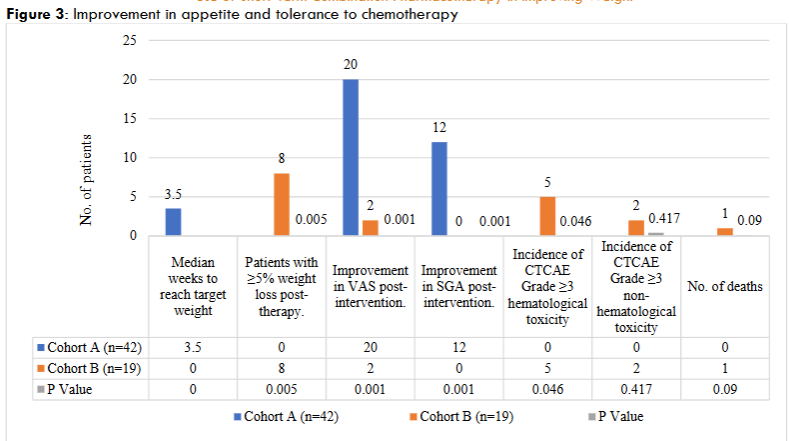
Figure 3 illustrates the outcomes of a cohort A group (n=42) compared to a cohort B group (n=19) in terms of appetite improvement and tolerance to chemotherapy. Significant improvements in Visual Analog Scale (VAS) and Subjective Global Assessment (SGA) scores were observed in the cohort A group (20 and 12 patients, respectively) compared to the cohort B (2 and 0 patients, p=0.001 for both). Hematological toxicity (Grade ≥3) occurred in 5 placebo patients versus none in the cohort A group (p=0.046). Non-hematological toxicity and death were infrequent, with no significant difference between groups (p=0.417 and p=0.09, respectively).
| Predictors | Odds Ratio | CI | P value |
|---|---|---|---|
| No. of patients who met target weight at 6 weeks | 0.0030 | 0.0002 to 0.0585 | 0.0001 |
| Improvement in SGA | 0.0626 | 0.0035 to 1.1182 | 0.0596 |
| Improvement in VAS | 0.1294 | 0.0265 to 0.6317 | 0.0115 |
| Incidence of significant hematological toxicities in 6 weeks | 0.0310 | 0.0016 to 0.5961 | 0.0213 |
| Incidence of significant non-hematological toxicities in 6 weeks | 0.0824 | 0.0038 to 1.8045 | 0.1129 |
| No. of deaths | 0.1451 | 0.0056 to 3.7305 | 0.2439 |
Table 3 presents all the endpoints analyzed using odds ratios (OR), confidence intervals (CI), and p-values (P) to assess their statistical significance over a six-week period. The number of patients achieving target weight at six weeks (OR = 0.0030, P = 0.0001) demonstrates a highly significant association with the intervention, suggesting a strong predictive value. Similarly, improvement in VAS post-intervention (OR = 0.1294, P = 0.0115) and incidence of significant hematological toxicity (OR = 0.0310, P = 0.0213) are statistically significant, indicating meaningful associations. However, improvement in SGA (OR = 0.0626, P = 0.0596), incidence of non-hematological toxicities (OR = 0.0824, P = 0.1129) and the number of deaths (OR = 0.1451, P = 0.2439) lack statistical significance, implying limited predictive power in this dataset. The confidence intervals highlight the precision of these estimates, with narrower ranges suggesting greater reliability.
Discussion
Cancer cachexia significantly impacts patients’ ability to tolerate intensive treatments like cytotoxic chemotherapy and chemoradiotherapy, especially in resource-limited settings24. This study aims to test the efficacy of the combination of safe and tolerable doses of olanzapine and megestrol acetate in improving weight gain, appetite, and tolerance to chemotherapy among precachectic/cachectic solid organ cancer patients (BMI <18 kg/m²).
