Improving Pediatric Congenital Hypothyroidism Outcomes
Improving Outcomes in Pediatric Congenital Hypothyroidism Through an Electronic Dashboard: A Quality Improvement Initiative
Atley Moberly¹; (Co-first author) Rob Gonsalves, DO¹; Linda Black, RN¹; Vinay Vaidya, MD¹; Mary Kay Walsh, RN¹; Chirag Kapadia, MD¹
- Phoenix Children’s Hospital, Phoenix, AZ
OPEN ACCESS
PUBLISHED 30 September 2025
CITATION Moberly, A., Gonsalves, R., et al., 2025. Improving Outcomes in Pediatric Congenital Hypothyroidism Through an Electronic Dashboard: A Quality Improvement Initiative. Medical Research Archives, [online] 13(9). https://doi.org/10.18103/mra.v13i9.6934
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i9.6934
ISSN 2375-1924
ABSTRACT
Congenital hypothyroidism (CH) is a prevalent endocrine disorder that can lead to intellectual disability, psychomotor dysfunction, and impaired growth if not treated promptly. Early diagnosis and consistent monitoring are critical, especially during the first three years of life. We developed a PowerBI dashboard integrated with the electronic medical record (EMR) to track thyroid-stimulating hormone (TSH) levels, clinic visits, and upcoming appointments for patients aged 3 months to 3 years. It enables real-time identification of patients not meeting clinical targets, facilitating timely interventions. Prior to dashboard implementation, 75% of patients achieved two or more normal TSH values annually. One year post-implementation, this figure rose to 91%, and further refinements increased it to 93% the following year. The dashboard also improved clinic efficiency by streamlining communication and reducing manual data tracking. This quality improvement (QI) initiative shows the effectiveness of EMR-integrated dashboards in improving clinical outcomes and operational efficiency. The model is scalable and adaptable for other chronic pediatric conditions requiring regular follow-up and laboratory monitoring. Our findings support broader adoption of similar PowerBI dashboards in Endocrinology and other specialties focused on chronic disease management.
Keywords
Congenital Hypothyroidism, Pediatric Endocrinology, Quality Improvement, Electronic Dashboard, TSH Monitoring, EMR Integration
Highlights
- Electronic dashboard implementation significantly improved clinical outcomes for pediatric patients with congenital hypothyroidism (CH) at Phoenix Children’s Hospital.
- TSH monitoring compliance increased from 75% to 93% over two years following dashboard deployment.
- The dashboard enabled real-time tracking of lab results, appointments, and patient demographics, streamlining care coordination.
- Quality improvement methodology using Plan-Do-Study-Act (PDSA) cycles facilitated iterative enhancements to the dashboard and clinical workflow.
- The project demonstrates a scalable model for other institutions to improve care for chronic pediatric conditions requiring frequent follow-up and lab monitoring.
Introduction
Congenital Hypothyroidism is a disorder leading to reduced levels of thyroid hormone (TH) at birth, which can lead to intellectual disability, psychomotor dysfunction, and impaired growth without treatment. CH affects approximately 1:2,000 to 1:4,000 live births a year. In the United States, the incidence rate of CH has significantly increased over the last few decades, which can be attributed to an increase in detection via newborn screening. A higher incidence has also been reported in Asian and Hispanic newborns. Possible reasons for these findings include environmental and genetic differences among populations.
Primarily, CH occurs due to abnormal embryonic thyroid growth or a lack of TH production at the thyroid level. However, CH can also transpire due to a lack of regulation from the hypothalamus or pituitary gland. Individuals can present with persistent CH or may experience a temporary deficiency of TH, known as transient CH. Causes of transient CH include premature delivery, iodine deficiency or exposure, maternal thyrotropin receptor blocking antibodies, maternal intake of anti-thyroid drugs, or other genetic factors.
The Newborn Screen (NBS) is a powerful tool used in the first 1 to 5 days of life to screen for various congenital disorders, including CH. This test is collected via heel stick and transferred as a dried blood spot on state-approved filter paper card specimens. In Arizona, the blood sample is tested via immunoassay for TSH. However, other test strategies can include free thyroxine (T4) or a combination of both tests. If an abnormal NBS is obtained, a confirmatory serum test is then collected measuring TSH and FT4 to assess treatment requirements.