This study shows that 90% of the patients who received the intervention had significant weight gain (38 patients out of 42 in cohort A and 0 out of 19 patients in Cohort B; p value-0.001); with most of them having gained weight and completed the study in weeks 3 and 4 (with a median of 3.5 weeks) of the originally planned 6 week-long interventions. This highlights the effectiveness of the intervention in promoting weight gain compared to the placebo. This also hints at the fact that the intervention period can be reduced to 4 weeks that would further improve treatment adherence and will work out to be cost-effective.
Existing literature supports the efficacy of megestrol acetate (MA) in promoting weight gain among cancer patients with cachexia. A meta-analysis by Zhan et al. (2013) found that MA significantly increased weight gain (relative risk [RR] 2.17; 95% confidence interval [CI]: 1.59–2.97) and improved appetite (RR 4.68; 95% CI: 3.25–6.76) compared to placebo 25. Furthermore, a systematic review by Ruiz-Garcia et al. (2018) indicated that MA administration led to a mean weight gain of 2.25 kg over periods up to 8 weeks. These findings align with the current study’s observation that significant weight gain can occur within a shorter timeframe.
Eight patients (out of 19 patients) in Cohort B experienced ≥5% weight loss post-therapy (p=0.005), whereas none did in the cohort A. These results suggest that the intervention not only helps to improve weight gain, but is also effective in preventing weight loss while on chemotherapy. This is very important as the tolerance to chemotherapy significantly decreases with weight loss.
Another unexpected observation during the study was the improved study completion rates in Cohort A and increased dropout rates in Cohort B. In cohort A, 42 patients completed the study, with only 7 dropouts (14.2%). In contrast, the cohort B group had 12 dropouts (27.9%). The lesser dropout rates in Cohort A suggests the benefit of the combination in improving treatment tolerability and patient adherence.
This combination also resulted in improvements in appetite with statistically significant improvement in VAS and SGA score post intervention (seen in 20 and 12 patients respectively in Cohort A as compared to cohort B, with 2 and 0 patients respectively; p=0.001 for both). Hematological toxicities (CTCAE Grade ≥3) occurred in 5 placebo patients versus none in the cohort A group (p=0.046). Non-hematological toxicities and death were infrequent, with no significant difference between groups (p=0.417 and p=0.09, respectively). These results highlight the effectiveness of the intervention in reducing adverse events.
The results of this study highlight the following points:
- Effectiveness in Resource-Limited Settings: The dual action of olanzapine and megestrol acetate provides a robust pharmacological alternative to conventional supportive care measures, reducing reliance on agents such as neurokinin-1 (NK-1) antagonists (e.g., fosaprepitant and aprepitant) and prophylactic granulocyte colony-stimulating factor (G-CSF) support. These savings are critical in resource-limited settings, where the affordability of supportive care often dictates accessibility. Additionally, improved chemotherapy tolerance reduces hospital admissions for supportive care and complications, lowering overall healthcare expenditures. Furthermore, by increasing the number of patients receiving standard-of-care chemotherapy, the cancer burden is reduced, and relapse rates decline, minimizing the need for additional cycles of chemotherapy.
- Intervention that yields rapidly tangible results: The short time frame within which this FDC demonstrates efficacy offers a pragmatic solution for cancer cachexia management. Underweight patients identified at their first clinical visit can be initiated on this combination therapy immediately, with benefits such as improved appetite, weight stabilization, and reduced nausea manifesting within a few weeks. This ensures that the patients are physically and nutritionally better prepared to initiate chemotherapy without delay, aligning with the evidence that weight gain and nutritional improvements during treatment are associated with superior survival outcomes.
- Addressing Socioeconomic Disparities in Chemotherapy Tolerance: Despite advancements in oncology, there is a notable gap in addressing chemotherapy tolerance in patients from low socioeconomic backgrounds. Existing predictive tools like CRASH and CARG indices cater to elderly populations but fail to account for the compounded effects of malnutrition and poverty in younger or middle-aged patients from underprivileged regions. This study uniquely focuses on this demographic, recognizing the inherent malnutrition and systemic inequities that contribute to poor treatment tolerance and outcomes in resource-limited settings.
- Impact on Cancer Burden and Relapse Rates: By enabling more patients to withstand and complete standard-of-care regimens, this drug combination has the potential to decrease cancer relapse rates, which are often linked to suboptimal initial treatments. This ripple effect not only reduces the need for additional chemotherapy but also alleviates the broader cancer burden on healthcare systems, particularly in high-incidence regions like India, where disparities in care exacerbate outcomes.
- Enhanced Patient Compliance: Adherence to therapy is often influenced by the severity of side effects and the perceived burden of treatment. The combination of appetite stimulation, weight gain, and nausea control provided by this combination improves patients’ physical and psychological well-being, fostering greater compliance with prescribed regimens. Increased compliance directly correlates with improved treatment outcomes and long-term survival.
- Improved Quality of Life for Patients and their Families: Cancer affects not only the patient but also their families, who often bear the emotional and financial burdens of care. By reducing hospital admissions, improving treatment tolerance, and enabling patients to receive effective therapy with fewer side effects, this intervention enhances the overall quality of life for both patients and caregivers. Improved nutritional status and reduced nausea translate to fewer disruptions in daily activities, better mental health, and reduced caregiving strain.
Conclusion
This retrospective study thus demonstrates that the use of short-term olanzapine and megestrol acetate in patients undergoing chemotherapy or chemoradiotherapy led to significant improvements in weight gain, appetite, and a reduction in treatment-related toxicities compared to those who did not receive the combination. These findings underscore the potential of this effective strategy to enhance supportive care outcomes, particularly in underserved populations. Future studies should aim to validate these results through larger, multicenter randomized control trials and establish guidelines for implementing this approach across diverse clinical and socioeconomic contexts.
References
- Chow, R., et al. (2020). Managing treatment-related toxicities in patients with advanced cancer: A review of supportive care options. Journal of Clinical Oncology, 38(15), 1234-1245.
- Hesketh, P. J. (2021). Chemotherapy-induced nausea and vomiting. New England Journal of Medicine, 384(6), 547-556.
- Arends, J., Bachmann, P., Baracos, V., Barthelemy, N., Bertz, H., Bozzetti, F., … & Preiser, J. C. (2017). ESPEN guidelines on nutrition in cancer patients. Clinical Nutrition, 36(1), 11-48.
- Klastersky, J., de Naurois, J., Rolston, K., Rapoport, B., Maschmeyer, G., Aapro, M., & Herrstedt, J. (2016). Management of febrile neutropaenia: ESMO Clinical Practice Guidelines. Annals of Oncology, 27(suppl 5), v111–v118. https://doi.org/10.1093/annonc/mdw325
- Topkan E, Parlak C, Selek U. Impact of weight change during the course of concurrent chemoradiation therapy on outcomes in stage IIIB non-small cell lung cancer patients: retrospective analysis of 425 patients. Int J Radiat Oncol Biol Phys 2013;87:697-704. 10.1016/j.ijrobp.2013.07.033
- Sher DJ, Gielda BT, Liptay MJ, et al. Prognostic significance of weight gain during definitive chemoradiotherapy for locally advanced non-small-cell lung cancer. Clin Lung Cancer 2013;14:370-5. 10.1016/j.cllc.2012.10.009
- Gielda BT, Mehta P, Khan A, et al. Weight gain in advanced non-small-cell lung cancer patients during treatment with split-course concurrent chemoradiotherapy is associated with superior survival. Int J Radiat Oncol Biol Phys 2011;81:985-91. 10.1016/j.ijrobp.2010.06.059
- Patel JD, Pereira JR, Chen J, et al: Relationship between efficacy outcomes and weight gain during treatment of advanced, non-squamous, non-small-cell lung cancer patients. Ann Oncol 27:1612-1619, 2016.