Deficiency of TH leads to a decline of neurocognitive development, which is most important in the first 3 years of life. This intellectual disability can be prevented or minimized with timely management and treatment with levothyroxine (L-T4), beginning at 10-15 mcg/kg daily. It is recommended treatment begins immediately upon positive screening while diagnostic tests are performed, and ideally no later than 2 weeks of age. L-T4 treatment aims to maintain serum FT4 in the mid to upper half and serum TSH in the mid to lower half of the pediatric reference ranges. Currently, L-T4 tablets are the most common treatment and the treatment available in most countries. The appropriate dosage of tablets can be crushed into powder and mixed with breastmilk, non-soy formula, or water for infants. The American Academy of Pediatrics (AAP) recommends serum TSH and FT4 measured 1 to 2 weeks after initiating L-T4 treatment and every 2 weeks until serum TSH level is normal. Then, monitoring may transition to every 1 to 2 months during the first 6 months of life and every 2 to 3 months in the second 6 months. Finally, serum levels should be collected every 3 to 4 months between the ages of 1 to 3 years, then every 6 to 12 months until growth is complete.
We recently demonstrated the use of electronic PowerBI dashboards in managing patients with type 1 diabetes mellitus (T1DM), including integration of data with the EMR, and ongoing collaboration with the information technology (IT) team. In that study, we showed increased patient compliance and glycemic control while decreasing hospital admissions. Given this success, we attempted to apply similar principles to our CH population. Previously, we had created and managed static data reports to contact patients with CH falling short of treatment goals.
Materials and Methods
DASHBOARD DEVELOPMENT AND DESCRIPTION
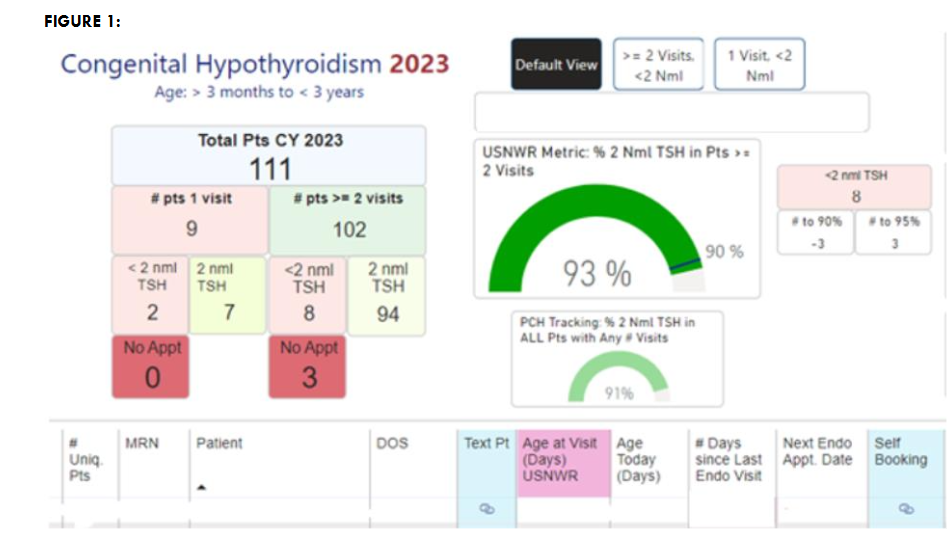
The dashboard, built on Microsoft PowerBI software, monitors TSH laboratory results, appointment scheduling, and patient demographics, pulling this information directly from the EMR. An overview of the main components is depicted in Figure 1. Specifically, target TSH values and the number of appointments within the year are tracked and recorded, and simple calculations, such as percentages of patients achieving target, are reported. It contains a patient list tab and a TSH results tab. On the patient list tab, we can monitor the date of service (DOS), next appointment date, number of days since the patient’s last visit, number of visits for the year, number of normal TSH values, and the date of lab collection. We currently monitor patients on the dashboard from 3 months to 3 years. Our electronic medical records (EMR) system continuously integrates lab values and upcoming appointments with our dashboard to create live data reports. Our EMR system also allows us to text patients and send appointment reminders.
INTERVENTION
With the use of our dashboard, we were able to begin implementing interventions specific to the CH population immediately. On the homepage of the dashboard interface, we identified the number of patients with CH between the ages of 3 months to 3 years and the number of patients reaching two visits within the calendar year. Furthermore, we assessed the number of patients with at least two normal TSH values and those with less than two normal TSH values. We could quickly and accurately identify patients falling short of our management goals and subsequently contact families to get labs, schedule appointments, discuss lab results, and adjust medication when needed.
Results
We saw 111 patients with CH meeting the age criteria described above in 2023. Before the development and implementation of our dashboard, 75% of patients had two or more normal TSH values within the calendar year. After the implementation of our dashboard in 2022, the percentage of patients reaching our goal increased to 91%. After further revisions to the dashboard and our process, we increased to 93% in 2023.
Discussion
We implemented use of the PowerBI dashboard, with data integration with our EMR, for CH patients below the age of 3 years. Our primary outcome was the percentage of patients seen at least twice yearly, with two or more normal TSH values in the calendar year. Secondary efforts, not as closely monitored, included scheduling patients needing follow-up appointments, and ensuring appropriate labs were ordered. Monthly meetings were held to review our processes and outcomes and changes to the dashboard, and these meetings involved Endocrine physicians and nursing, together with our IT team. Our approach was carried out as plan-do-study-act (PDSA) cycles.
The dashboard allowed for a better understanding of our clinic demographics and needs. It also served as a powerful tool for patient monitoring and intervention. We significantly increased the percentage of patients with two or more visits, plus two or more normal TSH values, just one year after dashboard implementation.
Our CH QI project has shown that by organizing patient data into a streamlined EMR-integrated dashboard, we were able to create meaningful improvements in clinic efficiency and patient care. Overall, this patient population is likely to have better developmental and growth outcomes as a result of this work, as inadequate control of thyroid conditions in the newborn period can compromise development and growth.
Given the successes of both our T1DM dashboard and now CH dashboard, we have additionally begun to develop and implement dashboards for the monitoring of patients with adrenal insufficiency, type 2 diabetes mellitus, and for children with conditions that adversely affect bone health.
PowerBI Data dashboards, integrated with the EMR, are a user-friendly tool that can be implemented at other institutions to similarly improve clinic efficiency and care for patients with CH and any chronic condition requiring laboratory monitoring and frequent follow-up.
Credit Author Statement
Atley Moberly: Writing and data analysis. Chirag Kapadia: Conceptualization; Methodology; Project Administration; Supervision; Writing – Original Draft; Writing – Review & Editing. Rob Gonsalves: Data Analysis; Monitoring; Writing. Linda Black: Data Monitoring; Patient Calls. Vinay Vaidya: Data Tool Development. Mary Kay Walsh: Data Tool Development.
Declaration of Interest Statement
The authors declare that they have no conflicts of interest to disclose in relation to the content of this manuscript.
References
- Rose, S. R., Wassner, A. J., Wintergerst, K. A., et al. (2023). Congenital hypothyroidism: Screening and management. Pediatrics, 151(1). https://doi.org/10.1542/peds.2022-060419
- Chen, C. Y., Lee, K. T., Lee, C. T., Lai, W. T., & Huang, Y. B. (2013). Epidemiology and clinical characteristics of congenital hypothyroidism in an Asian population: A nationwide population-based study. Journal of Epidemiology, 23(2), 85–94. https://doi.org/10.2188/jea.je20120113
- Mitchell, M. L., Hsu, H.-W., & Sahai, I. (2011). The increased incidence of congenital hypothyroidism: Fact or fancy? Clinical Endocrinology, 75(6), 806–810. https://doi.org/10.1111/j.1365-2265.2011.04128.x