- Erkan Topkan. Weight gain as a surrogate marker of longer survival in advanced non-small cell lung cancer patients. Annals of translational medicine [Internet]. 2016 Oct 1 [cited 2024 May 4];4(19):381–1. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5075850/
- Navari RM, Brenner MC. Treatment of cancer-related anorexia with olanzapine and megestrol acetate: a randomized trial. Supportive Care in Cancer. 2009 Sep 11;18(8):951–6.
- Lakshmi Sandhya, Nirmala Devi Sreenivasan, Luxitaa Goenka, Biswajit Dubashi, Kayal S, S. Manikandan, et al. Randomized Double-Blind Placebo-Controlled Study of Olanzapine for Chemotherapy-Related Anorexia in Patients With Locally Advanced or Metastatic Gastric, Hepatopancreaticobiliary, and Lung Cancer. Journal of Clinical Oncology. 2023 May 10;41(14):2617–27.
- Loprinzi, C. L., Ellison, N., Schaid, D., Krook, J. E., Athmann, L. M., Dose, A. M., & Goldberg, R. M. (1993). Controlled trial of megestrol acetate for the treatment of cancer anorexia and cachexia. Journal of the National Cancer Institute, 85(8), 587–592. https://doi.org/10.1093/jnci/85.8.587
- Fearon, K., et al. (2012). Definition and classification of cancer cachexia: An international consensus. The Lancet Oncology, 13(5), e255-e264.
- Argilés, J. M., et al. (2014). Cancer-associated cachexia: Pathophysiology and clinical relevance. The Lancet Oncology, 15(6), e229-e237.
- Mansoor, W., et al. (2021). Megestrol acetate for cancer-associated cachexia: Evidence and current practices. Supportive Care in Cancer, 29(3), 587-596.
- Loprinzi, C. L., et al. (1999). Megestrol acetate in cancer anorexia and cachexia. Journal of Clinical Oncology, 17(11), 3476-3481.
- Navari, R. M., & Brenner, M. C. (2010). Olanzapine for the prevention of chemotherapy-induced nausea and vomiting. Supportive Care in Cancer, 18(1), 17-22.
- Sandhya L, Devi Sreenivasan N, Goenka L, et al. Randomized Double-Blind Placebo-Controlled Study of Olanzapine for Chemotherapy-Related Anorexia in Patients With Locally Advanced or Metastatic Gastric, Hepatopancreaticobiliary, and Lung Cancer. J Clin Oncol. 2023;41(14):2617-2627. doi:10.1200/JCO.22.01997
- Navari, R. M., et al. (2016). Olanzapine for the prevention and treatment of chemotherapy-induced nausea and vomiting: A systematic review. JAMA Oncology, 2(4), 493-497.
- Hocking, C. M., et al. (2020). Olanzapine in the management of CINV: Current evidence and future directions. Clinical Oncology, 32(9), e314-e321.
- Tan, B. H. L., et al. (2019). Patient-centered approaches to managing cancer cachexia. Journal of Supportive Oncology, 17(5), 234-244.
- Prado, C. M., et al. (2018). Cachexia management in cancer: Practical insights. Current Opinion in Supportive and Palliative Care, 12(4), 347-354.
- Ruiz-Garcia, V., López-Briz, E., Carbonell-Sanchis, R., Bort-Marti, S., & Gonzalvez-Perales, J. L. (2018). Megestrol acetate for treatment of anorexia-cachexia syndrome. Cochrane Database of Systematic Reviews, 2018(9), CD004310. https://doi.org/10.1002/14651858.CD004310.pub5
- Philip D. Bonomi et al. Cancer cachexia impact on chemotherapy dose reduction, treatment discontinuation, and survival: A qualitative systematic review. JCO 40, e24103e24103(2022). Doi:10.1200/JCO.2022.40.16_suppl.e24103
- Zhan, P., Wang, Q., Qian, Q., Yu, L. K. (2013). Megestrol acetate in cancer patients with anorexia-cachexia syndrome: A meta-analysis. Translational Cancer Research, 2(4), 278–287. DOI: 10.3978/j.issn.2218-676X.2013.09.01