- Hinton, C. F., Harris, K. B., Borgfeld, L., et al. (2010). Trends in incidence rates of congenital hypothyroidism related to select demographic factors: Data from the United States, California, Massachusetts, New York, and Texas. Pediatrics, 125(Suppl. 2), S37–S47. https://doi.org/10.1542/peds.2009-1975D
- Rastogi, M. V., & LaFranchi, S. H. (2010). Congenital hypothyroidism. Orphanet Journal of Rare Diseases, 5(1), 17. https://doi.org/10.1186/1750-1172-5-17
- Kanike, N., Davis, A., & Shekhawat, P. S. (2017). Transient hypothyroidism in the newborn: To treat or not to treat. Translational Pediatrics, 6(4), 349–358. https://doi.org/10.21037/tp.2017.09.07
- Rose, S. R., & Brown, R. S. (2006). Update of newborn screening and therapy for congenital hypothyroidism. Pediatrics, 117(6), 2290–2303. https://doi.org/10.1542/peds.2006-0915
- Arizona Department of Health Services. (2024, May 20). State laboratory services – Newborn screening. https://www.azdhs.gov/preparedness/state-laboratory/newborn-screening/index.php#healthcare-providers-bloodspot
- Léger, J., Olivieri, A., Donaldson, M., et al. (2014). European Society for Paediatric Endocrinology consensus guidelines on screening, diagnosis, and management of congenital hypothyroidism. Hormone Research in Paediatrics, 81(2), 80–103. https://doi.org/10.1159/000358198
- Büyükgebiz, A. (2013). Newborn screening for congenital hypothyroidism. Journal of Clinical Research in Pediatric Endocrinology, 5(Suppl. 1), 8–12. https://doi.org/10.4274/jcrpe.845
- Shapira, S. K., Hinton, C. F., Held, P. K., Jones, E., Hannon, W. H., & Ojodu, J. (2015). Single newborn screen or routine second screening for primary congenital hypothyroidism. Molecular Genetics and Metabolism, 116(3), 125–132. https://doi.org/10.1016/j.ymgme.2015.08.003
- Murphy, N., Hume, R., van Toor, H., et al. (2004). The hypothalamic-pituitary-thyroid axis in preterm infants: Changes in the first 24 hours of postnatal life. The Journal of Clinical Endocrinology & Metabolism, 89(6), 2824–2831. https://doi.org/10.1210/jc.2003-030317
- Jonklaas, J., Bianco, A. C., Bauer, A. J., et al. (2014). Guidelines for the treatment of hypothyroidism: Prepared by the American Thyroid Association task force on thyroid hormone replacement. Thyroid, 24(12), 1670–1751. https://doi.org/10.1089/thy.2014.0028
- Bongers-Schokking, J. J., Koot, H. M., Wiersma, D., Verkerk, P. H., & de Muinck Keizer-Schrama, S. M. (2000). Influence of timing and dose of thyroid hormone replacement on development in infants with congenital hypothyroidism. Journal of Pediatrics, 136(3), 292–297. https://doi.org/10.1067/mpd.2000.103351
- Stagi, S., Municchi, G., Ferrari, M., & Wasniewska, M. G. (2022). An overview on different L-thyroxine (L-T4) formulations and factors potentially influencing the treatment of congenital hypothyroidism during the first 3 years of life. Frontiers in Endocrinology, 13, Article 859487. https://doi.org/10.3389/fendo.2022.859487
- Sandblom, L., Kapadia, C., Vaidya, V., et al. (2023). Electronic dashboard to improve outcomes in pediatric patients with type 1 diabetes mellitus. Journal of Diabetes Science and Technology. Advance online publication. https://doi.org/10.1177/19322968231159401
- Klosinska, M., Kaczynska, A., & Ben-Skowronek, I. (2022). Congenital hypothyroidism in preterm newborns – The challenges of diagnostics and treatment: A review. Frontiers in Endocrinology, 13, Article 860862. https://doi.org/10.3389/fendo.2022.860862
- Esposito, A., Vigone, M. C., Polizzi, M., Wasniewska, M. G., Cassio, A., Mussa, A., Gastaldi, R., Di Mase, R., Vincenzi, G., Pozzi, C., Peroni, E., Bravaccio, C., Capalbo, D., Bruzzese, D., & Salerno, M. (2022). Effect of initial levothyroxine dose on neurodevelopmental and growth outcomes in children with congenital hypothyroidism. Frontiers in Endocrinology, 13, Article 923448. https://doi.org/10.3389/fendo.2022.923448
- Uchida, K., & Suzuki, M. (2021). Congenital hypothyroidism and brain development: Association with other psychiatric disorders. Frontiers in Neuroscience, 15, Article 772382. https://doi.org/10.3389/fnins.2021.772382

